Breed Differences in PCV2 Uptake and Disintegration in Porcine Monocytes
Abstract
:1. Introduction
2. Materials and Methods
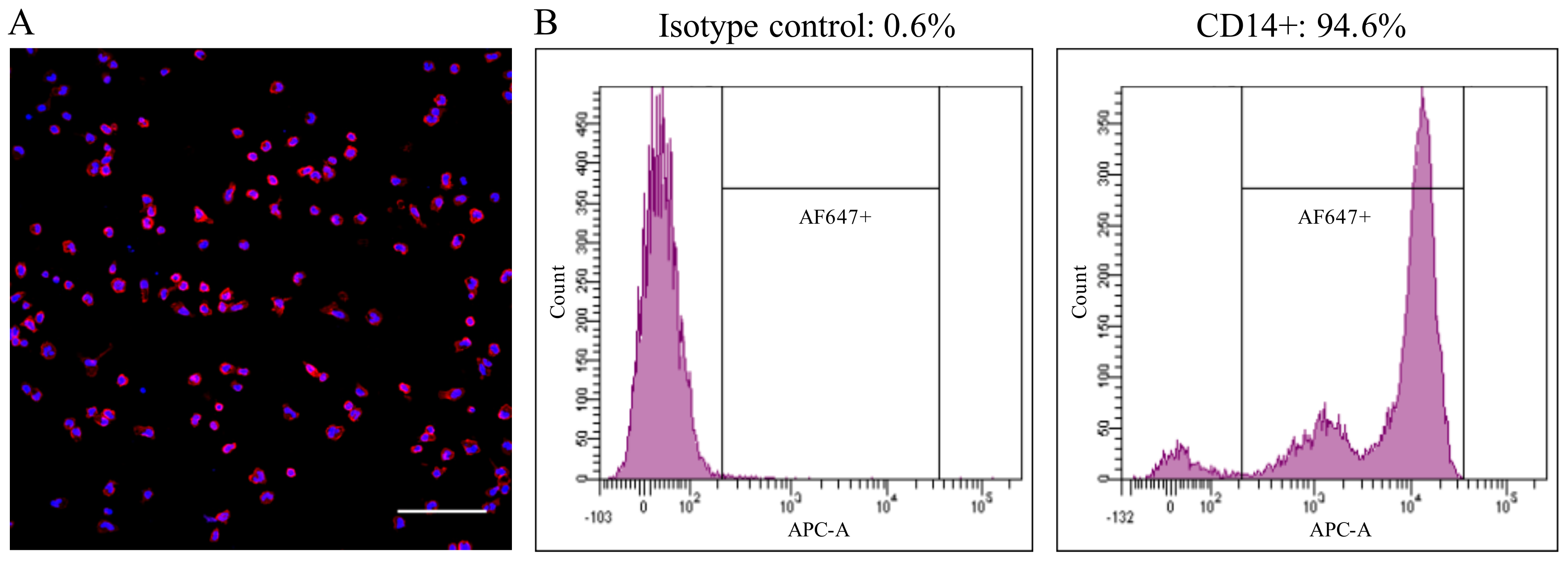
2.1. Preparation of Peripheral Blood Monocytes
2.2. Virus Preparation and Particle Quantification
2.3. The Fate of PCV2 Capsids in Blood Monocytes
2.4. Effect of Different Entry Inhibitors on PCV2 Uptake
2.5. PCV2 Trafficking in Blood Monocytes
2.6. The Fate of PCV2 Viral Genomes in Blood Monocytes
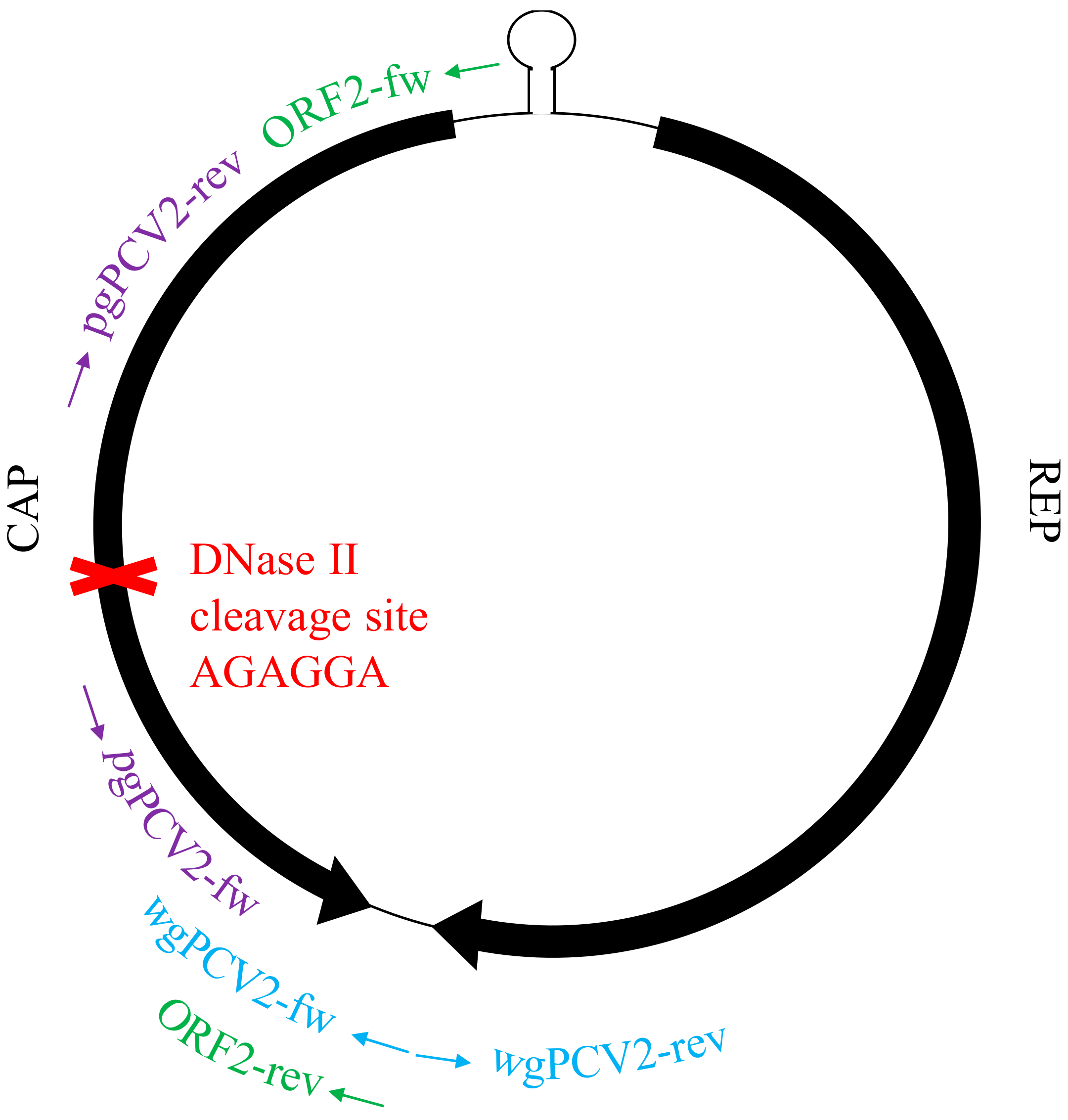
2.7. In Vitro Test of the Ability of Lysosomal Deoxyribonuclease II (DNase II) to Degrade PCV2 Genome
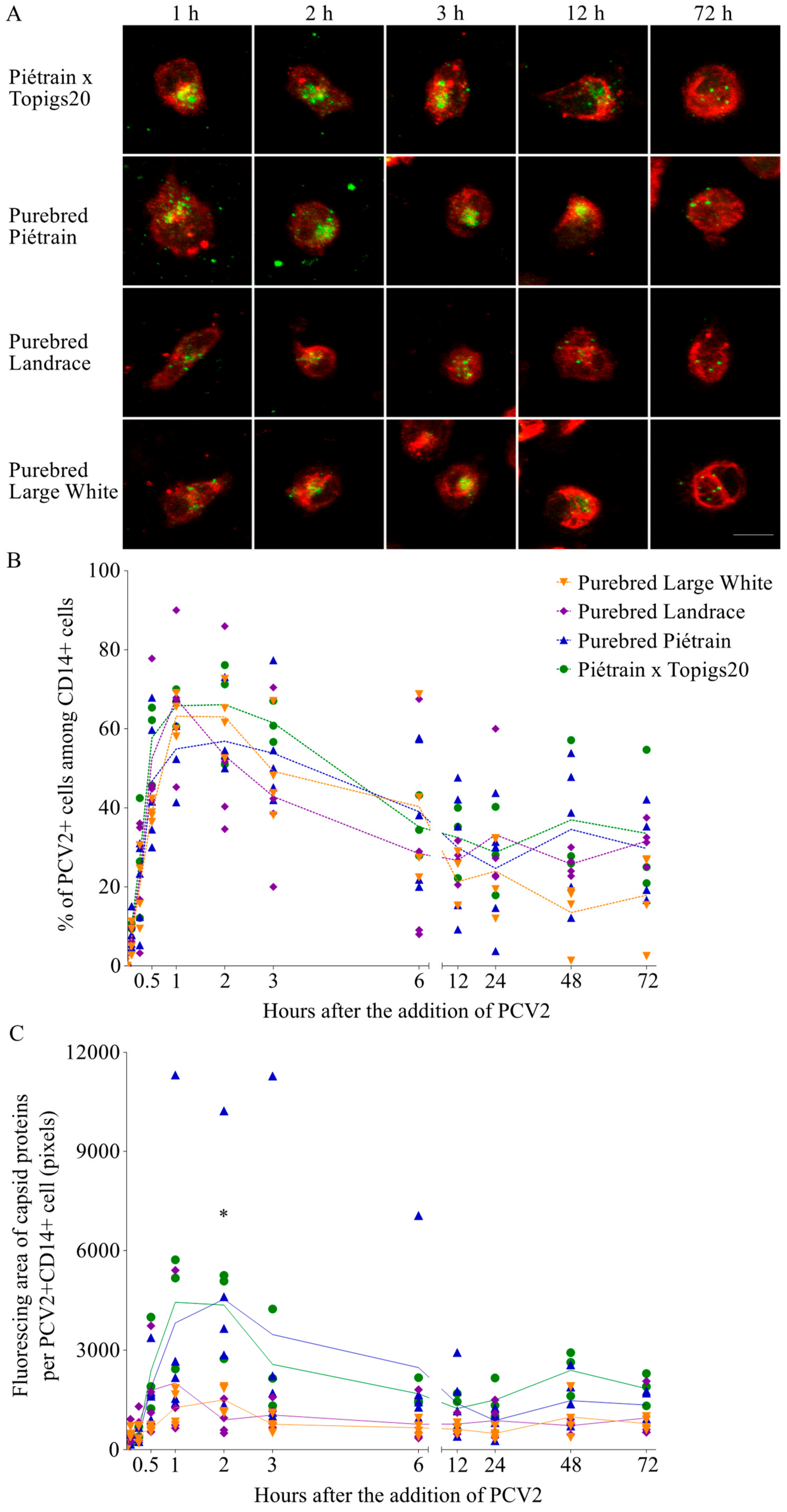
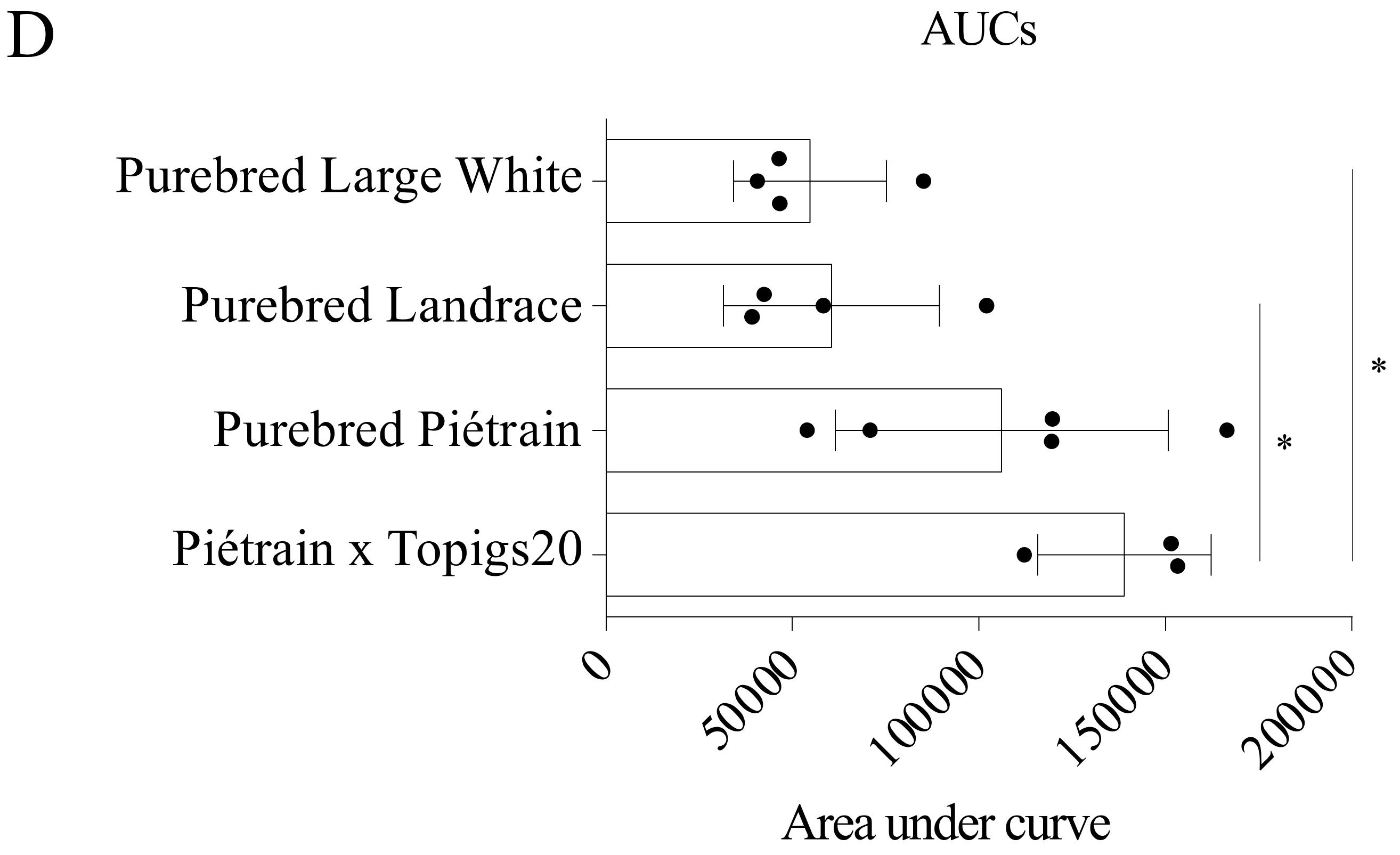
2.8. Kinetics of PCV2 Uptake and Disintegration in Blood Monocytes from Four Pig Breeds
2.9. Statistical Analysis
3. Results
3.1. The Uptake and Disintegration of PCV2 Capsids in Blood Monocytes
3.2. PCV2 Is Taken up by Blood Monocytes via Clathrin-Mediated Endocytosis
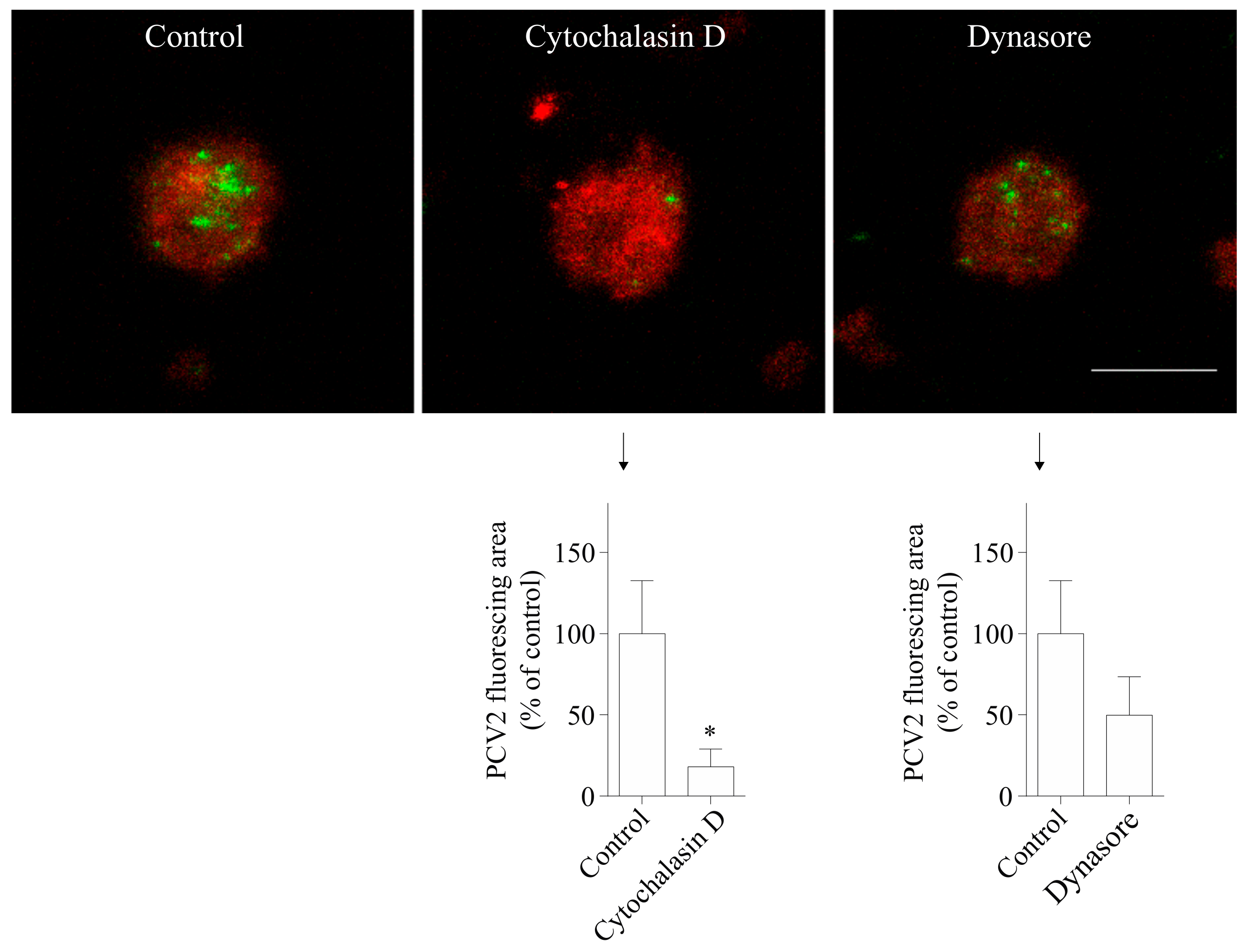
3.3. The Uptake of PCV2 by Blood Monocytes Requires Actin and Dynamin
3.4. PCV2 Traffics Along the Endosome-Lysosome Pathway
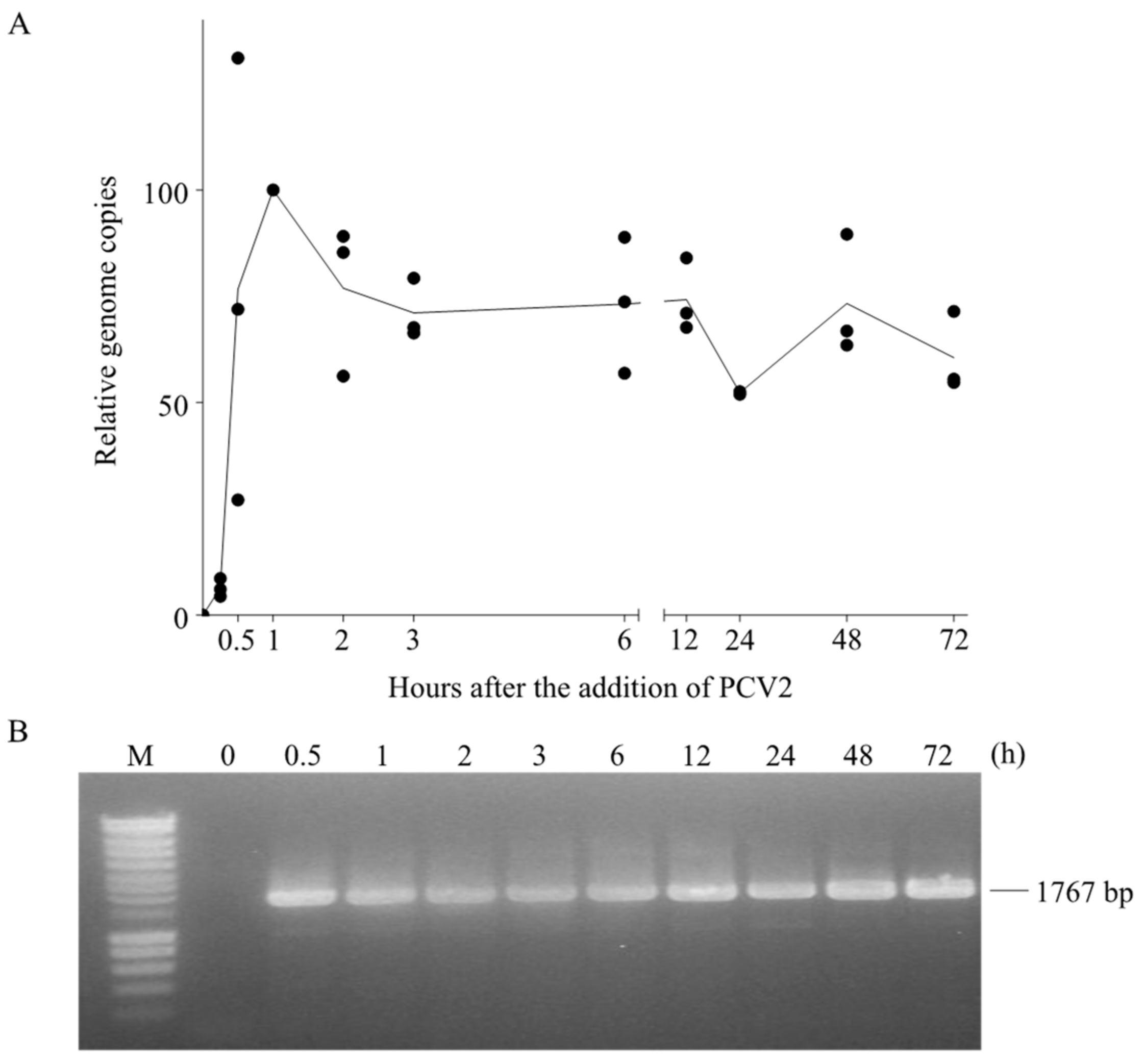
3.5. PCV2 Genome Persists in Blood Monocytes in Its Complete Form
3.6. Deoxyribonuclease II (DNase II) from Porcine Lysosome can Degrade PCV2 Genome
3.7. Blood Monocytes Differ in the Ability to Take up and Disintegrate PCV2
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Finsterbusch, T.; Mankertz, A. Porcine circoviruses—Small but powerful. Virus Res. 2009, 143, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Li, J.; Yuan, X.; Wang, G.; Shi, J.; Wu, J.; Cong, X.; Sun, W.; Du, Y.; Chai, T.; et al. Complete genome sequence of porcine circovirus 2B strain shandong. J. Virol. 2012, 86, 13885. [Google Scholar] [CrossRef] [PubMed]
- Crowther, R.A.; Berriman, J.A.; Curran, W.L.; Allan, G.M.; Todd, D. Comparison of the structures of three circoviruses: Chicken anemia virus, porcine circovirus type 2, and beak and feather disease virus. J. Virol. 2003, 77, 13036–13041. [Google Scholar] [CrossRef] [PubMed]
- Mankertz, A.; Mankertz, J.; Wolf, K.; Buhk, H.J. Identification of a protein essential for replication of porcine circovirus. J. Gen. Virol. 1998, 79, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Truong, C.; Mahe, D.; Blanchard, P.; Le Dimna, M.; Madec, F.; Jestin, A.; Albina, E. Identification of an immunorelevant ORF2 epitope from porcine circovirus type 2 as a serological marker for experimental and natural infection. Arch. Virol. 2001, 146, 1197–1211. [Google Scholar] [CrossRef] [PubMed]
