Sustainable Management of Metasequoia glyptostroboides Plantation Forests in Shanghai
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Model Selection, Calibration, Evaluation, and Sensitivity Analysis
2.3. Management Simulations
3. Results
3.1. Model Evaluation
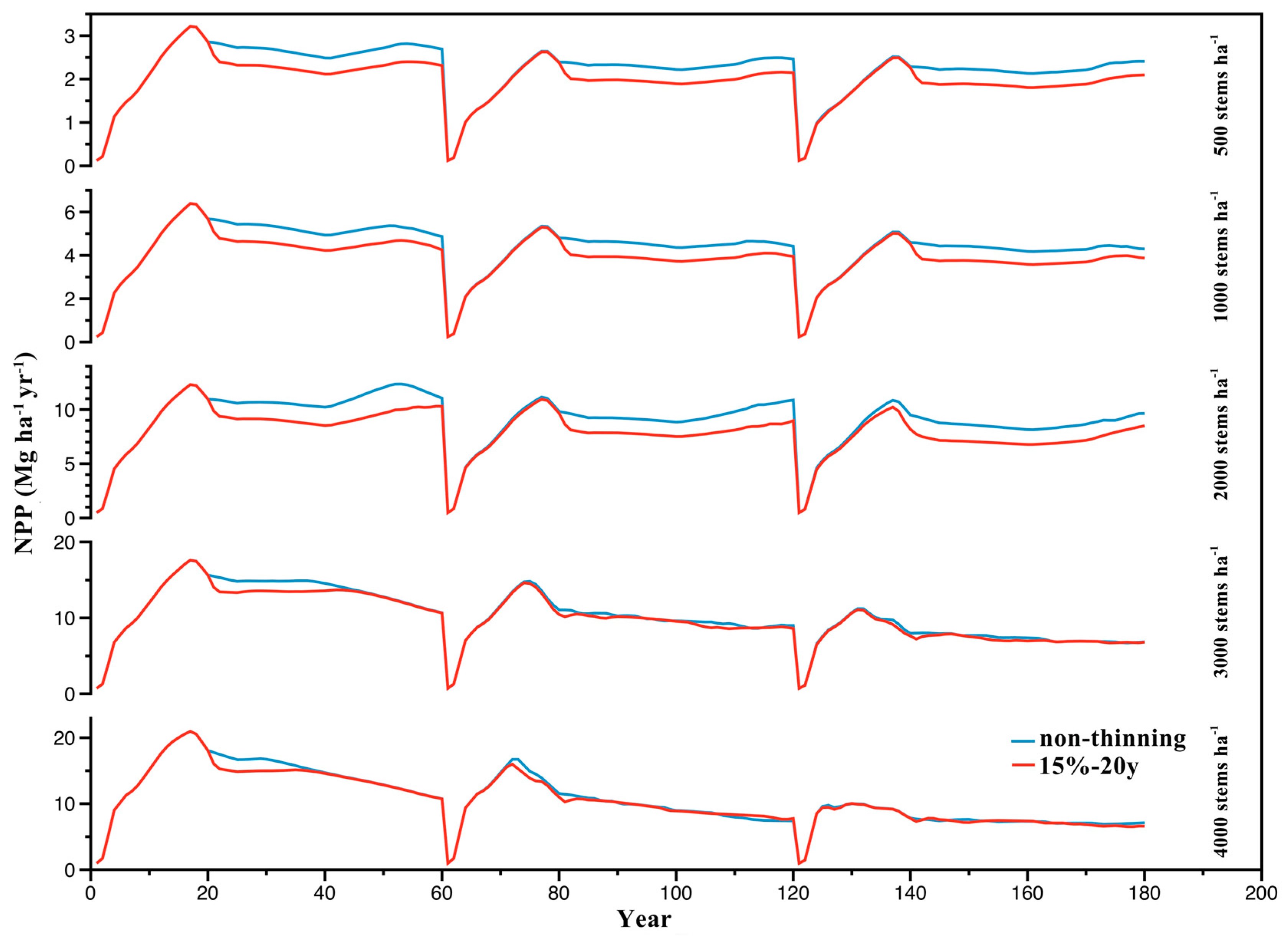
3.2. Effect of Stand Density on NPP
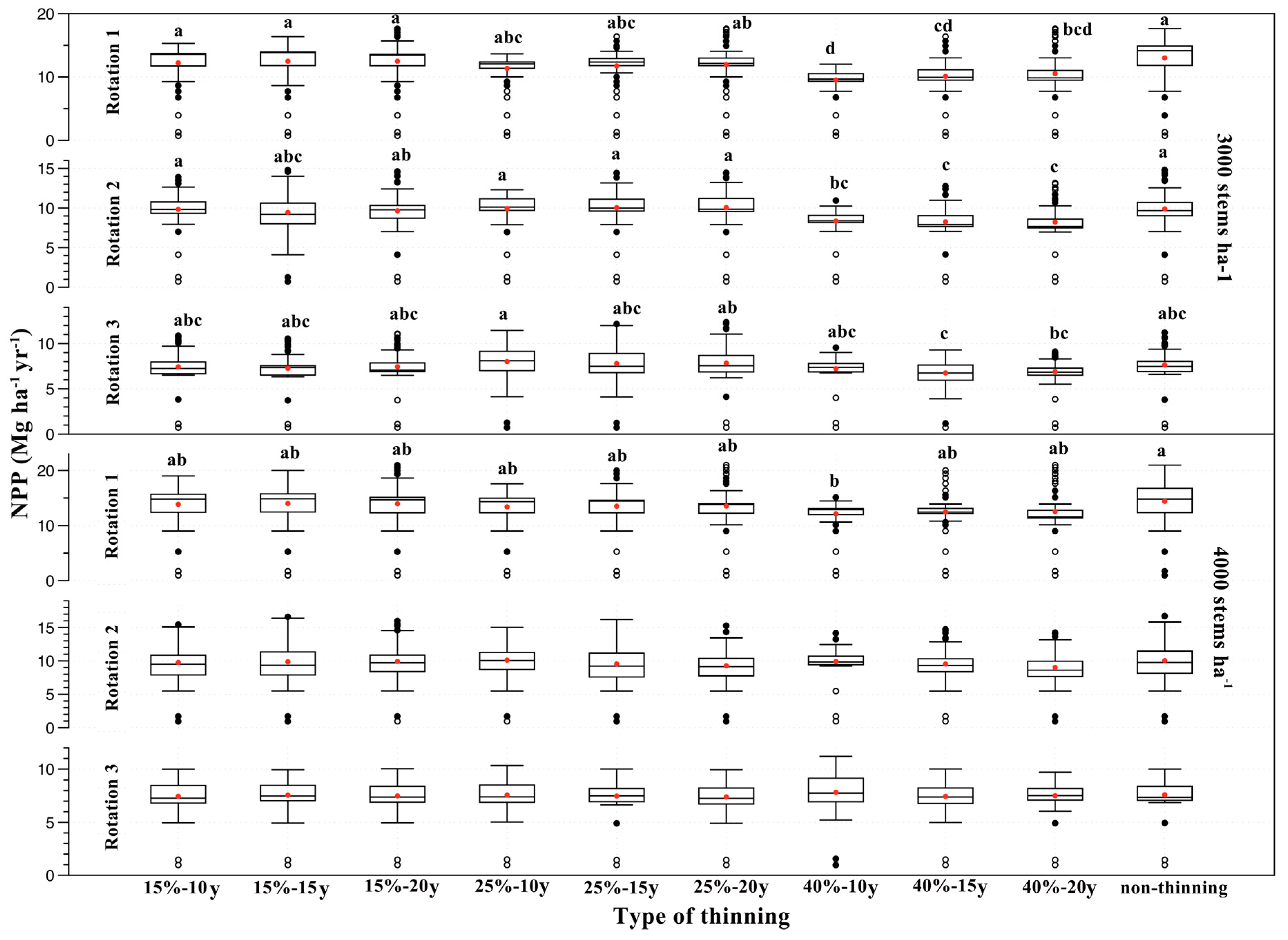
3.3. Effects of Thinning Treatments on NPP
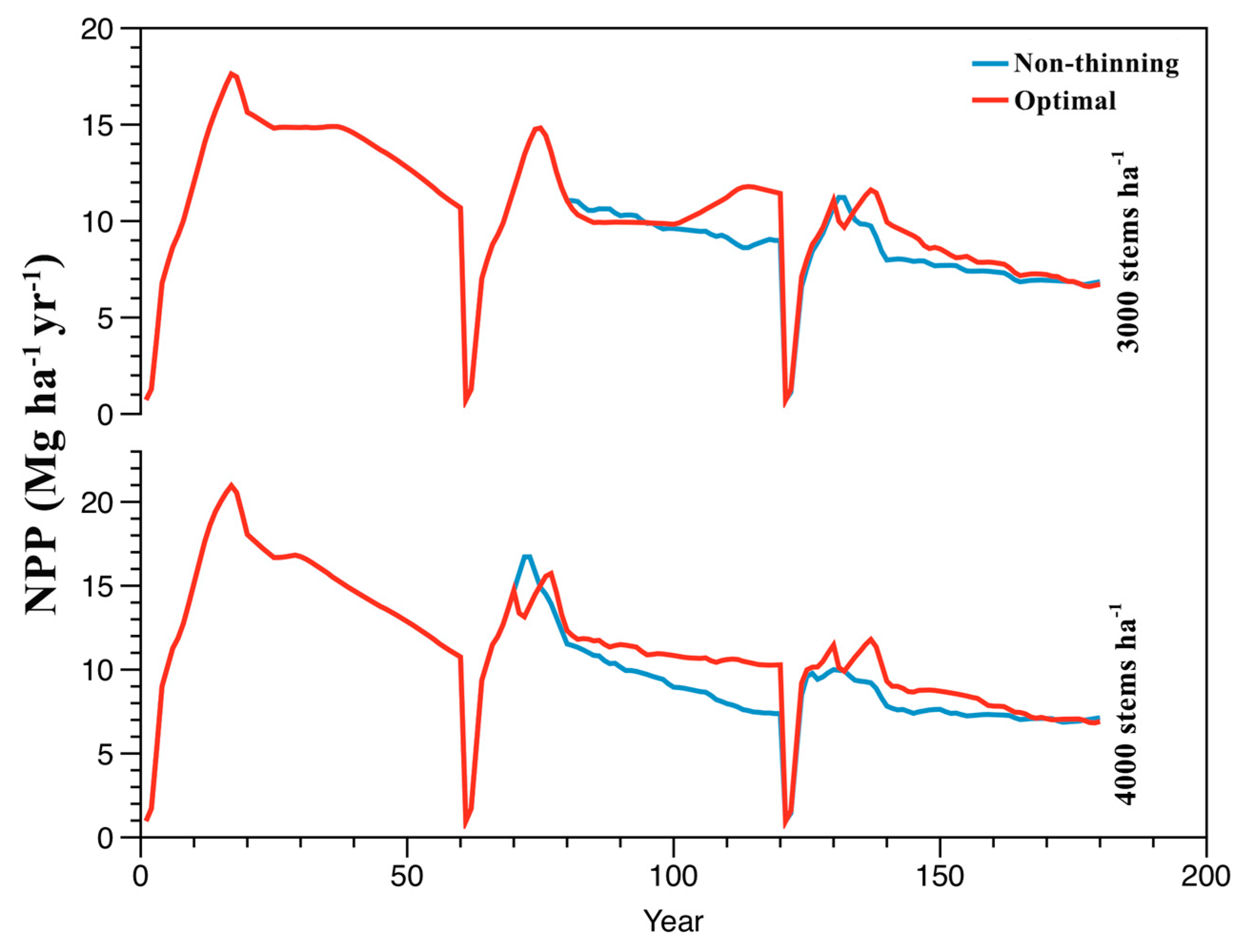
3.4. Optimal Management Prescriptions
4. Discussion
4.1. Model Evaluation
4.2. Effects of Thinning Treatments
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, C.; Li, H.; He, X.; Chen, W.; Xu, W.; Zhao, G.; Ning, Z. Concept discussion and analysis of urban forest. Chin. J. Ecol. 2003, 22, 146–149. [Google Scholar]
- Dwyer, J.F.; Mcpherson, E.G.; Schroeder, H.W.; Rowntree, R.A. Assessing the benefits and costs of the urban forest. Transp. Res. Econ. Policy 1992, 18, 751–768. [Google Scholar]
- Mcpherson, E.G.; Nowak, D.; Heisler, G.; Grimmond, S.; Souch, C.; Grant, R.; Rowntree, R. Quantifying urban forest structure, function, and value: The Chicago urban forest climate project. Urban Ecosyst. 1997, 1, 49–61. [Google Scholar] [CrossRef]
- Mcpherson, E.G. Urban forestry: The final frontier? J. For. 2003, 101, 20–25. [Google Scholar]
- Velasco, E.; Roth, M.; Norford, L.; Molina, L.T. Does urban vegetation enhance carbon sequestration? Landsc. Urban Plan. 2016, 148, 99–107. [Google Scholar] [CrossRef]
- Boukili, V.K.S.; Bebber, D.P.; Mortimer, T.; Venicx, G.; Lefcourt, D.; Chandler, M.; Eisenberg, C. Assessing the performance of urban forest carbon sequestration models using direct measurements of tree growth. Urban For. Urban Green. 2017, 24, 212–221. [Google Scholar] [CrossRef]
- Yoon, T.K.; Park, C.W.; Sun, J.L.; Ko, S.; Kim, K.N.; Son, Y.; Lee, K.H.; Oh, S.; Lee, W.K.; Son, Y. Allometric equations for estimating the aboveground volume of five common urban street tree species in Daegu, Korea. Urban For. Urban Green. 2013, 12, 344–349. [Google Scholar] [CrossRef]
- Gao, X.; Dai, Y.; Han, Y.; Liu, Z.; Niu, X.; Liu, C.; Yin, S.; Wang, B. Forest Ecology Continuous Inventory System Layout and Network Construction Research in Shanghai; China Forestry Publishing House: Beijing, China, 2016. [Google Scholar]
