Efficacy of FDA-Approved Anti-Inflammatory Drugs Against Venezuelan Equine Encephalitis Virus Infection
Abstract
1. Introduction
2. Material and Methods
2.1. Cell Lines, Viruses, and Reagents
2.2. Viral Infections and Inhibitor Studies
2.3. Viral Plaque Assays
2.4. Cell Viability Assay
2.5. Quantitative Real-Time Polymerase Chain Reaction
2.6. Statistical Analysis
3. Results
3.1. Anti-Inflammatory Drug Toxicity in HMC3 Microglia and U87 MG Astrocyte Cells
3.2. Anti-Inflammatory Drug Efficacy in VEEV Infection
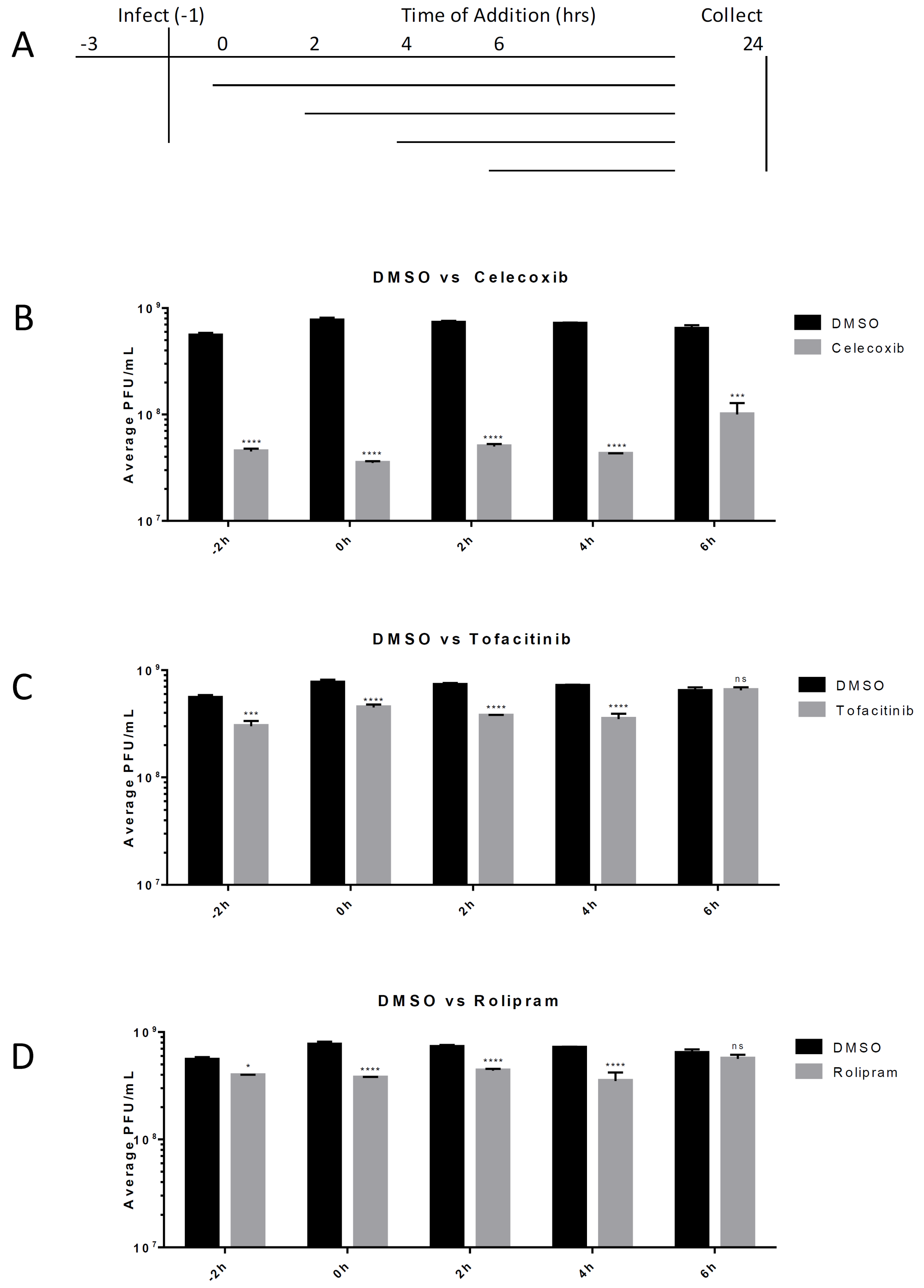
3.3. Efficacy of Inhibitors Against the Wild-Type VEEV TrD Strain
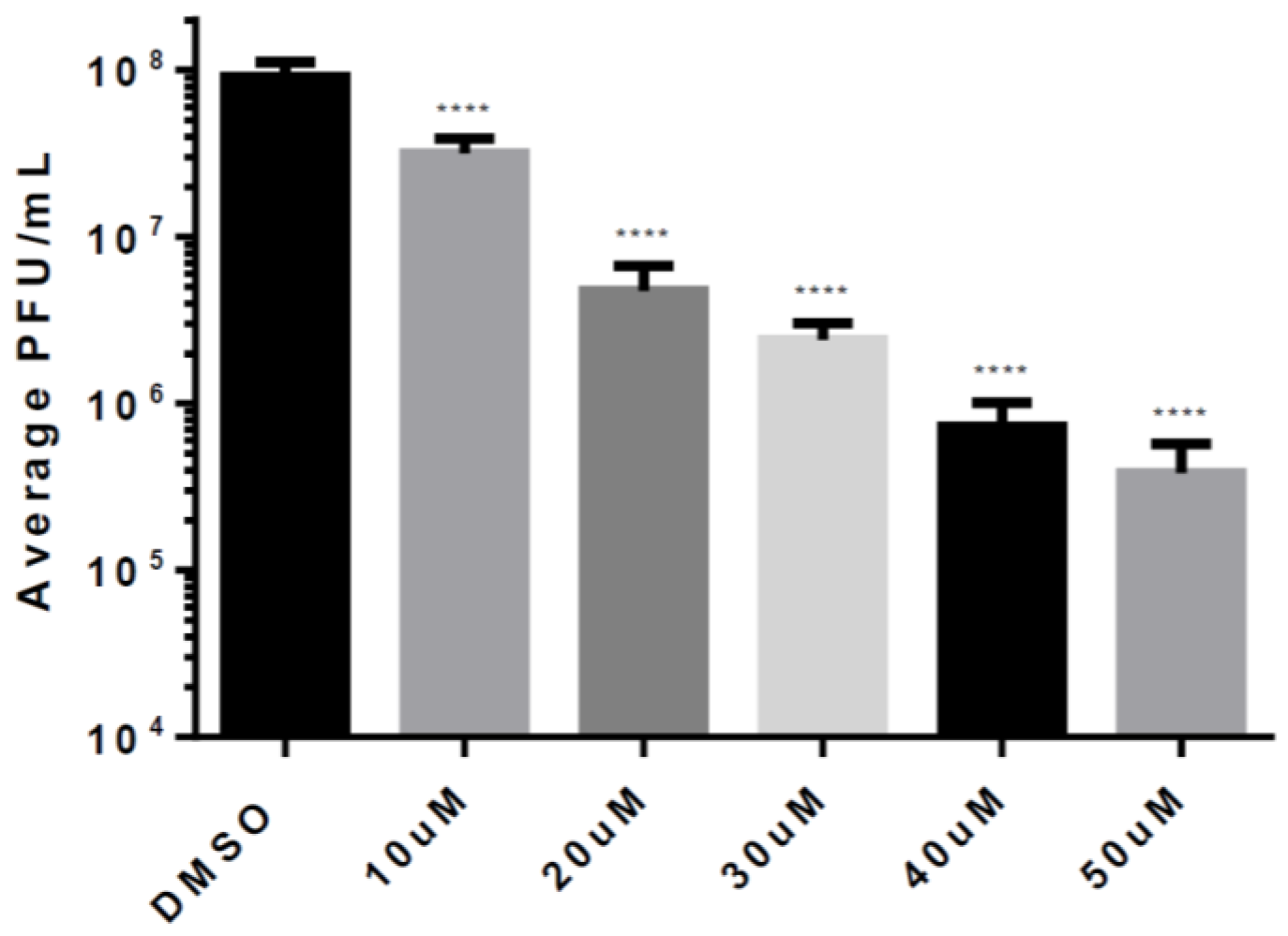
3.4. Post-Exposure Efficacy and Concentration Dependency of Celecoxib in HMC3s
3.5. Celecoxib Treatment Reduces Levels of Pro-Inflammatory Cytokines
3.6. Indirect Pro-Inflammatory Cytokines Increase in Viral Spread is Reduced by Celecoxib
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weaver, S.C.; Ferro, C.; Barrera, R.; Boshell, J.; Navarro, J.-C. Venezuelan Equine Encephalitis. Annu. Rev. Entomol. 2003, 49, 141–174. [Google Scholar] [CrossRef]
- Lundberg, L.; Carey, B.; Kehn-Hall, K. Venezuelan Equine Encephalitis Virus Capsid-The Clever Caper. Viruses 2017, 9, 279. [Google Scholar] [CrossRef]
- Rusnak, J.M.; Glass, P.J.; Weaver, S.C.; Sabourin, C.L.; Glenn, A.M.; Klimstra, W.; Badorrek, C.S.; Nasar, F.; Ward, L.A. Approach to Strain Selection and the Propagation of Viral Stocks for Venezuelan Equine Encephalitis Virus Vaccine Efficacy Testing under the Animal Rule. Viruses 2019, 11, 807. [Google Scholar] [CrossRef]
- Smith, D.R.; Aguilar, P.V.; Coffey, L.L.; Gromowski, G.D.; Wang, E.; Weaver, S.C. Venezuelan equine encephalitis virus transmission and effect on pathogenesis. Emerg. Infect. Dis. 2006, 12, 1190–1196. [Google Scholar] [CrossRef]
- Croddy, E.; Perez-Armendariz, C.; Hart, J.; Hart, J.D. Chemical and Biological Warfare: A Comprehensive Survey for the Concerned Citizen; Springer: New York, NY, USA, 2002. [Google Scholar]
