Resistance Analysis of a 3-Day Monotherapy Study with Glecaprevir or Pibrentasvir in Patients with Chronic Hepatitis C Virus Genotype 1 Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Compounds
2.2. Study Design
2.3. Viral Sequence Analysis
2.4. Effects of Amino Acid Substitutions on the Antiviral Activity of Different DAAs in HCV Replicon Cells
3. Results
3.1. Patients with Available HCV Sequencing Data
3.2. Prevalence of Baseline Polymorphisms
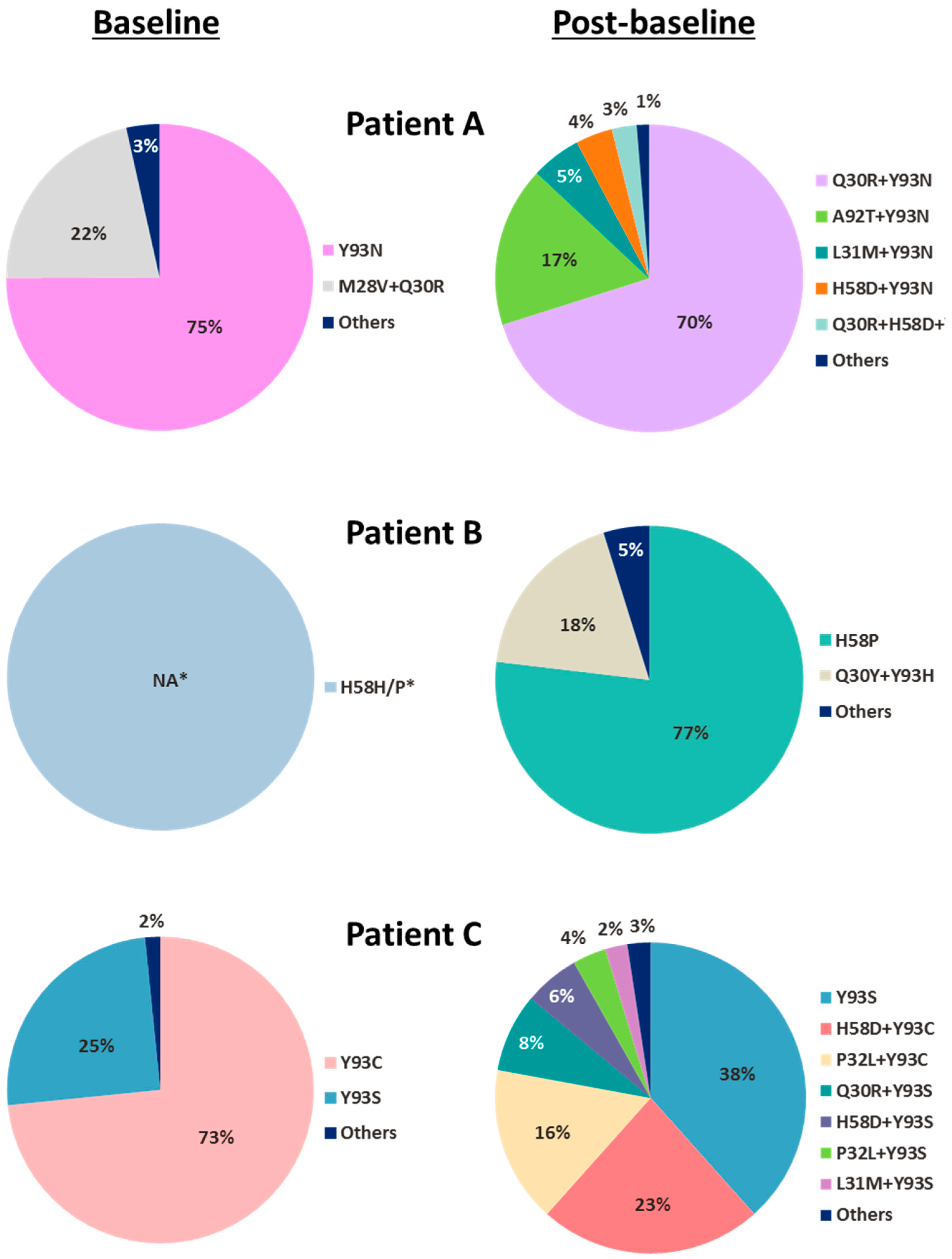
3.3. Treatment-Emergent Substitutions
3.4. Susceptibility of Baseline or Treatment-Emergent NS3 Substitutions to Glecaprevir
3.5. Susceptibility of Baseline or Treatment-Emergent NS5A Substitutions to Pibrentasvir
3.6. Susceptibility of NS5A Amino Acid Substitutions to Pibrentasvir and Other Approved NS5A Inhibitors
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Hepatitis Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Messina, J.P.; Humphreys, I.; Flaxman, A.; Brown, A.; Cooke, G.S.; Pybus, O.G.; Barnes, E. Global distribution and prevalence of hepatitis C virus genotypes. Hepatology 2015, 61, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.B.; Bukh, J.; Kuiken, C.; Muerhoff, A.S.; Rice, C.M.; Stapleton, J.T.; Simmonds, P. Expanded classification of hepatitis C virus into 7 genotypes and 67 subtypes: Updated criteria and genotype assignment web resource. Hepatology 2014, 59, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, F.; Kramer, J.; Asch, S.M.; Chayanupatkul, M.; Cao, Y.; El-Serag, H.B. Risk of hepatocellular cancer in HCV patients treated with direct-acting antiviral agents. Gastroenterology 2017, 153, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Van der Meer, A.J.; Veldt, B.J.; Feld, J.J.; Wedemeyer, H.; Dufour, J.F.; Lammert, F.; Duarte-Rojo, A.; Heathcote, E.J.; Manns, M.P.; Kuske, L.; et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. JAMA 2012, 308, 2584–2593. [Google Scholar] [CrossRef] [PubMed]
- Kwong, A.D. The HCV revolution did not happen overnight. ACS Med. Chem. Lett. 2014, 5, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Asselah, T.; Boyer, N.; Saadoun, D.; Martinot-Peignoux, M.; Marcellin, P. Direct-acting antivirals for the treatment of hepatitis C virus infection: Optimizing current IFN-free treatment and future perspectives. Liver Int. 2016, 36 (Suppl. S1), 47–57. [Google Scholar] [CrossRef] [PubMed]
- Falade-Nwulia, O.; Suarez-Cuervo, C.; Nelson, D.R.; Fried, M.W.; Segal, J.B.; Sulkowski, M.S. Oral direct-acting agent therapy for hepatitis C virus infection: A systematic review. Ann. Intern. Med. 2017, 166, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Gottwein, J.M.; Scheel, T.K.; Jensen, T.B.; Ghanem, L.; Bukh, J. Differential efficacy of protease inhibitors against HCV genotypes 2a; 3a; 5a; and 6a NS3/4A protease recombinant viruses. Gastroenterology 2011, 141, 1067–1079. [Google Scholar] [CrossRef] [PubMed]
