Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and Virus Production
2.2. Plant Extract Preparation
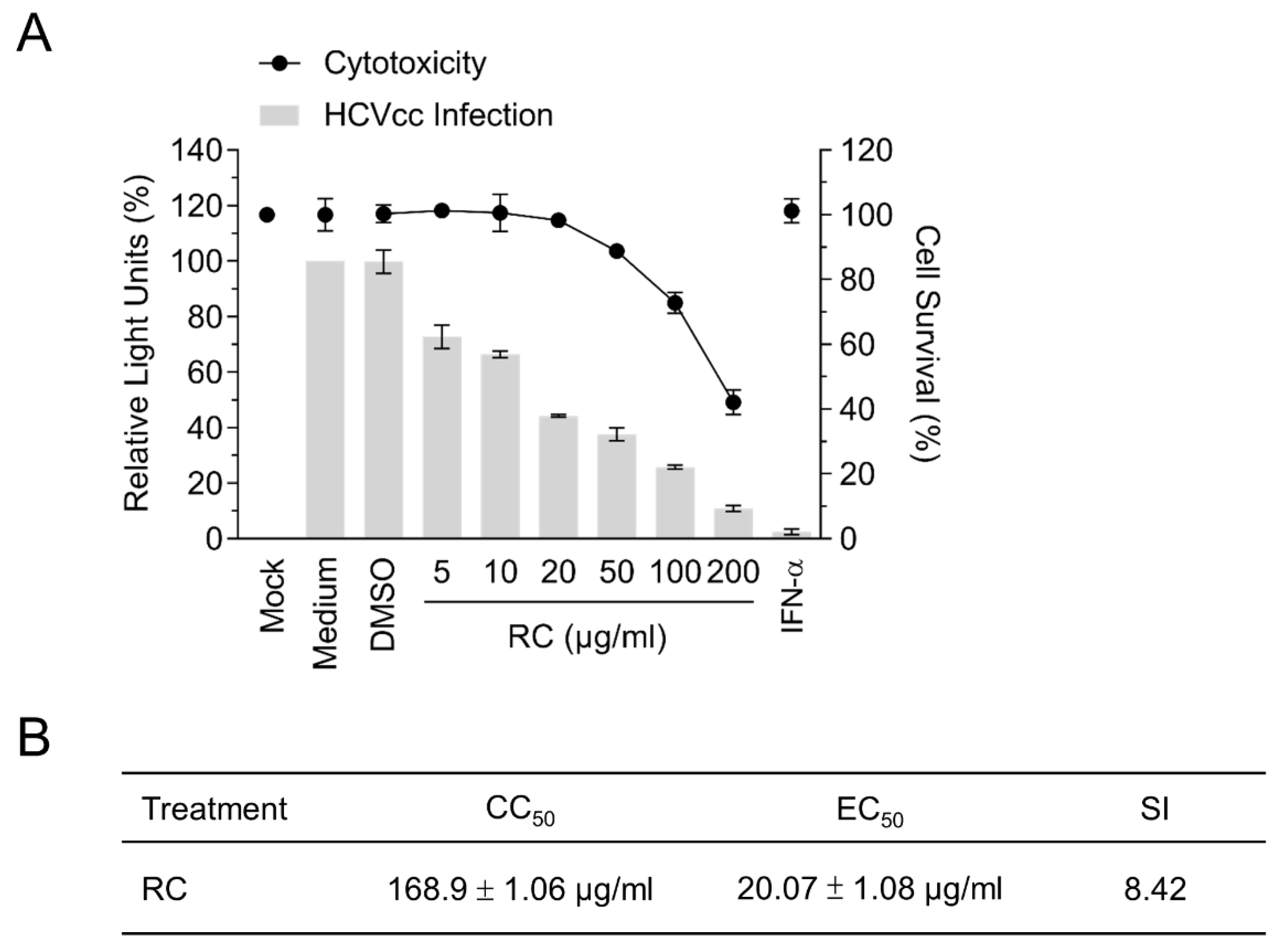
2.3. Cytotoxicity Assay and Antiviral Activity Analysis
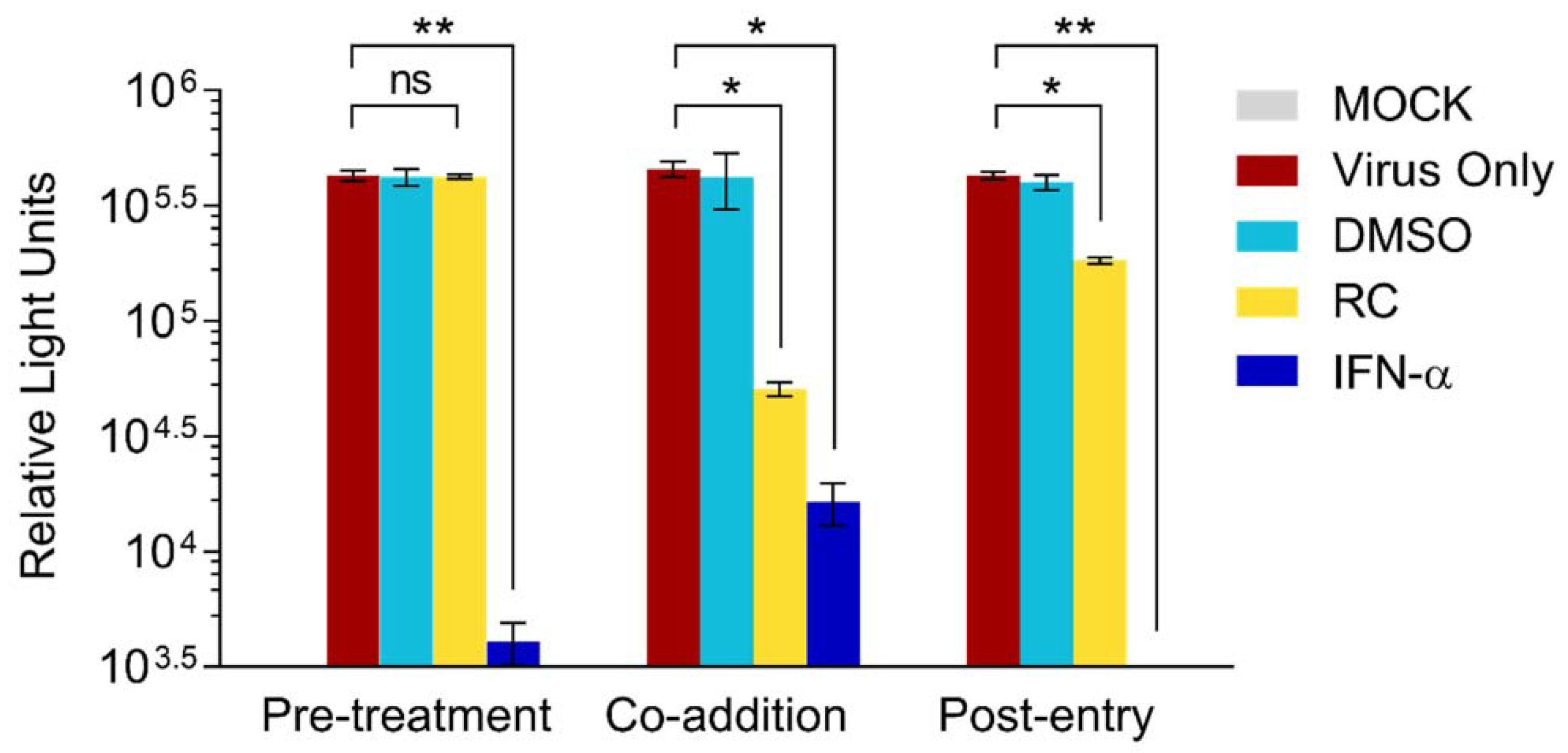
2.4. Time-of-Drug-Addition Assay
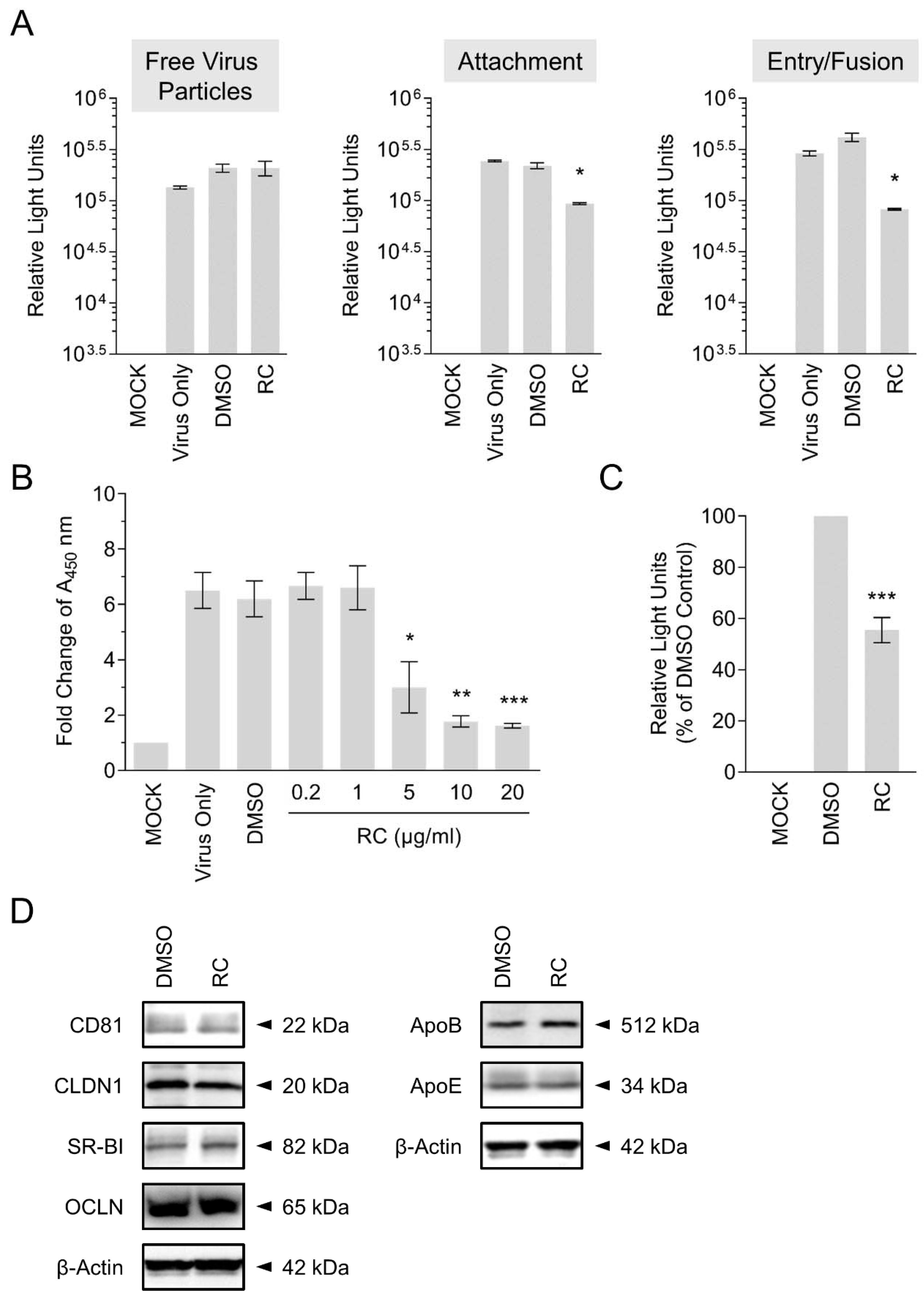
2.5. Synchronized Infection Analysis on Early Viral Entry
2.6. Binding Assay
2.7. Analysis Using HCV Pseudoparticles
2.8. Western Blot
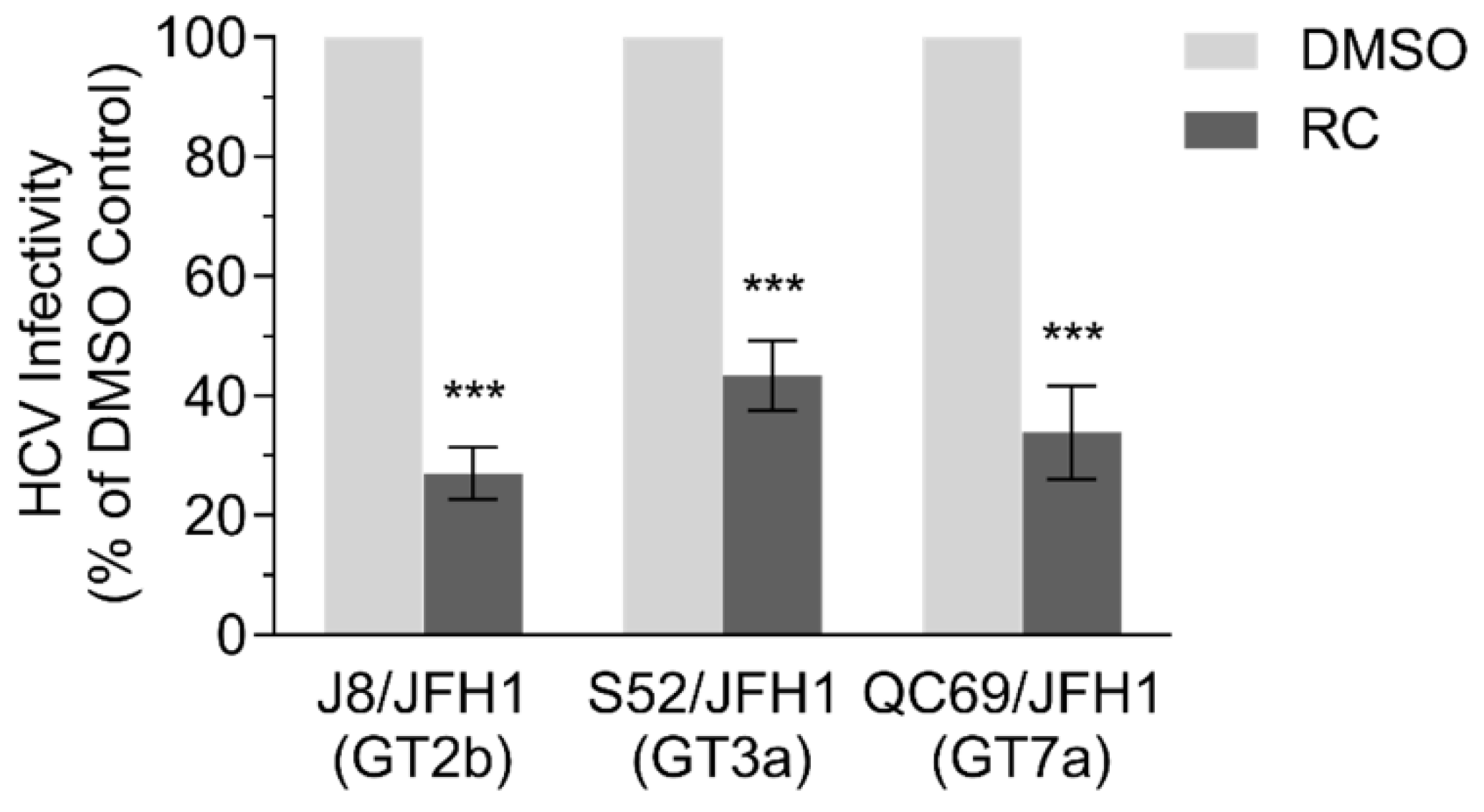
2.9. Inhibitory Effects Against Multiple Genotypes of HCV
2.10. Statistical Analysis
3. Results
3.1. RC is Capable of Inhibiting HCV Infection
3.2. RC Inhibits the Early Steps of HCV Entry
