Insight into the Tropism of Dengue Virus in Humans
Abstract
1. Importance
2. Introduction
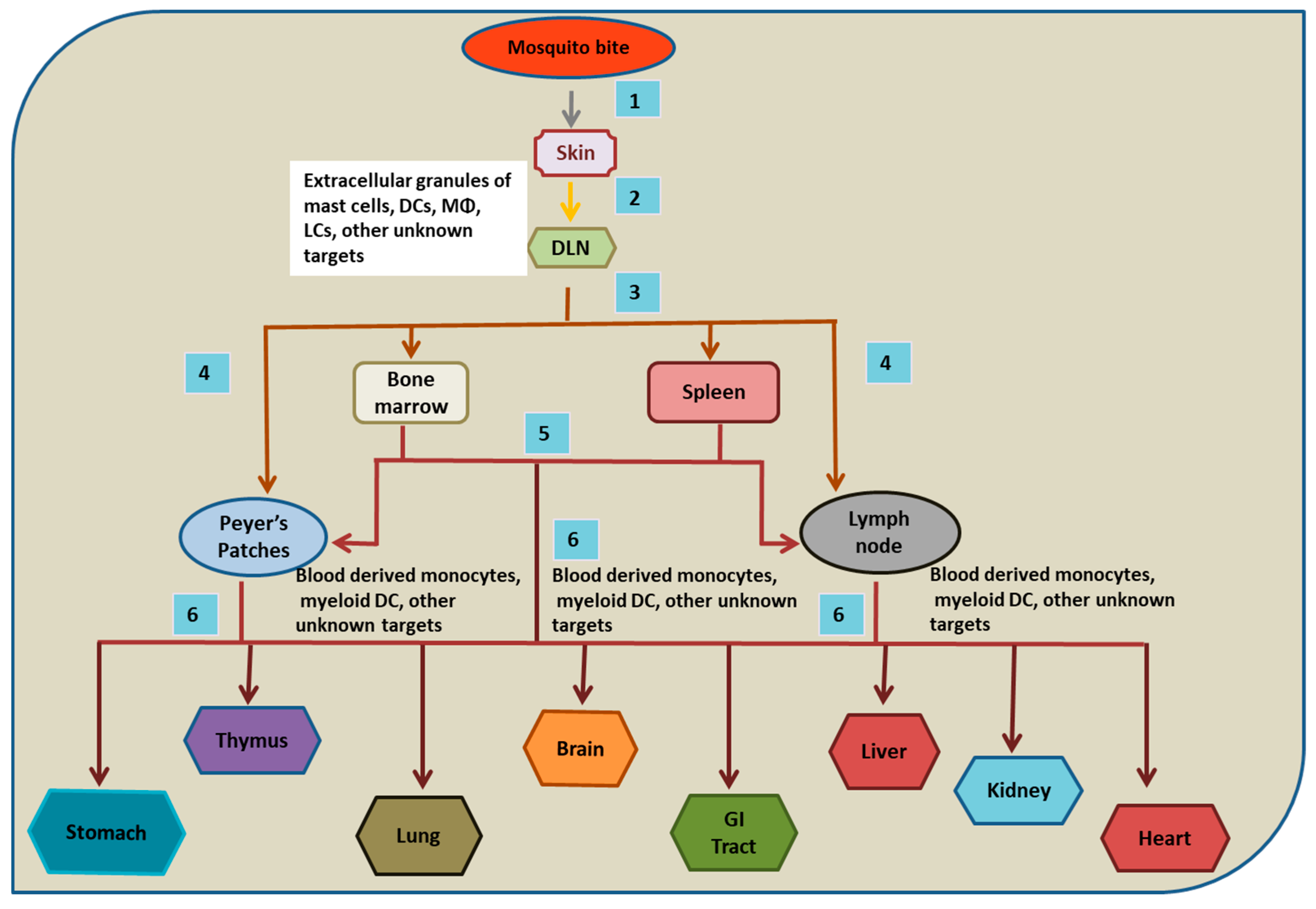
3. Dengue Tropism in Humans
4. Skin
5. Lymph Node and Thymus
6. Spleen
7. Liver
8. Bone Marrow
9. Central Nervous System (CNS)
10. Kidney
11. Lungs
12. Heart
13. Tropism in Mouse
14. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Santos Souza, H.F.; da Silva Almeida, B.; Boscardin, S.B. Early dengue virus interactions: The role of dendritic cells during infection. Virus Res. 2016, 223, 88–98. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Kuhn, R.J.; Rossmann, M.G. A structural perspective of the flavivirus life cycle. Nat. Rev. Microbiol 2005, 3, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Martina, B.E.E.; Koraka, P.; Osterhaus, A.D.M.E. Dengue virus pathogenesis: An integrated view. Clin. Microbiol. Rev. 2009, 22, 564–581. [Google Scholar] [CrossRef] [PubMed]
- WHO. Dengue Control Epidemiology; World Health Organization: Geneva, Switzerland, 2017; Available online: http://www.who.int/ (accessed on 31 August 2018).
- Sun, P.; Kochel, T.J. The battle between infection and host immune responses of dengue virus and its implication in dengue disease pathogenesis. Sci. World J. 2013, 2013, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ng, D.; Ghosh, A.; Jit, M.; Seneviratne, S.L. Mini-review: Can non-human leucocyte antigen genes determine susceptibility to severe dengue syndromes? Trans. R. Soc. Trop. Med. Hyg. 2017, 111, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Simmons, C.P.; Farrar, J.J.; Chau, N.v.V.; Wills, B. Dengue. N. Engl. J. Med. 2012, 366, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Kurane, I. Dengue hemorrhagic fever with special emphasis on immunopathogenesis. Comp. Immunol. Microbiol. Infect. Dis. 2007, 30, 329–340. [Google Scholar] [CrossRef] [PubMed]
- WHO. Dengue Haemorrhagic Fever: Diagnosis, Treatment and Control; World Health Organization: Geneva, Switzerland, 1997; pp. 12–23. [Google Scholar]
- Noisakran, S.; Onlamoon, N.; Songprakhon, P.; Hsiao, H.M.; Chokephaibulkit, K.; Perng, G.C. Cells in dengue virus infection in vivo. Adv. Virol. 2010, 2010, 1–15. [Google Scholar] [CrossRef]
- Acosta, E.G.; Talarico, L.B.; Damonte, E.B. Cell entry of dengue virus. Future Virol. 2008, 3, 471–479. [Google Scholar] [CrossRef]
- Cruz-Oliveira, C.; Freire, J.M.; Conceição, T.M.; Higa, L.M.; Castanho, M.A.; Da Poian, A.T. Receptors and routes of dengue virus entry into the host cells. FEMS Microbiol. Rev. 2015, 39, 155–170. [Google Scholar] [CrossRef]
- Kangwanpong, D.; Bhamarapravati, N.; Lucia, H.L. Diagnosing dengue virus infection in archived autopsy tissues by means of the in situ PCR method: A case report. Clin. Diagn. Virol. 1995, 3, 165–172. [Google Scholar] [CrossRef]
- Miagostovich, M.P.; Ramos, R.G.; Nicol, A.F.; Nogueira, R.M.; Cuzzi-Maya, T.; Oliveira, A.V.; Marchevsky, R.S.; Mesquita, R.P.; Schatzmayr, H.G. Retrospective study on dengue fatal cases. Clin. Neuropathol. 1997, 16, 204–208. [Google Scholar] [PubMed]
- Jessie, K.; Fong, M.Y.; Devi, S.; Lam, S.K.; Wong, K.T. Localization of dengue virus in naturally infected human tissues, by immunohistochemistry and in situ hybridization. J. Infect. Dis. 2004, 189, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Kyle, J.L.; Beatty, P.R.; Harris, E. Dengue virus infects macrophages and dendritic cells in a mouse model of infection. J. Infect. Dis. 2007, 195, 1808–1817. [Google Scholar] [CrossRef]
- Durbin, A.P.; Vargas, M.J.; Wanionek, K.; Hammond, S.N.; Gordon, A.; Rocha, C.; Balmaseda, A.; Harris, E. Phenotyping of peripheral blood mononuclear cells during acute dengue illness demonstrates infection and increased activation of monocytes in severe cases compared to classic dengue fever. Virology 2008, 376, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Balsitis, S.J.; Coloma, J.; Castro, G.; Alava, A.; Flores, D.; McKerrow, J.H.; Beatty, P.R.; Harris, E. Tropism of dengue virus in mice and humans defined by viral nonstructural protein 3-Specific immunostaining. Am. J. Trop. Med. Hyg. 2009, 80, 416–424. [Google Scholar] [CrossRef]
- Prestwood, T.R.; May, M.M.; Plummer, E.M.; Morar, M.M.; Yauch, L.E.; Shresta, S. Trafficking and replication patterns reveal splenic macrophages as major targets of dengue virus in mice. J. Virol. 2012, 86, 12138–12147. [Google Scholar] [CrossRef]
- Póvoa, T.F.; Alves, A.M.B.; Oliveira, C.A.B.; Nuovo, G.J.; Chagas, V.L.A.; Paes, M.V. The pathology of severe dengue in multiple organs of human fatal cases: Histopathology, ultrastructure and virus replication. PLoS ONE 2014, 9, 1–16. [Google Scholar] [CrossRef]
- Cerny, D.; Haniffa, M.; Shin, A.; Bigliardi, P.; Tan, B.K.; Lee, B.; Poidinger, M.; Tan, E.Y.; Ginhoux, F.; Fink, K. Selective susceptibility of human skin antigen presenting cells to productive dengue virus infection. PLoS Pathog. 2014, 10, 1–15. [Google Scholar] [CrossRef]
- Wang, X.N.; McGovern, N.; Gunawan, M.; Richardson, C.; Windebank, M.; Siah, T.; Lim, H.; Fink, K.; Li, J.L.Y.; Ng, L.G.; et al. A three-dimensional atlas of human dermal leukocytes, lymphatics, and blood vessels. J. Investig. Dermatol. 2014, 134, 965–974. [Google Scholar] [CrossRef]
