Spatial and Seasonal Variations of Standardized Photosynthetic Parameters under Different Environmental Conditions for Young Planted Larix olgensis Henry Trees
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
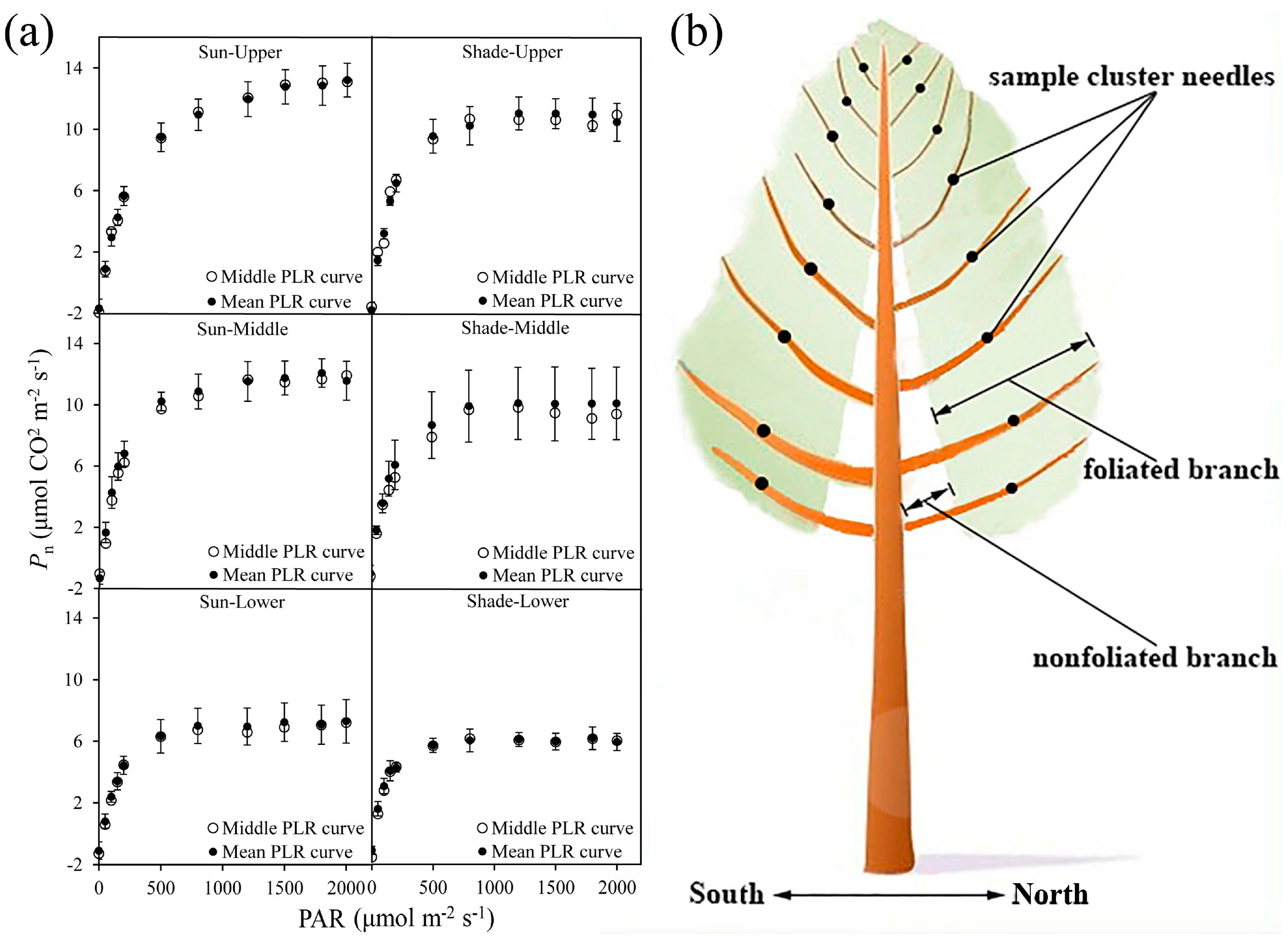
2.2. Sample Selection
2.3. Photosynthetic Gas Exchange Measurements
2.4. Photosynthetically Active Radiation Measurements
2.5. Leaf Morphology Measurements
2.6. Photosynthetic Parameters
2.7. Statistical Analysis
3. Results
3.1. Environmental Conditions
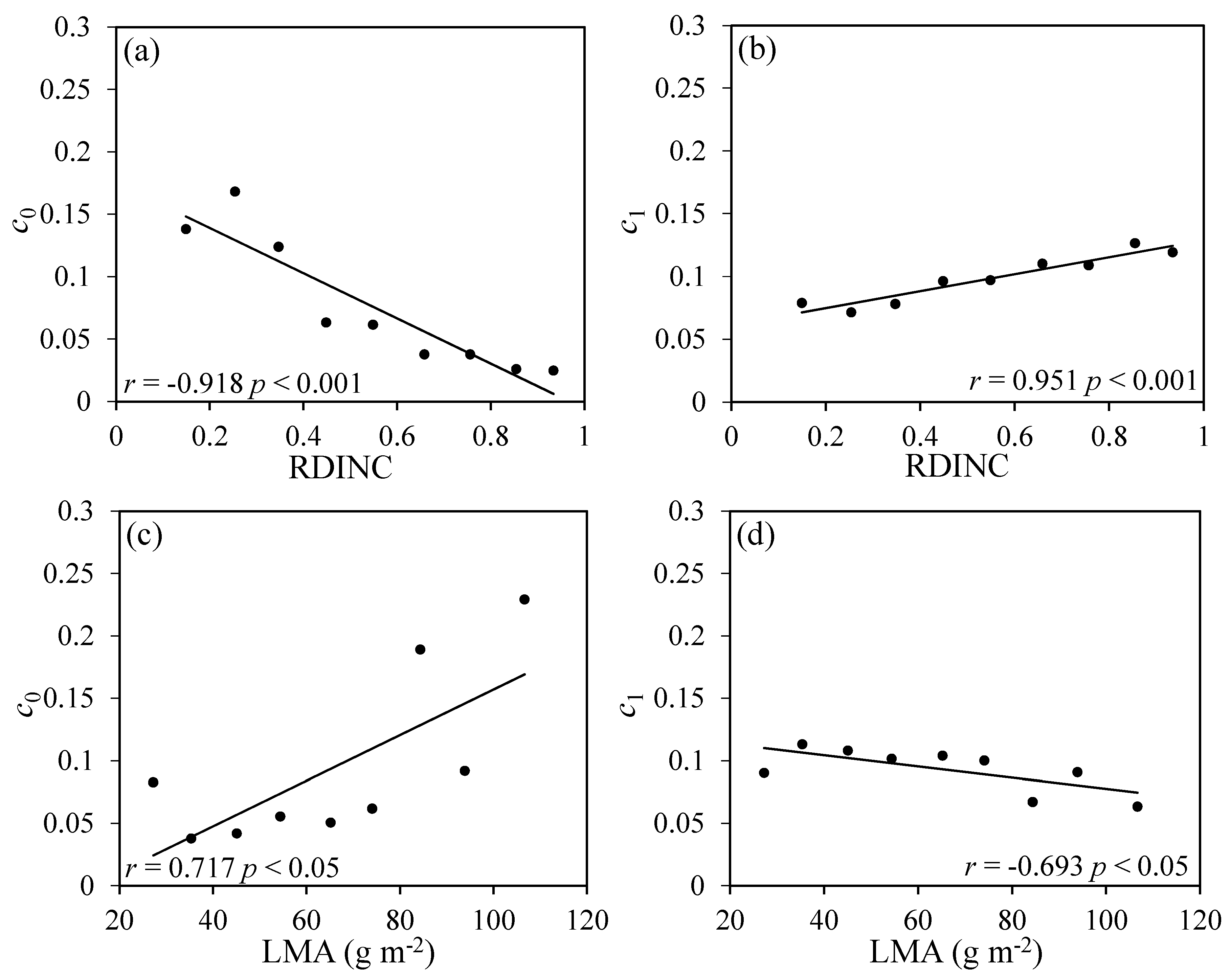
3.2. Relationships between Photosynthetic Parameters Versus Tleaf, VPD, LMA and RDINC
3.3. Standardization of Photosynthetic Parameters
3.4. Vertical Profiles of Standardized and Non-Standardized Photosynthetic Parameters during the Growing Season
3.5. The Response of Vertical Patterns of Standardized Photosynthetic Parameters to the Different Gradients of Environmental Conditions
3.6. Seasonal Evolution of Standardized and Non-Standardized Photosynthetic Parameters during the Growing Season
3.7. The Response of Seasonal Evolutions of Standardized Photosynthetic Parameters to the Different Gradients of Environmental Conditions
4. Discussion
4.1. Relationships between Photosynthetic Parameters and Environmental Factors
4.2. Relationships between Photosynthetic Parameters and LMA
4.3. Standardization of Photosynthetic Parameters
4.4. Spatial Variation in Standardized Leaf Photosynthetic Parameters
4.5. Seasonal Variations in Standardized Leaf Photosynthetic Parameters
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Grady, A.P.; Worledge, D.; Wilkinson, A.; Battaglia, M. Photosynthesis and respiration decline with light intensity in dominant and suppressed Eucalyptus globulus canopies. Funct. Plant Biol. 2008, 35, 439–447. [Google Scholar] [CrossRef]
- Cavaleri, M.A.; Oberbauer, S.F.; Clark, D.B.; Clark, D.A.; Ryan, M.G. Height is more important than light in determining leaf morphology in a tropical forest. Ecology 2010, 91, 1730–1739. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.M. Height-related decreases in mesophyll conductance, leaf photosynthesis and compensating adjustments associated with leaf nitrogen concentrations in Pinus densiflora. Tree Physiol. 2011, 31, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Kosugi, Y.; Takanashi, S.; Yokoyama, N.; Kamakura, M. Vertical variation in leaf gas exchange parameters for a Southeast Asian tropical rainforest in Peninsular Malaysia. J. Plant Res. 2012, 125, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Leuning, R.; Kelliher, F.M.; De Pury, D.G.G.; Schulze, E.D. Leaf nitrogen, photosynthesis, conductance and transpiration: Scaling from leaves to canopies. Plant Cell Environ. 1995, 18, 1183–1200. [Google Scholar] [CrossRef]
- Oliver, C.D.; Larson, B.C. Forest Stand Dynamics; John Wiley & sons, Inc.: New York, NY, USA, 1996. [Google Scholar]
- Li, F.R.; Wang, Z.F.; Wang, B.S. Studies on the effective crown development of Larix olgensis (I)—determination of the effective crown. J. Northeast For. Univ. 1996, 24, 1–8. (In Chinese) [Google Scholar]
- Xu, L.; Baldocchi, D.D. Seasonal trends in photosynthetic parameters and stomatal conductance of blue oak (Quercus douglasii) under prolonged summer drought and high temperature. Tree Physiol. 2003, 23, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Lam, F.; Barrett, J.D.; Nakajima, S. Influence of knot area ratio on the bending strength of Canadian Douglas fir timber used in Japanese post and beam housing. J. Wood Sci. 2005, 51, 18–25. [Google Scholar] [CrossRef]