3.3. RC Blocks HCV Viral Attachment and Entry/Fusion into the Host Cells
3.4. Confirmation of RC’s Antagonism Against HCV Cell Attachment by ELISA-Based Virus Binding Assay
3.5. RC Robustly Inhibits Infection by Pseudoparticles Bearing HCV Glycoproteins
3.6. RC Does Not Modulate Host Cell Entry Factors to HCV Infection
3.7. RC Inhibits Multiple HCV Genotypes
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moradpour, D.; Penin, F.; Rice, C.M. Replication of hepatitis C virus. Nat. Rev. Microbiol. 2007, 5, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Douam, F.; Lavillette, D.; Cosset, F.L. The mechanism of HCV entry into host cells. Prog. Mol. Biol. Transl. Sci. 2015, 129, 63–107. [Google Scholar] [PubMed]
- Burnouf, T.; Liu, C.H.; Lin, L.T. Strategies to Preclude Hepatitis C Virus Entry. In Advances in Treatment of Hepatitis C and B; Allam, N., Ed.; IntechOpen: Rijeka, Croatia, 2017; pp. 251–284. [Google Scholar]
- Fukuhara, T.; Ono, C.; Puig-Basagoiti, F.; Matsuura, Y. Roles of Lipoproteins and Apolipoproteins in Particle Formation of Hepatitis C Virus. Trends Microbiol. 2015, 23, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, X.; Chi, X.; Zhao, F.; Guo, J.; Ma, P.; Zhong, J.; Niu, J.; Pan, X.; Long, G. Neglected but Important Role of Apolipoprotein E Exchange in Hepatitis C Virus Infection. J. Virol. 2016, 90, 9632–9643. [Google Scholar] [CrossRef] [PubMed]