- Gottwein, J.M.; Pham, L.V.; Mikkelsen, L.S.; Ghanem, L.; Ramirez, S.; Scheel, T.K.H.; Carlsen, T.H.R.; Bukh, J. Efficacy of NS5A inhibitors against hepatitis C virus genotypes 1–7 and escape variants. Gastroenterology 2018, 154, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- Gao, M. Antiviral activity and resistance of HCV NS5A replication complex inhibitors. Curr. Opin. Virol. 2013, 3, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, I.M.; Asante-Appiah, E.; Wong, P.; Black, T.; Howe, A.; Wahl, J.; Robertson, M.N.; Nguyen, B.-Y.; Shaughnessy, M.; Hwang, P.; et al. Prevalence and impact of baseline NS5A resistance-associated variants (RAVs) on the efficacy of elbasvir/grazoprevir (EBR/GZR) against GT1a infection. Hepatology 2015, 62, 1393A–1394A. [Google Scholar]
- McPhee, F.; Hernandez, D.; Yu, F.; Ueland, J.; Monikowski, A.; Carifa, A.; Falk, P.; Wang, C.; Fridell, R.; Eley, T.; et al. Resistance analysis of hepatitis C virus genotype 1 prior treatment null responders receiving daclatasvir and asunaprevir. Hepatology 2013, 58, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Janssen Products. Olysio (Simeprevir). Highlights of Prescribing Information; Janssen Therapeutics: Titusville, NJ, USA, 2013. [Google Scholar]
- Hernandez, D.; Zhou, N.; Ueland, J.; Monikowski, A.; McPhee, F. Natural prevalence of NS5A polymorphisms in subjects infected with hepatitis C virus genotype 3 and their effects on the antiviral activity of NS5A inhibitors. J. Clin. Virol. 2013, 57, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Lontok, E.; Harrington, P.; Howe, A.; Kieffer, T.; Lennerstrand, J.; Lenz, O.; McPhee, F.; Mo, H.; Parkin, N.; Pilot-Matias, T.; et al. Hepatitis C virus drug resistance-associated substitutions: State of the art summary. Hepatology 2015, 62, 1623–1632. [Google Scholar] [CrossRef] [PubMed]
- Pawlotsky, J.M. Hepatitis C virus resistance to direct-acting antiviral drugs in interferon-free regimens. Gastroenterology 2016, 151, 70–86. [Google Scholar] [CrossRef] [PubMed]
- Romano, K.P.; Ali, A.; Aydin, C.; Soumana, D.; Ozen, A.; Deveau, L.M.; Silver, C.; Cao, H.; Newton, A.; Petropoulos, C.J.; et al. The molecular basis of drug resistance against hepatitis C virus NS3/4A protease inhibitors. PLoS Pathog. 2012, 8, e1002832. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.I.; Krishnan, P.; Pilot-Matias, T.; Kati, W.; Schnell, G.; Beyer, J.; Reisch, T.; Lu, L.; Dekhtyar, T.; Irvin, M.; et al. In Vitro antiviral activity and resistance profile of the next-generation hepatitis C virus NS5A inhibitor pibrentasvir. Antimicrob. Agents Chemother. 