- Bond, B.J.; Farnsworth, B.T.; Coulombe, R.A.; Winner, W.E. Foliage physiology and biochemistry in response to light gradients in conifers with varying shade tolerance. Oecologia 1999, 120, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Borghetti, M.; Vendramin, G.G.; Giannini, R. Specific leaf area and leaf area index distribution in a young Douglas. Can. J. For. Res. 1986, 16, 1283–1288. [Google Scholar] [CrossRef]
- Cescatti, A.; Zorer, R. Structural acclimation and radiation regime of silver fir (Abies alba Mill.) shoots along a light gradient. Plant Cell Environ. 2003, 26, 429–442. [Google Scholar] [CrossRef]
- Wyka, T.P.; Oleksyn, J.; Żytkowiak, R.; Karolewski, P.; Jagodziński, A.M.; Reich, P.B. Responses of leaf structure and photosynthetic properties to intra-canopy light gradients: A common garden test with four broadleaf deciduous angiosperm and seven evergreen conifer tree species. Oecologia 2012, 170, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Coble, A.P.; Cavaleri, M.A. Light drives vertical gradients of leaf morphology in a sugar maple (Acer saccharum) forest. Tree Physiol. 2014, 34, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Bond, B.J. Age-related changes in photosynthesis of woody plants. Trends Plant Sci. 2000, 5, 349–353. [Google Scholar] [CrossRef]
- Ishii, H.; Ford, E.D.; Boscolo, M.E.; Manriquez, A.C.; Wilson, M.E.; Hinckley, T.M. Variation in specific needle area of old-growth Douglas-fir in relation to needle age, within-crown position and epicormic shoot production. Tree Physiol. 2002, 22, 31–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, G.W.; Sillett, S.C.; Jennings, G.M.; Davis, S.D. The limits to tree height. Nature 2004, 428, 851–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oldham, A.R.; Sillett, S.C.; Tomescu, A.M.F.; Koch, G. The hydrostatic gradient, not light availability, drives height-related variation in Sequoia sempervirens (Cupressaceae) leaf anatomy. Am. J. Bot. 2010, 97, 1087–1097. [Google Scholar] [CrossRef] [PubMed]
- Coble, A.P.; Vanderwall, B.; Mau, A.; Cavaleri, M. How vertical patterns in leaf traits shift seasonally and the implications for modeling canopy photosynthesis in a temperate deciduous forest. Tree Physiol. 2016, 36, 1077–1091. [Google Scholar] [CrossRef] [PubMed]
- Field, C. Allocating leaf nitrogen for the maximization of carbon gain: Leaf age as a control on the allocation program. Oecologia 1983, 56, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Kenzo, T.; Ichie, T.; Watanabe, Y.; Yoneda, R.; Ninomiya, I.; Koike, T. Changes in photosynthesis and leaf characteristics with tree height in five dipterocarp species in a tropical rain forest. Tree Physiol. 2006, 26, 865–873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meinzer, F.C.; Bond, B.J.; Karanian, J.A. Biophysical constraints on leaf expansion in a tall conifer. Tree Physiol. 2008, 28, 197–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodruff, D.R.; Meinzer, F.C.; Lachenbruch, B.; Johnson, D.M. Coordination of leaf structure and gas exchange along a height gradient in a tall conifer. Tree Physiol. 2009, 29, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, D.R. The impacts of water stress on phloem transport in Douglas-fir trees. Tree Physiol. 2014, 34, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Benomar, L.; DesRochers, A.; Larocque, G.R. Changes in specific leaf area and photosynthetic nitrogen-use efficiency associated with physiological acclimation of two hybrid poplar clones to intraclonal competition. Can. J. For. Res. 2011, 41, 1465–1476. [Google Scholar] [CrossRef]
- Riikonen, J.; Oksanen, E.; Peltonen, P.; Holopainen, T.; Vapaavuori, E. Seasonal variation in physiological characteristics of two silver birch clones in the field. Can. J. For. Res. 2003, 33, 2164–2176. [Google Scholar] [CrossRef]