- Wang, Z.; Cui, X.; Yin, S.; Shen, G.; Han, Y.; Liu, C. Characteristics of carbon storage in Shanghai’s urban forest. Chin. Sci. Bull. 2013, 58, 1130–1138. [Google Scholar] [CrossRef]
- Da, L.; Yang, T.; Song, Y. Study on the urban ecological regionalization and forest distribution in Shanghai, China. Sci. Silvae Sin. 2004, 40, 84–88. [Google Scholar]
- Wu, Z.; Wang, J. Opportunities and challenges of our urban forestry. J. Chin. Urban For. 2016, 14, 7–9. [Google Scholar]
- Zhang, X.W.; Zheng, C.M.; Tang, X.J.; Zhang, W.D. Assessment of comprehensive index for forest eco-function in shanghai municipality. Subtrop. Soil Water Conserv. 2015, 27, 34–37. [Google Scholar]
- Oliver, C.D.; Larson, B.C. Forest Stand Dynamics, Update ed.; John Wiley: New York, NY, USA, 1996. [Google Scholar]
- Yang, Y.S.; Chen, G.S.; Xie, J.S.; Fan, Y.X.; Guo, J.F.; Yang, Z.J. Strategy for carbon management in China’s forests. J. For. Environ. 2015, 35, 297–303. [Google Scholar]
- Dan, R.; Jan, F. Designing the urban forest of tomorrow: Pre-commercial thinning adapted for use in urban areas in Sweden. Arboric. J. 1998, 22, 147–171. [Google Scholar]
- Kimmins, J.P.; Blanco, J.A. Issues facing forest management in Canada, and predictive ecosystem management tools for assessing possible futures. In Landscape Ecology in Forest Management and Conservation; Li, C., Lafortezza, R., Chen, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 46–72. [Google Scholar]
- Blanco, J.A.; González de Andrés, E.; San Emeterio, L.; Lo, Y.H. Modelling mixed forest stands: Methodological challenges and approaches. In Advanced Modelling Techniques Studying Glob. Changes in Environmental Sciences; Park, Y.S., Lek, S., Baehr, C., Jorgensen, S.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 186–223. [Google Scholar]
- Lo, Y.H.; Blanco, J.A.; Kimmins, J.P.; Seely, B.; Welham, C. Linking climate change and forest ecophysiology to project future trends in tree growth: A review of forest models. In Climate Change—Research and Technology for Adaptation and Mitigation; Blanco, J.A., Kheradmand, H., Eds.; InTech: Rijeka, Croatia, 2011; pp. 63–86. [Google Scholar]
