Expanding Repertoire of Plant Positive-Strand RNA Virus Proteases
Abstract
1. Introduction
2. Functional Protease Types
3. Chymotrypsin-Like Serine and Cysteine Proteases
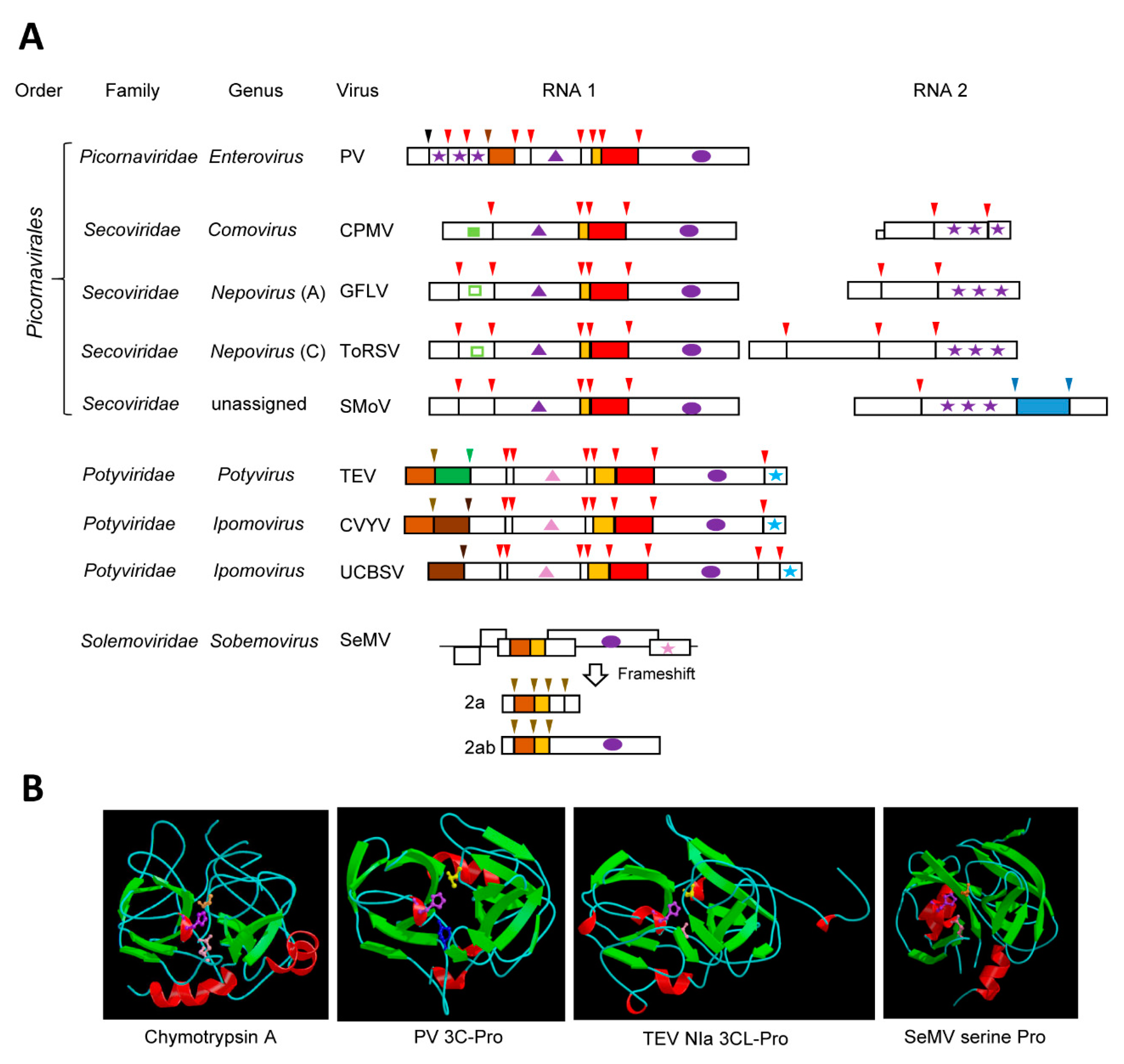
3.1. The Archetype Picornavirus 3C-Pros
3.2. The 3CL-Pros of Viruses in the Family Secoviridae and Their Diverse Cleavage Site Specificities
3.3. The well-characterized potyvirus NIa proteases
3.4. The Sobemovirus Serine Protease
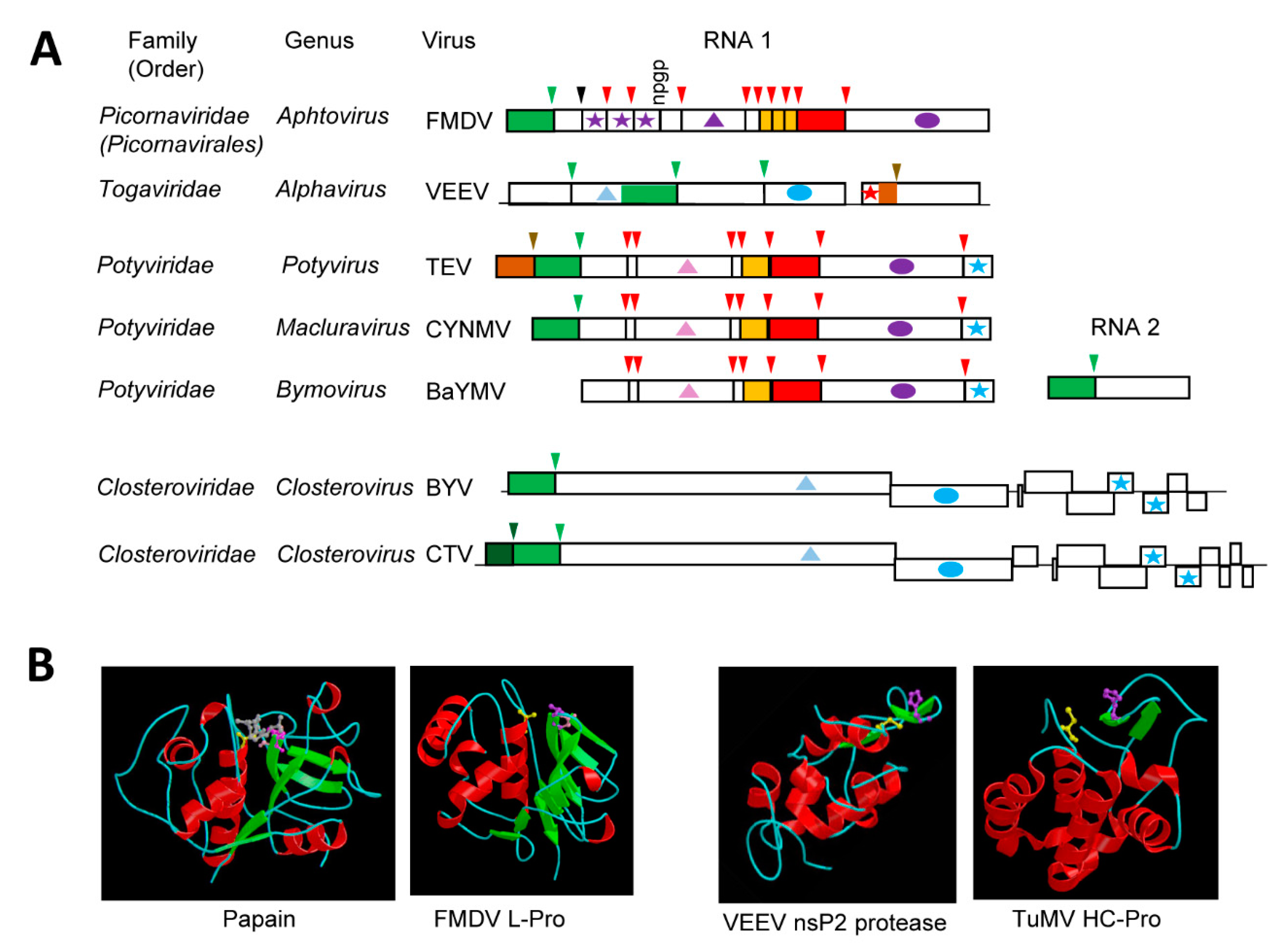
3.5. The Compact and Diverse Potyvirid P1 Serine Proteases and Their Intricate Regulatory Mechanisms
4. The Diverse “Papain-Like” Cysteine Proteases
4.1. The Multifunctional Potyvirid HC-Pro Protease with a Minimalistic Papain-Like Fold
4.2. The single or Tandem Closterovirus Leader Proteases
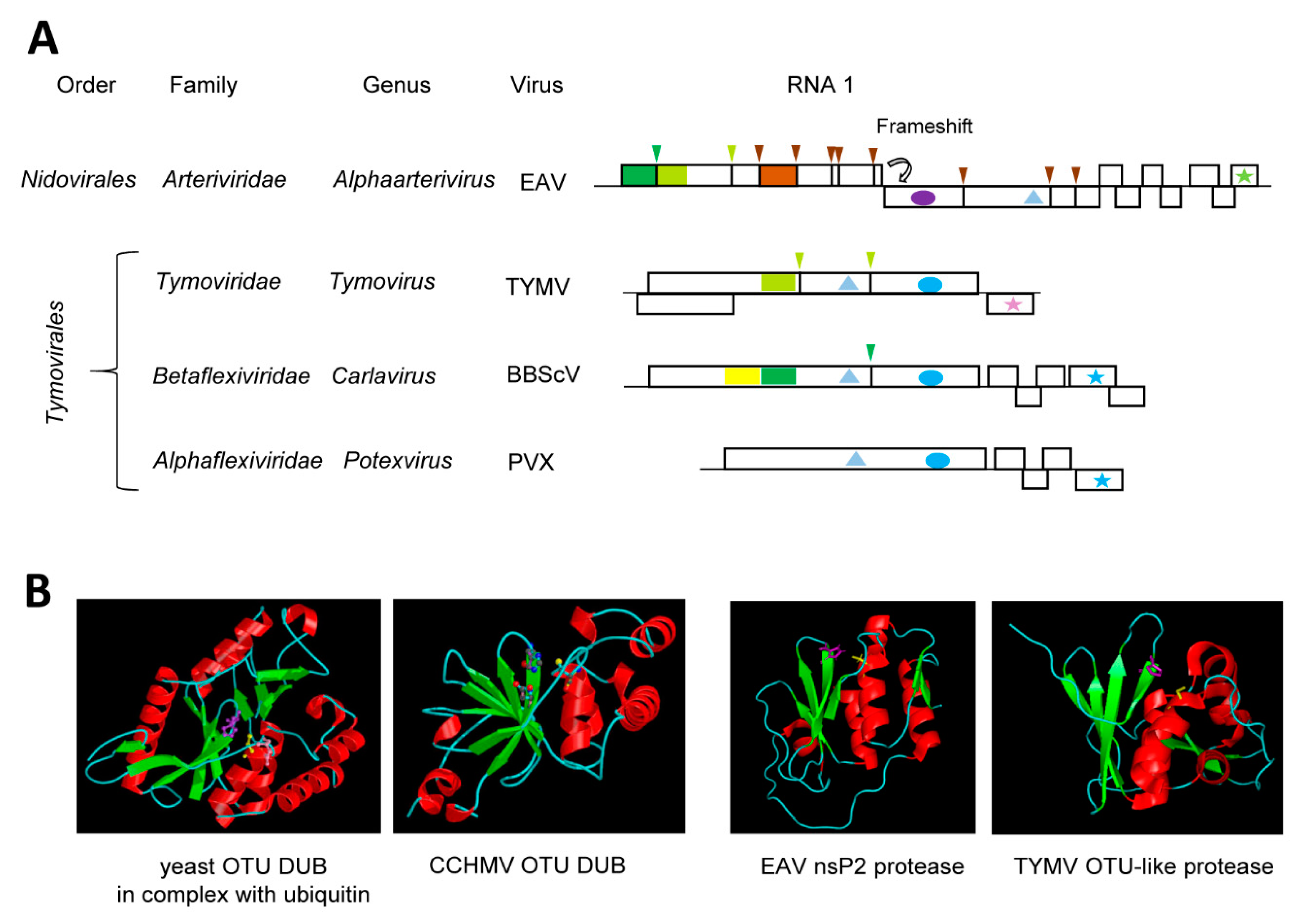
4.3. The Tymovirus Cysteine Protease with a Compact Ovarian-Tumor (OTU) Domain-Like Fold Driven by Its Dual Function as a Protease and Deubiquitinase
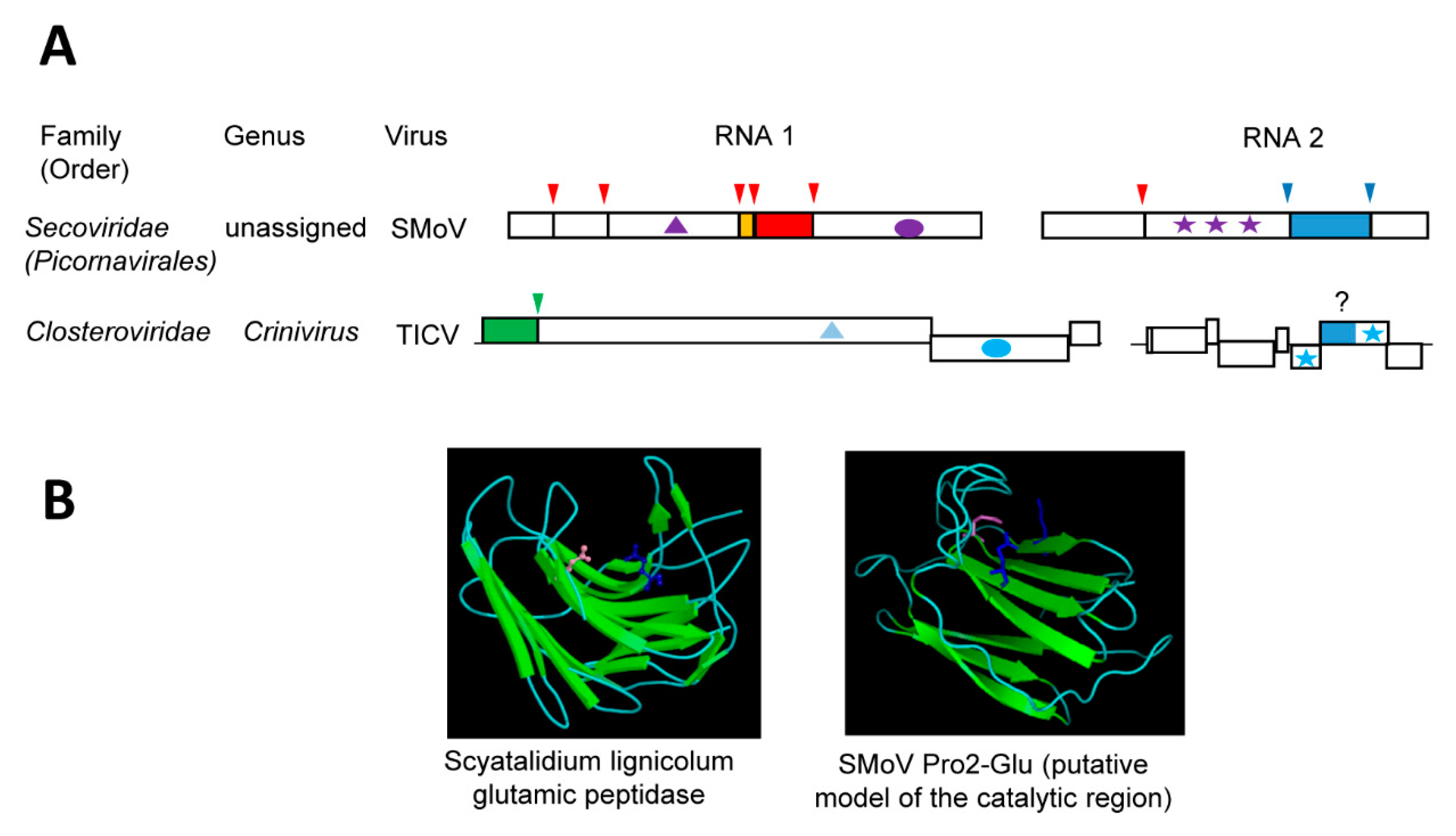
5. The Novel Glutamic Protease of Strawberry Mottle Virus (Family Secoviridae)
6. Aspartic Proteases Encoded by Reverse-Transcribing Viruses and by a Plant Negative-Strand RNA Virus, but Not (Yet?) by (+)-Strand RNA Viruses
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wolf, Y.I.; Kazlauskas, D.; Iranzo, J.; Lucia-Sanz, A.; Kuhn, J.H.; Krupovic, M.; Dolja, V.V.; Koonin, E.V. Origins and Evolution of the Global RNA Virome. MBio 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Krupovic, M.; Koonin, E.V. Multiple origins of viral capsid proteins from cellular ancestors. Proc. Natl. Acad. Sci. USA 2017, 114, E2401–E2410. [Google Scholar] [CrossRef] [PubMed]
- Koonin, E.V.; Dolja, V.V. Virus world as an evolutionary network of viruses and capsidless selfish elements. Microbiol. Mol. Biol. Rev. 2014, 78, 278–303. [Google Scholar] [CrossRef] [PubMed]
- Koonin, E.V.; Wolf, Y.I.; Nagasaki, K.; Dolja, V.V. The Big Bang of picorna-like virus evolution antedates the radiation of eukaryotic supergroups. Nat. Rev. Microbiol. 2008, 6, 925–939. [Google Scholar] [CrossRef] [PubMed]
- Koonin, E.V.; Dolja, V.V.; Krupovic, M. Origins and evolution of viruses of eukaryotes: The ultimate modularity. Virology 2015, 479, 2–25. [Google Scholar] [CrossRef]
- Dolja, V.V.; Koonin, E.V. Metagenomics reshapes the concepts of RNA virus evolution by revealing extensive horizontal virus transfer. Virus Res. 2018, 244, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Mascia, T.; Gallitelli, D. Synergies and antagonisms in virus interactions. Plant Sci. 2016, 252, 176–192. [Google Scholar] [CrossRef]
- Roossinck, M.J. Lifestyles of plant viruses. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1899–1905. [Google Scholar] [CrossRef]
- Dietzgen, R.G.; Mann, K.S.; Johnson, K.N. Plant Virus-Insect Vector Interactions: Current and Potential Future Research Directions. Viruses 2016, 8, 303. [Google Scholar] [CrossRef]
- Whitfield, A.E.; Falk, B.W.; Rotenberg, D. Insect vector-mediated transmission of plant viruses. Virology 2015, 479–480, 278–289. [Google Scholar] [CrossRef]
- Elena, S.F.; Fraile, A.; Garcia-Arenal, F. Evolution and emergence of plant viruses. Adv. Virus Res. 2014, 88, 161–191. [Google Scholar] [PubMed]
- Borderia, A.V.; Rozen-Gagnon, K.; Vignuzzi, M. Fidelity Variants and RNA Quasispecies. Curr. Top. Microbiol. Immunol. 2016, 392, 303–322. [Google Scholar] [PubMed]
- Borderia, A.V.; Stapleford, K.A.; Vignuzzi, M. RNA virus population diversity: Implications for inter-species transmission. Curr. Opin. Virol. 2011, 1, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C. Error thresholds and the constraints to RNA virus evolution. Trends Microbiol. 2003, 11, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Belshaw, R.; Gardner, A.; Rambaut, A.; Pybus, O.G. Pacing a small cage: Mutation and RNA viruses. Trends Ecol. Evol. 2008, 23, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Rodamilans, B.; Shan, H.; Pasin, F.; Garcia, J.A. Plant Viral Proteases: Beyond the Role of Peptide Cutters. Front. Plant Sci. 2018, 9, 666. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, O.H.; Svedin, E.; Kapell, S.; Nurminen, A.; Hytonen, V.P.; Flodstrom-Tullberg, M. Enteroviral proteases: Structure, host interactions and pathogenicity. Rev. Med. Virol. 2016, 26, 251–267. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Chen, S.; Cheng, A.; Wang, M. Roles of the Picornaviral 3C Proteinase in the Viral Life Cycle and Host Cells. Viruses 2016, 8, 82. [Google Scholar] [CrossRef]
- Jensen, L.M.; Walker, E.J.; Jans, D.A.; Ghildyal, R. Proteases of human rhinovirus: Role in infection. Meth. Mol. Biol. 2015, 1221, 129–141. [Google Scholar]
- Mielech, A.M.; Chen, Y.; Mesecar, A.D.; Baker, S.C. Nidovirus papain-like proteases: Multifunctional enzymes with protease, deubiquitinating and deISGylating activities. Virus Res. 2014, 194, 184–190. [Google Scholar] [CrossRef]