- Mullin, L.P.; Sillett, S.C.; Koch, G.; Tu, K.; Antoine, M.E. Physiological consequences of height-related morphological variation in Sequoia sempervirens foliage. Tree Physiol. 2009, 29, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Flexas, J.; Barbour, M.M.; Brendel, O.; Cabrera, H.M.; Carriquí, M.; Díaz-Espejo, A.; Douthe, C.; Dreyer, E.; Ferrio, J.P.; Gago, J.; et al. Mesophyll diffusion conductance to CO2: An unappreciated central player in photosynthesis. Plant Sci. 2012, 193, 70–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grassi, G.; Vicinelli, E.; Ponti, F.; Cantoni, L.; Magnani, F. Seasonal and interannual variability of photosynthetic capacity in relation to leaf nitrogen in a deciduous forest plantation in northern Italy. Tree Physiol. 2005, 25, 349–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strand, M.; Lundmark, T.; Söderbergh, I.; Mellander, P.E. Impacts of seasonal air and soil temperatures on photosynthesis in Scots pine trees. Tree Physiol. 2002, 22, 839–847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awada, T.; Radoglou, K.; Fotelli, M.N.; Constantinidou, H.I.A. Ecophysiology of seedlings of three Mediterranean pine species in contrasting light regimes. Tree Physiol. 2003, 23, 33–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Letts, M.G.; Phelan, C.A.; Johnson, D.R.E.; Rood, S. Seasonal photosynthetic gas exchange and leaf reflectance characteristics of male and female cottonwoods in a riparian woodland. Tree Physiol. 2008, 28, 1037–1048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wyka, T.P.; Żytkowiak, R.; Oleksyn, J. Seasonal dynamics of nitrogen level and gas exchange in different cohorts of Scots pine needles: A conflict between nitrogen mobilization and photosynthesis? Eur. J. For. Res. 2016, 135, 483–493. [Google Scholar] [CrossRef]
- Misson, L.; Tu, K.P.; Boniello, R.A.; Goldstein, A.H. Seasonality of photosynthetic parameters in a multi-specific and vertically complex forest ecosystem in the Sierra Nevada of California. Tree Physiol. 2006, 26, 729–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nouvellon, Y.; Laclau, J.P.; Epron, D.; Kinana, A.; Mabiala, A.; Roupsard, O.; Bonnefond, J.M.; Maire, G.I.; Marsden, C.; Bontemps, J.D.; et al. Within-stand and seasonal variations of specific leaf area in a clonal Eucalyptus, plantation in the Republic of Congo. For. Ecol. Manag. 2010, 259, 1796–1807. [Google Scholar] [CrossRef]
- Li, J.Y.; Ou-lee, T.M.; Raba, R.; Amundson, R.G.; Last, R.L. Arabidopsis flavonoid mutants are hypersensitive to UV-B irradiation. Plant Cell 1993, 5, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Coble, A.P.; Cavaleri, M.A. Light acclimation optimizes leaf functional traits despite height-related constraints in a canopy shading experiment. Oecologia 2015, 177, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Pardos, M.; Puértolas, J.; Madrigal, G.; Garriga, E.; Blas, S.D.; Calama, R. Seasonal changes in the physiological activity of regeneration under a natural light gradient in a Pinus pinea regular stand. For. Syst. 2010, 19, 367–380. [Google Scholar] [CrossRef]
- Craven, D.; Dent, D.; Braden, D.; Ashton, M.S.; Berlyn, G.P.; Hall, J.S. Seasonal variability of photosynthetic characteristics influences growth of eight tropical tree species at two sites with contrasting precipitation in Panama. For. Ecol. Manag. 2011, 261, 1643–1653. [Google Scholar] [CrossRef]
