Generation and Characterization of Anti-Filovirus Nucleoprotein Monoclonal Antibodies
Abstract
1. Introduction:
2. Methods
2.1. Cell Lines and Viruses
2.2. Production of Filovirus Antigens
2.3. Mice Immunization and Monoclonal Antibody (mAb) Production
2.4. Indirect ELISA
2.5. Western Blot Analysis
2.6. Confocal Microscopy (CM)
3. Results
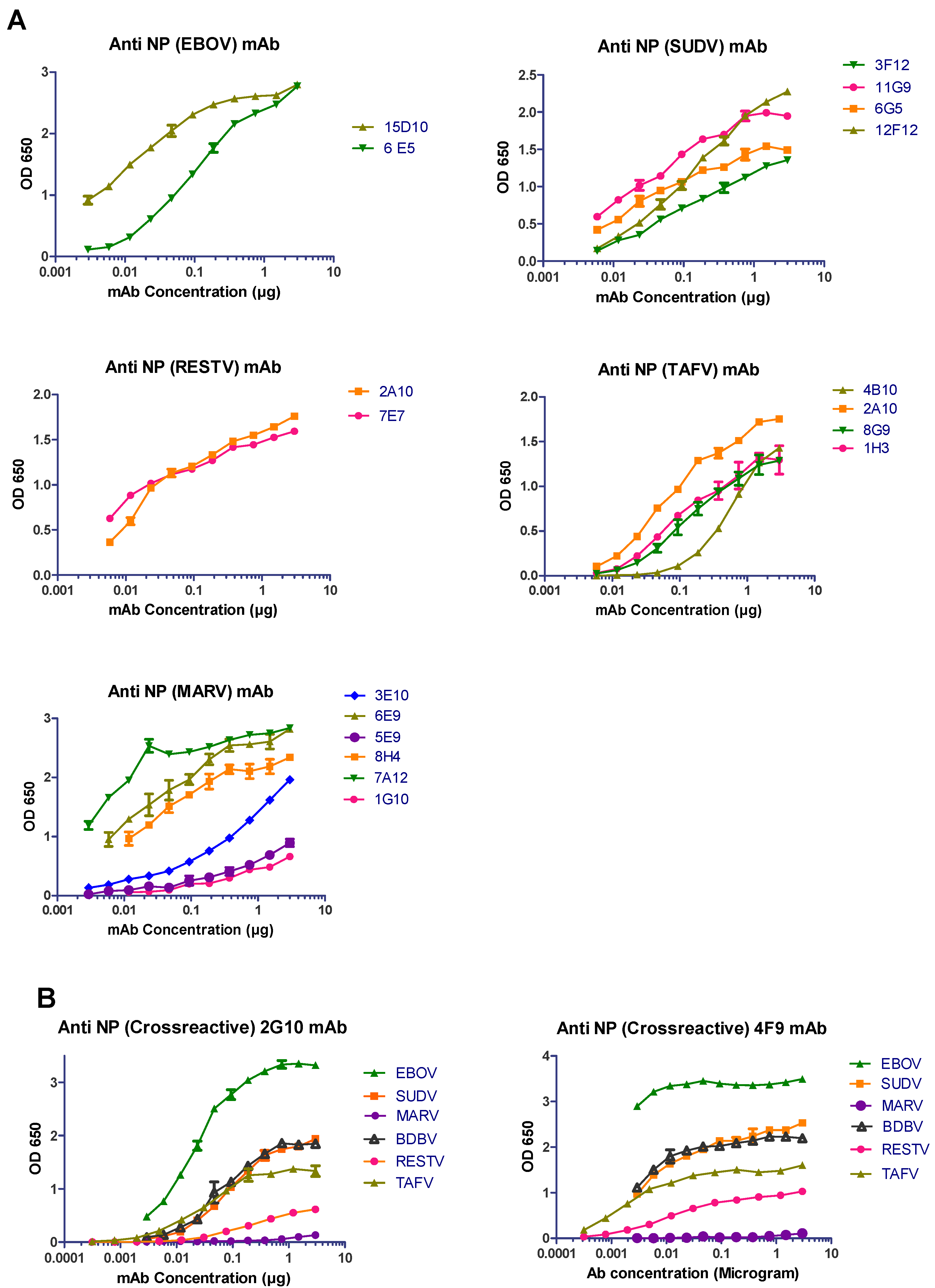
3.1. Anti-NP mAb Production and Isotyping
3.2. Characterization of Anti-NP mAb Reactivity
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Agua-Agum, J.; Allegranzi, B.; Ariyarajah, A.; Aylward, R.; Blake, I.M.; Barboza, P.; Bausch, D.; Brennan, R.J.; Clement, P.; Coffey, P.; et al. After Ebola in West Africa—Unpredictable Risks, Preventable Epidemics. N. Engl. J. Med. 2016, 375, 587–596. [Google Scholar] [PubMed]
- Messaoudi, I.; Amarasinghe, G.K.; Basler, C.F. Filovirus pathogenesis and immune evasion: Insights from Ebola virus and Marburg virus. Nat. Rev. Microbiol. 2015, 13, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Shears, P.; O’Dempsey, T.J. Ebola virus disease in Africa: Epidemiology and nosocomial transmission. J. Hosp. Infect. 2015, 90, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Wong, G.; Audet, J.; Bello, A.; Fernando, L.; Alimonti, J.B.; Fausther-Bovendo, H.; Wei, H.; Aviles, J.; Hiatt, E.; et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 2014, 514, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Audet, J.; Wong, G.; Pillet, S.; Bello, A.; Cabral, T.; Strong, J.E.; Plummer, F.; Corbett, C.R.; Alimonti, J.B.; et al. Successful treatment of ebola virus-infected cynomolgus macaques with monoclonal antibodies. Sci. Transl. Med. 2012, 4, 138ra181. [Google Scholar] [CrossRef] [PubMed]
- Wec, A.Z.; Herbert, A.S.; Murin, C.D.; Nyakatura, E.K.; Abelson, D.M.; Fels, J.M.; He, S.; James, R.M.; de La Vega, M.A.; Zhu, W.; et al. Antibodies from a Human Survivor Define Sites of Vulnerability for Broad Protection against Ebolaviruses. Cell 2017, 169, 878–890.e15. [Google Scholar] [CrossRef] [PubMed]
- Warren, T.K.; Jordan, R.; Lo, M.K.; Ray, A.S.; Mackman, R.L.; Soloveva, V.; Siegel, D.; Perron, M.; Bannister, R.; Hui, H.C.; et al. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature 2016, 531, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, P.; Marzi, A. Ebola and Marburg virus vaccines. Virus Genes 2017, 53, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Muhlberger, E. Filovirus replication and transcription. Future Virol. 2007, 2, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Changula, K.; Yoshida, R.; Noyori, O.; Marzi, A.; Miyamoto, H.; Ishijima, M.; Yokoyama, A.; Kajihara, M.; Feldmann, H.; Mweene, A.S.; et al. Mapping of conserved and species-specific antibody epitopes on the Ebola virus nucleoprotein. Virus Res. 2013, 176, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Becquart, P.; Mahlakoiv, T.; Nkoghe, D.; Leroy, E.M. Identification of continuous human B-cell epitopes in the VP35, VP40, nucleoprotein and glycoprotein of Ebola virus. PLoS ONE 2014, 9, e96360. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Liu, W.; Zhang, Q.; Cao, S.; Zhao, H.; Wang, T.; Qi, Z.; Han, Y.; Song, Y.; Wang, X.; et al. Yersinia pestis YopK Inhibits Bacterial Adhesion to Host Cells by Binding to the Extracellular Matrix Adaptor Protein Matrilin-2. Infect. Immun. 2017, 85, e01069-16. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.B.; Yang, R.; Zhang, Z.P.; Bi, L.J.; You, X.Y.; Wei, H.P.; Zhou, Y.F.; Yu, Z.; Zhang, X.E. Detection of B. anthracis spores and vegetative cells with the same monoclonal antibodies. PLoS ONE 2009, 4, e7810. [Google Scholar] [CrossRef]
- Perosa, F.; Carbone, R.; Ferrone, S.; Dammacco, F. Purification of human immunoglobulins by sequential precipitation with caprylic acid and ammonium sulphate. J. Immunol. Methods 1990, 128, 9–16. [Google Scholar] [CrossRef]
- Shi, W.; Huang, Y.; Sutton-Smith, M.; Tissot, B.; Panico, M.; Morris, H.R.; Dell, A.; Haslam, S.M.; Boyington, J.; Graham, B.S.; et al. A filovirus-unique region of Ebola virus nucleoprotein confers aberrant migration and mediates its incorporation into virions. J. Virol. 2008, 82, 6190–6199. [Google Scholar] [CrossRef] [PubMed]
- Bjorndal, A.S.; Szekely, L.; Elgh, F. Ebola virus infection inversely correlates with the overall expression levels of promyelocytic leukaemia (PML) protein in cultured cells. BMC Microbiol. 2003, 3, 6. [Google Scholar] [CrossRef]
- Yan, F.; He, S.; Banadyga, L.; Zhu, W.; Zhang, H.; Rahim, M.D.; Senthilkumaran, C.; Collignon, B.; Embury-Hyatt, C.; Qiu, X. Characterization of Reston virus infection in ferrets. Antivir. Res. 2019. [Google Scholar] [CrossRef]
- Dhillon, R.S.; Srikrishna, D.; Sachs, J. Controlling Ebola: Next steps. Lancet 2014, 384, 1409–1411. [Google Scholar] [CrossRef]
- Albarino, C.G.; Wiggleton Guerrero, L.; Chakrabarti, A.K.; Nichol, S.T. Transcriptional analysis of viral mRNAs reveals common transcription patterns in cells infected by five different filoviruses. PLoS ONE 2018, 13, e0201827. [Google Scholar] [CrossRef]
- Bharat, T.A.; Noda, T.; Riches, J.D.; Kraehling, V.; Kolesnikova, L.; Becker, S.; Kawaoka, Y.; Briggs, J.A. Structural dissection of Ebola virus and its assembly determinants using cryo-electron tomography. Proc. Natl. Acad. Sci. USA 2012, 109, 4275–4280. [Google Scholar] [CrossRef]
- Muhlberger, E.; Weik, M.; Volchkov, V.E.; Klenk, H.-D.; Becker, S. Comparison of the transcription and replication strategies of marburg virus and Ebola virus by using artificial replication systems. J. Virol. 1999, 73, 2333–2342. [Google Scholar] [PubMed]