- Yost, S.A.; Marcotrigiano, J. Viral precursor polyproteins: Keys of regulation from replication to maturation. Curr. Opin. Virol. 2013, 3, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Le Gall, O.; Christian, P.; Fauquet, C.M.; King, A.M.; Knowles, N.J.; Nakashima, N.; Stanway, G.; Gorbalenya, A.E. Picornavirales, a proposed order of positive-sense single-stranded RNA viruses with a pseudo-T = 3 virion architecture. Arch. Virol. 2008, 153, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Sanfacon, H.; Gorbalenya, A.E.; Knowles, N.J.; Chen, Y. Order Picornavirales. In Virus Taxonomy: Classification and Nomenclature of Viruses; Ninth Report of the International Committee on the Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Elseviers: San Diego, CA, USA, 2011; pp. 835–839. [Google Scholar]
- Martelli, G.P.; Adams, M.J.; Kreuze, J.F.; Dolja, V.V. Family Flexiviridae: A Case Study in Virion and Genome Plasticity. Annu. Rev. Phytopathol. 2007, 45, 73–100. [Google Scholar] [CrossRef] [PubMed]
- Dolja, V.V.; Kreuze, J.F.; Valkonen, J.P. Comparative and functional genomics of closteroviruses. Virus Res. 2006, 117, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Valli, A.A.; Gallo, A.; Rodamilans, B.; Lopez-Moya, J.J.; Garcia, J.A. The HCPro from the Potyviridae family: An enviable multitasking Helper Component that every virus would like to have. Mol. Plant Pathol. 2018, 19, 744–763. [Google Scholar] [CrossRef] [PubMed]
- Jupin, I.; Ayach, M.; Jomat, L.; Fieulaine, S.; Bressanelli, S. A mobile loop near the active site acts as a switch between the dual activities of a viral protease/deubiquitinase. PLoS Pathog. 2017, 13, e1006714. [Google Scholar] [CrossRef]
- Rawlings, N.D.; Barrett, A.J.; Thomas, P.D.; Huang, X.; Bateman, A.; Finn, R.D. The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res. 2018, 46, D624–D632. [Google Scholar] [CrossRef]
- King, A.M.Q.; Lefkowitz, E.J.; Mushegian, A.R.; Adams, M.J.; Dutilh, B.E.; Gorbalenya, A.E.; Harrach, B.; Harrison, R.L.; Junglen, S.; Knowles, N.J.; et al. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2018). Arch. Virol. 2018, 163, 2601–2631. [Google Scholar] [CrossRef]
- Thomas, E.L.; van der Hoorn, R.A.L. Ten Prominent Host Proteases in Plant-Pathogen Interactions. Int. J. Mol. Sci. 2018, 19, 639. [Google Scholar] [CrossRef]
- Van der Hoorn, R.A. Plant proteases: From phenotypes to molecular mechanisms. Annu. Rev. Plant Biol. 2008, 59, 191–223. [Google Scholar] [CrossRef]
- Tong, L. Viral proteases. Chem. Rev. 2002, 102, 4609–4626. [Google Scholar] [CrossRef] [PubMed]
- Verdaguer, N.; Ferrero, D.; Murthy, M. Viruses and viral proteins. IUCrJ 2014, 1, 492–504. [Google Scholar] [CrossRef] [PubMed]
- Babe, L.M.; Craik, C.S. Viral proteases: Evolution of diverse structural motifs to optimize function. Cell 1997, 91, 427–430. [Google Scholar] [CrossRef]
- Mann, K.S.; Chisholm, J.; Sanfacon, H. Strawberry mottle virus (family Secoviridae, order Picornavirales) encodes a novel glutamic protease to process the RNA2 polyprotein at two cleavage sites. J. Virol. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.D.; Flint, M. Virus-encoded proteinases of the picornavirus super-group. J. Gen. Virol. 1997, 78 (Pt 4), 699–723. [Google Scholar] [CrossRef] [PubMed]
- Seipelt, J.; Guarne, A.; Bergmann, E.; James, M.; Sommergruber, W.; Fita, I.; Skern, T. The structures of picornaviral proteinases. Virus Res. 1999, 62, 159–168. [Google Scholar] [CrossRef]
- Thompson, J.R.; Dasgupta, I.; Fuchs, M.; Iwanami, T.; Karasev, A.V.; Petrzik, K.; Sanfacon, H.; Tzanetakis, I.; van der Vlugt, R.; Wetzel, T.; et al. ICTV Virus Taxonomy Profile: Secoviridae. J. Gen. Virol. 2017, 98, 529–531. [Google Scholar] [CrossRef]
- Wylie, S.J.; Adams, M.; Chalam, C.; Kreuze, J.; Lopez-Moya, J.J.; Ohshima, K.; Praveen, S.; Rabenstein, F.; Stenger, D.; Wang, A.; et al. ICTV Virus Taxonomy Profile: Potyviridae. J. Gen. Virol. 2017, 98, 352–354. [Google Scholar] [CrossRef]
- Wellink, J.; van Kammen, A. Proteases involved in the processing of viral polyproteins. Brief review. Arch. Virol. 1988, 98, 1–26. [Google Scholar] [CrossRef]
- Dougherty, W.G.; Semler, B.L. Expression of virus-encoded proteinases: Functional and structural similarities with cellular enzymes. Microbiol. Rev. 1993, 57, 781–822. [Google Scholar]
- Gorbalenya, A.E.; Koonin, E.V.; Blinov, V.M.; Donchenko, A.P. Sobemovirus genome appears to encode a serine protease related to cysteine proteases of picornaviruses. FEBS Lett. 1988, 236, 287–290. [Google Scholar] [CrossRef]
- Gorbalenya, A.E.; Donchenko, A.P.; Blinov, V.M.; Koonin, E.V. Cysteine proteases of positive strand RNA viruses and chymotrypsin-like serine proteases. A distinct protein superfamily with a common structural fold. FEBS Lett. 1989, 243, 103–114. [Google Scholar] [CrossRef]
- Gorbalenya, A.E.; Blinov, V.M.; Donchenko, A.P. Poliovirus-encoded proteinase 3C: A possible evolutionary link between cellular serine and cysteine proteinase families. FEBS Lett. 1986, 194, 253–257. [Google Scholar] [CrossRef]