2017, 61, e02558-16. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.I.; Tripathi, R.; Reisch, T.; Lu, L.; Middleton, T.; Hopkins, T.A.; Pithawalla, R.; Irvin, M.; Dekhtyar, T.; Krishnan, P.; et al. In Vitro antiviral activity and resistance profile of the next-generation hepatitis C virus NS3/4A protease inhibitor glecaprevir. Antimicrob. Agents Chemother. 2018, 62, e01620-17. [Google Scholar] [CrossRef] [PubMed]
- Lawitz, E.J.; O’Riordan, W.D.; Asatryan, A.; Freilich, B.L.; Box, T.D.; Overcash, J.S.; Lovell, S.; Ng, T.I.; Liu, W.; Campbell, A.; et al. Potent antiviral activities of the direct-acting antivirals ABT-493 and ABT-530 with three-day monotherapy for hepatitis C virus genotype 1 infection. Antimicrob. Agents Chemother. 2016, 60, 1546–1555. [Google Scholar] [CrossRef] [PubMed]
- Pilot-Matias, T.; Tripathi, R.; Cohen, D.; Gaultier, I.; Dekhtyar, T.; Lu, L.; Reisch, T.; Irvin, M.; Hopkins, T.; Pithawalla, R.; et al. In Vitro and In Vivo antiviral activity and resistance profile of the hepatitis C virus NS3/4A protease inhibitor ABT-450. Antimicrob. Agents Chemother. 2015, 59, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, P.; Beyer, J.; Mistry, N.; Koev, G.; Reisch, T.; DeGoey, D.; Kati, W.; Campbell, A.; Williams, L.; Xie, W.; et al. In Vitro and In Vivo antiviral activity and resistance profile of ombitasvir; an inhibitor of hepatitis C virus NS5A. Antimicrob. Agents Chemother. 2015, 59, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Manos, M.M.; Shvachko, V.A.; Murphy, R.C.; Arduino, J.M.; Shire, N.J. Distribution of hepatitis C virus genotypes in a diverse US integrated health care population. J. Med. Virol. 2012, 84, 1744–1750. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.A.; Worth, A.; Martin, R.; Svarovskaia, E.; Brainard, D.M.; Lawitz, E.; Miller, M.D.; Mo, H. Characterization of hepatitis C virus resistance from a multiple-dose clinical trial of the novel NS5A inhibitor GS-5885. Antimicrob. Agents Chemother. 2013, 57, 6333–6340. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Curry, S.; McMonagle, P.; Yeh, W.W.; Ludmerer, S.W.; Jumes, P.A.; Marshall, W.L.; Kong, S.; Ingravallo, P.; Black, S.; et al. Susceptibilities of genotype 1a; 1b; and 3 hepatitis C virus variants to the NS5A inhibitor elbasvir. Antimicrob. Agents Chemother. 2015, 59, 6922–6929. [Google Scholar] [CrossRef] [PubMed]
- Harrington, P.R.; Komatsu, T.E.; Deming, D.J.; Donaldson, E.F.; O’Rear, J.J.; Naeger, L.K. Impact of hepatitis C virus polymorphisms on direct-acting antiviral treatment efficacy: Regulatory analyses and perspectives. Hepatology 2018, 67, 2430–2448. [Google Scholar] [CrossRef] [PubMed]
