Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Garlic (Allium sp.) Extract
2.2. Synthesis of Gold Nanoparticles
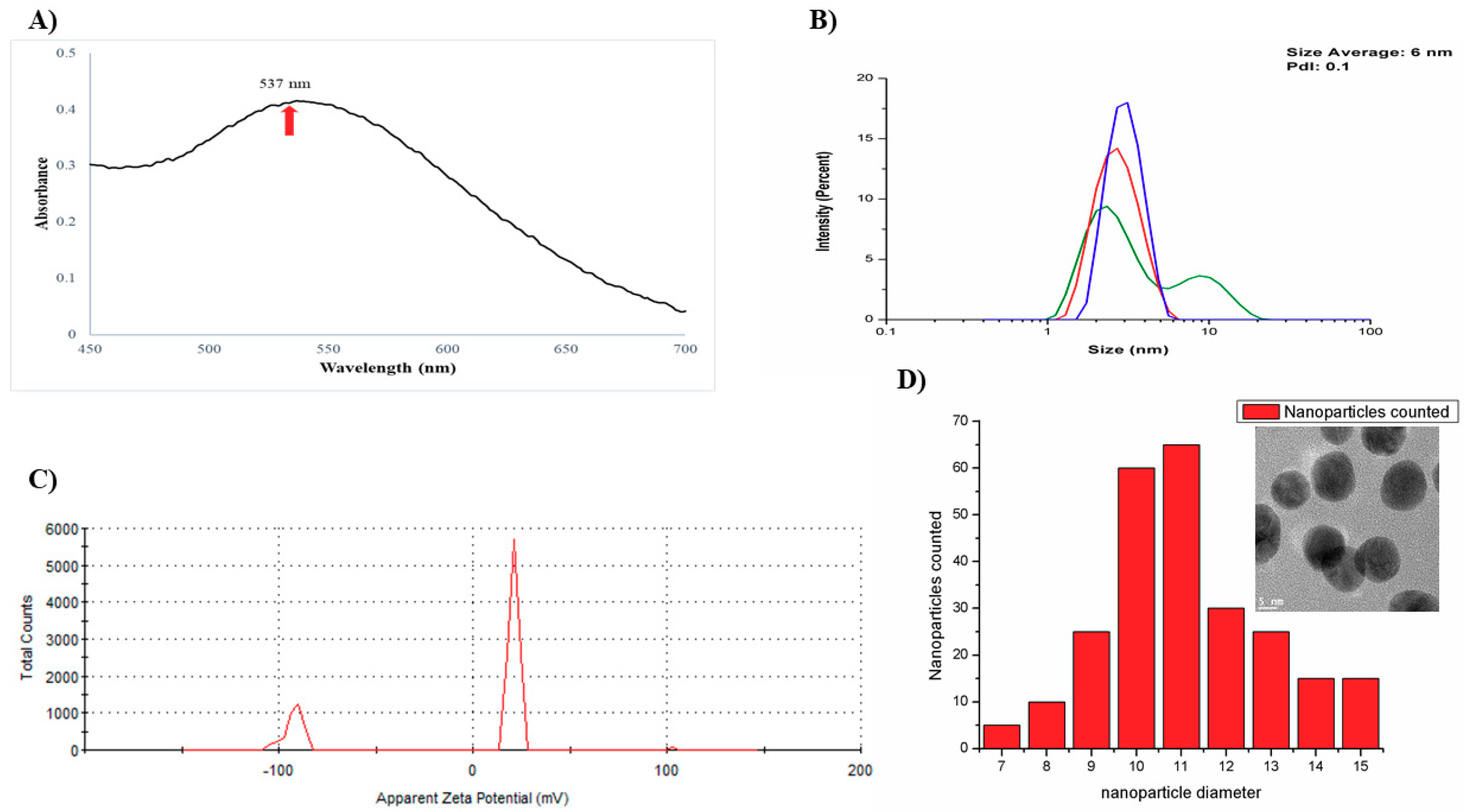
2.3. Characterization of AuNPs-As
2.4. Cell Culture and Virus Propagation
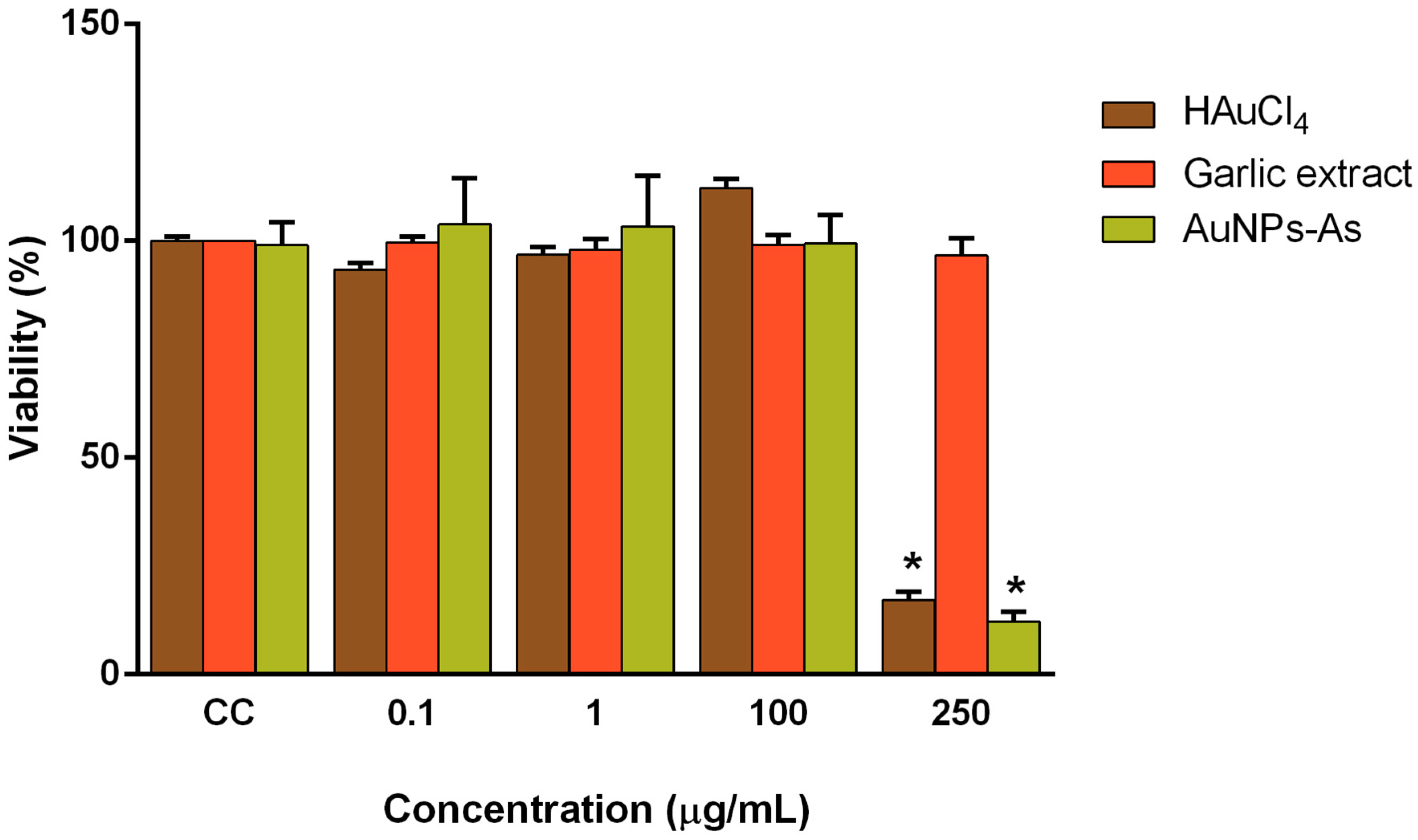
2.5. Viability Assays
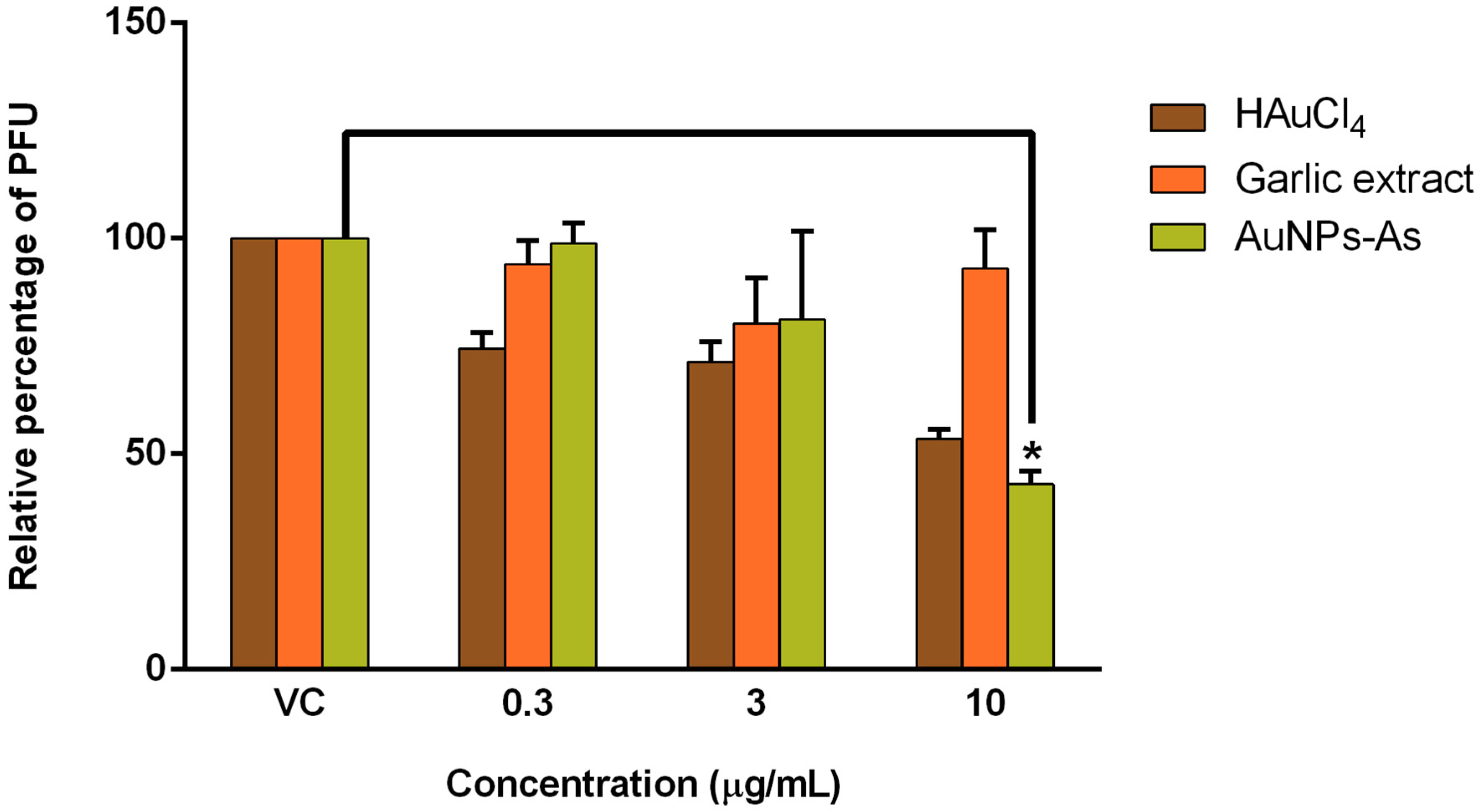
2.6. Antiviral Activity Assay
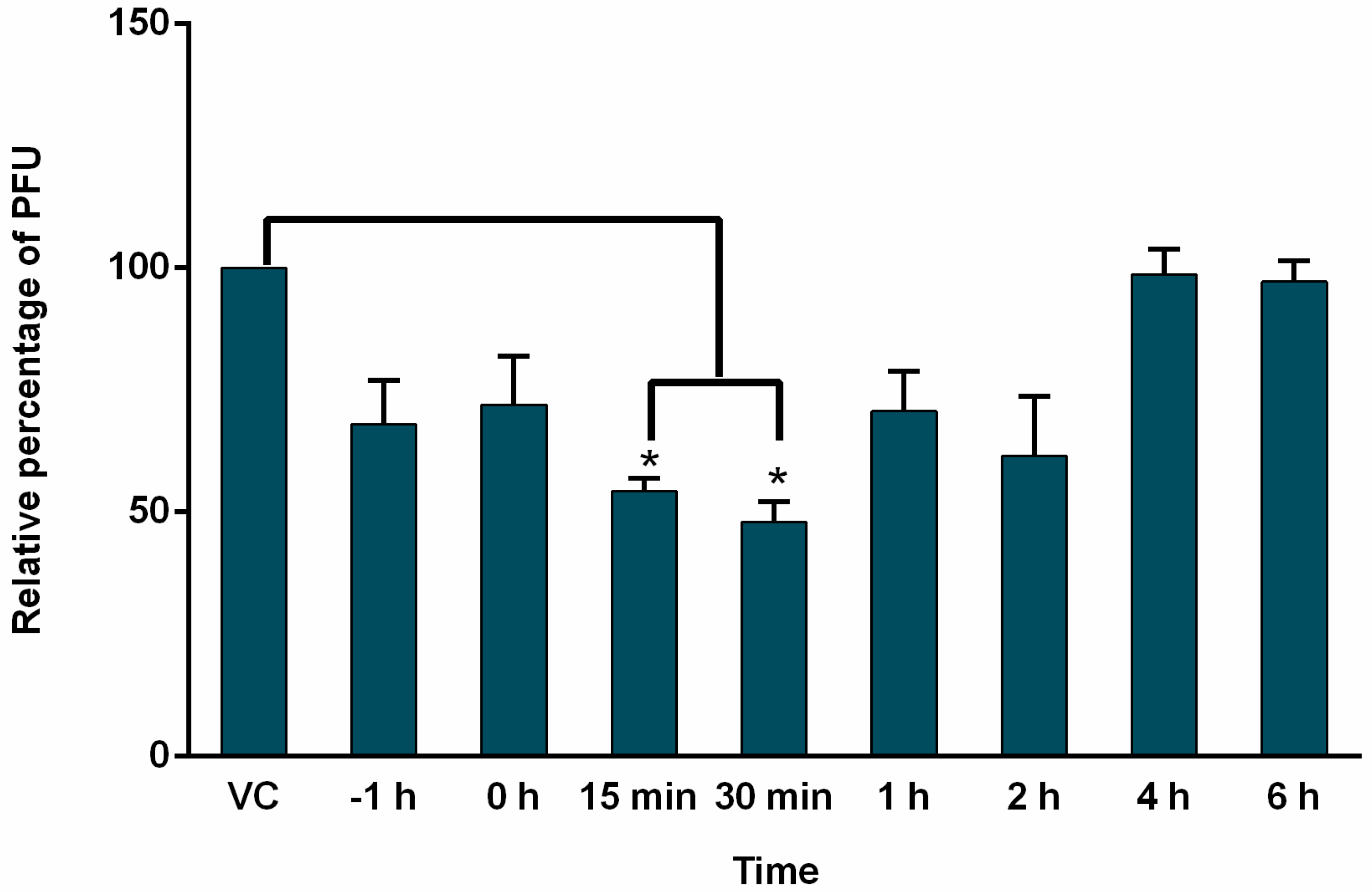
2.7. Time-of-Addition Assay
2.8. Virucidal Assay
2.9. RT-qPCR
2.10. Statistical Analysis
3. Results
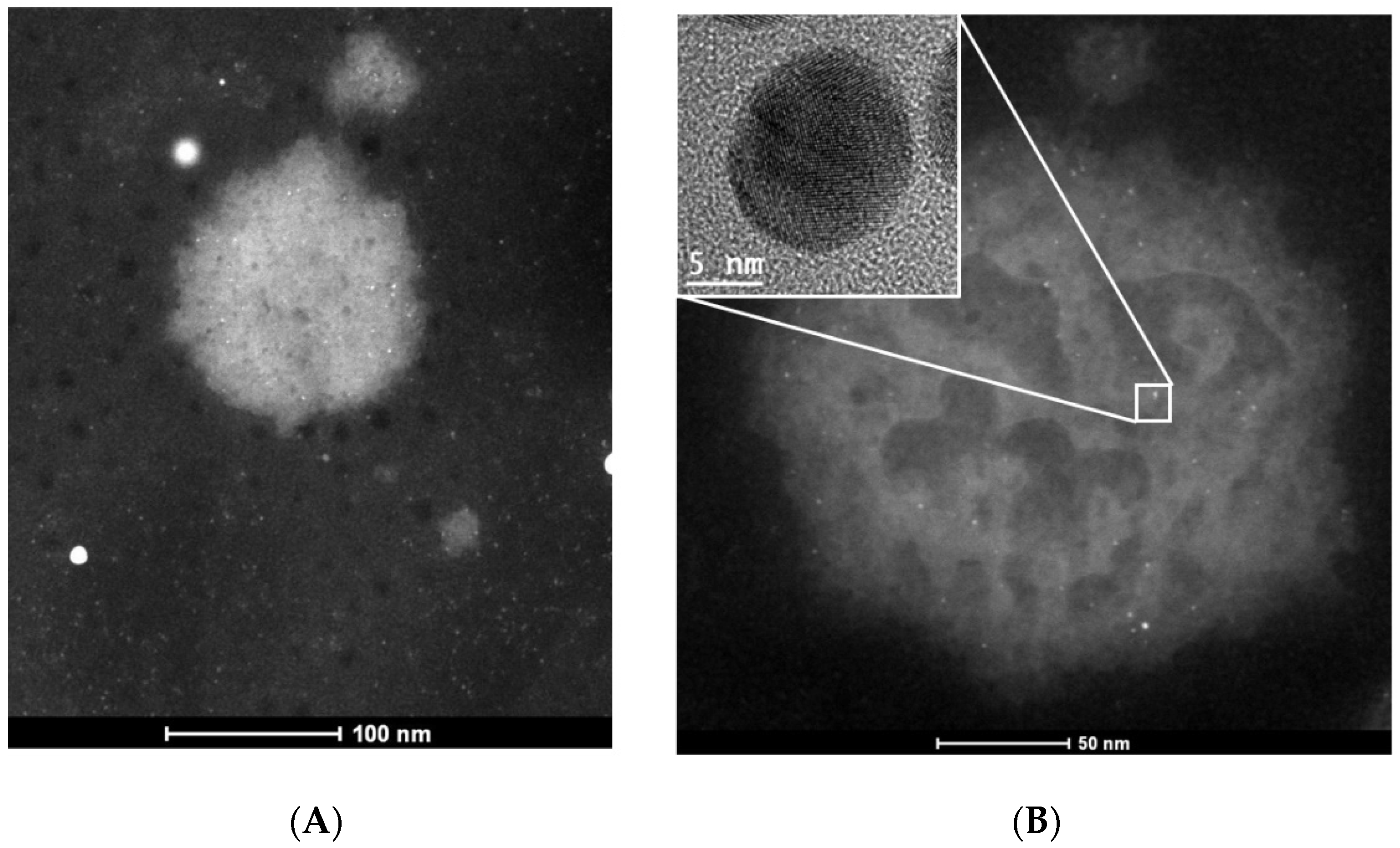
3.1. Characterization of AuNPs-As
3.2. Cytotoxicity of AuNPs
3.3. Antiviral Activity
3.4. Effect of AuNPs-As on Viral Infection Was Determined by Time of Addition Assays
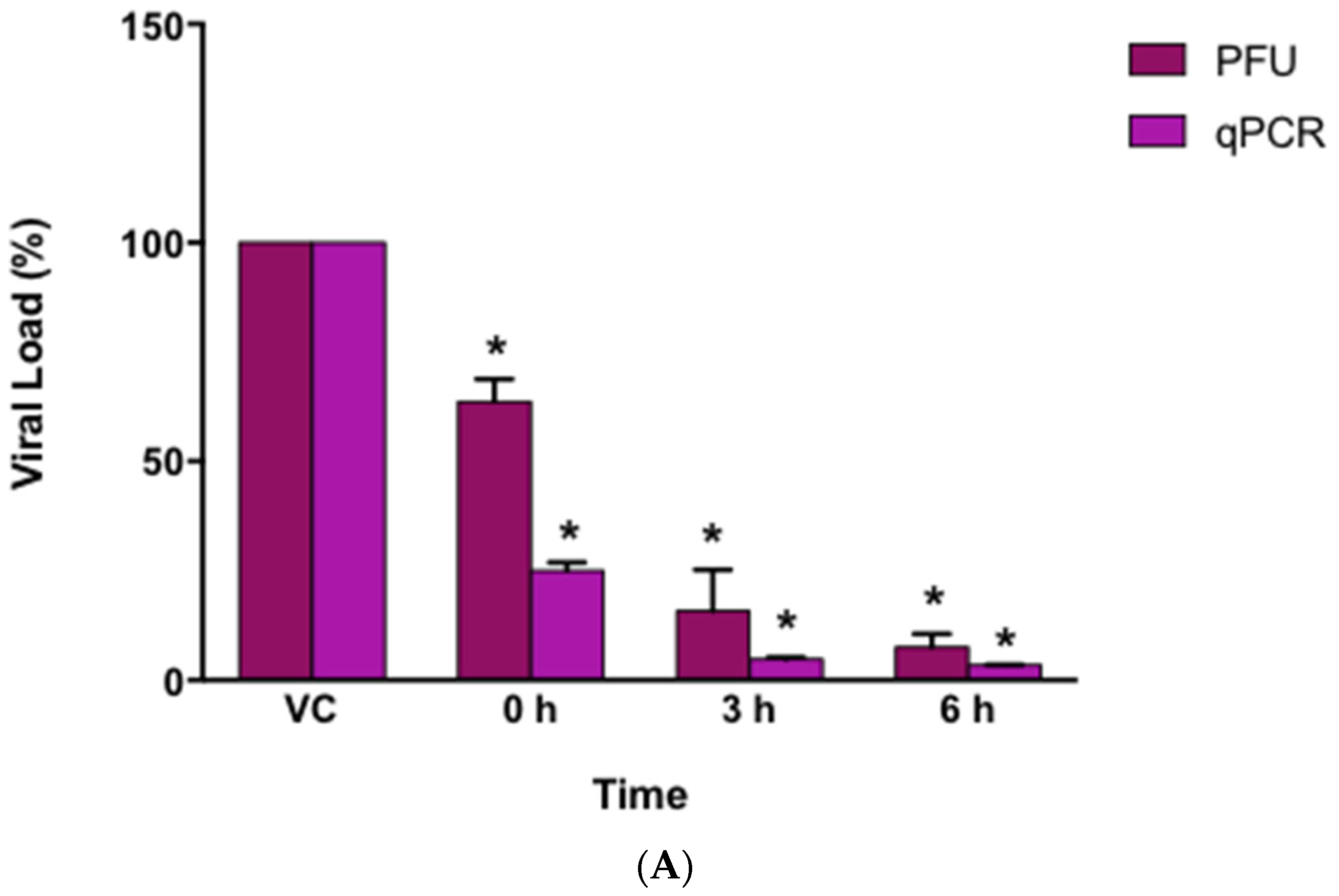
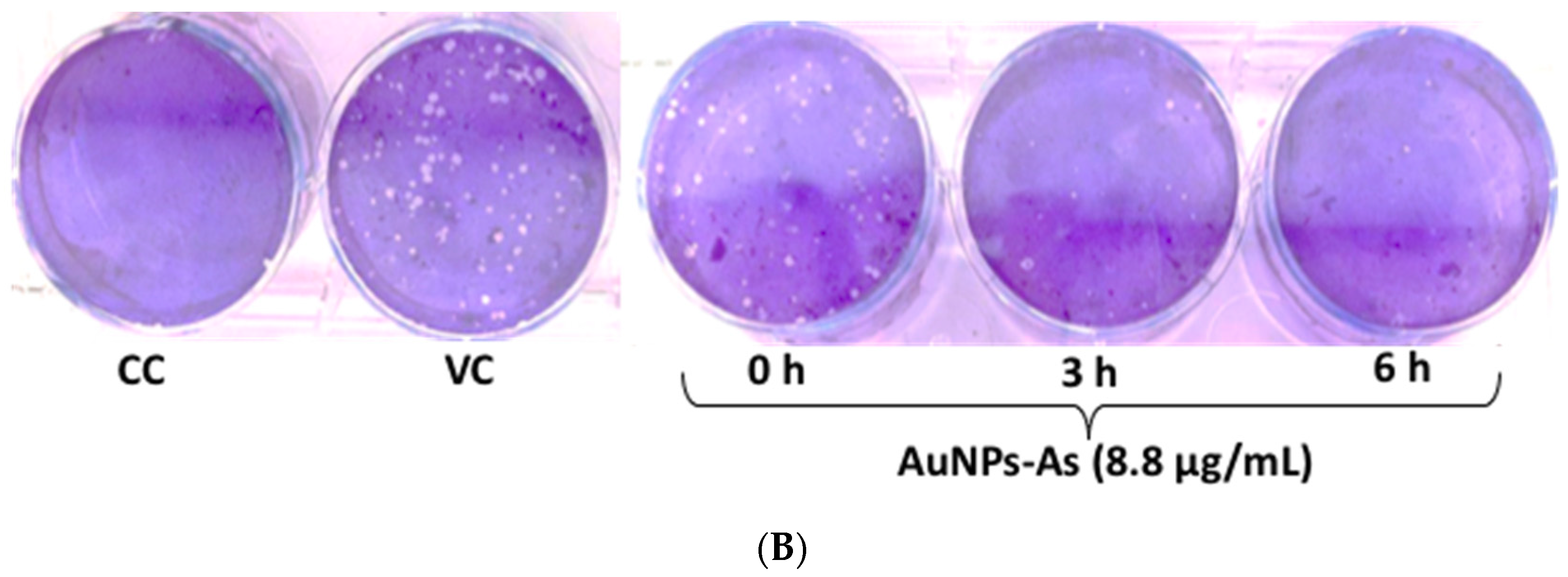
3.5. Virucidal Effect of AuNPs-As
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Griffin, D. Measles virus. Fields Virol. 2013, 1, 1042–1069. [Google Scholar]
- Durrheim, D.N.; Crowcroft, N.S.; Strebel, P.M. Measles—The epidemiology of elimination. Vaccine 2014, 32, 6880–6883. [Google Scholar] [CrossRef] [PubMed]