- Li, C.; Xu, G.; Zang, R.; Korpelainen, H.; Berninger, F. Sex-related differences in leaf morphological and physiological responses in Hippophae rhamnoides along an altitudinal gradient. Tree Physiol. 2007, 27, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.B.; Baldocchi, D.D.; Hanson, P.J. Leaf age affects the seasonal pattern of photosynthetic capacity and net ecosystem exchange of carbon in a deciduous forest. Plant Cell Environ. 2001, 24, 571–583. [Google Scholar] [CrossRef]
- Tanaka, K.; Kosugi, Y.; Nakamura, A. Impact of leaf physiological characteristics on seasonal variation in CO2, latent and sensible heat exchanges over a tree plantation. Agric. For. Meteorol. 2002, 114, 103–122. [Google Scholar] [CrossRef]
- Kosugi, Y.; Shibata, S.; Kobashi, S. Parameterization of the CO2 and H2O gas exchange of several temperate deciduous broad-leaved trees at the leaf scale considering seasonal changes. Plant Cell Environ. 2003, 26, 285–301. [Google Scholar] [CrossRef]
- Esprey, L.J.; Sands, P.J.; Smith, C.W. Understanding of 3-PG using a sensitivity analysis. For. Ecol. Manag. 2004, 193, 235–250. [Google Scholar] [CrossRef]
- Stape, J.L.; Ryan, M.G.; Binkley, D. Testing the utility of the 3-PG model for growth of Eucalyptus grandis × urophylla with natural and manipulated supplies of water and nutrients. For. Ecol. Manag. 2004, 193, 219–234. [Google Scholar] [CrossRef]
- Davidson, R.; Mauffette, Y.; Gagnon, D. Light requirements of seedlings: A method for selecting tropical trees for plantation forestry. Basic Appl. Ecol. 2002, 3, 209–220. [Google Scholar] [CrossRef]
- Lin, Y.S.; Medlyn, B.E.; Ellsworth, D.S. Temperature responses of leaf net photosynthesis: The role of component processes. Tree Physiol. 2012, 32, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Craven, D.; Braden, D.; Ashton, M.S.; Berlyn, G.P.; Wishnie, M.; Dent, D. Between and within-site comparisons of structural and physiological characteristics and foliar nutrient content of 14 tree species at a wet, fertile site and a dry, infertile site in Panama. For. Ecol. Manag. 2007, 238, 335–346. [Google Scholar] [CrossRef]
- Fellner, H.; Dirnberger, G.F.; Sterba, H. Specific leaf area of European Larch (Larix decidua Mill.). Trees 2016, 30, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Bauerle, W.L.; Oren, R.; Way, D.A.; Qian, S.S.; Stoy, P.C.; Thornton, P.E.; Bowden, J.D.; Hoffman, F.M.; Reynolds, R.F. Photoperiodic regulation of the seasonal pattern of photosynthetic capacity and the implications for carbon cycling. Proc. Nat. Acad. Sci. USA 2012, 109, 8612–8617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, Q.; Kawasaki, T.; Katahata, S.; Mukai, Y.; Chiba, Y. Horizontal and vertical variations in photosynthetic capacity in a Pinus densiflora crown in relation to leaf nitrogen allocation and acclimation to irradiance. Tree Physiol. 2003, 23, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, A.R.; Sillett, S.C.; Dawson, T.E. Effects of tree height on branch hydraulics, leaf structure and gas exchange in California redwoods. Plant Cell Environ. 2009, 32, 743–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rascher, U.; Bobich, E.G.; Lin, G.H.; Walter, A.; Morris, T.J.; Naumann, M.; Nichol, C.J.; Pierce, D.; Bil, K.; Kudeyarov, V.N.; et al. Functional diversity of photosynthesis during drought in a model tropical rainforest—the contributions of leaf area, photosynthetic electron transport and stomatal conductance to reduction in net ecosystem carbon exchange. Plant Cell Environ. 2004, 27, 1239–1256. [Google Scholar] [CrossRef]
- Broeckx, L.S.; Fichot, R.; Verlinden, M.S.; Ceulemans, R. Seasonal variations in photosynthesis, intrinsic water-use efficiency and stable isotope composition of poplar leaves in a short-rotation plantation. Tree Physiol. 2014, 34, 701–715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franco, A.C. Seasonal patterns of gas exchange, water relations and growth of Roupala montana, an evergreen savanna species. Plant Ecol. 1998, 136, 69–76. [Google Scholar] [CrossRef]