- Segales, J. Porcine circovirus type 2 (PCV2) infections: Clinical signs, pathology and laboratory diagnosis. Virus Res. 2012, 164, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Opriessnig, T.; Madson, D.M.; Prickett, J.R.; Kuhar, D.; Lunney, J.K.; Elsener, J.; Halbur, P.G. Effect of porcine circovirus type 2 (PCV2) vaccination on porcine reproductive and respiratory syndrome virus (PRRSV) and PCV2 coinfection. Vet. Microbiol. 2008, 131, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Segalés, J.; Domingo, M. Postweaning mulstisystemic wasting syndrome (PMWS) in pigs. A review. Veterinary Q. 2002, 24, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, R.E., Jr.; Meerts, P.; Nauwynck, H.J.; Ellis, J.A.; Pensaert, M.B. Characteristics of porcine circovirus-2 replication in lymphoid organs of pigs inoculated in late gestation or postnatally and possible relation to clinical and pathological outcome of infection. J. Vet. Diagn Investig. 2004, 16, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Allan, G.M.; Ellis, J.A. Porcine circoviruses: A review. J. Vet. Diagn Investig. 2000, 12, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, D.F.; McCullough, K.; Meehan, B.M.; McNeilly, F.; McNair, I.; Stevenson, L.S.; Foster, J.C.; Ellis, J.A.; Krakowka, S.; Adair, B.M.; et al. In vitro studies on the infection and replication of porcine circovirus type 2 in cells of the porcine immune system. Vet. Immunol. Immunopathol. 2003, 94, 149–161. [Google Scholar] [CrossRef]
- Chianini, F.; Majo, N.; Segales, J.; Dominguez, J.; Domingo, M. Immunohistochemical characterisation of PCV2 associate lesions in lymphoid and non-lymphoid tissues of pigs with natural postweaning multisystemic wasting syndrome (PMWS). Vet. Immunol. Immunopathol. 2003, 94, 63–75. [Google Scholar] [CrossRef]
- Rosell, C.; Segales, J.; Plana-Duran, J.; Balasch, M.; Rodriguez-Arrioja, G.M.; Kennedy, S.; Allan, G.M.; McNeilly, F.; Latimer, K.S.; Domingo, M. Pathological, immunohistochemical, and in-situ hybridization studies of natural cases of postweaning multisystemic wasting syndrome (PMWS) in pigs. J. Comp. Pathol. 1999, 120, 59–78. [Google Scholar] [CrossRef] [PubMed]