- Nestle, F.O.; Di Meglio, P.; Qin, J.Z.; Nickoloff, B.J. Skin immune sentinels in health and disease. Nat. Rev. Immunol. 2009, 9, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Briant, L.; Desprès, P.; Choumet, V.; Missé, D. Role of skin immune cells on the host susceptibility to mosquito-borne viruses. Virology 2014, 464, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.J.; Grouard-Vogel, G.; Sun, W.; Mascola, J.R.; Brachtel, E.; Putvatana, R.; Louder, M.K.; Filgueira, L.; Marovich, M.A.; Wong, H.K.; et al. Human skin Langerhans cells are targets of dengue virus infection. Nat. Med. 2000, 6, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.J.; Wang, J.J.; Shaio, M.F.; Kao, C.L.; Chang, D.M.; Han, S.W.; Lai, J.H. Infection of human dendritic cells by dengue virus causes cell maturation and cytokine production. J. Immunol. 2001, 166, 1499–1506. [Google Scholar] [CrossRef]
- Schaeffer, E.; Flacher, V.; Papageorgiou, V.; Decossas, M.; Fauny, J.D.; Kra¨mer, M.; Mueller, C.G. Dermal CD14 + dendritic cell and macrophage infection by dengue virus is stimulated by Interleukin-4. J. Investig. Dermatol. 2015, 135, 1743–1751. [Google Scholar] [CrossRef]
- Duangkhae, P.; Erdos, G.; Ryman, K.D.; Watkins, S.C.; Falo, L.D.; Marques, E.T.A.; Barratt-Boyes, S.M. Interplay between Keratinocytes and Myeloid Cells Drives Dengue Virus Spread in Human Skin. J. Investig. Dermatol. 2018, 138, 618–626. [Google Scholar] [CrossRef]
- Brown, M.G.; Hermann, L.L.; Issekutz, A.C.; Marshall, J.S.; Rowter, D.; Al-Afif, A.; Anderson, R. Dengue Virus Infection of Mast Cells Triggers Endothelial Cell Activation. J. Virol. 2010, 85, 1145–1150. [Google Scholar] [CrossRef]
- Brown, M.G.; King, C.A.; Sherren, C.; Marshall, J.S.; Anderson, R.A. Dominant role for FcγRII in antibody-enhanced dengue virus infection of human mast cells and associated CCL5 release. J. Leukoc. Biol. 2006, 80, 1242–1250. [Google Scholar] [CrossRef]
- Troupin, A.; Shirley, D.; Londono-Renteria, B.; Watson, A.M.; McHale, C.; Hall, A.; Hartstone-Rose, A.; Klimstra, W.B.; Gomez, G.; Colpitts, T.M. A Role for Human Skin Mast Cells in Dengue Virus Infection and Systemic Spread. J. Immunol. 2016, 197, 4382–4391. [Google Scholar] [CrossRef]
- Schmid, M.A.; Harris, E. Monocyte Recruitment to the Dermis and Differentiation to Dendritic Cells Increases the Targets for Dengue Virus Replication. PLoS Pathog. 2014, 10, 1–18. [Google Scholar] [CrossRef]
- Merad, M.; Manz, M.G.; Karsunky, H.; Wagers, A.; Peters, W.; Charo, I.; Weissman, I.L.; Cyster, J.G.; Engleman, E.G. Langerhans cells renew in the skin throughout life under steady-state conditions. Nat. Immunol. 2002, 3, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Bhoopat, L.; Bhamarapravati, N.; Attasiri, C.; Yoksarnl, S.; Chaiwun, B.; Khunamornpong, S.; Sirisanthana, V. Immunohistochemical characterization of a new monoclonal antibody reactive with dengue virus-infected cells in frozen tissue using immunoperoxidase technique. Asian Pac. J. Allergy Immunol. 1996, 14, 107–113. [Google Scholar] [PubMed]
- Aye, K.S.; Charngkaew, K.; Win, N.; Wai, K.Z.; Moe, K.; Punyadee, N.; Thiemmeca, S.; Suttitheptumrong, A.; Sukpanichnant, S.; Malasit, P.; et al. Pathologic highlights of dengue hemorrhagic fever in 13 autopsy cases from Myanmar. Hum. Pathol. 2014, 45, 1221–1233. [Google Scholar] [CrossRef] [PubMed]
- Killen, H.; O’sullivan, M.A. Detection of dengue virus by in situ hybridization. J. Virol. Methods 1993, 41, 135–146. [Google Scholar] [CrossRef]
- Blackley, S.; Kou, Z.; Chen, H.; Quinn, M.; Rose, R.C.; Schlesinger, J.J.; Coppage, M.; Jin, X. Primary human splenic macrophages, but not T or B Cells, are the principal target cells for dengue virus infection in vitro. J. Virol. 2007, 81, 13325–13334. [Google Scholar] [CrossRef][Green Version]
- Butt, N.; Abbassi, A.; Munir, S.M.; Ahmad, S.M.; Sheikh, Q.H. Haematological and biochemical indicators for the early diagnosis of dengue viral infection. J. Coll. Physicians Surg. Pak. 2008, 18, 282–285. [Google Scholar]
- Faridi, M.M.A.; Aggarwal, A.; Kumar, M.; Sarafrazul, A. Clinical and biochemical profile of dengue haemorrhagic fever in children in Delhi. Trop. Dr. 2008, 38, 28–30. [Google Scholar] [CrossRef]
- Mohan, B.; Patwari, A.K.; Anand, V.K. Hepatic dysfunction in childhood dengue infection. J. Trop. Pediatric 2000, 46, 40–43. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Lei, H.Y.; Nguyen, T.L.; Lin, Y.S.; Huang, K.J.; Lien, L.B.; Lin, C.F.; Yeh, T.M.; Ha, D.Q.; Huong, V.T.Q.; et al. Dengue hemorrhagic fever in infants: A study of clinical and cytokine profiles. J. Infect. Dis. 2004, 189, 221–232. [Google Scholar]
- Pancharoen, C.; Mekmullica, J.; Thisyakorn, U. Primary dengue infection: What are the clinical distinctions from secondary infection? Southeast Asian J. Trop. Med. Public Health 2001, 32, 476–480. [Google Scholar]
- Shah, G.S.; Islam, S.; Das, B.K. Clinical and laboratory profile of dengue infection in children. Kathmandu Univ. Med. J. 2006, 4, 40–43. [Google Scholar]
