Natural Variation in Resistance to Virus Infection in Dipteran Insects
Abstract
1. Introduction
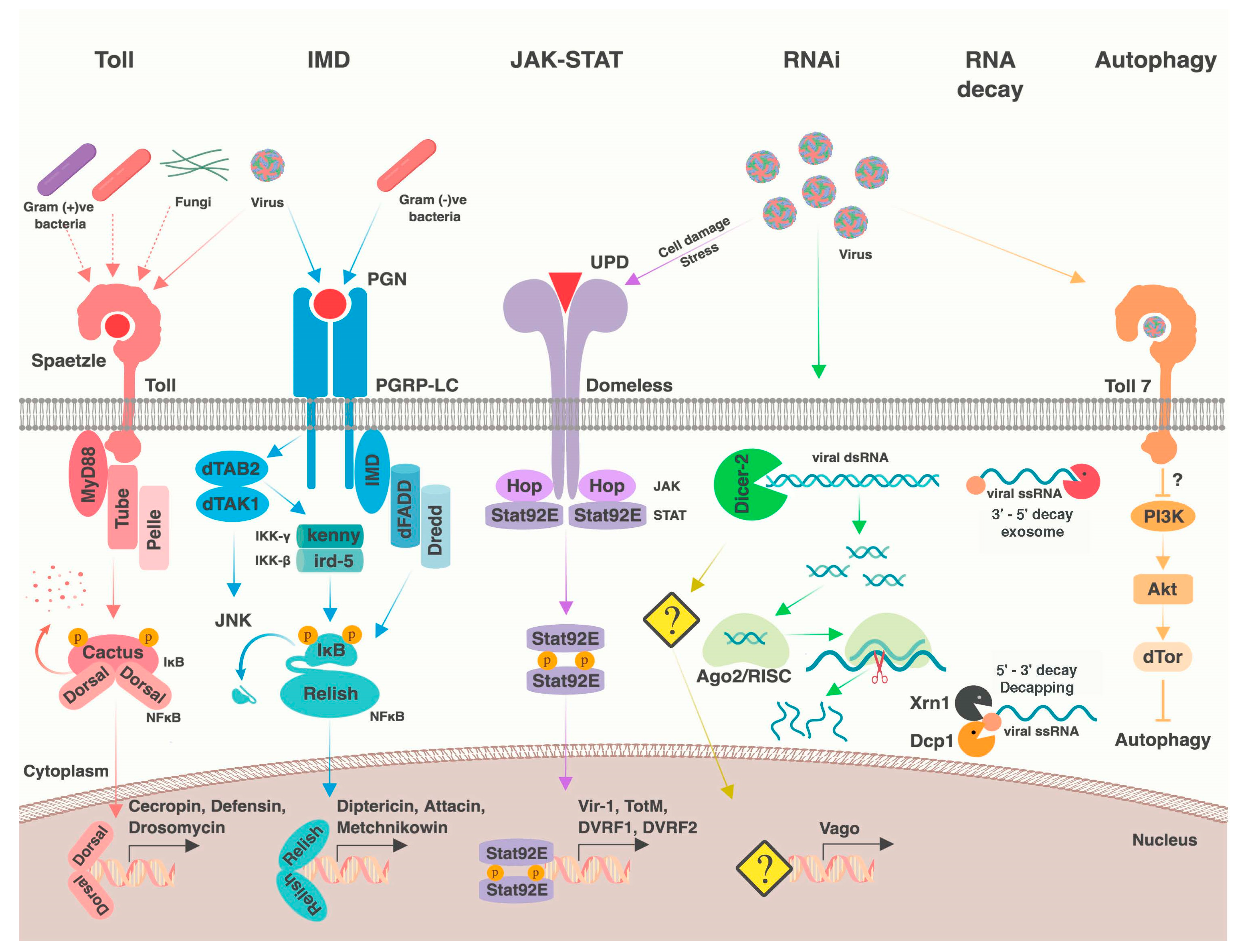
2. Antiviral Immune Processes
2.1. RNA Interference
2.2. Other RNA Processes
2.3. Nuclear Factor κB Pathways
2.4. JAK-STAT Pathway
2.5. Nutrient Signaling
2.6. Apoptosis, Phagocytosis, and Autophagy
3. Virus-Specific Responses
3.1. RNA Interference
3.2. Induced Immune Responses
3.3. Autophagy, Phagocytosis, and Apoptosis
4. Genetic Variation in Antiviral Immunity in Dipterans
4.1. Segregating Genetic Variants Associated with Viral Resistance in Drosophila
4.2. Evolution of Resistance Loci
4.3. Genetic Variation Associated with Viral Resistance in Mosquitoes
5. Microbe-Dependent Variation in Dipteran Antiviral Immunity
5.1. Wolbachia in Drosophila
5.2. Wolbachia in Mosquitoes
5.3. Gut Microbiota in Drosophila
5.4. Gut Microbiota in Mosquitoes
6. Other Sources of Variation
6.1. Abiotic Environment
6.2. Sexual Dimorphism and Mating
6.3. Infection History
6.4. Endogenous Viral Elements—Non-Retroviral Integrated RNA Virus Sequences
7. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Merkling, S.H.; van Rij, R.P. Beyond RNAi: Antiviral defense strategies in Drosophila and mosquito. J. Insect Physiol. 2013, 59, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Kingsolver, M.B.; Huang, Z.; Hardy, R.W. Insect antiviral innate immunity: Pathways, effectors, and connections. J. Mol. Biol. 2013, 425, 4921–4936. [Google Scholar] [CrossRef] [PubMed]
- Bronkhorst, A.W.; van Rij, R.P. The long and short of antiviral defense: Small RNA-based immunity in insects. Curr. Opin. Virol. 2014, 7, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Lamiable, O.; Imler, J.L. Induced antiviral innate immunity in Drosophila. Curr. Opin. Microbiol. 2014, 20, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Jupatanakul, N.; Dimopoulos, G. Mosquito immunity against arboviruses. Viruses 2014, 6, 4479–4504. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Cherry, S. Viruses and antiviral immunity in Drosophila. Dev. Comp. Immunol. 2014, 42, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Samuel, G.H.; Adelman, Z.N.; Myles, K.M. Antiviral Immunity and Virus-Mediated Antagonism in Disease Vector Mosquitoes. Trends Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.E.; Kantor, A.M.; Passarelli, A.L.; Clem, R.J. Tissue Barriers to Arbovirus Infection in Mosquitoes. Viruses 2015, 7, 3741–3767. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Chevillon, C.; Albright, R.G.; Thaisomboonsuk, B.; Richardson, J.H.; Jarman, R.G.; Scott, T.W. Genetic specificity and potential for local adaptation between dengue viruses and mosquito vectors. BMC Evol. Biol. 2009, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L. Quantitative genetics of Aedes aegypti vector competence for dengue viruses: Towards a new paradigm? Trends Parasitol. 2011, 27, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Brackney, D.E.; Beane, J.E.; Ebel, G.D. RNAi Targeting of West Nile Virus in Mosquito Midguts Promotes Virus Diversification. PLoS Pathog. 2009, 5, e1000502. [Google Scholar] [CrossRef] [PubMed]
- Forrester, N.L.; Coffey, L.L.; Weaver, S.C. Arboviral bottlenecks and challenges to maintaining diversity and fitness during mosquito transmission. Viruses 2014, 6, 3991–4004. [Google Scholar] [CrossRef] [PubMed]
- Lequime, S.; Fontaine, A.; Ar Gouilh, M.; Moltini-Conclois, I.; Lambrechts, L. Genetic Drift, Purifying Selection and Vector Genotype Shape Dengue Virus Intra-host Genetic Diversity in Mosquitoes. PLoS Genet. 2016, 12, e1006111. [Google Scholar] [CrossRef] [PubMed]
- Grubaugh, N.D.; Weger-Lucarelli, J.; Murrieta, R.A.; Fauver, J.R.; Garcia-Luna, S.M.; Prasad, A.N.; Black, W.C.; Ebel, G.D. Genetic Drift during Systemic Arbovirus Infection of Mosquito Vectors Leads to Decreased Relative Fitness during Host Switching. Cell Host Microbe 2016, 19, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Brault, A.C.; Powers, A.M.; Ortiz, D.; Estrada-Franco, J.G.; Navarro-Lopez, R.; Weaver, S.C. Venezuelan equine encephalitis emergence: Enhanced vector infection from a single amino acid substitution in the envelope glycoprotein. Proc. Natl. Acad. Sci. USA 2004, 101, 11344–11349. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Vanlandingham, D.L.; McGee, C.E.; Higgs, S. A single mutation in Chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007, 3, 1895–1906. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Weaver, S.C. Sequential adaptive mutations enhance efficient vector switching by chikungunya virus and its epidemic emergence. PLoS Pathog. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Chen, R.; Leal, G.; Forrester, N.; Higgs, S.; Huang, J.; Weaver, S.C. Chikungunya virus emergence is constrained in Asia by lineage-specific adaptive landscapes. Proc. Natl. Acad. Sci. USA 2011, 108, 7872–7877. [Google Scholar] [CrossRef] [PubMed]
- Vignuzzi, M.; Higgs, S. The Bridges and Blockades to Evolutionary Convergence on the Road to Predicting Chikungunya Virus Evolution. Annu. Rev. Virol. 2017, 4, 181–200. [Google Scholar] [CrossRef] [PubMed]
- Van Mierlo, J.T.; Bronkhorst, A.W.; Overheul, G.J.; Sadanandan, S.A.; Ekström, J.O.; Heestermans, M.; Hultmark, D.; Antoniewski, C.; van Rij, R.P. Convergent Evolution of Argonaute-2 Slicer Antagonism in Two Distinct Insect RNA Viruses. PLoS Pathog. 2012, 8, e1002872. [Google Scholar] [CrossRef] [PubMed]
- Kemp, C.; Mueller, S.; Goto, A.; Barbier, V.; Paro, S.; Bonnay, F.; Dostert, C.; Troxler, L.; Hetru, C.; Meignin, C.; et al. Broad RNA Interference-Mediated Antiviral Immunity and Virus-Specific Inducible Responses in Drosophila. J. Immunol. 2013, 190, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Olson, K.E.; Blair, C.D. Arbovirus-mosquito interactions: RNAi pathway. Curr. Opin. Virol. 2015, 15, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Goic, B.; Vodovar, N.; Mondotte, J.A.; Monot, C.; Frangeul, L.; Blanc, H.; Gausson, V.; Vera-Otarola, J.; Cristofari, G.; Saleh, M.C. RNA-mediated interference and reverse transcription control the persistence of RNA viruses in the insect model Drosophila. Nat. Immunol. 2013, 14, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Goic, B.; Stapleford, K.A.; Frangeul, L.; Doucet, A.J.; Gausson, V.; Blanc, H.; Schemmel-Jofre, N.; Cristofari, G.; Lambrechts, L.; Vignuzzi, M.; et al. Virus-derived DNA drives mosquito vector tolerance to arboviral infection. Nat. Commun. 2016, 7, 12410. [Google Scholar] [CrossRef] [PubMed]
- Tassetto, M.; Kunitomi, M.; Andino, R. Circulating Immune Cells Mediate a Systemic RNAi-Based Adaptive Antiviral Response in Drosophila. Cell 2017, 169, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Petit, M.; Mongelli, V.; Frangeul, L.; Blanc, H.; Jiggins, F.; Saleh, M.-C. piRNA pathway is not required for antiviral defense in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2016, 113, E4218–E4227. [Google Scholar] [CrossRef] [PubMed]
- Vodovar, N.; Bronkhorst, A.W.; van Cleef, K.W.R.; Miesen, P.; Blanc, H.; van Rij, R.P.; Saleh, M.-C. Arbovirus-Derived piRNAs Exhibit a Ping-Pong Signature in Mosquito Cells. PLoS ONE 2012, 7, e30861. [Google Scholar] [CrossRef] [PubMed]
- Morazzani, E.M.; Wiley, M.R.; Murreddu, M.G.; Adelman, Z.N.; Myles, K.M. Production of Virus-Derived Ping-Pong-Dependent piRNA-like Small RNAs in the Mosquito Soma. PLoS Pathog. 2012, 8, e1002470. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Black, W.C.; Hess, A.M.; Foy, B.D. Comparative genomics of small RNA regulatory pathway components in vector mosquitoes. BMC Genom. 2008, 9, 425. [Google Scholar] [CrossRef] [PubMed]
- Miesen, P.; Girardi, E.; van Rij, R.P. Distinct sets of PIWI proteins produce arbovirus and transposon-derived piRNAs in Aedes aegypti mosquito cells. Nucleic Acids Res. 2015, 43, 6545–6556. [Google Scholar] [CrossRef] [PubMed]
- Keene, K.M.; Foy, B.D.; Sanchez-Vargas, I.; Beaty, B.J.; Blair, C.D.; Olson, K.E. RNA interference acts as a natural antiviral response to O’nyong-nyong virus (Alphavirus; Togaviridae) infection of Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2004, 101, 17240–17245. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Donald, C.L.; Human, S.; Watson, M.; Siu, R.W.C.; McFarlane, M.; Fazakerley, J.K.; Kohl, A.; Fragkoudis, R. Knockdown of piRNA pathway proteins results in enhanced semliki forest virus production in mosquito cells. J. Gen. Virol. 2013, 94, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- Varjak, M.; Maringer, K.; Watson, M.; Sreenu, V.B.; Fredericks, A.C.; Pondeville, E.; Donald, C.L.; Sterk, J.; Kean, J.; Vazeille, M.; et al. Aedes aegypti Piwi4 Is a Noncanonical PIWI Protein Involved in Antiviral Responses. mSphere 2017, 2, e00144-17. [Google Scholar] [CrossRef] [PubMed]
- Shoemaker, C.J.; Green, R. Translation drives mRNA quality control. Nat. Struct. Mol. Biol. 2012, 19, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Schoenberg, D.R.; Maquat, L.E. Regulation of cytoplasmic mRNA decay. Nat. Rev. Genet. 2012, 13, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.L.; Wilusz, J. Cytoplasmic Viruses: Rage against the (Cellular RNA Decay) Machine. PLoS Pathog. 2013, 9, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Molleston, J.M.; Cherry, S. Attacked from all sides: RNA decay in antiviral defense. Viruses 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Orban, T.I.; Izaurralde, E. Decay of mRNAs targeted by RISC requires XRN1, the Ski complex, and the exosome. RNA 2005, 11, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, K.C.; McLane, L.M.; Maqbool, T.; Panda, D.; Gordesky-Gold, B.; Cherry, S. A genome-wide RNAi screen reveals that mRNA decapping restricts bunyaviral replication by limiting the pools of dcp2-accessible targets for cap-snatching. Genes Dev. 2013, 27, 1511–1525. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Magwire, M.M.; Bayer, F.; Jiggins, F.M. A Polymorphism in the Processing Body Component Ge-1 Controls Resistance to a Naturally Occurring Rhabdovirus in Drosophila. PLoS Pathog. 2016, 12. [Google Scholar] [CrossRef]
- Molleston, J.M.; Sabin, L.R.; Moy, R.H.; Menghani, S.V.; Rausch, K.; Gordesky-Gold, B.; Hopkins, K.C.; Zhou, R.; Jensen, T.H.; Wilusz, J.E.; et al. A conserved virus-induced cytoplasmic TRAMP-like complex recruits the exosome to target viral RNA for degradation. Genes Dev. 2016, 30, 1658–1670. [Google Scholar] [CrossRef] [PubMed]
