HIV-1 Integrase-DNA Recognition Mechanisms
Abstract
:1. Introduction
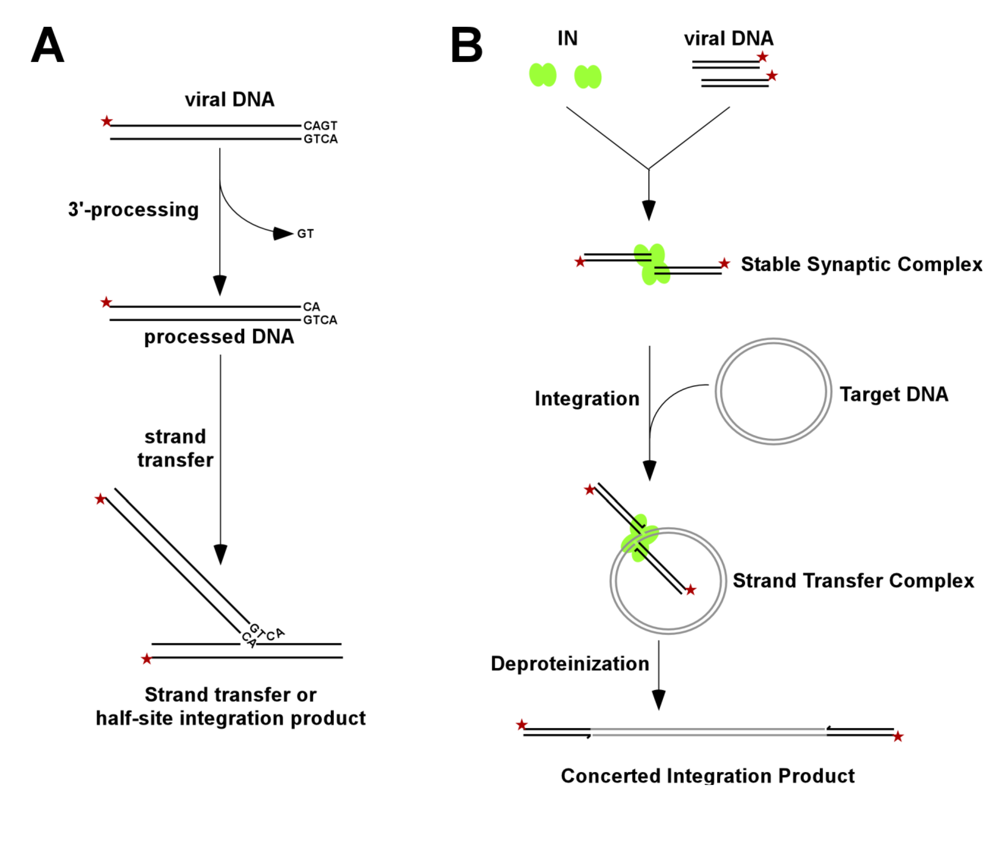
2. DNA Processing and Joining Reactions Catalyzed by HIV-1 IN
3. Structure of HIV-1 Integrase
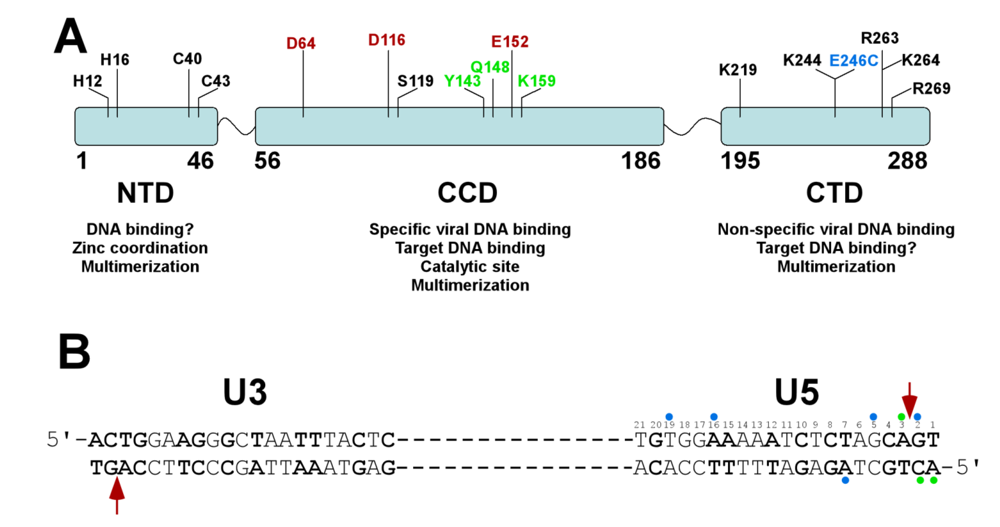
4. Sequence and Structure of Viral DNA
5. Mapping HIV-1 IN-Viral DNA Interactions
6. HIV-1 IN Interactions with the Target DNA
7. Concerted Integration Intermediates
8. LEDGF/p75 Strongly Modulates HIV-1 IN-DNA Interactions
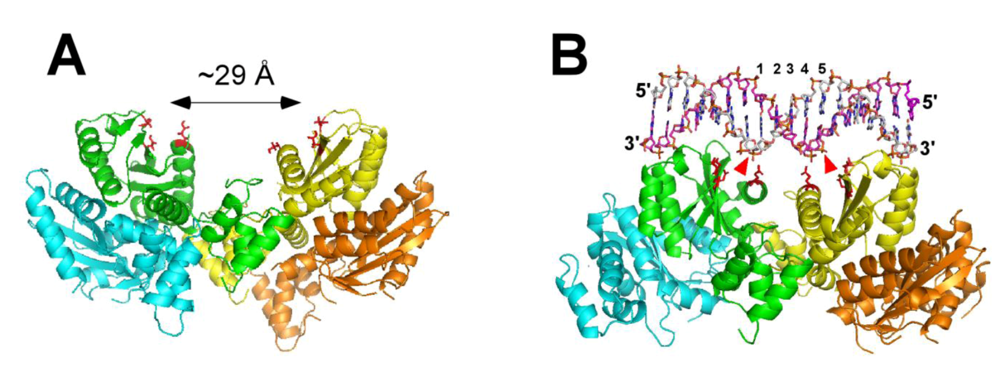
9. Molecular Modeling of the Functional Nucleoprotein Complexes
10. Remaining Questions and Outlook
Acknowledgments
References and Notes
- Brown, P.O. Integration. In Retroviruses; Coffin, J.M., Hughes, S.H., Varmus, H.E., Eds.; 1997; Cold Spring Harbor Laboratory: Plainview, NY, USA; pp. 161–204. [Google Scholar]
- Grobler, J.A.; Stillmock, K.; Hu, B.; Witmer, M.; Felock, P.; Espeseth, A.S.; Wolfe, A.; Egbertson, M.; Bourgeois, M.; Melamed, J.; Wai, J.S.; Young, S.; Vacca, J.; Hazuda, D.J. Diketo acid inhibitor mechanism and HIV-1 integrase: Implications for metal binding in the active site of phosphotransferase enzymes. Proc. Natl. Acad. Sci. USA 2002, 99, 6661–6666. [Google Scholar] [CrossRef]
- Hazuda, D.J.; Felock, P.; Witmer, M.; Wolfe, A.; Stillmock, K.; Grobler, J.A.; Espeseth, A.; Gabryelski, L.; Schleif, W.; Blau, C.; Miller, M.D. Inhibitors of strand transfer that prevent integration and inhibit HIV- 1 replication in cells. Science 2000, 287, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Summa, V.; Petrocchi, A.; Bonelli, F.; Crescenzi, B.; Donghi, M.; Ferrara, M.; Fiore, F.; Gardelli, C.; Gonzalez Paz, O.; Hazuda, D.J.; Jones, P.; Kinzel, O.; Laufer, R.; Monteagudo, E.; Muraglia, E.; Nizi, E.; Orvieto, F.; Pace, P.; Pescatore, G.; Scarpelli, R.; Stillmock, K.; Witmer, M.V.; Rowley, M. Discovery of raltegravir, a potent, selective orally bioavailable HIV-integrase inhibitor for the treatment of HIV-AIDS infection. J. Med. Chem. 2008, 51, 5843–5855. [Google Scholar] [CrossRef] [PubMed]
- Llano, M.; Saenz, D.T.; Meehan, A.; Wongthida, P.; Peretz, M.; Walker, W.H.; Teo, W.; Poeschla, E.M. An essential role for LEDGF/p75 in HIV integration. Science 2006, 314, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Lewinski, M.K.; Yamashita, M.; Emerman, M.; Ciuffi, A.; Marshall, H.; Crawford, G.; Collins, F.; Shinn, P.; Leipzig, J.; Hannenhalli, S.; Berry, C.C.; Ecker, J.R.; Bushman, F.D. Retroviral DNA integration: Viral and cellular determinants of target-site selection . PLoS Pathog. 2006, 2, e60. [Google Scholar] [CrossRef] [PubMed]
- Farnet, C.M.; Haseltine, W.A. Integration of human immunodeficiency virus type 1 DNA in vitro. Proc. Natl. Acad. Sci. USA 1990, 87, 4164–4168. [Google Scholar] [CrossRef]
- Bukrinsky, M.I.; Sharova, N.; McDonald, T.L.; Pushkarskaya, T.; Tarpley, W.G.; Stevenson, M. Association of integrase, matrix, and reverse transcriptase antigens of human immunodeficiency virus type 1 with viral nucleic acids following acute infection. Proc. Natl. Acad. Sci. USA 1993, 90, 6125–6129. [Google Scholar] [CrossRef]
- Buckman, J.S.; Bosche, W.J.; Gorelick, R.J. Human immunodeficiency virus type 1 nucleocapsid zn(2+) fingers are required for efficient reverse transcription, initial integration processes, and protection of newly synthesized viral DNA. J. Virol. 2003, 77, 1469–1480. [Google Scholar] [CrossRef] [PubMed]
- Carteau, S.; Batson, S.C.; Poljak, L.; Mouscadet, J.F.; de Rocquigny, H.; Darlix, J.L.; Roques, B.P.; Kas, E.; Auclair, C. Human immunodeficiency virus type 1 nucleocapsid protein specifically stimulates Mg2+-dependent DNA integration in vitro. J. Virol. 1997, 71, 6225–6229. [Google Scholar] [PubMed]
- Carteau, S.; Gorelick, R.J.; Bushman, F.D. Coupled integration of human immunodeficiency virus type 1 cDNA ends by purified integrase in vitro: Stimulation by the viral nucleocapsid protein. J. Virol. 1999, 73, 6670–6679. [Google Scholar] [PubMed]
- Chen, H.; Engelman, A. The barrier-to-autointegration protein is a host factor for HIV type 1 integration. Proc. Natl. Acad. Sci. USA 1998, 95, 15270–15274. [Google Scholar] [CrossRef]
- Lee, M.S.; Craigie, R. A previously unidentified host protein protects retroviral DNA from autointegration. Proc. Natl. Acad. Sci. USA 1998, 95, 1528–1533. [Google Scholar] [CrossRef]