- Corcuera, L.; Morales, F.; Abadía, A.; Pelegrín, G. Seasonal changes in photosynthesis and photoprotection in a Quercus ilex subsp. ballota woodland located in its upper altitudinal extreme in the Iberian Peninsula. Tree Physiol. 2005, 25, 599–608. [Google Scholar] [PubMed]
- Zhu, J.J.; Liu, Z.G.; Wang, H.X.; Yan, Q.L.; Fang, H.Y.; Hu, L.; Yu, L.Z. Effects of site preparation on emergence and early establishment of Larix olgensis, in montane regions of northeastern China. New For. 2008, 36, 247–260. [Google Scholar] [CrossRef]
- Wei, H.X.; Xu, C.Y.; Hawkins, B.J.; Ma, L.; Jiang, L. Organic amendment and inorganic fertilization affect soil properties and quality of Larix olgensis bareroot stock. New For. 2012, 43, 155–168. [Google Scholar] [CrossRef]
- Zhu, Y.; Dumroese, R.K.; Pinto, J.R.; Li, G.L.; Liu, Y. Fall fertilization enhanced nitrogen storage and translocation in Larix olgensis, seedlings. New For. 2013, 44, 849–861. [Google Scholar] [CrossRef]
- Eguchi, N.; Fukatsu, E.; Funada, R.; Tobita, H.; Kitao, M.; Maruyama, Y.; Koike, T. Changes in morphology, anatomy, and photosynthetic capacity of needles of Japanese Larch (Larix kaempferi) seedlings grown in high CO2 concentrations. Photosynthetica 2004, 42, 173–178. [Google Scholar] [CrossRef]
- Mao, Q.Z.; Watanabe, M.; Imori, M.; Kim, Y.S.; Kita, K.; Koike, T. Photosynthesis and nitrogen allocation in needles in the sun and shade crowns of hybrid larch saplings: Effect of nitrogen application. Photosynthetica 2012, 50, 422–428. [Google Scholar] [CrossRef]
- Wang, W.J.; Watanabe, Y.; Endo, I.; Kitaoka, S.; Koike, T. Seasonal changes in the photosynthetic capacity of cones on a larch (Larix kaempferi) canopy. Photosynthetica 2006, 44, 345–348. [Google Scholar] [CrossRef]
- Galvagno, M.; Rossini, M.; Migliavacca, M.; Cremonese, E.; Colombo, R.; Morra-di-Cella, U. Seasonal course of photosynthetic efficiency in Larix decidua Mill. in response to temperature and change in pigment composition during senescence. Int. J. Biometeorol. 2013, 57, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Kurachi, N.; Hagihara, A.; Hozumi, K. Canopy photosynthetic production in a Japanese larch stand. I. Seasonal and vertical changes of leaf characteristics along the light gradient in a canopy. Ecol. Res. 1992, 7, 255–265. [Google Scholar] [CrossRef]
- Liu, Q.; Dong, L.H.; Li, F.R.; Li, X. Spatial heterogeneity of canopy photosynthesis for Larix olgensis. Chin. J. Appl. Ecol. 2016, 27, 2789–2796. (In Chinese) [Google Scholar]
- Wu, C.; Wang, Z.X.; Sun, H.L.; Guo, S.L. Effects of different concentrations of Nitrogen and Phosphorus on Chlorophyll Biosynthesis, Chlorophyll a Fluorescence, and Photosynthetic Rate in Larix olgensis seedlings. Sci. Silv. Sin. 2005, 41, 31–36. (In Chinese) [Google Scholar]
- Zhao, X.Z.; Jiang, H.F.; Mao, Z.J. The studies of the photosynthetic characteristics of Larix olgensis Herry., Larix kaempferi Carr. and Larix gmelinii Rupr. seedlings. Bull. Bot. Res. 2007, 27, 361–366. (In Chinese) [Google Scholar]
- Luca, T.H.D. Relationship of 0.5 M K2SO4 extractable anthrone-reactive carbon to indices of microbial activity in forest soils. Soil Biol. Biochem. 1998, 30, 1293–1299. [Google Scholar] [CrossRef]
- Cluzeau, C.; Goff, N.L.; Ottorini, J.M. Development of primary branches and crown profile of Fraxinus excelsior. Can. J. For. Res. 1994, 24, 2315–2323. [Google Scholar] [CrossRef]