- Wahid, S.F.; Sanusi, S.; Zawawi, M.; Ali, R.A. A comparison of the pattern of liver involvement in dengue hemorrhagic fever with classic dengue fever. Southeast Asian J. Trop. Med. Public Health 2000, 31, 259–263. [Google Scholar] [PubMed]
- Ahmed, S.; Ali, N.; Ashraf, S.; Ilyas, M.; Tariq, W.U.; Chotani, R.A. Dengue fever outbreak: A clinical management experience. J. Coll. Physicians Surg. Pak. 2008, 18, 8–12. [Google Scholar] [PubMed]
- Itha, S.; Kashyap, R.; Krishnani, N.; Saraswat, V.A.; Choudhuri, G.; Aggarwal, R. Profile of liver involvement in dengue virus infection. Natl. Med. J. India 2005, 18, 127–130. [Google Scholar] [PubMed]
- Sai, P.M.V.; Dev, B.; Krishnan, R. Role of ultrasound in dengue fever. Br. J. Radiol. 2005, 78, 416–418. [Google Scholar]
- Pancharoen, C.; Rungsarannont, A.; Thisyakorn, U. Hepatic dysfunction in dengue patients with various severity. J. Med. Assoc. Thai. 2002, 85, 85. [Google Scholar]
- Kalayanarooj, S.; Vaughn, D.W.; Nimmannitya, S.; Green, S.; Suntayakorn, S.; Kunentrasai, N.; Viramitrachai, W.; Ratanachu-eke, S.; Kiatpolpoj, S.; Innis, B.L.; et al. Early Clinical and laboratory indicators of acute dengue illness. J. Infect. Dis. 1997, 176, 313–321. [Google Scholar] [CrossRef]
- Basílio-de-Oliveira, C.A.; Aguiar, G.R.; Baldanza, M.S.; Barth, O.M.; Eyer-Silva, W.A.; Paes, M.V. Pathologic study of a fatal case of dengue-3 virus infection in Rio de Janeiro, Brazil. Braz. J. Infect. Dis. 2005, 9, 341–347. [Google Scholar] [CrossRef]
- Couvelard, A.; Marianneau, P.; Bedel, C.; Drouet, M.T.; Vachon, F.; Henin, D.; Deubel, V. Report of a fatal case of dengue infection with hepatitis: Demonstration of dengue antigens in hepatocytes and liver apoptosis. Hum. Pathol. 1999, 30, 1106–1110. [Google Scholar] [CrossRef]
- De Macedo, F.C.; Nicol, A.F.; Cooper, L.D.; Yearsley, M.; Pires, A.R.; Nuovo, G.J. Histologic, viral, and molecular correlates of dengue fever infection of the liver using highly sensitive immunohistochemistry. Diagn. Mol. Pathol. 2006, 15, 223–228. [Google Scholar] [CrossRef]
- Hall, W.C.; Crowell, T.P.; Watts, D.M.; Barros, V.L.; Kruger, H.; Pinheiro, F.; Peters, C.J. Demonstration of yellow fever and dengue antigens in formalin-fixed paraffin-embedded human liver by immunohistochemical analysis. Am. J. Trop. Med. Hyg. 1991, 45, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Huerre, M.R.; Lan, N.T.; Marianneau, P.; Hue, N.B.; Khun, H.; Hung, N.T.; Khen, N.T.; Drouet, M.T.; Huong, V.T.; Ha, D.Q.; et al. Liver histopathology and biological correlates in five cases of fatal dengue fever in Vietnamese children. Virchows Arch. 2001, 438, 107–115. [Google Scholar] [PubMed]
- Limonta, D.; Capo, V.; Torres, G.; Perez, A.B.; Guzman, M.G. Apoptosis in tissues from fatal dengue shock syndrome. J. Clin. Virol. 2007, 40, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Miagostovich, M.P.; dos Santos, F.B.; de Araújo, E.S.; Dias, J.; Schatzmayr, H.G.; Nogueira, R.M. Diagnosis of Dengue by Using Reverse Transcriptase-Polymerase Chain Reaction. MemóriasInst. Oswaldo Cruz 1997, 92, 595–600. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marianneau, P.; Steffan, A.M.; Royer, C.; Drouet, M.T.; Jaeck, D.; Kirn, A.; Deubel, V. Infection of primary cultures of human Kupffer cells by Dengue virus, no viral progeny synthesis, but cytokine production is evident. J. Virol. 1999, 73, 5201–5206. [Google Scholar] [PubMed]
- Weiss, H.J.; Halstead, S.B. Studies of hemostasis in Thai hemorrhagic fever. J. Pediatric 1965, 66, 918–926. [Google Scholar] [CrossRef]
- La Russa, V.F.; Innis, B.L. Mechanisms of dengue virus induced bone marrow suppression. Baill. Clin. Haematol. 1995, 8, 249–270. [Google Scholar] [CrossRef]
- Nisalak, A.; Halstead, S.B.; Singharaj, P.; Udomsakdi, S.; Nye, S.W.; Vinijchaikul, K. Observations related to pathogenesis of dengue hemorrhagic fever. 3. Virologic studies of fatal disease. Yale J. Biol. Med. 1970, 42, 293–310. [Google Scholar]
- De Araújo, J.M.; Schatzmayr, H.G.; De Filippis, A.M.; Dos Santosa, F.B.; Cardoso, M.A.; Britto, C.; Coelho, J.M.C.O.; Nogueira, R.M.R. A retrospective survey of dengue virus infection in fatal cases from an epidemic in Brazil. J. Virol. Methods 2009, 155, 34–38. [Google Scholar] [CrossRef]
- Cattoretti, G.; Schiró, R.; Orazi, A.; Soligo, D.; Colombo, M.P. Bone marrow stroma in humans: Anti-nerve growth factor receptor antibodies selectively stain reticular cells in vivo and in vitro. Blood 1993, 81, 1726–1738. [Google Scholar] [CrossRef]
- Rothwell, S.W.; Putnak, R.; La Russa, V.F. Dengue-2 virus infection of human bone marrow: Characterization of dengue-2 antigen-positive stromal cells. Am. J. Trop. Med. Hyg. 1996, 54, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Nakao, S.; Lai, C.J.; Young, N.S. Dengue virus, a flavivirus, propagates in human bone marrow progenitors and hematopoietic cell lines. Blood 1989, 74, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, M.S.; Clavell, L.A. First report of dengue virus infection in a bone marrow transplant patient. Blood 2005, 106, 5331. [Google Scholar]