- Khong, A.; Jan, E. Modulation of Stress Granules and P Bodies during Dicistrovirus Infection. J. Virol. 2011, 85, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.L.; Anderson, J.R.; Kumagai, Y.; Wilusz, C.J.; Akira, S.; Khromykh, A.A.; Wilusz, J. A noncoding RNA produced by arthropod-borne flaviviruses inhibits the cellular exoribonuclease XRN1 and alters host mRNA stability. RNA 2012, 18, 2029–2040. [Google Scholar] [CrossRef] [PubMed]
- Samuel, C.E. ADARs: Viruses and innate immunity. Curr. Top. Microbiol. Immunol. 2012, 353, 163–195. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, J.A.; Keegan, L.P.; Wilfert, L.; O’Connell, M.A.; Jiggins, F.M. Evidence for ADAR-induced hypermutation of the Drosophila sigma virus (Rhabdoviridae). BMC Genet. 2009, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Piontkivska, H.; Matos, L.F.; Paul, S.; Scharfenberg, B.; Farmerie, W.G.; Miyamoto, M.M.; Wayne, M.L. Role of host-driven mutagenesis in determining genome evolution of sigma virus (DMelSV; Rhabdoviridae) in Drosophila melanogaster. Genome Biol. Evol. 2016, 8, 2952–2963. [Google Scholar] [CrossRef] [PubMed]
- Piontkivska, H.; Frederick, M.; Miyamoto, M.M.; Wayne, M.L. RNA editing by the host ADAR system affects the molecular evolution of the Zika virus. Ecol. Evol. 2017, 7, 4475–4485. [Google Scholar] [CrossRef] [PubMed]
- Valanne, S.; Wang, J.-H.; Ramet, M. The Drosophila Toll Signaling Pathway. J. Immunol. 2011, 186, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Myllymaki, H.; Valanne, S.; Ramet, M. The Drosophila Imd Signaling Pathway. J. Immunol. 2014, 192, 3455–3462. [Google Scholar] [CrossRef] [PubMed]
- Silverman, N.; Zhou, R.; Erlich, R.L.; Hunter, M.; Bernstein, E.; Schneider, D.; Maniatis, T. Immune activation of NF-kappaB and JNK requires Drosophila TAK1. J. Biol. Chem. 2003, 278, 48928–48934. [Google Scholar] [CrossRef] [PubMed]
- Delaney, J.R.; Stöven, S.; Uvell, H.; Anderson, K.V.; Engström, Y.; Mlodzik, M. Cooperative control of Drosophila immune responses by the JNK and NF-κB signaling pathways. EMBO J. 2006, 25, 3068–3077. [Google Scholar] [CrossRef] [PubMed]
- Zambon, R.A.; Nandakumar, M.; Vakharia, V.N.; Wu, L.P. The Toll pathway is important for an antiviral response in Drosophila. Proc. Natl. Acad. Sci. USA 2005, 102, 7257–7262. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008, 4, e1000098. [Google Scholar] [CrossRef] [PubMed]
- Avadhanula, V.; Weasner, B.P.; Hardy, G.G.; Kumar, J.P.; Hardy, R.W. A novel system for the launch of alphavirus RNA synthesis reveals a role for the Imd pathway in arthropod antiviral response. PLoS Pathog. 2009, 5, e1000582. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Jan, E.; Sarnow, P.; Schneider, D. The Imd pathway is involved in antiviral immune responses in Drosophila. PLoS ONE 2009, 4, e7436. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, M.; Moy, R.H.; Xu, J.; Bambina, S.; Yasunaga, A.; Shelly, S.S.; Gold, B.; Cherry, S. Virus Recognition by Toll-7 Activates Antiviral Autophagy in Drosophila. Immunity 2012, 36, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, Á.G.; Naylor, H.; Esteves, S.S.; Pais, I.S.; Martins, N.E.; Teixeira, L. The Toll-Dorsal Pathway Is Required for Resistance to Viral Oral Infection in Drosophila. PLoS Pathog. 2014, 10, e1004507. [Google Scholar] [CrossRef] [PubMed]
- Thoetkiattikul, H.; Beck, M.H.; Strand, M.R. Inhibitor kappaB-like proteins from a polydnavirus inhibit NF-kappaB activation and suppress the insect immune response. Proc. Natl. Acad. Sci. USA 2005, 102, 11426–11431. [Google Scholar] [CrossRef] [PubMed]
- Lamiable, O.; Kellenberger, C.; Kemp, C.; Troxler, L.; Pelte, N.; Boutros, M.; Marques, J.T.; Daeffler, L.; Hoffmann, J.A.; Roussel, A.; et al. Cytokine Diedel and a viral homologue suppress the IMD pathway in Drosophila. Proc. Natl. Acad. Sci. USA 2016, 113, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Luplertlop, N.; Surasombatpattana, P.; Patramool, D.; Dumas, E.; Wasinpiyamongkol, L.; Saune, L.; Hamel, R.; Bernard, E.; Sereno, D.; Thomas, F.R.; et al. Induction of a peptide with activity against a broad spectrum of pathogens in the Aedes aegypti salivary gland, following infection with Dengue Virus. PLoS Pathog. 2011, 7, e1001252. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Kingsolver, M.B.; Avadhanula, V.; Hardy, R.W. An Antiviral Role for Antimicrobial Peptides during the Arthropod Response to Alphavirus Replication. J. Virol. 2013, 87, 4272–4280. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Pan, P.C.; Govind, S. A role for the Drosophila Toll/Cactus pathway in larval hematopoiesis. Development 1998, 125, 1909–1920. [Google Scholar] [PubMed]
- Georgel, P.; Naitza, S.; Kappler, C.; Ferrandon, D.; Zachary, D.; Swimmer, C.; Kopczynski, C.; Duyk, G.; Reichhart, J.M.; Hoffmann, J.A. Drosophila Immune Deficiency (IMD) Is a Death Domain Protein that Activates Antibacterial Defense and Can Promote Apoptosis. Dev. Cell 2001, 1, 503–514. [Google Scholar] [CrossRef]
- Liu, B.; Behura, S.K.; Clem, R.J.; Schneemann, A.; Becnel, J.; Severson, D.W.; Zhou, L. P53-Mediated Rapid Induction of Apoptosis Conveys Resistance to Viral Infection in Drosophila melanogaster. PLoS Pathog. 2013, 9, e1003137. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Hopkins, K.; Sabin, L.; Yasunaga, A.; Subramanian, H.; Lamborn, I.; Gordesky-Gold, B.; Cherry, S. ERK signaling couples nutrient status to antiviral defense in the insect gut. Proc. Natl. Acad. Sci. USA 2013, 110, 15025–15030. [Google Scholar] [CrossRef] [PubMed]
- Sansone, C.L.; Cohen, J.; Yasunaga, A.; Xu, J.; Osborn, G.; Subramanian, H.; Gold, B.; Buchon, N.; Cherry, S. Microbiota-dependent priming of antiviral intestinal immunity in Drosophila. Cell Host Microbe 2015, 18, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Arbouzova, N.I.; Zeidler, M.P. JAK/STAT signalling in Drosophila: Insights into conserved regulatory and cellular functions. Development 2006, 133, 2605–2616. [Google Scholar] [CrossRef] [PubMed]
- Deddouche, S.; Matt, N.; Budd, A.; Mueller, S.; Kemp, C.; Galiana-Arnoux, D.; Dostert, C.; Antoniewski, C.; Hoffmann, J.A.; Imler, J.L. The DExD/H-box helicase Dicer-2 mediates the induction of antiviral activity in Drosophila. Nat. Immunol. 2008, 9, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Paradkar, P.N.; Trinidad, L.; Voysey, R.; Duchemin, J.-B.; Walker, P.J. Secreted Vago restricts West Nile virus infection in Culex mosquito cells by activating the Jak-STAT pathway. Proc. Natl. Acad. Sci. USA 2012, 109, 18915–18920. [Google Scholar] [CrossRef] [PubMed]
- Jupatanakul, N.; Sim, S.; Angleró-Rodríguez, Y.I.; Souza-Neto, J.; Das, S.; Poti, K.E.; Rossi, S.L.; Bergren, N.; Vasilakis, N.; Dimopoulos, G. Engineered Aedes aegypti JAK/STAT Pathway-Mediated Immunity to Dengue Virus. PLoS Negl. Trop. Dis. 2017, 11, e0005187. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Jouanguy, E.; Irving, P.; Troxler, L.; Galiana-Arnoux, D.; Hetru, C.; Hoffmann, J.A.; Imler, J.L. The Jak-STAT signaling pathway is required but not sufficient for the antiviral response of Drosophila. Nat. Immunol. 2005, 6, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Merkling, S.H.; Bronkhorst, A.W.; Kramer, J.M.; Overheul, G.J.; Schenck, A.; Van Rij, R.P. The Epigenetic Regulator G9a Mediates Tolerance to RNA Virus Infection in Drosophila. PLoS Pathog. 2015, 11, e1004692. [Google Scholar] [CrossRef] [PubMed]
- Souza-Neto, J.A.; Sim, S.; Dimopoulos, G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc. Natl. Acad. Sci. USA 2009, 106, 17841–17846. [Google Scholar] [CrossRef] [PubMed]
- Lazzaro, B.P.; Little, T.J. Immunity in a variable world. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Ponton, F.; Wilson, K.; Holmes, A.J.; Cotter, S.C.; Raubenheimer, D.; Simpson, S.J. Integrating nutrition and immunology: A new frontier. J. Insect Physiol. 2013, 59, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Partridge, L.; Alic, N.; Bjedov, I.; Piper, M.D.W. Ageing in Drosophila: The role of the insulin/Igf and TOR signalling network. Exp. Gerontol. 2011, 46, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Shelly, S.; Lukinova, N.; Bambina, S.; Berman, A.; Cherry, S. Autophagy Is an Essential Component of Drosophila Immunity against Vesicular Stomatitis Virus. Immunity 2009, 30, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Moy, R.H.; Gold, B.; Molleston, J.M.; Schad, V.; Yanger, K.; Salzano, M.V.; Yagi, Y.; Fitzgerald, K.A.; Stanger, B.Z.; Soldan, S.S.; et al. Antiviral autophagy restricts rift valley fever virus infection and is conserved from flies to mammals. Immunity 2014, 40, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Lamiable, O.; Arnold, J.; da Silva de Faria, I.J.; Proveti Olmo, R.; Bergami, F.; Meignin, C.; Hoffmann, J.A.; Marques, J.T.; Imler, J.-L. Analysis of the contribution of hemocytes and autophagy to Drosophila antiviral immunity. J. Virol. 2016, 90, JVI.00238-16. [Google Scholar] [CrossRef] [PubMed]
- DiAngelo, J.R.; Bland, M.L.; Bambina, S.; Cherry, S.; Birnbaum, M.J. The immune response attenuates growth and nutrient storage in Drosophila by reducing insulin signaling. Proc. Natl. Acad. Sci. USA 2009, 106, 20853–20858. [Google Scholar] [CrossRef] [PubMed]
- Becker, T.; Loch, G.; Beyer, M.; Zinke, I.; Aschenbrenner, A.C.; Carrera, P.; Inhester, T.; Schultze, J.L.; Hoch, M. FOXO-dependent regulation of innate immune homeostasis. Nature 2010, 463, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Spellberg, M.J.; Marr, M.T. FOXO regulates RNA interference in Drosophila and protects from RNA virus infection. Proc. Natl. Acad. Sci. USA 2015, 112, 14587–14592. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.K.; Hardy, R.W. Role for the Phosphatidylinositol 3-Kinase-Akt-TOR Pathway during Sindbis Virus Replication in Arthropods. J. Virol. 2012, 86, 3595–3604. [Google Scholar] [CrossRef] [PubMed]
- Everett, H.; McFadden, G. Apoptosis: An innate immune response to virus infection. Trends Microbiol. 1999, 7, 160–165. [Google Scholar] [CrossRef]
- Steller, H. Regulation of apoptosis in Drosophila. Cell Death Differ. 2008, 15, 1132–1138. [Google Scholar] [CrossRef] [PubMed]
- Vandergaast, R.; Mitchell, J.K.; Byers, N.M.; Friesen, P.D. Insect Inhibitor-of-Apoptosis (IAP) Proteins Are Negatively Regulated by Signal-Induced N-Terminal Degrons Absent within Viral IAP Proteins. J. Virol. 2015, 89, 4481–4493. [Google Scholar] [CrossRef] [PubMed]