- Bazan, J.F.; Fletterick, R.J. Viral cysteine proteases are homologous to the trypsin-like family of serine proteases: Structural and functional implications. Proc. Natl. Acad. Sci. USA 1988, 85, 7872–7876. [Google Scholar] [CrossRef]
- Allaire, M.; Chernaia, M.M.; Malcolm, B.A.; James, M.N. Picornaviral 3C cysteine proteinases have a fold similar to chymotrypsin-like serine proteinases. Nature 1994, 369, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, E.M.; Mosimann, S.C.; Chernaia, M.M.; Malcolm, B.A.; James, M.N. The refined crystal structure of the 3C gene product from hepatitis A virus: Specific proteinase activity and RNA recognition. J. Virol. 1997, 71, 2436–2448. [Google Scholar] [PubMed]
- Mosimann, S.C.; Cherney, M.M.; Sia, S.; Plotch, S.; James, M.N. Refined X-ray crystallographic structure of the poliovirus 3C gene product. J. Mol. Biol. 1997, 273, 1032–1047. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.A.; Smith, W.W.; Ferre, R.A.; Condon, B.; Budahazi, G.; Sisson, W.; Villafranca, J.E.; Janson, C.A.; McElroy, H.E.; Gribskov, C.L.; et al. Structure of human rhinovirus 3C protease reveals a trypsin-like polypeptide fold, RNA-binding site, and means for cleaving precursor polyprotein. Cell 1994, 77, 761–771. [Google Scholar] [CrossRef]
- Valli, A.; Lopez-Moya, J.J.; Garcia, J.A. Recombination and gene duplication in the evolutionary diversification of P1 proteins in the family Potyviridae. J. Gen. Virol. 2007, 88, 1016–1028. [Google Scholar] [CrossRef]
- Adams, M.J.; Antoniw, J.F.; Beaudoin, F. Overview and analysis of the polyprotein cleavage sites in the family Potyviridae. Mol. Plant Pathol. 2005, 6, 471–487. [Google Scholar] [CrossRef]
- Ypma-Wong, M.F.; Filman, D.J.; Hogle, J.M.; Semler, B.L. Structural domains of the poliovirus polyprotein are major determinants for proteolytic cleavage at Gln-Gly pairs. J. Biol. Chem. 1988, 263, 17846–17856. [Google Scholar] [PubMed]
- Clark, A.J.; Bertens, P.; Wellink, J.; Shanks, M.; Lomonossoff, G.P. Studies on hybrid comoviruses reveal the importance of three-dimensional structure for processing of the viral coat proteins and show that the specificity of cleavage is greater in trans than in cis. Virology 1999, 263, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, M.F.; Baltimore, D. Polypeptide cleavages in the formation of poliovirus proteins. Proc. Natl. Acad. Sci. USA 1968, 61, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Gorbalenya, A.E.; Svitkin, Y.V.; Kazachkov, Y.A.; Agol, V.I. Encephalomyocarditis virus-specific polypeptide p22 is involved in the processing of the viral precursor polypeptides. FEBS Lett. 1979, 108, 1–5. [Google Scholar] [CrossRef]
- Shih, D.S.; Shih, C.T.; Zimmern, D.; Rueckert, R.R.; Kaesberg, P. Translation of encephalomyocarditis virus RNA in reticulocyte lysates: Kinetic analysis of the formation of virion proteins and a protein required for processing. J. Virol. 1979, 30, 472–480. [Google Scholar]
- Palmenberg, A.C.; Pallansch, M.A.; Rueckert, R.R. Protease required for processing picornaviral coat protein resides in the viral replicase gene. J. Virol. 1979, 32, 770–778. [Google Scholar]
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV Virus Taxonomy Profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef]
- Daros, J.A.; Carrington, J.C. RNA binding activity of NIa proteinase of tobacco etch potyvirus. Virology 1997, 237, 327–336. [Google Scholar] [CrossRef]
- Sanfacon, H.; Wellink, J.; Le Gall, O.; Karasev, A.; van der Vlugt, R.; Wetzel, T. Secoviridae: A proposed family of plant viruses within the order Picornavirales that combines the families Sequiviridae and Comoviridae, the unassigned genera Cheravirus and Sadwavirus, and the proposed genus Torradovirus. Arch. Virol. 2009, 154, 899–907. [Google Scholar] [CrossRef]
- Isogai, M.; Tatuto, N.; Ujiie, C.; Watanabe, M.; Yoshikawa, N. Identification and characterization of blueberry latent spherical virus, a new member of subgroup C in the genus Nepovirus. Arch. Virol. 2012, 157, 297–303. [Google Scholar] [CrossRef]
- Verver, J.; Goldbach, R.; Garcia, J.A.; Vos, P. In vitro expression of a full-length DNA copy of cowpea mosaic virus B RNA: Identification of the B RNA encoded 24-kd protein as a viral protease. EMBO J. 1987, 6, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hans, F.; Sanfacon, H. Tomato ringspot nepovirus protease: Characterization and cleavage site specificity. J. Gen. Virol. 1995, 76, 917–927. [Google Scholar] [CrossRef] [PubMed]
- Margis, R.; Viry, M.; Pinck, M.; Pinck, L. Cloning and in vitro characterization of the grapevine fanleaf virus proteinase cistron. Virology 1991, 185, 779–787. [Google Scholar] [CrossRef]
- Fuchs, M.; Schmitt-Keichinger, C.; Sanfacon, H. A Renaissance in Nepovirus Research Provides New Insights Into Their Molecular Interface With Hosts and Vectors. Adv. Virus Res. 2017, 97, 61–105. [Google Scholar] [PubMed]