- Puccioni-Sohler, M.; Soares, C.N.; Papaiz-Alvarenga, R.; Castro, M.J.; Faria, L.C.; Peralta, J.M. Neurologic dengue manifestations associated with intrathecal specific immune response. Neurology 2009, 73, 1413–1417. [Google Scholar] [CrossRef]
- Carod-Artal, F.J.; Wichmann, O.; Farrar, J.; Gascón, J. Neurological complications of dengue virus infection. Lancet Neurol. 2013, 12, 906–919. [Google Scholar] [CrossRef]
- Cam, B.V.; Fonsmark, L.; Hue, N.B.; Phuong, N.T.; Poulsen, A.; Heegaard, E.D. Prospective case-control study of encephalopathy in children with dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 2001, 65, 848–851. [Google Scholar] [CrossRef]
- Nogueira, R.M.; Schatzmayr, H.G.; De Filippis, A.M.; Dos Santos, F.B.; Da Cunha, R.V.; Coelho, J.O.; De Souza, L.J.; Guimarães, F.R.; De Araújo, E.S.M.; De Simone, T.S.; et al. Dengue virus type 3, Brazil, 2002. Emerg. Infect. Dis. 2005, 11, 1376–1381. [Google Scholar] [CrossRef]
- Domingues, R.B.; Kuster, G.W.; Onuki-Castro, F.L.; Souza, V.A.; Levi, J.E.; Pannuti, C.S. Involvement of the central nervous system in patients with dengue virus infection. J. Neurol. Sci. 2008, 267, 36–40. [Google Scholar] [CrossRef]
- Kumar, R.; Tripathi, S.; Tambe, J.J.; Arora, V.; Srivastava, A.; Nag, V.L. Dengue encephalopathy in children in Northern India: Clinical features and comparison with non dengue. J. Neurol. Sci. 2008, 269, 41–48. [Google Scholar] [CrossRef]
- Araújo, F.M.; Araújo, M.S.; Nogueira, R.M.; Brilhante, R.S.N.; Oliveira, D.N.; Rocha, M.F.G.; Cordeiro, R.A.; Arau´jo, R.M.C.; Sidrim, J.J.C. Central nervous system involvement in dengue, A study in fatal cases from a dengue endemic area. Neurology 2012, 78, 736–742. [Google Scholar] [CrossRef]
- Li, G.H.; Ning, Z.J.; Liu, Y.M.; Li, X.H. Neurological manifestations of dengue infection. Lancet 2000, 355, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Ramos, C.; Sánchez, G.; Pando, R.H.; Baquera, J.; Hernandez, D.; Mota, J.; Ramos, J.; Flores, A.; LlausBs, E. Dengue virus in the brain of a fatal case of hemorrhagic dengue fever. J. Neurovirol. 1998, 4, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Janssen, H.L.; Bienfait, H.P.; Jansen, C.L.; Van Duinen, S.G.; Vriesendorp, R.; Schimsheimer, R.J.; Groen, J.; Osterhaus, A.D.M.E. Fatal cerebral oedema associated with primary dengue infection. J. Infect. 1998, 36, 344–346. [Google Scholar] [CrossRef]
- Lombardi, R.; Yu, L.; Younes-Ibrahim, M.; Schor, N.; Burdmann, E.A. Epidemiology of Acute Kidney Injury in Latin America. Semin. Nephrol. 2008, 28, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.A.M.; Tan, J.; Khalil, M.A.U.; Awan, S.; Rangasami, M. Predictors of hospital stay and mortality in dengue virus infection-experience from Aga Khan University Hospital Pakistan. BMC Res. Notes 2014, 7, 1–7. [Google Scholar] [CrossRef]
- Mohsin, N.; Mohamed, E.; Gaber, M.; Obaidani, I.; Budruddin, M.; Al Busaidy, S. Acute tubular necrosis associated with non-hemorrhagic dengue fever: A case report. Ren. Fail. 2009, 31, 736–739. [Google Scholar] [CrossRef]
- Repizo, L.P.; Malheiros, D.M.; Yu, L.; Barros, R.T.; Burdmann, E.A. Biopsy Proven Acute Tubular Necrosis Due To Rhabdomyolysis in a Dengue Fever Patient: A Case Report and Review of Literature. Rev. Inst. Med. Trop. Sao Paulo 2014, 56, 85–88. [Google Scholar] [CrossRef]
- Upadhaya, B.K.; Sharma, A.; Khaira, A.; Dinda, A.K.; Agarwal, S.K.; Tiwari, S.C. Transient IgA nephropathy with acute kidney injury in a patient with dengue fever. Saudi J. Kidney Dis. Transpl. 2010, 21, 521–525. [Google Scholar]
- Lizarraga, K.J.; Nayer, A. Dengue-associated kidney disease. J. Nephropathol. 2014, 3, 57–62. [Google Scholar]
- Bhagat, M.; Zaki, S.A.; Sharma, S.; Manglani, M.V. Acute glomerulonephritis in dengue haemorrhagic fever in the absence of shock, sepsis, haemolysis or rhabdomyolysis. Paediatric Int. Child Health 2012, 32, 161–163. [Google Scholar] [CrossRef]
- Lima, E.Q.; Gorayeb, F.S.; Zanon, J.R.; Nogueira, M.L.; Ramalho, H.J.; Burdmann, E.A. Dengue haemorrhagic fever-induced acute kidney injury without hypotension, haemolysis or rhabdomyolysis. Nephrol. Dial. Transpl. 2007, 22, 3322–3326. [Google Scholar] [CrossRef]
- Nair, V.R.; Unnikrishnan, D.; Satish, B.; Sahadulla, M.I. Acute renal failure in dengue fever in the absence of bleeding manifestations or shock. Infect. Dis. Clin. Pract. 2005, 13, 142–143. [Google Scholar] [CrossRef]
- Bosch, X.; Poch, E.; Grau, J.M. Rhabdomyolysis and acute kidney injury. N. Engl. J. Med. 2009, 361, 62–72. [Google Scholar] [CrossRef]
- Wiwanitkit, V. Acute renal failure in the fatal cases of dengue hemorrhagic fever, a summary in Thai death cases. Ren. Fail. 2005, 27, 647. [Google Scholar] [CrossRef]
- Davis, J.S.; Bourke, P. Rhabdomyolysis associated with dengue virus infection. Clin. Infect. Dis. 2004, 38, 109–111. [Google Scholar] [CrossRef]
- Finsterer, J.; Kongchan, K. Severe, persisting, steroid-responsive Dengue myositis. J. Clin. Virol. 2006, 35, 426–428. [Google Scholar] [CrossRef]
- Lim, M.; Goh, H.K. Rhabdomyolysis following dengue virus infection. Singap. Med. J. 2005, 46, 645–646. [Google Scholar]
- Boonpucknavig, S.; Vuttiviroj, O.; Boonpucknavig, V. Infection of young adult mice with dengue virus type 2. Trans. R. Soc. Trop. Med. Hyg. 1981, 75, 647–653. [Google Scholar] [CrossRef]
