Species Richness of the Family Ericaceae along an Elevational Gradient in Yunnan, China
Abstract
:1. Introduction
2. Materials and Methods
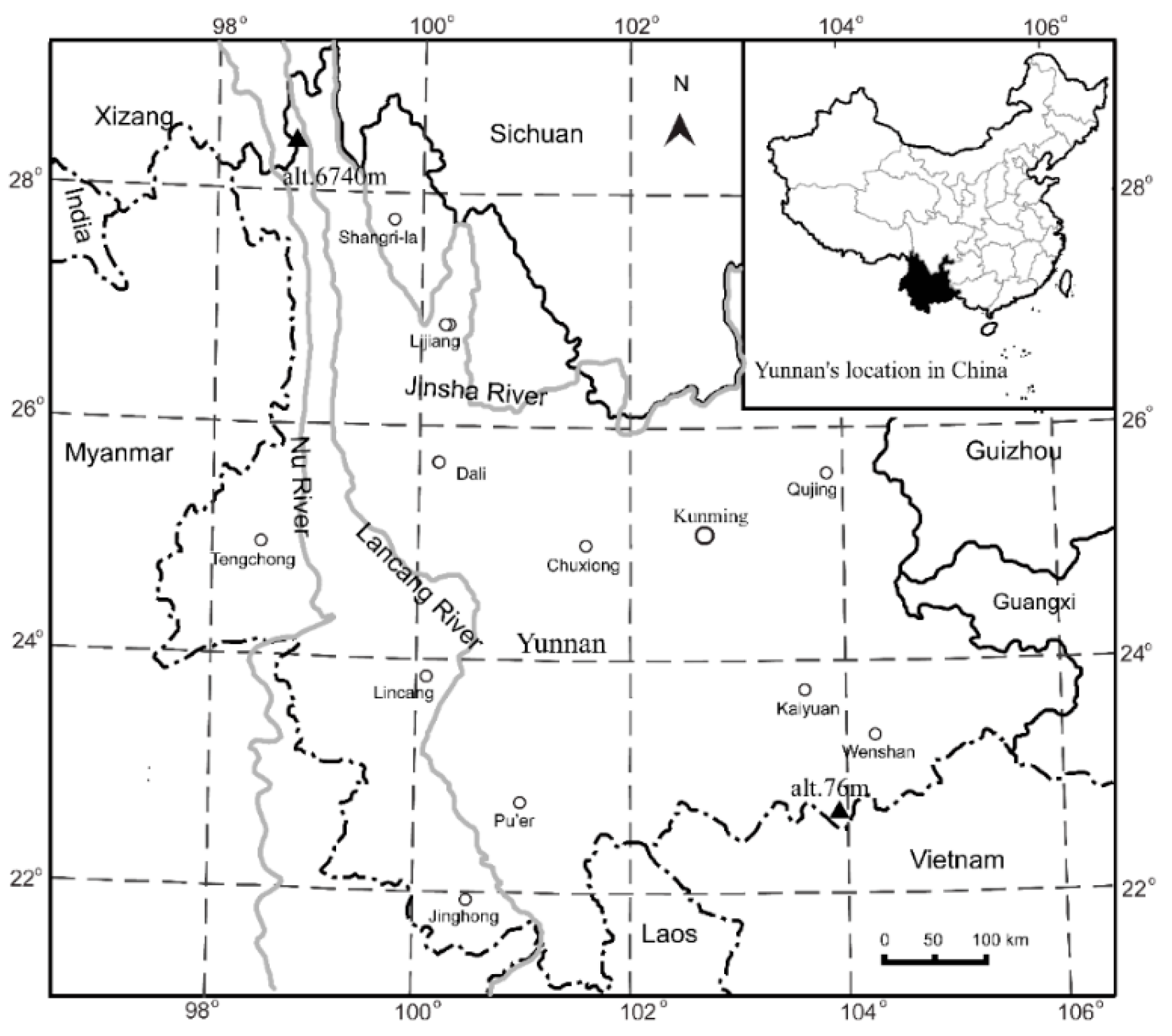
2.1. Study Area
2.2. Data Sources
2.3. Data Analysis
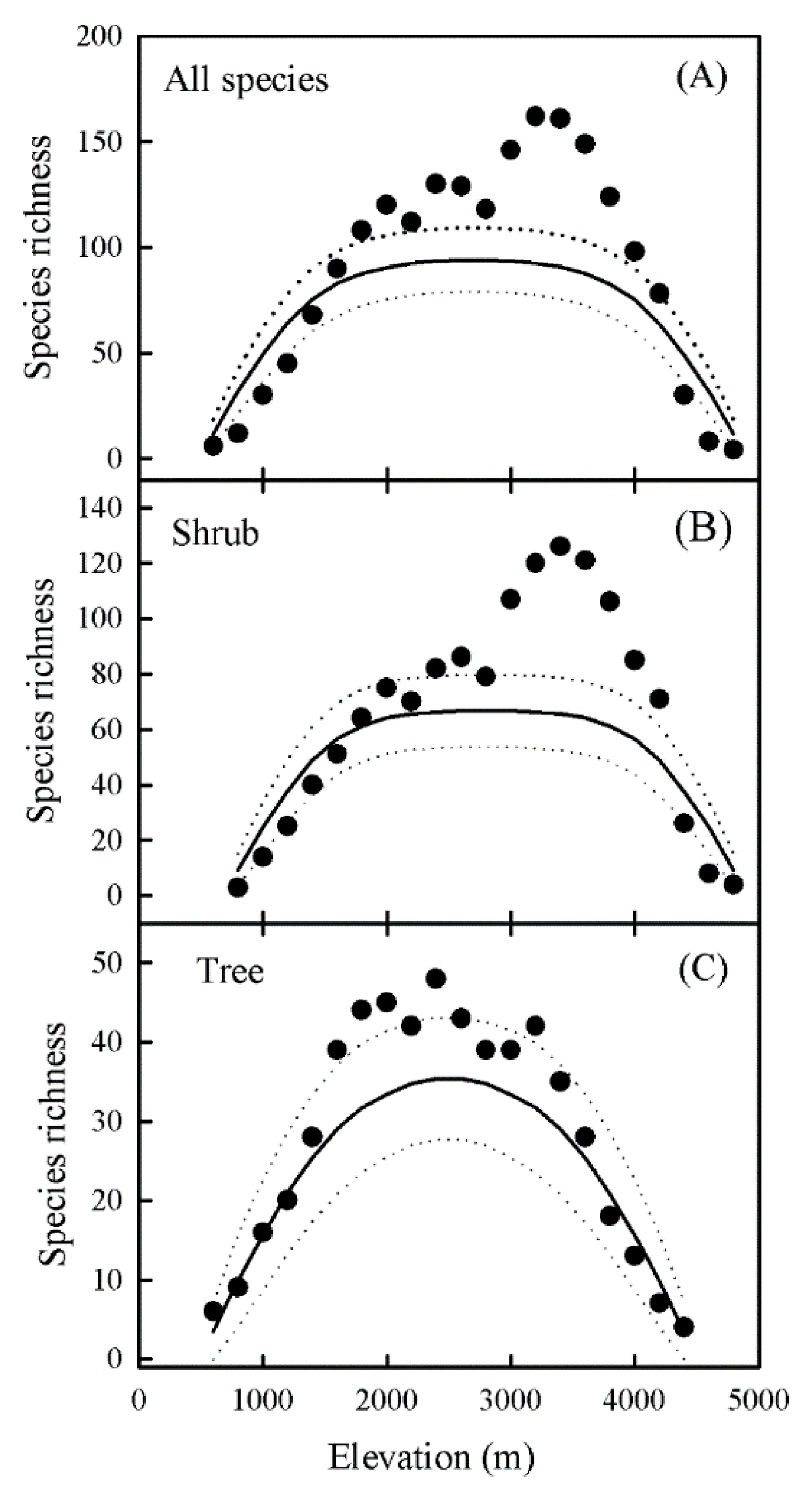
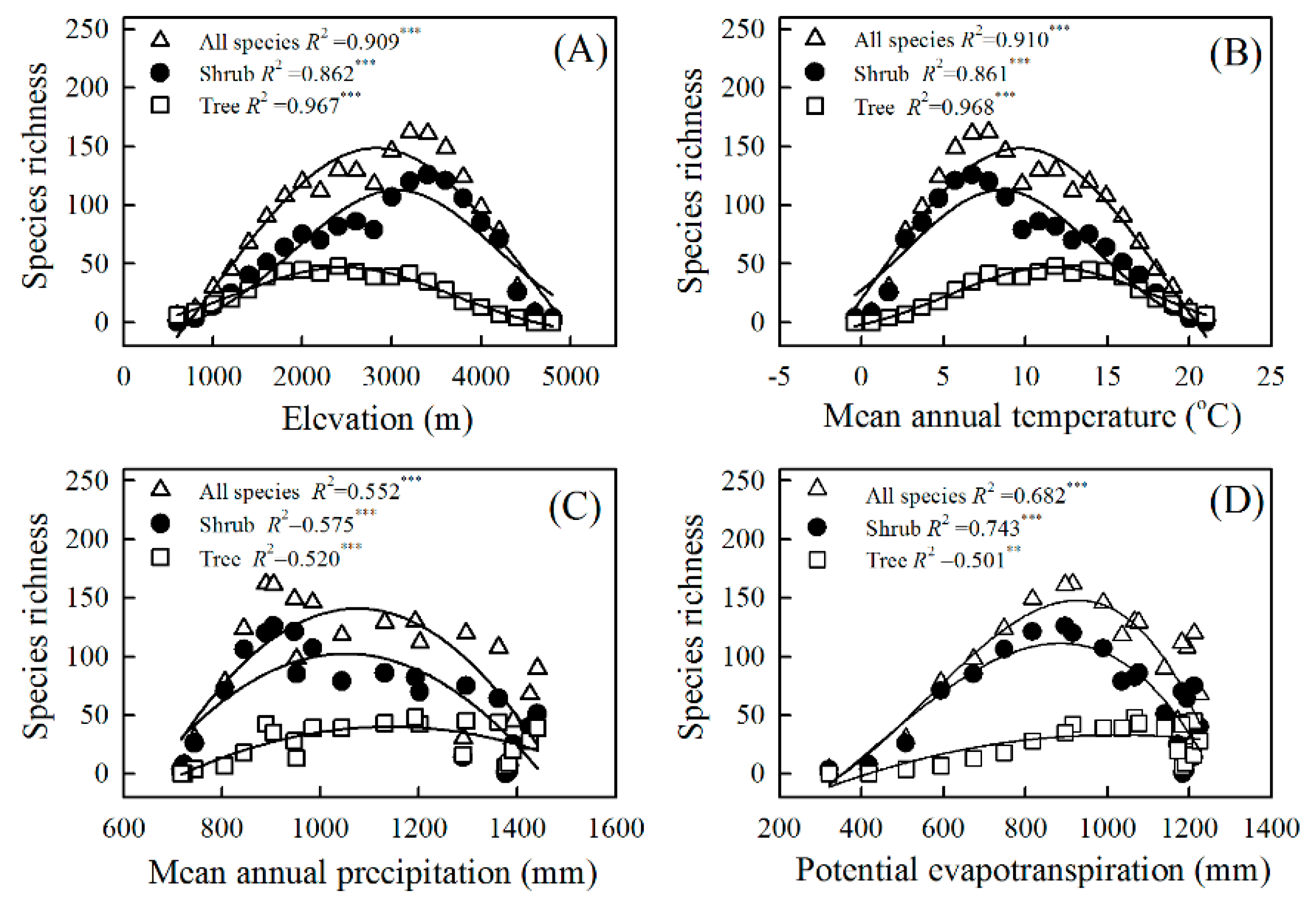
3. Results
3.1. Elevational Patterns of Species Richness
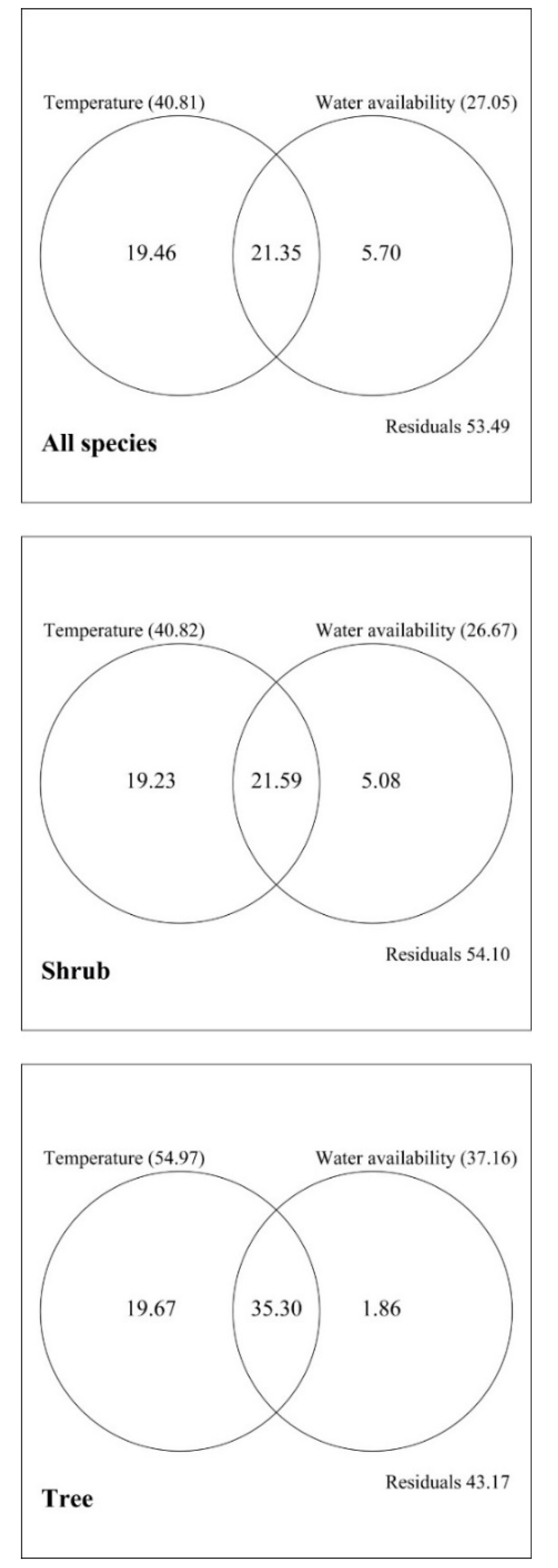
3.2. Explanatory Powers of the MDE, the SAR, and Climate for Elevational Patterns of Richness
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grytnes, J.A.; Vetaas, O.R. Species richness and altitude: A comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient, Nepal. Am. Nat. 2002, 159, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Song, C.Y.; Cao, M.C. Relationships between plant species richness and terrain in middle sub-tropical eastern China. Forests 2017, 8, 344. [Google Scholar] [CrossRef]
- Tang, Z.Y.; Fang, J.Y. A review on the elevational patterns of plant species diversity. Biodivers. Sci. 2004, 12, 20–28. [Google Scholar]
- Zhang, S.-B.; Chen, W.-Y.; Huang, J.-L.; Bi, Y.-F.; Yang, X.-F. Orchid species richness along elevational and environmental gradients in Yunnan, China. PLoS ONE 2015, 10, e0142621. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.F.; Kelt, D.A.; Sun, Z.Y.; Liu, H.X.; Hu, L.J.; Ren, H.; Wen, J. Global variation in elevational diversity patterns. Sci. Rep. 2013, 3, 3007. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. The use of ‘altitude’ in ecological research. Trends Ecol. Evol. 2007, 22, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Rahbek, C. The elevational gradient of species richness: A uniform pattern? Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- Doležal, J.; Šrůtek, M. Altitudinal changes in composition and structure of mountain-temperate vegetation: A case study from the Western Carpathians. Plant Ecol. 2002, 158, 201–221. [Google Scholar] [CrossRef]
- Lomolino, M.V. Elevation gradients of species-density: Historical and prospective views. Glob. Ecol. Biogeogr. 2001, 10, 3–13. [Google Scholar] [CrossRef]
- McCain, C.M. Elevational gradients in diversity of small mammals. Ecology 2005, 86, 366–372. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M.; Robert, R.; Dunn, R.R. What drives elevational patterns of diversity? A test of geometric constraints, climate and species pool effects for pteridophytes on an elevational gradient in Costa Rica. Glob. Ecol. Biogeogr. 2006, 15, 358–371. [Google Scholar] [CrossRef]
- Rai, H.; Khare, R.; Baniya, C.B.; Upreti, D.K.; Gupta, R.K. Elevational gradients of terricolous lichen species richness in the Western Himalaya. Biodivers. Conserv. 2015, 24, 1155–1174. [Google Scholar] [CrossRef]
- Fu, C.Z.; Hua, X.; Li, J.; Chang, Z.; Pu, Z.C.; Chen, J.K. Elevational patterns of frog species richness and endemic richness in the Hengduan Mountains, China: Geometric constraints, area and climate effects. Ecography 2006, 29, 919–927. [Google Scholar] [CrossRef]
- Trigas, P.; Panitsa, M.; Tsiftsis, S. Elevational gradient of vascular plant species richness and endemism in Crete—The effect of post-isolation mountain uplift on a continental island system. PLoS ONE 2013, 8, e59425. [Google Scholar] [CrossRef] [PubMed]
- Colwell, R.K.; Rahbek, C.; Gotelli, N.J. The mid-domain effect and species richness patterns: What have we learned so far? Am. Nat. 2004, 163, E1–E23. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, C. The environmental control of plant species density on a Himalayan elevation gradient. J. Biogeogr. 2005, 32, 999–1018. [Google Scholar] [CrossRef]
- Bachman, S.; Baker, W.J.; Brummitt, N.; Dransfield, J.; Moat, J. Elevational gradients, area and tropical island diversity: An example from the palms of New Guinea. Ecography 2004, 27, 299–310. [Google Scholar] [CrossRef]
- Elsen, P.R.; Tingley, M.W. Global mountain topography and the fate of montane species under climate change. Nat. Clim. Chang. 2015, 5, 772–776. [Google Scholar] [CrossRef]
- Currie, D.J.; Kerr, J.T. Tests of the mid-domain hypothesis: A review of the evidence. Ecol. Monogr. 2008, 78, 3–18. [Google Scholar] [CrossRef]
- Vetaas, O.R. Realized and potential climate niches: A comparison of four Rhododendron tree species. J. Biogeogr. 2002, 29, 545–554. [Google Scholar] [CrossRef]
- Körner, C. Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystem, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Wang, Z.H.; Fang, J.Y.; Tang, Z.Y.; Lin, X. Patterns, determinants and models of woody plant diversity in China. Proc. R. Soc. B. Biol. Sci. 2010. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.H.; Lomolino, M.V. Biogeography, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 1998. [Google Scholar]
- Wang, G.H.; Zhou, G.S.; Yang, L.M.; Li, Z.Q. Distribution, species diversity and life-form spectra of plant communities along an altitudinal gradient in the northern slopes of Qilianshan Mountains, Gansu, China. Plant Ecol. 2003, 165, 169–181. [Google Scholar] [CrossRef]
- Bhattarai, K.R.; Vetaas, O.R.; Grytnes, J.A. Fern species richness along a central Himalayan elevational gradient, Nepal. J. Biogeogr. 2004, 31, 389–400. [Google Scholar] [CrossRef]
- Grime, J.P. Plant Strategies and Vegetation Processes; John Wiley & Sons, Ltd.: Chichester, UK, 1979. [Google Scholar]
- Kubota, Y.; Murata, H.; Kikuzawa, K. Effects of topographic heterogeneity on tree species richness and stand dynamics in a subtropical forest in Okinawa Island, southern Japan. J. Ecol. 2004, 92, 230–240. [Google Scholar] [Green Version]
- Michalet, R.; Maalouf, J.-P.; Choler, P.; Clément, B.; Rosebery, D.; Royer, J.-M.; Schöb, C.; Lortie, C.J. Competition, facilitation and environmental severity shape the relationship between local and regional species richness in plant communities. Ecography 2015, 38, 335–345. [Google Scholar] [CrossRef] [Green Version]
- Whittaker, R.J.; Katherine, J.W. Scale and species richness: Towards a general, hierarchical theory of species diversity. J. Biogeogr. 2001, 28, 453–470. [Google Scholar] [CrossRef]
- Ergeton, J.J.G.; Wilson, S.D. Plant competition over winter in alpine shrubland and grassland, Snowy Mountains, Australia. Arct. Alp. Res. 1993, 25, 124–129. [Google Scholar]
- Tang, Z.-Y.; Ke, J.-H. Altitudinal patterns of plant species diversity in Mt. Niubeiliang, Qinling Mountains. Biodivers. Sci. 2004, 12, 108–114. [Google Scholar]
- Wang, Z.; Tang, Z.; Fang, J. Altitudinal patterns of seed plant richness in the Gaoligong Mountains, south-east Tibet, China. Divers. Distrib. 2007, 13, 845–854. [Google Scholar] [CrossRef]
- Aiba, S.; Kohyama, T. Tree species stratification in relation to allometry and demography in a warm-temperate rain forest. J. Ecol. 1996, 84, 207–218. [Google Scholar] [CrossRef]
- Ryan, M.G.; Yoder, B.J. Hydraulic limits to tree height and tree growth. Bioscience 1997, 47, 235–242. [Google Scholar] [CrossRef]
- Singh, K.K.; Kumar, S.; Rai, L.K.; Krishn, A.P. Rhododendrons conservation in the Sikkim Himalaya. Curr. Sci. 2003, 85, 602–606. [Google Scholar]
- Fang, M.; Fang, R.; He, M.; Hu, L.; Yang, H.; Qin, H.; Min, T.; Chamberlain, D.F.; Stevens, P.F.; Wallace, G.D.; et al. Flora of China; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 2005; Volume 14, pp. 242–517. [Google Scholar]
- Wu, Z.Y. Flora Yunnanica (Tomus 4); Science Press: Beijing, China, 1986. [Google Scholar]
- Kessler, M. Elevational gradients in species richness and endemism of selected plant groups in the central Bolivian Andes. Plant Ecol. 2000, 149, 181–193. [Google Scholar] [CrossRef]
- Yang, Y.M.; Tian, K.; Hao, J.M.; Pei, S.J.; Yang, Y.X. Biodiversity and biodiversity conservation in Yunnan, China. Biodivers. Conserv. 2004, 13, 813–826. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Zhu, Y.C. Vegetation of Yunnan; Science Press: Beijing, China, 1987. [Google Scholar]
- Wu, Z.Y. Flora Yunnanica (Tomus 5); Science Press: Beijing, China, 1991. [Google Scholar]
- Wolf, J.H.D.; Flamenco-Sandoval, A. Patterns in species richness and distribution of vascular epiphytes in Chiapas, Mexico. J. Biogeogr. 2003, 30, 1689–1707. [Google Scholar] [CrossRef]
- Qian, H.; Ricklefs, R.E. Global concordance in diversity patterns of vascular plants and terrestrial vertebrates. Ecol. Lett. 2008, 11, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration-Guidelines for Computing Crop Water Requirements-FAO Irrigation and Drainage Paper 56; FAO-Food and Agriculture Organization of the United Nations: Rome, Italy, 1998; Volume 156, p. 178. [Google Scholar]
- Colwell, R.K. RangeModel: Tools for exploring and assessing geometric constraints on species richness (the mid-domain effect) along transects. Ecography 2008, 31, 4–7. [Google Scholar] [CrossRef]
- Borcard, D.; Legendre, P.; Drapeau, P. Partialling out the spatial component of ecological variation. Ecology 1992, 73, 1045–1055. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; O’Hara, R.B.; Simpson, GL.; Solymos, P.; Stevens, M.H.; Wagner, H. Vegan: Community Ecology Package, R Package Version 2.4-3. 2010. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 16 May 2017).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: http://www.R-project.org/ (accessed on 12 May 2017).
- Legendre, P.; Gallagher, E.D. Ecologically meaningful transformations for ordination of species data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peres-Neto, P.; Legendre, P.; Dray, S.; Borcard, D. Variation partitioning of species data matrices: Estimation and comparison of fractions. Ecology 2006, 87, 2614–2625. [Google Scholar] [CrossRef]
- Tao, J.; Zang, R.G.; Yu, C.Y. Altitudinal patterns of plant communities and species diversity in the Habaxueshan Mountains, Yunnan, China. Sci. Silvae Sin. 2011, 47, 1–6. [Google Scholar]
- Li, X.-H.; Wang, D.-D.; Li, H. Feature of Ericaceae flora in Gaoligong Mountains. J. West China For. Sci. 2017, 46 (Suppl. II), 112–118. Available online: http://159.226.100.28/ZK/detail.aspx?id=674277914 (accessed on 26 July 2018). (In Chinese).
- O’Brien, E.M.; Field, R.; Whittaker, R.J. Climatic gradients in woody plant (tree and shrub) diversity: Water-energy dynamics, residual variation, and topography. Oikos 2000, 89, 588–600. [Google Scholar] [CrossRef]
- Smith, D.D.; Sperry, J.S. Coordination between water transport capacity, biomass growth, metabolic scaling and species stature in co-occurring shrub and tree species. Plant Cell Environ. 2014, 37, 2679–2690. [Google Scholar] [CrossRef] [PubMed]
- Harms, K.H.; Condit, R.; Hubbell, S.P.; Foster, R.B. Habitat associations of trees and shrubs in a 50-ha neotropical forest plot. J. Ecol. 2001, 89, 947–959. [Google Scholar] [CrossRef] [Green Version]
- Brodribb, T.J.; Field, T.S. Stem hydraulic supply is linked to leaf photosynthetic capacity: Evidence from New Caledonian and Tasmanian rainforests. Plant Cell Environ. 2000, 23, 1381–1388. [Google Scholar] [CrossRef]
- O’Brien, E.M. Water-energy dynamics, climate, and prediction of woody plant species richness: An interim general model. J. Biogeogr. 1998, 25, 379–398. [Google Scholar] [CrossRef]
- Cordero, R.A.; Nilsen, R.T. Effects of summer drought and winter freezing on stem hydraulic conductivity of Rhododendron species from contrasting climates. Tree Physiol. 2002, 22, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Wang, J.J.; Lai, L.M.; Jiang, L.H.; Zhuang, P.; Zhang, L.H.; Zheng, Y.R.; Baskin, J.M.; Baskin, C.C. Geographic variation in seed traits within and among forty-two species of Rhododendron (Ericaceae) on the Tibetan plateau: Relationships with altitude, habitat, plant height, and phylogeny. Ecol. Evol. 2014, 4, 1913–1923. [Google Scholar] [CrossRef] [PubMed]
- Francis, A.P.; Currie, D.J. A globally consistent richness-climate relationship for angiosperms. Am. Nat. 2003, 161, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Fernàndez-Martínez, J.; Fransi, M.A.; Fleck, I. Ecophysiological responses of Betula pendula, Pinus uncinata and Rhododendron ferrugineum in the Catalan Pyrenees to low summer rainfall. Tree Physiol. 2016, 36, 1520–1535. [Google Scholar] [CrossRef] [PubMed]
- Caroline, M.W.; Hugh, A.L.H.; Brent, J.S. Cold truths: How winter drives responses of terrestrial organisms to climate change. Biol. Rev. 2015, 90, 214–235. [Google Scholar]
- Rajaniemi, T.K. Why does fertilization reduce plant species diversity? Testing three competition-based hypotheses. J. Ecol. 2002, 90, 316–324. [Google Scholar] [CrossRef] [Green Version]
- Brown, R.L.; Jacobs, L.A.; Peet, R.K. Species richness: Small scale. In Encyclopedia of Life Sciences; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2007. [Google Scholar]
- Dobrowski, S.Z.; Swanson, A.K.; Abatzoglou, J.T.; Holden, Z.A.; Safford, H.D.; Schwartz, M.K.; Gavin, D.G. Forest structure and species traits mediate projected recruitment declines in western US tree species. Glob. Ecol. Biogeogr. 2015, 24, 917–927. [Google Scholar] [CrossRef]

| Intercept | Lifeform | Elevation | MAT | MAP | PET | Interaction | Adjusted R2 (%) | AIC |
|---|---|---|---|---|---|---|---|---|
| −132.83 | 150.42 | 0.20 | - | - | - | −0.20 | 77.82 | 210.5 |
| 816.91 | −811.15 | - | −39.44 | - | - | 39.93 | 77.91 | 210.1 |
| 1199.63 | −1347.50 | - | - | −0.89 | - | 1.11 | 72.02 | 215.3 |
| 914.41 | −1002.73 | - | - | - | −0.79 | 1.02 | 77.05 | 210.9 |
| −93.16 | −51.62 | - | −21.53 | −0.02 | 0.49 | - | 83.64 | 204.2 |
| −116.35 | - | - | −18.22 | −0.05 | 0.47 | - | 83.94 | 203.1 |
| −316.60 | - | - | 40.18 | 0.46 | - | −0.04 | 84.24 | 202.7 |
| −142.50 | - | - | 10.72 | - | 0.39 | −0.02 | 93.01 | 184.8 |
| −547.20 | - | - | - | 0.61 | 0.83 | −0.0007 | 68.37 | 218.0 |
| Environmental Variable | All Species | Shrubs | Trees |
|---|---|---|---|
| Temperature of the coldest month | 0.840 *** | 0.781 *** | 0.838 *** |
| Temperature of the warmest month | 0.905 *** | 0.826 *** | 0.911 *** |
| Temperature seasonality | 0.662 *** | 0.632 *** | 0.667 *** |
| Precipitation of the driest month | 0.497 ** | 0.478 ** | 0.604 *** |
| Precipitation of the wettest month | 0.758 *** | 0.734 *** | 0.691 *** |
| Precipitation seasonality | 0.036 | 0.026 | 0.065 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.-H.; Cai, Y.-F.; Zhang, L.; Xu, C.-K.; Zhang, S.-B. Species Richness of the Family Ericaceae along an Elevational Gradient in Yunnan, China. Forests 2018, 9, 511. https://doi.org/10.3390/f9090511
Wang J-H, Cai Y-F, Zhang L, Xu C-K, Zhang S-B. Species Richness of the Family Ericaceae along an Elevational Gradient in Yunnan, China. Forests. 2018; 9(9):511. https://doi.org/10.3390/f9090511
Chicago/Turabian StyleWang, Ji-Hua, Yan-Fei Cai, Lu Zhang, Chuan-Kun Xu, and Shi-Bao Zhang. 2018. "Species Richness of the Family Ericaceae along an Elevational Gradient in Yunnan, China" Forests 9, no. 9: 511. https://doi.org/10.3390/f9090511