- Lo, Y.H.; Blanco, J.A.; Welham, C.; Wang, M. Maintaining ecosystem function by restoring forest biodiversity: Reviewing decision-support tools that link biology, hydrology and geochemistry. In Biodiversity in Ecosystems: Linking Structure and Function; Lo, Y.H., Blanco, J.A., Roy, S., Eds.; InTech: Rijeka, Croatia, 2015; pp. 143–167. [Google Scholar]
- Blanco, J.A.; Gonzalez, E. Exploring the sustainability of current management prescriptions for Pinus caribaea plantations in Cuba: A modelling approach. J. Trop. For. Sci. 2010, 22, 139–154. [Google Scholar]
- Kimmins, J.P. Forest Ecology: A Foundation for Sustainable Forest Management and Environmental Ethics in Forestry, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2004. [Google Scholar]
- DeAngelis, D.L.; Mooij, W.M. In praise of mechanistically rich models. In Models in Ecosystem Science; Canham, C.D., Cole, J.J., Lauenroth, W.K., Eds.; Princeton University Press: Princeton, NY, USA, 2003; p. 476. [Google Scholar]
- Kimmins, J.P.; Blanco, J.A.; Seely, B.; Welham, C.; Scoullar, K. Forecasting Forest Futures: A Hybrid Modelling Approach to the Assessment of Sustainability of Forest Ecosystems and Their Values; Earthscan: London, UK, 2010. [Google Scholar]
- Seely, B.; Welham, C.; Blanco, J.A. Towards the application of soil organic matter as an indicator of forest ecosystem productivity: Deriving thresholds, developing monitoring systems, and evaluating practices. Ecol. Indic. 2010, 10, 999–1008. [Google Scholar] [CrossRef]
- Kimmins, J.P.; Blanco, J.A.; Seely, B.; Welham, C.; Scoullar, K. Complexity in modelling forest ecosystems: How much is enough? For. Ecol. Manag. 2008, 256, 1646–1658. [Google Scholar] [CrossRef]
- Chu, K.-L.; Cooper, W.S. An ecological reconnaissance in the native home of Metasequoia Glyptostroboides. Ecology 1950, 31, 260–278. [Google Scholar] [CrossRef]
- Wilczyński, S.; Muter, E.; Wertz, B. The application of the tree-ring chronologies in assessing ecological requirements of Metasequoia glyptostroboides growing in southern Poland. Geochronometria 2014, 41, 129–135. [Google Scholar] [CrossRef]