- Vandergaast, R.; Schultz, K.L.W.; Cerio, R.J.; Friesen, P.D. Active Depletion of Host Cell Inhibitor-of-Apoptosis Proteins Triggers Apoptosis upon Baculovirus DNA Replication. J. Virol. 2011, 85, 8348–8358. [Google Scholar] [CrossRef] [PubMed]
- Nainu, F.; Tanaka, Y.; Shiratsuchi, A.; Nakanishi, Y. Protection of Insects against Viral Infection by Apoptosis-Dependent Phagocytosis. J. Immunol. 2015, 195, 5696–5706. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Zhang, X. Involvement of Ran in the regulation of phagocytosis against virus infection in S2 cells. Dev. Comp. Immunol. 2013, 41, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Zhang, X. The Wnt signaling pathway is involved in the regulation of phagocytosis of virus in Drosophila. Sci. Rep. 2013, 3, 2069. [Google Scholar] [CrossRef] [PubMed]
- Moy, R.H.; Cherry, S. Antimicrobial autophagy: A conserved innate immune response in Drosophila. J. Innate Immun. 2013, 5, 444–455. [Google Scholar] [CrossRef] [PubMed]
- Yasunaga, A.; Hanna, S.L.; Li, J.; Cho, H.; Rose, P.P.; Spiridigliozzi, A.; Gold, B.; Diamond, M.S.; Cherry, S. Genome-Wide RNAi Screen Identifies Broadly-Acting Host Factors That Inhibit Arbovirus Infection. PLoS Pathog. 2014, 10, e1003914. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.G.; Weaver, S.C.; Tesh, R.B.; Vasilakis, N. Insect-specific virus discovery: Significance for the arbovirus community. Viruses 2015, 7, 4911–4928. [Google Scholar] [CrossRef] [PubMed]
- Webster, C.L.; Waldron, F.M.; Robertson, S.; Crowson, D.; Ferrari, G.; Quintana, J.F.; Brouqui, J.-M.; Bayne, E.H.; Longdon, B.; Buck, A.H.; et al. The Discovery, Distribution, and Evolution of Viruses Associated with Drosophila melanogaster. PLoS Biol. 2015, 13, e1002210. [Google Scholar] [CrossRef] [PubMed]
- Webster, C.L.; Longdon, B.; Lewis, S.H.; Obbard, D.J. Twenty-Five New Viruses Associated with the Drosophilidae (Diptera). Evol. Bioinform. Online 2016, 12, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Medd, N.C.; Fellous, S.; Nakai, M.; Xuereb, A.; Cross, J.V.; Obbard, D.J. The virome of Drosophila suzukii, an invasive pest of soft fruit. bioRxiv 2017, 190322. [Google Scholar] [CrossRef]
- Li, H.; Li, W.X.; Ding, S.W. Induction and suppression of RNA silencing by an animal virus. Science 2002, 296, 1319–1321. [Google Scholar] [CrossRef] [PubMed]
- Van Rij, R.P.; Saleh, M.C.; Berry, B.; Foo, C.; Houk, A.; Antoniewski, C.; Andino, R. The RNA silencing endonuclease Argonaute 2 mediates specific antiviral immunity in Drosophila melanogaster. Genes Dev. 2006, 20, 2985–2995. [Google Scholar] [CrossRef] [PubMed]
- Nayak, A.; Berry, B.; Tassetto, M.; Kunitomi, M.; Acevedo, A.; Deng, C.; Krutchinsky, A.; Gross, J.; Antoniewski, C.; Andino, R. Cricket paralysis virus antagonizes Argonaute 2 to modulate antiviral defense in Drosophila. Nat. Struct. Mol. Biol. 2010, 17, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Bronkhorst, A.W.; Van Cleef, K.W.R.; Venselaar, H.; Van Rij, R.P. A dsRNA-binding protein of a complex invertebrate DNA virus suppresses the Drosophila RNAi response. Nucleic Acids Res. 2014, 42, 12237–12248. [Google Scholar] [CrossRef] [PubMed]
- Van Cleef, K.W.R.; Van Mierlo, J.T.; Miesen, P.; Overheul, G.J.; Fros, J.J.; Schuster, S.; Marklewitz, M.; Pijlman, G.P.; Junglen, S.; Van Rij, R.P. Mosquito and Drosophila entomobirnaviruses suppress dsRNA- and siRNA-induced RNAi. Nucleic Acids Res. 2014, 42, 8732–8744. [Google Scholar] [CrossRef] [PubMed]
- Van Mierlo, J.T.; Overheul, G.J.; Obadia, B.; van Cleef, K.W.R.; Webster, C.L.; Saleh, M.C.; Obbard, D.J.; van Rij, R.P. Novel Drosophila Viruses Encode Host-Specific Suppressors of RNAi. PLoS Pathog. 2014, 10, e1004256. [Google Scholar] [CrossRef] [PubMed]
- Csorba, T.; Kontra, L.; Burgyán, J. Viral silencing suppressors: Tools forged to fine-tune host-pathogen coexistence. Virology 2015, 479–480, 85–103. [Google Scholar] [CrossRef] [PubMed]
- Habayeb, M.S.; Ekström, J.O.; Hultmark, D. Nora virus persistent infections are not affected by the RNAi machinery. PLoS ONE 2009, 4, e5731. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Grant, G.; Sabin, L.R.; Gordesky-Gold, B.; Yasunaga, A.; Tudor, M.; Cherry, S. Transcriptional pausing controls a rapid antiviral innate immune response in Drosophila. Cell Host Microbe 2012, 12, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.W.; McGraw, E.A.; Ammar, E.-D.; Dietzgen, R.G.; Hogenhout, S.A. Drosophila melanogaster Mounts a Unique Immune Response to the Rhabdovirus Sigma virus. Appl. Environ. Microbiol. 2008, 74, 3251–3256. [Google Scholar] [CrossRef] [PubMed]
- Palmer, W.H.; Medd, N.; Beard, P.M.; Obbard, D.J. Isolation of a natural DNA virus of Drosophila melanogaster, and characterisation of host resistance and immune responses. bioRxiv 2017, 215111. [Google Scholar] [CrossRef]
- Ramirez, J.L.; Dimopoulos, G. The Toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev. Comp. Immunol. 2010, 34, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Fragkoudis, R.; Chi, Y.; Siu, R.W.C.; Barry, G.; Attarzadeh-Yazdi, G.; Merits, A.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Semliki Forest virus strongly reduces mosquito host defence signaling. Insect Mol. Biol. 2008, 17, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Waldock, J.; Olson, K.E.; Christophides, G.K. Anopheles gambiae Antiviral Immune Response to Systemic O’nyong-nyong Infection. PLoS Negl. Trop. Dis. 2012, 6, e1565. [Google Scholar] [CrossRef] [PubMed]
- Carissimo, G.; Pondeville, E.; McFarlane, M.; Dietrich, I.; Mitri, C.; Bischoff, E.; Antoniewski, C.; Bourgouin, C.; Failloux, A.-B.; Kohl, A.; et al. Antiviral immunity of Anopheles gambiae is highly compartmentalized, with distinct roles for RNA interference and gut microbiota. Proc. Natl. Acad. Sci. USA 2015, 112, E176–E185. [Google Scholar] [CrossRef] [PubMed]
- Mondotte, J.A.; Saleh, M.C. Antiviral Immune Response and the Route of Infection in Drosophila melanogaster. In Advances in Virus Research, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Prasad, A.N.; Brackney, D.E.; Ebel, G.D. The role of innate immunity in conditioning mosquito susceptibility to West Nile virus. Viruses 2013, 5, 3142–3170. [Google Scholar] [CrossRef] [PubMed]
- Colpitts, T.M.; Cox, J.; Vanlandingham, D.L.; Feitosa, F.M.; Cheng, G.; Kurscheid, S.; Wang, P.; Krishnan, M.N.; Higgs, S.; Fikrig, E. Alterations in the Aedes aegypti transcriptome during infection with west nile, dengue and yellow fever viruses. PLoS Pathog. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, R.; Zhang, B.; Zhao, T.; Wang, P.; Liang, G.; Cheng, G. Blood meal acquisition enhances arbovirus replication in mosquitoes through activation of the GABAergic system. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Settles, E.W.; Friesen, P.D. Flock House Virus Induces Apoptosis by Depletion of Drosophila Inhibitor-of-Apoptosis Protein DIAP1. J. Virol. 2008, 82, 1378–1388. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.M.; May, R.M. Coevolution of Hosts and Parasites. Parasitology 1982, 85, 411–426. [Google Scholar] [CrossRef] [PubMed]
- Flor, H.H. The complementary genetic system in flax and flax rust. Adv. Gen. 1956, 8, 29–54. [Google Scholar]
- Grosberg, R.K.; Hart, M.W. Mate selection and the evolution of highly polymorphic self/nonself recognition genes. Science 2000, 289, 2111–2114. [Google Scholar] [CrossRef] [PubMed]
- Sessions, O.M.; Barrows, N.J.; Souza-Neto, J.A.; Robinson, T.J.; Hershey, C.L.; Rodgers, M.A.; Ramirez, J.L.; Dimopoulos, G.; Yang, P.L.; Pearson, J.L.; et al. Discovery of insect and human dengue virus host factors. Nature 2009, 458, 1047–1050. [Google Scholar] [CrossRef] [PubMed]
- Longdon, B.; Jiggins, F.M. Vertically transmitted viral endosymbionts of insects: Do sigma viruses walk alone? Proc. R. Soc. B Biol. Sci. 2012, 279, 3889–3898. [Google Scholar] [CrossRef] [PubMed]
- Fleuriet, A. Presence of the Hereditary Rhabdovirus Sigma and Polymorphism for a Gene for Resistance to This Virus in Natural Populations of Drosophila Melanogaster. Evolution 1976, 30, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Wayne, M.L.; Contamine, D.; Kreitman, M. Molecular population genetics of ref(2)P, a locus which confers viral resistance in Drosophila. Mol. Biol. Evol. 1996, 13, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Avila, A.; Silverman, N.; Diaz-Meco, M.T.; Moscat, J. The Drosophila Atypical Protein Kinase C-Ref(2)P Complex Constitutes a Conserved Module for Signaling in the Toll Pathway. Mol. Cell. Biol. 2002, 22, 8787–8795. [Google Scholar] [CrossRef] [PubMed]
- Carré-Mlouka, A.; Gaumer, S.; Gay, P.; Petitjean, A.M.; Coulondre, C.; Dru, P.; Bras, F.; Dezélée, S.; Contamine, D. Control of sigma virus multiplication by the ref(2)P gene of Drosophila melanogaster: An in vivo study of the PB1 domain of Ref(2)P. Genetics 2007, 176, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Nezis, I.P.; Simonsen, A.; Sagona, A.P.; Finley, K.; Gaumer, S.; Contamine, D.; Rusten, T.E.; Stenmark, H.; Brech, A. Ref(2)P, the Drosophila melanogaster homologue of mammalian p62, is required for the formation of protein aggregates in adult brain. J. Cell Biol. 2008, 180, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Contamine, D.; Petitjean, A.M.; Ashburner, M. Genetic resistance to viral infection: The molecular cloning of a Drosophila gene that restricts infection by the rhabdovirus sigma. Genetics 1989, 123, 525–533. [Google Scholar] [PubMed]
- Gay, P. Drosophila genes which intervene in multiplication of sigma virus (author’s transl). Mol. Gen. Genet. 1978, 159, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Wilfert, L.; Jiggins, F.M. Disease association mapping in Drosophila can be replicated in the wild. Biol. Lett. 2010, 6, 666–668. [Google Scholar] [CrossRef] [PubMed]
- Magwire, M.M.; Fabian, D.K.; Schweyen, H.; Cao, C.; Longdon, B.; Bayer, F.; Jiggins, F.M. Genome-Wide Association Studies Reveal a Simple Genetic Basis of Resistance to Naturally Coevolving Viruses in Drosophila melanogaster. PLoS Genet. 2012, 8. [Google Scholar] [CrossRef] [PubMed]
- Magwire, M.M.; Bayer, F.; Webster, C.L.; Cao, C.; Jiggins, F.M. Successive increases in the resistance of Drosophila to viral infection through a transposon insertion followed by a duplication. PLoS Genet. 2011, 7, e1002337. [Google Scholar] [CrossRef] [PubMed]