- Pouwels, J.; Carette, J.E.; Van Lent, J.; Wellink, J. Cowpea mosaic virus: Effects on host cell processes. Mol. Plant Pathol. 2002, 3, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.A.; Voorhorst, W.G.; Wery, J.; Wellink, J.; van Kammen, A. A regulatory role for the 32K protein in proteolytic processing of cowpea mosaic virus polyproteins. Virology 1992, 191, 81–89. [Google Scholar] [CrossRef]
- Wetzel, T.; Chisholm, J.; Bassler, A.; Sanfacon, H. Characterization of proteinase cleavage sites in the N-terminal region of the RNA1-encoded polyprotein from Arabis mosaic virus (subgroup A nepovirus). Virology 2008, 375, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Sanfacon, H. Proteolytic processing at a novel cleavage site in the N-terminal region of the tomato ringspot nepovirus RNA-1-encoded polyprotein in vitro. J. Gen. Virol. 2000, 81, 2771–2781. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, O.; Greif, C.; Dufourcq, P.; Reinbolt, J.; Fritsch, C. Functional characterization of the proteolytic activity of the tomato black ring nepovirus RNA-1-encoded polyprotein. Virology 1995, 206, 362–371. [Google Scholar] [CrossRef]
- Margis, R.; Viry, M.; Pinck, M.; Bardonnet, N.; Pinck, L. Differential proteolytic activities of precursor and mature forms of the 24K proteinase of grapevine fanleaf nepovirus. Virology 1994, 200, 79–86. [Google Scholar] [CrossRef]
- Chisholm, J.; Wieczorek, A.; Sanfacon, H. Expression and partial purification of recombinant tomato ringspot nepovirus 3C-like proteinase: Comparison of the activity of the mature proteinase and the VPg-proteinase precursor. Virus Res. 2001, 79, 153–164. [Google Scholar] [CrossRef]
- Wellink, J.; Rezelman, G.; Goldbach, R.; Beyreuther, K. Determination of the proteolytic processing sites in the polyprotein encoded by the bottom-component RNA of Cowpea mosaic virus. J. Virol. 1986, 59, 50–58. [Google Scholar] [PubMed]
- Carrier, K.; Hans, F.; Sanfacon, H. Mutagenesis of amino acids at two tomato ringspot nepovirus cleavage sites: Effect on proteolytic processing in cis and in trans by the 3C-like protease. Virology 1999, 258, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Phan, J.; Zdanov, A.; Evdokimov, A.G.; Tropea, J.E.; Peters, H.K., 3rd; Kapust, R.B.; Li, M.; Wlodawer, A.; Waugh, D.S. Structural basis for the substrate specificity of tobacco etch virus protease. J. Biol. Chem. 2002, 277, 50564–50572. [Google Scholar] [CrossRef]
- Dougherty, W.G.; Carrington, J.C.; Cary, S.M.; Parks, T.D. Biochemical and mutational analysis of a plant virus polyprotein cleavage site. EMBO J. 1988, 7, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Kapust, R.B.; Tozser, J.; Copeland, T.D.; Waugh, D.S. The P1’ specificity of tobacco etch virus protease. Biochem. Biophys. Res. Commun. 2002, 294, 949–955. [Google Scholar] [CrossRef]
- Nautiyal, K.; Kuroda, Y. A SEP tag enhances the expression, solubility and yield of recombinant TEV protease without altering its activity. New Biotechnol. 2018, 42, 77–84. [Google Scholar] [CrossRef]
- Raran-Kurussi, S.; Cherry, S.; Zhang, D.; Waugh, D.S. Removal of Affinity Tags with TEV Protease. Meth. Mol. Biol. 2017, 1586, 221–230. [Google Scholar]
- Taxis, C.; Knop, M. TIPI: TEV protease-mediated induction of protein instability. Meth. Mol. Biol. 2012, 832, 611–626. [Google Scholar]
- Rubio, V.; Shen, Y.; Saijo, Y.; Liu, Y.; Gusmaroli, G.; Dinesh-Kumar, S.P.; Deng, X.W. An alternative tandem affinity purification strategy applied to Arabidopsis protein complex isolation. Plant J. 2005, 41, 767–778. [Google Scholar] [CrossRef]
- Kapust, R.B.; Waugh, D.S. Controlled intracellular processing of fusion proteins by TEV protease. Protein Expr. Purif. 2000, 19, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Ceriani, M.F.; Marcos, J.F.; Hopp, H.E.; Beachy, R.N. Simultaneous accumulation of multiple viral coat proteins from a TEV-NIa based expression vector. Plant Mol. Biol. 1998, 36, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Marcos, J.F.; Beachy, R.N. In vitro characterization of a cassette to accumulate multiple proteins through synthesis of a self-processing polypeptide. Plant Mol. Biol. 1994, 24, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Puckette, M.; Smith, J.D.; Gabbert, L.; Schutta, C.; Barrera, J.; Clark, B.A.; Neilan, J.G.; Rasmussen, M. Production of foot-and-mouth disease virus capsid proteins by the TEV protease. J. Biotechnol. 2018, 275, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.A.; Riechmann, J.L.; Martin, M.T.; Lain, S. Proteolytic activity of the plum pox potyvirus NIa-protein on excess of natural and artificial substrates in Escherichia coli. FEBS Lett. 1989, 257, 269–273. [Google Scholar] [CrossRef]
- Garcia, J.A.; Lain, S.; Cervera, M.T.; Riechmann, J.L.; Martin, M.T. Mutational analysis of plum pox potyvirus polyprotein processing by the NIa protease in Escherichia coli. J. Gen. Virol. 1990, 71 (Pt 12), 2773–2779. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.T.; Lopez Otin, C.; Lain, S.; Garcia, J.A. Determination of polyprotein processing sites by amino terminal sequencing of nonstructural proteins encoded by plum pox potyvirus. Virus Res. 1990, 15, 97–106. [Google Scholar] [CrossRef]
