Priorities, Barriers, and Facilitators towards International Guidelines for the Delivery of Supportive Clinical Care during an Ebola Outbreak: A Cross-Sectional Survey
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Frame
2.2. Questionnaire Development
2.3. Questionnaire Testing
2.4. Questionnaire Administration
2.5. Statistical Analyses
2.6. Funding and Ethical Approval
2.7. Patient Involvement
3. Results
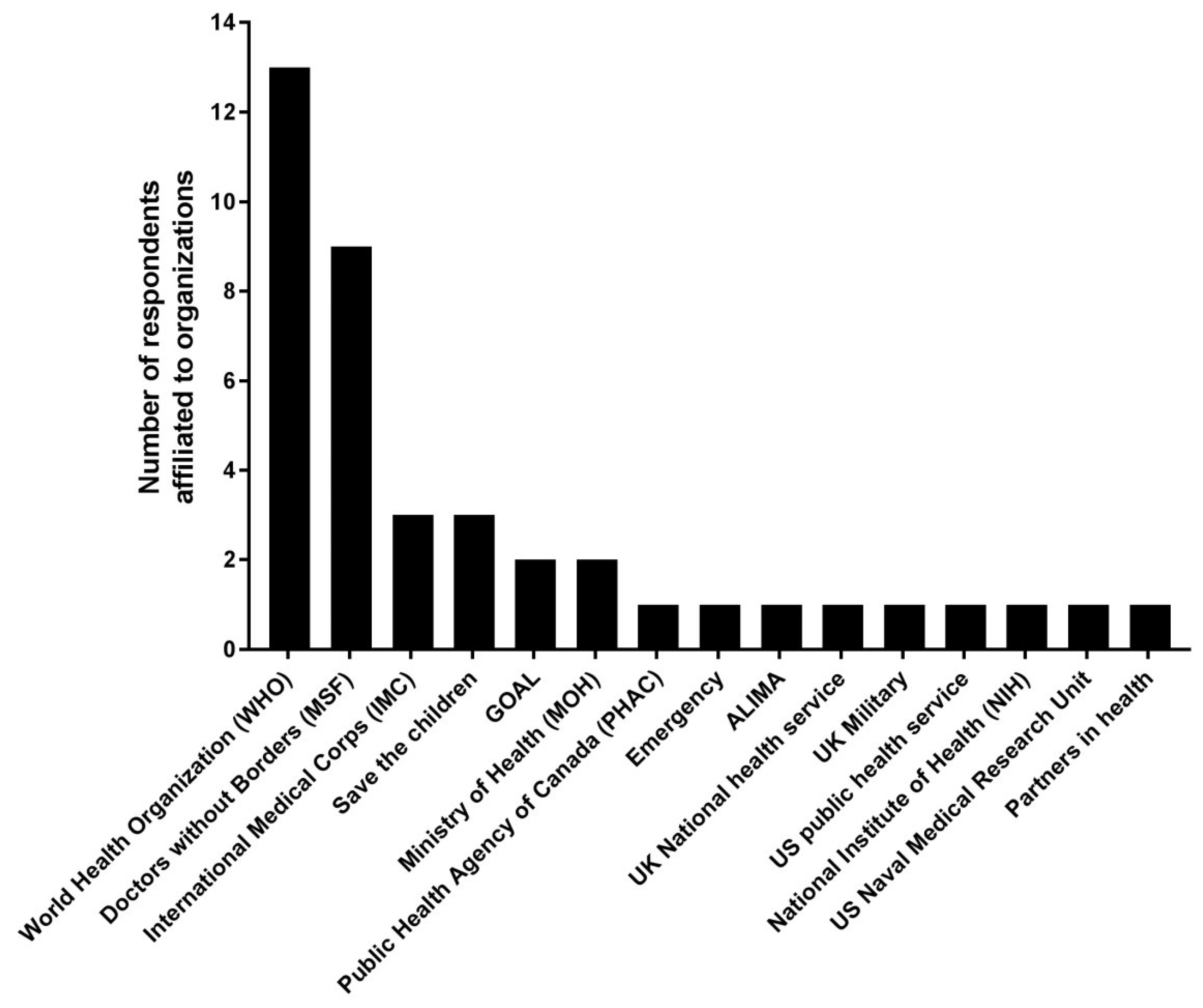
3.1. Respondent Characteristics
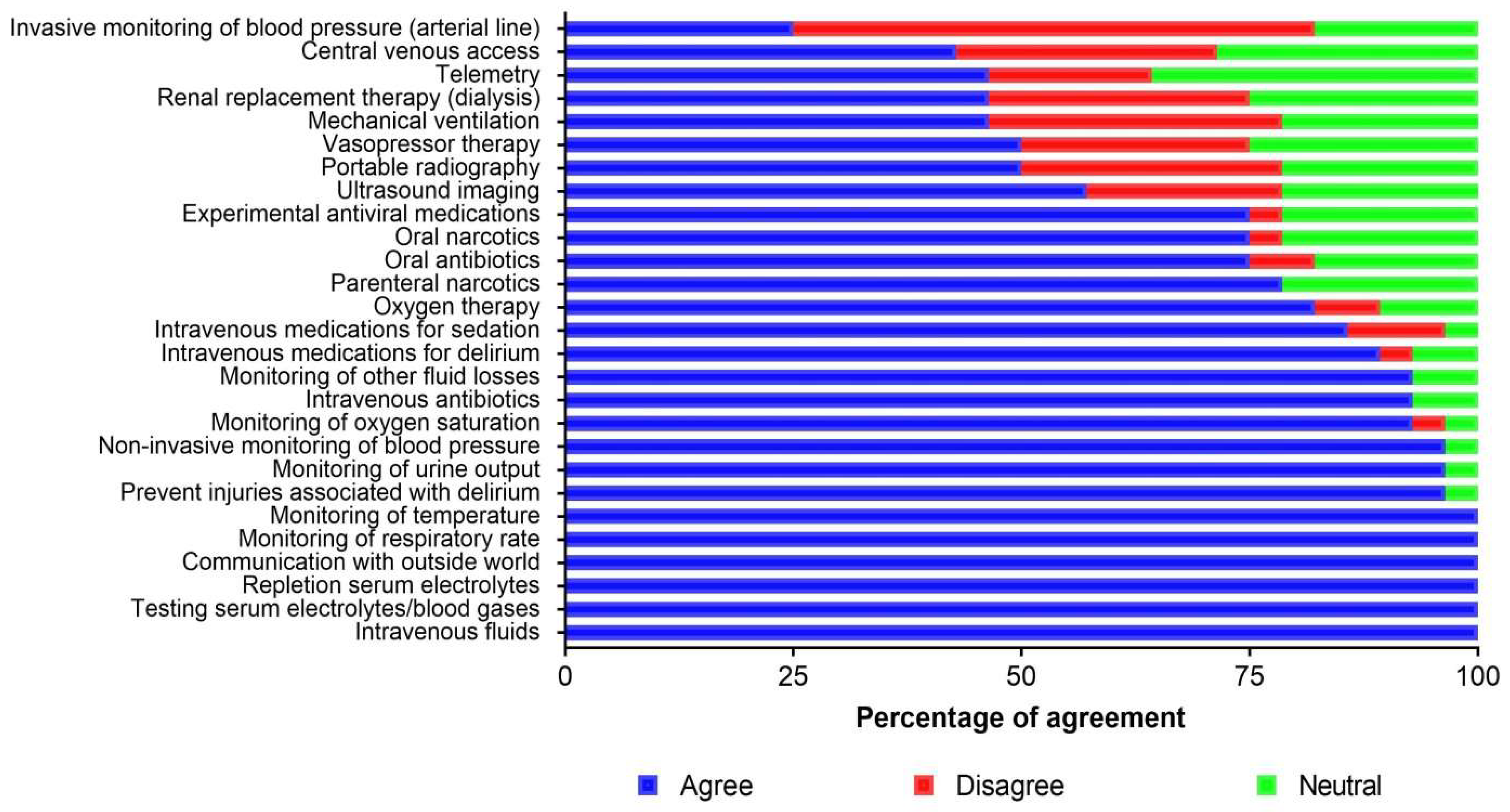
3.2. Barriers, Facilitators, and Supportive Care Priorities
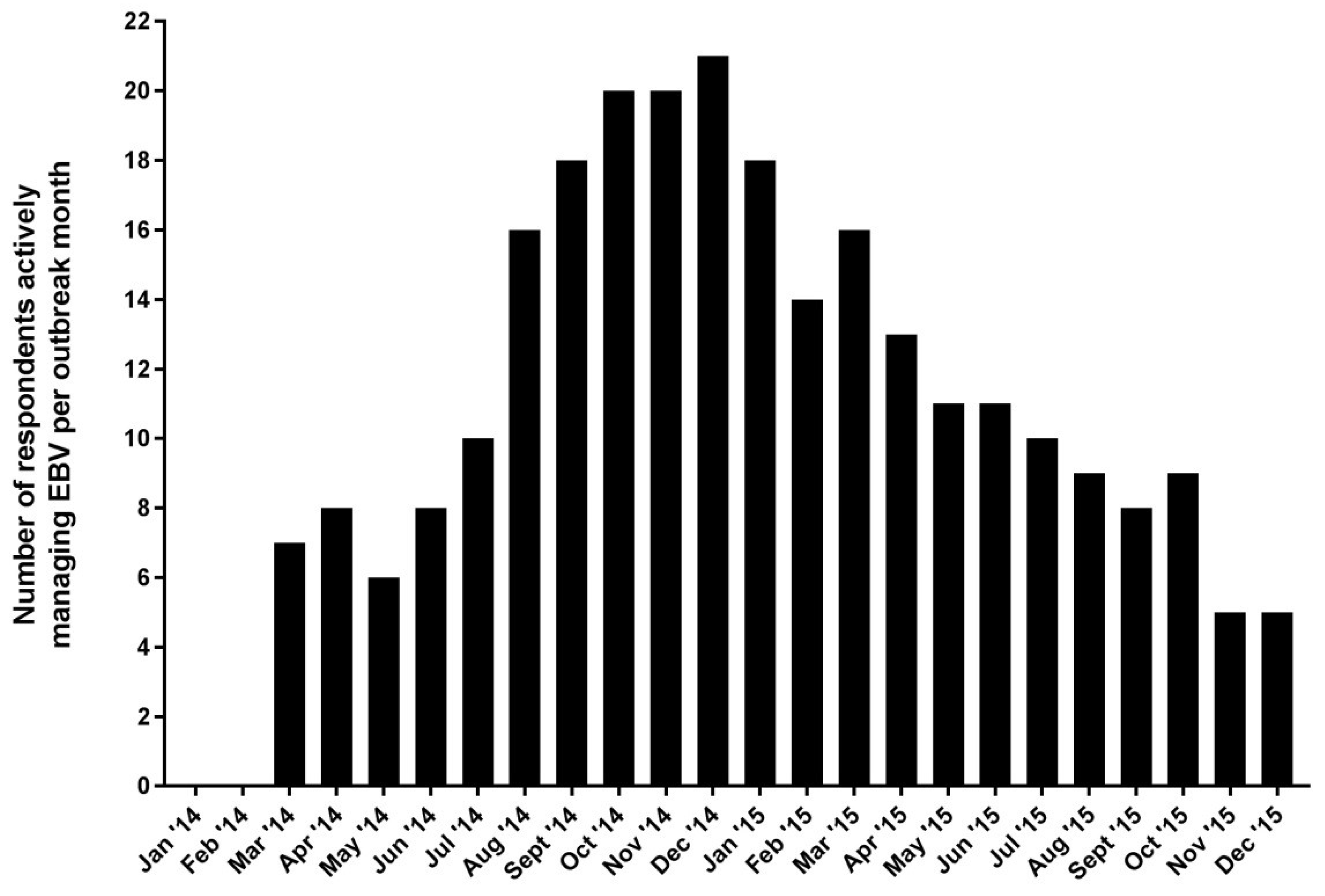
3.3. Auditing the Delivery of Care in ETUs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Brett-Major, D.M.; Jacob, S.T.; Jacquerioz, F.A.; Risi, G.F.; Fischer, W.A., II; Kato, Y.; Houlihan, C.F.; Crozier, I.; Bosa, H.K.; Lawler, J.V.; et al. Being ready to treat Ebola virus disease patients. Am. J. Trop. Med. Hyg. 2015, 92, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Fowler, R.A.; Fletcher, T.; Fischer, W.A., II; Lamontagne, F.; Jacob, S.; Brett-Major, D.; Lawler, J.V.; Jacquerioz, F.A.; Houlihan, C.; O’Dempsey, T.; et al. Caring for critically ill patients with ebola virus disease. Perspectives from West Africa. Am. J. Respir. Crit. Care. Med. 2014, 190, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne, F.; Clement, C.; Fletcher, T.; Jacob, S.T.; Fischer, W.A., II; Fowler, R.A. Doing today’s work superbly well--treating Ebola with current tools. N. Engl. J. Med. 2014, 371, 1565–1566. [Google Scholar] [CrossRef] [PubMed]