- Zhang, W.; Feng, Z.; Wang, X.; Niu, J. Impacts of elevated ozone on growth and photosynthesis of metasequoia glyptostroboides hu et cheng. Plant Sci. 2014, 226, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Lee, S.-H. Developing the volume models for 5 major species of street trees in gwangju metropolitan city of Korea. Urban For. Urban Green. 2016, 18, 53–58. [Google Scholar] [CrossRef]
- Tang, C.Q.; Yang, Y.; Ohsawa, M.; Momohara, A.; Hara, M.; Cheng, S.; Fan, S. Population structure of relict Metasequoia glyptostroboides and its habitat fragmentation and degradation in south-central China. Biol. Conserv. 2011, 144, 279–289. [Google Scholar] [CrossRef]
- Field, C.B. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, N.; Karlsson, P.E.; Bergh, J.; Nilsson, U. Impact of ozone on sequestration of carbon by swedish forests under a changing climate: A modeling study. For. Sci. 2014, 28, 101–110. [Google Scholar] [CrossRef]
- Wei, X.; Blanco, J.A. Significant increase in ecosystem c can be achieved with sustainable forest management in subtropical plantation forests. PLoS ONE 2014, 9, e89688. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Zheng, J.; Liu, G.; Liu, S.; Wang, W.; Liu, Y.; BLANCO. The concept and application of carbon sequestration potentials in plantation forests. Acta Ecol. Sin. 2015, 35, 2–8. [Google Scholar]
- Kang, E.; Lu, L.; Xu, Z. Vegetation and carbon sequestration and their relation to water resources in an inland river basin of northwest China. J. Environ. Manag. 2007, 85, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Dai, E.; Wu, Z.; Ge, Q.; Xi, W.; Wang, X. Predicting the responses of forest distribution and aboveground biomass to climate change under RCP scenarios in southern China. Glob. Chang. Biol. 2016, 22, 3642–3661. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Guo, Z.; Hu, H.; Kato, T.; Muraoka, H.; Son, Y. Forest biomass carbon sinks in east Asia, with special reference to the relative contributions of forest expansion and forest growth. Glob. Chang. Biol. 2014, 20, 2019–2030. [Google Scholar] [CrossRef] [PubMed]
- Chinese Standard GB/T 33027-2016. In Methodology for Field Long-Term Observation of Forest Ecosystem; Standards Press of China: Beijing, China, 2016.