- Cogni, R.; Cao, C.; Day, J.P.; Bridson, C.; Jiggins, F.M. The genetic architecture of resistance to virus infection in Drosophila. Mol. Ecol. 2016, 25, 5228–5241. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.E.; Faria, V.G.; Nolte, V.; Schlotterer, C.; Teixeira, L.; Sucena, E.; Magalhaes, S. Host adaptation to viruses relies on few genes with different cross-resistance properties. Proc. Natl. Acad. Sci. USA 2014, 111, 5938–5943. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Cogni, R.; Barbier, V.; Jiggins, F.M. Complex coding and regulatory polymorphisms in a restriction factor determine the susceptibility of Drosophila to viral infection. Genetics 2017, 206, 2159–2173. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Quillery, E.; Noël, V.; Richardson, J.H.; Jarman, R.G.; Thomas, W.; Chevillon, C.; Scott, T.W. Specificity of resistance to dengue virus isolates is associated with genotypes of the mosquito antiviral gene Dicer-2 Specificity of resistance to dengue virus isolates is associated with genotypes of the mosquito antiviral gene Dicer-2. Proc. R. Soc. B 2013, 280, 20122437. [Google Scholar] [CrossRef] [PubMed]
- Fansiri, T.; Fontaine, A.; Diancourt, L.; Caro, V.; Thaisomboonsuk, B.; Richardson, J.H.; Jarman, R.G.; Ponlawat, A.; Lambrechts, L. Genetic Mapping of Specific Interactions between Aedes aegypti Mosquitoes and Dengue Viruses. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.F.; Fulton, R.E.; Salasek, M.L.; Beaty, B.J.; Black, W.C., IV. Quantitative trait loci that control vector competence for dengue-2 virus in the mosquito Aedes aegypti. Genetics 2000, 156, 687–698. [Google Scholar] [PubMed]
- Graham, D.H.; Holmes, J.L.; Beaty, B.J.; Black, W.C., IV. Quantitative trait loci conditioning transovarial transmission of La Crosse virus in the eastern treehole mosquito, Ochlerotatus triseriatus. Insect Mol. Biol. 2003, 12, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Aminetzach, Y.T.; Macpherson, J.M.; Petrov, D.A. Evolution: Pesticide resistance via transposition-mediated adaptive gene truncation in Drosophila. Science 2005, 309, 764–767. [Google Scholar] [CrossRef] [PubMed]
- Lentz, T.L.; Burrage, T.G.; Smith, L.; Tignor, G.H. The acetylcholine receptor as a cellular receptor for rabies virus. Yale J. Biol. Med. 1983, 56, 315–322. [Google Scholar] [PubMed]
- Yu, J.H.; Yang, W.-H.; Gulick, T.; Bloch, K.D.; Bloch, D.B. Ge-1 is a central component of the mammalian cytoplasmic mRNA processing body. RNA 2005, 11, 1795–1802. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Eulalio, A.; Lingel, A.; Helms, S.; Conti, E.; Izaurralde, E. The C-terminal region of Ge-1 presents conserved structural features required for P-body localization. RNA 2008, 14, 1991–1998. [Google Scholar] [CrossRef] [PubMed]
- Brun, P.; Plus, N. The viruses of Drosophila. In The Genetics and Biology of Drosophila; Academic Press: London, UK, 1980; pp. 625–702. [Google Scholar]
- Bangham, J.; Knott, S.A.; Kim, K.W.; Young, R.S.; Jiggins, F.M. Genetic variation affecting host-parasite interactions: Major-effect quantitative trait loci affect the transmission of sigma virus in Drosophila melanogaster. Mol. Ecol. 2008, 17, 3800–3807. [Google Scholar] [CrossRef] [PubMed]
- Fricke, R.; Gohl, C.; Dharmalingam, E.; Grevelhörster, A.; Zahedi, B.; Harden, N.; Kessels, M.; Qualmann, B.; Bogdan, S. Drosophila Cip4/Toca-1 Integrates Membrane Trafficking and Actin Dynamics through WASP and SCAR/WAVE. Curr. Biol. 2009, 19, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.; Shackelton, L.A.; Holmes, E.C. Rates of evolutionary change in viruses: Patterns and determinants. Nat. Rev. Genet. 2008, 9, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Ebert, D. Host-parasite coevolution: Insights from the Daphnia-parasite model system. Curr. Opin. Microbiol. 2008, 11, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Obbard, D.J.; Dudas, G. The genetics of host-virus coevolution in invertebrates. Curr. Opin. Virol. 2014, 8, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Dru, P.; Bras, F.; Dezelee, S.; Gay, P.; Petitjean, A.M.; Pierre-Deneubourg, A.; Teninges, D.; Contamine, D. Unusual variability of the Drosophila melanogaster ref(2)P protein which controls the multiplication of sigma rhabdovirus. Genetics 1993, 133, 943–954. [Google Scholar] [PubMed]
- Fleuriet, A. Perpetuation of the hereditary sigma virus in populations of its host, Drosophila melanogaster. Geographical analysis of correlated polymorphisms. Genetica 1986, 70, 167–177. [Google Scholar] [CrossRef]
- Fleuriet, A.; Sperlich, D. Evolution of the Drosophila melanogaster-sigma virus system in a natural population from Tuebingen. Theor. Appl. Genet. 1992, 85, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Fleuriet, A.; Periquet, G. Evolution of the Drosophila melanogaster-sigma virus system in natural populations from Languedoc (southern France). Arch. Virol. 1993, 129, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Bangham, J.; Obbard, D.J.; Kim, K.-W.; Haddrill, P.R.; Jiggins, F.M. The age and evolution of an antiviral resistance mutation in Drosophila melanogaster. Proc. Biol. Sci. 2007, 274, 2027–2034. [Google Scholar] [CrossRef] [PubMed]
- Wilfert, L.; Jiggins, F.M. The dynamics of reciprocal selective sweeps of host resistance and a parasite counter-adaptation in Drosophila. Evolution 2013, 67, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Howick, V.M.; Lazzaro, B.P. Convergent Balancing Selection on an Antimicrobial Peptide in Drosophila. Curr. Biol. 2016, 26, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Lazzaro, B.P. The potential for adaptive maintenance of diversity in insect antimicrobial peptides. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371, 20150291. [Google Scholar] [CrossRef] [PubMed]
- Hain, D.; Bettencourt, B.R.; Okamura, K.; Csorba, T.; Meyer, W.; Jin, Z.; Biggerstaff, J.; Siomi, H.; Hutvagner, G.; Lai, E.C.; et al. Natural Variation of the Amino-Terminal Glutamine-Rich Domain in Drosophila Argonaute2 Is Not Associated with Developmental Defects. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Palmer, W.H.; Obbard, D.J. Variation and Evolution in the Glutamine-Rich Repeat Region of Drosophila Argonaute-2. G3 Genes Genomes Genet. 2016, 6, 2563–2572. [Google Scholar] [CrossRef] [PubMed]
- Obbard, D.J.; Jiggins, F.M.; Halligan, D.L.; Little, T.J. Natural selection drives extremely rapid evolution in antiviral RNAi genes. Curr. Biol. 2006, 16, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Obbard, D.J.; Welch, J.J.; Kim, K.W.; Jiggins, F.M. Quantifying adaptive evolution in the Drosophila immune system. PLoS Genet. 2009, 5, e1000698. [Google Scholar] [CrossRef] [PubMed]
- Palmer, W.H.; Hadfield, J.D.; Obbard, D.J. RNA Interference Pathways Display High Rates of Adaptive Protein Evolution in Multiple Invertebrates. Genetics 2018. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowski, B.; Hupalo, D.N.; Kern, A.D. Recurrent adaptation in RNA interference genes across the Drosophila phylogeny. Mol. Biol. Evol. 2011, 28, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Obbard, D.J.; Jiggins, F.M.; Bradshaw, N.J.; Little, T.J. Recent and recurrent selective sweeps of the antiviral RNAi gene argonaute-2 in three species of Drosophila. Mol. Biol. Evol. 2011, 28, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.H.; Webster, C.L.; Salmela, H.; Obbard, D.J. Repeated duplication of Argonaute2 is associated with strong selection and testis specialization in Drosophila. Genetics 2016, 204, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Schlenke, T.A.; Begun, D.J. Natural selection drives Drosophila immune system evolution. Genetics 2003, 164, 1471–1480. [Google Scholar] [PubMed]
- Sackton, T.B.; Lazzaro, B.P.; Schlenke, T.A.; Evans, J.D.; Hultmark, D.; Clark, A.G. Dynamic evolution of the innate immune system in Drosophila. Nat. Genet. 2007, 39, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Qin, S.; Song, X.; Li, Y.; Jin, P.; Chen, L.; Ma, F. Evolutionary rate patterns of genes involved in the Drosophila Toll and Imd signaling pathway. BMC Evol. Biol. 2013, 13, 245. [Google Scholar] [CrossRef] [PubMed]
- Juneja, P.; Lazzaro, B.P. Haplotype structure and expression divergence at the Drosophila cellular immune gene eater. Mol. Biol. Evol. 2010, 27, 2284–2299. [Google Scholar] [CrossRef] [PubMed]
- Nene, V.; Wortman, J.R.; Lawson, D.; Haas, B.; Kodira, C.; Tu, Z.; Loftus, B.; Xi, Z.; Megy, K.; Grabherr, M.; et al. Genome sequence of Aedes aegypti, a major arbovirus vector. Science 2007, 316, 1718–1723. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.; Beaty, B.; Black, W.C. Quantitative genetics of vector competence for dengue-2 virus in Aedes aegypti. Am. J. Trop. Med. Hyg. 1998, 59, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.E.; Beaty, B.J.; Black, W.C. Selection of D2S3, an Aedes aegypti (Diptera: Culicidae) Strain with High Oral Susceptibility to Dengue 2 Virus and D2MEB, a Strain with a Midgut Barrier to Dengue 2 Escape. J. Med. Entomol. 2005, 42, 110–119. [Google Scholar] [CrossRef]
- Gomez-Machorro, C.; Bennett, K.E.; Munoz, M.D.L.; Black, W.C., IV. Quantitative trait loci affecting dengue midgut infection barriers in an advanced intercross line of Aedes aegypti. Insect Mol. Biol. 2004, 13, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Noriega, F.G.; Pennington, J.E.; Barillas-Mury, C.; Wang, X.Y.; Wells, M.A. Aedes aegypti midgut early trypsin is post-transcriptionally regulated by blood feeding. Insect Mol. Biol. 1996, 5, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Gorrochotegui-Escalante, N.; Lozano-Fuentes, S.; Bennett, K.E.; Molina-Cruz, A.; Beaty, B.J.; Black, W.C., IV. Association mapping of segregating sites in the early trypsin gene and susceptibility to dengue-2 virus in the mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2005, 35, 771–788. [Google Scholar] [CrossRef] [PubMed]
- Molina-Cruz, A.; Gupta, L.; Richardson, J.; Bennett, K.; Black, W.C., IV; Barillas-Mury, C. Effect of mosquito midgut trypsin activity on dengue-2 virus infection and dissemination in Aedes aegypti. Am. J. Trop. Med. Hyg. 2005, 72, 631–637. [Google Scholar] [PubMed]
- Brackney, D.E.; Foy, B.D.; Olson, K.E. The effects of midgut serine proteases on dengue virus type 2 infectivity of Aedes aegypti. Am. J. Trop. Med. Hyg. 2008, 79, 267–274. [Google Scholar] [PubMed]
