Comparative Virological and Pathogenic Characteristics of Avian Influenza H5N8 Viruses Detected in Wild Birds and Domestic Poultry in Egypt during the Winter of 2016/2017
Abstract
:1. Introduction
2. Materials and Methods
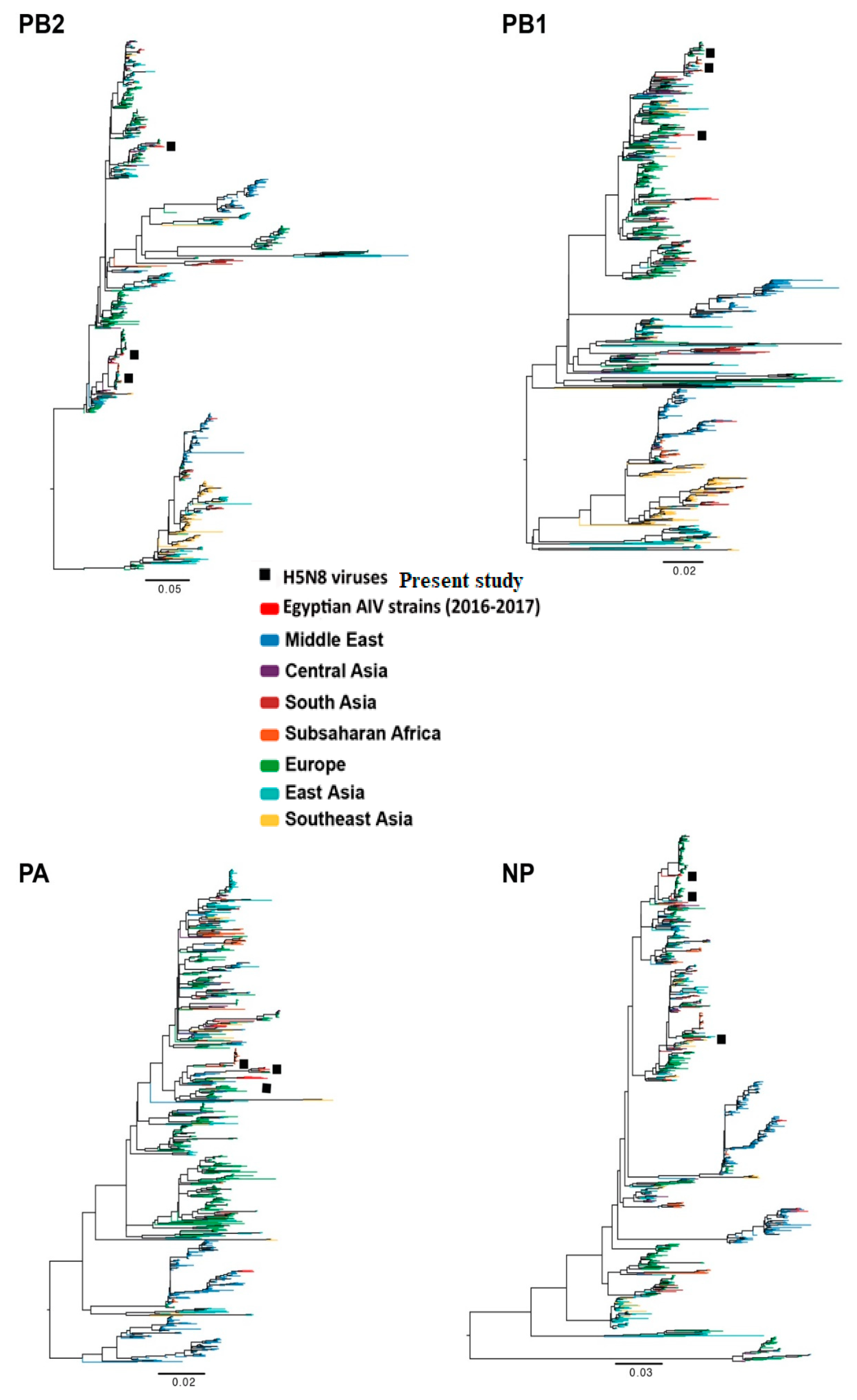
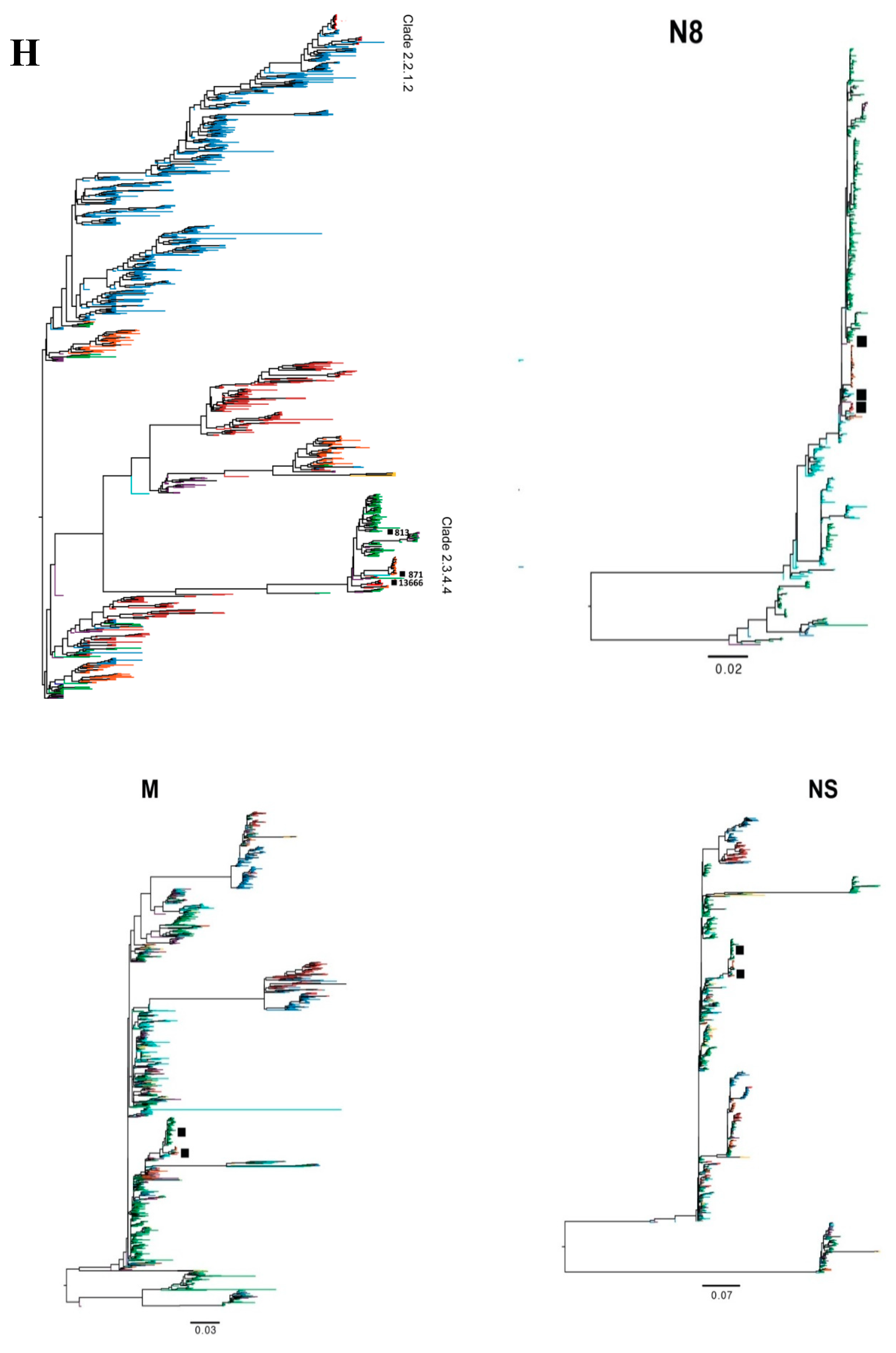
2.1. Phylogenetic Analysis of 2.3.4.4 A (H5N8) Egyptian Influenza Virus Genes
2.2. Cells
2.3. Viruses
2.4. Growth Kinetics of H5N8 Viruses in Mammalian Cells
2.5. Replication Rate of H5N8 Viruses in SPF-ECEs
2.6. Animal Experiments
2.7. Pathogenicity in Mice
2.8. Pathogenicity in Chickens and Ducks
2.9. Histopathology
2.10. Statistical Analysis