- Strebel, P.M.; Cochi, S.L.; Hoekstra, E.; Rota, P.A.; Featherstone, D.; Bellini, W.J.; Katz, S.L. A world without measles. J. Infect. Dis. 2011, 204, S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.T.; Murray, J.S.; Gacic-Dobo, M.; Dabbagh, A.; Mulders, M.N.; Strebel, P.M.; Okwo-Bele, J.-M.; Rota, P.A.; Goodson, J.L. Progress toward regional measles elimination—Worldwide, 2000–2014. Morb. Mortal. Wkly. Rep. 2015, 64, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E.; Li, G. Approved antiviral drugs over the past 50 years. Clin. Microbiol. Rev. 2016, 29, 695–747. [Google Scholar] [CrossRef]
- Kannan, R.; Stirk, W.; Van Staden, J. Synthesis of silver nanoparticles using the seaweed Codium capitatum PC Silva (Chlorophyceae). S. Afr. J. Bot. 2013, 86, 1–4. [Google Scholar] [CrossRef]
- Sangeetha, N.; Saravanan, K. Biogenic silver nanoparticles using marine seaweed (Ulva lactuca) and evaluation of its antibacterial activity. J. Nanosci. Nanotechnol. 2014, 2, 99–102. [Google Scholar]
- Rai, M.; Deshmukh, S.D.; Ingle, A.P.; Gupta, I.R.; Galdiero, M.; Galdiero, S. Metal nanoparticles: The protective nanoshield against virus infection. Crit. Rev. Microbiol. 2016, 42, 46–56. [Google Scholar] [CrossRef]
- Kimling, J.; Maier, M.; Okenve, B.; Kotaidis, V.; Ballot, H.; Plech, A. Turkevich method for gold nanoparticle synthesis revisited. J. Phys. Chem. B 2006, 110, 15700–15707. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Morán-Santibañez, K.; Cruz-Suárez, L.E.; Ricque-Marie, D.; Robledo, D.; Freile-Pelegrín, Y.; Peña-Hernández, M.A.; Rodríguez-Padilla, C.; Trejo-Avila, L.M. Synergistic effects of sulfated polysaccharides from Mexican seaweeds against measles virus. BioMed Res. Int. 2016, 2016, 8502123. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Torres, A.C.; Zarate-Triviño, D.G.; Lorenzo-Anota, H.Y.; Ávila-Ávila, A.; Rodríguez-Abrego, C.; Rodríguez-Padilla, C. Chitosan gold nanoparticles induce cell death in HeLa and MCF-7 cells through reactive oxygen species production. Int. J. Nanomed. 2018, 13, 3235. [Google Scholar] [CrossRef] [PubMed]
- Meléndez-Villanueva, M.A. Grado y nivel de acción de nanopartículas de metal y compuestos bioactivos naturales contra el virus de sarampión. Master’s Thesis, Universidad Autónoma de Nuevo León, San Nicolás de los Garza, Mexico, 2017. [Google Scholar]
- Geraldes, A.N.; da Silva, A.A.; Leal, J.; Estrada-Villegas, G.M.; Lincopan, N.; Katti, K.V.; Lugão, A.B. Green nanotechnology from plant extracts: Synthesis and characterization of gold nanoparticles. Adv. Nanopart. 2016, 5, 176. [Google Scholar] [CrossRef]