- Xu, N.; Zhang, T.; Wang, X.; Liu, H. Soil organic carbon storage changes in Yangtze delta region, China. Environ. Earth Sci. 2011, 63, 1021–1028. [Google Scholar]
- Zhao, D.; Kane, M.; Teskey, R.; Fox, T.R.; Albaugh, T.J.; Allen, H.L.; Rubilar, R. Maximum response of loblolly pine plantations to silvicultural management in the southern united states. For. Ecol. Manag. 2016, 375, 105–111. [Google Scholar] [CrossRef]
- Kimmins, J.P. Scientific Foundations for the Simulation of Ecosystem Function and Management in Forcyte-11; Information Report NOR-X-328; Northwest Region, Northern Forestry Centre: Edmonton, AB, Canada, 1993.
- Kimmins, J.P.; Mailly, D.; Seely, B. Modelling forest ecosystem net primary production: The hybrid simulation approach used in forecast. Ecol. Model. 1999, 122, 195–224. [Google Scholar] [CrossRef]
- Xiao, C.B.; Wang, H.; Fan, K.F.; Xavier, B.; Han, Y.J.; Kang, H.Z.; Liu, C.J. Carbon storage of metasequioa glyptostroboides plantation ecosystems at different age stages in Chongming island, east China. J. Shanghai Jiaotong Univ. (Agric. Sci.) 2010, 28, 30–34. [Google Scholar]
- Ding, Y.X.; Chen, J.L. Effect of continuous plantation of Chinese fir on soil fertility. Pedosphere 1995, 5, 57–66. [Google Scholar]
- Liao, L.P.; Ma, Y.Q.; Wang, S.L.; Gao, H.; Yu, X.J. Decomposition of leaf litter of Chinese-fir in mixture with major associated broad-leaved plantation species. Acta Phytoecol. Sin. 2000, 24, 27–33. [Google Scholar]
- Liu, F.; Luo, R.; Jiang, J. Quantities and effects of various phosphorus compounds in soil under chinese-fir plantation. J. Nanjing For. Univ. 1991, 4, 7–12. [Google Scholar]
- Tian, D.L. Study on bio-geochemistry cycling in Chinese-fir ecosystem. In studies on forest ecosystems in China. In Studies on Forest Ecosystems in China; Forestry Ministry of China Publishing House of Northeast Forestry University: Harbin, China, 1994; pp. 136–145. [Google Scholar]
- Tian, D.L.; Zhao, K. Studies on the litter in a Chinese-fir plantation ecosystem: (i) amounts, composition and dynamics of litter. J. Cent. South For. Inst. 1989, 9, 38–43. [Google Scholar]
- Yang, Y.S.; Chen, G.S.; Huang, B.L. Variation in the soil water and nutrients between different rotation stands of chinese-fir. J. Nanjing For. Univ. 2000, 24, 25–28. [Google Scholar]
- Zhou, X.; Luo, R.; Ye, J. Effect of continuous cropping with Chinese-fir upon soil nutrients and its feedback. J. Nanjing For. Univ. 1991, 3, 47–52. [Google Scholar]
- Hashimoto, S.; Wattenbach, M.; Smith, P. A new scheme for initializing process-based ecosystem models by scaling soil carbon pools. Ecol. Model. 2011, 222, 3598–3602. [Google Scholar] [CrossRef]
- Shi, M.; Yang, Z.-L.; Lawrence, D.M.; Dickinson, R.E.; Subin, Z.M. Spin-up processes in the community land model version 4 with explicit carbon and nitrogen components. Ecol. Model. 2013, 263, 308–325. [Google Scholar] [CrossRef]
- Guan, B.T.; Wright, W.E.; Chung, C.-H.; Chang, S.-T. ENSO and PDO strongly influence Taiwan spruce height growth. For. Ecol. Manag. 2012, 267, 50–57. [Google Scholar] [CrossRef]
- Theil, H. Applied Econometric Forecasting; North-Holland: Amsterdam, The Netherlands, 1966. [Google Scholar]
- Vanclay, J.K.; Skovsgaard, J.P. Evaluating forest growth models. Ecol. Model. 1997, 98, 1–12. [Google Scholar] [CrossRef]
- Freese, F. Testing accuracy. For. Sci. 1960, 6, 139–145. [Google Scholar]
- Reynolds, M.R. Estimating the error in model predictions. For. Sci. 1984. [Google Scholar]
- Blanco, J.A. Forests may need centuries to recover their original productivity after continuous intensive management: An example from douglas-fir stands. Sci. Total Environ. 2012, 437, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Chinese Standard GB/T 18337.3-2001. In Non-Commercial Forest Construction-Technical Regulation; Standards Press of China: Beijing, China, 2001.