- Pawlotsky, J.M. NS5A inhibitors in the treatment of hepatitis C. J. Hepatol. 2013, 59, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Zeuzem, S.; Mizokami, M.; Pianko, S.; Mangia, A.; Han, K.H.; Martin, R.; Svarovskaia, E.; Dvory-Sobol, H.; Doehle, B.; Hedskog, C.; et al. NS5A resistance-associated substitutions in patients with genotype 1 hepatitis C virus: Prevalence and effect on treatment outcome. J. Hepatol. 2017, 66, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Lawitz, E.J.; Dvory-Sobol, H.; Doehle, B.P.; Worth, A.S.; McNally, J.; Brainard, D.M.; Link, J.O.; Miller, M.D.; Mo, H. Clinical resistance to velpatasvir (GS-5816); a novel pan-genotypic inhibitor of the hepatitis C virus NS5A protein. Antimicrob. Agents Chemother. 2016, 60, 5368–5378. [Google Scholar] [CrossRef] [PubMed]
- McPhee, F.; Hernandez, D.; Zhou, N.; Yu, F.; Ueland, J.; Monikowski, A.; Chayama, K.; Toyota, J.; Izumi, N.; Yokosuka, O.; et al. Virological escape in HCV genotype-1-infected patients receiving daclatasvir plus ribavirin and peginterferon alfa-2a or alfa-2b. Antivir. Ther. 2014, 19, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Wyles, D.; Dvory-Sobol, H.; Svarovskaia, E.S.; Doehle, B.P.; Martin, R.; Afdhal, N.H.; Kowdley, K.V.; Lawitz, E.; Brainard, D.M.; Miller, M.D.; et al. Post-treatment resistance analysis of hepatitis C virus from phase II and III clinical trials of ledipasvir/sofosbuvir. J. Hepatol. 2017, 66, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Fridell, R.A.; Wang, C.; Sun, J.H.; O’Boyle, D.R., II; Nower, P.; Valera, L.; Qiu, D.; Roberts, S.; Huang, X.; Kienzle, B.; et al. Genotypic and phenotypic analysis of variants resistant to hepatitis C virus nonstructural protein 5A replication complex inhibitor BMS-790052 in humans: In Vitro and In Vivo correlations. Hepatology 2011, 54, 1924–1935. [Google Scholar] [CrossRef] [PubMed]
- Verbinnen, T.; Van Marck, H.; Vandenbroucke, I.; Vijgen, L.; Claes, M.; Lin, T.I.; Simmen, K.; Neyts, J.; Fanning, G.; Lenz, O. Tracking the evolution of multiple In Vitro hepatitis C virus replicon variants under protease inhibitor selection pressure by 454 deep sequencing. J. Virol. 2010, 84, 11124–11133. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, C.; Kieffer, T.L.; Bartels, D.; Hanzelka, B.; Müh, U.; Welker, M.; Wincheringer, D.; Zhou, Y.; Chu, H.M.; Lin, C.; et al. Dynamic hepatitis C virus genotypic and phenotypic changes in patients treated with the protease inhibitor telaprevir. Gastroenterology 2007, 132, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- Strizki, J.M.; Barnard, R.J.O.; Cheney, C.; McHale, C.; Graham, D.; Himmelberger, A.; Petry, A.; Fraser, I.; Nachbar, R.B.; Hazuda, D.J. Evaluation of NS3 amino acid variants in a phase 1b study of genotype 1 (GT1) and GT3 infected patients with the HCV protease inhibitor; MK-5172. J. Hepatol. 2012, 56, S479. [Google Scholar] [CrossRef]