- Cherepanov, P.; Maertens, G.; Proost, P.; Devreese, B.; Van Beeumen, J.; Engelborghs, Y.; De Clercq, E.; Debyser, Z. HIV-1 integrase forms stable tetramers and associates with LEDGF/p75 protein in human cells. J. Biol. Chem. 2003, 278, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Shun, M.C.; Raghavendra, N.K.; Vandegraaff, N.; Daigle, J.E.; Hughes, S.; Kellam, P.; Cherepanov, P.; Engelman, A. LEDGF/p75 functions downstream from preintegration complex formation to effect gene-specific HIV-1 integration. Genes Dev. 2007, 21, 1767–1778. [Google Scholar] [CrossRef] [PubMed]
- Engelman, A. Host cell factors and HIV-1 integration. Future HIV Ther. 2007, 1, 415–426. [Google Scholar] [CrossRef]
- Lee, M.S.; Craigie, R. Protection of retroviral DNA from autointegration: Involvement of a cellular factor. Proc. Natl. Acad. Sci. USA 1994, 91, 9823–9827. [Google Scholar] [CrossRef]
- Wu, W.; Henderson, L.E.; Copeland, T.D.; Gorelick, R.J.; Bosche, W.J.; Rein, A.; Levin, J.G. Human immunodeficiency virus type 1 nucleocapsid protein reduces reverse transcriptase pausing at a secondary structure near the murine leukemia virus polypurine tract. J. Virol. 1996, 70, 7132–7142. [Google Scholar] [PubMed]
- Chen, H.; Wei, S.Q.; Engelman, A. Multiple integrase functions are required to form the native structure of the human immunodeficiency virus type I intasome. J. Biol. Chem. 1999, 274, 17358–17364. [Google Scholar] [CrossRef] [PubMed]
- Bowerman, B.; Brown, P.O.; Bishop, J.M.; Varmus, H.E. A nucleoprotein complex mediates the integration of retroviral DNA. Genes Dev. 1989, 3, 469–478. [Google Scholar] [CrossRef]
- Brown, P.O.; Bowerman, B.; Varmus, H.E.; Bishop, J.M. Correct integration of retroviral DNA in vitro. Cell 1987, 49, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, T.; Mizuuchi, K. Retroviral DNA integration: Structure of an integration intermediate. Cell 1988, 54, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Ellison, V.; Abrams, H.; Roe, T.; Lifson, J.; Brown, P. Human immunodeficiency virus integration in a cell-free system. J. Virol. 1990, 64, 2711–2715. [Google Scholar] [PubMed]
- Lee, Y.M.; Coffin, J.M. Efficient autointegration of avian retrovirus DNA in vitro. J. Virol. 1990, 64, 5958–5965. [Google Scholar] [PubMed]
- Engelman, A. Isolation and analysis of HIV-1 preintegration complexes. Methods Mol. Biol. 2009, 485, 135–149. [Google Scholar] [PubMed]
- Li, M.; Craigie, R. Processing of viral DNA ends channels the HIV-1 integration reaction to concerted integration. J. Biol. Chem. 2005, 280, 29334–29339. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Mizuuchi, M.; Burke, T.R.; Craigie, R. Retroviral DNA integration: Reaction pathway and critical intermediates . Embo J. 2006, 25, 1295–1304. [Google Scholar] [PubMed]
- Li, M.; Craigie, R. Nucleoprotein complex intermediates in HIV-1 integration. Methods 2009, 47, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Pursley, M.H.; Grandgenett, D.P. Efficient concerted integration by recombinant human immunodeficiency virus type 1 integrase without cellular or viral cofactors. J. Virol. 2002, 76, 3105–3113. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.K.; Sinha, S.; Grandgenett, D.P. Transcriptional coactivator LEDGF/p75 modulates human immunodeficiency virus type 1 integrase-mediated concerted integration. J. Virol. 2007, 81, 3969–3979. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Grandgenett, D.P. Recombinant human immunodeficiency virus type 1 integrase exhibits a capacity for full-site integration in vitro that is comparable to that of purified preintegration complexes from virus-infected cells. J. Virol. 2005, 79, 8208–8216. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.A.; Vincent, K.A.; Ellison, V.; Brown, P.O. Reversal of integration and DNA splicing mediated by integrase of human immunodeficiency virus. Science 1992, 255, 723–726. [Google Scholar] [PubMed]
- Delelis, O.; Parissi, V.; Leh, H.; Mbemba, G.; Petit, C.; Sonigo, P.; Deprez, E.; Mouscadet, J.F. Efficient and specific internal cleavage of a retroviral palindromic DNA sequence by tetrameric HIV-1 integrase . PLoS One 2007, 2, e608. [Google Scholar] [CrossRef] [PubMed]