- Feeney, E.R.; Chung, R.T. Antiviral treatment of hepatitis C. BMJ 2014, 348, g3308. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Labarga, P.; Barreiro, P.; Fernandez-Montero, J.V.; de Mendoza, C.; Esposito, I.; Benitez-Gutierrez, L.; Pena, J.M. Drug interactions with new hepatitis C oral drugs. Expert Opin. Drug Metab. Toxicol. 2015, 11, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Gritsenko, D.; Hughes, G. Ledipasvir/Sofosbuvir (harvoni): Improving options for hepatitis C virus infection. P T. 2015, 40, 256–276. [Google Scholar] [PubMed]
- Zoulim, F.; Liang, T.J.; Gerbes, A.L.; Aghemo, A.; Deuffic-Burban, S.; Dusheiko, G.; Fried, M.W.; Pol, S.; Rockstroh, J.K.; Terrault, N.A.; et al. Hepatitis C virus treatment in the real world: Optimising treatment and access to therapies. Gut 2015, 64, 1824–1833. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Monographs on Selected Medicinal Plants-Volume 1. Available online: http://apps.who.int/medicinedocs/en/d/Js2200e/13.html#Js2200e.13 (accessed on 9 July 2018).
- Meng, F.C.; Wu, Z.F.; Yin, Z.Q.; Lin, L.G.; Wang, R.; Zhang, Q.W. Coptidis rhizoma and its main bioactive components: Recent advances in chemical investigation, quality evaluation and pharmacological activity. Chin. Med. 2018, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- MOHW. Taiwan Herbal Pharmacopeia English Version, 2nd ed.; English Version; Ministry of Health and Welfare: Taipei, Taiwan, 2016.