- Lawitz, E.; Yang, J.C.; Stamm, L.M.; Taylor, J.G.; Cheng, G.; Brainard, D.M.; Miller, M.D.; Mo, H.; Dvory-Sobol, H. Characterization of HCV resistance from a 3-day monotherapy study of voxilaprevir; a novel pangenotypic NS3/4A protease inhibitor. Antivir. Ther. 2017. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, I.M.; Lawitz, E.; Gane, E.J.; Willems, B.E.; Ruane, P.J.; Nahass, R.G.; Borgia, S.M.; Shafran, S.D.; Workowski, K.A.; Pearlman, B.; et al. Efficacy of 8 weeks of sofosbuvir; velpatasvir; and voxilaprevir in patients with chronic HCV infection: 2 phase 3 randomized trials. Gastroenterology 2017, 153, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Zeuzem, S.; Foster, G.R.; Wang, S.; Asatryan, A.; Gane, E.; Feld, J.J.; Asselah, T.; Bourliere, M.; Ruane, P.J.; Wedemeyer, H.; et al. Glecaprevir-pibrentasvir for 8 or 12 weeks in HCV genotype 1 or 3 infection. N. Engl. J. Med. 2018, 378, 354–369. [Google Scholar] [CrossRef] [PubMed]
- Howe, A.Y.; Black, S.; Curry, S.; Ludmerer, S.W.; Liu, R.; Barnard, R.J.; Newhard, W.; Hwang, P.M.; Nickle, D.; Gilbert, C.; et al. Virologic resistance analysis from a phase 2 study of MK-5172 combined with pegylated interferon/ribavirin in treatment-naive patients with hepatitis C virus genotype 1 infection. Clin. Infect. Dis. 2014, 59, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Center for Drug Evaluation and Research, Zepatier Microbiology/Virology Review. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/208261Orig1s000MicroR.pdf (accessed on 1 May 2018).
- Bartlett, S.R.; Grebely, J.; Eltahla, A.A.; Reeves, J.D.; Howe, A.Y.M.; Miller, V.; Ceccherini-Silberstein, F.; Bull, R.A.; Douglas, M.W.; Dore, G.J.; et al. Sequencing of hepatitis C virus for detection of resistance to direct-acting antiviral therapy: A systematic review. Hepatol. Commun. 2017, 1, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Feld, J.J. Resistance testing: Interpretation and incorporation into HCV treatment algorithms. Clin. Liver Dis. 2017, 9, 115–120. [Google Scholar] [CrossRef]
- Krishnan, P.; Schnell, G.; Tripathi, R.; Ng, T.; Reisch, T.; Beyer, J.; Dekhtyar, T.; Irvin, M.; Xie, W.; Larsen, L.; et al. Pooled resistance analysis in HCV genotype 1–6-infected patients treated with glecaprevir/pibrentasvir in phase 2 and 3 clinical trials. J. Hepatol. 2017, 66, 01249-18. [Google Scholar] [CrossRef]