- Bordi, L.; Carletti, F.; Lalle, E.; Colavita, F.; Meschi, S.; Di Caro, A.; Nicastri, E.; Scognamiglio, P.; Vairo, F.; Di Lallo, D.; et al. Molecular Characterization of Autochthonous Chikungunya Cluster in Latium Region, Italy. Emerg. Infect. Dis. 2018, 24, 178–180. [Google Scholar] [CrossRef]
- Ryman, K.D.; Klimstra, W.B. Host responses to alphavirus infection. Immunol. Rev. 2008, 225, 27–45. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Siman-Tov, G.; Keck, F.; Kortchak, S.; Bakovic, A.; Risner, K.; Lu, T.K.; Bhalla, N.; De La Fuente-Nunez, C.; Narayanan, A. Human cathelicidin peptide LL-37 as a therapeutic antiviral targeting Venezuelan equine encephalitis virus infections. Antivir. Res. 2019, 164, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Cain, M.; Salimi, H.; Gong, Y.; Yang, L.; Hamilton, S.; Heffernan, J.R.; Hou, J.; Miller, M.J.; Klein, R.S. Virus entry and replication in the brain precedes blood-brain barrier disruption during intranasal alphavirus infection. J. Neuroimmunol. 2017, 308, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Bhomia, M.; Honnold, S.P.; Maheshwari, R.K. Role of adhesion molecules and inflammation in Venezuelan equine encephalitis virus infected mouse brain. Virol. J. 2011, 8, 197. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Rossiter, J.P. Apoptotic cell death is an important cause of neuronal injury in experimental Venezuelan equine encephalitis virus infection of mice. Acta Neuropathol. 1997, 93, 349–353. [Google Scholar] [CrossRef]
- Schoneboom, B.A.; Catlin, K.M.K.; Marty, A.M.; Grieder, F.B. Inflammation is a component of neurodegeneration in response to Venezuelan equine encephalitis virus infection in mice. J. Neuroimmunol. 2000, 109, 132–146. [Google Scholar] [CrossRef]
- Phillips, A.T.; Rico, A.B.; Stauft, C.B.; Hammond, S.L.; Aboellail, T.A.; Tjalkens, R.B.; Olson, K.E. Entry Sites of Venezuelan and Western Equine Encephalitis Viruses in the Mouse Central Nervous System following Peripheral Infection. J. Virol. 2016, 90, 5785–5796. [Google Scholar] [CrossRef] [PubMed]