- Kang, H.; Lee, Y.J.; Goo, J.H.; Park, W.J. Determination of the substrate specificity of turnip mosaic virus NIa protease using a genetic method. J. Gen. Virol. 2001, 82, 3115–3117. [Google Scholar] [CrossRef]
- Tozser, J.; Tropea, J.E.; Cherry, S.; Bagossi, P.; Copeland, T.D.; Wlodawer, A.; Waugh, D.S. Comparison of the substrate specificity of two potyvirus proteases. FEBS J. 2005, 272, 514–523. [Google Scholar] [CrossRef]
- Sun, P.; Austin, B.P.; Tozser, J.; Waugh, D.S. Structural determinants of tobacco vein mottling virus protease substrate specificity. Protein Soc. 2010, 19, 2240–2251. [Google Scholar] [CrossRef]
- Garcia, J.A.; Lain, S. Proteolytic activity of plum pox virus-tobacco etch virus chimeric NIa proteases. FEBS Lett. 1991, 281, 67–72. [Google Scholar] [CrossRef]
- Mathur, C.; Jimsheena, V.K.; Banerjee, S.; Makinen, K.; Gowda, L.R.; Savithri, H.S. Functional regulation of PVBV Nuclear Inclusion protein-a protease activity upon interaction with Viral Protein genome-linked and phosphorylation. Virology 2012, 422, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Sabharwal, P.; Srinivas, S.; Savithri, H.S. Mapping the domain of interaction of PVBV VPg with NIa-Pro: Role of N-terminal disordered region of VPg in the modulation of structure and function. Virology 2018, 524, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Schaad, M.C.; Haldeman-Cahill, R.; Cronin, S.; Carrington, J.C. Analysis of the VPg-proteinase (NIa) encoded by tobacco etch potyvirus: Effects of mutations on subcellular transport, proteolytic processing, and genome amplification. J. Virol. 1996, 70, 7039–7048. [Google Scholar] [PubMed]
- Rantalainen, K.I.; Uversky, V.N.; Permi, P.; Kalkkinen, N.; Dunker, A.K.; Makinen, K. Potato virus A genome-linked protein VPg is an intrinsically disordered molten globule-like protein with a hydrophobic core. Virology 2008, 377, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.; Rodrigo, G.; Aragones, V.; Ruiz, M.; Lodewijk, I.; Fernandez, U.; Elena, S.F.; Daros, J.A. Interaction network of tobacco etch potyvirus NIa protein with the host proteome during infection. BMC Genom. 2016, 17, 87. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Laliberte, J.F. The genome-linked protein VPg of plant viruses-a protein with many partners. Curr. Opin. Virol. 2011, 1, 347–354. [Google Scholar] [CrossRef]
- Somera, M.; Sarmiento, C.; Truve, E. Overview on Sobemoviruses and a Proposal for the Creation of the Family Sobemoviridae. Viruses 2015, 7, 3076–3115. [Google Scholar] [CrossRef]
- Li, X.; Ryan, M.D.; Lamb, J.W. Potato leafroll virus protein P1 contains a serine proteinase domain. J. Gen. Virol. 2000, 81, 1857–1864. [Google Scholar] [CrossRef]
- Sadowy, E.; Juszczuk, M.; David, C.; Gronenborn, B.; Hulanicka, M.D. Mutational analysis of the proteinase function of Potato leafroll virus. J. Gen. Virol. 2001, 82, 1517–1527. [Google Scholar] [CrossRef]
- Nair, S.; Savithri, H.S. Processing of SeMV polyproteins revisited. Virology 2010, 396, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Halpin, C.; Ryan, M.D. A novel cleavage site within the potato leafroll virus P1 polyprotein. J. Gen. Virol. 2007, 88, 1620–1623. [Google Scholar] [CrossRef] [PubMed]
- Gayathri, P.; Satheshkumar, P.S.; Prasad, K.; Nair, S.; Savithri, H.S.; Murthy, M.R. Crystal structure of the serine protease domain of Sesbania mosaic virus polyprotein and mutational analysis of residues forming the S1-binding pocket. Virology 2006, 346, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Satheshkumar, P.S.; Gayathri, P.; Prasad, K.; Savithri, H.S. “Natively unfolded” VPg is essential for Sesbania mosaic virus serine protease activity. J. Biol. Chem. 2005, 280, 30291–30300. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Gayathri, P.; Murthy, M.R.; Savithri, H.S. Stacking interactions of W271 and H275 of SeMV serine protease with W43 of natively unfolded VPg confer catalytic activity to protease. Virology 2008, 382, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Verchot, J.; Koonin, E.V.; Carrington, J.C. The 35-kDa protein from the N-terminus of the potyviral polyprotein functions as a third virus-encoded proteinase. Virology 1991, 185, 527–535. [Google Scholar] [CrossRef]
- Verchot, J.; Herndon, K.L.; Carrington, J.C. Mutational analysis of the tobacco etch potyviral 35-kDa proteinase: Identification of essential residues and requirements for autoproteolysis. Virology 1992, 190, 298–306. [Google Scholar] [CrossRef]
- Valli, A.; Martin-Hernandez, A.M.; Lopez-Moya, J.J.; Garcia, J.A. RNA silencing suppression by a second copy of the P1 serine protease of Cucumber vein yellowing ipomovirus, a member of the family Potyviridae that lacks the cysteine protease HCPro. J. Virol. 2006, 80, 10055–10063. [Google Scholar] [CrossRef]
- Rodamilans, B.; Valli, A.; Garcia, J.A. Mechanistic divergence between P1 proteases of the family Potyviridae. J. Gen. Virol. 2013, 94, 1407–1414. [Google Scholar] [CrossRef]