- Rastogi, L.; Arunachalam, J. Green synthesis route for the size controlled synthesis of biocompatible gold nanoparticles using aqueous extract of garlic (Allium sativum). Adv. Mater. Lett. 2013, 4, 548–555. [Google Scholar] [CrossRef]
- Farooq, U.; Tweheyo, M.T.; Sjøblom, J.; Øye, G. Surface characterization of model, outcrop, and reservoir samples in low salinity aqueous solutions. J. Dispers. Sci. Technol. 2011, 32, 519–531. [Google Scholar] [CrossRef]
- Puttipipatkhachorn, S.; Nunthanid, J.; Yamamoto, K.; Peck, G. Drug physical state and drug–polymer interaction on drug release from chitosan matrix films. J. Control. Release 2001, 75, 143–153. [Google Scholar] [CrossRef]
- Kim, J.O.; Kabanov, A.V.; Bronich, T.K. Polymer micelles with cross-linked polyanion core for delivery of a cationic drug doxorubicin. J. Control. Release 2009, 138, 197–204. [Google Scholar] [CrossRef]
- Honary, S.; Zahir, F. Effect of zeta potential on the properties of nano-drug delivery systems-a review (Part 2). Trop. J. Pharm. Res. 2013, 12, 265–273. [Google Scholar]
- Fatima, M.; Zaidi, N.U.S.S.; Amraiz, D.; Afzal, F. In vitro antiviral activity of Cinnamomum cassia and its nanoparticles against H7N3 influenza a virus. J. Microbiol. Biotechnol. 2016, 26, 151–159. [Google Scholar] [CrossRef]