- Power, M. The predictive validation of ecological and environmental models. Ecol. Model. 1993, 68, 33–50. [Google Scholar] [CrossRef]
- Blanco, J.A.; Seely, B.; Welham, C.; Kimmins, J.P.H.; Seebacher, T.M. Testing the performance of a forest ecosystem model (FORECAST) against 29 years of field data in a Pseudotsuga menziesii plantation. Can. J. For. Res. 2007, 37, 1808–1820. [Google Scholar] [CrossRef]
- Zeide, B. Thinning and growth: A full turnaround. J. For. 2001, 99, 20–25. [Google Scholar]
- Pelletier, G.; Pitt, D.G. Silvicultural responses of two spruce plantations to midrotation commercial thinning in New Brunswick. Can. J. For. Res. 2008, 38, 851–867. [Google Scholar] [CrossRef]
- Beers, T.W. Components of forest growth. J. For. 1962, 60, 245–248. [Google Scholar]
- Marshall, D.D.; Curtis, R.O. Levels-of-Growing-Stock Cooperative Study in Douglas-Fir; Report No. 15—The Hoskins Study; U.S. Department of Agriculture: Washington, DC, USA, 2002; pp. 1963–1998.
- Soucy, M.; Lussier, J.-M.; Lavoie, L. Long-term effects of thinning on growth and yield of an upland black spruce stand. Can. J. For. Res. 2012, 42, 1669–1677. [Google Scholar] [CrossRef]
- Loftus, J. Economics and Biology of Commercial Thinning in Coastal British Columbia. In Proceedings of the Commercial Thinning Workshop, Whitecourt, AB, Canada, 17–18 October 1996; pp. 24–28. [Google Scholar]
- Bi, J.; Blanco, J.A.; Seely, B.; Kimmins, J.P.; Ding, Y.; Welham, C. Yield decline in Chinese-fir plantations: A simulation investigation with implications for model complexity. Can. J. For. Res. 2007, 37, 1615–1630. [Google Scholar] [CrossRef]
- Wei, X.; Blanco, J.A.; Jiang, H.; Kimmins, J.P.H. Effects of nitrogen deposition on carbon sequestration in chinese fir forest ecosystems. Sci. Total Environ. 2012, 416, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Jack, S.B.; Long, J.N. Linkages between silviculture and ecology: An analysis of density management diagrams. For. Ecol. Manag. 1996, 86, 205–220. [Google Scholar] [CrossRef]
- Vose, J.M.; Allen, H.L. Leaf area, stemwood growth, and nutrition relationships in loblolly pine. For. Sci. 1988, 34, 547–563. [Google Scholar]
- Binkley, D.; Reid, P. Long-term responses of stem growth and leaf area to thinning and fertilization in a Douglas-fir plantation. Can. J. For. Res. 1984, 14, 656–660. [Google Scholar] [CrossRef]
- Perry, D. The competition process in forest stands. In Attributes of Trees as Crop Plants; Cannell, M.G.R., Jackson, J.E., Eds.; Institute of Terrestrial Ecology: Abbotts Ripton, UK, 1985; pp. 481–506. [Google Scholar]
- Brix, H. Effects of thinning and nitrogen fertilization on growth of Douglas-fir: Relative contribution of foliage quantity and efficiency. Can. J. For. Res. 1983, 13, 167–175. [Google Scholar] [CrossRef]
- Oren, R.; Waring, R.H.; Stafford, S.G.; Barrett, J.W. Twenty-four years of ponderosa pine growth in relation to canopy leaf area and understory competition. For. Sci. 1987, 33, 538–547. [Google Scholar]
- Velazquez-Martinez, A.; Perry, D.A.; Bell, T.E. Response of aboveground biomass increment, growth efficiency, and foliar nutrients to thinning, fertilization, and pruning in young Douglas-fir plantations in the central Oregon Cascades. Can. J. For. Res. 1992, 22, 1278–1289. [Google Scholar] [CrossRef]
- Pretzsch, H.; Mette, T. Linking stand-level self-thinning allometry to the tree-level leaf biomass allometry. Trees 2008, 22, 611–622. [Google Scholar] [CrossRef]
- Boivin-Dompierre, S.; Achim, A.; Pothier, D. Functional response of coniferous trees and stands to commercial thinning in eastern Canada. For. Ecol. Manag. 2017, 384, 6–16. [Google Scholar] [CrossRef]
- Morris, D.M.; Hamish Kimmins, J.P.; Duckert, D.R. The use of soil organic matter as a criterion of the relative sustainability of forest management alternatives: A modelling approach using forecast. For. Ecol. Manag. 1997, 94, 61–78. [Google Scholar] [CrossRef]
- Farley, K.A.; Kelly, E.F. Effects of afforestation of a páramo grassland on soil nutrient status. For. Ecol. Manag. 2004, 195, 281–290. [Google Scholar] [CrossRef]
- Polglase, P.J.; Attiwill, P.M. Nitrogen and phosphorus cycling in relation to stand age of Ecucalyptus regnans F. Muell. Plant Soil 1992, 142, 157–166. [Google Scholar] [CrossRef]
- Sun, Z.; Liu, L.; Peng, S.; Peñuelas, J.; Zeng, H.; Piao, S. Age-related modulation of the nitrogen resorption efficiency response to growth requirements and soil nitrogen availability in a temperate pine plantation. Ecosystems 2017, 19, 698–709. [Google Scholar] [CrossRef]
- Seely, B.; Welham, C.; Kimmins, H. Carbon sequestration in a boreal forest ecosystem: Results from the ecosystem simulation model, forecast. For. Ecol. Manag. 2002, 169, 123–135. [Google Scholar] [CrossRef]
- Parker, J.L.; Fernandez, I.J.; Rustad, L.E.; Norton, S.A. Effects of nitrogen enrichment, wildfire, and harvesting on forest-soil carbon and nitrogen. Soil Sci. Soc. Am. J. 2001, 65, 1248–1255. [Google Scholar] [CrossRef]
- Zhang, J.; Webster, J.; Young, D.H.; Fiddler, G.O. Effect of thinning and soil treatments on Pinus ponderosa plantations: 15-year results. For. Ecol. Manag. 2016, 368, 123–132. [Google Scholar] [CrossRef]
- LeBauer, D.S.; Treseder, K.K. Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 2008, 89, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Zhou, X.; Luo, Y.; Yang, Y.; Fang, C.; Chen, J.; Li, B. Minor stimulation of soil carbon storage by nitrogen addition: A meta-analysis. Agric. Ecosyst. Environ. 2011, 140, 234–244. [Google Scholar] [CrossRef]
- Environmental Protection Agency (EPA). Integrated Science Assessment for Oxides of Nitrogen and Sulfur—Environmental Criteria; National Center for Environmental Assessment, U.S. Environmental Protection Agency: Research Triangle Park, NC, USA, 2008.
- Thomas, R.Q.; Canham, C.D.; Weathers, K.C.; Goodale, C.L. Increased tree carbon storage in response to nitrogen deposition in the US. Nat. Geosci. 2010, 3, 229–244. [Google Scholar]
- Niu, S.; Classen, A.T.; Dukes, J.S.; Kardol, P.; Liu, L.; Luo, Y.; Rustad, L.; Sun, J.; Tang, J.; Templer, P.H.; et al. Global patterns and substrate-based mechanisms of the terrestrial nitrogen cycle. Ecol. Lett. 2016, 19, 697–709. [Google Scholar] [CrossRef] [PubMed]
- Seely, B.; Welham, C.; Scoullar, K. Application of a hybrid forest growth model to evaluate climate change impacts on productivity, nutrient cycling and mortality in a montane forest ecosystem. PLoS ONE 2015, 10, e0135034. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wei, X.; Liao, W.; Blanco, J.A.; Liu, Y.; Liu, S.; Liu, G.; Zhang, L.; Guo, X.; Guo, S. Evaluation of the effects of forest management strategies on carbon sequestration in evergreen broad-leaved (Phoebe bournei) plantation forests using forecast ecosystem model. For. Ecol. Manag. 2013, 300, 21–32. [Google Scholar] [CrossRef]
- Reyer, C. Forest productivity under environmental change—A review of stand-scale modeling studies. Curr. For. Rep. 2015, 1, 53–68. [Google Scholar] [CrossRef]
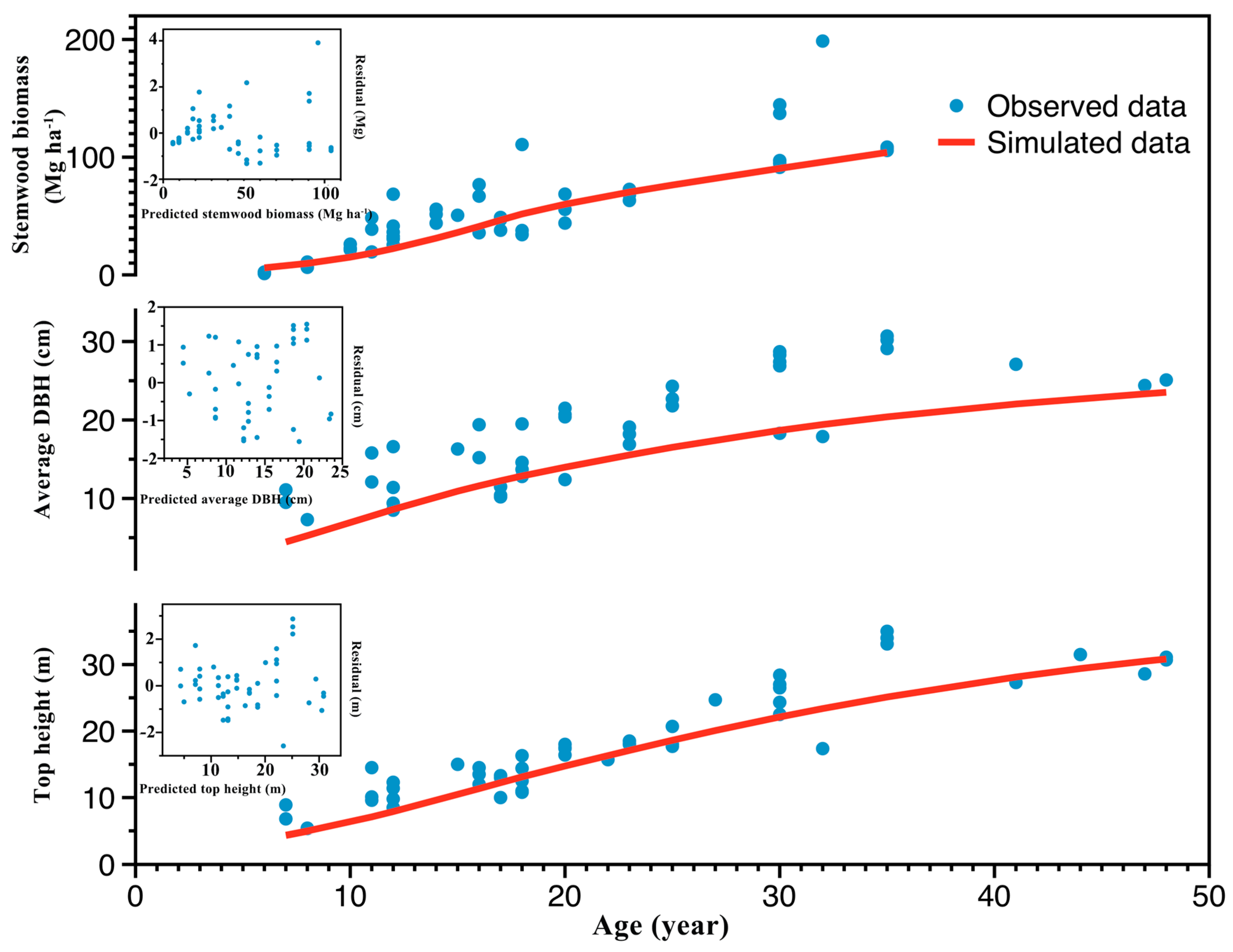

| Index | Stemwood Biomass | DBH | Top Height |
|---|---|---|---|
| Average bias | 10.33 Mg ha−1 | 1.84 cm | 3.96 m |
| MAE | 14.91 Mg ha−1 | 2.61 cm | 4.39 m |
| R2 | 0.71 | 0.70 | 0.87 |
| ME | 0.67 | 0.52 | 0.83 |
| Theil’s U | 0.36 | 0.17 | 0.28 |
| e*α = 0.05 | 40.94 Mg ha−1 | 9.20 cm | 5.89 m |
| e*α = 0.20 | 28.88 Mg ha−1 | 6.51 cm | 4.14 m |
| Stand Density (stems ha−1) | Rotation | Difference in Accumulated NPP between Thinning and Non-Thinning Treatments (Mg ha−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 15%-10y | 15%-15y | 15%-20y | 25%-10y | 25%-15y | 25%-20y | 40%-10y | 40%-15y | 40%-20y | ||
| 3000 | 1 | −47.20 | −31.03 | −30.90 | −98.96 | −75.11 | −63.54 | −208.88 | −175.10 | −148.63 |
| 2 | −2.88 | −26.08 | −14.19 | 2.40 | 9.68 | 10.49 | −92.23 | −97.57 | −99.31 | |
| 3 | −12.42 | −22.11 | −11.56 | 22.71 | 9.67 | 12.69 | −24.65 | −52.56 | −43.77 | |
| 4000 | 1 | −32.80 | −23.51 | −26.34 | −60.13 | −53.29 | −51.28 | −133.88 | −116.82 | −110.12 |
| 2 | −16.29 | −10.21 | −7.18 | 4.00 | −30.14 | −46.49 | −8.04 | −30.62 | −61.60 | |
| 3 | −8.33 | −2.00 | −6.15 | −1.52 | −7.00 | −12.01 | 13.90 | −8.91 | −5.16 | |
| Stand Density (stems ha−1) | Rotation | Thinning Treatment | Difference in Accumulated NPP (Mg ha−1) | |
|---|---|---|---|---|
| Per Rotation | In Three Rotations | |||
| 3000 | 2 | 25%-20y | 32.98 | 65.55 |
| 3 | 25%-10y | 32.57 | ||
| 4000 | 2 | 25%-10y | 69.01 *,a | 114.84 |
| 3 | 40%-10y | 45.83 *,b | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, J.; Blanco, J.A.; Wei, X.; Liu, C. Sustainable Management of Metasequoia glyptostroboides Plantation Forests in Shanghai. Forests 2018, 9, 64. https://doi.org/10.3390/f9020064
Zheng J, Blanco JA, Wei X, Liu C. Sustainable Management of Metasequoia glyptostroboides Plantation Forests in Shanghai. Forests. 2018; 9(2):64. https://doi.org/10.3390/f9020064
Chicago/Turabian StyleZheng, Ji, Juan A. Blanco, Xiaohua Wei, and Chunjiang Liu. 2018. "Sustainable Management of Metasequoia glyptostroboides Plantation Forests in Shanghai" Forests 9, no. 2: 64. https://doi.org/10.3390/f9020064
APA StyleZheng, J., Blanco, J. A., Wei, X., & Liu, C. (2018). Sustainable Management of Metasequoia glyptostroboides Plantation Forests in Shanghai. Forests, 9(2), 64. https://doi.org/10.3390/f9020064