- Maliogka, V.I.; Salvador, B.; Carbonell, A.; Saenz, P.; Leon, D.S.; Oliveros, J.C.; Delgadillo, M.O.; Garcia, J.A.; Simon-Mateo, C. Virus variants with differences in the P1 protein coexist in a Plum pox virus population and display particular host-dependent pathogenicity features. Mol. Plant Pathol. 2012, 13, 877–886. [Google Scholar] [CrossRef]
- Pasin, F.; Simon-Mateo, C.; Garcia, J.A. The hypervariable amino-terminus of P1 protease modulates potyviral replication and host defense responses. PLoS Pathog. 2014, 10, e1003985. [Google Scholar] [CrossRef] [PubMed]
- Shan, H.; Pasin, F.; Valli, A.; Castillo, C.; Rajulu, C.; Carbonell, A.; Simon-Mateo, C.; Garcia, J.A.; Rodamilans, B. The Potyviridae P1a leader protease contributes to host range specificity. Virology 2015, 476, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Shan, H.; Pasin, F.; Tzanetakis, I.E.; Simon-Mateo, C.; Garcia, J.A.; Rodamilans, B. Truncation of a P1 leader proteinase facilitates potyvirus replication in a non-permissive host. Mol. Plant Pathol. 2018, 19, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.A.; Schrijvers, L.; Tan, A.; Vos, P.; Wellink, J.; Goldbach, R. Proteolytic activity of the cowpea mosaic virus encoded 24K protein synthesized in Escherichia coli. Virology 1987, 159, 67–75. [Google Scholar] [CrossRef]
- Drenth, J.; Jansonius, J.N.; Koekoek, R.; Swen, H.M.; Wolthers, B.G. Structure of papain. Nature 1968, 218, 929–932. [Google Scholar] [CrossRef]
- Vernet, T.; Tessier, D.C.; Chatellier, J.; Plouffe, C.; Lee, T.S.; Thomas, D.Y.; Storer, A.C.; Menard, R. Structural and functional roles of asparagine 175 in the cysteine protease papain. J. Biol. Chem. 1995, 270, 16645–16652. [Google Scholar] [CrossRef]
- Novinec, M.; Lenarcic, B. Papain-like peptidases: Structure, function, and evolution. Biomol. Concepts 2013, 4, 287–308. [Google Scholar] [CrossRef]
- Gorbalenya, A.E.; Koonin, E.V.; Lai, M.M. Putative papain-related thiol proteases of positive-strand RNA viruses. Identification of rubi- and aphthovirus proteases and delineation of a novel conserved domain associated with proteases of rubi-, alpha- and coronaviruses. FEBS Lett. 1991, 288, 201–205. [Google Scholar] [CrossRef]
- Guarne, A.; Tormo, J.; Kirchweger, R.; Pfistermueller, D.; Fita, I.; Skern, T. Structure of the foot-and-mouth disease virus leader protease: A papain-like fold adapted for self-processing and eIF4G recognition. EMBO J. 1998, 17, 7469–7479. [Google Scholar] [CrossRef]
- Russo, A.T.; White, M.A.; Watowich, S.J. The crystal structure of the Venezuelan equine encephalitis alphavirus nsP2 protease. Structure 2006, 14, 1449–1458. [Google Scholar] [CrossRef]
- Carrington, J.C.; Cary, S.M.; Parks, T.D.; Dougherty, W.G. A second proteinase encoded by a plant potyvirus genome. EMBO J. 1989, 8, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Carrington, J.C.; Freed, D.D.; Sanders, T.C. Autocatalytic processing of the potyvirus helper component proteinase in Escherichia coli and in vitro. J. Virol. 1989, 63, 4459–4463. [Google Scholar] [PubMed]
- Plisson, C.; Drucker, M.; Blanc, S.; German-Retana, S.; Le Gall, O.; Thomas, D.; Bron, P. Structural characterization of HC-Pro, a plant virus multifunctional protein. J. Biol. Chem. 2003, 278, 23753–23761. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Lin, J.; Ye, K. Structure of the autocatalytic cysteine protease domain of potyvirus helper-component proteinase. J. Biol. Chem. 2011, 286, 21937–21943. [Google Scholar] [CrossRef] [PubMed]
- Hasiow-Jaroszewska, B.; Fares, M.A.; Elena, S.F. Molecular evolution of viral multifunctional proteins: The case of potyvirus HC-Pro. J. Mol. Evol. 2014, 78, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Agranovsky, A. Closteroviruses: Molecular biology, evolution and interactions with cells. In Plant Viruses: Evolution and Management; Springer: Singapore, 2016; pp. 231–252. [Google Scholar]
- Agranovsky, A.A.; Koonin, E.V.; Boyko, V.P.; Maiss, E.; Frotschl, R.; Lunina, N.A.; Atabekov, J.G. Beet yellows closterovirus: Complete genome structure and identification of a leader papain-like thiol protease. Virology 1994, 198, 311–324. [Google Scholar] [CrossRef]
- Zinovkin, R.A.; Erokhina, T.N.; Lesemann, D.E.; Jelkmann, W.; Agranovsky, A.A. Processing and subcellular localization of the leader papain-like proteinase of Beet yellows closterovirus. J. Gen. Virol. 2003, 84, 2265–2270. [Google Scholar] [CrossRef]
- Peng, C.W.; Napuli, A.J.; Dolja, V.V. Leader proteinase of beet yellows virus functions in long-distance transport. J. Virol. 2003, 77, 2843–2849. [Google Scholar] [CrossRef]
- Peremyslov, V.V.; Hagiwara, Y.; Dolja, V.V. Genes required for replication of the 15.5-kilobase RNA genome of a plant closterovirus. J. Virol. 1998, 72, 5870–5876. [Google Scholar]
- Liu, Y.P.; Peremyslov, V.V.; Medina, V.; Dolja, V.V. Tandem leader proteases of Grapevine leafroll-associated virus-2: Host-specific functions in the infection cycle. Virology 2009, 383, 291–299. [Google Scholar] [CrossRef]