- Muluye, R.A.; Bian, Y.; Alemu, P.N. Anti-inflammatory and Antimicrobial Effects of Heat-Clearing Chinese Herbs: A Current Review. J. Tradit. Complement. Med. 2014, 4, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Chin, L.W.; Cheng, Y.W.; Lin, S.S.; Lai, Y.Y.; Lin, L.Y.; Chou, M.Y.; Chou, M.C.; Yang, C.C. Anti-herpes simplex virus effects of berberine from Coptidis rhizoma, a major component of a Chinese herbal medicine, Ching-Wei-San. Arch. Virol. 2010, 155, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Shin, H.S.; Park, H.; Kim, Y.C.; Yun, Y.G.; Park, S.; Shin, H.J.; Kim, K. In vitro inhibition of coronavirus replications by the traditionally used medicinal herbal extracts, Cimicifuga rhizoma, Meliae cortex, Coptidis rhizoma, and Phellodendron cortex. J. Clin. Virol. 2008, 41, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-H.; Chathuranga, K.; Uddin, M.B.; Weeratunga, P.; Kim, M.S.; Cho, W.-K.; Kim, H.I.; Ma, J.Y.; Lee, J.-S. Coptidis Rhizoma extract inhibits replication of respiratory syncytial virus in vitro and in vivo by inducing antiviral state. J. Microbiol. 2017, 55, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.T.; Chen, T.Y.; Lin, S.C.; Chung, C.Y.; Lin, T.C.; Wang, G.H.; Anderson, R.; Lin, C.C.; Richardson, C.D. Broad-spectrum antiviral activity of chebulagic acid and punicalagin against viruses that use glycosaminoglycans for entry. BMC Microbiol. 2013, 13, 187. [Google Scholar] [CrossRef] [PubMed]
- The Plant List-Version 1.1. Available online: http://www.theplantlist.org/ (accessed on 14 July 2018).
- Ren, L.; Xue, X.; Liang, X. Characterization of protoberberine alkaloids in Coptidis Rhizoma (Huanglian) by HPLC with ESI-MS/MS. J. Sep. Sci. 2013, 36, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zhang, T.; Zhang, R.; Ito, Y. Application of analytical and preparative high-speed counter-current chromatography for separation of alkaloids from Coptis chinensis Franch. J. Chromatogr. A. 1998, 829, 137–141. [Google Scholar] [CrossRef]
- Lin, L.T.; Chen, T.Y.; Chung, C.Y.; Noyce, R.S.; Grindley, T.B.; McCormick, C.; Lin, T.C.; Wang, G.H.; Lin, C.C.; Richardson, C.D. Hydrolyzable tannins (chebulagic acid and punicalagin) target viral glycoprotein-glycosaminoglycan interactions to inhibit herpes simplex virus 1 entry and cell-to-cell spread. J. Virol. 2011, 85, 4386–4398. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.C.; Chang, S.P.; Lin, L.C.; Li, C.L.; Richardson, C.D.; Lin, C.C.; Lin, L.T. Limonium sinense and gallic acid suppress hepatitis C virus infection by blocking early viral entry. Antiviral Res. 2015, 118, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.T.; Chung, C.Y.; Hsu, W.C.; Chang, S.P.; Hung, T.C.; Shields, J.; Russell, R.S.; Lin, C.C.; Li, C.F.; Yen, M.H.; et al. Saikosaponin b2 is a naturally occurring terpenoid that efficiently inhibits hepatitis C virus entry. J. Hepatol. 2015, 62, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Bartosch, B.; Cosset, F.L. Studying HCV cell entry with HCV pseudoparticles (HCVpp). Methods Mol. Biol. 2009, 510, 279–293. [Google Scholar] [PubMed]
- Gottwein, J.M.; Scheel, T.K.; Hoegh, A.M.; Lademann, J.B.; Eugen-Olsen, J.; Lisby, G.; Bukh, J. Robust hepatitis C genotype 3a cell culture releasing adapted intergenotypic 3a/2a (S52/JFH1) viruses. Gastroenterology 2007, 133, 1614–1626. [Google Scholar] [CrossRef] [PubMed]