- Keck, F.; Kortchak, S.; Bakovic, A.; Roberts, B.; Agrawal, N.; Narayanan, A. Direct and indirect pro-inflammatory cytokine response resulting from TC-83 infection of glial cells. Virulence 2018, 9, 1403–1421. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.M.; McLachlan, A.J.; Day, R.O.; Williams, K.M. Clinical Pharmacokinetics and Pharmacodynamics of Celecoxib. Clin. Pharmacokinet. 2000, 38, 225–242. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-Y.; Yang, Y.-P.; Huang, P.-I.; Li, W.-C.; Huang, M.-C.; Kao, C.-L.; Chen, Y.-J.; Chen, M.-T. Exercise suppresses COX-2 pro-inflammatory pathway in vestibular migraine. Brain Res. Bull. 2015, 116, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Tuure, L.; Hämäläinen, M.; Moilanen, E. PDE4 inhibitor rolipram inhibits the expression of microsomal prostaglandin E synthase-1 by a mechanism dependent on MAP kinase phosphatase-1. Pharmacol. Res. Perspect. 2017, 5, e00363. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Mix, E.; Winblad, B. The Antidepressant and Antiinflammatory Effects of Rolipram in the Central Nervous System. CNS Drug Rev. 2006, 7, 387–398. [Google Scholar] [CrossRef]
- Banner, K.H.; Trevethick, M.A. PDE4 inhibition: A novel approach for the treatment of inflammatory bowel disease. Trends Pharmacol. Sci. 2004, 25, 430–436. [Google Scholar] [CrossRef]
- Li, H.; Zuo, J.; Tang, W. Phosphodiesterase-4 Inhibitors for the Treatment of Inflammatory Diseases. Front. Pharmacol. 2018, 9, 1–21. [Google Scholar] [CrossRef]
- Rodig, S.J.; Meraz, M.A.; White, J.M.; Lampe, P.A.; Riley, J.K.; Arthur, C.D.; King, K.L.; Sheehan, K.C.F.; Yin, L.; Pennica, D.; et al. Disruption of the Jak1 Gene Demonstrates Obligatory and Nonredundant Roles of the Jaks in Cytokine-Induced Biologic Responses. Cell 1998, 93, 373–383. [Google Scholar] [CrossRef]
- Julander, J.G.; Skirpstunas, R.; Siddharthan, V.; Shafer, K.; Hoopes, J.D.; Smee, D.F.; Morrey, J.D. C3H/HeN mouse model for the evaluation of antiviral agents for the treatment of Venezuelan equine encephalitis virus infection. Antivir. Res. 2008, 78, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Keck, F.; Khan, D.; Roberts, B.; Agrawal, N.; Bhalla, N.; Narayanan, A. Mitochondrial-Directed Antioxidant Reduces Microglial-Induced Inflammation in Murine In Vitro Model of TC-83 Infection. Viruses 2018, 10, 606. [Google Scholar] [CrossRef] [PubMed]