- Cagno, V.; Andreozzi, P.; D’Alicarnasso, M.; Silva, P.J.; Mueller, M.; Galloux, M.; Le Goffic, R.; Jones, S.T.; Vallino, M.; Hodek, J. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat. Mater. 2018, 17, 195. [Google Scholar] [CrossRef]
- Pritchett, J.C.; Naesens, L.; Montoya, J. Treating HHV-6 infections: The laboratory efficacy and clinical use of ati-HHV-6 agents. In Human Herpesviruses HHV-6A, HHV-6B & HHV-7 Diagnosis and Clinical Management; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Morán-Santibañez, K.; Peña-Hernández, M.; Cruz-Suárez, L.; Ricque-Marie, D.; Skouta, R.; Vasquez, A.; Rodríguez-Padilla, C.; Trejo-Avila, L. Virucidal and synergistic activity of polyphenol-rich extracts of seaweeds against measles virus. Viruses 2018, 10, 465. [Google Scholar] [CrossRef] [PubMed]
- Crance, J.M.; Scaramozzino, N.; Jouan, A.; Garin, D. Interferon, ribavirin, 6-azauridine and glycyrrhizin: Antiviral compounds active against pathogenic flaviviruses. Antivir. Res. 2003, 58, 73–79. [Google Scholar] [CrossRef]
- Ahmed, E.M.; Solyman, S.M.; Mohamed, N.; Boseila, A.A.; Hanora, A. Antiviral activity of Ribavirin nano-particles against measles virus. Cell. Mol. Biol. (Noisy-le-Gd. Fr.) 2018, 64, 24–32. [Google Scholar] [CrossRef]