- Gane, E.; Lawitz, E.; Pugatch, D.; Papatheodoridis, G.; Brau, N.; Brown, A.; Pol, S.; Leroy, V.; Persico, M.; Moreno, C.; et al. Glecaprevir and pibrentasvir in patients with HCV and severe renal impairment. N. Engl. J. Med. 2017, 377, 1448–1455. [Google Scholar] [CrossRef] [PubMed]
| Arm a | Dose | Mean Maximal Decrease in HCV RNA (Log10 IU/mL) | n/N (%) b | |
|---|---|---|---|---|
| Genotype 1a | Genotype 1b | |||
| Glecaprevir monotherapy | ||||
| 1 | 100 mg | 4.1 | 5/7 | 0/1 |
| 2 | 400 mg | 4.0 | 4/6 | 2/2 |
| 3 | 700 mg | 4.3 | 6/8 | - |
| 4 | 200 mg | 4.2 | 6/7 | 0/1 |
| 5 c | 200 mg | 3.9 | 4/5 | 3/3 |
| 11 d | 300 mg | 3.8 | 5/7 | - |
| Pibrentasvir monotherapy | ||||
| 6 | 15 mg | 3.4 | 3/6 | 2/2 |
| 7 | 120 mg | 4.5 | 3/7 | 0/1 |
| 8 | 400 mg | 4.3 | 1/8 | - |
| 9 | 40 mg | 4.1 | 3/6 | 1/2 |
| 10 c | 120 mg | 3.9 | 4/6 | 2/2 |
| HCV Subtype | Number of Patient(s) a | Arm (Dose, mg) | Baseline NS3 Polymorphism b | Post-Baseline | |
|---|---|---|---|---|---|
| Treatment-Emergent NS3 Substitution b,c | Visit Day d | ||||
| 1a | 1 | 3(700) | T54S, V55I | None | 2 |
| 1 | 3(700) | V55A | None | 3 | |
| 1 | 11(300) | V55A, Q80K | None | 2 | |
| 3 | 2(400)/3(700)/4(200) | Q80K | NA | NA | |
| 8 | Multiple | Q80K | None | Multiple | |
| 1 | 4(200) | Q80K | K80Q | 2 | |
| 1 | 1(100) | Q80K | T54S | 2 | |
| 1 | 3(700) | Q80K | A156T | 3 | |
| 1 | 3(700) | Q80K, S122G | K/Q80L e | 2 | |
| 1 | 11(300) | Q80K, I170V | None | 2 | |
| 1 | 3(700) | Q80L | NA | NA | |
| 1 | 1(100) | Q80L, I170V | NA | NA | |
| 1 | 1(100) | I170V | None | 2 | |
| 1b | 1 | 1(100) | Y56F, S122T, V170I | NA | NA |
| 1 | 2(400) | Y56F, S122T, V170I | None | 3 | |
| 1 | 5(200) | Q80L, V170I | None | 2 | |
| 1 | 4(200) | V170I | NA | NA | |
| HCV Subtype | Number of Patient a | Arm (Dose, mg) | Baseline NS5A Polymorphism b | Post-Baseline | |
|---|---|---|---|---|---|
| Treatment-Emergent NS5A Substitution b,c | Visit Day d | ||||
| 1a | 1 | 8(400) | M28V, Q30R, H58P | NA | NA |
| 1 e | 8(400) | M28V, Q30R, Y93N | L31M, H58D, A92T | 4 | |
| 1 | 7(120) | H58N/P/T | None | 2 | |
| 1 f | 10(120) | H58P | Q30Y, Y93H | 3 | |
| 1 | 8(400) | E62D | NA | NA | |
| 1 | 10(120) | E62D | None | 3 | |
| 1 g | 10(120) | Y93C/S | Q30R, P32L, H58D | 4 | |
| 1b | 1 | 7(120) | L31M, P58S | NA | NA |
| 1 | 10(120) | P58T, Q62E | None | 2 | |
| 1 | 6(15) | Y93H | None | 4 | |
| 1 | 9(40) | P58S | None | 4 | |
| 1 | 9(40) | P58T | NA | NA | |
| HCV Subtype | NS3 Substitutions a | Glecaprevir EC50 (Mean ± SD, nM) | Fold Change in EC50 b | Replication Efficiency c (%) |
|---|---|---|---|---|
| 1a (H77) | Wild-type | 0.21 ± 0.08 | - | 100 |
| T54S | 0.20 ± 0.06 | 1.0 | 6.2 | |
| V55I | 0.05 ± 0.01 | 0.22 | 81 | |