- Pensaert, M.B.; Sanchez, R.E., Jr.; Ladekjaer-Mikkelsen, A.S.; Allan, G.M.; Nauwynck, H.J. Viremia and effect of fetal infection with porcine viruses with special reference to porcine circovirus 2 infection. Vet. Microbiol. 2004, 98, 175–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez, R.E., Jr.; Meerts, P.; Nauwynck, H.J.; Pensaert, M.B. Change of porcine circovirus 2 target cells in pigs during development from fetal to early postnatal life. Vet. Microbiol. 2003, 95, 15–25. [Google Scholar] [CrossRef]
- Vincent, I.E.; Carrasco, C.P.; Herrmann, B.; Meehan, B.M.; Allan, G.M.; Summerfield, A.; McCullough, K.C. Dendritic cells harbor infectious porcine circovirus type 2 in the absence of apparent cell modulation or replication of the virus. J. Virol. 2003, 77, 13288–13300. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, D.J.; Meerts, P.; Costers, S.; Misinzo, G.; Barbe, F.; Van Reeth, K.; Nauwynck, H.J. Increased porcine circovirus type 2 replication in porcine leukocytes in vitro and in vivo by concanavalin a stimulation. Vet. Microbiol. 2008, 132, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Nauwynck, H.J.; Sanchez, R.; Meerts, P.; Lefebvre, D.J.; Saha, D.; Huang, L.; Misinzo, G. Cell tropism and entry of porcine circovirus 2. Virus Res. 2012, 164, 43–45. [Google Scholar] [CrossRef] [PubMed]
- Misinzo, G.; Meerts, P.; Bublot, M.; Mast, J.; Weingartl, H.M.; Nauwynck, H.J. Binding and entry characteristics of porcine circovirus 2 in cells of the porcine monocytic line 3D4/31. J. Gen. Virol. 2005, 86, 2057–2068. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Soria, S.; Segales, J.; Nofrarias, M.; Calsamiglia, M.; Ramirez, H.; Minguez, A.; Serrano, I.M.; Marin, O.; Callen, A. Genetic influence on the expression of PCV disease. Vet. Rec. 2004, 155, 504. [Google Scholar] [PubMed]
- Opriessnig, T.; Fenaux, M.; Thomas, P.; Hoogland, M.J.; Rothschild, M.F.; Meng, X.J.; Halbur, P.G. Evidence of breed-dependent differences in susceptibility to porcine circovirus type-2-associated disease and lesions. Vet. Pathol. 2006, 43, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Opriessnig, T.; Patterson, A.R.; Madson, D.M.; Pal, N.; Rothschild, M.; Kuhar, D.; Lunney, J.K.; Juhan, N.M.; Meng, X.J.; Halbur, P.G. Difference in severity of porcine circovirus type two-induced pathological lesions between landrace and pietrain pigs1. J. Anim. Sci. 2009, 87, 1582–1590. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, D.J.; Costers, S.; Van Doorsselaere, J.; Misinzo, G.; Delputte, P.L.; Nauwynck, H.J. Antigenic differences among porcine circovirus type 2 strains, as demonstrated by the use of monoclonal antibodies. J. Gen. Virol. 2008, 89, 177–187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spitzer, S.; Eckstein, F. Inhibition of deoxyribonucleases by phosphorothioate groups in oligodeoxyribonucleotides. Nucleic Acids Res. 1988, 16, 11691–11704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Doorsselaere, J.; Lefebvre, D.J.; Nauwynck, H.J.; Delputte, P.L. Detection of truncated circular DNA species in Escherichia coli with a pcv2-containing plasmid with a single PCV2 origin of replication. Intervirology 2010, 53, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Kaksonen, M.; Toret, C.P.; Drubin, D.G. Harnessing actin dynamics for clathrin-mediated endocytosis. Nat. Rev. Mol. Cell. Biol. 2006, 7, 404. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.; Warrington, A.; Taylor, K.A.; Svitkina, T. Structural organization of the actin cytoskeleton at sites of clathrin-mediated endocytosis. Curr. Biol. CB 2011, 21, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Loebrich, S. The role of F-actin in modulating clathrin-mediated endocytosis: Lessons from neurons in health and neuropsychiatric disorder. Commun. Integr. Biol. 2014, 7, e28740. [Google Scholar] [CrossRef] [PubMed]
- Hinshaw, J.E.; Schmid, S.L. Dynamin self-assembles into rings suggesting a mechanism for coated vesicle budding. Nature 1995, 374, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Macia, E.; Ehrlich, M.; Massol, R.; Boucrot, E.; Brunner, C.; Kirchhausen, T. Dynasore, a cell-permeable inhibitor of dynamin. Dev. Cell. 2006, 10, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Pelkmans, L.; Helenius, A. Insider information: What viruses tell us about endocytosis. Curr. Opin. Cell. Biol. 2003, 15, 414–422. [Google Scholar] [CrossRef]