- Cabezas, R.; ÃVila, M.; Gonzalez, J.; El-Bachã, R.S.; Bãez, E.; GarcãA-Segura, L.M.; Jurado Coronel, J.C.; Capani, F.; Cardona-Gomez, G.P.; Barreto, G.E. Astrocytic modulation of blood brain barrier: Perspectives on Parkinson’s disease. Front. Cell. Neurosci. 2014, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, R.S.; Kothari, S.T.; Gohil, D.J.; D’Souza, M.; Chowdhary, A.S. Novel evidence of microglial immune response in impairment of Dengue infection of CNS. Immunobiology 2015, 220, 1170–1176. [Google Scholar] [CrossRef]
- Das, T.; Hoarau, J.J.; Bandjee, M.C.J.; Maquart, M.; Gasque, P. Multifaceted innate immune responses engaged by astrocytes, microglia and resident dendritic cells against Chikungunya neuroinfection. J. Gen. Virol. 2015, 96, 294–310. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Risner, K.; Ahmed, A.; Bakovic, A.; Kortchak, S.; Bhalla, N.; Narayanan, A. Efficacy of FDA-Approved Anti-Inflammatory Drugs Against Venezuelan Equine Encephalitis Virus Infection. Viruses 2019, 11, 1151. https://doi.org/10.3390/v11121151
Risner K, Ahmed A, Bakovic A, Kortchak S, Bhalla N, Narayanan A. Efficacy of FDA-Approved Anti-Inflammatory Drugs Against Venezuelan Equine Encephalitis Virus Infection. Viruses. 2019; 11(12):1151. https://doi.org/10.3390/v11121151
Chicago/Turabian StyleRisner, Kenneth, Aslaa Ahmed, Allison Bakovic, Stephanie Kortchak, Nishank Bhalla, and Aarthi Narayanan. 2019. "Efficacy of FDA-Approved Anti-Inflammatory Drugs Against Venezuelan Equine Encephalitis Virus Infection" Viruses 11, no. 12: 1151. https://doi.org/10.3390/v11121151
APA StyleRisner, K., Ahmed, A., Bakovic, A., Kortchak, S., Bhalla, N., & Narayanan, A. (2019). Efficacy of FDA-Approved Anti-Inflammatory Drugs Against Venezuelan Equine Encephalitis Virus Infection. Viruses, 11(12), 1151. https://doi.org/10.3390/v11121151