| Q80K | 0.19 ± 0.05 | 0.91 | 91 | |
| Q80L | 0.44 ± 0.33 | 2.1 | 38 | |
| A156T | 286 ± 93 | 1361 | 5.2 | |
| I170V | 0.21 ± 0.03 | 1.0 | 77 | |
| 1b (Con 1) | Wild-type | 0.47 ± 0.13 | - | 100 |
| Y56F | NA | NA | 1.0 | |
| Q80L | 0.30 ± 0.07 | 0.64 | 123 |
| HCV Subtype | NS5A Substitutions a | Pibrentasvir EC50 (Mean ± SD, pM) | Fold Change in EC50 b | Replication Efficiency c (%) |
|---|---|---|---|---|
| 1a (H77) | Wild-type | 0.72 ± 0.45 | - | 100 |
| M28V | 1.3 ± 0.86 | 1.8 | 87 | |
| Q30R | 1.2 ± 0.62 | 1.7 | 60 | |
| Q30Y | 0.55 ± 0.11 | 0.77 | 21 | |
| L31M | 0.76 ± 0.11 | 1.1 | 141 | |
| P32L | 1.2 ± 0.43 | 1.7 | 19 | |
| H58D | 0.80 ± 0.17 | 1.1 | 66 | |
| H58P | 0.46 ± 0.06 | 0.64 | 129 | |
| E62D | 0.46 ± 0.06 | 0.64 | 104 | |
| A92T | 0.28 ± 0.03 | 0.39 | 4.1 | |
| Y93C | 1.2 ± 0.57 | 1.7 | 24 | |
| Y93H | 4.8 ± 1.5 | 6.7 | 18 | |
| Y93N | 5.1 ± 2.1 | 7.1 | 35 | |
| Y93S | 1.2 ± 0.20 | 1.6 | 3.4 | |
| M28V + Q30R d | 0.82 ± 0.04 | 1.1 | 17 | |
| Q30R + Y93N | 95 ± 16 | 131 | 3.6 | |
| Q30R + Y93S | NA | NA | <0.5 | |
| L31M + Y93N | 140 ± 34 | 195 | 31 | |
| P32L + Y93C | NA | NA | 0.5 | |
| H58D + Y93C | 168 ± 32 | 233 | 13 | |
| H58D + Y93S | 1058 ± 457 | 1469 | 2.1 | |
| H58D + Y93N | 1418 ± 279 | 1969 | 21 | |
| A92T + Y93N | NA | NA | <0.5 | |
| 1b (Con 1) | Wild-type | 1.9 ± 0.80 | - | 100 |
| L31M | 2.9 ± 1.2 | 1.5 | 119 | |
| P58S | 2.4 ± 1.3 | 1.2 | 80 | |
| Y93H | 1.1 ± 0.27 | 0.60 | 38 |
| Genotype 1a (H77) | Replication Efficiency a (%) | Daclatasvir | Ombitasvir | Elbasvir | Ledipasvir | Velpatasvir | Pibrentasvir |
| WT EC50 (pM) | 7.9 ± 2.0 | 2.7 ± 0.80 | 4.8 ± 1.7 | 11 ± 4.0 | 4.1 ± 1.6 | 0.72 ± 0.45 | |
| NS5A substitutions | Fold change in EC50 b | ||||||
| M28T | 89 | 437 | 8965 | 21 | 108 | 13 | 2.0 |
| M28V | 87 | 0.95 | 58 | 1.0 | 0.72 | 0.82 | 1.8 |
| Q30E | 70 | 10,400 | 1326 | 58 | 2611 | 17 | 2.4 |
| Q30H | 64 | 154 | 2.8 | 5.8 | 367 | 4.3 | 1.0 |
| Q30R | 60 | 178 | 800 | 14 | 400 | 4.3 | 1.7 |
| L31M | 141 | 140 | 1.8 | 5.2 | 339 | 8.2 | 1.1 |
| P32L | 19 | 388 | 44 | 6.4 | 202 | 15 | 1.7 |
| H58D | 66 | 124 | 243 | 4.5 | 130 | 2.1 | 1.1 |
| A92K | 2.3 | 74,055 | 11,995 | 7188 | 112,034 | 54,665 | 0.85 |
| Y93C | 24 | 383 | 1675 | 19 | 811 | 19 | 1.7 |
| Y93H | 18 | 2324 | 41,383 | 201 | 2753 | 409 | 6.7 |
| Y93N | 35 | 8641 | 66,740 | 605 | 6561 | 3133 | 7.1 |
| Y93S | 3.4 | 2395 | 7790 | 63 | 2347 | 53 | 1.6 |
| K24R + Q30R | 83 | 1209 | 950 | 65 | 2757 | 5.9 | 0.41 |
| M28T + Q30R | 32 | 8462 | 3,537,179 | 546 | 2224 | 34 | 1.6 |
| Q30H + Y93H | 35 | 11,270 | 33,490 | 786 | 2778 | 872 | 17 |
| Q30R + L31M | 49 | 16,785 | 504 | 2842 | 17,537 | 150 | 3.0 |
| Q30R + H58D | 50 | 64,004 | 320,751 | 4066 | 21,207 | 66 | 126 |