- Gottwein, J.M.; Scheel, T.K.; Jensen, T.B.; Lademann, J.B.; Prentoe, J.C.; Knudsen, M.L.; Hoegh, A.M.; Bukh, J. Development and characterization of hepatitis C virus genotype 1–7 cell culture systems: Role of CD81 and scavenger receptor class B type I and effect of antiviral drugs. Hepatology 2009, 49, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Lin, C.C.; Hsu, W.C.; Chung, C.Y.; Lin, C.C.; Jassey, A.; Chang, S.P.; Tai, C.J.; Tai, C.J.; Shields, J.; et al. Highly bioavailable silibinin nanoparticles inhibit HCV infection. Gut 2017, 66, 1853–1861. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.Y.; Liu, C.H.; Burnouf, T.; Wang, G.H.; Chang, S.P.; Jassey, A.; Tai, C.J.; Tai, C.J.; Huang, C.J.; Richardson, C.D.; et al. Activity-based and fraction-guided analysis of Phyllanthus urinaria identifies loliolide as a potent inhibitor of hepatitis C virus entry. Antiviral Res. 2016, 130, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Verna, E.C.; Brown, R.S., Jr. Hepatitis C and liver transplantation: Enhancing outcomes and should patients be retransplanted. Clin. Liver Dis. 2008, 12, 637–659, ix-x. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Fofana, I.; Thumann, C.; Mailly, L.; Alles, R.; Robinet, E.; Meyer, N.; Schaeffer, M.; Habersetzer, F.; Doffoel, M.; et al. Synergy of entry inhibitors with direct-acting antivirals uncovers novel combinations for prevention and treatment of hepatitis C. Gut 2015, 64, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement Med. 2014, 4, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Wagoner, J.; Negash, A.; Kane, O.J.; Martinez, L.E.; Nahmias, Y.; Bourne, N.; Owen, D.M.; Grove, J.; Brimacombe, C.; McKeating, J.A.; et al. Multiple effects of silymarin on the hepatitis C virus lifecycle. Hepatology 2010, 51, 1912–1921. [Google Scholar] [CrossRef] [PubMed]
- Blaising, J.; Levy, P.L.; Gondeau, C.; Phelip, C.; Varbanov, M.; Teissier, E.; Ruggiero, F.; Polyak, S.J.; Oberlies, N.H.; Ivanovic, T.; et al. Silibinin inhibits hepatitis C virus entry into hepatocytes by hindering clathrin-dependent trafficking. Cell Microbiol. 2013, 15, 1866–1882. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.Y.; Liu, C.H.; Wang, G.H.; Jassey, A.; Li, C.L.; Chen, L.; Yen, M.H.; Lin, C.C.; Lin, L.T. (4R,6S)-2-Dihydromenisdaurilide is a Butenolide that Efficiently Inhibits Hepatitis C Virus Entry. Sci. Rep. 2016, 6, 29969. [Google Scholar] [CrossRef] [PubMed]
- Haid, S.; Novodomska, A.; Gentzsch, J.; Grethe, C.; Geuenich, S.; Bankwitz, D.; Chhatwal, P.; Jannack, B.; Hennebelle, T.; Bailleul, F.; et al. A plant-derived flavonoid inhibits entry of all HCV genotypes into human hepatocytes. Gastroenterology 2012, 143, 213–222. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, T.-C.; Jassey, A.; Lin, C.-J.; Liu, C.-H.; Lin, C.-C.; Yen, M.-H.; Lin, L.-T. Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection. Viruses 2018, 10, 669. https://doi.org/10.3390/v10120669
Hung T-C, Jassey A, Lin C-J, Liu C-H, Lin C-C, Yen M-H, Lin L-T. Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection. Viruses. 2018; 10(12):669. https://doi.org/10.3390/v10120669
Chicago/Turabian StyleHung, Ting-Chun, Alagie Jassey, Chien-Ju Lin, Ching-Hsuan Liu, Chun-Ching Lin, Ming-Hong Yen, and Liang-Tzung Lin. 2018. "Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection" Viruses 10, no. 12: 669. https://doi.org/10.3390/v10120669
APA StyleHung, T.-C., Jassey, A., Lin, C.-J., Liu, C.-H., Lin, C.-C., Yen, M.-H., & Lin, L.-T. (2018). Methanolic Extract of Rhizoma Coptidis Inhibits the Early Viral Entry Steps of Hepatitis C Virus Infection. Viruses, 10(12), 669. https://doi.org/10.3390/v10120669