- Harris, J.; Cottrell, S.; Plummer, S.; Lloyd, D. Antimicrobial properties of Allium sativum (garlic). Appl. Microbiol. Biotechnol. 2001, 57, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Kaushik, S.; Pandit, P.; Dhull, D.; Yadav, J.P.; Kaushik, S. Green synthesis of silver nanoparticles from medicinal plants and evaluation of their antiviral potential against chikungunya virus. Appl. Microbiol. Biotechnol. 2019, 103, 881–891. [Google Scholar] [CrossRef] [PubMed]
- Avilala, J.; Golla, N. Antibacterial and antiviral properties of silver nanoparticles synthesized by marine actinomycetes. Int. J. Pharm. Sci. Res. 2019, 10, 1223–1228. [Google Scholar]
- Haggag, E.G.; Elshamy, A.M.; Rabeh, M.A.; Gabr, N.M.; Salem, M.; Youssif, K.A.; Samir, A.; Muhsinah, A.B.; Alsayari, A.; Abdelmohsen, U.R. Antiviral potential of green synthesized silver nanoparticles of Lampranthus coccineus and Malephora lutea. Int. J. Nanomed. 2019, 14, 6217. [Google Scholar] [CrossRef]
- Di Gianvincenzo, P.; Marradi, M.; Martínez-Ávila, O.M.; Bedoya, L.M.; Alcamí, J.; Penadés, S. Gold nanoparticles capped with sulfate-ended ligands as anti-HIV agents. Bioorg. Med. Chem. Lett. 2010, 20, 2718–2721. [Google Scholar] [CrossRef]
- Mehrbod, P.; Motamed, N.; Tabatabaian, M.; Estyar, R.S.; Amini, E.; Shahidi, M.; Kheiri, M. In vitro antiviral effect of” Nanosilver” on influenza virus. DARU J. Pharm. Sci. 2015, 17, 88–93. [Google Scholar]