- Barón, O.L.; Ursic-Bedoya, R.J.; Lowenberger, C.A.; Ocampo, C.B. Differential Gene Expression from Midguts of Refractory and Susceptible Lines of the Mosquito, Aedes aegypti, Infected with Dengue-2 Virus. J. Insect Sci. 2010, 10, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.R.; Schneider, J.R.; Grimstad, P.R.; Severson, D.W. Quantitative genetics of vector competence for La Crosse virus and body size in Ochlerotatus hendersoni and Ochlerotatus triseriatus interspecific hybrids. Genetics 2005, 169, 1529–1539. [Google Scholar] [CrossRef] [PubMed]
- Borucki, M.K.; Kempf, B.J.; Blitvich, B.J.; Blair, C.D.; Beaty, B.J. La Crosse virus: Replication in vertebrate and invertebrate hosts. Microbes Infect. 2002, 4, 341–350. [Google Scholar] [CrossRef]
- Tran, K.T.; Vazeille-Falcoz, M.; Mousson, L.; Tran, H.H.; Rodhain, F.; Ngugen, T.H.; Failloux, A.B. Aedes aegypti in Ho Chi Minh City (Viet Nam): Susceptibility to dengue 2 virus and genetic differentiation. Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 581–586. [Google Scholar] [PubMed]
- Mousson, L.; Vazeille, M.; Chawprom, S.; Prajakwong, S.; Rodhain, F.; Failloux, A.-B. Genetic structure of Aedes aegypti populations in Chiang Mai (Thailand) and relation with dengue transmission. Trop. Med. Int. Health 2002, 7, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, S.A.; Simmons, M.P.; Olson, K.E.; Beaty, B.J.; Blair, C.D.; Black, W.C. Rapid Intraspecific Evolution of miRNA and siRNA Genes in the Mosquito Aedes aegypti. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Halbach, R.; Junglen, S.; van Rij, R.P. Mosquito-specific and mosquito-borne viruses: Evolution, infection, and host defense. Curr. Opin. Insect Sci. 2017, 22, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Behura, S.K.; Gomez-Machorro, C.; Harker, B.W.; deBruyn, B.; Lovin, D.D.; Hemme, R.R.; Mori, A.; Romero-Severson, J.; Severson, D.W. Global cross-talk of genes of the mosquito Aedes aegypti in response to dengue virus infection. PLoS Negl. Trop. Dis. 2011, 5. [Google Scholar] [CrossRef] [PubMed]
- Ocampo, C.B.; Caicedo, P.A.; Jaramillo, G.; Ursic Bedoya, R.; Baron, O.; Serrato, I.M.; Cooper, D.M.; Lowenberger, C. Differential Expression of Apoptosis Related Genes in Selected Strains of Aedes aegypti with Different Susceptibilities to Dengue Virus. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Clem, R.J. Arboviruses and apoptosis: The role of cell death in determining vector competence. J. Gen. Virol. 2016, 97, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, R.; Scott, T.W. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 2006, 11, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Girard, Y.A.; Schneider, B.S.; McGee, C.E.; Wen, J.; Han, V.C.; Popov, V.; Mason, P.W.; Higgs, S. Salivary gland morphology and virus transmission during long-term cytopathologic West Nile virus infection in Culex mosquitoes. Am. J. Trop. Med. Hyg. 2007, 76, 118–128. [Google Scholar] [PubMed]
- Mercado-Curiel, R.F.; Black, W.C., IV; Mũoz, M.D.L. A dengue receptor as possible genetic marker of vector competence in Aedes aegypti. BMC Microbiol. 2008, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Jupatanakul, N.; Ramirez, J.L.; Kang, S.; Romero-Vivas, C.M.; Mohammed, H.; Dimopoulos, G. Transcriptomic Profiling of Diverse Aedes aegypti Strains Reveals Increased Basal-level Immune Activation in Dengue Virus-refractory Populations and Identifies Novel Virus-vector Molecular Interactions. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Angleró-Rodríguez, Y.I.; Talyuli, O.A.; Blumberg, B.J.; Kang, S.; Demby, C.; Shields, A.; Carlson, J.; Jupatanakul, N.; Dimopoulos, G. An Aedes aegypti-associated fungus increases susceptibility to dengue virus by modulating gut trypsin activity. eLife 2017, 6, e28844. [Google Scholar] [CrossRef] [PubMed]
- Nishi, T.; Forgac, M. The vacuolar (H+)-ATPases—Nature’s most versatile proton pumps. Nat. Rev. Mol. Cell Biol. 2002, 3, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Smit, J.M.; Moesker, B.; Rodenhuis-Zybert, I.; Wilschut, J. Flavivirus cell entry and membrane fusion. Viruses 2011, 3, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Wolbachia and virus protection in insects. Science 2008, 322, 702. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Ferreira, Á.; Ashburner, M. The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. PLoS Biol. 2008, 6, 2753–2763. [Google Scholar] [CrossRef] [PubMed]
- Serbus, L.R.; Casper-Lindley, C.; Landmann, F.; Sullivan, W. The Genetics and Cell Biology of Wolbachia-Host Interactions. Annu. Rev. Genet. 2008, 42, 683–707. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.E.; Leong, Y.S.; O’Neill, S.L.; Johnson, K.N. Variation in antiviral protection mediated by different Wolbachia strains in Drosophila simulans. PLoS Pathog. 2009, 5, e1000656. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Bian, G.; Pan, X.; Xi, Z. Wolbachia induces density-dependent inhibition to dengue virus in mosquito cells. PLoS Negl. Trop. Dis. 2012, 6, e1754. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.; Longdon, B.; Bauer, S.; Chan, Y.S.; Miller, W.J.; Bourtzis, K.; Teixeira, L.; Jiggins, F.M. Symbionts Commonly Provide Broad Spectrum Resistance to Viruses in Insects: A Comparative Analysis of Wolbachia Strains. PLoS Pathog. 2014, 10, e1004369. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.; Tolosana, I.; Ok, S.; Smith, S.; Snoeck, K.; Day, J.P.; Jiggins, F.M. Symbiont strain is the main determinant of variation in Wolbachia-mediated protection against viruses across Drosophila species. Mol. Ecol. 2017, 26, 4072–4084. [Google Scholar] [CrossRef] [PubMed]
- Terradas, G.; McGraw, E.A. Wolbachia-mediated virus blocking in the mosquito vector Aedes aegypti. Curr. Opin. Insect Sci. 2017, 22, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Caragata, E.P.; Rancès, E.; Hedges, L.M.; Gofton, A.W.; Johnson, K.N.; O’Neill, S.L.; McGraw, E.A. Dietary Cholesterol Modulates Pathogen Blocking by Wolbachia. PLoS Pathog. 2013, 9, e1003459. [Google Scholar] [CrossRef] [PubMed]
- Caragata, E.P.; Rezende, F.O.; Simões, T.C.; Moreira, L.A. Diet-Induced Nutritional Stress and Pathogen Interference in Wolbachia-Infected Aedes aegypti. PLoS Negl. Trop. Dis. 2016, 10, e0005158. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Hussain, M.; O’Neill, S.L.; Asgari, S. Wolbachia uses a host microRNA to regulate transcripts of a methyltransferase, contributing to dengue virus inhibition in Aedes aegypti. Proc. Natl. Acad. Sci. USA 2013, 110, 10276–10281. [Google Scholar] [CrossRef] [PubMed]
- Wong, Z.S.; Brownlie, J.C.; Johnson, K.N. Oxidative stress correlates with Wolbachia-mediated antiviral protection in Wolbachia-Drosophila associations. Appl. Environ. Microbiol. 2015, 81, 3001–3005. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, T.; Newton, I.L.G.; Hardy, R.W. Wolbachia elevates host methyltransferase expression to block an RNA virus early during infection. PLoS Pathog. 2017, 13, e1006427. [Google Scholar] [CrossRef] [PubMed]
- Terradas, G.; Joubert, D.A.; McGraw, E.A. The RNAi pathway plays a small part in Wolbachia-mediated blocking of dengue virus in mosquito cells. Sci. Rep. 2017, 7, 43847. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Yamada, R.; O’Neill, S.L.; Johnson, K.N. The small interfering RNA pathway is not essential for wolbachia-mediated antiviral protection in Drosophila melanogaster. Appl. Environ. Microbiol. 2012, 78, 6773–6776. [Google Scholar] [CrossRef] [PubMed]
- Rancès, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; O’Neill, S.L. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLoS Pathog. 2012, 8. [Google Scholar] [CrossRef] [PubMed]
- Rances, E.; Johnson, T.K.; Popovici, J.; Iturbe-Ormaetxe, I.; Zakir, T.; Warr, C.G.; O’Neill, S.L. The Toll and Imd Pathways Are Not Required for Wolbachia-Mediated Dengue Virus Interference. J. Virol. 2013, 87, 11945–11949. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Marialva, M.S.P.; Yamada, R.; O’Neill, S.L.; Teixeira, L. High anti-viral protection without immune upregulation after interspecies Wolbachia transfer. PLoS ONE 2014, 9, e99025. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Marialva, M.S.P.; Esteves, S.S.; Weinert, L.A.; Martinez, J.; Jiggins, F.M.; Teixeira, L. Wolbachia Variants Induce Differential Protection to Viruses in Drosophila melanogaster: A Phenotypic and Phylogenomic Analysis. PLoS Genet. 2013, 9, e1003896. [Google Scholar] [CrossRef] [PubMed]
- Min, K.-T.; Benzer, S. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc. Natl. Acad. Sci. USA 1997, 94, 10792–10796. [Google Scholar] [CrossRef] [PubMed]
- Woolfit, M.; Iturbe-Ormaetxe, I.; Brownlie, J.C.; Walker, T.; Riegler, M.; Seleznev, A.; Popovici, J.; Rancès, E.; Wee, B.A.; Pavlides, J.; et al. Genomic evolution of the pathogenic Wolbachia strain, wMelPop. Genome Biol. Evol. 2013, 5, 2189–2204. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Teixeira, L. Mutualism Breakdown by Amplification of Wolbachia Genes. PLoS Biol. 2015, 13, e1002065. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Teixeira, L. Within host selection for faster replicating bacterial symbionts. bioRxiv 2017, 222240. [Google Scholar] [CrossRef]
- Rohrscheib, C.E.; Frentiu, F.D.; Horn, E.; Ritchie, F.K.; van Swinderen, B.; Weible, M.W.; O’Neill, S.L.; Brownlie, J.C. Intensity of Mutualism Breakdown Is Determined by Temperature Not Amplification of Wolbachia Genes. PLoS Pathog. 2016, 12, e1005888. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Lane, A.M.; Fong, A.W.C.; Voronin, D.A.; Iturbe-Ormaetxe, I.; Yamada, R.; McGraw, E.A.; O’Neill, S.L. Host adaptation of a Wolbachia strain after long-term serial passage in mosquito cell lines. Appl. Environ. Microbiol. 2008, 74, 6963–6969. [Google Scholar] [CrossRef] [PubMed]
- Engelstädter, J.; Hurst, G.D.D. The Ecology and Evolution of Microbes that Manipulate Host Reproduction. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 127–149. [Google Scholar] [CrossRef]
- Martinez, J.; Ok, S.; Smith, S.; Snoeck, K.; Day, J.P.; Jiggins, F.M. Should Symbionts Be Nice or Selfish? Antiviral Effects of Wolbachia Are Costly but Reproductive Parasitism Is Not. PLoS Pathog. 2015, 11, e1005021. [Google Scholar] [CrossRef] [PubMed]