- Leligdowicz, A.; Fischer, W.A., II; Uyeki, T.M.; Fletcher, T.E.; Adhikari, N.K.; Portella, G.; Lamontagne, F.; Clement, C.; Jacob, S.T.; Rubinson, L.; et al. Ebola virus disease and critical illness. Crit. Care. 2016, 20, 217. [Google Scholar] [CrossRef] [PubMed]
- Murthy, S.; Ebola Clinical Care authors, g. Ebola and provision of critical care. Lancet 2015, 385, 1392–1393. [Google Scholar] [CrossRef]
- Greenberg, A.; Michlig, G.J.; Larson, E.; Varallyay, I.; Chang, K.; Enobun, B.; Schenk, E.; Whong, B.; Surkan, P.; Kennedy, C.E.; et al. “I Knew I Could Make a Difference”: Motivations and Barriers to Engagement in Fighting the West African Ebola Outbreak Among U.S.-Based Health Professionals. Qual. Health. Res. 2018, 29, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Rutkow, L.; Paul, A.; Taylor, H.A.; Barnett, D.J. Perceived Facilitators and Barriers to Local Health Department Workers’ Participation in Infectious Disease Emergency Responses. J. Public Health Manag. Pract. 2017, 23, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Lindblade, K.A.; Kateh, F.; Nagbe, T.K.; Neatherlin, J.C.; Pillai, S.K.; Attfield, K.R.; Dweh, E.; Barradas, D.T.; Williams, S.G.; Blackley, D.J.; et al. Decreased Ebola Transmission after Rapid Response to Outbreaks in Remote Areas, Liberia, 2014. Emerg. Infect. Dis. 2015, 21, 1800–1807. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne, F.; Fowler, R.A.; Adhikari, N.K.; Murthy, S.; Brett-Major, D.M.; Jacobs, M.; Uyeki, T.M.; Vallenas, C.; Norris, S.L.; Fischer, W.A., II; et al. Evidence-based guidelines for supportive care of patients with Ebola virus disease. Lancet 2018, 391, 700–708. [Google Scholar] [CrossRef]