- Delelis, O.; Carayon, K.; Saib, A.; Deprez, E.; Mouscadet, J.F. Integrase and integration: Biochemical activities of HIV-1 integrase. Retrovirology 2008, 5, 114. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Zheng, R.; Caffrey, M.; Craigie, R.; Clore, G.M.; Gronenborn, A.M. Solution structure of the N-terminal zinc binding domain of HIV-1 integrase. Nat. Struct. Biol. 1997, 4, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Dyda, F.; Hickman, A.B.; Jenkins, T.M.; Engelman, A.; Craigie, R.; Davies, D.R. Crystal structure of the catalytic domain of HIV-1 integrase: Similarity to other polynucleotidyl transferases. Science 1994, 266, 1981–1986. [Google Scholar] [PubMed]
- Goldgur, Y.; Dyda, F.; Hickman, A.B.; Jenkins, T.M.; Craigie, R.; Davies, D.R. Three new structures of the core domain of HIV-1 integrase: An active site that binds magnesium. Proc. Natl. Acad. Sci. USA 1998, 95, 9150–9154. [Google Scholar] [CrossRef]
- Eijkelenboom, A.P.; Lutzke, R.A.; Boelens, R.; Plasterk, R.H.; Kaptein, R.; Hard, K. The DNA-binding domain of HIV-1 integrase has an SH3-like fold. Nat. Struct. Biol. 1995, 2, 807–810. [Google Scholar] [CrossRef]
- Lodi, P.J.; Ernst, J.A.; Kuszewski, J.; Hickman, A.B.; Engelman, A.; Craigie, R.; Clore, G.M.; Gronenborn, A.M. Solution structure of the DNA binding domain of HIV-1 integrase. Biochemistry 1995, 34, 9826–9833. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.C.; Krucinski, J.; Miercke, L.J.; Finer-Moore, J.S.; Tang, A.H.; Leavitt, A.D.; Stroud, R.M. Crystal structure of the HIV-1 integrase catalytic core and C-terminal domains: A model for viral DNA binding. Proc. Natl. Acad. Sci. USA 2000, 97, 8233–8238. [Google Scholar] [CrossRef]
- Wang, J.Y.; Ling, H.; Yang, W.; Craigie, R. Structure of a two-domain fragment of HIV-1 integrase: Implications for domain organization in the intact protein. Embo J. 2001, 20, 7333–7343. [Google Scholar] [CrossRef] [PubMed]
- Goldgur, Y.; Craigie, R.; Cohen, G.H.; Fujiwara, T.; Yoshinaga, T.; Fujishita, T.; Sugimoto, H.; Endo, T.; Murai, H.; Davies, D.R. Structure of the HIV-1 integrase catalytic domain complexed with an inhibitor: A platform for antiviral drug design. Proc. Natl. Acad. Sci. USA 1999, 96, 13040–13043. [Google Scholar] [CrossRef]
- Bushman, F.D.; Engelman, A.; Palmer, I.; Wingfield, P.; Craigie, R. Domains of the integrase protein of human immunodeficiency virus type 1 responsible for polynucleotidyl transfer and zinc binding. Proc. Natl. Acad. Sci. USA 1993, 90, 3428–3432. [Google Scholar] [CrossRef]
- Zheng, R.; Jenkins, T.M.; Craigie, R. Zinc folds the N-terminal domain of HIV-1 integrase, promotes multimerization, and enhances catalytic activity. Proc. Natl. Acad. Sci. USA 1996, 93, 13659–13664. [Google Scholar] [CrossRef]
- Lee, S.P.; Xiao, J.; Knutson, J.R.; Lewis, M.S.; Han, M.K. Zn2+ promotes the self-association of human immunodeficiency virus type-1 integrase in vitro. Biochemistry 1997, 36, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Engelman, A.; Craigie, R. Identification of conserved amino acid residues critical for human immunodeficiency virus type 1 integrase function in vitro. J. Virol. 1992, 66, 6361–6369. [Google Scholar] [PubMed]
- Hare, S.; Di Nunzio, F.; Labeja, A.; Wang, J.; Engelman, A.; Cherepanov, P. Structural basis for functional tetramerization of lentiviral integrase . PLoS Pathog. 2009, 5, e1000515. [Google Scholar] [CrossRef] [PubMed]
- McKee, C.J.; Kessl, J.J.; Shkriabai, N.; Dar, M.J.; Engelman, A.; Kvaratskhelia, M. Dynamic modulation of HIV-1 integrase structure and function by cellular lens epithelium-derived growth factor (LEDGF) protein. J. Biol. Chem. 2008, 283, 31802–31812. [Google Scholar] [CrossRef] [PubMed]
- Fayet, O.; Ramond, P.; Polard, P.; Prere, M.F.; Chandler, M. Functional similarities between retroviruses and the IS3 family of bacterial insertion sequences? Mol. Microbiol. 1990, 4, 1771–1777. [Google Scholar] [CrossRef]
- Kulkosky, J.; Jones, K.S.; Katz, R.A.; Mack, J.P.; Skalka, A.M. Residues critical for retroviral integrative recombination in a region that is highly conserved among retroviral/retrotransposon integrases and bacterial insertion sequence transposases. Mol. Cell. Biol. 1992, 12, 2331–2338. [Google Scholar] [PubMed]
- Hazuda, D.J.; Wolfe, A.L.; Hastings, J.C.; Robbins, H.L.; Graham, P.L.; LaFemina, R.L.; Emini, E.A. Viral long terminal repeat substrate binding characteristics of the human immunodeficiency virus type 1 integrase. J. Biol. Chem. 1994, 269, 3999–4004. [Google Scholar] [PubMed]
- Leavitt, A.D.; Robles, G.; Alesandro, N.; Varmus, H.E. Human immunodeficiency virus type 1 integrase mutants retain in vitro integrase activity yet fail to integrate viral DNA efficiently during infection. J. Virol. 1996, 70, 721–728. [Google Scholar] [PubMed]
- Yang, W.; Steitz, T.A. Recombining the structures of HIV integrase, RuvC and RNase H. Structure 1995, 3, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Beese, L.S.; Steitz, T.A. Structural basis for the 3'-5' exonuclease activity of Escherichia coli DNA polymerase I: A two metal ion mechanism. Embo J. 1991, 10, 25–33. [Google Scholar] [PubMed]
- Lins, R.D.; Straatsma, T.P.; Briggs, J.M. Similarities in the HIV-1 and ASV integrase active sites upon metal cofactor binding. Biopolymers 2000, 53, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Robinson, H.; Gao, Y.G.; McCrary, B.S.; Edmondson, S.P.; Shriver, J.W.; Wang, A.H. The hyperthermophile chromosomal protein Sac7d sharply kinks DNA. Nature 1998, 392, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Krueger, J.K.; McCrary, B.S.; Wang, A.H.; Shriver, J.W.; Trewhella, J.; Edmondson, S.P. The solution structure of the Sac7d/DNA complex: A small-angle X-ray scattering study. Biochemistry 1999, 38, 10247–10255. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.G.; Su, S.Y.; Robinson, H.; Padmanabhan, S.; Lim, L.; McCrary, B.S.; Edmondson, S.P.; Shriver, J.W.; Wang, A.H. The crystal structure of the hyperthermophile chromosomal protein Sso7d bound to DNA. Nat. Struct. Biol. 1998, 5, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Lutzke, R.A.; Plasterk, R.H. Structure-based mutational analysis of the C-terminal DNA-binding domain of human immunodeficiency virus type 1 integrase: Critical residues for protein oligomerization and DNA binding. J. Virol. 1998, 72, 4841–4848. [Google Scholar] [PubMed]
- Esposito, D.; Craigie, R. Sequence specificity of viral end DNA binding by HIV-1 integrase reveals critical regions for protein-DNA interaction. Embo J. 1998, 17, 5832–5843. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; McKee, C.J.; Kessl, J.J.; Santos, W.L.; Daigle, J.E.; Engelman, A.; Verdine, G.; Kvaratskhelia, M. Subunit-specific protein footprinting reveals significant structural rearrangements and a role for N-terminal Lys-14 of HIV-1 Integrase during viral DNA binding. J. Biol. Chem. 2008, 283, 5632–5641. [Google Scholar] [CrossRef] [PubMed]
- Bera, S.; Pandey, K.K.; Vora, A.C.; Grandgenett, D.P. Molecular Interactions between HIV-1 integrase and the two viral DNA ends within the synaptic complex that mediates concerted integration. J. Mol. Biol. 2009, 389, 183–198. [Google Scholar] [CrossRef] [PubMed]
- LaFemina, R.L.; Callahan, P.L.; Cordingley, M.G. Substrate specificity of recombinant human immunodeficiency virus integrase protein. J. Virol. 1991, 65, 5624–5630. [Google Scholar] [PubMed]
- van Gent, D.C.; Elgersma, Y.; Bolk, M.W.; Vink, C.; Plasterk, R.H. DNA binding properties of the integrase proteins of human immunodeficiency viruses types 1 and 2. Nucleic Acids Res. 1991, 19, 3821–3827. [Google Scholar] [CrossRef] [PubMed]
- Smerdon, S.J.; Jager, J.; Wang, J.; Kohlstaedt, L.A.; Chirino, A.J.; Friedman, J.M.; Rice, P.A.; Steitz, T.A. Structure of the binding site for nonnucleoside inhibitors of the reverse transcriptase of human immunodeficiency virus type 1. Proc. Natl. Acad. Sci. USA 1994, 91, 3911–3915. [Google Scholar] [CrossRef]
- Bujacz, G.; Alexandratos, J.; Wlodawer, A.; Merkel, G.; Andrake, M.; Katz, R.A.; Skalka, A.M. Binding of different divalent cations to the active site of avian sarcoma virus integrase and their effects on enzymatic activity. J. Biol. Chem. 1997, 272, 18161–18168. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Wong, S.; Bushman, F. Metal binding by the D,DX35E motif of human immunodeficiency virus type 1 integrase: Selective rescue of Cys substitutions by Mn2+ in vitro. J. Virol. 2004, 78, 6715–6722. [Google Scholar] [CrossRef] [PubMed]
- Hazuda, D.J.; Felock, P.J.; Hastings, J.C.; Pramanik, B.; Wolfe, A.L. Differential divalent cation requirements uncouple the assembly and catalytic reactions of human immunodeficiency virus type 1 integrase. J. Virol. 1997, 71, 7005–7011. [Google Scholar] [PubMed]
- Lee, S.P.; Censullo, M.L.; Kim, H.G.; Han, M.K. Substrate-length-dependent activities of human immunodeficiency virus type 1 integrase in vitro: Differential DNA binding affinities associated with different lengths of substrates. Biochemistry 1995, 34, 10215–10223. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Asante-Appiah, E.; Skalka, A.M. Divalent cations stimulate preferential recognition of a viral DNA end by HIV-1 integrase. Biochemistry 1999, 38, 8458–8468. [Google Scholar] [CrossRef] [PubMed]
- Vercammen, J.; Maertens, G.; Gerard, M.; De Clercq, E.; Debyser, Z.; Engelborghs, Y. DNA-induced polymerization of HIV-1 integrase analyzed with fluorescence fluctuation spectroscopy. J. Biol. Chem. 2002, 277, 38045–38052. [Google Scholar] [CrossRef] [PubMed]
- Brin, E.; Leis, J. HIV-1 integrase interaction with U3 and U5 terminal sequences in vitro defined using substrates with random sequences. J. Biol. Chem. 2002, 277, 18357–18364. [Google Scholar] [CrossRef] [PubMed]
- Renisio, J.G.; Cosquer, S.; Cherrak, I.; El Antri, S.; Mauffret, O.; Fermandjian, S. Pre-organized structure of viral DNA at the binding-processing site of HIV-1 integrase. Nucleic Acids Res. 2005, 33, 1970–1981. [Google Scholar] [CrossRef] [PubMed]
- Katz, R.A.; DiCandeloro, P.; Kukolj, G.; Skalka, A.M. Role of DNA end distortion in catalysis by avian sarcoma virus integrase. J. Biol. Chem. 2001, 276, 34213–34220. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Sayer, J.M.; Yagi, H.; Patil, S.S.; Debart, F.; Maier, M.A.; Corey, D.R.; Vasseur, J.J.; Burke, T.R.; Marquez, V.E.; Jerina, D.M.; Pommier, Y. Effect of DNA modifications on DNA processing by HIV-1 integrase and inhibitor binding: Role of DNA backbone flexibility and an open catalytic site . J. Biol. Chem. 2006, 281, 32428–32438. [Google Scholar] [PubMed]
- Agapkina, J.; Smolov, M.; Barbe, S.; Zubin, E.; Zatsepin, T.; Deprez, E.; Le Bret, M.; Mouscadet, J.F.; Gottikh, M. Probing of HIV-1 integrase/DNA interactions using novel analogs of viral DNA. J. Biol. Chem. 2006, 281, 11530–11540. [Google Scholar] [CrossRef] [PubMed]
- Engelman, A.; Bushman, F.D.; Craigie, R. Identification of discrete functional domains of HIV-1 integrase and their organization within an active multimeric complex. Embo J. 1993, 12, 3269–3275. [Google Scholar] [PubMed]
- van den Ent, F.M.; Vos, A.; Plasterk, R.H. Dissecting the role of the N-terminal domain of human immunodeficiency virus integrase by trans-complementation analysis. J. Virol. 1999, 73, 3176–3183. [Google Scholar] [PubMed]
- van Gent, D.C.; Vink, C.; Groeneger, A.A.; Plasterk, R.H. Complementation between HIV integrase proteins mutated in different domains. Embo J. 1993, 12, 3261–3267. [Google Scholar] [PubMed]
- Vincent, K.A.; Ellison, V.; Chow, S.A.; Brown, P.O. Characterization of human immunodeficiency virus type 1 integrase expressed in Escherichia coli and analysis of variants with amino-terminal mutations. J. Virol. 1993, 67, 425–437. [Google Scholar] [PubMed]
- Jenkins, T.M.; Engelman, A.; Ghirlando, R.; Craigie, R. A soluble active mutant of HIV-1 integrase: Involvement of both the core and carboxyl-terminal domains in multimerization. J. Biol. Chem. 1996, 271, 7712–7718. [Google Scholar] [CrossRef] [PubMed]
- Deprez, E.; Tauc, P.; Leh, H.; Mouscadet, J.F.; Auclair, C.; Brochon, J.C. Oligomeric states of the HIV-1 integrase as measured by time-resolved fluorescence anisotropy. Biochemistry 2000, 39, 9275–9284. [Google Scholar] [CrossRef] [PubMed]
- Deprez, E.; Barbe, S.; Kolaski, M.; Leh, H.; Zouhiri, F.; Auclair, C.; Brochon, J.C.; Le Bret, M.; Mouscadet, J.F. Mechanism of HIV-1 integrase inhibition by styrylquinoline derivatives in vitro. Mol. Pharmacol. 2004, 65, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Baranova, S.; Tuzikov, F.V.; Zakharova, O.D.; Tuzikova, N.A.; Calmels, C.; Litvak, S.; Tarrago-Litvak, L.; Parissi, V.; Nevinsky, G.A. Small-angle X-ray characterization of the nucleoprotein complexes resulting from DNA-induced oligomerization of HIV-1 integrase. Nucleic Acids Res. 2007, 35, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Faure, A.; Calmels, C.; Desjobert, C.; Castroviejo, M.; Caumont-Sarcos, A.; Tarrago-Litvak, L.; Litvak, S.; Parissi, V. HIV-1 integrase crosslinked oligomers are active in vitro. Nucleic Acids Res. 2005, 33, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Guiot, E.; Carayon, K.; Delelis, O.; Simon, F.; Tauc, P.; Zubin, E.; Gottikh, M.; Mouscadet, J.F.; Brochon, J.C.; Deprez, E. Relationship between the oligomeric status of HIV-1 integrase on DNA and enzymatic activity. J. Biol. Chem. 2006, 281, 22707–22719. [Google Scholar] [CrossRef] [PubMed]
- Bao, K.K.; Wang, H.; Miller, J.K.; Erie, D.A.; Skalka, A.M.; Wong, I. Functional oligomeric state of avian sarcoma virus integrase. J. Biol. Chem. 2003, 278, 1323–1327. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Gao, K.; Bushman, F.D.; Yeager, M. Single-particle image reconstruction of a tetramer of HIV integrase bound to DNA. J. Mol. Biol. 2007, 366, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Heuer, T.S.; Brown, P.O. Photo-cross-linking studies suggest a model for the architecture of an active human immunodeficiency virus type 1 integrase-DNA complex. Biochemistry 1998, 37, 6667–6678. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.M.; Esposito, D.; Engelman, A.; Craigie, R. Critical contacts between HIV-1 integrase and viral DNA identified by structure-based analysis and photo-crosslinking. Embo J. 1997, 16, 6849–6859. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, A.; Neamati, N.; Pilon, A.A.; Sunder, S.; Pommier, Y. Chemical trapping of ternary complexes of human immunodeficiency virus type 1 integrase, divalent metal, and DNA substrates containing an abasic site. Implications for the role of lysine 136 in DNA binding. J. Biol. Chem. 1996, 271, 27330–27338. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Santos, W.; Pais, G.C.; Marchand, C.; Amin, R.; Burke, T.R.; Verdine, G.; Pommier, Y. Integration requires a specific interaction of the donor DNA terminal 5'-cytosine with glutamine 148 of the HIV-1 integrase flexible loop . J. Biol. Chem. 2006, 281, 461–467. [Google Scholar] [PubMed]
- Heuer, T.S.; Brown, P.O. Mapping features of HIV-1 integrase near selected sites on viral and target DNA molecules in an active enzyme-DNA complex by photo-cross- linking. Biochemistry 1997, 36, 10655–10665. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Butler, S.L.; Bushman, F. Human immunodeficiency virus type 1 integrase: Arrangement of protein domains in active cDNA complexes. Embo J. 2001, 20, 3565–3576. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Weber, I.T.; Harrison, R.W.; Leis, J. Identification of amino acids in HIV-1 and avian sarcoma virus integrase subsites required for specific recognition of the long terminal repeat Ends. J. Biol. Chem. 2006, 281, 4173–4182. [Google Scholar] [CrossRef] [PubMed]
- Dolan, J.; Chen, A.; Weber, I.T.; Harrison, R.W.; Leis, J. Defining the DNA substrate binding sites on HIV-1 integrase. J. Mol. Biol. 2009, 385, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Limon, A.; Devroe, E.; Silver, P.A.; Cherepanov, P.; Engelman, A. Class II integrase mutants with changes in putative nuclear localization signals are primarily blocked at a postnuclear entry step of human immunodeficiency virus type 1 replication. J. Virol. 2004, 78, 12735–12746. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Limon, A.; Ghory, H.Z.; Engelman, A. Genetic analyses of DNA-binding mutants in the catalytic core domain of human immunodeficiency virus type 1 integrase. J. Virol. 2005, 79, 2493–2505. [Google Scholar] [CrossRef] [PubMed]
- Asante-Appiah, E.; Skalka, A.M. A metal-induced conformational change and activation of HIV-1 integrase. J. Biol. Chem. 1997, 272, 16196–16205. [Google Scholar] [CrossRef] [PubMed]
- Puglia, J.; Wang, T.; Smith-Snyder, C.; Cote, M.; Scher, M.; Pelletier, J.N.; John, S.; Jonsson, C.B.; Roth, M.J. Revealing domain structure through linker-scanning analysis of the murine leukemia virus (MuLV) RNase H and MuLV and human immunodeficiency virus type 1 integrase proteins. J. Virol. 2006, 80, 9497–9510. [Google Scholar] [CrossRef] [PubMed]
- Bor, Y.C.; Miller, M.D.; Bushman, F.D.; Orgel, L.E. Target-sequence preferences of HIV-1 integration complexes in vitro. Virology 1996, 222, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Goodarzi, G.; Chiu, R.; Brackmann, K.; Kohn, K.; Pommier, Y.; Grandgenett, D.P. Host site selection for concerted integration by human immunodeficiency virus type-1 virions in vitro. Virology 1997, 231, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, M.L.; Grandgenett, D.P. Retroviral integration: In vitro host site selection by avian integrase. J. Virol. 1994, 68, 4314–4321. [Google Scholar] [PubMed]
- Carteau, S.; Hoffmann, C.; Bushman, F. Chromosome structure and human immunodeficiency virus type 1 cDNA integration: Centromeric alphoid repeats are a disfavored target. J. Virol. 1998, 72, 4005–4014. [Google Scholar] [PubMed]
- Holman, A.G.; Coffin, J.M. Symmetrical base preferences surrounding HIV-1, avian sarcoma/leukosis virus, and murine leukemia virus integration sites. Proc. Natl. Acad. Sci. USA 2005, 102, 6103–6107. [Google Scholar] [CrossRef]
- Stevens, S.W.; Griffith, J.D. Sequence analysis of the human DNA flanking sites of human immunodeficiency virus type 1 integration. J. Virol. 1996, 70, 6459–6462. [Google Scholar] [PubMed]
- Wu, X.; Li, Y.; Crise, B.; Burgess, S.M.; Munroe, D.J. Weak palindromic consensus sequences are a common feature found at the integration target sites of many retroviruses. J. Virol. 2005, 79, 5211–5214. [Google Scholar] [CrossRef] [PubMed]
- Ciuffi, A.; Bushman, F.D. Retroviral DNA integration: HIV and the role of LEDGF/p75. Trends Genet. 2006, 22, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Engelman, A.; Cherepanov, P. The lentiviral integrase binding protein LEDGF/p75 and HIV-1 replication . PLoS Pathog. 2008, 4, e1000046. [Google Scholar] [CrossRef] [PubMed]
- Pruss, D.; Reeves, R.; Bushman, F.D.; Wolffe, A.P. The influence of DNA and nucleosome structure on integration events directed by HIV integrase. J. Biol. Chem. 1994, 269, 25031–25041. [Google Scholar] [PubMed]
- Pruss, D.; Bushman, F.D.; Wolffe, A.P. Human immunodeficiency virus integrase directs integration to sites of severe DNA distortion within the nucleosome core. Proc. Natl. Acad. Sci. USA 1994, 91, 5913–5917. [Google Scholar] [CrossRef]
- Katz, R.A.; Gravuer, K.; Skalka, A.M. A preferred target DNA structure for retroviral integrase in vitro. J. Biol. Chem. 1998, 273, 24190–24195. [Google Scholar] [CrossRef] [PubMed]
- Pryciak, P.M.; Sil, A.; Varmus, H.E. Retroviral integration into minichromosomes in vitro. Embo J. 1992, 11, 291–303. [Google Scholar] [PubMed]
- Pryciak, P.M.; Varmus, H.E. Nucleosomes, DNA-binding proteins, and DNA sequence modulate retroviral integration target site selection. Cell 1992, 69, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.L.; Skinner, L.M.; Sudol, M.; Katzman, M. Use of patient-derived human immunodeficiency virus type 1 integrases to identify a protein residue that affects target site selection. J. Virol. 2001, 75, 7756–7762. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.L.; Sudol, M.; Katzman, M. An amino acid in the central catalytic domain of three retroviral integrases that affects target site selection in nonviral DNA. J. Virol. 2003, 77, 3838–3845. [Google Scholar] [CrossRef] [PubMed]
- Lutzke, R.A.; Vink, C.; Plasterk, R.H. Characterization of the minimal DNA-binding domain of the HIV integrase protein. Nucleic Acids Res. 1994, 22, 4125–4131. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.L.; Zhang, Y.; Shkriabai, N.; Karki, R.G.; Nicklaus, M.C.; Kotrikadze, N.; Hess, S.; Le Grice, S.F.; Craigie, R.; Pathak, V.K.; Kvaratskhelia, M. Mass spectrometric analysis of the HIV-1 integrase-pyridoxal 5'-phosphate complex reveals a new binding site for a nucleotide inhibitor. J. Biol. Chem. 2005, 280, 7949–7955. [Google Scholar] [CrossRef] [PubMed]
- van Gent, D.C.; Groeneger, A.A.; Plasterk, R.H. Mutational analysis of the integrase protein of human immunodeficiency virus type 2. Proc. Natl. Acad. Sci. USA 1992, 89, 9598–9602. [Google Scholar] [CrossRef]
- Hare, S.; Shun, M.C.; Gupta, S.S.; Valkov, E.; Engelman, A.; Cherepanov, P. A novel co-crystal structure affords the design of gain-of-function lentiviral integrase mutants in the presence of modified PSIP1/LEDGF/p75 . PLoS Pathog. 2009, 5, e1000259. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, N.K.; Engelman, A. LEDGF/p75 interferes with the formation of synaptic nucleoprotein complexes that catalyze full-site HIV-1 DNA integration in vitro: Implications for the mechanism of viral cDNA integration. Virology 2007, 360, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Priet, S.; Gros, N.; Navarro, J.M.; Boretto, J.; Canard, B.; Querat, G.; Sire, J. HIV-1-associated uracil DNA glycosylase activity controls dUTP misincorporation in viral DNA and is essential to the HIV-1 life cycle. Mol. Cell 2005, 17, 479–490. [Google Scholar] [CrossRef] [PubMed]
- De Rijck, J.; Vandekerckhove, L.; Gijsbers, R.; Hombrouck, A.; Hendrix, J.; Vercammen, J.; Engelborghs, Y.; Christ, F.; Debyser, Z. Overexpression of the lens epithelium-derived growth factor/p75 integrase binding domain inhibits human immunodeficiency virus replication. J. Virol. 2006, 80, 11498–11509. [Google Scholar] [CrossRef] [PubMed]
- Cherepanov, P. LEDGF/p75 interacts with divergent lentiviral integrases and modulates their enzymatic activity in vitro. Nucleic Acids Res. 2007, 35, 113–124. [Google Scholar] [CrossRef] [PubMed]
- De Luca, L.; Vistoli, G.; Pedretti, A.; Barreca, M.L.; Chimirri, A. Molecular dynamics studies of the full-length integrase-DNA complex. Biochem. Biophys. Res. Commun. 2005, 336, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Karki, R.G.; Tang, Y.; Burke, T.R.; Nicklaus, M.C. Model of full-length HIV-1 integrase complexed with viral DNA as template for anti-HIV drug design . J. Comput. Aided Mol. Des. 2004, 18, 739–760. [Google Scholar] [CrossRef] [PubMed]
- Podtelezhnikov, A.A.; Gao, K.; Bushman, F.D.; McCammon, J.A. Modeling HIV-1 integrase complexes based on their hydrodynamic properties. Biopolymers 2003, 68, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Wielens, J.; Crosby, I.T.; Chalmers, D.K. A three-dimensional model of the human immunodeficiency virus type 1 integration complex. J. Comput. Aided Mol. Des. 2005, 19, 301–317. [Google Scholar] [CrossRef] [PubMed]
- Michel, F.; Crucifix, C.; Granger, F.; Eiler, S.; Mouscadet, J.F.; Korolev, S.; Agapkina, J.; Ziganshin, R.; Gottikh, M.; Nazabal, A.; Emiliani, S.; Benarous, R.; Moras, D.; Schultz, P.; Ruff, M. Structural basis for HIV-1 DNA integration in the human genome, role of the LEDGF/P75 cofactor. Embo J. 2009, 28, 980–991. [Google Scholar] [CrossRef] [PubMed]
- Steiniger-White, M.; Rayment, I.; Reznikoff, W.S. Structure/function insights into Tn5 transposition. Curr. Opin. Struct. Biol. 2004, 14, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Alian, A.; Griner, S.L.; Chiang, V.; Tsiang, M.; Jones, G.; Birkus, G.; Geleziunas, R.; Leavitt, A.D.; Stroud, R.M. Catalytically-active complex of HIV-1 integrase with a viral DNA substrate binds anti-integrase drugs. Proc. Natl. Acad. Sci. USA 2009, 106, 8192–8197. [Google Scholar] [CrossRef]
- Valkov, E.; Gupta, S.S.; Hare, S.; Helander, A.; Roversi, P.; McClure, M.; Cherepanov, P. Functional and structural characterization of the integrase from the prototype foamy virus. Nucleic Acids Res. 2009, 37, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Botbol, Y.; Raghavendra, N.K.; Rahman, S.; Engelman, A.; Lavigne, M. Chromatinized templates reveal the requirement for the LEDGF/p75 PWWP domain during HIV-1 integration in vitro. Nucleic Acids Res. 2008, 36, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Kessl, J.J.; Eidahl, J.O.; Shkriabai, N.; Zhao, Z.; McKee, C.J.; Hess, S.; Burke, T.R.; Kvaratskhelia, M. An allosteric mechanism for inhibiting HIV-1 integrase with a small molecule . Mol. Pharmacol. 2009, 76, 824–832. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Kessl, J.J.; McKee, C.J.; Eidahl, J.O.; Shkriabai, N.; Katz, A.; Kvaratskhelia, M. HIV-1 Integrase-DNA Recognition Mechanisms. Viruses 2009, 1, 713-736. https://doi.org/10.3390/v1030713
Kessl JJ, McKee CJ, Eidahl JO, Shkriabai N, Katz A, Kvaratskhelia M. HIV-1 Integrase-DNA Recognition Mechanisms. Viruses. 2009; 1(3):713-736. https://doi.org/10.3390/v1030713
Chicago/Turabian StyleKessl, Jacques J., Christopher J. McKee, Jocelyn O. Eidahl, Nikolozi Shkriabai, Ari Katz, and Mamuka Kvaratskhelia. 2009. "HIV-1 Integrase-DNA Recognition Mechanisms" Viruses 1, no. 3: 713-736. https://doi.org/10.3390/v1030713
APA StyleKessl, J. J., McKee, C. J., Eidahl, J. O., Shkriabai, N., Katz, A., & Kvaratskhelia, M. (2009). HIV-1 Integrase-DNA Recognition Mechanisms. Viruses, 1(3), 713-736. https://doi.org/10.3390/v1030713



