Soil Aggregation and Organic Carbon Dynamics in Poplar Plantations
Abstract
1. Introduction
2. Materials and Methods
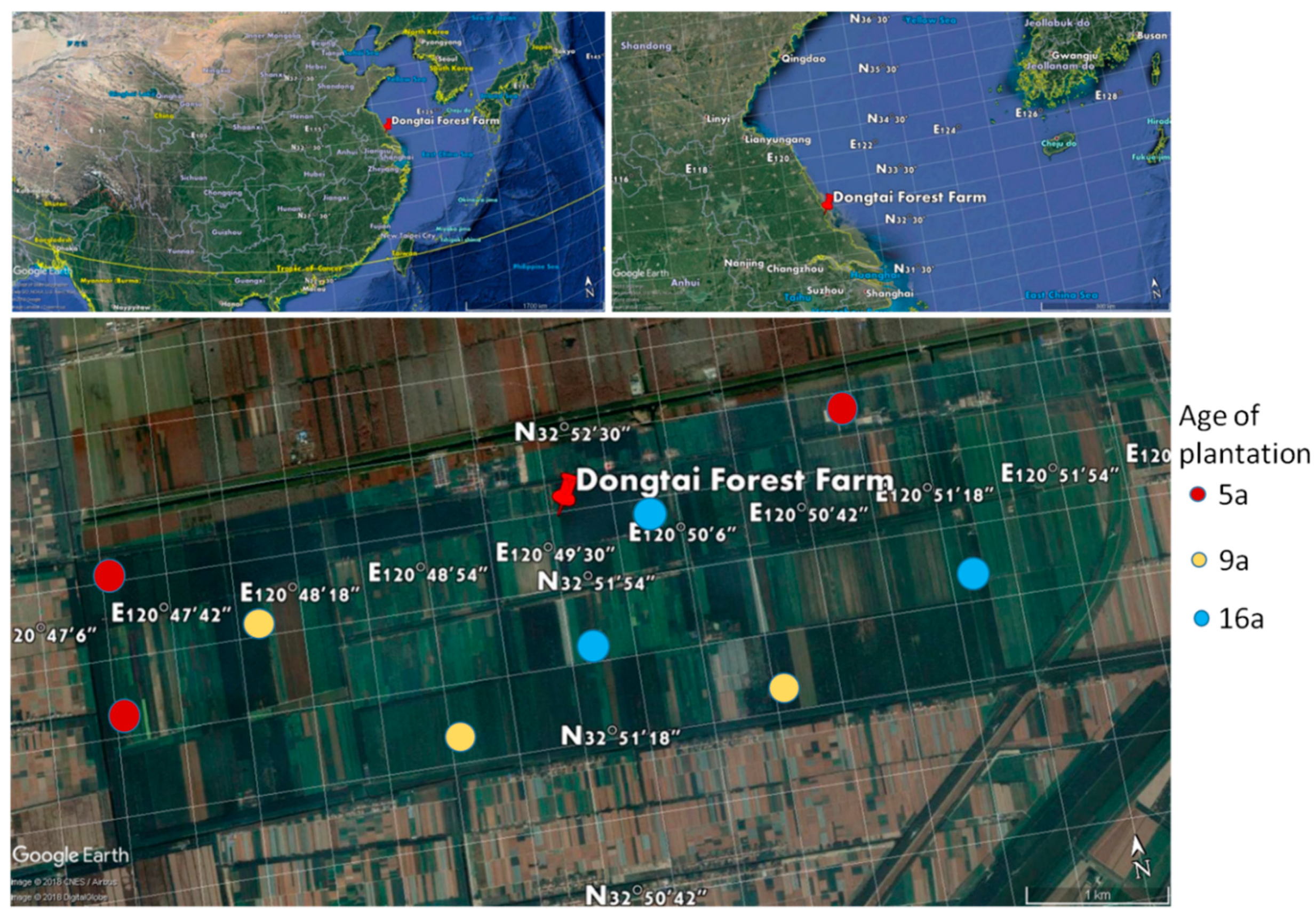
2.1. Study Area and Sites
2.2. Sampling Design
2.3. Field Measurements and Sampling
2.4. Laboratory Analyses
2.4.1. Water-Stable Aggregates
2.4.2. SOC
2.4.3. Soil Microbial Biomass Carbon
2.4.4. Litterfall, Fine-Root Biomass, and SMBC
2.5. Statistical Analysis
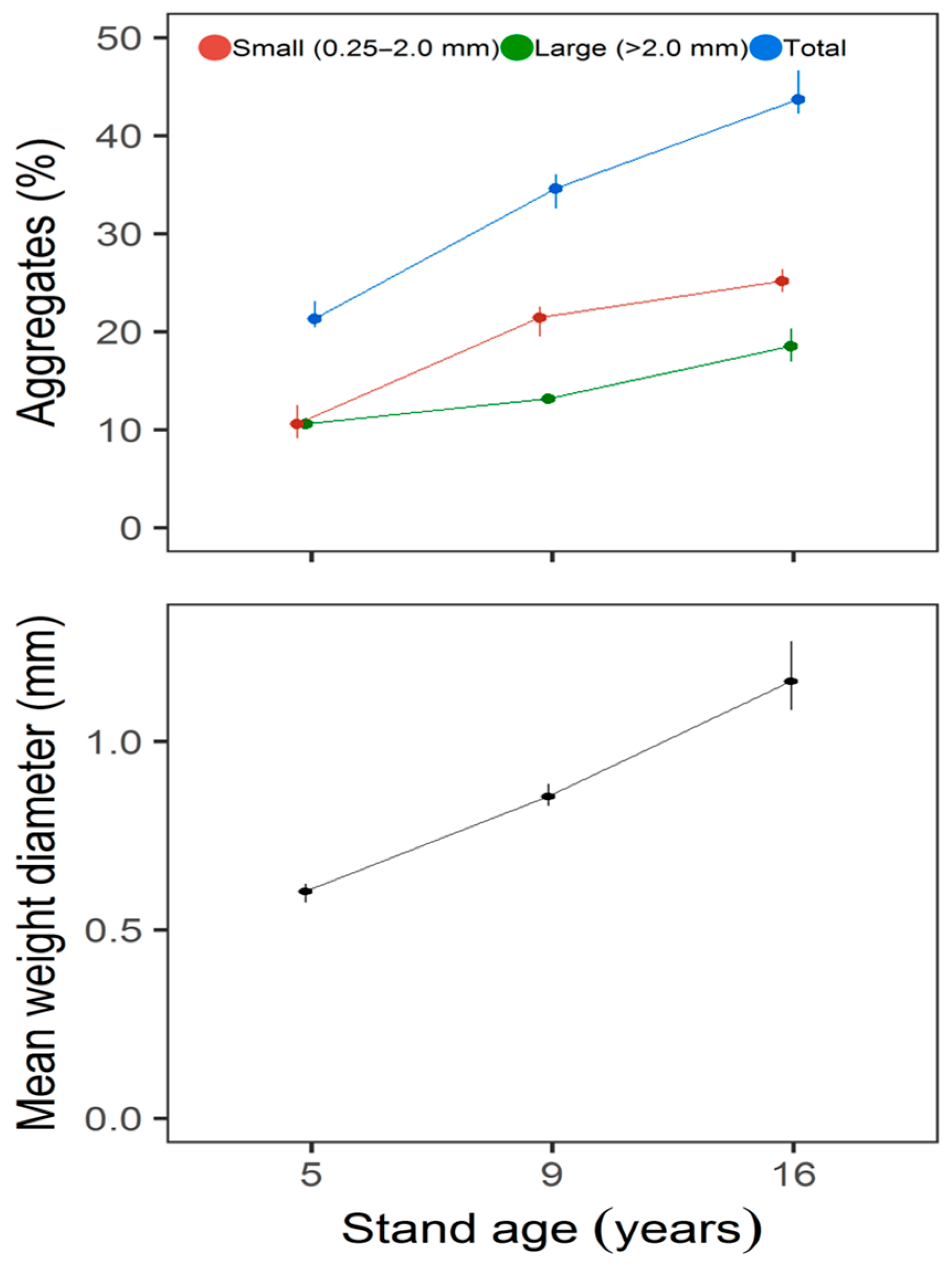
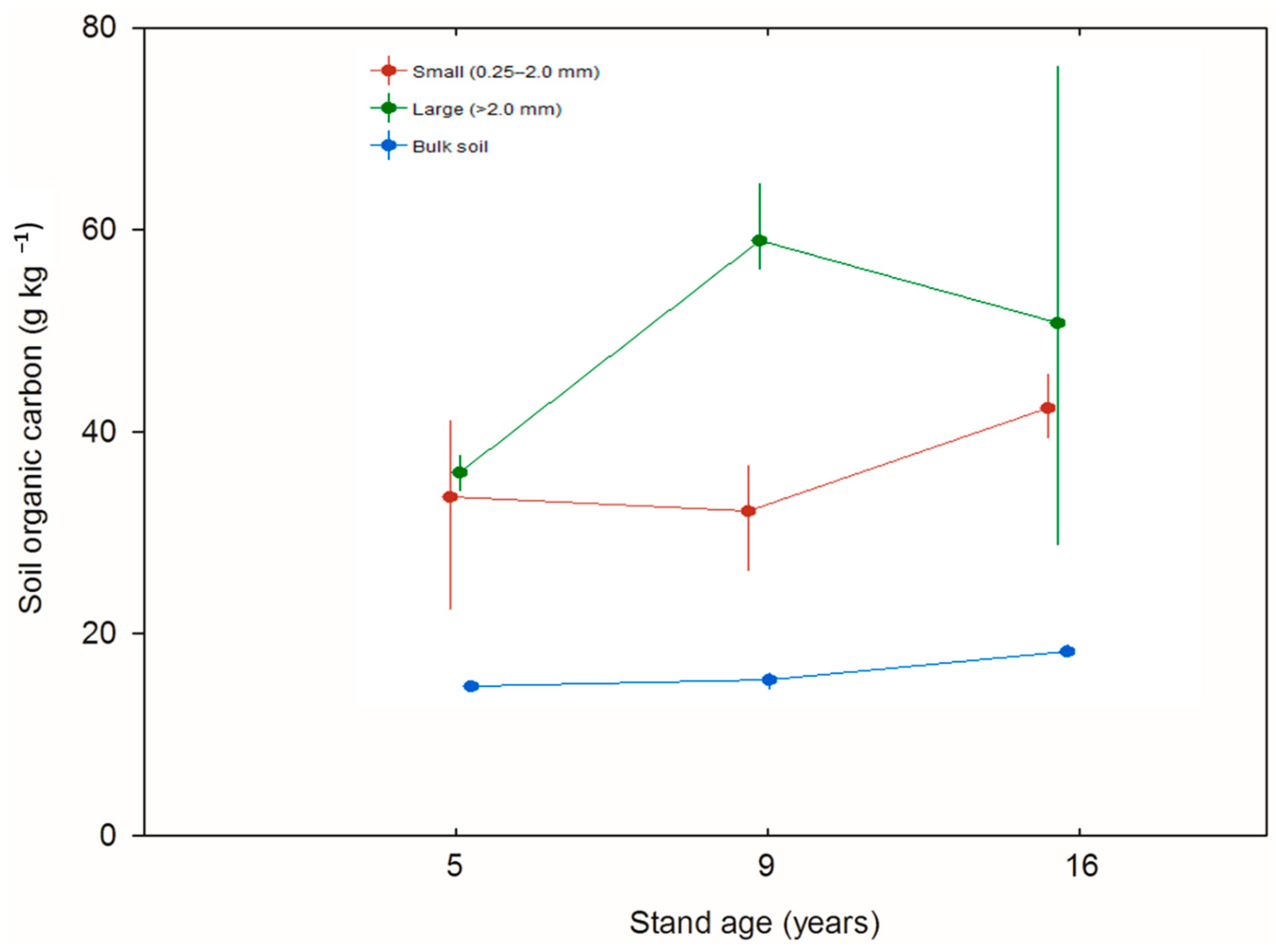
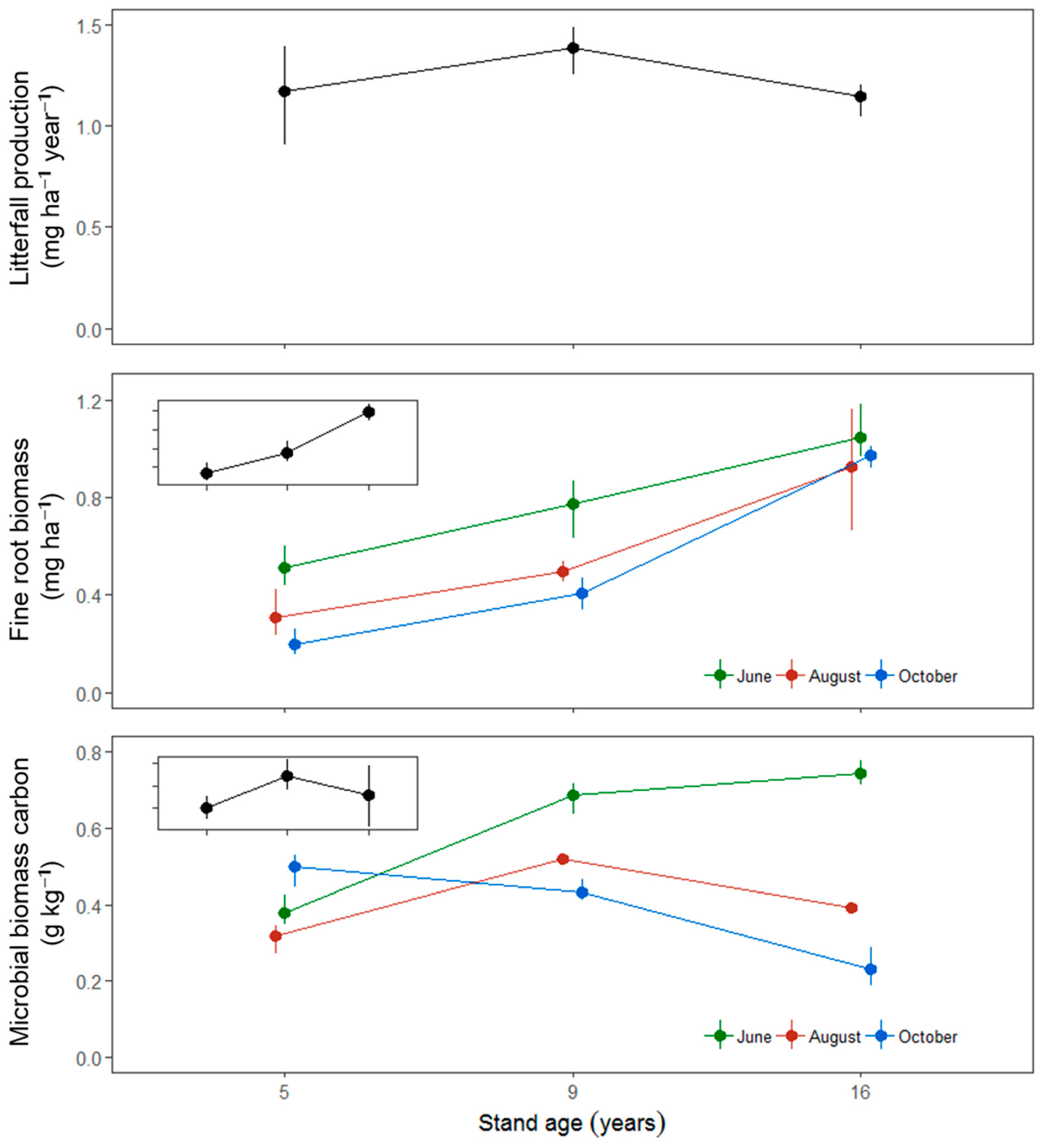
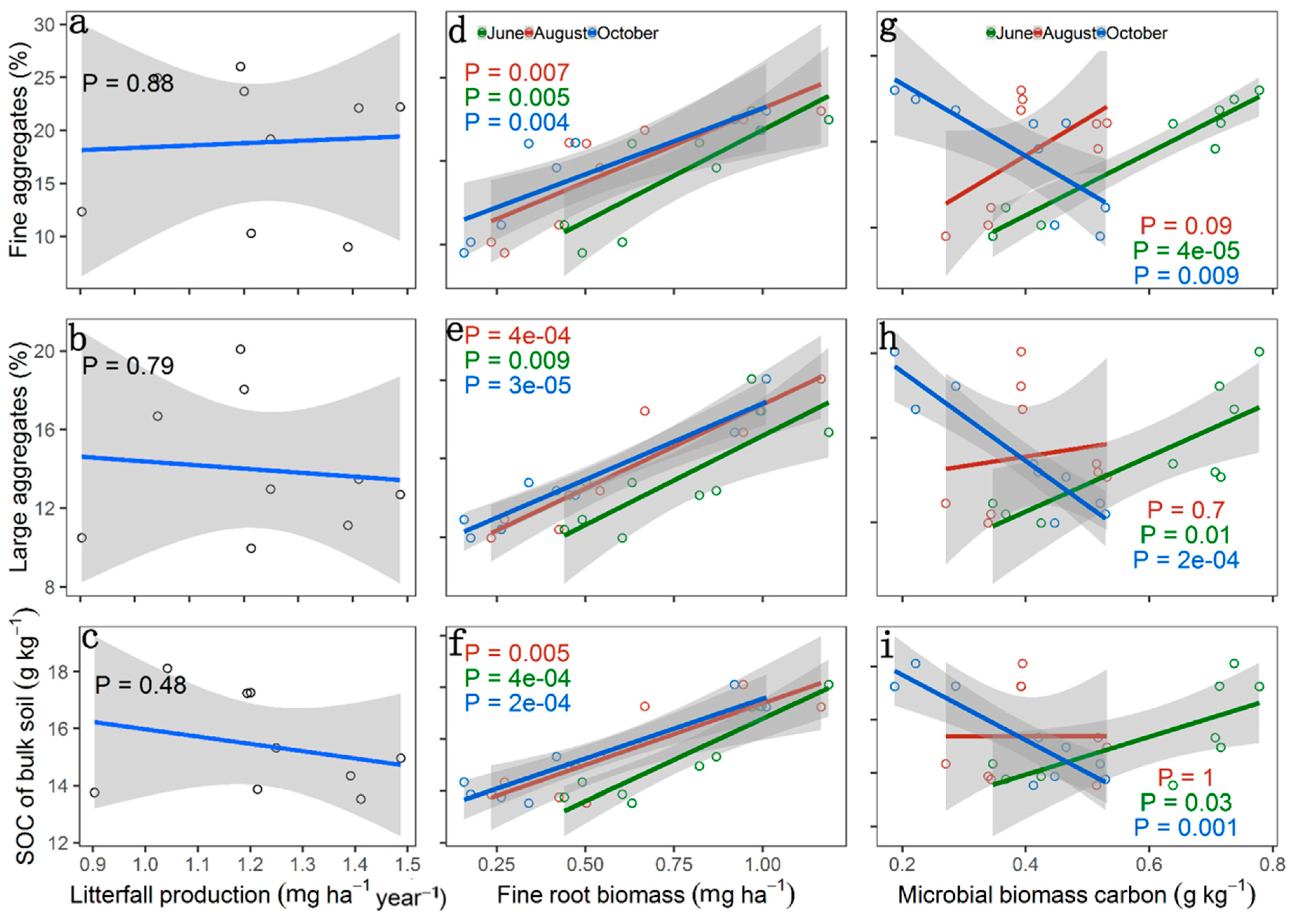
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amezketa, E. Soil aggregate stability: A review. J. Sustain. Agric. 1999, 14, 83–151. [Google Scholar] [CrossRef]
- Bronick, C.J.; Lal, R. Soil structure and management: A review. Geoderma 2005, 124, 3–22. [Google Scholar] [CrossRef]
- Plaza-Bonilla, D.; Cantero-Martínez, C.; Viñas, P.; Álvaro-Fuentes, J. Soil aggregation and organic carbon protection in a no-tillage chronosequence under Mediterranean conditions. Geoderma 2013, 193, 76–82. [Google Scholar] [CrossRef]
- Tisdall, J.; Oades, J.M. Organic matter and water-stable aggregates in soils. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Spaccini, R.; Piccolo, A. Effects of field managements for soil organic matter stabilization on water-stable aggregate distribution and aggregate stability in three agricultural soils. J. Geochem. Explor. 2013, 129, 45–51. [Google Scholar] [CrossRef]
- Chai, Y.J.; Zeng, X.B.; E, S.Z.; Bai, L.Y.; Su, S.M.; Huang, T. Effects of freeze-thaw on aggregate stability and the organic carbon and nitrogen enrichment ratios in aggregate fractions. Soil Use Manag. 2014, 30, 507–516. [Google Scholar] [CrossRef]
- Chen, C.F.; Liu, W.J.; Jiang, X.J.; Wu, J.E. Effects of rubber-based agroforestry systems on soil aggregation and associated soil organic carbon: Implications for land use. Geoderma 2017, 299, 13–24. [Google Scholar] [CrossRef]
- Nzeyimana, I.; Hartemink, A.E.; Ritsema, C.; Stroosnijder, L.; Lwanga, E.H.; Geissen, V. Mulching as a strategy to improve soil properties and reduce soil erodibility in coffee farming systems of Rwanda. Catena 2017, 149, 43–51. [Google Scholar] [CrossRef]
- Wang, S.Q.; Li, T.X.; Zheng, Z.C. Distribution of microbial biomass and activity within soil aggregates as affected by tea plantation age. Catena 2017, 153, 1–8. [Google Scholar] [CrossRef]
- Zhong, X.L.; Li, J.T.; Li, X.J.; Ye, Y.C.; Liu, S.S.; Hallett, P.D.; Ogden, M.R.; Naveed, M. Physical protection by soil aggregates stabilizes soil organic carbon under simulated N deposition in a subtropical forest of China. Geoderma 2017, 285, 323–332. [Google Scholar] [CrossRef]
- Wu, H.; Guo, Z.; Peng, C. Land use induced changes of organic carbon storage in soils of China. Glob. Chang. Biol. 2003, 9, 305–315. [Google Scholar] [CrossRef]
- Meersmans, J.; De Ridder, F.; Canters, F.; De Baets, S.; Van Molle, M. A multiple regression approach to assess the spatial distribution of Soil Organic Carbon (SOC) at the regional scale (Flanders, Belgium). Geoderma 2008, 143, 1–13. [Google Scholar] [CrossRef]
- Hajabbasi, M.A.; Jalalian, A.; Karimzadeh, H.R. Deforestation effects on soil physical and chemical properties, Lordegan, Iran. Plant Soil 1997, 190, 301–308. [Google Scholar] [CrossRef]
- Zheng, F.L.; He, X.B.; Gao, X.T.; Zhang, C.E.; Tang, K.L. Effects of erosion patterns on nutrient loss following deforestation on the Loess Plateau of China. Agric. Ecosyst. Environ. 2005, 108, 85–97. [Google Scholar] [CrossRef]
- Karhu, K.; Wall, A.; Vanhala, P.; Liski, J.; Esala, M.; Regina, K. Effects of afforestation and deforestation on boreal soil carbon stocks—Comparison of measured C stocks with Yasso07 model results. Geoderma 2011, 164, 33–45. [Google Scholar] [CrossRef]
- Chen, H.Y.H.; Shrestha, B.M. Stand age, fire and clearcutting affect soil organic carbon and aggregation of mineral soils in boreal forests. Soil Biol. Biochem. 2012, 50, 149–157. [Google Scholar] [CrossRef]
- Murty, D.; Kirschbaum, M.U.F.; McMurtrie, R.E.; McGilvray, H. Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Glob. Chang. Biol. 2002, 8, 105–123. [Google Scholar] [CrossRef]
- Wei, X.; Li, X.; Jia, X.; Shao, M. Accumulation of soil organic carbon in aggregates after afforestation on abandoned farmland. Biol. Fertil. Soil 2013, 49, 637–646. [Google Scholar] [CrossRef]
- Blankinship, J.C.; Fonte, S.J.; Six, J.; Schimel, J.P. Plant versus microbial controls on soil aggregate stability in a seasonally dry ecosystem. Geoderma 2016, 272, 39–50. [Google Scholar] [CrossRef]
- Denef, K.; Six, J.; Bossuyt, H.; Frey, S.D.; Elliott, E.T.; Merckx, R.; Paustian, K. Influence of dry-wet cycles on the interrelationship between aggregate, particulate organic matter, and microbial community dynamics. Soil Biol. Biochem. 2001, 33, 1599–1611. [Google Scholar] [CrossRef]
- Garcia-Franco, N.; Albaladejo, J.; Almagro, M.; Martínez-Mena, M. Beneficial effects of reduced tillage and green manure on soil aggregation and stabilization of organic carbon in a Mediterranean agroecosystem. Soil Tillage Res. 2015, 153, 66–75. [Google Scholar] [CrossRef]
- Demenois, J.; Carriconde, F.; Bonaventure, P.; Maeght, J.L.; Stokes, A.; Rey, F. Impact of plant root functional traits and associated mycorrhizas on the aggregate stability of a tropical Ferralsol. Geoderma 2018, 312, 6–16. [Google Scholar] [CrossRef]
- Winsome, T.S.; Mccoll, J.G. Changes in chemistry and aggregation of a California forest soil worked by the earthworm Argilophilus papillifer eisen (megascolecidae). Soil Biol. Biochem. 1998, 30, 1677–1687. [Google Scholar] [CrossRef]
- Xiang, H.; Zhang, L.; Wen, D. Change of soil carbon fractions and water-stable aggregates in a forest ecosystem succession in South China. Forests 2015, 8, 2703–2718. [Google Scholar] [CrossRef]
- Zeglin, L.H.; Stursova, M.; Sinsabaugh, R.L.; Collins, S.L. Microbial responses to nitrogen addition in three contrasting grassland ecosystems. Oecologia 2007, 154, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Bardgett, R.D.; Freeman, C.; Ostle, N.J. Microbial contributions to climate change through carbon cycle feedbacks. ISME J. 2008, 2, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Pei, Z.; Su, J.; Zhang, J.; Zheng, Y.; Ni, J.; Xiao, C.; Wang, R. Comparing soil organic carbon dynamics in perennial grasses and shrubs in a saline-alkaline arid region, northwestern China. PLoS ONE 2012, 7, e42927. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.B.; Lee, C.H.; Jung, K.Y.; Park, K.D.; Lee, D.; Kim, P.J. Changes of soil organic carbon and its fractions in relation to soil physical properties in a long-term fertilized paddy. Soil Tillage Res. 2009, 104, 227–232. [Google Scholar] [CrossRef]
- Linsler, D.; Geisseler, D.; Loges, R.; Taube, F.; Ludwig, B. Temporal dynamics of soil organic matter composition and aggregate distribution in permanent grassland after a single tillage event in a temperate climate. Soil Tillage Res. 2013, 126, 90–99. [Google Scholar] [CrossRef]
- Six, J.; Elliott, E.; Paustian, K. Soil macroaggregate turnover and microaggregate formation: A mechanism for C sequestration under no-tillage agriculture. Soil Biol. Biochem. 2000, 32, 2099–2103. [Google Scholar] [CrossRef]
- Ussiri, D.; Lal, R.; Jacinthe, P. Soil properties and carbon sequestration of afforested pastures in reclaimed minesoils of Ohio. Soil Sci. Soc. Am. J. 2006, 70, 1797–1806. [Google Scholar] [CrossRef]
- Asaye, Z.; Zewdie, S. Fine root dynamics and soil carbon accretion under thinned and un-thinned Cupressus lusitanica stands in, Southern Ethiopia. Plant Soil 2013, 366, 261–271. [Google Scholar] [CrossRef]
- Lehmann, A.; Rillig, M.C. Are there temporal trends in root architecture and soil aggregation for Hordeum vulgare breeding lines? Appl. Soil Ecol. 2013, 65, 31–34. [Google Scholar] [CrossRef]
- Yuan, Z.Y.; Chen, H.Y.H. Fine root biomass, production, turnover rates, and nutrient contents in boreal forest ecosystems in relation to species, climate, fertility, and stand age: Literature review and meta-analyses. Crit. Rev. Plant Sci. 2010, 29, 204–221. [Google Scholar] [CrossRef]
- Brassard, B.W.; Chen, H.Y.H.; Cavard, X.; Laganière, J.; Reich, P.B.; Bergeron, Y.; Paré, D.; Yuan, Z.; Chen, H. Tree species diversity increases fine root productivity through increased soil volume filling. J. Ecol. 2013, 101, 210–219. [Google Scholar] [CrossRef]
- Barbhuiya, A.; Arunachalam, A.; Pandey, H.; Khan, M.; Arunachalam, K. Fine root dynamics in undisturbed and disturbed stands of a tropical wet evergreen forest in northeast India. Trop. Ecol. 2012, 53, 69–79. [Google Scholar]
- Miller, R.; Jastrow, J. Hierarchy of root and mycorrhizal fungal interactions with soil aggregation. Soil Biol. Biochem. 1990, 22, 579–584. [Google Scholar] [CrossRef]
- Ruiz-Peinado, R.; Bravo-Oviedo, A.; López-Senespleda, E.; Montero, G.; Río, M. Do thinnings influence biomass and soil carbon stocks in Mediterranean maritime pinewoods? Eur. J. For. Res. 2012, 132, 253–262. [Google Scholar] [CrossRef]
- Liang, W.; Hu, H.; Liu, F.; Zhang, D. Research advance of biomass and carbon storage of poplar in China. J. For. Res. 2006, 17, 75–79. [Google Scholar] [CrossRef]
- Li, Y.; Su, X.; Zhang, B.; Huang, Q.; Zhang, X.; Huang, R. Expression of jasmonic ethylene responsive factor gene in transgenic poplar tree leads to increased salt tolerance. Tree Physiol. 2009, 29, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Ju, X. Effect of unrestricted nitrogen and irrigation application on soil carbon and nitrogen pools in greenhouse vegetable systems. Better Crops Plant Food 2010, 94, 29–31. [Google Scholar]
- National Meteorological Information Center. Annual Data Sets of Meteorological Observation in China. Available online: http://data.cma.cn/ (accessed on 4 May 2010).
- Johnson, E.A.; Miyanishi, K. Testing the assumptions of chronosequences in succession. Ecol. Lett. 2008, 11, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.R.; Wardle, D.A.; Bardgett, R.D.; Clarkson, B.D. The use of chronosequences in studies of ecological succession and soil development. J. Ecol. 2010, 98, 725–736. [Google Scholar] [CrossRef]
- Cavard, X.; Bergeron, Y.; Chen, H.Y.H.; Pare, D.; Laganiere, J.; Brassard, B. Competition and facilitation between tree species change with stand development. Oikos 2011, 120, 1683–1695. [Google Scholar] [CrossRef]
- Gee, G.W.; Bauder, J.W. Particle-Size Analysis—1; Soil Science Society of America, American Society of Agronomy: Madison, WI, USA, 1986; pp. 383–411. [Google Scholar]
- Jennings, S.B.; Brown, N.D.; Sheil, D. Assessing forest canopies and understorey illumination: Canopy closure, canopy cover and other measures. Forests 1999, 72, 59–74. [Google Scholar] [CrossRef]
- Franzluebbers, A.; Arshad, M. Soil microbial biomass and mineralizable carbon of water-stable aggregates. Soil Sci. Soc. Am. J. 1997, 61, 1090–1097. [Google Scholar]
- Su, Y.; Wang, F.; Suo, D.; Zhang, Z.; Du, M. Long-term effect of fertilizer and manure application on soil-carbon sequestration and soil fertility under the wheat–wheat–maize cropping system in northwest China. Nutr. Cycl. Agroecosyst. 2006, 75, 285–295. [Google Scholar] [CrossRef]
- Adesodun, J.; Adeyemi, E.; Oyegoke, C. Distribution of nutrient elements within water-stable aggregates of two tropical agro-ecological soils under different land uses. Soil Tillage Res. 2007, 92, 190–197. [Google Scholar] [CrossRef]
- Chen, G.S.; Yang, Z.J.; Gao, R.; Xie, J.S.; Guo, J.F.; Huang, Z.Q.; Yang, Y.S. Carbon storage in a chronosequence of Chinese fir plantations in southern China. For. Ecol. Manag. 2013, 300, 68–76. [Google Scholar] [CrossRef]
- Marin, S.; Andrea, L.E.; Ramona, S.L. Assessment of metals bioavailability to vegetables under field conditions using DGT, single extractions and multivariate statistics. Chem. Cent. J. 2012, 6, 119. [Google Scholar]
- Vance, E.; Brookes, P.; Jenkinson, D. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Yuan, Z.Y.; Chen, H.Y.H.; Ostle, N. Fine root dynamics with stand development in the boreal forest. Funct. Ecol. 2012, 26, 991–998. [Google Scholar] [CrossRef]
- Beyer, F.; Hertel, D.; Jung, K.; Fender, A.C.; Leuschner, C. Competition effects on fine root survival of Fagus sylvatica and Fraxinus excelsior. For. Ecol. Manag. 2013, 302, 14–22. [Google Scholar] [CrossRef]
- Canty, A.; Ripley, B. Package ‘Boot’. Available online: http://cran.r-project.org/web/packages/boot/index.html (accessed on 1 March 2017).
- Bates, D.; Bolker, B.; Walker, S.; Christensen, R.H.B.; Singmann, H.; Dai, B.; Grothendieck, G. Lme4: Linear mixed-effects models using Eigen and S4. R Package Version 2017, 1, 1–13. [Google Scholar]
- Moritz, S.; Cule, E. Package ‘Ridge’: Ridge Regression with Automatic Selection of the Penalty Parameter. Available online: http://github.com/SteffenMoritz/ridge (accessed on 25 March 2017).
- R Development Core Team. R: A Language and Environment for Statistical Computing, Version 3.3.1; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Qiu, L.; Wei, X.; Gao, J.; Zhang, X. Dynamics of soil aggregate-associated organic carbon along an afforestation chronosequence. Plant Soil 2015, 391, 237–251. [Google Scholar] [CrossRef]
- Jackson, R.B.; Lajtha, K.; Crow, S.E.; Hugelius, G.; Kramer, M.G.; Piñeiro, G. The Ecology of Soil Carbon: Pools, Vulnerabilities, and Biotic and Abiotic Controls. Annu. Rev. Ecol. Evolut. Syst. 2017, 48, 419–445. [Google Scholar] [CrossRef]
- Olupot, G.; Daniel, H.; Lockwood, P.; Mchenry, M.; Mcleod, M.; Gilkes, R.J.; Prakongkep, N. Root contributions to long-term storage of soil organic carbon: Theories, mechanisms and gaps. In Proceedings of the 19th World Congress of Soil Science: Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; pp. 112–115. [Google Scholar]
- Garcia-Franco, N.; Wiesmeier, M.; Goberna, M.; Martínez-Mena, M.; Albaladejo, J. Carbon dynamics after afforestation of semiarid shrublands: Implications of site preparation techniques. For. Ecol. Manag. 2014, 319, 107–115. [Google Scholar] [CrossRef]
- Querejeta, J.I.; Roldan, A.; Albaladejo, J.; Castillo, V. The role of mycorrhizae, site preparation, and organic amendment in the afforestation of a semi-arid Mediterranean site with Pinus halepensis. For. Sci. 1998, 44, 203–211. [Google Scholar]
- Jastrow, J. Soil aggregate formation and the accrual of particulate and mineral-associated organic matter. Soil Biol. Biochem. 1996, 28, 665–676. [Google Scholar] [CrossRef]
- Helfrich, M.; Ludwig, B.; Thoms, C.; Gleixner, G.; Flessa, H. The role of soil fungi and bacteria in plant litter decomposition and macroaggregate formation determined using phospholipid fatty acids. Appl. Soil Ecol. 2015, 96, 261–264. [Google Scholar] [CrossRef]
- Gupta, V.V.S.R.; Germida, J.J. Soil aggregation: Influence on microbial biomass and implications for biological processes. Soil Biol. Biochem. 2015, 80, A3–A9. [Google Scholar] [CrossRef]
- McNally, S.R.; Laughlin, D.C.; Rutledge, S.; Dodd, M.B.; Six, J.; Schipper, L.A. Root carbon inputs under moderately diverse sward and conventional ryegrass-clover pasture: Implications for soil carbon sequestration. Plant Soil 2015, 392, 289–299. [Google Scholar] [CrossRef]
- Mardhiah, U.; Caruso, T.; Gurnell, A.; Rillig, M.C. Just a matter of time: Fungi and roots significantly and rapidly aggregate soil over four decades along the Tagliamento River, NE Italy. Soil Biol. Biochem. 2014, 75, 133–142. [Google Scholar] [CrossRef]
- Imhoff, M.L.; Bounoua, L.; Ricketts, T.; Loucks, C.; Harriss, R.; Lawrence, W.T. Global patterns in human consumption of net primary production. Nature 2004, 429, 870–873. [Google Scholar] [CrossRef] [PubMed]
- Six, J.; Callewaert, P.; Lenders, S.; De Gryze, S.; Morris, S.; Gregorich, E.; Paul, E.; Paustian, K. Measuring and understanding carbon storage in afforested soils by physical fractionation. Soil Sci. Soc. Am. J. 2002, 66, 1981–1987. [Google Scholar] [CrossRef]
- Makkonen, K.; Helmisaari, H.S. Fine root biomass and production in Scots pine stands in relation to stand age. Tree Physiol. 2001, 21, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Mu, X.; Yuan, Z.; Deng, Q.; Chen, Y.; Yuan, L.Y.; Ryan, L.T.; Kallenbach, R.L. Soil nutrients and water affect the age-related fine root biomass but not production in two plantation forests on the Loess Plateau, China. J. Arid Environ. 2016, 135, 173–180. [Google Scholar] [CrossRef]
- Deng, Q.; Cheng, X.; Hui, D.; Zhang, Q.; Li, M.; Zhang, Q. Soil microbial community and its interaction with soil carbon and nitrogen dynamics following afforestation in central China. Sci. Total Environ. 2016, 541, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Zhang, M.; Xiao, R.; Cui, Y.; Yu, F. Changes in soil microbial biomass and community composition in coastal wetlands affected by restoration projects in a Chinese delta. Geoderma 2017, 289, 124–134. [Google Scholar] [CrossRef]
- Gupta, V.V.S.R.; Germida, J.J. Distribution of microbial biomass and its activity in different soil aggregate size classes as affected by cultivation. Soil Biol. Biochem. 1988, 20, 777–786. [Google Scholar] [CrossRef]
- Mvan, G.; Merckx, R.; Vlassak, K. Spatial distribution of microbial biomass in microaggregates of a silty-loam soil and the relation with the resistance of microorganisms to soil drying. Soil Biol. Biochem. 1996, 28, 503–510. [Google Scholar]
- Handayani, I.P.; Coyne, M.S.; Tokosh, R.S. Soil organic matter fractions and aggregate distribution in response to tall fescue stands. Int. J. Soil Sci. 2010, 5, 1–10. [Google Scholar] [CrossRef]
- Manna, M.C.; Swarup, A.; Wanjari, R.H.; Singh, Y.V.; Ghosh, P.K.; Singh, K.N.; Tripathi, A.K.; Saha, M.N. Soil organic matter in a West Bengal Inceptisol after 30 years of multiple cropping and fertilization. Soil Sci. Soc. Am. J. 2006, 70, 121. [Google Scholar] [CrossRef]
- Six, J.; Paustian, K.; Elliott, E.T.; Combrink, C. Soil structure and organic matter. Soil Sci. Soc. Am. J. 2000, 64, 681–689. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, N.; Ge, T.; Kuzyakov, Y.; Wang, Z.-L.; Li, Z.; Tang, Z.; Chen, Y.; Wu, C.; Lou, Y. Soil aggregation regulates distributions of carbon, microbial community and enzyme activities after 23-year manure amendment. Appl. Soil Ecol. 2017, 111, 65–72. [Google Scholar]
- Chen, X.; Li, Z.; Liu, M.; Jiang, C.; Che, Y. Microbial community and functional diversity associated with different aggregate fractions of a paddy soil fertilized with organic manure and/or NPK fertilizer for 20 years. J. Soils Sediment 2015, 15, 292–301. [Google Scholar] [CrossRef]
- Piao, H.C.; Liu, G.S.; Wu, Y.Y.; Xu, W.B. Relationships of soil microbial biomass carbon and organic carbon with environmental parameters in mountainous soils of southwest China. Biol. Fertil. Soil 2001, 33, 347–350. [Google Scholar] [CrossRef]
| Age of Plantation (Years) | 5 | 9 | 16 |
|---|---|---|---|
| Height (m) | 18.1 ± 0.8 | 21.3 ± 1.7 | 23.6 ± 0.4 |
| Diameter at breast height (cm) | 16.2 ± 0.5 | 23.1 ± 1.2 | 28.6 ± 1.7 |
| Canopy closure (%) | 43.6 ± 4.8 | 53.6 ± 4.8 | 75.0 ± 4.1 |
| Understory biomass (g/m2) | 101.0 ± 3.9 | 97.8 ± 2.3 | 98.7 ± 3.5 |
| pH | 8.34 ± 0.15 | 8.25 ± 0.17 | 8.31 ± 0.13 |
| C/N | 13.2 ± 0.16 | 13.5 ± 0.23 | 17.3 ± 0.20 |
| Sand (g/kg) | 719.7 ± 17.2 | 719.0 ± 17.5 | 722.3 ± 23.5 |
| Silt (g/kg) | 162.0 ± 12.8 | 153.0 ± 4.5 | 161.3 ± 6.3 |
| Clay (g/kg) | 130.2 ± 0.8 | 119.8 ± 2.2 | 116.5 ± 1.7 |
| Stand desity (stems/ha) | 625 | 313 | 305 |
| Year of land reclamation | 1997 | 1993 | 1986 |
| Year of plantation establishment | 2007 | 2003 | 1996 |
| Attribute | Sum of Squares | df | f | p |
|---|---|---|---|---|
| Small aggregates | 1329.8 | 2, 6 | 17.61 | <0.001 |
| Large aggregates | 374.4 | 2, 6 | 19.37 | <0.001 |
| Total aggregates | 2962.2 | 2, 6 | 23.29 | <0.001 |
| Mean weight diameter | 1.7 | 2, 6 | 23.92 | <0.001 |
| Organic C content in bulk soil | 21.6 | 2, 6 | 26.12 | 0.001 |
| Organic C content in fine aggregates | 191.5 | 2, 6 | 2.00 | 0.216 |
| Organic C content in large aggregates | 841.6 | 2, 6 | 2.02 | 0.213 |
| Attribute | n | Age | Date | Age × Date |
|---|---|---|---|---|
| Litterfall | 12 | 0.241 | ||
| Fine-root biomass | 36 | <0.001 | <0.001 | 0.274 |
| SMBC | 36 | <0.001 | <0.001 | <0.001 |
| Predictor | Estimate | Scaled Estimate | Std. Error (Scaled) | t Value (Scaled) | Pr (>|t|) |
|---|---|---|---|---|---|
| Weighted percentage of fine aggregates | |||||
| (Intercept) | 0.263 | ||||
| Root | 3.628 | 2.582 | 2.806 | 0.920 | 0.357 |
| SMBC | 29.631 | 14.485 | 2.820 | 5.136 | <0.001 |
| Litterfall | −1.700 | −0.883 | 2.081 | 0.424 | 0.671 |
| Weighted percentage of large aggregates | |||||
| (Intercept) | 7.613 | ||||
| Root | 5.088 | 3.622 | 1.753 | 2.066 | 0.039 |
| SMBC | 8.269 | 4.042 | 1.753 | 2.306 | 0.021 |
| Litterfall | −2.121 | −1.102 | 1.765 | 0.624 | 0.532 |
| Organic C content in bulk soil | |||||
| (Intercept) | 13.047 | ||||
| Root | 5.319 | 3.786 | 0.797 | 4.751 | <0.001 |
| SMBC | 0.559 | 0.273 | 0.800 | 0.341 | 0.733 |
| Litterfall | −1.741 | −0.905 | 0.638 | 1.419 | 0.156 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ge, Z.; Fang, S.; Chen, H.Y.H.; Zhu, R.; Peng, S.; Ruan, H. Soil Aggregation and Organic Carbon Dynamics in Poplar Plantations. Forests 2018, 9, 508. https://doi.org/10.3390/f9090508
Ge Z, Fang S, Chen HYH, Zhu R, Peng S, Ruan H. Soil Aggregation and Organic Carbon Dynamics in Poplar Plantations. Forests. 2018; 9(9):508. https://doi.org/10.3390/f9090508
Chicago/Turabian StyleGe, Zhiwei, Shuiyuan Fang, Han Y.H. Chen, Rongwei Zhu, Sili Peng, and Honghua Ruan. 2018. "Soil Aggregation and Organic Carbon Dynamics in Poplar Plantations" Forests 9, no. 9: 508. https://doi.org/10.3390/f9090508
APA StyleGe, Z., Fang, S., Chen, H. Y. H., Zhu, R., Peng, S., & Ruan, H. (2018). Soil Aggregation and Organic Carbon Dynamics in Poplar Plantations. Forests, 9(9), 508. https://doi.org/10.3390/f9090508