- Oliveira, J.F.P.; Burdmann, E.A. Dengue-associated acute kidney injury. Clin. Kidney J. 2015, 8, 681–685. [Google Scholar] [CrossRef]
- Garcia, S.; Morales, R.; Hunter, R.F. Dengue fever with thrombocytopenia: Studies towards defining vulnerability of bleeding. Medica de Puerto Rico 1995, 87, 2–7. [Google Scholar]
- Futrakul, P.; Poshyachinda, V.; Mitrakul, C.; Kun-Anake, C.; Boonpucknavig, V.; Boompucknavig, S.; Bhamarapravati, N. Renal involvement and reticulo-endothelial-system clearance in dengue hemorrhagic fever. J. Med. Assoc. Thai. 1973, 56, 33–39. [Google Scholar]
- Barreto, D.F.; Takiya, C.M.; Paes, M.V.; Farias-Filho, J.; Pinhão, A.T.; Alves, A.M.; Costa, S.M.; Barth, O.M. Histopathological aspects of Dengue-2 virus infected mice tissues and complementary virus isolation. J. Submicrosc. Cytol. Pathol. 2004, 36, 121–130. [Google Scholar]
- Boonpucknavig, V.; Bhamarapravati, N.; Boonpucknavig, S.; Futrakul, P.; Tanpaichitr, P. Glomerular changes in dengue hemorrhagic fever. Arch. Pathol. Lab. Med. 1976, 100, 206–212. [Google Scholar]
- Guzmán, M.G.; Alvarez, M.; Rodríguez, R.; Rosario, D.; Vázquez, S.; Valds, L.; Cabrera, M.V.; Kourí, G. Fatal dengue hemorrhagic fever in Cuba, 1997. Int. J. Infect. Dis. 1999, 3, 130–135. [Google Scholar] [CrossRef]
- Limonta, D.; Falcón, V.; Torres, G.; Capó, V.; Menéndez, I.; Rosario, D.; Castellanos, Y.; Alvarez, M.; Rodríguez-Roche, R.; De La Rosa, M.C.; et al. Dengue virus identification by transmission electron microscopy and molecular methods in fatal dengue hemorrhagic fever. Infection 2012, 40, 689–694. [Google Scholar] [CrossRef]
- Rodrigues, R.S.; Brum, A.L.G.; Paes, M.V.; Póvoa, T.F.; Basilio-de-Oliveira, C.A.; Marchiori, E.; Borghi, D.P.; Ramos, G.V.; Bozza, F.A. Lung in dengue: Computed tomography findings. PLoS ONE 2014, 9, e96313. [Google Scholar] [CrossRef]
- Shivanthan, M.C.; Navinan, M.R.; Constantine, G.R.; Rajapakse, S. Cardiac involvement in dengue infection. J. Infect. Dev. Ctries 2015, 9, 338–346. [Google Scholar] [CrossRef]
- Miranda, C.H.; Borges Mde, C.; Matsuno, A.K.; Vilar, F.C.; Gali, L.G.; Volpe, G.J.; Schmidt, A.; Pazin-Filho, A.; Silva, F.M.; Castro-Jorge, L.A.; et al. Evaluation of cardiac involvement during dengue viral infection. Clin. Infect. Dis. 2013, 57, 812–819. [Google Scholar] [CrossRef]
- Yadav, R.K.; Kumar, S. To study cardiac manifestations in patients presenting with dengue infection and to find out the correlation of cardiac manifestations to warning signs of dengue. Int. J. Adv. Med. 2017, 4, 323–328. [Google Scholar] [CrossRef]
- Virk, H.U.H.; Inayat, F.; Rahman, Z.U. Complete Heart Block in Association with Dengue Hemorrhagic Fever. Korean Circ. J. 2016, 46, 866–869. [Google Scholar] [CrossRef]
- Weerakoon, K.G.; Kularatne, S.A.; Edussuriya, D.H.; Kodikara, S.K.; Gunatilake, L.P.; Pinto, V.G.; Seneviratne, A.B.; Gunasena, S. Histopathological diagnosis of myocarditis in a dengue outbreak in Sri Lanka, 2009. BMC Res. Notes 2011, 4, 2–7. [Google Scholar] [CrossRef]
- Salgado, D.M.; Eltit, J.M.; Mansfield, K.; Panqueba, C.; Castro, D.; Vega, M.R.; Xhaja, K.; Schmidt, D.; Martin, K.J.; Allen, P.D.; et al. Heart and skeletal muscle are targets of dengue virus infection. Pediatric Infect. Dis. J. 2010, 29, 238–242. [Google Scholar] [CrossRef]
- Ashour, J.; Morrison, J.; Laurent-Rolle, M.; Belicha-Villanueva, A.; Plumlee, C.R.; Bernal-Rubio, D.; Williams, K.L.; Eva Harris, E.; Fernandez-Sesma, A.; Christian Schindler, C.; et al. Mouse STAT2 restricts early Dengue virus replication. Cell Host Microbe 2010, 8, 410–421. [Google Scholar] [CrossRef]
- Morrison, J.; Laurent-Rolle, M.; Maestre, A.M.; Rajsbaum, R.; Pisanelli, G.; Simon, V.; Mulder, L.C.F.; Fernandez-Sesma, A.; Garcı´a-Sastre, A. Dengue virus co-opts UBR4 to degrade STAT2 and antagonize type I interferon signaling. PLoS Pathog. 2013, 9, 1–13. [Google Scholar] [CrossRef]
- Stabell, A.; Meyerson, N.; Gullberg, R.C.; Gilchrist, A.R.; Webb, K.J.; Old, W.M.; Perera, R.; Sawyer, S.L. Dengue viruses cleave STING in humans but not in nonhuman primates, their presumed natural reservoir. eLife 2018, 7, e31919. [Google Scholar] [CrossRef]
- Aguirre, S.; Maestre, A.M.; Pagni, S.; Patel, J.R.; Savage, T.; Gutman, D.; Maringer, K.; Bernal-Rubio, D.; Shabman, R.S.; Simon, V.; et al. DENV inhibits type I IFN production in infected cells by cleaving human STING. PLoS Pathog. 2012, 8, 1–14. [Google Scholar] [CrossRef]
- Yu, C.; Chang, T.; Liang, J.; Chiang, R.; Lee, Y.; Liao, C.; Lin, Y. Dengue virus targets the adaptor protein MITA to subvert host innate immunity. PLoS Pathog. 2012, 8, 1–13. [Google Scholar] [CrossRef]
- Prestwood, T.R.; Morar, M.M.; Zellweger, R.M.; Miller, R.; May, M.M.; Yauch, L.E.; Lada, S.M.; Shresta, S. Gamma interferon (IFN-γ) receptor restricts systemic dengue virus replication and prevents paralysis in IFN-α/β receptor-deficient mice. J. Virol. 2012, 86, 12561–12570. [Google Scholar] [CrossRef]