- Riegler, M.; Sidhu, M.; Miller, W.J.; O’Neill, S.L. Evidence for a global Wolbachia replacement in Drosophila melanogaster. Curr. Biol. 2005, 15, 1428–1433. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.D.S.; Nolte, V.; Schlötterer, C. Nonrandom Wolbachia infection status of Drosophila melanogaster strains with different mtDNA haplotypes. Mol. Biol. Evol. 2008, 25, 2493–2498. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.F.; Weinert, L.A.; Welch, J.J.; Linheiro, R.S.; Magwire, M.M.; Jiggins, F.M.; Bergman, C.M. Population Genomics of the Wolbachia Endosymbiont in Drosophila melanogaster. PLoS Genet. 2012, 8, e1003129. [Google Scholar] [CrossRef] [PubMed]
- Turelli, M.; Hoffmann, A.A. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 1991, 353, 440–442. [Google Scholar] [CrossRef] [PubMed]
- James, A.C.; Ballard, J.W.O. Expression of cytoplasmic incompatibility in Drosophila simulans and its impact on infection frequencies and distribution of Wolbachia pipientis. Evolution 2000, 54, 1661–1672. [Google Scholar] [CrossRef] [PubMed]
- Kriesner, P.; Hoffmann, A.A.; Lee, S.F.; Turelli, M.; Weeks, A.R. Rapid Sequential Spread of Two Wolbachia Variants in Drosophila simulans. PLoS Pathog. 2013, 9, e1003607. [Google Scholar] [CrossRef] [PubMed]
- Mousson, L.; Zouache, K.; Arias-Goeta, C.; Raquin, V.; Mavingui, P.; Failloux, A.-B. The Native Wolbachia Symbionts Limit Transmission of Dengue Virus in Aedes albopictus. PLoS Negl. Trop. Dis. 2012, 6, e1989. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A Wolbachia Symbiont in Aedes aegypti Limits Infection with Dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Jiggins, F.M. The spread of Wolbachia through mosquito populations. PLoS Biol. 2017, 15, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Montgomery, B.L.; Popovici, J.; Iturbe-Ormaetxe, I.; Johnson, P.H.; Muzzi, F.; Greenfield, M.; Durkan, M.; Leong, Y.S.; Dong, Y.; et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 2011, 476, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Iturbe-Ormaetxe, I.; Callahan, A.G.; Phillips, B.L.; Billington, K.; Axford, J.K.; Montgomery, B.; Turley, A.P.; O’Neill, S.L. Stability of the wMel Wolbachia Infection following Invasion into Aedes aegypti Populations. PLoS Negl. Trop. Dis. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Carrington, L.B.; Tran, B.C.N.; Le, N.T.H.; Luong, T.T.H.; Nguyen, T.T.; Nguyen, P.T.; Nguyen, C.V.V.; Nguyen, H.T.C.; Vu, T.T.; Vo, L.T.; et al. Field- and clinically derived estimates of Wolbachia -mediated blocking of dengue virus transmission potential in Aedes aegypti mosquitoes. Proc. Natl. Acad. Sci. USA 2018, 115, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.M.; Pfeiffer, J.K. Viruses and the Microbiota. Annu. Rev. Virol. 2014, 1, 55–69. [Google Scholar] [CrossRef] [PubMed]
- Jakubowska, A.K.; Vogel, H.; Herrero, S. Increase in Gut Microbiota after Immune Suppression in Baculovirus-infected Larvae. PLoS Pathog. 2013, 9, e1003379. [Google Scholar] [CrossRef] [PubMed]
- Sansone, C.; Cohen, J.; Gold, B.; Zhu, W.; Misic, A.M.; Beiting, D.P.; Winter, S.E.; Cherry, S. Aging-associated dysbiosis increases susceptibility to enteric viral infection in Drosophila. bioRxiv 2017, 156455. [Google Scholar] [CrossRef]
- Buchon, N.; Broderick, N.A.; Chakrabarti, S.; Lemaitre, B. Invasive and indigenous microbiota impact intestinal stem cell activity through multiple pathways in Drosophila. Genes Dev. 2009, 23, 2333–2344. [Google Scholar] [CrossRef] [PubMed]
- Corby-Harris, V.; Pontaroli, A.C.; Shimkets, L.J.; Bennetzen, J.L.; Habel, K.E.; Promislow, D.E.L. Geographical distribution and diversity of bacteria associated with natural populations of Drosophila melanogaster. Appl. Environ. Microbiol. 2007, 73, 3470–3479. [Google Scholar] [CrossRef] [PubMed]
- Chandler, J.A.; Lang, J.; Bhatnagar, S.; Eisen, J.A.; Kopp, A. Bacterial communities of diverse Drosophila species: Ecological context of a host-microbe model system. PLoS Genet. 2011, 7, e1002272. [Google Scholar] [CrossRef] [PubMed]
- Van Tol, S.; Dimopoulos, G. Influences of the Mosquito Microbiota on Vector Competence. Adv. Insect Physiol. 2016, 51, 243–291. [Google Scholar]
- Ramirez, J.L.; Souza-Neto, J.; Cosme, R.T.; Rovira, J.; Ortiz, A.; Pascale, J.M.; Dimopoulos, G. Reciprocal tripartite interactions between the Aedes aegypti midgut microbiota, innate immune system and dengue virus influences vector competence. PLoS Negl. Trop. Dis. 2012, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Short, S.M.; Bahia, A.C.; Saraiva, R.G.; Dong, Y.; Kang, S.; Tripathi, A.; Mlambo, G.; Dimopoulos, G. Chromobacterium Csp_P Reduces Malaria and Dengue Infection in Vector Mosquitoes and Has Entomopathogenic and In Vitro Anti-pathogen Activities. PLoS Pathog. 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- Apte-Deshpande, A.; Paingankar, M.; Gokhale, M.D.; Deobagkar, D.N. Serratia odorifera a midgut inhabitant of Aedes aegypti mosquito enhances its susceptibility to dengue-2 virus. PLoS ONE 2012, 7, e40401. [Google Scholar] [CrossRef] [PubMed]
- Apte-Deshpande, A.D.; Paingankar, M.S.; Gokhale, M.D.; Deobagkar, D.N. Serratia odorifera mediated enhancement in susceptibility of Aedes aegypti for chikungunya virus. Indian J. Med. Res. 2014, 139, 762–768. [Google Scholar] [PubMed]
- Muturi, E.J.; Bara, J.J.; Rooney, A.P.; Hansen, A.K. Midgut fungal and bacterial microbiota of Aedes triseriatus and Aedes japonicus shift in response to La Crosse virus infection. Mol. Ecol. 2016, 25, 4075–4090. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.D.; Nogueira, J.R.; Bales, A.A.; Pittman, K.E.; Anderson, J.R. Interactions Between La Crosse Virus and Bacteria Isolated From the Digestive Tract of Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2011, 48, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Shirasu-Hiza, M.M.; Dionne, M.S.; Pham, L.N.; Ayres, J.S.; Schneider, D.S. Interactions between circadian rhythm and immunity in Drosophila melanogaster. Curr. Biol. 2007, 17, R353–R355. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Edery, I. Circadian Regulation in the Ability of Drosophila to Combat Pathogenic Infections. Curr. Biol. 2008, 18, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Stone, E.F.; Fulton, B.O.; Ayres, J.S.; Pham, L.N.; Ziauddin, J.; Shirasu-Hiza, M.M. The circadian clock protein timeless regulates phagocytosis of bacteria in Drosophila. PLoS Pathog. 2012, 8, e1002445. [Google Scholar] [CrossRef] [PubMed]
- Siva-Jothy, M.T.; Thompson, J.J.W. Short-term nutrient deprivation affects immune function. Physiol. Entomol. 2002, 27, 206–212. [Google Scholar] [CrossRef]
- Fellous, S.; Lazzaro, B.P. Larval food quality affects adult (but not larval) immune gene expression independent of effects on general condition. Mol. Ecol. 2010, 19, 1462–1468. [Google Scholar] [CrossRef] [PubMed]
- Povey, S.; Cotter, S.C.; Simpson, S.J.; Wilson, K. Dynamics of macronutrient self-medication and illness-induced anorexia in virally infected insects. J. Anim. Ecol. 2014, 83, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Rottschaefer, S.M.; Lazzaro, B.P. The Complex Contributions of Genetics and Nutrition to Immunity in Drosophila melanogaster. PLoS Genet. 2015, 11, e1005030. [Google Scholar] [CrossRef] [PubMed]
- Muturi, E.J.; Kim, C.H.; Alto, B.W.; Berenbaum, M.R.; Schuler, M.A. Larval environmental stress alters Aedes aegypti competence for Sindbis virus. Trop. Med. Int. Health 2011, 16, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Horne, K.M.E.; Vanlandingham, D.L. Bunyavirus-vector interactions. Viruses 2014, 6, 4373–4397. [Google Scholar] [CrossRef] [PubMed]
- Alto, B.W.; Reiskind, M.H.; Lounibos, L.P. Size Alters Susceptibility of Vectors to Dengue Virus Infection and Dissemination. Am. J. Trop. Med. Hyg. 2008, 79, 688–695. [Google Scholar] [PubMed]
- Schneider, J.R.; Mori, A.; Romero-Severson, J.; Chadee, D.D.; Severson, D.W. Investigations of dengue-2 susceptibility and body size among Aedes aegypti populations. Med. Vet. Entomol. 2007, 21, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Bara, J.J.; Parker, A.T.; Muturi, E.J. Comparative Susceptibility of Ochlerotatus japonicus, Ochlerotatus triseriatus, Aedes albopictus, and Aedes aegypti (Diptera: Culicidae) to La crosse virus. J. Med. Entomol. 2016, 53, 1415–1421. [Google Scholar] [CrossRef] [PubMed]
- Alto, B.W.; Lounibos, L.P.; Mores, C.N.; Reiskind, M.H. Larval competition alters susceptibility of adult Aedes mosquitoes to dengue infection. Proc. R. Soc. B Biol. Sci. 2008, 275, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.S.; Alcalay, Y.; Lovin, D.D.; Cunningham, J.M.; Eng, M.W.; Chadee, D.D.; Severson, D.W. Larval stress alters dengue virus susceptibility in Aedes aegypti (L.) adult females. Acta Trop. 2017, 174, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Kay, B.H.; Fanning, I.D.; Mottram, P. Rearing temperature influences flavivirus vector competence of mosquitoes. Med. Vet. Entomol. 1989, 3, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Dohm, D.J.; O’Guinn, M.L.; Turell, M.J. Effect of environmental temperature on the ability of Culex pipiens (Diptera: Culicidae) to transmit West Nile virus. J. Med. Entomol. 2002, 39, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.L.; Mores, C.N.; Lord, C.C.; Tabachnick, W.J. Impact of Extrinsic Incubation Temperature and Virus Exposure on Vector Competence of Culex pipiens quinquefasciatus Say (Diptera: Culicidae) for West Nile Virus. Vector-Borne Zoonotic Dis. 2007, 7, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, A.M.; Meola, M.A.; Moudy, R.M.; Kramer, L.D. Temperature, viral genetics, and the transmission of West Nile virus by Culex pipiens mosquitoes. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- Rohani, A.; Wong, Y.C.; Zamre, I.; Lee, H.L.; Zurainee, M.N. The effect of extrinsic incubation temperature on development of dengue serotype 2 and 4 viruses in Aedes aegypti (L.). Southeast Asian J. Trop. Med. Public Health 2009, 40, 942–950. [Google Scholar] [PubMed]