- Peng, C.W.; Peremyslov, V.V.; Mushegian, A.R.; Dawson, W.O.; Dolja, V.V. Functional specialization and evolution of leader proteinases in the family Closteroviridae. J. Virol. 2001, 75, 12153–12160. [Google Scholar] [CrossRef]
- Kang, S.H.; Atallah, O.O.; Sun, Y.D.; Folimonova, S.Y. Functional diversification upon leader protease domain duplication in the Citrus tristeza virus genome: Role of RNA sequences and the encoded proteins. Virology 2018, 514, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Makarova, K.S.; Aravind, L.; Koonin, E.V. A novel superfamily of predicted cysteine proteases from eukaryotes, viruses and Chlamydia pneumoniae. Trends Biochem. Sci. 2000, 25, 50–52. [Google Scholar] [CrossRef]
- Bailey-Elkin, B.A.; van Kasteren, P.B.; Snijder, E.J.; Kikkert, M.; Mark, B.L. Viral OTU deubiquitinases: A structural and functional comparison. PLoS Pathog. 2014, 10, e1003894. [Google Scholar] [CrossRef] [PubMed]
- Bransom, K.L.; Dreher, T.W. Identification of the essential cysteine and histidine residues of the turnip yellow mosaic virus protease. Virology 1994, 198, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Rozanov, M.N.; Drugeon, G.; Haenni, A.L. Papain-like proteinase of turnip yellow mosaic virus: A prototype of a new viral proteinase group. Arch. Virol. 1995, 140, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, C.; Ayach, M.; Beaurepaire, L.; Chenon, M.; Andreani, J.; Guerois, R.; Jupin, I.; Bressanelli, S. A compact viral processing proteinase/ubiquitin hydrolase from the OTU family. PLoS Pathog. 2013, 9, e1003560. [Google Scholar] [CrossRef]
- Jakubiec, A.; Drugeon, G.; Camborde, L.; Jupin, I. Proteolytic processing of Turnip yellow mosaic virus replication proteins and functional impact on infectivity. J. Virol. 2007, 81, 11402–11412. [Google Scholar] [CrossRef]
- Chenon, M.; Camborde, L.; Cheminant, S.; Jupin, I. A viral deubiquitylating enzyme targets viral RNA-dependent RNA polymerase and affects viral infectivity. EMBO J. 2012, 31, 741–753. [Google Scholar] [CrossRef]
- Van Kasteren, P.B.; Bailey-Elkin, B.A.; James, T.W.; Ninaber, D.K.; Beugeling, C.; Khajehpour, M.; Snijder, E.J.; Mark, B.L.; Kikkert, M. Deubiquitinase function of arterivirus papain-like protease 2 suppresses the innate immune response in infected host cells. Proc. Natl. Acad. Sci. USA 2013, 110, E838–E847. [Google Scholar] [CrossRef]
- Akutsu, M.; Ye, Y.; Virdee, S.; Chin, J.W.; Komander, D. Molecular basis for ubiquitin and ISG15 cross-reactivity in viral ovarian tumor domains. Proc. Natl. Acad. Sci. USA 2011, 108, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Capodagli, G.C.; McKercher, M.A.; Baker, E.A.; Masters, E.M.; Brunzelle, J.S.; Pegan, S.D. Structural analysis of a viral ovarian tumor domain protease from the Crimean-Congo hemorrhagic fever virus in complex with covalently bonded ubiquitin. J. Virol. 2011, 85, 3621–3630. [Google Scholar] [CrossRef] [PubMed]
- James, T.W.; Frias-Staheli, N.; Bacik, J.P.; Levingston Macleod, J.M.; Khajehpour, M.; Garcia-Sastre, A.; Mark, B.L. Structural basis for the removal of ubiquitin and interferon-stimulated gene 15 by a viral ovarian tumor domain-containing protease. Proc. Natl. Acad. Sci. USA 2011, 108, 2222–2227. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, D.M.; Rozanov, M.N.; Hillman, B.I. Autocatalytic processing of the 223-kDa protein of blueberry scorch carlavirus by a papain-like proteinase. Virology 1995, 207, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Mann, K.S.; Walker, M.; Sanfaçon, H. Identification of cleavage sites recognized by the 3C-like cysteine protease within the two polyproteins of strawberry mottle virus. Front. Microbiol. 2017, 8, 745. [Google Scholar] [CrossRef] [PubMed]
- Chandra, N.R.; Prabu, M.M.; Suguna, K.; Vijayan, M. Structural similarity and functional diversity in proteins containing the legume lectin fold. Protein Eng. 2001, 14, 857–866. [Google Scholar] [CrossRef]
- Fujinaga, M.; Cherney, M.M.; Oyama, H.; Oda, K.; James, M.N. The molecular structure and catalytic mechanism of a novel carboxyl peptidase from Scytalidium lignicolum. Proc. Natl. Acad. Sci. USA 2004, 101, 3364–3369. [Google Scholar] [CrossRef]
- Kataoka, Y.; Takada, K.; Oyama, H.; Tsunemi, M.; James, M.N.; Oda, K. Catalytic residues and substrate specificity of scytalidoglutamic peptidase, the first member of the eqolisin in family (G1) of peptidases. FEBS Lett. 2005, 579, 2991–2994. [Google Scholar] [CrossRef]
- Kondo, M.Y.; Okamoto, D.N.; Santos, J.A.; Juliano, M.A.; Oda, K.; Pillai, B.; James, M.N.; Juliano, L.; Gouvea, I.E. Studies on the catalytic mechanism of a glutamic peptidase. J. Biol. Chem. 2010, 285, 21437–21445. [Google Scholar] [CrossRef]
- Pillai, B.; Cherney, M.M.; Hiraga, K.; Takada, K.; Oda, K.; James, M.N. Crystal structure of scytalidoglutamic peptidase with its first potent inhibitor provides insights into substrate specificity and catalysis. J. Mol. Biol. 2007, 365, 343–361. [Google Scholar] [CrossRef]
- Sasaki, H.; Kubota, K.; Lee, W.C.; Ohtsuka, J.; Kojima, M.; Iwata, S.; Nakagawa, A.; Takahashi, K.; Tanokura, M. The crystal structure of an intermediate dimer of aspergilloglutamic peptidase that mimics the enzyme-activation product complex produced upon autoproteolysis. J. Biochem. 2012, 152, 45–52. [Google Scholar] [CrossRef]
- Wintermantel, W.M. Transmission efficiency and epidemiology of criniviruses. In Bemisia: Bionomics and Management of a Global Pest; Springer: Dordrecht, The Netherlands; Heildelberg, Germany; London, UK; New York, NY, USA, 2009; pp. 319–331. [Google Scholar]
- Kiss, Z.A.; Medina, V.; Falk, B. Crinivirus replication and host interactions. Front. Microbiol. 2013, 4, 99. [Google Scholar] [CrossRef] [PubMed]
- Agranovsky, A.A.; Lesemann, D.E.; Maiss, E.; Hull, R.; Atabekov, J.G. “Rattlesnake” structure of a filamentous plant RNA virus built of two capsid proteins. Proc. Natl. Acad. Sci. USA 1995, 92, 2470–2473. [Google Scholar] [CrossRef]
- Killiny, N.; Harper, S.; Alfaress, S.; El Mohtar, C.; Dawson, W. Minor coat and heat-shock proteins are involved in binding of citrus tristeza virus to the foregut of its aphid vector, Toxoptera citricida. Appl. Environ. Microbiol. 2016. [Google Scholar] [CrossRef]
- Stewart, L.R.; Medina, V.; Tian, T.; Turina, M.; Falk, B.W.; Ng, J.C. A mutation in the Lettuce infectious yellows virus minor coat protein disrupts whitefly transmission but not in planta systemic movement. J. Virol. 2010, 84, 12165–12173. [Google Scholar] [CrossRef]
- Krupovic, M.; Blomberg, J.; Coffin, J.M.; Dasgupta, I.; Fan, H.; Geering, A.D.; Gifford, R.; Harrach, B.; Hull, R.; Johnson, W.; et al. Ortervirales: New Virus Order Unifying Five Families of Reverse-Transcribing Viruses. J. Virol. 2018, 92, e00515-18. [Google Scholar] [CrossRef] [PubMed]
- Hayward, A. Origin of the retroviruses: When, where, and how? Curr. Opin. Virol. 2017, 25, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Dunn, B.M.; Goodenow, M.M.; Gustchina, A.; Wlodawer, A. Retroviral proteases. Genome Biol. 2002, 3, 3006.1–3006.7. [Google Scholar] [CrossRef]
- Wlodawer, A.; Miller, M.; Jaskolski, M.; Sathyanarayana, B.K.; Baldwin, E.; Weber, I.T.; Selk, L.M.; Clawson, L.; Schneider, J.; Kent, S. Conserved folding in retroviral proteases: Crystal structure of a synthetic HIV-1 protease. Science 1989, 245, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Navia, M.A.; Fitzgerald, P.M.; McKeever, B.M.; Leu, C.T.; Heimbach, J.C.; Herber, W.K.; Sigal, I.S.; Darke, P.L.; Springer, J.P. Three-dimensional structure of aspartyl protease from human immunodeficiency virus HIV-1. Nature 1989, 337, 615–620. [Google Scholar] [CrossRef]
- Torruella, M.; Gordon, K.; Hohn, T. Cauliflower mosaic virus produces an aspartic proteinase to cleave its polyproteins. EMBO J. 1989, 8, 2819–2825. [Google Scholar] [CrossRef] [PubMed]
- Champagne, J.; Benhamou, N.; Leclerc, D. Localization of the N-terminal domain of cauliflower mosaic virus coat protein precursor. Virology 2004, 324, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Karsies, A.; Merkle, T.; Szurek, B.; Bonas, U.; Hohn, T.; Leclerc, D. Regulated nuclear targeting of cauliflower mosaic virus. J. Gen. Virol. 2002, 83, 1783–1790. [Google Scholar] [CrossRef]
- Luna, G.R.; Peña, E.J.; Borniego, M.B.; Heinlein, M.; García, M.L. Citrus psorosis virus movement protein contains an aspartic protease required for autocleavage and the formation of tubule-like structures at plasmodesmata. J. Virol. 2018, 92, e00355-18. [Google Scholar] [CrossRef] [PubMed]
- Kondo, H.; Hirano, S.; Chiba, S.; Andika, I.B.; Hirai, M.; Maeda, T.; Tamada, T. Characterization of burdock mottle virus, a novel member of the genus Benyvirus, and the identification of benyvirus-related sequences in the plant and insect genomes. Virus Res. 2013, 177, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Pascon, R.C.; Kitajima, J.P.; Breton, M.C.; Assumpcao, L.; Greggio, C.; Zanca, A.S.; Okura, V.K.; Alegria, M.C.; Camargo, M.E.; Silva, G.G.; et al. The complete nucleotide sequence and genomic organization of Citrus Leprosis associated Virus, Cytoplasmatic type (CiLV-C). Virus Genes 2006, 32, 289–298. [Google Scholar] [CrossRef] [PubMed]
© 2019 by Her Majesty the Queen in Right of Canada as Represented by the Minister of Agriculture and Agri-Food Canada. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mann, K.S.; Sanfaçon, H. Expanding Repertoire of Plant Positive-Strand RNA Virus Proteases. Viruses 2019, 11, 66. https://doi.org/10.3390/v11010066
Mann KS, Sanfaçon H. Expanding Repertoire of Plant Positive-Strand RNA Virus Proteases. Viruses. 2019; 11(1):66. https://doi.org/10.3390/v11010066
Chicago/Turabian StyleMann, Krin S., and Hélène Sanfaçon. 2019. "Expanding Repertoire of Plant Positive-Strand RNA Virus Proteases" Viruses 11, no. 1: 66. https://doi.org/10.3390/v11010066
APA StyleMann, K. S., & Sanfaçon, H. (2019). Expanding Repertoire of Plant Positive-Strand RNA Virus Proteases. Viruses, 11(1), 66. https://doi.org/10.3390/v11010066