- Bassman, J.H.; Zwier, J.C. Gas exchange characteristics of Populus trichocarpa, Populus deltoides and Populus trichocarpa × P. deltoids clones. Tree Physiol. 1991, 8, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, J.; Taylor, J.A. On the Temperature dependence of soil respiration. Funct. Ecol. 1994, 8, 315–323. [Google Scholar] [CrossRef]
- Rodríguez-calcerrada, J.; Atkin, O.K.; Robson, T.M.; Zaragoza-castells, J.; Gil, L.; Aranda, I. Thermal acclimation of leaf dark respiration of beech seedlings experiencing summer drought in high and low light environments. Tree Physiol. 2010, 30, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Weerasinghe, L.K.; Creek, D.; Crous, K.Y.; Xiang, S.; Liddell, M.J.; Turnbull, M.H.; Atkin, O.K. Canopy position affects the relationships between leaf respiration and associated traits in a tropical rainforest in Far North Queensland. Tree Physiol. 2014, 34, 564–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, N.; Meyer, H.; Welp, G.; Amelung, W. Soil respiration and its temperature sensitivity (Q10): Rapid acquisition using mid-infrared spectroscopy. Geoderma 2018, 323, 31–40. [Google Scholar] [CrossRef]
- Sage, R.F.; Kubien, D.S. The temperature response of C3 and C4 photosynthesis. Plant Cell Environ. 2007, 30, 1086–1106. [Google Scholar] [CrossRef] [PubMed]
- Way, D.A.; Sage, R.F. Thermal acclimation of photosynthesis in black spruce [Picea mariana (Mill.) B.S.P.]. Plant Cell Environ. 2008, 31, 1250–1262. [Google Scholar] [CrossRef] [PubMed]
- Wieser, G.; Oberhuber, W.; Walder, L.; Spieler, D.; Gruber, A. Photosynthetic temperature adaptation of Pinus cembra, within the timberline ecotone of the Central Austrian Alps. Ann. For. Sci. 2010, 67, 201. [Google Scholar] [CrossRef] [PubMed]
- Calama, R.; Puértolas, J.; Madrigal, G.; Pardos, M. Modeling the environmental response of leaf net photosynthesis in Pinus pinea L. natural regeneration. Ecol. Model. 2013, 251, 9–21. [Google Scholar] [CrossRef]
- Mayoral, C.; Calama, R.; Sánchez-González, M.; Pardos, M. Modelling the influence of light, water and temperature on photosynthesis in young trees of mixed Mediterranean forests. New For. 2015, 46, 485–506. [Google Scholar] [CrossRef]
- Lloyd, J.; Farquhar, G.D. Effects of rising temperatures and [CO2] on the physiology of tropical forest trees. Phil. Trans. Biol. Sci. 2008, 363, 1811–1817. [Google Scholar] [CrossRef] [PubMed]
- Hikosaka, K.; Nabeshima, E.; Hiura, T. Seasonal changes in the temperature response of photosynthesis in canopy leaves of Quercus crispula in a cool-temperate forest. Tree Physiol. 2007, 27, 1035–1041. [Google Scholar] [CrossRef] [PubMed]
- Tjoelker, M.G.; Oleksyn, J.; Reich, P.B. Modelling respiration of vegetation: Evidence for a general temperature-dependent Q10. Glob. Chang. Biol. 2001, 7, 223–230. [Google Scholar] [CrossRef]
- Griffin, K.L.; Tissue, D.T.; Turnbull, M.H.; Schuster, W.; Whitehead, D. Leaf dark respiration as a function of canopy position in Nothofagus fusca trees grown at ambient and elevated CO2 partial pressures for 5 years. Funct. Ecol. 2001, 15, 497–505. [Google Scholar] [CrossRef]
- Griffin, K.L.; Turnbull, M.; Murthy, R. Canopy position affects the temperature response of leaf respiration in Populus deltoides. New Phytol. 2002, 154, 609–619. [Google Scholar] [CrossRef]
- Tissue, D.T.; Lewis, J.D.; Wullschleger, S.D.; Amthor, J.S.; Griffin, K.L.; Anderson, O.R. Leaf respiration in sweetgum (Liquidambar styraciflua) at different canopy positions grown in ambient and elevated CO2 in the field. Tree Physiol. 2002, 22, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.D.; Lucash, M.; Olszyk, D.; Tingey, D.T. Seasonal patterns of photosynthesis in Douglas-fir seedlings during the third and fourth year of exposure to elevated CO2, and temperature. Plant Cell Environ. 2001, 24, 539–548. [Google Scholar] [CrossRef]