- Muhlberger, E.; Lotfering, B.; Klenk, H.D.; Becker, S. Three of the four nucleocapsid proteins of Marburg virus, NP, VP35, and L, are sufficient to mediate replication and transcription of Marburg virus-specific monocistronic minigenomes. J. Virol. 1998, 72, 8756–8764. [Google Scholar] [PubMed]

| Target | Amino Acid Sequences | mAbs Generated |
|---|---|---|
| EBOV-NP | aa 421–440: YDDDDDIPFPGPINDDDNPG | 4F9, 2G10 |
| EBOV-NP | aa 491–510: DDEDTKPVPNRSTKGGQQKN | 6E5, 15D10 |
| SUDV-NP | aa 631–644: QGSESEALPINSKK | 6G5, 3F12, 11G9, 12F12 |
| TAFV-NP | aa 630–643: NQVSGSENTDNKPH | 1H3, 8G9, 4B10, 2A10 |
| RESTV-NP | aa 630–643: TSQLNEDPDIGQSK | 7E7, 2A10 |
| MARV-NP | aa 635–652: RVVTKKGRTFLYPNDLLQ | 1G10, 3E10, 5E9, 6E9, 8H4, 7A12 |
| mAb | Type | Reactivity | ELISA (Irradiated Virus) | Western Blot (Irradiated Virus) | Confocal Microscopy (Wild-Type Infection) |
|---|---|---|---|---|---|
| 15D10 | IgG1 | EBOV | ++ | ++ | ++ |
| 6E5 | IgG1 | ++ | ++ | ++ | |
| 3F12 | IgG1 | SUDV | ++ | +++ | + |
| 11G9 | IgG1 | +++ | +++ | ++ | |
| 6G5 | IgG1 | ++ | +++ | ++ | |
| 12F12 | IgG1 | +++ | + | ++ | |
| 2A10 | IgG1 | RESTV | ++ | ++ | ++ |
| 7E7 | IgG1 | ++ | ++ | ++ | |
| 4B10 | IgG2b | TAFV | ++ | +++ | + |
| 2A10 | IgG1 | ++ | +++ | + | |
| 8G9 | IgG1 | ++ | +++ | − | |
| 1H3 | IgG1 | ++ | +++ | +++ | |
| 3E10 | IgM | MARV | ++ | +++ | +/− |
| 6E9 | IgG2b | +++ | +++ | ++ | |
| 5E9 | IgM | + | +++ | + | |
| 8H4 | IgG1 | +++ | +++ | +++ | |
| 7A12 | IgG1 | +++ | +++ | +++ | |
| 1G10 | IgG1 | + | + | − | |
| 2G10 | IgG1 | EBOV | +++ | +++ | +++ |
| SUDV | ++ | + | ++ | ||
| TAFV | ++ | + | ++ | ||
| RESTV | + | − | +++ | ||
| MARV | − | − | − | ||
| BDBV | ++ | +++ | ND | ||
| 4F9 | IgM | EBOV | +++ | +++ | +++ |
| SUDV | +++ | + | ++ | ||
| TAFV | ++ | + | ND | ||
| RESTV | + | − | +++ | ||
| MARV | − | − | + | ||
| BDBV | ++ | +++ | ND |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahim, M.N.; Wang, M.; Wang, T.; He, S.; Griffin, B.D.; Kobasa, D.; Yang, R.; Du, Z.; Qiu, X. Generation and Characterization of Anti-Filovirus Nucleoprotein Monoclonal Antibodies. Viruses 2019, 11, 259. https://doi.org/10.3390/v11030259
Rahim MN, Wang M, Wang T, He S, Griffin BD, Kobasa D, Yang R, Du Z, Qiu X. Generation and Characterization of Anti-Filovirus Nucleoprotein Monoclonal Antibodies. Viruses. 2019; 11(3):259. https://doi.org/10.3390/v11030259
Chicago/Turabian StyleRahim, Md Niaz, Min Wang, Tong Wang, Shihua He, Bryan D. Griffin, Darwyn Kobasa, Ruifu Yang, Zongmin Du, and Xiangguo Qiu. 2019. "Generation and Characterization of Anti-Filovirus Nucleoprotein Monoclonal Antibodies" Viruses 11, no. 3: 259. https://doi.org/10.3390/v11030259
APA StyleRahim, M. N., Wang, M., Wang, T., He, S., Griffin, B. D., Kobasa, D., Yang, R., Du, Z., & Qiu, X. (2019). Generation and Characterization of Anti-Filovirus Nucleoprotein Monoclonal Antibodies. Viruses, 11(3), 259. https://doi.org/10.3390/v11030259