- Lara, H.H.; Ayala-Nuñez, N.V.; Ixtepan-Turrent, L.; Rodriguez-Padilla, C. Mode of antiviral action of silver nanoparticles against HIV-1. J. Nanobiotechnol. 2010, 8, 1. [Google Scholar] [CrossRef]
- Brügger, B.; Glass, B.; Haberkant, P.; Leibrecht, I.; Wieland, F.T.; Kräusslich, H.-G. The HIV lipidome: A raft with an unusual composition. Proc. Natl. Acad. Sci. USA 2006, 103, 2641–2646. [Google Scholar] [CrossRef] [PubMed]
- Kalvodova, L.; Sampaio, J.L.; Cordo, S.; Ejsing, C.S.; Shevchenko, A.; Simons, K. The lipidomes of vesicular stomatitis virus, semliki forest virus, and the host plasma membrane analyzed by quantitative shotgun mass spectrometry. J. Virol. 2009, 83, 7996–8003. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, T.; Kajikawa, M.; Maita, N.; Takeda, M.; Kuroki, K.; Sasaki, K.; Kohda, D.; Yanagi, Y.; Maenaka, K. Crystal structure of measles virus hemagglutinin provides insight into effective vaccines. Proc. Natl. Acad. Sci. USA 2007, 104, 19535–19540. [Google Scholar] [CrossRef] [PubMed]
| Treatment | CC50 (μg/mL) | CE50 (μg/mL) | SI |
|---|---|---|---|
| AuNPs-As | 141.75 | 8.829 | 16.05 |
| HAuCl4 | 231.74 | 31.4 | 7.4 |
| Garlic extract | >1500 | Undetermined | Undetermined |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meléndez-Villanueva, M.A.; Morán-Santibañez, K.; Martínez-Sanmiguel, J.J.; Rangel-López, R.; Garza-Navarro, M.A.; Rodríguez-Padilla, C.; Zarate-Triviño, D.G.; Trejo-Ávila, L.M. Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract. Viruses 2019, 11, 1111. https://doi.org/10.3390/v11121111
Meléndez-Villanueva MA, Morán-Santibañez K, Martínez-Sanmiguel JJ, Rangel-López R, Garza-Navarro MA, Rodríguez-Padilla C, Zarate-Triviño DG, Trejo-Ávila LM. Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract. Viruses. 2019; 11(12):1111. https://doi.org/10.3390/v11121111
Chicago/Turabian StyleMeléndez-Villanueva, Mayra A., Karla Morán-Santibañez, Juan J. Martínez-Sanmiguel, Raúl Rangel-López, Marco A. Garza-Navarro, Cristina Rodríguez-Padilla, Diana G. Zarate-Triviño, and Laura M. Trejo-Ávila. 2019. "Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract" Viruses 11, no. 12: 1111. https://doi.org/10.3390/v11121111
APA StyleMeléndez-Villanueva, M. A., Morán-Santibañez, K., Martínez-Sanmiguel, J. J., Rangel-López, R., Garza-Navarro, M. A., Rodríguez-Padilla, C., Zarate-Triviño, D. G., & Trejo-Ávila, L. M. (2019). Virucidal Activity of Gold Nanoparticles Synthesized by Green Chemistry Using Garlic Extract. Viruses, 11(12), 1111. https://doi.org/10.3390/v11121111