- Xiao, F.-Z.; Zhang, Y.; Deng, Y.-Q.; He, S.; Xie, H.-G.; Zhou, X.-N.; Yan, Y.-S. The effect of temperature on the extrinsic incubation period and infection rate of dengue virus serotype 2 infection in Aedes albopictus. Arch. Virol. 2014, 159, 3053–3057. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.D.; Hardy, J.L.; Presser, S.B. Effect of temperature of extrinsic incubation on the vector competence of Culex tarsalis for western equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1983, 32, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Westbrook, C.J.; Reiskind, M.H.; Pesko, K.N.; Greene, K.E.; Lounibos, L.P. Larval environmental temperature and the susceptibility of Aedes albopictus Skuse (Diptera: Culicidae) to Chikungunya virus. Vector Borne Zoonotic Dis. 2010, 10, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Adelman, Z.N.; Anderson, M.A.E.; Wiley, M.R.; Murreddu, M.G.; Samuel, G.H.; Morazzani, E.M.; Myles, K.M. Cooler Temperatures Destabilize RNA Interference and Increase Susceptibility of Disease Vector Mosquitoes to Viral Infection. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Samuel, G.H.; Adelman, Z.N.; Myles, K.M. Temperature-dependent effects on the replication and transmission of arthropod-borne viruses in their insect hosts. Curr. Opin. Insect Sci. 2016, 16, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Paaijmans, K.P.; Fansiri, T.; Carrington, L.B.; Kramer, L.D.; Thomas, M.B.; Scott, T.W. Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2011, 108, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Carrington, L.B.; Armijos, M.V.; Lambrechts, L.; Scott, T.W. Fluctuations at a Low Mean Temperature Accelerate Dengue Virus Transmission by Aedes aegypti. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Carrington, L.B.; Seifert, S.N.; Armijos, M.V.; Lambrechts, L.; Scott, T.W. Reduction of Aedes aegypti vector competence for dengue virus under large temperature fluctuations. Am. J. Trop. Med. Hyg. 2013, 88, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Zuk, M. The Sicker Sex. PLoS Pathog. 2009, 5, e1000267. [Google Scholar] [CrossRef] [PubMed]
- Duneau, D.F.; Kondolf, H.C.; Im, J.H.; Ortiz, G.A.; Chow, C.; Fox, M.A.; Eugénio, A.T.; Revah, J.; Buchon, N.; Lazzaro, B.P. The Toll pathway underlies host sexual dimorphism in resistance to both Gram-negative and Gram-positive bacteria in mated Drosophila. BMC Biol. 2017, 15, 124. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Stewart, C.O.; Rund, S.S.C.; Monteith, K.; Vale, P.F. Costs and benefits of sublethal Drosophila C virus infection. J. Evol. Biol. 2017, 30, 1325–1335. [Google Scholar] [CrossRef] [PubMed]
- Rolff, J. Bateman’s principle and immunity. Proc. R. Soc. B Biol. Sci. 2002, 269, 867–872. [Google Scholar] [CrossRef] [PubMed]
- Zuk, M.; Stoehr, A.M. Immune Defense and Host Life History. Am. Nat. 2002, 160, S9–S22. [Google Scholar] [CrossRef] [PubMed]
- Stoehr, A.M.; Kokko, H. Sexual dimorphism in immunocompetence: What does life-history theory predict? Behav. Ecol. 2006, 17, 751–756. [Google Scholar] [CrossRef]
- Nunn, C.L.; Lindenfors, P.; Pursall, E.R.; Rolff, J. On sexual dimorphism in immune function. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Regan, J.C.; Khericha, M.; Dobson, A.J.; Bolukbasi, E.; Rattanavirotkul, N.; Partridge, L. Sex difference in pathology of the ageing gut mediates the greater response of female lifespan to dietary restriction. eLife 2016, 5, e10956. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zipperlen, P.; Kubli, E. Drosophila Sex-Peptide Stimulates Female Innate Immune System after Mating via the Toll and Imd Pathways. Curr. Biol. 2005, 15, 1690–1694. [Google Scholar] [CrossRef] [PubMed]
- Fedorka, K.M.; Linder, J.E.; Winterhalter, W.; Promislow, D. Post-mating disparity between potential and realized immune response in Drosophila melanogaster. Proc. R. Soc. B Biol. Sci. 2007, 274, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Short, S.M.; Lazzaro, B.P. Female and male genetic contributions to post-mating immune defence in female Drosophila melanogaster. Proc. R. Soc. B Biol. Sci. 2010, 277, 3649–3657. [Google Scholar] [CrossRef] [PubMed]
- Schwenke, R.A.; Lazzaro, B.P. Juvenile Hormone Suppresses Resistance to Infection in Mated Female Drosophila melanogaster. Curr. Biol. 2017, 27, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Lawniczak, M.K.N.; Barnes, A.I.; Linklater, J.R.; Boone, J.M.; Wigby, S.; Chapman, T. Mating and immunity in invertebrates. Trends Ecol. Evol. 2007, 22, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Rolff, J.; Siva-Jothy, M.T. Copulation corrupts immunity: A mechanism for a cost of mating in insects. Proc. Natl. Acad. Sci. USA 2002, 99, 9916–9918. [Google Scholar] [CrossRef] [PubMed]
- Flatt, T.; Heyland, A.; Rus, F.; Porpiglia, E.; Sherlock, C.; Yamamoto, R.; Garbuzov, A.; Palli, S.R.; Tatar, M.; Silverman, N. Hormonal regulation of the humoral innate immune response in Drosophila melanogaster. J. Exp. Biol. 2008, 211, 2712–2724. [Google Scholar] [CrossRef] [PubMed]
- Flatt, T.; Tu, M.-P.; Tatar, M. Hormonal pleiotropy and the juvenile hormone regulation of Drosophila development and life history. BioEssays 2005, 27, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Moshitzky, P.; Fleischmann, I.; Chaimov, N.; Saudan, P.; Klauser, S.; Kubli, E.; Applebaum, S.W. Sex-peptide activates juvenile hormone biosynthesis in the Drosophila melanogaster corpus allatum. Arch. Insect Biochem. Physiol. 1996, 32, 363–374. [Google Scholar] [CrossRef]
- Burand, J.P.; Kim, W.; Afonso, C.L.; Tulman, E.R.; Kutish, G.F.; Lu, Z.; Rock, D.L. Analysis of the genome of the sexually transmitted insect virus Helicoverpa zea nudivirus 2. Viruses 2012, 4, 28–61. [Google Scholar] [CrossRef] [PubMed]
- Takatsuka, J.; Nakai, M.; Shinoda, T. A virus carries a gene encoding juvenile hormone acid methyltransferase, a key regulatory enzyme in insect metamorphosis. Sci. Rep. 2017, 7, 13522. [Google Scholar] [CrossRef] [PubMed]
- Palmer, W.H.; Medd, N.; Obbard, D.J. University of Edinburgh, Edinburgh, UK. PCR survey for viruses in Drosophila melanogaster. Unpublished work. 2016. [Google Scholar]
- Chen, Y.; Zhao, Y.; Hammond, J.; Hsu, H.T.; Evans, J.; Feldlaufer, M. Multiple virus infections in the honey bee and genome divergence of honey bee viruses. J. Invertebr. Pathol. 2004, 87, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Washburn, J.O.; Haas-Stapleton, E.J.; Tan, F.F.; Beckage, N.E.; Volkman, L.E. Co-infection of Manduca sexta larvae with polydnavirus from Cotesia congregata increases susceptibility to fatal infection by Autographa californica M Nucleopolyhedrovirus. J. Insect Physiol. 2000, 46, 179–190. [Google Scholar] [CrossRef]
- Berry, B.; Deddouche, S.; Kirschner, D.; Imler, J.-L.; Antoniewski, C. Viral Suppressors of RNA Silencing Hinder Exogenous and Endogenous Small RNA Pathways in Drosophila. PLoS ONE 2009, 4, e5866. [Google Scholar] [CrossRef] [PubMed]
- Kuwata, R.; Isawa, H.; Hoshino, K.; Sasaki, T.; Kobayashi, M.; Maeda, K.; Sawabe, K. Analysis of mosquito-borne flavivirus superinfection in culex tritaeniorhynchus (Diptera: Culicidae) cells persistently infected with culex flavivirus (Flaviviridae). J. Med. Entomol. 2015, 52, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.M.; Carey, D.E. Concurrent Isolation from Patient of Two Arboviruses, Chikungunya and Dengue Type 2. Science 1967, 157, 1307–1308. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.F.; Su, C.L.; Shu, P.Y.; Yang, C.F.; Liao, T.L.; Cheng, C.H.; Hu, H.C.; Huang, J.H. Concurrent isolation of chikungunya virus and dengue virus from a patient with coinfection resulting from a trip to Singapore. J. Clin. Microbiol. 2010, 48, 4586–4589. [Google Scholar] [CrossRef] [PubMed]
- Parreira, R.; Centeno-Lima, S.; Lopes, A.; Portugal-Calisto, D.; Constantino, A.; Nina, J. Dengue virus serotype 4 and Chikungunya virus coinfection in a traveller returning from Luanda, Angola, January 2014. Eurosurveillance 2014, 19, 6–9. [Google Scholar] [CrossRef]
- Villamil-Gómez, W.E.; González-Camargo, O.; Rodriguez-Ayubi, J.; Zapata-Serpa, D.; Rodriguez-Morales, A.J. Dengue, chikungunya and Zika co-infection in a patient from Colombia. J. Infect. Public Health 2016, 9, 684–686. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Mousson, L.; Martin, E.; Failloux, A.B. Orally co-infected Aedes albopictus from La Reunion Island, Indian Ocean, can deliver both dengue and chikungunya infectious viral particles in their saliva. PLoS Negl. Trop. Dis. 2010, 4, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Rückert, C.; Weger-Lucarelli, J.; Garcia-Luna, S.M.; Young, M.C.; Byas, A.D.; Murrieta, R.A.; Fauver, J.R.; Ebel, G.D. Impact of simultaneous exposure to arboviruses on infection and transmission by Aedes aegypti mosquitoes. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Göertz, G.P.; Vogels, C.B.F.; Geertsema, C.; Koenraadt, C.J.M.; Pijlman, G.P. Mosquito co-infection with Zika and chikungunya virus allows simultaneous transmission without affecting vector competence of Aedes aegypti. PLoS Negl. Trop. Dis. 2017, 11, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Blitvich, B.J.; Firth, A.E. Insect-specific flaviviruses: A systematic review of their discovery, host range, mode of transmission, superinfection exclusion potential and genomic organization. Viruses 2015, 7, 1927–1959. [Google Scholar] [CrossRef] [PubMed]
- Hobson-Peters, J.; Yam, A.W.Y.; Lu, J.W.F.; Setoh, Y.X.; May, F.J.; Kurucz, N.; Walsh, S.; Prow, N.A.; Davis, S.S.; Weir, R.; et al. A New Insect-Specific Flavivirus from Northern Australia Suppresses Replication of West Nile Virus and Murray Valley Encephalitis Virus in Co-infected Mosquito Cells. PLoS ONE 2013, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hall-Mendelin, S.; McLean, B.J.; Bielefeldt-Ohmann, H.; Hobson-Peters, J.; Hall, R.A.; Van Den Hurk, A.F. The insect-specific Palm Creek virus modulates West Nile virus infection in and transmission by Australian mosquitoes. Parasites Vectors 2016, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kenney, J.L.; Solberg, O.D.; Langevin, S.A.; Brault, A.C. Characterization of a novel insect-specific flavivirus from Brazil: Potential for inhibition of infection of arthropod cells with medically important flaviviruses. J. Gen. Virol. 2014, 95, 2796–2808. [Google Scholar] [CrossRef] [PubMed]
- Goenaga, S.; Kenney, J.L.; Duggal, N.K.; Delorey, M.; Ebel, G.D.; Zhang, B.; Levis, S.C.; Enria, D.A.; Brault, A.C. Potential for co-infection of a mosquito-specific flavivirus, nhumirim virus, to block west nile virus transmission in mosquitoes. Viruses 2015, 7, 5801–5812. [Google Scholar] [CrossRef] [PubMed]
- Nasar, F.; Erasmus, J.H.; Haddow, A.D.; Tesh, R.B.; Weaver, S.C. Eilat virus induces both homologous and heterologous interference. Virology 2015, 484, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.G.; Olea-Popelka, F.J.; Eisen, L.; Moore, C.G.; Blair, C.D. Transmission dynamics of an insect-specific flavivirus in a naturally infected Culex pipiens laboratory colony and effects of co-infection on vector competence for West Nile virus. Virology 2012, 427, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Longdon, B.; Cao, C.; Martinez, J.; Jiggins, F.M. Previous Exposure to an RNA Virus Does Not Protect against Subsequent Infection in Drosophila melanogaster. PLoS ONE 2013, 8, e73833. [Google Scholar] [CrossRef] [PubMed]
- Tidbury, H.J.; Pedersen, A.B.; Boots, M. Within and transgenerational immune priming in an insect to a DNA virus. Proc. R. Soc. B Biol. Sci. 2011, 278, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Crochu, S.; Cook, S.; Attoui, H.; Charrel, R.N.; De Chesse, R.; Belhouchet, M.; Lemasson, J.J.; de Micco, P.; de Lamballerie, X. Sequences of flavivirus-related RNA viruses persist in DNA form integrated in the genome of Aedes spp. mosquitoes. J. Gen. Virol. 2004, 85, 1971–1980. [Google Scholar] [CrossRef] [PubMed]
- Nag, D.K.; Brecher, M.; Kramer, L.D. DNA forms of arboviral RNA genomes are generated following infection in mosquito cell cultures. Virology 2016, 498, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Katzourakis, A.; Gifford, R.J. Endogenous viral elements in animal genomes. PLoS Genet. 2010, 6. [Google Scholar] [CrossRef]
- Fort, P.; Albertini, A.; Van-Hua, A.; Berthomieu, A.; Roche, S.; Delsuc, F.; Pasteur, N.; Capy, P.; Gaudin, Y.; Weill, M. Fossil rhabdoviral sequences integrated into arthropod genomes: Ontogeny, evolution, and potential functionality. Mol. Biol. Evol. 2012, 29, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Frangeul, L.; Dickson, L.B.; Blanc, H.; Verdier, Y.; Vinh, J.; Lambrechts, L.; Saleh, M.-C. Uncovering the Repertoire of Endogenous Flaviviral Elements in Aedes Mosquito Genomes. J. Virol. 2017, 91, e00571-17. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, Z.J.; Dolan, P.T.; Kunitomi, M.; Tassetto, M.; Seetin, M.G.; Oh, S.; Heiner, C.; Paxinos, E.; Andino, R. The Diversity, Structure, and Function of Heritable Adaptive Immunity Sequences in the Aedes aegypti Genome. Curr. Biol. 2017, 27, 3511–3519. [Google Scholar] [CrossRef] [PubMed]
- Palatini, U.; Miesen, P.; Carballar-Lejarazu, R.; Ometto, L.; Rizzo, E.; Tu, Z.; van Rij, R.P.; Bonizzoni, M. Comparative genomics shows that viral integrations are abundant and express piRNAs in the arboviral vectors Aedes aegypti and Aedes albopictus. BMC Genom. 2017, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Miesen, P.; Joosten, J.; van Rij, R.P. PIWIs Go Viral: Arbovirus-Derived piRNAs in Vector Mosquitoes. PLoS Pathog. 2016, 12, e1006017. [Google Scholar] [CrossRef] [PubMed]
- Dudchenko, O.; Batra, S.S.; Omer, A.D.; Nyquist, S.K.; Hoeger, M.; Durand, N.C.; Shamim, M.S.; Machol, I.; Lander, E.S.; Aiden, A.P.; et al. De novo assembly of the Aedes aegypti genome using Hi-C yields chromosome-length scaffolds. Science 2017, 356, 92–95. [Google Scholar] [CrossRef] [PubMed]
| Virus Name | Abbreviation | Genome | Family | Host Restriction |
|---|---|---|---|---|
| Cricket paralysis virus | CrPV | (+) ssRNA | Dicistroviridae | Insect-specific |
| Drosophila C virus | DCV | (+) ssRNA | Dicistroviridae | Insect-specific |
| Culex flavivirus | CxFV | (+) ssRNA | Flaviviridae | Insect-specific |
| Nhumirim virus | NHUMV | (+) ssRNA | Flaviviridae | Insect-specific |
| Palm Creek virus | PCV | (+) ssRNA | Flaviviridae | Insect-specific |
| Flock House virus | FHV | (+) ssRNA | Nodaviridae | Insect-specific |
| Eilat virus | EILV | (+) ssRNA | Togaviridae | Insect-specific |
| Nora virus | Nora | (+) ssRNA | Unclassified Picornavirales | Insect-specific |
| Drosophila melanogaster sigma virus | DmelSV | (−) ssRNA | Rhabdoviridae | Insect-specific |
| Drosophila X virus | DXV | dsRNA | Birnaviridae | Insect-specific |
| Invertebrate iridescent virus 6 | IIV6 | dsDNA | Iridoviridae | Insect-specific |
| Kallithea virus | Kallithea | dsDNA | Nudiviridae | Insect-specific |
| Dengue virus | DENV | (+) ssRNA | Flaviviridae | Arbovirus |
| Japanese encephalitis virus | JEV | (+) ssRNA | Flaviviridae | Arbovirus |
| Murray Valley encephalitis virus | MVEV | (+) ssRNA | Flaviviridae | Arbovirus |
| St Louis encephalitis virus | SLEV | (+) ssRNA | Flaviviridae | Arbovirus |
| West Nile virus | WNV | (+) ssRNA | Flaviviridae | Arbovirus |
| Yellow fever virus | YFV | (+) ssRNA | Flaviviridae | Arbovirus |
| Zika virus | ZIKV | (+) ssRNA | Flaviviridae | Arbovirus |
| Chikungunya virus | CHIKV | (+) ssRNA | Togaviridae | Arbovirus |
| O’nyong’nyong virus | ONNV | (+) ssRNA | Togaviridae | Arbovirus |
| Semliki Forest virus | SFV | (+) ssRNA | Togaviridae | Arbovirus |
| Sindbis virus | SINV | (+) ssRNA | Togaviridae | Arbovirus |
| Venezuelan equine encephalitis virus | VEEV | (+) ssRNA | Togaviridae | Arbovirus |
| Western equine encephalitis virus | WEEV | (+) ssRNA | Togaviridae | Arbovirus |
| La Crosse encephalitis virus | LACV | (−) ssRNA | Peribunyaviridae | Arbovirus |
| Rift Valley fever virus | RVFV | (−) ssRNA | Phenuiviridae | Arbovirus |
| Vesicular stomatitis virus | VSV | (−) ssRNA | Rhabdoviridae | Arbovirus |
| Host Factor | Virus | Host | Associated Mutation | Phenotype (Homozygotes) | Population Frequency (Resistant Allele) | Related Processes | Methodology | References |
|---|---|---|---|---|---|---|---|---|
| ref(2)P | DmelSV | Dmel | NS polymorphism | 24% reduction in infection rate | 24% | Autophagy | Genetic mapping, mutagenesis, GWAS | [127,128,129,130] |
| CHKov1/CHKov2 | DmelSV | Dmel | TE insertion, rearrangement | 52% reduction in infection rate | TE: 83%; rearrangement <0.5% | Predicted acetylcholine esterase | Genetic mapping, GWAS | [130,131] |
| Ge-1 | DmelSV | Dmel | 26 aa deletion | 97% reduction in infection rate | 1% | RNA decay | Genetic mapping | [40] |
| ref(1)H QTL | DmelSV | Dmel | Unknown | Unknown | Unknown | Unknown | Genetic mapping | [128] |
| ref(3)O QTL | DmelSV | Dmel | Unknown | Unknown | Unknown | Unknown | Genetic mapping | [128] |
| ref(3)V QTL | DmelSV | Dmel | Unknown | Unknown | Unknown | Unknown | Genetic mapping | [128] |
| X13 QTL | DmelSV | Dmel | Unknown | 38% reduction in infection rate | Unknown | Unknown | Genetic mapping | [132] |
| X65 QTL | DmelSV | Dmel | Unknown | 13% reduction in infection rate | Unknown | Unknown | Genetic mapping | [132] |
| 2R70 QTL | DmelSV | Dmel | Unknown | 11% reduction in infection rate | Unknown | Unknown | Genetic mapping | [132] |
| 3R64 QTL | DmelSV | Dmel | Unknown | 12% reduction in infection rate | Unknown | Unknown | Genetic mapping | [132] |
| pastrel | DCV | Dmel | 7 alleles: cis-regulatory, structural, NS variants | 16%, 57%, or 80% increase in survival | NS: 7–33%, Structural variants: 4–51% | Unknown | GWAS, experimental evolution | [130,133,134] |
| 2R69 QTL | DCV | Dmel | Unknown | 2.5 day increase in survival time | Unknown | Unknown | Recombinant inbred line | [132] |
| 2L18 QTL | DCV | Dmel | Unknown | 0.75 day increase in survival time | Unknown | Unknown | Recombinant inbred line | [132] |
| APC7 | DCV | Dmel | Synonymous polymorphism | 95% increase in survival | 3% | Cell cycle | GWAS | [130] |
| Ubc-E2H | DCV | Dmel | Intronic polymorphism | Not reported | 27% (lab-maintained population) | Predicted ubiquitin ligase | Experimental evolution | [133] |
| Cip4 | Kallithea | Dmel | NS polymorphism | 27% increase in survival | 77% | Membrane trafficking | GWAS | [107] |
| Dicer-2 | DENV-1 | Aaeg | Unknown | Explains 17.8% of viral dissemination | Unknown | RNAi | Genetic mapping | [135] |
| 71CGT1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains up to 7.6% variation in MIB | Unknown | Unknown | Genetic mapping | [136] |
| 335CGA1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 8.1% variation in MIB and ≤21.4% in dissemination | Unknown | Unknown | Genetic mapping | [136] |
| 88CA1/88GAA1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 12% variation in MIB and ≤75.6% in titer | Unknown | Unknown | Genetic mapping | [136] |
| 301CT1/301ACG1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 11.4% variation in MIB | Unknown | Unknown | Genetic mapping | [136] |
| B19 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 6.2% variation in MIB | Unknown | Unknown | Genetic mapping | [136] |
| 69TGA1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 22.6% variation in dissemination and ≤ 8.9% in titer | Unknown | Unknown | Genetic mapping | [136] |
| 201AAT1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 15.2% variation in titer | Unknown | Unknown | Genetic mapping | [136] |
| 470CT2/470AG1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 12.3% variation in titer | Unknown | Unknown | Genetic mapping | [136] |
| 17ATA1 QTL | DENV-1/3 | Aaeg | Unknown | QTL explains ≤ 13.7% variation in titer | Unknown | Unknown | Genetic mapping | [136] |
| early trypsin QTL | DENV-2 | Aaeg | Unknown | 86% decrease in infection dissemination | Unknown | Blood meal digestion | Genetic mapping | [137] |
| B18.621 QTL | DENV-2 | Aaeg | Unknown | 87% decrease in infection dissemination | Unknown | Unknown | Genetic mapping | [137] |
| B20.392 QTL | LACV | Otri | Unknown | Transovarial transmission rate increased from 0 to 60% | Unknown | Unknown | Genetic mapping | [138] |
| C01.385/C13.5573 QTL | LACV | Otri | 2 linked QTL with unknown mutations | Transovarial transmission rate increased from 0 to 60% | Unknown | Unknown | Genetic mapping | [138] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmer, W.H.; Varghese, F.S.; Van Rij, R.P. Natural Variation in Resistance to Virus Infection in Dipteran Insects. Viruses 2018, 10, 118. https://doi.org/10.3390/v10030118
Palmer WH, Varghese FS, Van Rij RP. Natural Variation in Resistance to Virus Infection in Dipteran Insects. Viruses. 2018; 10(3):118. https://doi.org/10.3390/v10030118
Chicago/Turabian StylePalmer, William H., Finny S. Varghese, and Ronald P. Van Rij. 2018. "Natural Variation in Resistance to Virus Infection in Dipteran Insects" Viruses 10, no. 3: 118. https://doi.org/10.3390/v10030118
APA StylePalmer, W. H., Varghese, F. S., & Van Rij, R. P. (2018). Natural Variation in Resistance to Virus Infection in Dipteran Insects. Viruses, 10(3), 118. https://doi.org/10.3390/v10030118