- Burns, K.E.; Duffett, M.; Kho, M.E.; Meade, M.O.; Adhikari, N.K.; Sinuff, T.; Cook, D.J.; Group, A. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ 2008, 179, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Boozary, A.S.; Farmer, P.E.; Jha, A.K. The Ebola outbreak, fragile health systems, and quality as a cure. JAMA 2014, 312, 1859–1860. [Google Scholar] [CrossRef] [PubMed]
- Chertow, D.S.; Kleine, C.; Edwards, J.K.; Scaini, R.; Giuliani, R.; Sprecher, A. Ebola virus disease in West Africa--clinical manifestations and management. N. Engl. J. Med. 2014, 371, 2054–2057. [Google Scholar] [CrossRef] [PubMed]
- Loignon, C.; Nouvet, E.; Couturier, F.; Benhadj, L.; Adhikari, N.K.J.; Murthy, S.; Fowler, R.A.; Lamontagne, F. Barriers to supportive care during the Ebola virus disease outbreak in West Africa: Results of a qualitative study. PLoS ONE 2018, 13, e0201091. [Google Scholar] [CrossRef] [PubMed]
- Den Boon, S.; Vallenas, C.; Ferri, M.; Norris, S.L. Incorporating health workers’ perspectives into a WHO guideline on personal protective equipment developed during an Ebola virus disease outbreak. F1000Res. 2018, 7, 45. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Respondents (n = 29) | 1Provided consent, but did not completed questionnaire (n = 9) | 1Declined invitation or did not respond to invitation (n = 19) | ||
|---|---|---|---|---|---|
| Age–median [IQR] | 40 [34, 48] | Footnote 4 | |||
