Effect of Predation, Competition, and Facilitation on Tree Survival and Growth in Abandoned Fields: Towards Precision Restoration
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Experimental Design
2.2.1. Survival and Growth
2.2.2. Foliar Measurements
2.2.3. Soil Water Content and Light
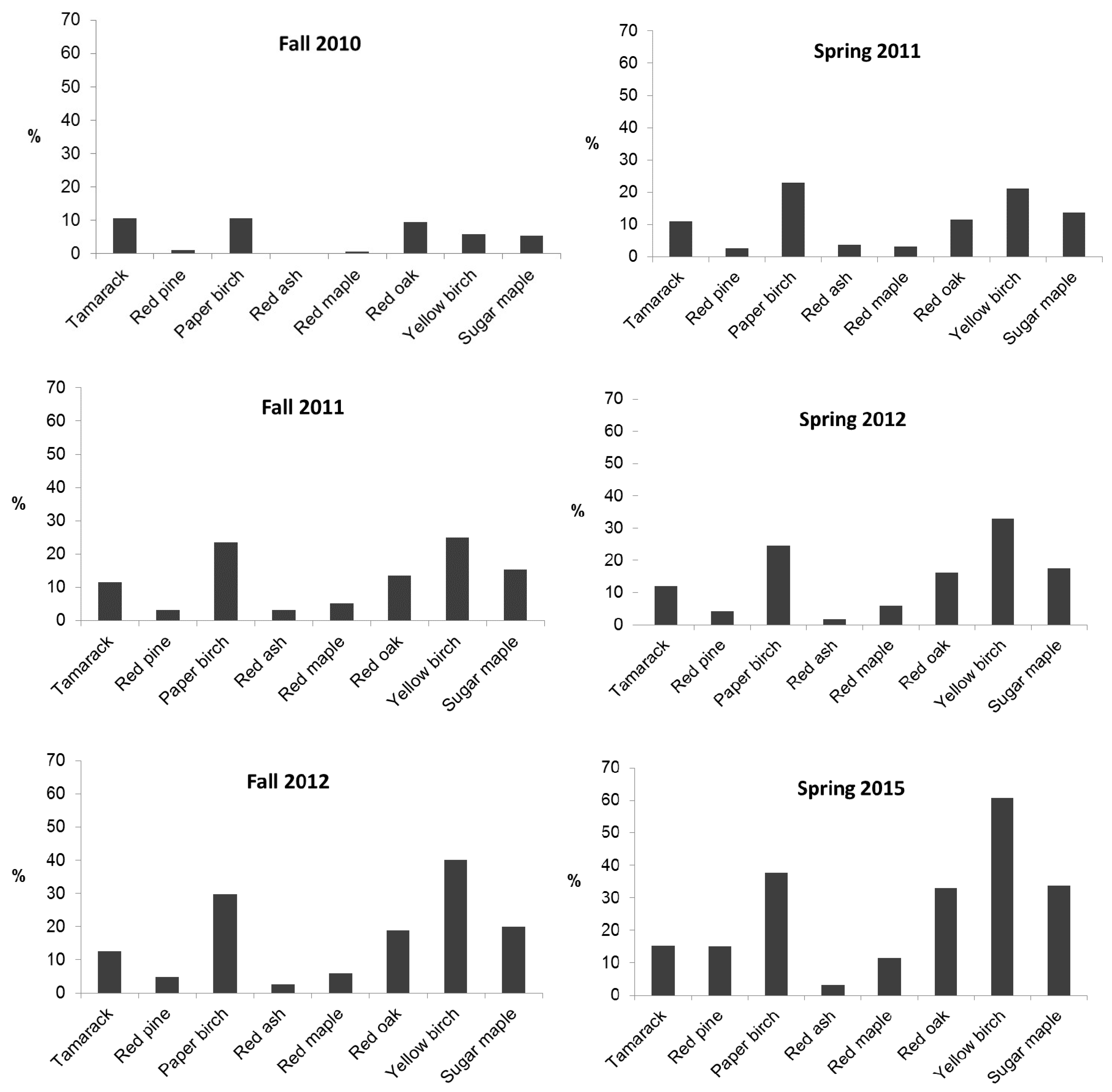
2.2.4. Herbaceous Vegetation
2.3. Statistical Analysis
3. Results
3.1. General
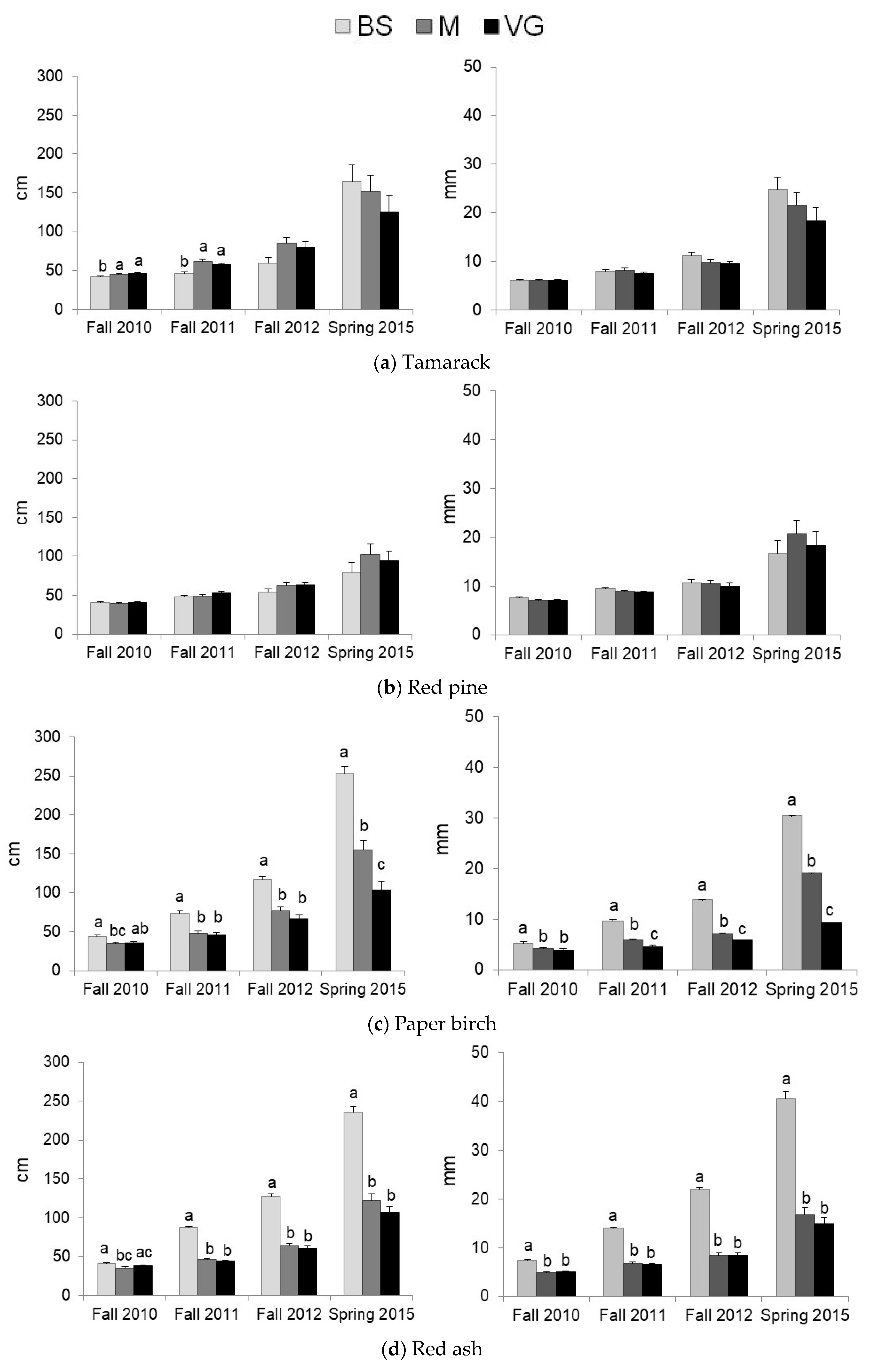
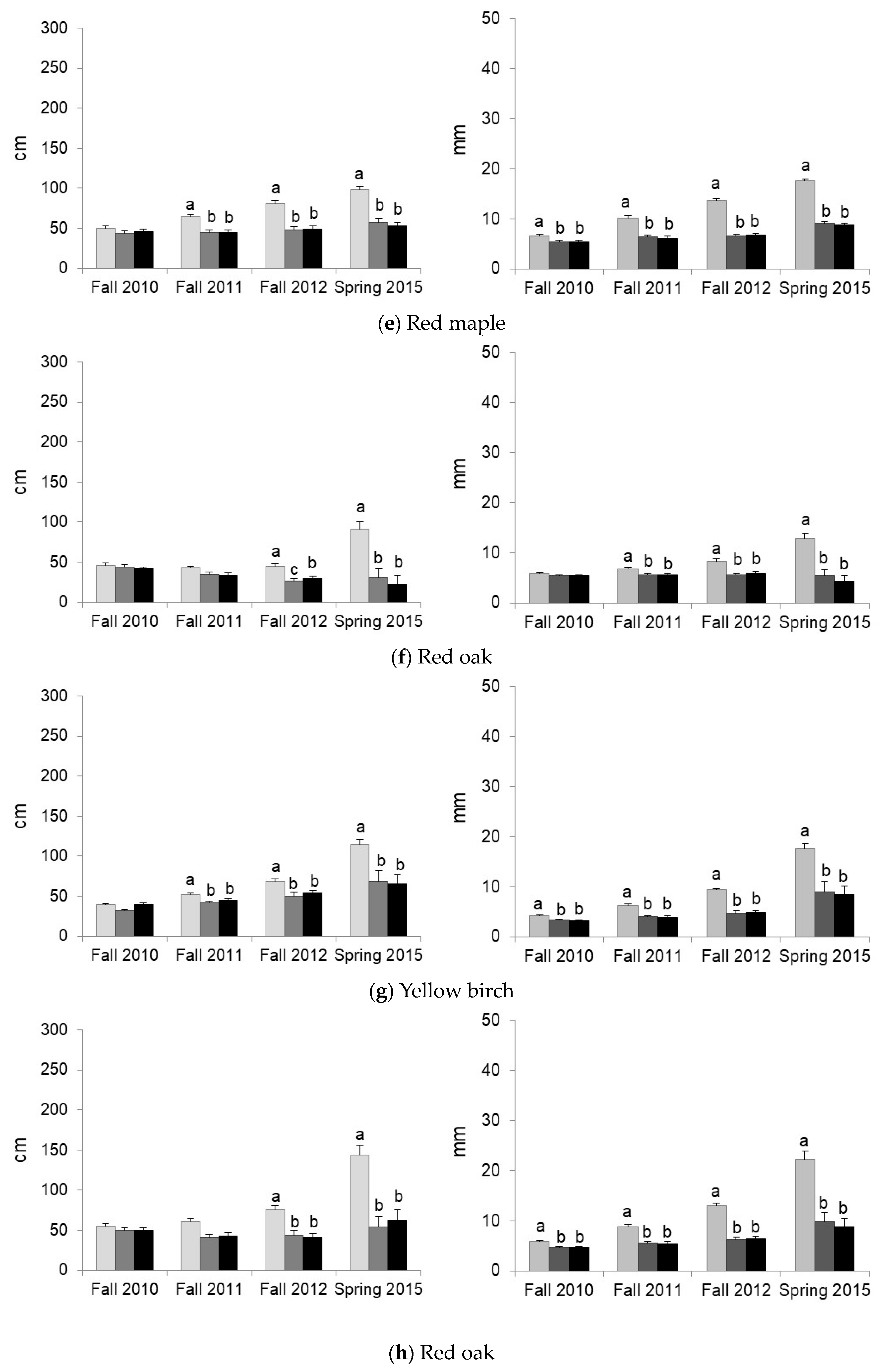
3.2. Predation and Herbaceous Vegetation Effects on Tree Survival and Growth
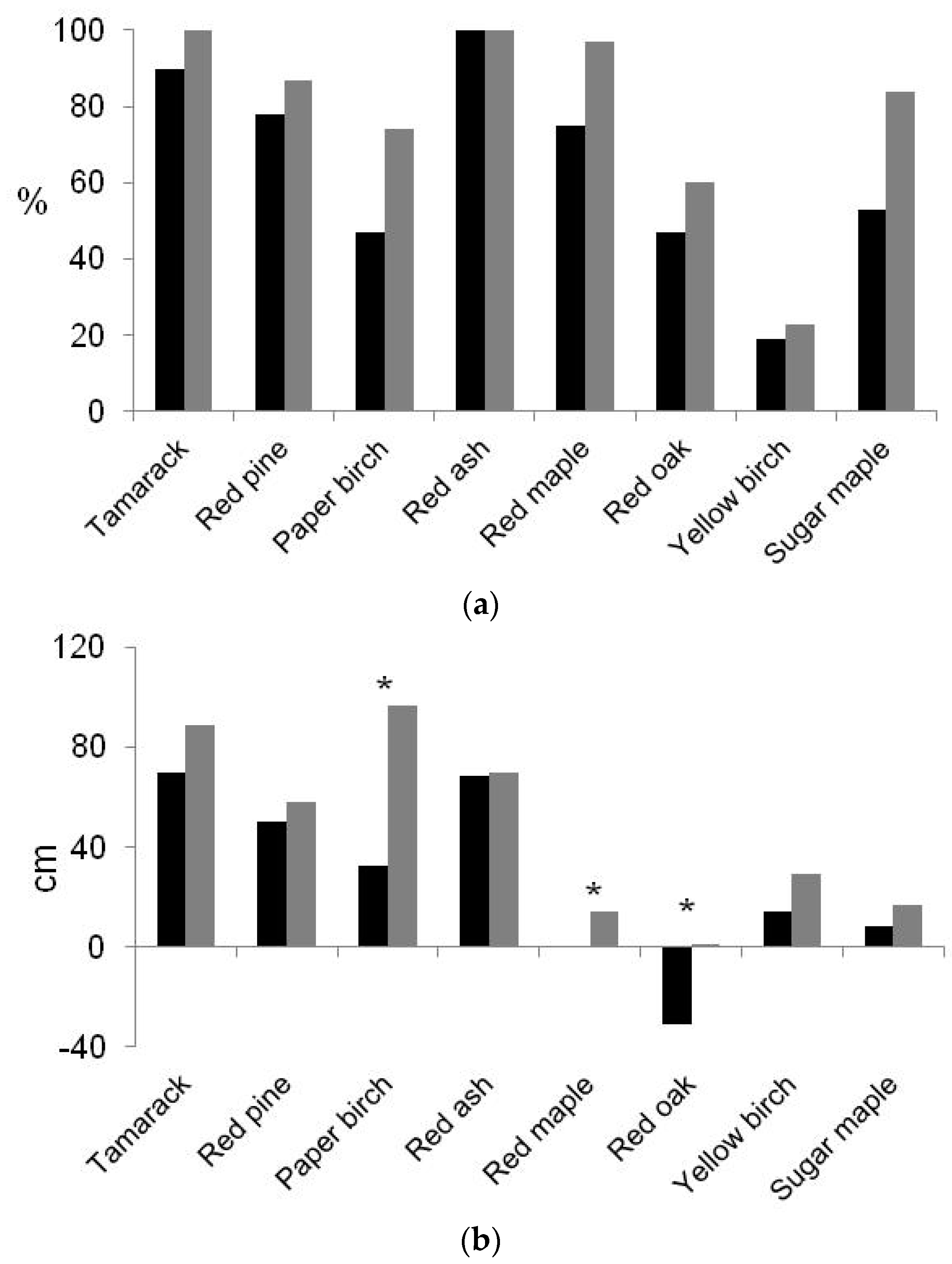
3.3. Foliar Attributes, Soil Water Content, and the Influence of Herb Communities
4. Discussion
4.1. Predation
4.2. Competition, Tolerance, and Facilitation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Padilla, F.M.; Pugnaire, F.I. The role of nurse plants in the restoration of degraded environments. Front. Ecol. Environ. 2006, 4, 196–202. [Google Scholar] [CrossRef]
- Gómez-Aparicio, L. The role of plant interactions in the restoration of degraded ecosystems: A meta-analysis across life-forms and ecosystems. J. Ecol. 2009, 97, 1202–1214. [Google Scholar] [CrossRef]
- Goldberg, D.E.; Rajaniemi, T.; Gurevitch, J.; Stewart-Oaten, A. Empirical approaches to quantifying interaction intensity: Competition and facilitation along productivity gradients. Ecology 1999, 80, 1118–1131. [Google Scholar] [CrossRef]
- De Steven, D. Experiments on mechanisms of tree establishment in old-field succession: Seedling emergence. Ecology 1991, 72, 1066–1075. [Google Scholar] [CrossRef]
- De Steven, D. Experiments on mechanisms of tree establishment in old-field succession: Seedling survival and growth. Ecology 1991, 72, 1076–1088. [Google Scholar] [CrossRef]
- Gill, D.S.; Marks, P.L. Tree and shrub seedling colonization of old fields in central New York. Ecol. Monogr. 1991, 61, 183–205. [Google Scholar] [CrossRef]
- Berkowitz, A.R.; Canham, C.D.; Kelly, V.R. Competition vs. Facilitation of tree seedling growth and survival in early successional communities. Ecology 1995, 76, 1156–1168. [Google Scholar] [CrossRef]
- Callaway, R.M. Positive interactions in plant communities and the individualistic-continuum concept. Oecologia 1997, 112, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Niering, W.A.; Goodwin, R.H. Creation of relatively stable shrublands with herbicides: Arresting “Succession” on rights-of-way and pastureland. Ecology 1974, 55, 784–795. [Google Scholar] [CrossRef]
- Benjamin, K.; Domon, G.; Bouchard, A. Vegetation composition and succession of abandoned farmland: Effects of ecological, historical and spatial factors. Landsc. Ecol. 2005, 20, 627–647. [Google Scholar] [CrossRef]
- Burton, P.J.; Bazzaz, F.A. Ecophysiological Responses of Tree Seedlings Invading Different Patches of Old-Field Vegetation. J. Ecol. 1995, 83, 99–112. [Google Scholar] [CrossRef]
- Davis, M.A.; Wrage, K.J.; Reich, P.B. Competition between tree seedlings and herbaceous vegetation: Support for a theory of resource supply and demand. J. Ecol. 1998, 86, 652–661. [Google Scholar] [CrossRef]
- Davis, M.A.; Wrage, K.J.; Reich, P.B.; Tjoelker, M.G.; Schaeffer, T.; Muermann, C. Survival, growth, and photosynthesis of tree seedlings competing with herbaceous vegetation along a water-light-nitrogen gradient. Plant Ecol. 1999, 145, 341–350. [Google Scholar] [CrossRef]
- Laliberté, E.; Bouchard, A.; Cogliastro, A. Optimizing hardwood reforestation in old fields: The effects of treeshelters and environmental factors on tree seedling growth and physiology. Rest. Ecol. 2008, 16, 270–280. [Google Scholar] [CrossRef]
- Gale, M.R.; Grigal, D.F. Vertical root distributions of northern tree species in relation to successional status. Can. J. For. Res. 1987, 17, 829–834. [Google Scholar] [CrossRef]
- Coll, L.; Potvin, C.; Messie, C.; Delagrange, S. Root architecture and allocation patterns of eight native tropical species with different successional status used in open-grown mixed plantations in Panama. Trees 2008, 22, 585–596. [Google Scholar] [CrossRef]
- Hooper, E.; Condit, R.; Legendre, P. Responses of 20 native tree species to reforestation strategies for abandoned farmland in Panama. Ecol. Appl. 2002, 12, 1626–1641. [Google Scholar] [CrossRef]
- Doust, S.J.; Erskine, P.D.; Lamb, D. Direct seeding to restore rainforest species: Microsite effects on the early establishment and growth of rainforest tree seedlings on degraded land in the wet tropics of Australia. For. Ecol. Manag. 2006, 234, 333–343. [Google Scholar] [CrossRef]
- Doust, S.J.; Erskine, P.D.; Lamb, D. Restoring rainforest species by direct seeding: Tree seedling establishment and growth performance on degraded land in the wet tropics of Australia. For. Ecol. Manag. 2008, 256, 1178–1188. [Google Scholar] [CrossRef]
- Caldwell, M.M.; Richards, H.J. Competing root systems: Morphology and models of absorption. In On the Economy of Plant Form and Function; Givnish, T.J., Ed.; Cambridge University Press: Cambridge, UK, 1986; pp. 251–273. [Google Scholar]
- Pywell, R.F.; Bullock, J.M.; Roy, D.B.; Warman, L.I.Z.; Walker, K.J.; Rothery, P. Plant traits as predictors of performance in ecological restoration. J. Appl. Ecol. 2003, 40, 65–77. [Google Scholar] [CrossRef]
- Horsley, S.B. Allelopathic inhibition of black cherry by fern, grass, goldenrod, and aster. Can. J. For. Res. 1977, 7, 205–216. [Google Scholar] [CrossRef]
- De Blois, S.; Brisson, J.; Bouchard, A. Herbaceous covers to control tree invasion in rights-of-way: Ecological concepts and applications. Environ. Manag. 2004, 33, 606–619. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Canham, C.D. Effects of meadow vole population density on tree seedling survival in old fields. Ecology 1993, 74, 1792–1801. [Google Scholar] [CrossRef]
- Pusenius, J.; Ostfeld, R.S.; Keesing, F. Patch selection and tree-seedling predation by resident vs. immigrant meadow voles. Ecology 2000, 81, 2951–2956. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Manson, R.H.; Canham, C.D. Effects of rodents on survival of tree seeds and seedlings invading old fields. Ecology 1997, 78, 1531–1542. [Google Scholar] [CrossRef]
- Stange, E.E.; Shea, K.L. Effects of deer browsing, fabric mats, and tree shelters on Quercus rubra seedings. Rest. Ecol. 1998, 16, 29–34. [Google Scholar]
- Moore, N.P.; Hart, J.D.; Langton, S.D. Factors influencing browsing by fallow deer Dama dama in young broad-leaved plantations. Biol. Conserv. 1999, 87, 255–260. [Google Scholar] [CrossRef]
- Moore, N.P.; Hart, J.D.; Kelly, P.F.; Langton, S.D. Browsing by fallow deer (Dama dama) in young broadleaved plantations: Seasonality, and the effects of previous browsing and bud eruption. Forestry 2000, 73, 437–445. [Google Scholar] [CrossRef]
- McPherson, G.R. Effects of herbivory and herb interference on oak establishment in a semi-arid temperate savanna. J. Veg. Sci. 1993, 4, 687–692. [Google Scholar] [CrossRef]
- Ward, J.S.; Gent, M.P.; Stephens, G.R. Effects of planting stock quality and browse protection-type on height growth of northern red oak and eastern white pine. For. Ecol. Manag. 2000, 127, 205–216. [Google Scholar] [CrossRef]
- Sweeney, B.W.; Czapka, S.J.; Yerkes, T. Riparian forest restoration: Increasing success by reducing plant competition and herbivory. Rest. Ecol. 2002, 10, 392–400. [Google Scholar] [CrossRef]
- Groninger, J.W. Increasing the impact of bottomland hardwood afforestation. J. For. 2005, 103, 184–188. [Google Scholar] [CrossRef]
- Cogliastro, A.; Benjamin, K.; Bouchard, A. Effects of full and partial clearing, with and without herbicide, on weed cover, light availability, and establishment success of white ash in shrub communities of abandoned pastureland in southwestern Quebec, Canada. New For. 2006, 32, 197–210. [Google Scholar] [CrossRef]