- Howell, D.P.; Krieser, R.J.; Eastman, A.; Barry, M.A. Deoxyribonuclease II is a lysosomal barrier to transfection. Mol. Ther. 2003, 8, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.J.; Aguilera, R.J. Dnase II: Genes, enzymes and function. Gene 2003, 322, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Weingartl, H.M.; Sabara, M.; Pasick, J.; van Moorlehem, E.; Babiuk, L. Continuous porcine cell lines developed from alveolar macrophages: Partial characterization and virus susceptibility. J. Virol. Methods 2002, 104, 203–216. [Google Scholar] [CrossRef]
- Misinzo, G.; Delputte, P.L.; Lefebvre, D.J.; Nauwynck, H.J. Porcine circovirus 2 infection of epithelial cells is clathrin-, caveolae- and dynamin-independent, actin and rho-gtpase-mediated, and enhanced by cholesterol depletion. Virus Res. 2009, 139, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Vincent, I.E.; Carrasco, C.P.; Guzylack-Piriou, L.; Herrmann, B.; McNeilly, F.; Allan, G.M.; Summerfield, A.; McCullough, K.C. Subset-dependent modulation of dendritic cell activity by circovirus type 2. Immunology 2005, 115, 388–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meier, O.; Greber, U.F. Adenovirus endocytosis. J. Gene Med. 2004, 6 (Suppl. 1), S152–S163. [Google Scholar] [CrossRef] [Green Version]
- Blanchard, E.; Belouzard, S.; Goueslain, L.; Wakita, T.; Dubuisson, J.; Wychowski, C.; Rouillé, Y. Hepatitis c virus entry depends on clathrin-mediated endocytosis. J. Virol. 2006, 80, 6964–6972. [Google Scholar] [CrossRef] [PubMed]
- Rust, M.J.; Lakadamyali, M.; Zhang, F.; Zhuang, X. Assembly of endocytic machinery around individual influenza viruses during viral entry. Nat. Struct. Mol. Biol. 2004, 11, 567–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nauwynck, H.J.; Duan, X.; Favoreel, H.W.; Van Oostveldt, P.; Pensaert, M.B. Entry of porcine reproductive and respiratory syndrome virus into porcine alveolar macrophages via receptor-mediated endocytosis. J. Gen. Virol. 1999, 80, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Saha, D.; Huang, L.; Bussalleu, E.; Lefebvre, D.J.; Fort, M.; Van Doorsselaere, J.; Nauwynck, H.J. Antigenic subtyping and epitopes’ competition analysis of porcine circovirus type 2 using monoclonal antibodies. Vet. Microbiol. 