| Sex–n female (%) | 8 (28) | 1 (11) | 7 (37) | ||
| English–n (%) | 25 (86) | Footnote4 | |||
| Residents of African countries–n (%) | All countries | 11 (38) | Footnote4 | ||
| Guinea, Liberia or Sierra Leone | 5 (17) | ||||
| Expertise–n (%) | Medicine 2 | Public health & epidemiology | 5 (17) | 6 (67) | 16 (84) |
| Infectious diseases | 4 (14) | ||||
| Other (anaesthesiology, emergency medicine, pediatrics) | 3 (10) | ||||
| Unspecified | 14 (48) | ||||
| Nursing | 3 (10) | 1 (11) | 2 (11) | ||
| Project management—coordination | 3 (10) | 2 (22) | 1 (5) | ||
| Years of experience–median [IQR] | 13 [6, 17] | Footnote4 | |||
| 3Country of usual professional activities–n (%) | United States of America | 9 (31) | |||
| Sierra Leone | 8 (28) | ||||
| Guinea | 5 (17) | ||||
| United Kingdom | 5 (17) | ||||
| Uganda | 4 (14) | ||||
| Switzerland | 3 (10) | ||||
| Canada | 3 (10) | ||||
| Other (Peru, Kenya, Malawi, India, Haiti, Honduras, Ethiopia, South Sudan, Croatia, Sudan, Italy, Afghanistan, Turkey, Ireland, Australia, Belgium, Liberia, France, Senegal, Mali, Niger, Democratic Republic of Congo, Chad, Burkina Faso, Cameroon) | 8 (28) | ||||
| Number of affiliations–n (%) | Single affiliation | 19 (66) | Footnote4 | ||
| Two affiliations | 8 (28) | ||||
| Three affiliations | 2 (7) | ||||
| Main affiliation type | Governmental | 5 (17) | 3 (33) | 6 (32) | |