- Sampaio, A.B.; Holl, K.D.; Scariot, A. Does restoration enhance regeneration of seasonal deciduous forests in pastures in central Brazil? Rest. Ecol. 2007, 15, 462–471. [Google Scholar] [CrossRef]
- Barea, J.M.; Azcon-Aguilar, C.; Azcon, R. Interactions between mycorrhizal fungi and rhisosphere micro-organisms within the context of sustainable soil-plant systems. In Multitrophic Interactions in Terrestrial Systems; Gange, A.C., Brown, V.K., Eds.; Blackwell Science: Cambridge, MA, USA, 1997; pp. 65–78. [Google Scholar]
- Alguacil, M.M.; Lumini, E.; Roldán, A.; Salinas-García, J.R.; Bonfante, P.; Bianciotto, V. The impact of tillage practices on arbuscular mycorrhizal fungal diversity in subtropical crops. Ecol. Appl. 2008, 18, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Cogliastro, A.; Gagnon, D.; Coderre, D.; Bhereur, P. Responses of seven hardwood tree species to herbicide, rototilling, and legume cover at two southern Quebec plantation sites. Can. J. For. Res. 1990, 20, 1172–1182. [Google Scholar] [CrossRef]
- Coll, L.; Messier, C.; Delagrange, S.; Berninger, F. Growth, allocation and leaf gas exchanges of hybrid poplar plants in their establishment phase on previously forested sites: Effect of different vegetation management techniques. Ann. For. Sci. 2007, 64, 275–285. [Google Scholar] [CrossRef]
- Davies, R.J. The importance of weed control and the use of tree shelters for establishing broadleaved trees on grass-dominated sites in England. Forestry 1985, 58, 167–180. [Google Scholar] [CrossRef]
- