Wood and Fiber Quality of Plantation-Grown Conifers: A Summary of Research with an Emphasis on Loblolly and Radiata Pine
Abstract
:1. Introduction
- X-ray densitometry (density);
- X-ray diffraction (MFA);
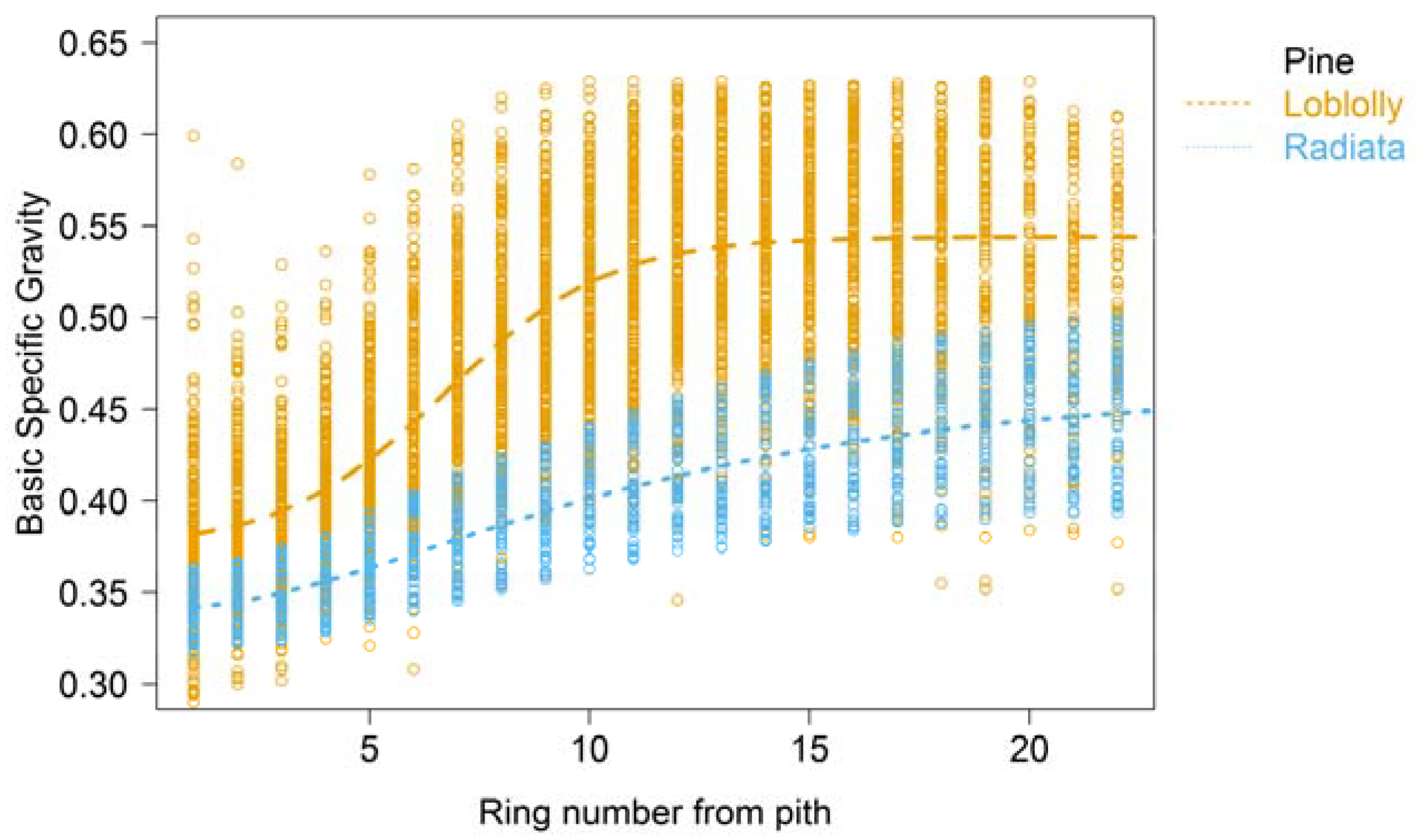
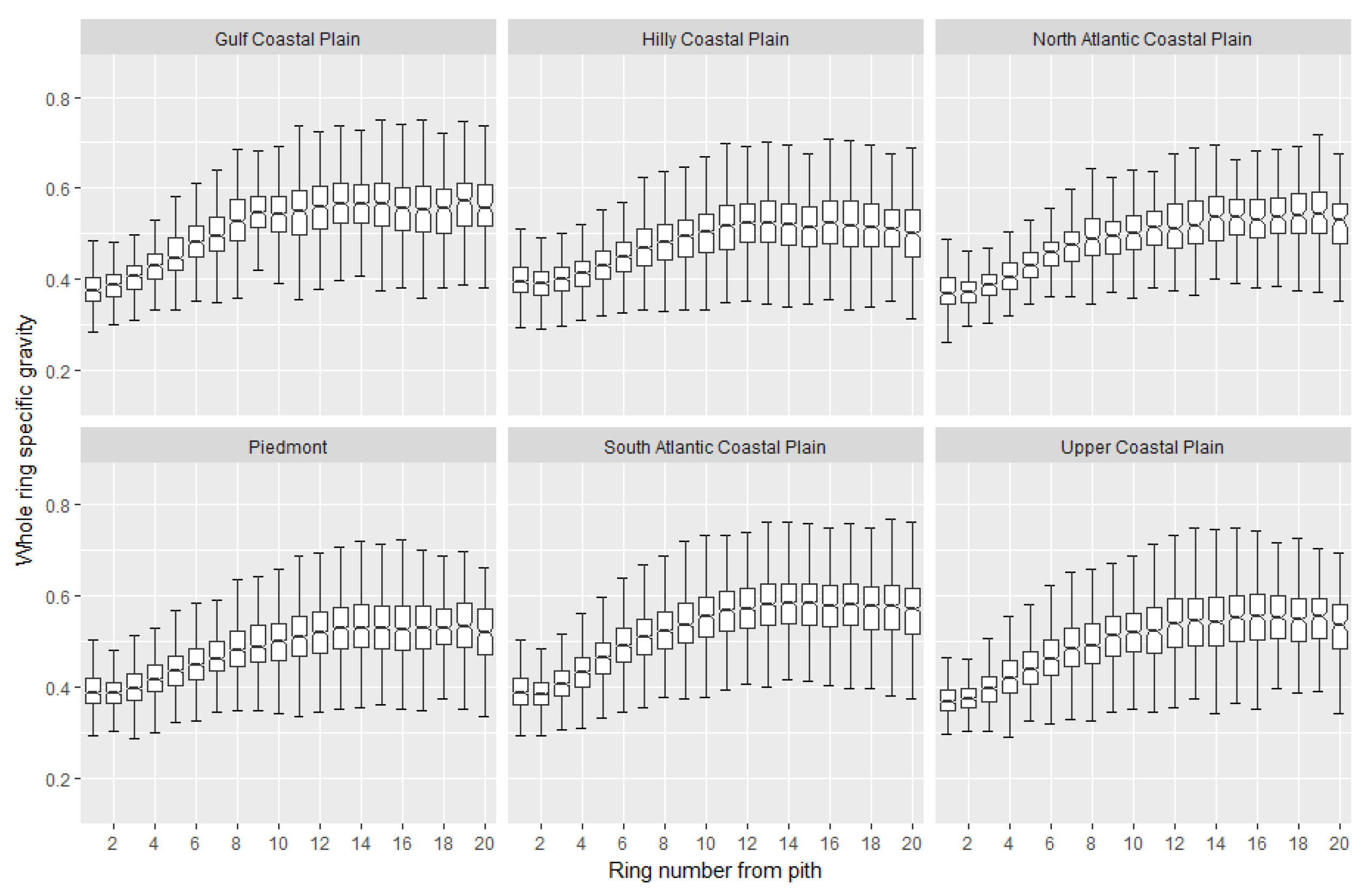
2. Wood Property Variation
3. Silviculture and Wood Quality
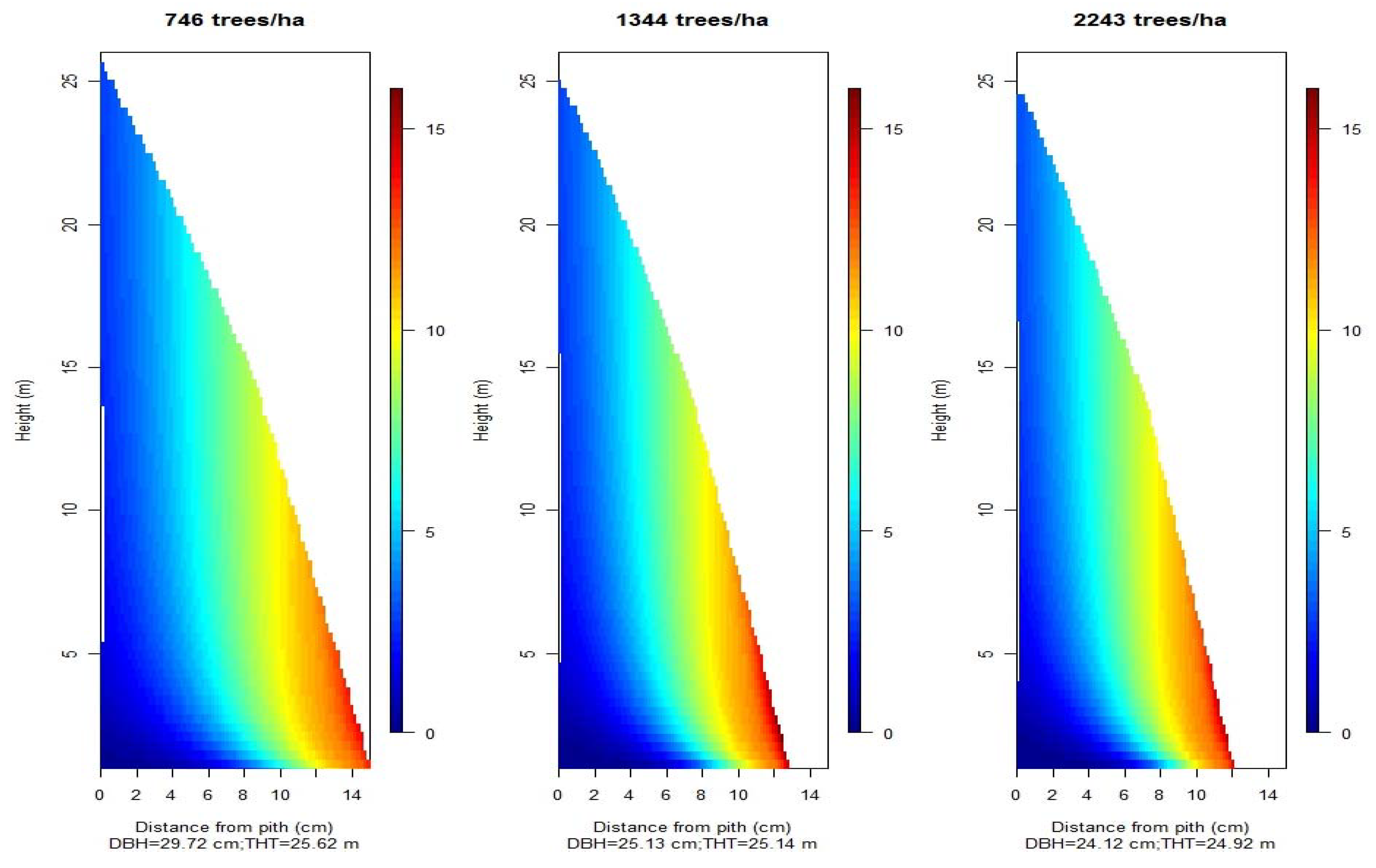
3.1. Planting Density
3.2. Fertilization and Competition Control upon Planting
3.3. Mid-Rotation Thinning and Fertilization
3.4. Irrigation
4. Wood Quality and Durability
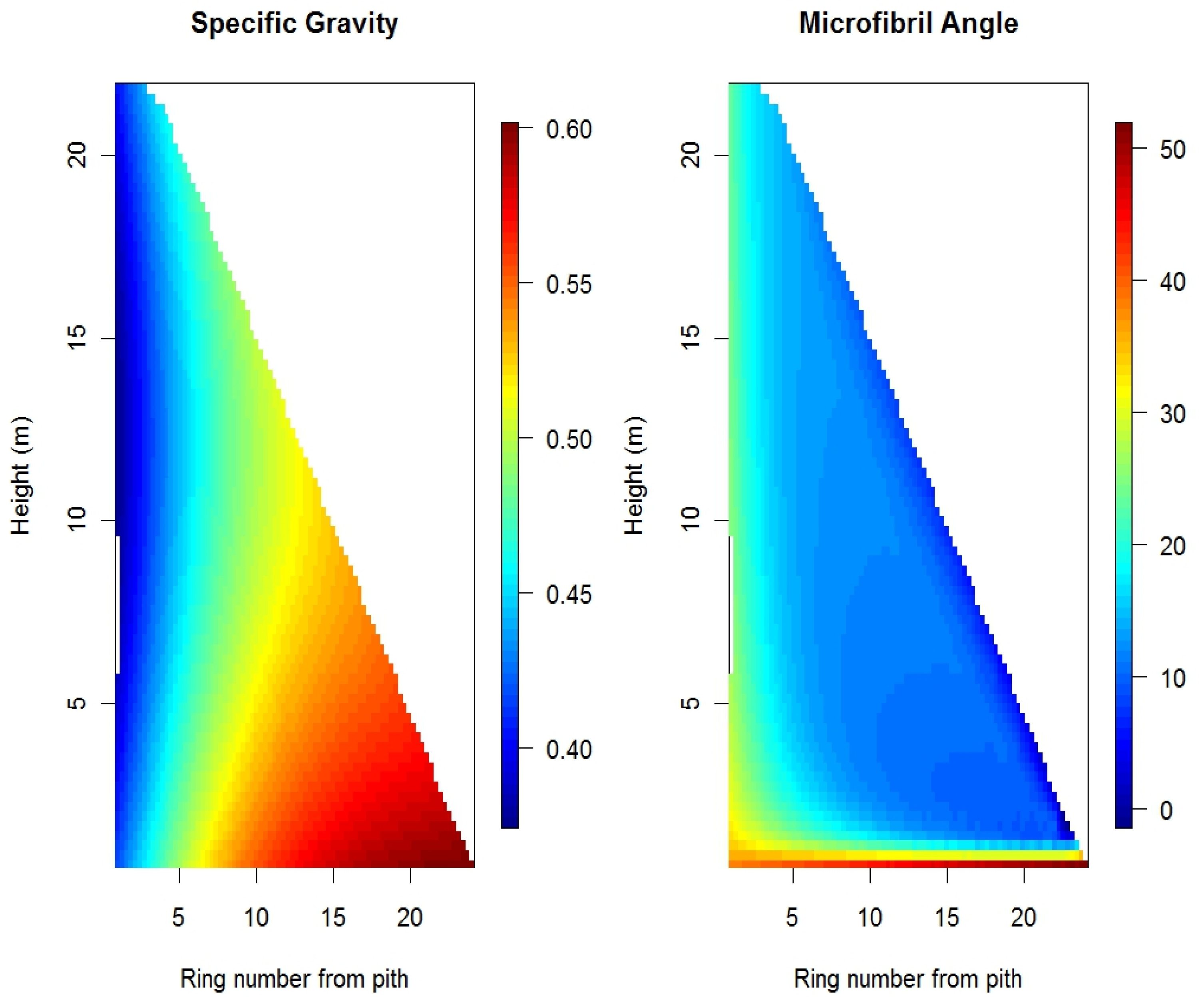
5. Mathematical Models for Explaining Variation in Wood Properties and Wood Quality
6. Mathematical Models Predicting Silvicultural Responses in Wood Properties
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Payn, T.; Carnus, J.-M.; Freer-Smith, P.; Kimberley, M.; Kollert, W.; Liu, S.; Orazio, C.; Rodriguez, L.; Silva, L.N.; Wingfield, M.J. Changes in planted forests and future global implications. For. Ecol. Manag. 2015, 352, 57–67. [Google Scholar] [CrossRef]
- Portin, A.; Lehtonen, P. Strategic Review of the Future of Forest Plantations; Report A12–86069 Prepared for the Forest Stewardship Council; Indufor: Helsinki, Finland, 2012; p. 111. [Google Scholar]
- McKeand, S.; Mullin, T.; White, T. Deployment of genetically improved loblolly and slash pines in the south. J. For. 2003, 101, 32–37. [Google Scholar]
- Aspinwall, M.J.; McKeand, S.E.; King, J.S. Carbon sequestration from 40 years of planting genetically improved loblolly pine across the southeast United States. For. Sci. 2012, 58, 446–546. [Google Scholar] [CrossRef]
- Fox, T.R.; Jokela, E.J.; Allen, H.L. The development of pine plantation silviculture in the southern United States. J. For. 2007, 105, 337–347. [Google Scholar]
- Shula, R.G. The upper limits of stem-volume production in radiata pine in New Zealand. N. Zeal. For. 1989, 34, 19–22. [Google Scholar]
- Kimberley, M.O.; Moore, J.R.; Dungey, H.S. Quantification of realised genetic gain in radiata pine and its incorporation into growth and yield modelling systems. Can. J. For. Res. 2015, 45, 1676–1687. [Google Scholar] [CrossRef]
- Moore, J.; Clinton, P. Enhancing the productivity of radiata pine forestry within environmental limits. N. Zeal. J. For. 2015, 60, 35–41. [Google Scholar]
- Moore, J.R.; Cown, D.J. Corewood (Juvenile Wood) and Its Impact on Wood Utilisation. Curr. For. Rep. 2017, 3, 107–118. [Google Scholar] [CrossRef]
- Briggs, D.G.; Smith, W.R. Effects of silvicultural pratices on wood properties of conifers: A review. In Douglas-fir Stand Management for the Future; Oliver, C., Hanley, D., Johnson, J., Eds.; University of Washington Press: Seattle, WA, USA, 1986; pp. 108–116. [Google Scholar]
- Panshin, A.J.; de Zeeuw, C. Textbook of Wood Technology, 4th ed.; McGraw-Hill: New York, NY, USA, 1980; p. 772. [Google Scholar]
- Smook, G.A. Handbook for Pulp and Paper Technologists, 3rd ed.; Angus Wilde Publications Inc.: Vancouver, BC, Canada, 2002. [Google Scholar]
- Saranpää, P. Wood Density and Growth. In Wood Quality and Its Biological Basis; Barnett, J.R., Jeronimidis, G., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2003; pp. 87–117. [Google Scholar]
- Meylan, B.A. The influence of microfibril angle on the longitudinal shrinkage-moisture content relationship. Wood Sci. Technol. 1972, 6, 293–301. [Google Scholar] [CrossRef]
- Meylan, B.A. Cause of high longitudinal shrinkage in wood. For. Prod. J. 1968, 18, 75–78. [Google Scholar]
- Cave, I.D.; Walker, J.C.F. Stiffness of wood in fast-grown plantation softwoods: The influence of microfibril angle. For. Prod. J. 1994, 44, 42–48. [Google Scholar]
- Evans, R.; Ilic, J. Rapid prediction of wood stiffness from microfibril angle and density. For. Prod. J. 2001, 51, 53–57. [Google Scholar]
- Jones, T.G.; Emms, G.W. Influence of acoustic velocity, density, and knots on the stiffness grade outturn of radiata pine logs. Wood Fiber Sci. 2010, 42, 1–9. [Google Scholar]
- Olsson, A.; Oscarsson, J.; Johansson, M.; Källsner, B. Prediction of timber bending strength on basis of bending stiffness and material homogeneity assessed from dynamic excitation. Wood Sci. Technol. 2012, 46, 667–683. [Google Scholar] [CrossRef]
- Olsson, A.; Oscarsson, J.; Serrano, E.; Källsner, B.; Johansson, M.; Enquist, B. Prediction of timber bending strength and in-member cross-sectional stiffness variation on the basis of local wood fibre orientation. Eur. J. Wood Wood Prod. 2013, 71, 319–333. [Google Scholar] [CrossRef]
- Da Silva Perez, D.; Fauchon, T. Wood quality for pulp and paper. In Wood Quality and Its Biological Basis; Barnett, J.R., Jeronimidis, G., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2003; pp. 157–186. [Google Scholar]
- Pereira, H.; Graca, J.; Rodrigues, J.C. Wood chemistry in relation to quality. In Wood Quality and It’s Biological Basis; Barnett, J.R., Jeronimidis, G., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2003; pp. 53–86. [Google Scholar]
- Evans, R. A variance approach to the X-ray diffractometric estimation of microfibril angle in wood. Appita J. 1999, 52, 283–289. [Google Scholar]
- Evans, R. Rapid measurement of the transverse dimensions of tracheids in radial wood sections from Pinus radiata. Holzforschung 1994, 48, 168–172. [Google Scholar] [CrossRef]
- Evans, R. Wood stiffness by X-ray diffractometry. In Characterization of the Cellulosic Cell Wall; Stokke, D.D., Groom, L.H., Eds.; Blackwell Publishing: Hoboken, NJ, USA, 2006; pp. 138–146. [Google Scholar]
- So, C.-L.; Via, B.K.; Groom, L.H.; Schimleck, L.R.; Shupe, T.F.; Kelley, S.S.; Rials, T.G. Near infrared (NIR) spectroscopy in the forest products industry. For. Prod. J. 2004, 54, 6–16. [Google Scholar]
- Tsuchikawa, S. A review of recent near infrared research for wood and paper. Appl. Spectrosc. Rev. 2007, 42, 43–71. [Google Scholar] [CrossRef]
- Wang, X.; Ross, R.J.; Erickson, J.R.; Ligon, J.B. Nondestructive evaluation of trees. Exp. Tech. 2000, 24, 27–29. [Google Scholar] [CrossRef]
- Huang, C.-L.; Lindström, H.; Nakada, R.; Ralston, J. Cell wall structure and wood properties determined by acoustics—A selective review. Holz Als Roh-und Werkstoff 2003, 61, 321–335. [Google Scholar] [CrossRef]
- Bocur, V. Acoustics of Wood. In Springer Series in Wood Science; Springer: Berlin, Germany, 2006; p. 394. [Google Scholar]
- Gao, S.; Wang, X.; Wiemann, M.C.; Brashaw, B.K.; Ross, R.J.; Wang, L. A critical analysis of methods for rapid and nondestructive determination of wood density in standing trees. Ann. For. Sci. 2017, 74, 27. [Google Scholar] [CrossRef]
- Cown, D.J. Comparison of the Pilodyn and Torsiometer methods for the rapid assessment of wood density in living trees. N. Zeal. J. For. Sci. 1978, 8, 384–391. [Google Scholar]
- Watt, M.S.; Garnett, B.T.; Walker, J.C.F. The use of the pilodyn for assessing outerwood density in New Zealand radiata pine. For. Prod. J. 1996, 46, 101–106. [Google Scholar]
- Isik, F.; Li, B. Rapid assessment of wood density of live trees using the Resistograph for selection in tree improvement programs. Can. J. For. Res. 2003, 33, 2426–2435. [Google Scholar] [CrossRef]
- Jordan, L.; Clark, A.; Schimleck, L.R.; Hall, D.B.; Daniels, R.F. Regional variation in wood specific gravity of planted loblolly pine in the United States. Can. J. For. Res. 2008, 38, 698–710. [Google Scholar] [CrossRef]
- Kimberley, M.O.; Cown, D.J.; McKinley, R.B.; Moore, J.R.; Dowling, L.J. Modelling variation in wood density within and among trees in stands of New Zealand-grown radiata pine. N. Zeal. J. For. Sci. 2015, 45, 22. [Google Scholar] [CrossRef]
- Antony, F.; Jordan, L.; Schimleck, L.R.; Clark, A., III; Souter, R.A.; Daniels, R.F. Regional variation in wood modulus of elasticity (stiffness) and modulus of rupture (strength) of planted loblolly pine in the United States. Can. J. For. Res. 2011, 41, 1522–1533. [Google Scholar] [CrossRef]
- Jordan, L.; Daniels, R.F.; He, R. Multilevel nonlinear mixed-effects models for the modeling of earlywood and latewood microfibril angle. For. Sci. 2005, 51, 357–371. [Google Scholar]
- Palmer, D.J.; Kimberley, M.O.; Cown, D.J.; McKinley, R.B. Assessing prediction accuracy in a regression kriging surface of Pinus radiata outerwood density across New Zealand. For. Ecol. Manag. 2013, 308, 9–16. [Google Scholar] [CrossRef]
- Tian, X.; Cown, D.J.; McConchie, D.L. Modelling of radiata pine wood properties. Part 2: Wood density. N. Zeal. J. For. Sci. 1995, 25, 214–230. [Google Scholar]
- Watt, M.S.; Kimberley, M.O.; Harrington, J.J.; Riddell, M.J.C.; Cown, D.J.; Moore, J.R. Differences in intra-tree variation in spiral grain angle for radiata pine. N. Zeal. J. For. Sci. 2013, 43, 12. [Google Scholar] [CrossRef]
- Moore, J.R.; Cown, D.J.; McKinley, R.B. Modelling microfibril angle variation in New Zealand-grown radiata pine. N. Zeal. J. For. Sci. 2014, 44, 25. [Google Scholar] [CrossRef]
- Moore, J.R.; Cown, D.J.; McKinley, R.B. Modelling spiral grain angle variation in New Zealand-grown radiata pine. N. Zeal. J. For. Sci. 2015, 45, 15. [Google Scholar] [CrossRef]
- Walker, J. Wood quality: A perspective from New Zealand. Forests 2013, 4, 234–250. [Google Scholar] [CrossRef]
- Burdon, R.D.; Kibblewhite, R.P.; Walker, J.C.F.; Megraw, R.A.; Evans, R.; Cown, D.J. Juvenile versus mature wood: A new concept, orthogonal to corewood versus outerwood, with special reference to Pinus radiata and P. Taeda. For. Sci. 2004, 50, 399–415. [Google Scholar]
- Zobel, B.J.; Jett, J.B. Genetics of Wood Production; Springer: Berlin, Germany, 1995; p. 352. [Google Scholar]
- Kimberley, M.O.; Moore, J.R.; Dungey, H.S. Modelling the effects of genetic improvement on radiata pine wood density. N. Zeal. J. For. Sci. 2016, 46, 8. [Google Scholar] [CrossRef]
- Burdon, R.D.; Li, Y.; Suontama, M.; Dungey, H.S. Genotype × site × silviculture interactions in radiata pine: Knowledge, working hypotheses and pointers for research. N. Zeal. J. For. Sci. 2017, 47, 6. [Google Scholar] [CrossRef]
- Drew, D.M.; Downes, G.M.; Grady, A.P.O.; Read, J.; Worledge, D. High resolution temporal variation in wood properties in irrigated and nonirrigated Eucalyptus globulus. Ann. For. Sci. 2009, 66, 406. [Google Scholar] [CrossRef]
- Fox, T.R. Sustained productivity in intensively managed forest plantations. For. Ecol. Manag. 2000, 138, 187–202. [Google Scholar] [CrossRef]
- Baldwin, V.C.; Peterson, K.D.; Clark, A., III; Ferguson, R.B.; Strub, M.R.; Bower, D.R. The effects of spacing and thinning on stand and tree characteristics of 38-year-old loblolly pine. For. Ecol. Manag. 2000, 137, 91–102. [Google Scholar] [CrossRef]
- Sharma, M.; Burkhart, H.E.; Amateis, R.L. Modeling the effect of density on the growth of loblolly pine trees. South. J. Appl. For. 2002, 26, 124–133. [Google Scholar]
- Clark, A., III; Saucier, J.R.; Baldwin, V.C.; Bower, D.R. Influence of initial planting density, geographic location, and species on juvenile wood formation in southern pine. For. Prod. J. 1989, 39, 42–48. [Google Scholar]
- Clark, A., III; Saucier, J.R.; Baldwin, V.C.; Bower, D.R. Effect of initial spacing and thinning on lumber grade, yield, and strength of loblolly pine. For. Prod. J. 1994, 44, 14–20. [Google Scholar]
- Antony, F.; Schimleck, L.R.; Jordan, L.; Daniels, R.F.; Clark, A., III. Modeling the effect of initial planting density on within tree variation of stiffness in loblolly pine. Ann. For. Sci. 2012, 69, 641–650. [Google Scholar] [CrossRef]
- Clark, A., III; McAlister, R.H. Visual tree grading systems for estimating lumber yields in young and mature southern pine. For. Prod. J. 1998, 48, 59–67. [Google Scholar]
- Clark, A., III; Jordan, L.; Schimleck, L.; Daniels, R.F. Effect of initial planting spacing on wood properties of unthinned loblolly pine at age 21. For. Prod. J. 2008, 58, 78–83. [Google Scholar]
- Moore, J.R.; Dash, J.P.; Lee, J.R.; McKinley, R.B.; Dungey, H.S. Quantifying the influence of seedlot and stand density on growth, wood properties and the economics of growing radiata pine. Forestry 2017, 91, 327–340. [Google Scholar] [CrossRef]
- Butler, M.A.; Dahlen, J.; Daniels, R.F.; Eberhardt, T.L.; Antony, F. Bending strength and stiffness of loblolly pine lumber from intensively managed stands located on the Georgia Lower Coastal Plain. Eur. J. Wood Wood Prod. 2016, 74, 91–100. [Google Scholar] [CrossRef]
- Kretschmann, D.E.; Bendtsen, B.A. Ultimate tensile strength and modulus of elasticity of fast-grown plantation loblolly pine lumber. Wood Fiber Sci. 1992, 24, 189–203. [Google Scholar]
- Macdonald, E.; Hubert, J. A review of the effects of silviculture on timber quality of Sitka spruce. Forestry 2002, 75, 107–137. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Chauret, G.; Ren, H.Q.; Desjardins, R. Impact of initial spacing on plantation black spruce lumber grade yield, bending properties, and MSR yield. Wood Fiber Sci. 2002, 34, 460–475. [Google Scholar]
- Moore, J.; Achim, A.; Lyon, A.; Mochan, S.; Gardiner, B. Effects of early re-spacing on the physical and mechanical properties of Sitka spruce structural timber. For. Ecol. Manag. 2009, 258, 1174–1180. [Google Scholar] [CrossRef]
- Waghorn, M.J.; Watt, M.S.; Mason, E.G. Influence of tree morphology, genetics, and initial stand density on outerwood modulus of elasticity of 17-year-old Pinus radiata. For. Ecol. Manag. 2007, 244, 86–92. [Google Scholar] [CrossRef]
- Lasserre, J.-P.; Mason, E.G.; Watt, M.S.; Moore, J.R. Influence of initial planting spacing and genotype on microfibril angle, wood density, fibre properties and modulus of elasticity in Pinus radiata D. Don corewood. For. Ecol. Manag. 2009, 258, 1924–1931. [Google Scholar] [CrossRef]
- Lasserre, J.-P.; Mason, E.G.; Watt, M.S. The effects of genotype and spacing on Pinus radiata [D. Don] corewood stiffness in an 11-year old experiment. For. Ecol. Manag. 2005, 205, 375–383. [Google Scholar] [CrossRef]
- Soto, L.; Valenzuela, L.; Lasserre, J.P. Effect of initial planting density in dynamic modulus of elasticity in standing trees and logs of 28 years old radiata pine plantation in sandy soil, Chile. Maderas. Ciencia y Tecnologia 2012, 14, 209–220. [Google Scholar] [CrossRef]
- Watt, M.S.; Zoric, B.; Kimberley, M.O.; Harrington, J. Influence of stocking on radial and longitudinal variation in modulus of elasticity, microfibril angle, and density in a 24-year-old. Pinus radiata thinning trial. Can. J. For. Res. 2011, 41, 1422–1431. [Google Scholar]
- Watt, M.S.; Zoric, B. Development of a model describing modulus of elasticity across environmental and stand density gradients in plantation-grown Pinus radiata within New Zealand. Can. J. For. Res. 2010, 40, 1558–1566. [Google Scholar] [CrossRef]
- Amateis, R.L.; Burkhart, H.E.; Jeong, G.Y. Modulus of elasticity declines with decreasing planting density for loblolly pine (Pinus taeda) plantations. Ann. For. Sci. 2013, 70, 743–750. [Google Scholar] [CrossRef]
- Clark, A.; Daniels, R.F.; Miller, J.H. Effect of controlling herbaceous and woody competing vegetation on wood quality of planted loblolly pine. For. Prod. J. 2006, 56, 40–46. [Google Scholar]
- Antony, F.; Schimleck, L.R.; Jordan, L.; Clark, A.; Daniels, R.F. Effect of early age woody and herbaceous competition control on wood properties of loblolly pine. For. Ecol. Manag. 2011, 262, 1639–1647. [Google Scholar] [CrossRef]
- Mora, C.R. Effects of Early Intensive Silviculture on Wood Properties of Loblolly Pine; North Carolina State University: Raleigh, NC, USA, 2003. [Google Scholar]
- Mora, C.R.; Allen, H.L.; Daniels, R.F.; Clark, A. Modeling corewood–outerwood transition in loblolly pine using wood specific gravity. Can. J. For. Res. 2007, 37, 999–1011. [Google Scholar] [CrossRef]
- Clark, A.; Borders, B.E.; Daniels, R.F. Impact of vegetation control and annual fertilization on properties of loblolly pine wood at age 12. For. Prod. J. 2004, 54, 90–96. [Google Scholar]
- Watt, M.S.; Downes, G.M.; Whitehead, D.; Mason, E.G.; Richardson, B.; Grace, J.C.; Moore, J.R. Wood properties of juvenile Pinus radiata growing in the presence and absence of competing understorey vegetation at a dryland site. Trees Struct. Funct. 2005, 19, 580–586. [Google Scholar] [CrossRef]
- Mason, E.G. Interactions between influences of genotype and grass competition on growth and wood stiffness of juvenile radiata pine in a summer-dry environment. Can. J. For. Res. 2006, 36, 2454–2463. [Google Scholar] [CrossRef]
- Watt, M.S.; Clinton, P.C.; Parfitt, R.L.; Ross, C.; Coker, G. Modelling the influence of site and weed competition on juvenile modulus of elasticity in Pinus radiata across broad environmental gradients. For. Ecol. Manag. 2009, 258, 1479–1488. [Google Scholar] [CrossRef]
- Antony, F.; Jordan, L.; Daniels, R.F.; Schimleck, L.R.; Clark, A.; HalL, D.B. Effect of midrotation fertilization on growth and specific gravity of loblolly pine. Can. J. For. Res. 2009, 39, 928–935. [Google Scholar] [CrossRef]
- Antony, F.; Jordan, L.; Schimleck, L.R.; Daniels, R.F.; Clark, A. The effect of mid-rotation fertilization on the wood properties of loblolly pine (Pinus taeda). IAWA J. 2009, 30, 49–58. [Google Scholar] [CrossRef]
- Nyakuengama, J.G.; Downes, G.M.; Ng, J. Growth and wood density responses to later-age fertilizer applications in Pinus radiata. IAWA J. 2002, 23, 431–448. [Google Scholar]
- Love-Myers, K.R.; Clark, A.; Schimleck, L.R.; Jokela, E.J.; Daniels, R.F. Specific gravity responses of slash and loblolly pine following mid-rotation fertilization. For. Ecol. Manag. 2009, 257, 2342–2349. [Google Scholar] [CrossRef]
- Cown, D.J.; McConchie, D.L. Effects of thinning and fertiliser application on wood properties of Pinus radiata. N. Zeal. J. For. Sci. 1981, 11, 79–91. [Google Scholar]
- Beets, P.N.; Gilchrist, K.; Jeffreys, M.P. Wood density of radiata pine: Effect of nitrogen supply. For. Ecol. Manag. 2001, 145, 173–180. [Google Scholar] [CrossRef]
- Beets, P.N.; Kimberley, M.O.; McKinley, R.B. Predicting wood density of Pinus radiata annual growth increments. N. Zeal. J. For. Sci. 2007, 37, 241–266. [Google Scholar]
- Antony, F.; Schimleck, L.R.; Daniels, R.F. Effect of mid-rotation fertilization on stiffness and strength of loblolly pine wood. IAWA J. 2013, 34, 127–134. [Google Scholar] [CrossRef]
- Antony, F.; Schimleck, L.R.; Daniels, R.F.; Clark, A. Effect of fertilization on growth and wood properties of thinned and unthinned midrotation loblolly pine (Pinus taeda L.) stands. South. J. Appl. For. 2011, 35, 142–147. [Google Scholar]
- Wang, H.; Kimberley, M.O.; Magesan, G.N.; McKinley, R.B.; Lee, J.R.; Lavery, J.M.; Hodgkiss, P.D.F.; Payn, T.W.; Wilks, P.J.; Fisher, C.R.; et al. Midrotation effects of biosolids application on tree growth and wood properties in a Pinus radiata plantation. Can. J. For. Res. 2006, 36, 1921–1930. [Google Scholar] [CrossRef]
- Nyakuengama, J.G.; Downes, G.M.; Ng, J. Changes caused by mid-rotation fertilizer application to the fibre anatomy of Pinus radiata. IAWA J. 2003, 24, 397–409. [Google Scholar] [CrossRef]
- Love-Myers, K.R.; Clark, A.; Schimleck, L.R.; Dougherty, P.M.; Daniels, R.F. The effects of irrigation and fertilization on specific gravity of loblolly pine. For. Sci. 2010, 56, 484–493. [Google Scholar]
- Gonzalez-Benecke, C.A.; Martin, T.A.; Peter, G.F. Water availability and genetic effects on wood properties of loblolly pine (Pinus taeda). Can. J. For. Res. 2010, 40, 2265–2277. [Google Scholar] [CrossRef]
- Nicholls, J.W.P.; Waring, H.D. The effect of environmental factors on wood characteristics IV. Irrigation and partial droughting of Pinus radiata. Silvae Genet. 1977, 26, 107–111. [Google Scholar]
- Scheffer, T.C.; Morrell, J.J. Natural durability of wood: A worldwide checklist of species. In Forest Research Laboratory Research Contribution 22; College of Forestry, Oregon State University: Corvallis, OR, USA, 1998. [Google Scholar]
- Taylor, A.M.; Gartner, B.; Morrell, J.J. Heartwood formation and natural durability–a review. Wood Fiber Sci. 2002, 34, 587–611. [Google Scholar]
- Sellin, A. Sapwood amount in Picea abies (L.) Karst. determined by tree age and radial growth rate. Holzforschung 1996, 50, 291–296. [Google Scholar] [CrossRef]
- DeBell, D.S.; Morrell, J.J.; Gartner, B.L. Within-stem variation in tropolone content and decay resistance of second-growth western redcedar. For. Sci. 1999, 45, 101–107. [Google Scholar]
- Clark, J.W.; Scheffer, T.C. Natural decay resistance of the heartwod of coast redwood Sequoia sempervirens (D.Don) Endl. For. Prod. J. 1983, 33, 15–20. [Google Scholar]
- Ajuong, E.; Freitag, C.; Morrell, J.J. Decay resistance and extractive content of second-growth Port Orford cedar (Chamaecyparis lawsoniana) wood. Wood Fiber Sci. 2014, 46, 502–509. [Google Scholar]
- Taylor, A.M.; Gartner, B.L.; Morrell, J.J. Co-incident variations in growth rate and heartwood extractive concentration in Douglas-fir. For. Ecol. Manag. 2003, 186, 257–260. [Google Scholar] [CrossRef]
- Taylor, A.M.; Gartner, B.L.; Morrell, J.J. Western redcedar extractives: Is there a role for the silviculturist? For. Prod. J. 2006, 56, 58–63. [Google Scholar]
- Paul, B.H. Variability in wood of southern pines as influenced by silvicultural practices. In Forest Products Laboratory Report 1923; United States Department of Agriculture Forest Service: Madison, WI, USA, 1957; p. 9. [Google Scholar]
- Cown, D.J. New Zealand pine and Douglas-fir: Suitability for processing. In FRI Bulletin 216; New Zealand Forest Research Institute: Rotorua, New Zealand, 1999; p. 72. [Google Scholar]
- Haygreen, J.G.; Bowyer, J.L. Forest Products and Wood Science: An Introduction, 3rd ed.; Iowa State University Press: Ames, IA, USA, 1996. [Google Scholar]
- Wahlgren, H.D.; Fassnacht, D.L. Estimating tree specific gravity from a single increment core. In Report 2146; United States Department of Agriculture Forest Service, Forest Products Laboratory: Madison, WI, USA, 1959. [Google Scholar]
- Zobel, B.; Henson, F.; Webb, C. Estimation of certain wood properties of loblolly and slash pine trees from breast height sampling. For. Sci. 1960, 6, 155–162. [Google Scholar]
- Taras, M.A.; Wahlgren, H.D. A comparison of increment core sampling methods for estimating tree specific gravity. In Research Paper SE-7; United States Department of Agriculture Forest Service, Southeastern Forest Experiment Station: Ashville, NC, USA, 1963; p. 16. [Google Scholar]
- Aspinwall, M.J. Relating Breast Height Wood Properties to Whole Stem Wood Properties in Loblolly Pine; North Carolina State University: Raleigh, NC, USA, 2007. [Google Scholar]
- Evans, R.; Kibblewhite, R.P.; Stringer, S. Kraft pulp fibre property prediction from wood properties in elevan radiata pine clones. Appita J. 1997, 50, 25–33. [Google Scholar]
- Cown, D.J.; McConchie, D.L.; Young, G.D. Radiata pine wood properties survey. In FRI Bulletin 50; Revised edition; Ministry of Forestry, Forest Research Institute: Rotorua, New Zealand, 1991; p. 50. [Google Scholar]
- Tassissa, G.; Burkhart, H.E. Modeling thinning effects on ring specific gravity of loblolly pine (Pinus taeda L.). For. Sci. 1998, 44, 212–223. [Google Scholar]
- Daniels, R.F.; He, R.; Clark, A.; Souter, R.A. Modelling wood properties of planted loblolly pine from pith to bark and stump to tip. In Proceedings of the Fourth Workshop-Connection between Forest Resources and Wood Quality: Modeling Approaches and Simulation Software, Harrison Hot Springs, BC, Canada, 8–15 September 2002. [Google Scholar]
- He, R. Mixed Effects Modeling of Wood Properties of Loblolly Pine in the Southeastern United States; University of Georgia: Athens, GA, USA, 2004. [Google Scholar]
- Phillips, K.M. Modeling within Tree Changes in Wood Specific Gravity and Moisture Content for Loblolly Pine in Georgia; University of Georgia: Athens, GA, USA, 2002. [Google Scholar]
- Antony, F.; Schimleck, L.R.; Daniels, R.F.; Clark, A.; Hall, D.B. Modeling the longitudinal variation in wood specific gravity of planted loblolly pine (Pinus taeda) in the United States. Can. J. For. Res. 2010, 40, 2439–2451. [Google Scholar] [CrossRef]
- Jordan, L.; He, R.; Hall, D.B.; Clark, A.; Daniels, R.F. Variation in loblolly pine ring microfibril angle in the southeastern United States. Wood Fiber Sci. 2007, 39, 352–363. [Google Scholar]
- Jordan, L.; Re, R.; Hall, D.B.; Clark, A.; Daniels, R.F. Variation in loblolly pine cross-sectional microfibril angle with tree height and physiographic region. Wood Fiber Sci. 2006, 38, 390–398. [Google Scholar]
- Tian, X.; Cown, D.J.; Lausberg, M.J.F. Modelling of radiata pine wood properties. Part 1: Spiral grain. N. Zeal. J. For. Sci. 1995, 25, 200–213. [Google Scholar]
- Leban, J.M.; Daquitaine, R.; Daquitaine, F.; Saint-André, L. Linking models for tree growth and wood quality in Norway spruce. Part 1: Validations of predictions for sawn properties, ring width, wod density and knottiness. In Proceedings of the Second Workshop Connection between Silviculture and Wood Quality through Modellin Approaches and Simulation Software, Berg-en-Dal, South Africa, 26–31 August 1996; pp. 220–228. [Google Scholar]
- Wilhelmsson, L.; Arlinger, J.; Arlinger, K.; Lundqvist, S.-O.; Lundqvist, T.; Lundqvist, O.; Olsson, L. Models for predicting wood properties in stems of Picea abies and Pinus sylvestris in Sweden. Scand. J. For. Res. 2002, 17, 330–350. [Google Scholar] [CrossRef]
- Ikonen, V.-P.; Peltola, H.; Wilhelmsson, L.; Kilpeläinen, A.; Väisänen, H.; Nuutinen, T.; Kellomäki, S. Modelling the distribution of wood properties along the stems of Scots pine (Pinus sylvestris L.) and Norway spruce (Picea abies (L.) Karst.) as affected by silvicultural management. For. Ecol. Manag. 2008, 256, 1356–1371. [Google Scholar] [CrossRef]
- Auty, D.; Achim, A.; Macdonald, E.; Macdonald, A.D.; Gardiner, B.A. Models for predicting wood density variation in Scots pine. Forestry 2014, 87, 449–458. [Google Scholar] [CrossRef]
- Auty, D.; Gardiner, B.A.; Achim, A.; Moore, J.R.; Cameron, A.D. Models for predicting microfibril angle variation in Scots pine. Ann. For. Sci. 2013, 70, 209–218. [Google Scholar] [CrossRef]
- Ioanna, M. Modelling Spiral Angle in Picea Sitchensis; University of York: York, UK, 2007. [Google Scholar]
- Fonweban, J.; Mavrou, I.; Gardiner, B.; Macdonald, E. Modelling the effect of spacing and site exposure on spiral grain angle on Sitka spruce (Picea sitchensis (Bong.) Carr.) in Northern Britain. Forestry 2013, 86, 331–342. [Google Scholar] [CrossRef]
- Gardiner, B.; Leban, J.M.; Auty, D.; Auty, H. Models for predicting wood density of British-grown Sitka spruce. Forestry 2011, 84, 119–132. [Google Scholar] [CrossRef]
- Antony, F.; Schimleck, L.R.; Hall, D.B.; Clark, A. Modeling the effect of midrotation fertilization on specific gravity of loblolly pine (Pinus taeda L.). For. Sci. 2011, 57, 145–152. [Google Scholar]
- Filipescu, C.N.; Stoehr, M.U.; Pigott, D.R. Variation of lumber properties in genetically improved full-sib families of Douglas-fir in British Columbia, Canada. Forestry 2018, 91, 320–326. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schimleck, L.; Antony, F.; Dahlen, J.; Moore, J. Wood and Fiber Quality of Plantation-Grown Conifers: A Summary of Research with an Emphasis on Loblolly and Radiata Pine. Forests 2018, 9, 298. https://doi.org/10.3390/f9060298
Schimleck L, Antony F, Dahlen J, Moore J. Wood and Fiber Quality of Plantation-Grown Conifers: A Summary of Research with an Emphasis on Loblolly and Radiata Pine. Forests. 2018; 9(6):298. https://doi.org/10.3390/f9060298
Chicago/Turabian StyleSchimleck, Laurence, Finto Antony, Joseph Dahlen, and John Moore. 2018. "Wood and Fiber Quality of Plantation-Grown Conifers: A Summary of Research with an Emphasis on Loblolly and Radiata Pine" Forests 9, no. 6: 298. https://doi.org/10.3390/f9060298
APA StyleSchimleck, L., Antony, F., Dahlen, J., & Moore, J. (2018). Wood and Fiber Quality of Plantation-Grown Conifers: A Summary of Research with an Emphasis on Loblolly and Radiata Pine. Forests, 9(6), 298. https://doi.org/10.3390/f9060298