3. Results
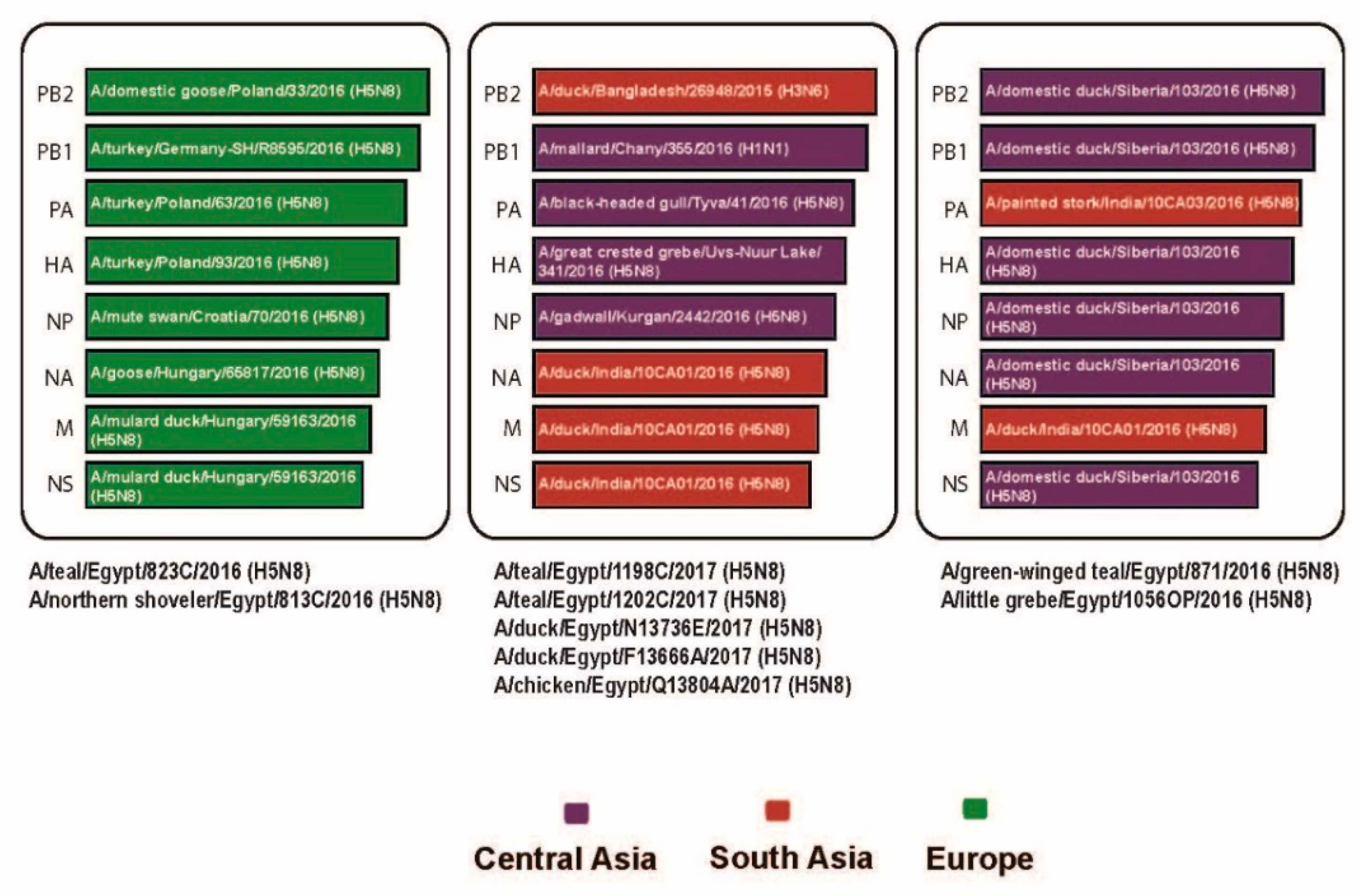
3.1. Source of Introductions of Egyptian H5N8 Genes
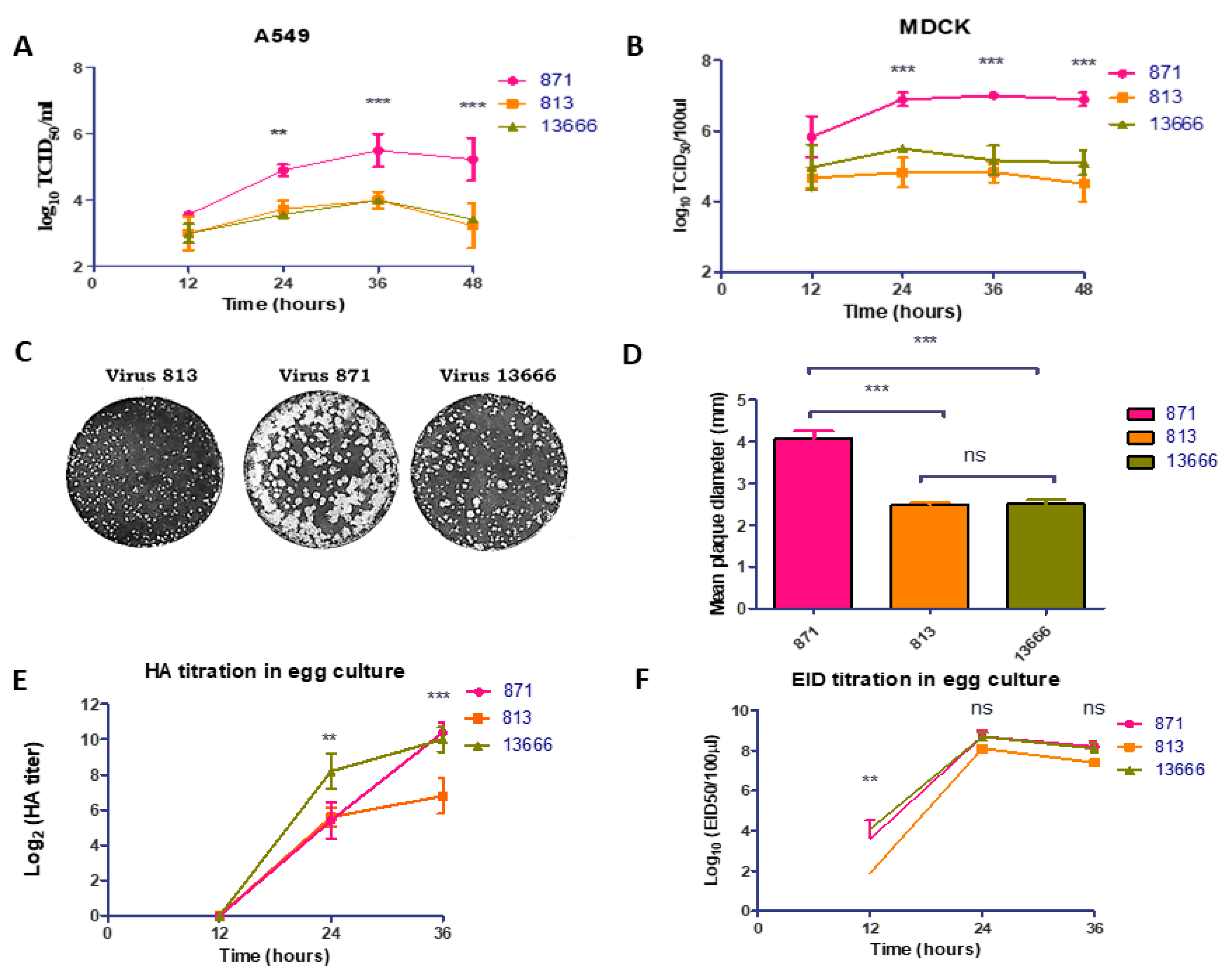
3.2. Growth Kinetics of H5N8 Viruses in Mammalian Cells and SPF-ECEs
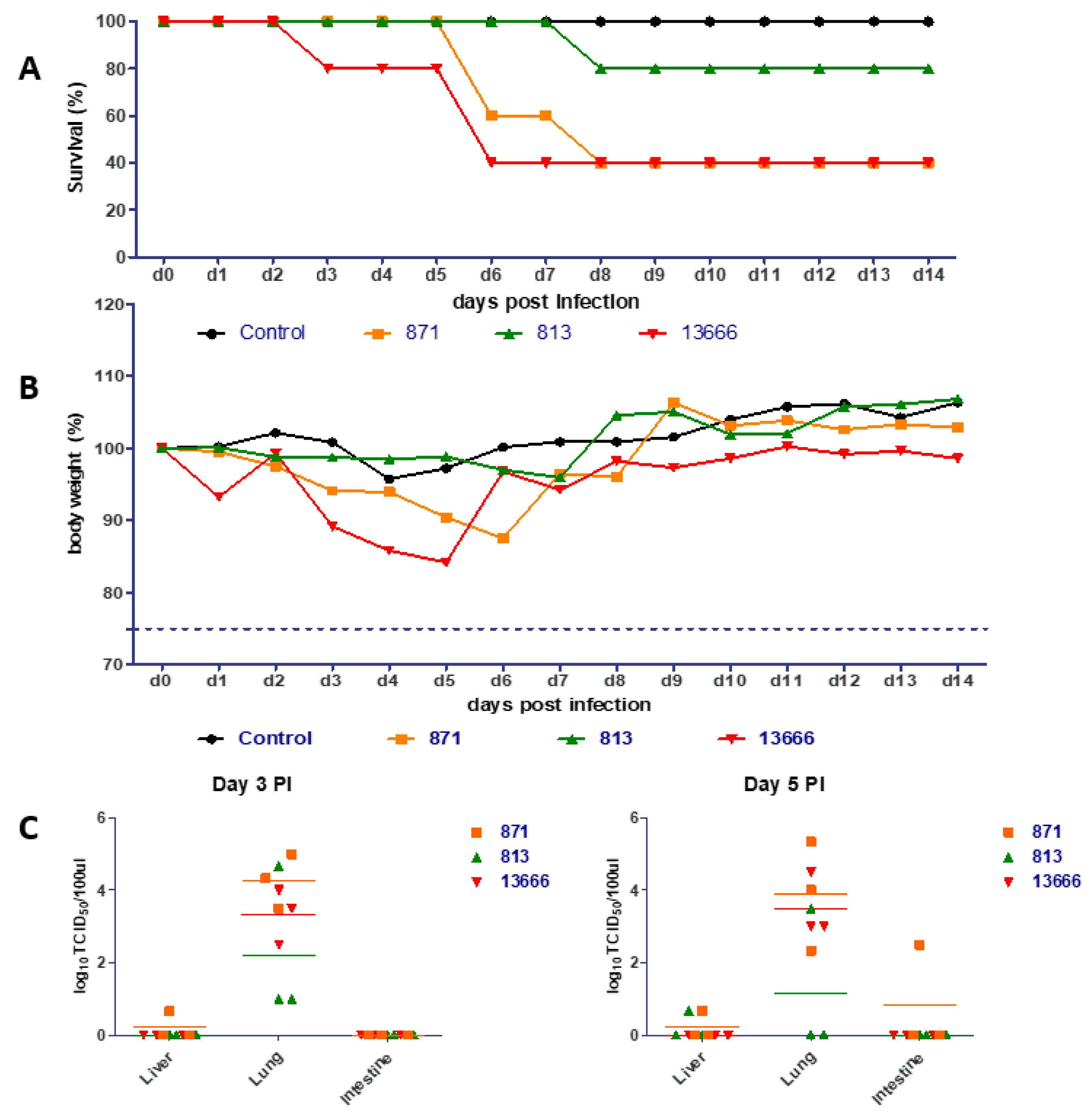
3.3. Pathogenicity in Mice
3.4. Titer in Organs of Mice
3.5. Viral Replication and Pathogenicity in Ducks and Chickens
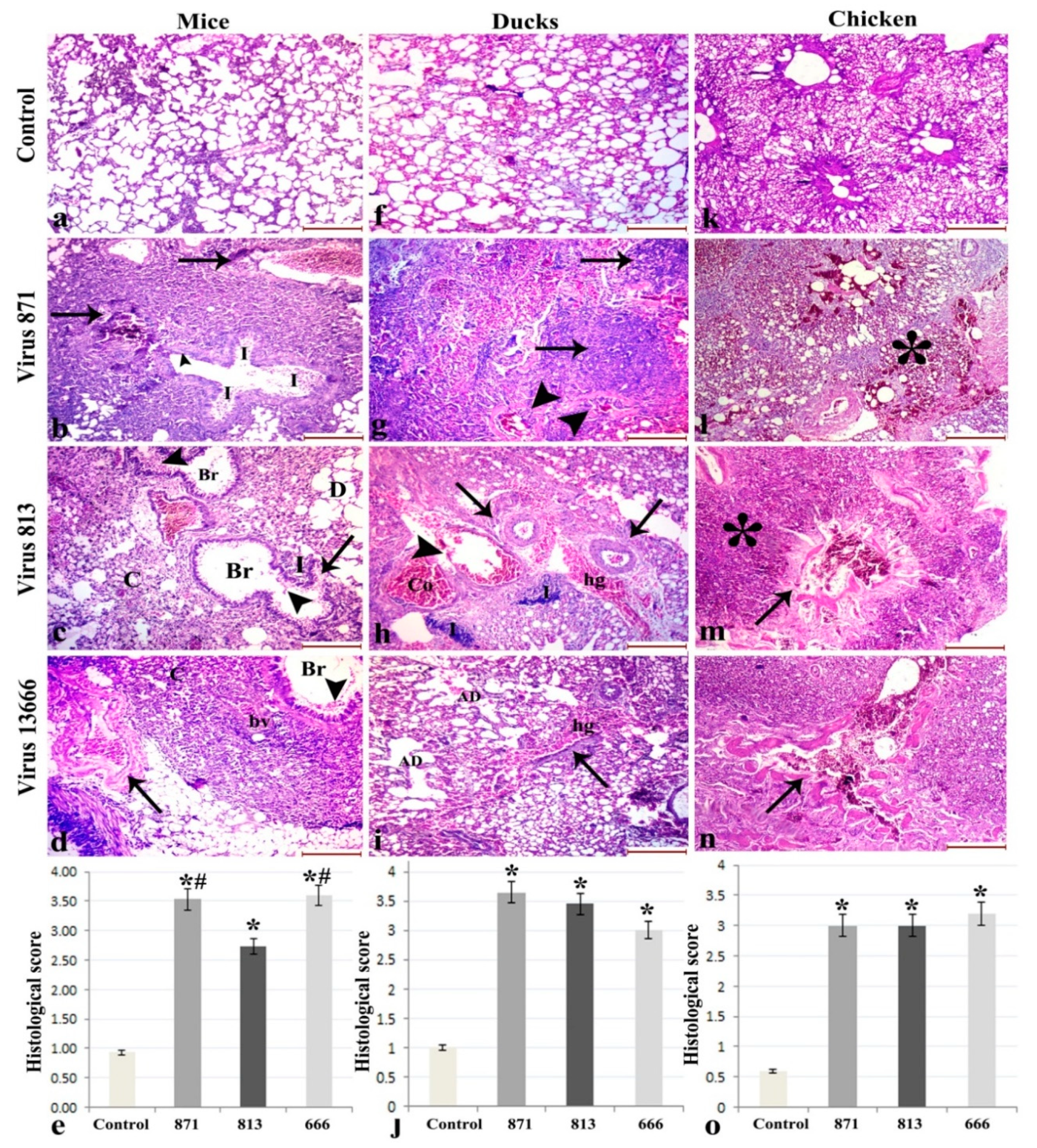
3.6. Histopathology
3.6.1. Lung
3.6.2. Liver
3.6.3. Intestine
3.6.4. Other Organs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Alexander, D.J. A review of avian influenza in different bird species. Vet. Microbiol. 2000, 74, 3–13. [Google Scholar] [CrossRef]
- Webster, R.G.; Bean, W.J.; Gorman, O.T.; Chambers, T.M.; Kawaoka, Y. Evolution and ecology of influenza A viruses. Microbiol. Mol. Boil. Rev. 1992, 56, 152–179. [Google Scholar]
- WHO/OIE/FAO. Continued evolution of highly pathogenic avian influenza A (H5N1): Updated nomenclature. Influenza Other Respir. Viru. 2012, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- De Vries, E.; Guo, H.; Dai, M.; Rottier, P.J.; van Kuppeveld, F.J.; de Haan, C.A. Rapid Emergence of Highly Pathogenic Avian Influenza Subtypes from a Subtype H5N1 Hemagglutinin Variant. Emerg. Infect. Dis. 2015, 21, 842–846. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.H.; Sharshov, K.; Swayne, D.E.; Kurskaya, O.; Sobolev, I.; Kabilov, M.; Alekseev, A.; Irza, V.; Shestopalov, A. Novel Reassortant Clade 2.3.4.4 Avian Influenza A(H5N8) Virus in Wild Aquatic Birds, Russia, 2016. Emerg. Infect. Dis. 2017, 23, 359–360. [Google Scholar] [CrossRef] [Green Version]
- W.H.O. (WHO). Assessment of Risk Associated with Influenza A (H5N8) Virus. Available online: https://www.who.int/influenza/human_animal_interface/avian_influenza/riskassessment_AH5N8_201611/en/ (accessed on 17 November 2016).
- Kandeil, A.; Kayed, A.; Moatasim, Y.; Webby, R.J.; McKenzie, P.P.; Kayali, G.; Ali, M.A. Genetic characterization of highly pathogenic avian influenza A H5N8 viruses isolated from wild birds in Egypt. J. Gen. Virol. 2017, 98, 1573. [Google Scholar] [CrossRef]
- Selim, A.A.; Erfan, A.M.; Hagag, N.; Zanaty, A.; Samir, A.H.; Samy, M.; Abdelhalim, A.; Arafa, A.A.; Soliman, M.A.; Shaheen, M.; et al. Highly Pathogenic Avian Influenza Virus (H5N8) Clade 2.3.4.4 Infection in Migratory Birds, Egypt. Emerg. Infect. Dis. 2017, 23, 1048–1051. [Google Scholar] [CrossRef] [Green Version]
- OIE Update on Avian Influenza in Animals. Available online: http://www.oie.int/wahis_2/public%5C..%5Ctemp%5Creports/en_fup_0000023232_20170314_163139.pdf (accessed on 14 March 2017).
- Yehia, N.; Naguib, M.M.; Li, R.; Hagag, N.; El-Husseiny, M.; Mosaad, Z.; Nour, A.; Rabea, N.; Hasan, W.M.; Hassan, M.K.; et al. Multiple introductions of reassorted highly pathogenic avian influenza viruses (H5N8) clade 2.3.4.4b causing outbreaks in wild birds and poultry in Egypt. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2018, 58, 56–65. [Google Scholar] [CrossRef]
- Salaheldin, A.H.; El-Hamid, H.S.; Elbestawy, A.R.; Veits, J.; Hafez, H.M.; Mettenleiter, T.C.; Abdelwhab, E.M. Multiple Introductions of Influenza A(H5N8) Virus into Poultry, Egypt, 2017. Emerg. Infect. Dis. 2018, 24, 943. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Yang, Z. Phylogenetic analysis using parsimony and likelihood methods. J. Mol. Evol. 1996, 42, 294–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints12. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Zanin, M.; Kocer, Z.A.; Poulson, R.L.; Gabbard, J.D.; Howerth, E.W.; Jones, C.A.; Friedman, K.; Seiler, J.; Danner, A.; Kercher, L.; et al. Potential for Low-Pathogenic Avian H7 Influenza A Viruses To Replicate and Cause Disease in a Mammalian Model. J. Virol. 2017, 91, e01934-16. [Google Scholar] [CrossRef] [PubMed]
- Volmer, C.; Soubies, S.M.; Grenier, B.; Guerin, J.L.; Volmer, R. Immune response in the duck intestine following infection with low-pathogenic avian influenza viruses or stimulation with a Toll-like receptor 7 agonist administered orally. J. Gen. Virol. 2011, 92, 534–543. [Google Scholar] [CrossRef]
- Zhang, F.; Yu, M.; Weiland, E.; Morrissy, C.; Zhang, N.; Westbury, H.; Wang, L.F. Characterization of epitopes for neutralizing monoclonal antibodies to classical swine fever virus E2 and Erns using phage-displayed random peptide library. Arch. Virol. 2006, 151, 37–54. [Google Scholar] [CrossRef]
- Kandeil, A.; Hicks, J.T.; Young, S.G.; El Taweel, A.N.; Kayed, A.S.; Moatasim, Y.; Kutkat, O.; Bagato, O.; McKenzie, P.P.; Cai, Z.; et al. Active surveillance and genetic evolution of avian influenza viruses in Egypt, 2016–2018. Emerg. Microbes Infect. 2019, 8, 1370–1382. [Google Scholar] [CrossRef]
- Cheng, Y.Y.; Yang, S.R.; Wang, Y.T.; Lin, Y.H.; Chen, C.J. Amino Acid Residues 68–71 Contribute to Influenza A Virus PB1-F2 Protein Stability and Functions. Front. Microbiol. 2017, 8, 692. [Google Scholar] [CrossRef]
- Schrauwen, E.J.; Herfst, S.; Leijten, L.M.; van Run, P.; Bestebroer, T.M.; Linster, M.; Bodewes, R.; Kreijtz, J.H.; Rimmelzwaan, G.F.; Osterhaus, A.D.; et al. The multibasic cleavage site in H5N1 virus is critical for systemic spread along the olfactory and hematogenous routes in ferrets. J. Virol. 2012, 86, 3975–3984. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, Y.; Sun, H.; Pu, J.; Bi, Y.; Shi, Y.; Lu, X.; Li, J.; Zhu, Q.; Gao, G.F.; et al. A single amino acid at the hemagglutinin cleavage site contributes to the pathogenicity and neurovirulence of H5N1 influenza virus in mice. J. Virol. 2012, 86, 6924–6931. [Google Scholar] [CrossRef]
- Kajihara, M.; Sakoda, Y.; Soda, K.; Minari, K.; Okamatsu, M.; Takada, A.; Kida, H. The PB2, PA, HA, NP, and NS genes of a highly pathogenic avian influenza virus A/whooper swan/Mongolia/3/2005 (H5N1) are responsible for pathogenicity in ducks. Virol. J. 2013, 10, 45. [Google Scholar] [CrossRef]
- Kim, Y.I.; Pascua, P.N.; Kwon, H.I.; Lim, G.J.; Kim, E.H.; Yoon, S.W.; Park, S.J.; Kim, S.M.; Choi, E.J.; Si, Y.J.; et al. Pathobiological features of a novel, highly pathogenic avian influenza A(H5N8) virus. Emerg. Microbes Infect. 2014, 3, e75. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Kwon, J.H.; Noh, J.Y.; Park, J.K.; Yuk, S.S.; Erdene-Ochir, T.O.; Lee, J.B.; Park, S.Y.; Choi, I.S.; Lee, S.W.; et al. Pathogenicity of the Korean H5N8 highly pathogenic avian influenza virus in commercial domestic poultry species. Avian Pathol. J. WVPA 2016, 45, 208–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, H.M.; Lee, E.K.; Song, B.M.; Jeong, J.; Choi, J.G.; Jeong, J.; Moon, O.K.; Yoon, H.; Cho, Y.; Kang, Y.M.; et al. Novel reassortant influenza A(H5N8) viruses among inoculated domestic and wild ducks, South Korea, 2014. Emerg. Infect. Dis. 2015, 21, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Pulit-Penaloza, J.A.; Sun, X.; Creager, H.M.; Zeng, H.; Belser, J.A.; Maines, T.R.; Tumpey, T.M. Pathogenesis and Transmission of Novel Highly Pathogenic Avian Influenza H5N2 and H5N8 Viruses in Ferrets and Mice. J. Virol. 2015, 89, 10286–10293. [Google Scholar] [CrossRef] [Green Version]
- Kaplan, B.S.; Russier, M.; Jeevan, T.; Marathe, B.; Govorkova, E.A.; Russell, C.J.; Kim-Torchetti, M.; Choi, Y.K.; Brown, I.; Saito, T.; et al. Novel Highly Pathogenic Avian A(H5N2) and A(H5N8) Influenza Viruses of Clade 2.3.4.4 from North America Have Limited Capacity for Replication and Transmission in Mammals. Msphere 2016, 1, e00003-16. [Google Scholar] [CrossRef] [Green Version]
- Koçer, Z.A.; Fan, Y.; Huether, R.; Obenauer, J.; Webby, R.J.; Zhang, J.; Webster, R.G.; Wu, G. Survival analysis of infected mice reveals pathogenic variations in the genome of avian H1N1 viruses. Sci. Rep. 2014, 4, 7455. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Chen, H.; Huang, J.; Chen, Y.; Gu, M.; Wang, X.; Hu, S.; Liu, X.; Liu, X. A nonpathogenic duck-origin H9N2 influenza A virus adapts to high pathogenicity in mice. Arch. Virol. 2014, 159, 2243–2252. [Google Scholar] [CrossRef]
- Teng, Q.; Zhang, X.; Xu, D.; Zhou, J.; Dai, X.; Chen, Z.; Li, Z. Characterization of an H3N2 canine influenza virus isolated from Tibetan mastiffs in China. Vet. Microbiol. 2013, 162, 345–352. [Google Scholar] [CrossRef]
- Youk, S.S.; Lee, D.H.; Leyson, C.M.; Smith, D.; Criado, M.F.; DeJesus, E.; Swayne, D.E.; Pantin-Jackwood, M.J. Loss of Fitness of Mexican H7N3 Highly Pathogenic Avian Influenza Virus in Mallards after Circulating in Chickens. J. Virol. 2019, 93, e00543-19. [Google Scholar] [CrossRef]
- Marjuki, H.; Scholtissek, C.; Franks, J.; Negovetich, N.J.; Aldridge, J.R.; Salomon, R.; Finkelstein, D.; Webster, R.G. Three amino acid changes in PB1-F2 of highly pathogenic H5N1 avian influenza virus affect pathogenicity in mallard ducks. Arch. Virol. 2010, 155, 925–934. [Google Scholar] [CrossRef] [Green Version]
- James, J.; Howard, W.; Iqbal, M.; Nair, V.K.; Barclay, W.S.; Shelton, H. Influenza A virus PB1-F2 protein prolongs viral shedding in chickens lengthening the transmission window. J. Gen. Virol. 2016, 97, 2516–2527. [Google Scholar] [CrossRef] [PubMed]
- Kamal, R.P.; Alymova, I.V.; York, I.A. Evolution and Virulence of Influenza A Virus Protein PB1-F2. Int. J. Mol. Sci. 2018, 19, 96. [Google Scholar] [CrossRef] [PubMed]
- Schmolke, M.; Manicassamy, B.; Pena, L.; Sutton, T.; Hai, R.; Varga, Z.T.; Hale, B.G.; Steel, J.; Pérez, D.R.; García-Sastre, A. Differential Contribution of PB1-F2 to the Virulence of Highly Pathogenic H5N1 Influenza A Virus in Mammalian and Avian Species. PLoS Pathog. 2011, 7, e1002186. [Google Scholar] [CrossRef] [PubMed]
- Conenello, G.M.; Zamarin, D.; Perrone, L.A.; Tumpey, T.; Palese, P. A Single Mutation in the PB1-F2 of H5N1 (HK/97) and 1918 Influenza A Viruses Contributes to Increased Virulence. PLoS Pathog. 2007, 3, e141. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.L.; Hornung, F.; Boyd, K.L.; Smith, A.M.; McKeon, R.; Bennink, J.; Yewdell, J.W.; McCullers, J.A. Expression of the 1918 influenza A virus PB1-F2 enhances the pathogenesis of viral and secondary bacterial pneumonia. Cell Host Microbe 2007, 2, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Alymova, I.V.; Green, A.M.; van de Velde, N.; McAuley, J.L.; Boyd, K.L.; Ghoneim, H.E.; McCullers, J.A. Immunopathogenic and antibacterial effects of H3N2 influenza A virus PB1-F2 map to amino acid residues 62, 75, 79, and 82. J. Virol. 2011, 85, 12324–12333. [Google Scholar] [CrossRef]
- Naffakh, N.; Tomoiu, A.; Rameix-Welti, M.A.; van der Werf, S. Host restriction of avian influenza viruses at the level of the ribonucleoproteins. Annu. Rev. Microbiol. 2008, 62, 403–424. [Google Scholar] [CrossRef]
- Chen, G.W.; Chang, S.C.; Mok, C.K.; Lo, Y.L.; Kung, Y.N.; Huang, J.H.; Shih, Y.H.; Wang, J.Y.; Chiang, C.; Chen, C.J.; et al. Genomic signatures of human versus avian influenza A viruses. Emerg. Infect. Dis. 2006, 12, 1353–1360. [Google Scholar] [CrossRef]
- Jackson, D.; Hossain, M.J.; Hickman, D.; Perez, D.R.; Lamb, R.A. A new influenza virus virulence determinant: The NS1 protein four C-terminal residues modulate pathogenicity. Proc. Natl. Acad. Sci. USA 2008, 105, 4381–4386. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.J.; Chen, G.W.; Wang, C.H.; Huang, C.H.; Wang, Y.C.; Shih, S.R. Differential Localization and Function of PB1-F2 Derived from Different Strains of Influenza A Virus. J. Virol. 2010, 84, 10051–10062. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Sun, Y.; Xu, Q.; Tan, Y.; Pu, J.; Yang, H.; Brown, E.G.; Liu, J. Mouse-adapted H9N2 influenza A virus PB2 protein M147L and E627K mutations are critical for high virulence. PLoS ONE 2012, 7, e40752. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, E.C.; Wise, H.M.; Kudryavtseva, K.; Curran, M.D.; Digard, P. Characterisation of influenza A viruses with mutations in segment 5 packaging signals. Vaccine 2009, 27, 6270–6275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stoute, S.; Chin, R.; Crossley, B.; Gabriel Senties-Cue, C.; Bickford, A.; Pantin-Jackwood, M.; Breitmeyer, R.; Jones, A.; Carnaccini, S.; Shivaprasad, H.L. Highly Pathogenic Eurasian H5N8 Avian Influenza Outbreaks in Two Commercial Poultry Flocks in California. Avian Dis. 2016, 60, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Prokopyeva, E.A.; Zinserling, V.A.; Bae, Y.C.; Kwon, Y.; Kurskaya, O.G.; Sobolev, I.A.; Kozhin, P.M.; Komissarov, A.; Fadeev, A.; Petrov, V.; et al. Pathology of A(H5N8) (Clade 2.3.4.4) Virus in Experimentally Infected Chickens and Mice. Interdiscip. Perspect. Infect. Dis. 2019, 2019, 4124865. [Google Scholar] [CrossRef] [PubMed]


| Virus | 871 | 813 | 13666 | |
|---|---|---|---|---|
| Organ | ||||
| Liver | 2 (1/3) b | 1 (1/3) | 1.5 (1/3) | |
| Spleen | 2.25 ± 0.35 (2/3) | 1.5 ± 0 (2/3) | 1.5 (1/3) | |
| Intestine | 2 ± 0 (3/3) | 1.33 ± 0.3 (3/3) | 1 (1/3) | |
| Kidney | 1.5 ± 0 (3/3) | 1.5 ± 0 (3/3) | 1 (1/3) | |
| Lung | 2 ± 0.7 (2/3) | 1.5 ± 0 (2/3) | 1.5 (1/3) | |
| Trachea | 1.16 ± 0.6 (3/3) | 1.7 ± 0.8 (3/3) | 1 ± 0.7 (2/3) | |
| Brain | 1.75 ± 0.35 (2/3) | 1.7 ± 0.3 (3/3) | 1.5(1/3) | |
| Virus | 871 | 813 | 13666 | |
|---|---|---|---|---|
| Organ | ||||
| Liver | 2.5 ± 0.8 (3/4) b | 3.375 ± 0.25 (4/4) | 2.875 ± 0.25 (4/4) | |
| Spleen | 3.875 ± 1.7 (3/4) | 3 ± 0.57 (4/4) | 2 ± 1.4 (2/4) | |
| Intestine | 3.5 ± 0 (3/4) | 3.5 ± 0 (4/4) | 3 ± 0 (4/4) | |
| Kidney | 3.625 ± 0.25 (4/4) | 3.5 ± 0 (4/4) | 3.5 ± 0 (4/4) | |
| Lung | 3 ± 1.3 (4/4) | 3.875 ± 0.25 (4/4) | 2.75 ± 0.5 (4/4) | |
| Trachea | 3 ± 1.3 (4/4) | 3.5 ± 0 (4/4) | 2.625 ± 0.75 (4/4) | |
| Brain | 3.1 ± 1.04 (3/4) | 3.5 ± 0 (4/4) | 3.5 ± 0 (4/4) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moatasim, Y.; Kandeil, A.; Aboulhoda, B.E.; El-Shesheny, R.; Alkhazindar, M.; AbdElSalam, E.T.; Kutkat, O.; Kamel, M.N.; El Taweel, A.N.; Mostafa, A.; et al. Comparative Virological and Pathogenic Characteristics of Avian Influenza H5N8 Viruses Detected in Wild Birds and Domestic Poultry in Egypt during the Winter of 2016/2017. Viruses 2019, 11, 990. https://doi.org/10.3390/v11110990
Moatasim Y, Kandeil A, Aboulhoda BE, El-Shesheny R, Alkhazindar M, AbdElSalam ET, Kutkat O, Kamel MN, El Taweel AN, Mostafa A, et al. Comparative Virological and Pathogenic Characteristics of Avian Influenza H5N8 Viruses Detected in Wild Birds and Domestic Poultry in Egypt during the Winter of 2016/2017. Viruses. 2019; 11(11):990. https://doi.org/10.3390/v11110990
Chicago/Turabian StyleMoatasim, Yassmin, Ahmed Kandeil, Basma Emad Aboulhoda, Rabeh El-Shesheny, Maha Alkhazindar, Elsayed Tarek AbdElSalam, Omnia Kutkat, Mina Nabil Kamel, Ahmed Nageh El Taweel, Ahmed Mostafa, and et al. 2019. "Comparative Virological and Pathogenic Characteristics of Avian Influenza H5N8 Viruses Detected in Wild Birds and Domestic Poultry in Egypt during the Winter of 2016/2017" Viruses 11, no. 11: 990. https://doi.org/10.3390/v11110990
APA StyleMoatasim, Y., Kandeil, A., Aboulhoda, B. E., El-Shesheny, R., Alkhazindar, M., AbdElSalam, E. T., Kutkat, O., Kamel, M. N., El Taweel, A. N., Mostafa, A., Hicks, J. T., Abd elghaffar, S. K., Kayali, G., & Ali, M. A. (2019). Comparative Virological and Pathogenic Characteristics of Avian Influenza H5N8 Viruses Detected in Wild Birds and Domestic Poultry in Egypt during the Winter of 2016/2017. Viruses, 11(11), 990. https://doi.org/10.3390/v11110990