2012, 157, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Settembre, C.; Fraldi, A.; Medina, D.L.; Ballabio, A. Signals for the lysosome: A control center for cellular clearance and energy metabolism. Nat. Rev. Mol. Cell Biol. 2013, 14, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Hellevik, T.; Martinez, I.; Olsen, R.; Toh, B.H.; Webster, P.; Smedsrod, B. Transport of residual endocytosed products into terminal lysosomes occurs slowly in rat liver endothelial cells. Hepatology 1998, 28, 1378–1389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahraus, A.; Storrie, B.; Griffiths, G.; Desjardins, M. Evidence for retrograde traffic between terminal lysosomes and the prelysosomal/late endosome compartment. J. Cell Sci. 1994, 107, 145. [Google Scholar] [PubMed]
- Carrillo, C.; Borca, M.V.; Afonso, C.L.; Onisk, D.V.; Rock, D.L. Long-term persistent infection of swine monocytes/macrophages with African swine fever virus. J. Virol. 1994, 68, 580–583. [Google Scholar] [PubMed]
- Peluso, R.; Haase, A.; Stowring, L.; Edwards, M.; Ventura, P. A Trojan horse mechanism for the spread of visna virus in monocytes. Virology 1985, 147, 231–236. [Google Scholar] [CrossRef]
- Chu, Y.; Sperber, K.; Mayer, L.; Hsu, M.T. Persistent infection of human adenovirus type 5 in human monocyte cell lines. Virology 1992, 188, 793–800. [Google Scholar] [CrossRef]
- Lefebvre, D.J.; van Doorsselaere, J.; Delputte, P.L.; Nauwynck, H.J. Recombination of two porcine circovirus type 2 strains. Arch. Virol. 2009, 154, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Csagola, A.; Kecskemeti, S.; Kardos, G.; Kiss, I.; Tuboly, T. Genetic characterization of type 2 porcine circoviruses detected in Hungarian wild boars. Arch. Virol. 2006, 151, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Fenaux, M.; Halbur, P.G.; Haqshenas, G.; Royer, R.; Thomas, P.; Nawagitgul, P.; Gill, M.; Toth, T.E.; Meng, X.J. Cloned genomic DNA of type 2 porcine circovirus is infectious when injected directly into the liver and lymph nodes of pigs: Characterization of clinical disease, virus distribution, and pathologic lesions. J. Virol. 2002, 76, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.; Helenius, A. Virus entry: Open sesame. Cell 2006, 124, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Wiethoff, C.M.; Wodrich, H.; Gerace, L.; Nemerow, G.R. Adenovirus protein VI mediates membrane disruption following capsid disassembly. J. Virol. 2005, 79, 1992–2000. [Google Scholar] [CrossRef] [PubMed]
- Chandran, K.; Farsetta, D.L.; Nibert, M.L. Strategy for nonenveloped virus entry: A hydrophobic conformer of the reovirus membrane penetration protein micro 1 mediates membrane disruption. J. Virol. 2002, 76, 9920–9933. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, R.; Blaas, D. Uncoating of human rhinoviruses. Rev. Med. Virol. 2010, 20, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.; Schwartz, A.; Corey, S.; Orandle, M.; Kennedy, W.; Thompson, B.; Alvarez, X.; Brown, C.; Gartner, S.; Lackner, A. Proliferating cellular nuclear antigen expression as a marker of perivascular macrophages in simian immunodeficiency virus encephalitis. Am. J. Pathol. 2002, 161, 575–585. [Google Scholar] [CrossRef]
- Lin, C.-M.; Jeng, C.-R.; Hsiao, S.-H.; Lee, Y.; Tsai, Y.-C.; Chia, M.-Y.; Pang, V.F. Monocyte-derived dendritic cells enhance cell proliferation and porcine circovirus type 2 replication in concanavalin a-stimulated swine peripheral blood lymphocytes in vitro. Vet. Immunol. Immunopathol. 2012, 145, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Perez-Martin, E.; Rovira, A.; Calsamiglia, M.; Mankertz, A.; Rodriguez, F.; Segales, J. A new method to identify cell types that support porcine circovirus type 2 replication in formalin-fixed, paraffin-embedded swine tissues. J. Virol. Methods 2007, 146, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Meerts, P.; Misinzo, G.; McNeilly, F.; Nauwynck, H.J. Replication kinetics of different porcine circovirus 2 strains in PK-15 cells, fetal cardiomyocytes and macrophages. Arch. Virol. 2005, 150, 427–441. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.W.; Pang, V.F.; Chen, L.J.; Chia, M.Y.; Tsai, Y.C.; Jeng, C.R. Bacterial lipopolysaccharide induces porcine circovirus type 2 replication in swine alveolar macrophages. Vet. Microbiol. 2006, 115, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, L.T.; Mateu, E.; Sibila, M.; Fort, M.; Andaluz, A.; McNeilly, F.; Allan, G.; Sanchez, A.; Segales, J.; Stevenson, L. Lack of in vitro and in vivo effects of lipopolysaccharide on porcine circovirus type 2 infection. Viral Immunol. 2007, 20, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Opriessnig, T.; Patterson, A.R.; Meng, X.J.; Halbur, P.G. Porcine circovirus type 2 in muscle and bone marrow is infectious and transmissible to naive pigs by oral consumption. Vet. Microbiol. 2009, 133, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Dubin, G.; Toussaint, J.F.; Cassart, J.P.; Howe, B.; Boyce, D.; Friedland, L.; Abu-Elyazeed, R.; Poncelet, S.; Han, H.H.; Debrus, S. Investigation of a regulatory agency enquiry into potential porcine circovirus type 1 contamination of the human rotavirus vaccine, rotarix: Approach and outcome. Hum. Vaccin Immunother. 2013, 9, 2398–2408. [Google Scholar] [CrossRef] [PubMed]
- Esona, M.D.; Mijatovic-Rustempasic, S.; Yen, C.; Parashar, U.D.; Gentsch, J.R.; Bowen, M.D.; LaRussa, P. Detection of PCV-2 DNA in stool samples from infants vaccinated with rotateq(R). Hum. Vaccin Immunother. 2014, 10, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Burbelo, P.D.; Ragheb, J.A.; Kapoor, A.; Zhang, Y. The serological evidence in humans supports a negligible risk of zoonotic infection from porcine circovirus type 2. Biologicals 2013, 41, 430–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hattermann, K.; Roedner, C.; Schmitt, C.; Finsterbusch, T.; Steinfeldt, T.; Mankertz, A. Infection studies on human cell lines with porcine circovirus type 1 and porcine circovirus type 2. Xenotransplantation 2004, 11, 284–294. [Google Scholar] [CrossRef] [PubMed]




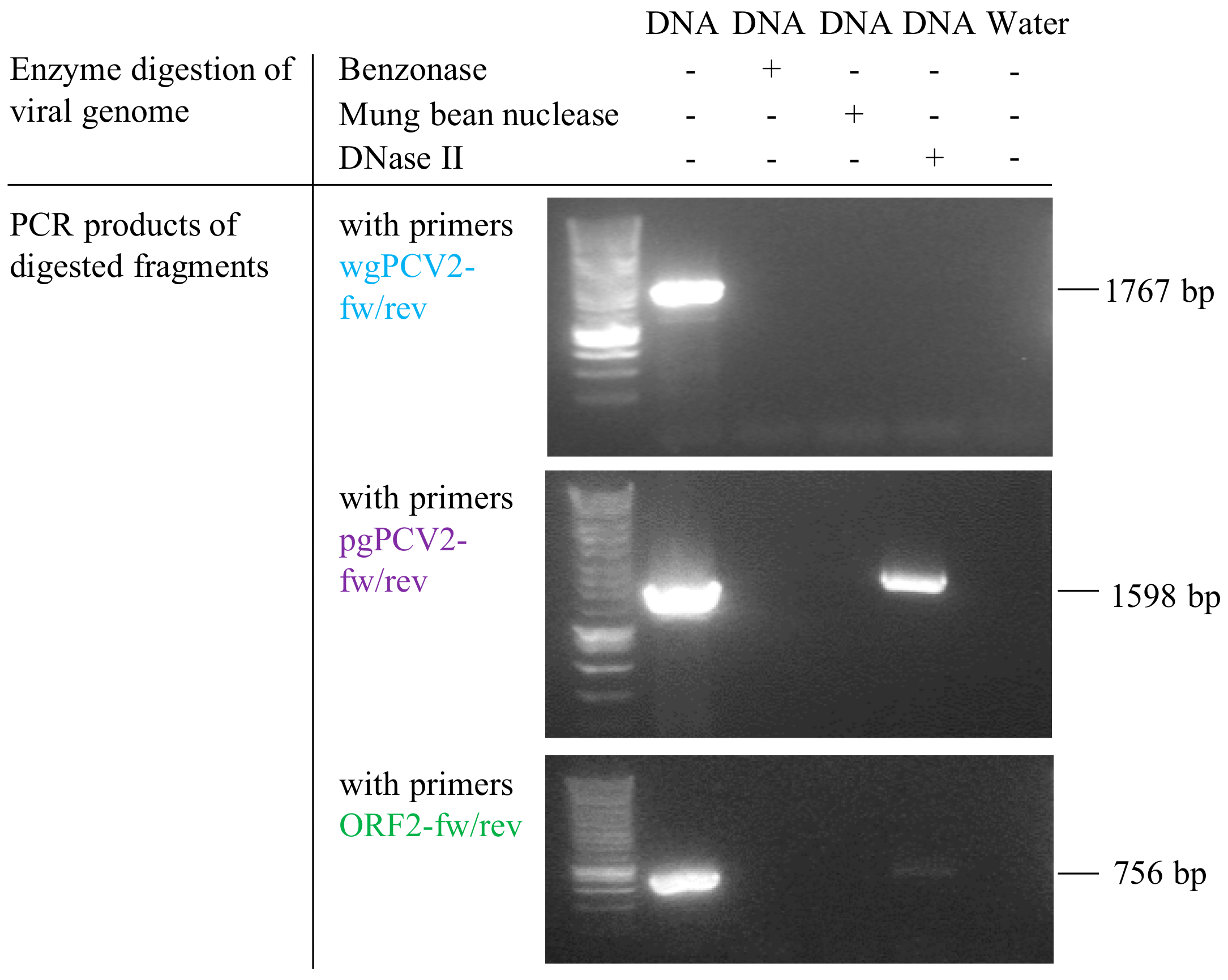
| Primer Set | Forward Primer (5′ to 3′) | Reverse Primer (5′ to 3′) | Product |
| wgPCV2-fw/rev | ggaagcttcagtaatttatttcatatggaa | ggaagcttttttatcacttcgtaatggtt | 1767 bp (whole PCV2 genome) |
| ORF2-fw/rev | gcgcacttcttttcgttttcag | gaatgcggccgcttatcacttcgtaatggtttttattattca | 756 bp (DNase II cleavage site included) |
| pgPCV2-fw/rev | ggctgtggcctttgktac | tgtrgaccacgtaggcctcg | 1598 bp (DNase II cleavage site not included) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, R.; Trus, I.; Yang, B.; Huang, L.; Nauwynck, H.J. Breed Differences in PCV2 Uptake and Disintegration in Porcine Monocytes. Viruses 2018, 10, 562. https://doi.org/10.3390/v10100562
Wei R, Trus I, Yang B, Huang L, Nauwynck HJ. Breed Differences in PCV2 Uptake and Disintegration in Porcine Monocytes. Viruses. 2018; 10(10):562. https://doi.org/10.3390/v10100562
Chicago/Turabian StyleWei, Ruifang, Ivan Trus, Bo Yang, Liping Huang, and Hans J. Nauwynck. 2018. "Breed Differences in PCV2 Uptake and Disintegration in Porcine Monocytes" Viruses 10, no. 10: 562. https://doi.org/10.3390/v10100562
APA StyleWei, R., Trus, I., Yang, B., Huang, L., & Nauwynck, H. J. (2018). Breed Differences in PCV2 Uptake and Disintegration in Porcine Monocytes. Viruses, 10(10), 562. https://doi.org/10.3390/v10100562