- Dhole, P.; Nakayama, E.E.; Saito, A.; Limkittikul, K.; Phanthanawiboon, S.; Shioda, T.; Kurosu, T. Sequence diversity of dengue virus type 2 in brain and thymus of infected interferon receptor ko mice: Implications for dengue virulence. Virol. J. 2016, 13, 1–8. [Google Scholar] [CrossRef][Green Version]
- Fink, K.; Ng, C.; Nkenfou, C.; Vasudevan, S.G.; Rooijen, N.V.; Schul, W. Depletion of macrophages in mice results in higher dengue virus titers and highlights the role of macrophages for virus control. Eur. J. Immunol. 2009, 39, 2809–2821. [Google Scholar] [CrossRef]
- Shresta, S.; Sharar, K.L.; Prigozhin, D.M.; Beatty, P.R.; Harris, E. Murine model for dengue virus-induced lethal disease with increased vascular permeability. J. Virol. 2006, 80, 10208–10217. [Google Scholar] [CrossRef]
- Zompi, S.; Santich, B.H.; Beatty, P.R.; Harris, E. Protection from Secondary Dengue Virus Infection In A Mouse Model Reveals The Role Of Serotype Cross-Reactive B And T Cells. J. Immunol. 2012, 188, 404–416. [Google Scholar] [CrossRef]
- Tan, G.; Jowin, K.; Trasti, S.L.; Schul, W.; Yip, G.; Alonso, S. A non mouse-adapted dengue virus strain as a new model of severe dengue infection in AG129 mice. PLoS Negl. Trop Dis. 2010, 4, 1–10. [Google Scholar] [CrossRef]
- Grant, D.; Tan, G.; Qing, M.; Ng, J.K.W.; Yip, A.; Zou, G.; Xie, X.; Yuan, Z.; Schreiber, M.J.; Schul, W.; et al. A single amino acid in non structural protein NS4B confers virulence to dengue virus in AG129 mice through enhancement of viral RNA synthesis. J. Virol. 2011, 85, 7775–7787. [Google Scholar] [CrossRef]
- Calvert, A.; Huang, C.; Kinney, R.; Roehrig, J.T. Non-structural proteins of dengue 2 virus offer limited protection to interferon-deficient mice after dengue 2 virus challenge. J. Gen. Virol. 2006, 87, 339–346. [Google Scholar] [CrossRef]
- Mota, J.; Rico-Hesse, R. Humanized mice show clinical signs of dengue fever according to infecting virus genotype. J. Virol. 2009, 83, 8638–8645. [Google Scholar] [CrossRef]
- Yam-Puc, J.C.; García-Cordero, J.; Calderón-Amador, J.; Donis-Maturano, L.; Cedillo-Barrón, L.; Flores-Romo, L. Germinal center reaction following cutaneous dengue virus infection in immune-competent mice. Front. Immunol. 2015, 6, 1–9. [Google Scholar] [CrossRef]
- Pham, A.M.; Langlois, R.A.; Tenoever, B.R. Replication in Cells of Hematopoietic Origin Is Necessary for Dengue Virus Dissemination. PLoS Pathog. 2012, 8, 1–10. [Google Scholar] [CrossRef]
- Surasombatpattana, P.; Hamel, R.; Patramool, S.; Luplertlop, N.; Thomas, F.; Desprès, P.; Briant, L.; Yssel, H.; Missé, D. Dengue virus replication in infected human keratinocytes leads to activation of antiviral innate immune responses. Infect. Genet. Evol. 2011, 11, 1664–1673. [Google Scholar] [CrossRef]
- Rathore, A.P.S.; St John, A.L. Immune responses to dengue virus in the skin. Open Biol. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- King, C.A.; Marshall, J.S.; Alshurafa, H.; Anderson, R. Release of Vasoactive Cytokines by Antibody-Enhanced Dengue Virus Infection of a Human Mast Cell/Basophil Line. J. Virol. 2000, 74, 7146–7150. [Google Scholar] [CrossRef]
- Sun, P.; Fernandez, S.; Marovich, M.A.; Palmer, D.R.; Celluzzi, C.M.; Boonnak, K.; Liang, Z.; Subramanian, H.; Porter, K.R.; Sun, W.; et al. Functional characterization of ex vivo blood myeloid and plasmacytoid dendritic cells after infection with dengue virus. Virology 2009, 383, 207–215. [Google Scholar] [CrossRef]
- Chen, Y.C.; Wang, S.Y.; King, C.C. Bacterial lipopolysaccharide inhibits dengue virus infection of primary human monocytes/macrophages by blockade of virus entry via a CD14-dependent mechanism. J. Virol. 1999, 73, 2650–2657. [Google Scholar]
- Carnec, X.; Meertens, L.; Dejarnac, O.; Perera-Lecoin, M.; Hafirassou, M.L.; Kitaura, J.; Ramdasi, R.; Schwartz, O.; Amara, A. The phosphatidylserine and phosphatidylethanolamine receptor CD300a binds dengue virus and enhances infection. J. Virol. 2015, 90, 92–102. [Google Scholar] [CrossRef]
- Kou, Z.; Quinn, M.; Chen, H.; Rodrigo, W.W.S.I.; Rose, R.C.; Schlesinger, J.J.; Jin, X. Monocytes, But Not T or B Cells, Are the Principal Target Cells for Dengue Virus (DV) Infection Among Human Peripheral Blood Mononuclear Cells. J. Med. Virol. 2008, 80, 134–146. [Google Scholar] [CrossRef]
- Zhang, J.L.; Wang, J.L.; Gao, N.; Chen, Z.T.; Tian, Y.P.; An, J. Up-regulated expression of β3 integrin induced by dengue virus serotype 2 infection associated with virus entry into human dermal microvascular endothelial cells. Biochem. Biophys. Res. Commun. 2007, 356, 763–768. [Google Scholar] [CrossRef]
- Talavera, D.; Castillo, A.M.; Dominguez, M.C.; Gutierrez, A.E.; Meza, I. IL8 release, tight junction and cytoskeleton dynamic reorganization conducive to permeability increase are induced by dengue virus infection of microvascular endothelial monolayers. J. Gen. Virol. 2004, 85, 1801–1813. [Google Scholar] [CrossRef]
- Dalrymple, N.; Mackow, E.R. Productive dengue virus infection of human endothelial cells is directed by heparansulfate-containing proteoglycan receptors. J. Virol. 2011, 85, 9478–9485. [Google Scholar] [CrossRef]