| Non-governmental | 24 (83) | 6 (66) | 13 (68) | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Battista, M.-C.; Loignon, C.; Benhadj, L.; Nouvet, E.; Murthy, S.; Fowler, R.; Adhikari, N.K.J.; Haj-Moustafa, A.; Salam, A.P.; Chan, A.K.; et al. Priorities, Barriers, and Facilitators towards International Guidelines for the Delivery of Supportive Clinical Care during an Ebola Outbreak: A Cross-Sectional Survey. Viruses 2019, 11, 194. https://doi.org/10.3390/v11020194
Battista M-C, Loignon C, Benhadj L, Nouvet E, Murthy S, Fowler R, Adhikari NKJ, Haj-Moustafa A, Salam AP, Chan AK, et al. Priorities, Barriers, and Facilitators towards International Guidelines for the Delivery of Supportive Clinical Care during an Ebola Outbreak: A Cross-Sectional Survey. Viruses. 2019; 11(2):194. https://doi.org/10.3390/v11020194
Chicago/Turabian StyleBattista, Marie-Claude, Christine Loignon, Lynda Benhadj, Elysee Nouvet, Srinivas Murthy, Robert Fowler, Neill K. J. Adhikari, Adnan Haj-Moustafa, Alex P. Salam, Adrienne K. Chan, and et al. 2019. "Priorities, Barriers, and Facilitators towards International Guidelines for the Delivery of Supportive Clinical Care during an Ebola Outbreak: A Cross-Sectional Survey" Viruses 11, no. 2: 194. https://doi.org/10.3390/v11020194
APA StyleBattista, M.-C., Loignon, C., Benhadj, L., Nouvet, E., Murthy, S., Fowler, R., Adhikari, N. K. J., Haj-Moustafa, A., Salam, A. P., Chan, A. K., Mishra, S., Couturier, F., Hudon, C., Horby, P., Bedell, R., Rekart, M., Hajek, J., & Lamontagne, F. (2019). Priorities, Barriers, and Facilitators towards International Guidelines for the Delivery of Supportive Clinical Care during an Ebola Outbreak: A Cross-Sectional Survey. Viruses, 11(2), 194. https://doi.org/10.3390/v11020194