| Q30R + Y93C | 6.2 | 5010 | 43,352 | 328 | 1668 | 117 | 3.8 |
| Q30R + Y93H | 21 | 17,018 | 351,081 | 4263 | 7740 | 6949 | 260 |
| L31M + Y93C | 32 | 32,979 | 1973 | 2813 | 17,564 | 2965 | 6.1 |
| L31M + Y93H | 11 | 21,165 | ND | 4266 | 24,840 | 18,323 | 75 |
| L31V + Y93H | 73 | 275,021 | 32,495 | 13,736 | 61,297 | 166,675 | 94 |
| Genotype 1b (Con 1) | Replication Efficiency a (%) | Daclatasvir | Ombitasvir | Elbasvir | Ledipasvir | Velpatasvir | Pibrentasvir |
| WT EC50 (pM) | 11 ± 2.0 | 0.79 ± 0.25 | 3.2 ± 1.4 | 1.6 ± 0.62 | 4.8 ± 0.77 | 1.9 ± 0.80 | |
| NS5A substitutions | Fold change in EC50 b | ||||||
| L31V | 86 | 2.5 | 8.4 | 0.96 | 22 | 2.1 | 0.77 |
| P58D | 69 | 3.2 | 577 | 5.1 | 122 | 3.2 | 1.2 |
| Y93H | 38 | 7.3 | 77 | 7.1 | 345 | 3.0 | 0.60 |
| Y93N | 52 | 7.8 | 220 | 2.5 | 250 | 3.8 | 0.62 |
| Y93S | 23 | 0.82 | 12 | 0.66 | 60 | 0.49 | 0.39 |
| L31I + Y93H | 44 | 311 | ND | 176 | 63,257 | 88 | 1.2 |
| L31M + Y93H | 11 | 1166 | 142 | 626 | 13,940 | 143 | 0.70 |
| L31V + Y93H | 24 | 1259 | 12,328 | 1040 | 67,323 | 2201 | 0.87 |
| P58S + Y93H | 34 | 51 | 1401 | 25 | 1101 | 26 | 0.78 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ng, T.I.; Pilot-Matias, T.; Tripathi, R.; Schnell, G.; Krishnan, P.; Reisch, T.; Beyer, J.; Dekhtyar, T.; Irvin, M.; Lu, L.; et al. Resistance Analysis of a 3-Day Monotherapy Study with Glecaprevir or Pibrentasvir in Patients with Chronic Hepatitis C Virus Genotype 1 Infection. Viruses 2018, 10, 462. https://doi.org/10.3390/v10090462
Ng TI, Pilot-Matias T, Tripathi R, Schnell G, Krishnan P, Reisch T, Beyer J, Dekhtyar T, Irvin M, Lu L, et al. Resistance Analysis of a 3-Day Monotherapy Study with Glecaprevir or Pibrentasvir in Patients with Chronic Hepatitis C Virus Genotype 1 Infection. Viruses. 2018; 10(9):462. https://doi.org/10.3390/v10090462
Chicago/Turabian StyleNg, Teresa I., Tami Pilot-Matias, Rakesh Tripathi, Gretja Schnell, Preethi Krishnan, Thomas Reisch, Jill Beyer, Tatyana Dekhtyar, Michelle Irvin, Liangjun Lu, and et al. 2018. "Resistance Analysis of a 3-Day Monotherapy Study with Glecaprevir or Pibrentasvir in Patients with Chronic Hepatitis C Virus Genotype 1 Infection" Viruses 10, no. 9: 462. https://doi.org/10.3390/v10090462
APA StyleNg, T. I., Pilot-Matias, T., Tripathi, R., Schnell, G., Krishnan, P., Reisch, T., Beyer, J., Dekhtyar, T., Irvin, M., Lu, L., Asatryan, A., Campbell, A., Yao, B., Lovell, S., Mensa, F., Lawitz, E. J., Kort, J., & Collins, C. (2018). Resistance Analysis of a 3-Day Monotherapy Study with Glecaprevir or Pibrentasvir in Patients with Chronic Hepatitis C Virus Genotype 1 Infection. Viruses, 10(9), 462. https://doi.org/10.3390/v10090462