- Yang, J.; Zou, L.; Yang, Y.; Yuan, J.; Hu, Z.; Liu, H.; Peng, H.; Shang, W.; Zhang, X.; Zhu, J.; et al. Superficial vimentin mediates DENV-2 infection of vascular endothelial cells. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Wan, S.W.; Lin, C.F.; Lu, Y.T.; Lei, H.Y.; Anderson, R.; Lin, Y.S. Endothelial cell surface expression of protein disulfideisomerase activates β1 and β3 integrins and facilitates dengue virus infection. J. Cell. Biochem. 2012, 113, 1681–1691. [Google Scholar]
- Salazar, M.I.; del Angel, R.M.; Lanz-Mendoza, H.; Ludert, J.E.; Pando-Robles, V. The role of cell proteins in dengue virus infection. J. Proteom. 2014, 111, 6–15. [Google Scholar] [CrossRef]
- Da Conceição, T.M.; Rust, N.M.; Berbel, A.C.; Martins, N.B.; Santos, C.A.N.; Da Poian, A.T.; De Arruda, L.B. Essential role of RIG-I in the activation of endothelial cells by dengue virus. Virology 2013, 435, 281–292. [Google Scholar] [CrossRef]
- Meertens, L.; Carnec, X.; Lecoin, M.P.; Ramdasi, R.; Guivel-Benhassine, F.; Lew, E.; Lemke, G.; Schwartz, O.; Amara, A. The TIM and TAM families of phosphatidylserine receptors mediate dengue virus entry. Cell Host Microbe 2012, 12, 544–557. [Google Scholar] [CrossRef]
- Gao, F.; Duan, X.; Lu, X.; Liu, Y.; Zheng, L.; Ding, Z.; Li, J. Novel binding between pre-membrane protein and claudin-1 is required for efficient dengue virus entry. Biochem. Biophys. Res. Commun. 2010, 391, 952–957. [Google Scholar] [CrossRef]
- Jindadamrongwech, S.; Thepparit, C.; Smith, D.R. Identification of GRP 78 (BiP) as a liver cell expressed receptor element for dengue virus serotype 2. Arch. Virol. 2004, 149, 915–927. [Google Scholar] [CrossRef]
- Upanan, S.; Kuadkitkan, A.; Smith, D.R. Identification of dengue virus binding proteins using affinity chromatography. J. Virol. Methods 2008, 151, 325–328. [Google Scholar] [CrossRef]
- Che, P.; Tang, H.; Li, Q. The interaction between claudin-1 and dengue viral prM/M protein for its entry. Virol 2013, 446, 303–313. [Google Scholar] [CrossRef]
- Azizan, A.; Sweat, J.; Espino, C.; Gemmer, J.; Stark, L.; Kazanis, D. Differential proinflammatory and angiogenesis-specific cytokine production in human pulmonary endothelial cells, HPMEC-ST1.6R infected with dengue-2 and dengue-3 virus. J. Virol. Methods 2006, 138, 211–217. [Google Scholar] [CrossRef]
- Lin, Y.W.; Wang, K.J.; Lei, H.Y.; Lin, Y.S.; Yeh, T.M.; Liu, H.S.; Liu, C.C.; Chen, S.H. Virus replication and cytokine production in dengue virus-infected human B lymphocytes. J. Virol. 2002, 76, 12242–12249. [Google Scholar] [CrossRef]
- Boonpucknavig, S.; Bhamarapravati, N.; Nimmannitya, S.; Phalavadhtana, A.; Siripont, J. Immunofluorescent staining of the surfaces of lymphocytes in suspension from patients with dengue hemorrhagic fever. Am. J. Pathol. 1976, 85, 37–48. [Google Scholar]
- Bielefeldt-Ohmann, H.; Meyer, M.; Fitzpatrick, D.R.; Mackenzie, J.S. Dengue virus binding to human leukocyte cell lines, receptor usage differs between cell types and virus strains. Virus Res. 2001, 73, 81–89. [Google Scholar] [CrossRef]
- Theofilopoulos, A.N.; Brandt, W.E.; Russell, P.K.; Dixon, F.T. Replication of Dengue-2 Virus in Cultured Human Lymphoblastoid Cells and Subpopulations of Human Peripheral Leukocytes. J. Immunol. 1976, 117, 953–961. [Google Scholar]
- Kurane, I.; Kontny, U.; Janus, J.; Ennis, F.A. Dengue-2 virus infection of human mononuclear cell lines and establishment of persistent infections. Arch. Virol. 1990, 110, 91–101. [Google Scholar] [CrossRef]
- Silveira, G.F. Human T lymphocytes are permissive for dengue virus replication. J. Virol. 2018, 92, 1–19. [Google Scholar] [CrossRef]
- Mentor, N.A.; Kurane, I. Dengue virus infection of human T lymphocytes. Acta Virol. 1997, 41, 175–176. [Google Scholar]
- Reyes-del Valle, J.; Chavez-Salinas, S.; Medina, F.; del Angel, R.M. Heat Shock Protein 90 and Heat Shock Protein 70 Are Components of Dengue Virus Receptor Complex in Human Cells. J. Virol. 2005, 79, 4557–4567. [Google Scholar] [CrossRef]
- Puerta-Guardo, H.; Mosso, C.; Medina, F.; Liprandi, F.; Ludert, J.E.; del Angel, R.M. Antibody-dependent enhancement of dengue virus infection in U937 cells requires cholesterol-rich membrane microdomains. J. Gen. Virol. 2010, 91, 394–403. [Google Scholar] [CrossRef]
- Reyes-del Valle, J.; Salas-Benito, J.; Soto-Acosta, R.; del Angel, R.M. Dengue Virus Cellular Receptors and Tropism. Curr. Trop. Med. Rep. 2014, 1, 36–43. [Google Scholar] [CrossRef]
- Tassaneetrithep, B.; Burgess, T.H.; Granelli-Piperno, A.; Trumpfheller, C.; Finke, J.; Sun, W.; Eller, M.A.; Pattanapanyasat, K.; Sarasombath, S.; Birx, D.L.; et al. DC-SIGN (CD209) Mediates Dengue Virus Infection of Human Dendritic Cells. J. Exp. Med. 2003, 197, 823–829. [Google Scholar] [CrossRef]
- Marovich, M.; Grouard-Vogel, G.; Louder, M.; Eller, M.; Sun, W.; Wu, S.J.; Putvatana, R.; Murphy, G.; Tassaneetrithep, B.; Burgess, T.; et al. Human dendritic cells as targets of dengue virus infection. J. Investig. Dermatol. Symp. Proc. 2001, 6, 219–224. [Google Scholar] [CrossRef]
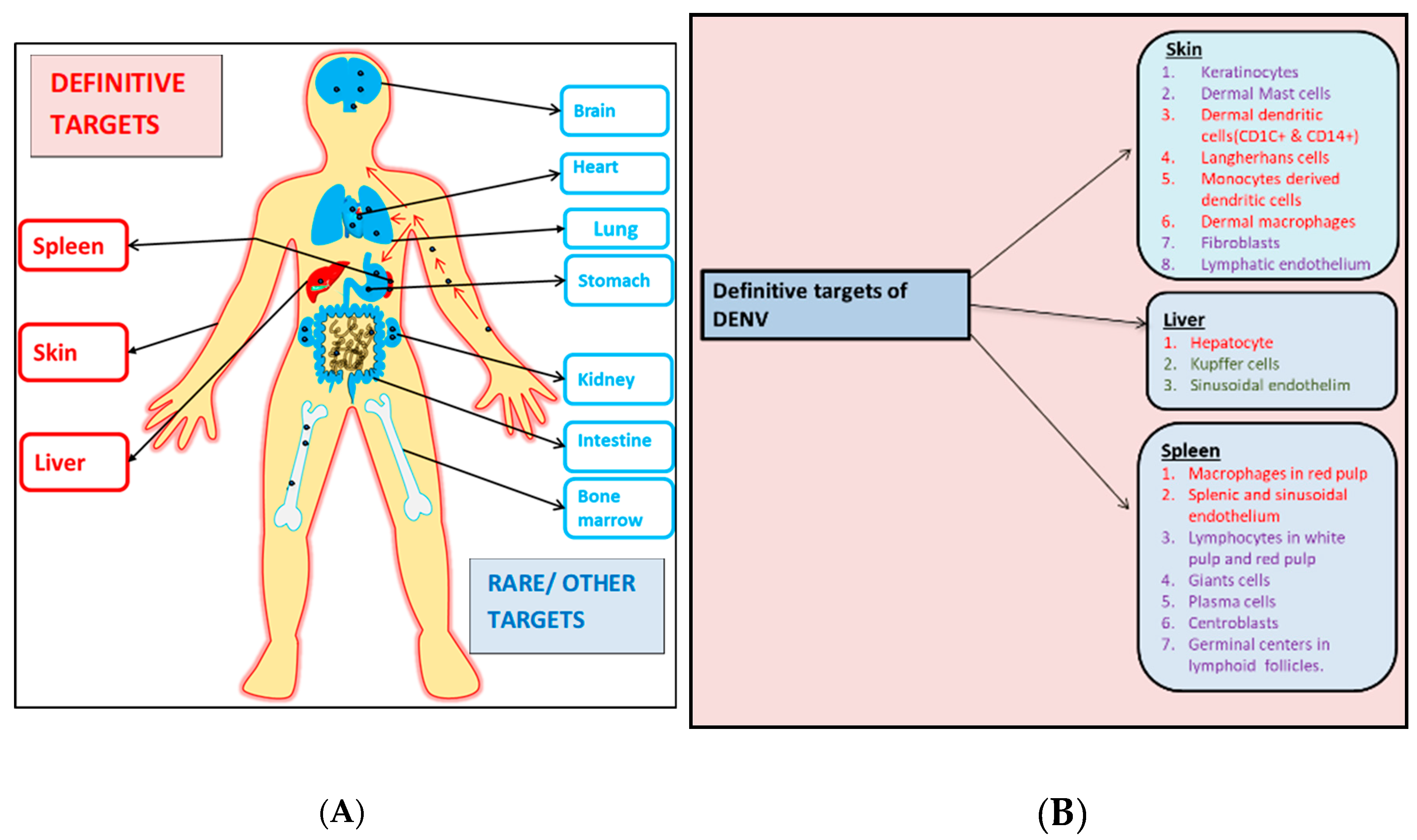
| Human Organs/Tissues | Primary Cells/Cell Lines | Presence of DENV Antigens/RNA | References |
|---|---|---|---|
| Skin | a. Primary epidermal keratinocytes and HaCaT cells | Negative sense (−) RNA, envelope (E) and non-structural (NS) proteins NS5 and NS3 | [28,121,122] |
| b. Primary dermal mast cells, HMC1 and KU812 cell lines | Positive-sense (+) RNA and viral capsid (C) | [29,30,31,123] | |
| c. Dermal DCs (CD1c+ and CD14+) and Langerhan cells | NS1, E and positive-sense (+) RNA | [21,25,27] | |
| d. Monocyte-derived DCs in dermis | NS1 and E | [25,26,124] | |
| e. Dermal macrophages | Positive-sense (+) RNA and E | [21,27,125,126,127] | |
| f. Fibroblasts | NS3 | [28] | |
| g. Lymphatic endothelium | NS3 | [28] | |
| h. Human microvascular endothelium line-1 (HMEC-1) | E and NS1 | [128,129,130,131,132,133] | |
| Draining Lymph nodes | a. Macrophages | DENV RNA and NS3 | [13,18,34] |
| b. Immunoblasts | Unspecified | [34] | |
| c. Lymphocytes | Unspecified | [34] | |
| d. Plasma cells | Unspecified | [34] | |
| Spleen | a. Macrophages in red pulp | Negative-sense (−) and positive-sense (+) RNA, NS3, E and NS1 | [13,14,15,18,20,34,35,37] |
| b. Splenic endothelium and sinusoidal endothelium | NS3 | [18,130,131] | |
| c. Immunoblasts in white pulp | Positive-sense (+) RNA and an unspecified DENV antigen | [15,34] | |
| d. Lymphocytes in white pulp | Positive-sense (+) RNA and an unspecified DENV antigen | [15,34] | |
| e. Lymphocytes in red pulp | Positive-sense (+) RNA and an unspecified DENV antigen | [15] | |
| f. Giant cells (binucleated or multinucleated) | Positive-sense (+) RNA and an unspecified DENV antigen | [15] | |
| g. Plasma cells | Unspecified | [34] | |
| h. Mononuclear cells (macrophages and DCs) in white pulp | NS3, NS1 and E | [18,35] | |
| i. Germinal centers in lymphoid follicle | Positive-sense (+) RNA and an unspecified DENV antigen | [15] | |
| j. Centroblasts in white pulp | Positive-sense (+) RNA and an unspecified DENV antigen | [15] | |
| Central Nervous System | a. Vascular endothelium and human brain microvascular endothelial cells (HBMEC) | Positive-sense (+) RNA | [74,130,131,133,134] |
| b. Neurons | Positive-sense (+) RNA and NS3 | [34,74,133] | |
| c. Astrocytes | Positive-sense (+) RNA and NS3 | [18,34,74,135] | |
| d. Microglia | Positive-sense (+) RNA | [34,74] | |
| e. Lymphocytes, Purkinje cells and granular cells | Unspecified | [34] | |
| Kidney | a. Immunoblasts | Unspecified | [34] |
| b. Histiocytes | Unspecified | [34] | |
| c. Plasma cells | Unspecified | [34] | |
| d. Lymphocytes | Unspecified | [34] | |
| Liver | a. Hepatocytes, HepG2, Huh7, Huh75.1 and Huh7.5 | Negative-sense (−) and positive-sense (+) RNA and NS3 | [13,14,20,34,135,136,137,138,139] |
| b. Kuppfer cells | Negative-sense (−) and positive-sense (+) RNA and NS3 | [13,14,15,20,34] | |
| c. Vascular/sinusoidal endothelium | Negative-sense (−) and positive-sense (+) RNA and NS3 | [13,14,15,20,34,130] | |
| Lung | a. Alveolar macrophages | Negative-sense (−) RNA and NS3 | [14,15,20,34] |
| b. Type II pneumocytes | Negative-sense (−) RNA and NS3 | [14,20,34] | |
| c. Vascular endothelium and human pulmonary endothelial cell (EC) line (HPMEC-ST1.6R) | Negative-sense (−) and positive-sense (+) RNA and NS3 | [14,15,20,34,130,140] | |
| Intestine | a. Plasma cells in lamina propria | Unspecified | [34] |
| b. Mononuclear cells in lamina propria | Unspecified | [34] | |
| Heart | a. Myocardial endothelium | Negative-sense (−) RNA and NS3 | [20,104] |
| b. Myocardial interstitial cells | Unspecified | [104] | |
| c. Cardiomyocytes | Negative-sense (−) RNA and NS3 | [20,104] | |
| d. Monocytes/macrophages | Negative-sense (−) RNA and NS3 | [20] | |
| Stomach | a. Lymphocytes in mucosa | Unspecified | [34] |
| b. Plasma cells in lamina propria | Unspecified | [34] | |
| Blood | a. Peripheral blood monocyte/macrophage (PBMC)-derived primary B cells and B cell lines (Raji cells, Wil 2WT, BM, LK63, Daudi and 8866) | Negative-sense (−) RNA, an unspecified DENV antigen (primary B cells) and DENV RNA (BM and LK63, Daudi, Raji) | [141,142,143,144,145] |
| b. PBMC-derived T cells and T cell lines (JK44, JK49, CB2.8, CB6.17, HSB-2, Molt-4 and Jurkat) | (Wil 2WT and 8866) DENV NS3, C, NS1 (Primary T cells), DENV RNA (Molt-4 and Jurkat) and an unspecified antigen (JK44, JK49, CB2.8, CB6.17 and HSB-2) | [143,145,146,147] | |
| c. Activated monocytes and U937 cells | prM and NS3 | [17,125,127,128,148,149,150] | |
| d. Blood-derived DCs | Negative-sense (−) and positive-sense (+) RNA, DENV E and NS1 | [25,124,126,151,152] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Begum, F.; Das, S.; Mukherjee, D.; Mal, S.; Ray, U. Insight into the Tropism of Dengue Virus in Humans. Viruses 2019, 11, 1136. https://doi.org/10.3390/v11121136
Begum F, Das S, Mukherjee D, Mal S, Ray U. Insight into the Tropism of Dengue Virus in Humans. Viruses. 2019; 11(12):1136. https://doi.org/10.3390/v11121136
Chicago/Turabian StyleBegum, Feroza, Sandeepan Das, Debica Mukherjee, Sweety Mal, and Upasana Ray. 2019. "Insight into the Tropism of Dengue Virus in Humans" Viruses 11, no. 12: 1136. https://doi.org/10.3390/v11121136
APA StyleBegum, F., Das, S., Mukherjee, D., Mal, S., & Ray, U. (2019). Insight into the Tropism of Dengue Virus in Humans. Viruses, 11(12), 1136. https://doi.org/10.3390/v11121136