- Sperlich, D.; Chang, C.T.; Penuelas, J.; Gracia, C.; Sabaté, S. Seasonal variability of foliar photosynthetic and morphological traits and drought impacts in a Mediterranean mixed forest. Tree Physiol. 2015, 35, 501–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitamura, M.; Yamamura, Y.; Nakano, T. Large-scale canopy opening causes decreased photosynthesis in the saplings of shade-tolerant conifer, Abies veitchii. Tree Physiol. 2009, 29, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.D.; Berlyn, G.P.; Ashton, P.M.S.; Thadani, R.; Cameron, I.R. Foliar plasticity of hybrid spruce in relation to crown position and stand age. Can. J. Bot. 2000, 78, 305–317. [Google Scholar]
- Zhang, Y.; Zheng, Q.; Tyree, M.T. Factors controlling plasticity of leaf morphology in Robinia pseudoacacia L. I: Height-associated variation in leaf structure. Ann. For. Sci. 2012, 69, 29–37. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Equiza, M.A.; Zheng, Q.S.; Tyree, M.T. Factors controlling plasticity of leaf morphology in Robinia pseudoacacia L. II: The impact of water stress on leaf morphology of seedlings grown in a controlled environment chamber. Ann. For. Sci. 2012, 69, 39–47. [Google Scholar] [CrossRef]
- Xu, C.Y.; Griffin, K.L. Seasonal variation in the temperature response of leaf respiration in Quercus rubra: Foliage respiration and leaf properties. Funct. Ecol. 2006, 20, 778–789. [Google Scholar] [CrossRef]
- Yuko, Y.; Kouki, H.; Tadaki, H. Seasonal changes in photosynthesis, nitrogen content and nitrogen partitioning in Lindera umbellata leaves grown in high or low irradiance. Tree Physiol. 2006, 26, 1315–1323. [Google Scholar]
- Radoglou, K.; Teskey, R.O. Changes in rates of photosynthesis and respiration during needle development of loblolly pine. Tree Physiol. 1997, 17, 485–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collier, D.E.; Thibodeau, B.A. Changes in respiration and chemical content during autumnal senescence of Populus tremuloides and Quercus rubra leaves. Tree Physiol. 1995, 15, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Vose, J.M.; Ryan, M.G. Seasonal respiration of foliage, fine roots, and woody tissues in relation to growth, tissue N, and photosynthesis. Glob. Chang. Biol. 2010, 8, 182–193. [Google Scholar] [CrossRef]





| Sample Trees | Sample Plots | |||||||
|---|---|---|---|---|---|---|---|---|
| Tree NO. | DBH (cm) | H (m) | Plot NO. | Range of DBH (cm) | Range of H (m) | Quadratic Mean Diameter (cm) | Mean H (m) | Stand Density (trees ha−1) |
| I | 10.75 | 10.33 | P1 | 5.0–16.0 | 6.0–12.8 | 9.92 | 9.23 | 2200 |
| II | 11.15 | 10.52 | P2 | 5.3–15.9 | 5.3–13.2 | 10.59 | 10.19 | 2400 |
| III | 10.90 | 10.81 | P3 | 5.2–14.1 | 7.1–10.5 | 9.73 | 9.62 | 3200 |
| IV | 10.80 | 10.35 | P4 | 5.0–16.7 | 5.8–13.0 | 10.13 | 9.63 | 2920 |
| V | 11.69 | 10.80 | P5 | 5.2–16.5 | 6.3–12.7 | 10.92 | 10.3 | 2430 |
| RDINC | Statistics | Photosynthetic Parameters | Leaf Morphology | Environmental Conditions | ||||
|---|---|---|---|---|---|---|---|---|
| Pmax (μmol m−2 s−1) | Rd (μmol m−2 s−1) | gs-sat (mol m−2 s−1) | LMA (g m−2) | Tleaf (°C) | VPD (kPa) | PAR (μmol m−2 s−1) | ||
| 0.1–0.2 | Sample number | n = 1299 (99 PLR curves) | n = 104 | |||||
| Mean | 14.67 | 1.48 | 0.25 | 83.61 | 29.62 | 1.74 | 913.85 | |
| Std | 4.9 | 0.6 | 0.13 | 19.47 | 3.33 | 0.66 | 427.96 | |
| Max | 28.77 | 2.93 | 0.61 | 127.53 | 38.15 | 4.05 | 1879.79 | |
| Min | 5.57 | 0.58 | 0.05 | 29.18 | 19.53 | 0.78 | 58.04 | |
| 0.2–0.3 | Sample number | n = 1026 (80 PLR curves) | n = 348 | |||||
| Mean | 11.47 | 1.5 | 0.17 | 74.44 | 30.32 | 2.06 | 836.3 | |
| Std | 3.33 | 0.59 | 0.09 | 15.42 | 2.92 | 0.82 | 472.67 | |
| Max | 18.41 | 2.85 | 0.51 | 108.67 | 39.37 | 4.32 | 1998.87 | |
| Min | 5.23 | 0.31 | 0.04 | 31.07 | 25.07 | 0.97 | 104.9 | |
| 0.3–0.4 | Sample number | n = 953 (73 PLR curves) | n = 1406 | |||||
| Mean | 10.96 | 1.17 | 0.16 | 65.44 | 28.43 | 1.79 | 663.86 | |
| Std | 3.31 | 0.5 | 0.08 | 14.63 | 3.19 | 0.73 | 517.09 | |
| Max | 17.79 | 2.51 | 0.41 | 90.94 | 35.58 | 3.55 | 2006.43 | |
| Min | 4.7 | 0.18 | 0.04 | 26.64 | 19.74 | 0.83 | 14.27 | |
| 0.4–0.5 | Sample number | n = 912 (69 PLR curves) | n = 1910 | |||||
| Mean | 9.9 | 1.03 | 0.14 | 60.4 | 28.53 | 1.77 | 550.91 | |
| Std | 2.53 | 0.52 | 0.07 | 13.99 | 3.42 | 0.79 | 496.74 | |
| Max | 15.02 | 2.15 | 0.3 | 84.9 | 36.07 | 3.64 | 1804.97 | |
| Min | 3.21 | 0.31 | 0.02 | 24.85 | 20.42 | 0.65 | 7.86 | |
| 0.5–0.6 | Sample number | n = 835 (62 PLR curves) | n = 2076 | |||||
| Mean | 8.78 | 0.96 | 0.12 | 56.01 | 27.83 | 1.84 | 444.87 | |
| Std | 2.17 | 0.46 | 0.06 | 12.39 | 3.51 | 0.72 | 465.05 | |
| Max | 13.24 | 2.29 | 0.29 | 75.77 | 34.54 | 3.55 | 1856.4 | |
| Min | 4.41 | 0.19 | 0.03 | 28.85 | 19.71 | 0.67 | 7.45 | |
| 0.6–0.7 | Sample number | n = 802 (61 PLR curves) | n = 2793 | |||||
| Mean | 7.57 | 0.8 | 0.11 | 49.96 | 27.19 | 1.7 | 276.86 | |
| Std | 1.82 | 0.34 | 0.05 | 9.96 | 3.42 | 0.6 | 348.78 | |
| Max | 13.69 | 1.65 | 0.28 | 69.83 | 34.2 | 3.76 | 1781.77 | |
| Min | 4.33 | 0.3 | 0.03 | 29.3 | 18.96 | 0.81 | 6.44 | |
| 0.7–0.8 | Sample number | n = 884 (66 PLR curves) | n = 2841 | |||||
| Mean | 6.79 | 0.75 | 0.1 | 45.17 | 26.91 | 1.66 | 198.29 | |
| Std | 1.91 | 0.36 | 0.04 | 8.73 | 3.42 | 0.55 | 305.57 | |
| Max | 14.87 | 1.91 | 0.24 | 64.23 | 34.2 | 3.33 | 1609.52 | |
| Min | 3.17 | 0.25 | 0.03 | 26.22 | 18.5 | 0.75 | 2.79 | |
| 0.8–0.9 | Sample number | n = 816 (63 PLR curves) | n = 2572 | |||||
| Mean | 6.55 | 0.85 | 0.1 | 40.6 | 26.85 | 1.62 | 119.02 | |
| Std | 1.81 | 0.46 | 0.05 | 7.04 | 3.93 | 0.51 | 230.04 | |
| Max | 11.92 | 2.13 | 0.23 | 54.49 | 33.46 | 3.01 | 1463.72 | |
| Min | 3.47 | 0.2 | 0.03 | 26.86 | 15.45 | 0.64 | 1.54 | |
| 0.9–1.0 | Sample number | n = 907 (67 PLR curves) | n = 2282 | |||||
| Mean | 5.55 | 0.66 | 0.09 | 37.53 | 26.79 | 1.59 | 76.58 | |
| Std | 1.71 | 0.4 | 0.04 | 7 | 3.63 | 0.52 | 180.25 | |
| Max | 10.99 | 1.84 | 0.24 | 58.73 | 34.38 | 3.07 | 1477.64 | |
| Min | 2.1 | 0.17 | 0.03 | 23.98 | 17.8 | 0.64 | 1.04 | |
| Photosynthetic Parameters | R2 | RMSE |
|---|---|---|
| Pmax (μmol m−2 s−1) | 0.769 | 2.00 |
| Rd (μmol m−2 s−1) | 0.575 | 0.37 |
| gs-sat (mol m−2 s−1) | 0.546 | 0.06 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Q.; Li, F. Spatial and Seasonal Variations of Standardized Photosynthetic Parameters under Different Environmental Conditions for Young Planted Larix olgensis Henry Trees. Forests 2018, 9, 522. https://doi.org/10.3390/f9090522
Liu Q, Li F. Spatial and Seasonal Variations of Standardized Photosynthetic Parameters under Different Environmental Conditions for Young Planted Larix olgensis Henry Trees. Forests. 2018; 9(9):522. https://doi.org/10.3390/f9090522
Chicago/Turabian StyleLiu, Qiang, and Fengri Li. 2018. "Spatial and Seasonal Variations of Standardized Photosynthetic Parameters under Different Environmental Conditions for Young Planted Larix olgensis Henry Trees" Forests 9, no. 9: 522. https://doi.org/10.3390/f9090522
APA StyleLiu, Q., & Li, F. (2018). Spatial and Seasonal Variations of Standardized Photosynthetic Parameters under Different Environmental Conditions for Young Planted Larix olgensis Henry Trees. Forests, 9(9), 522. https://doi.org/10.3390/f9090522





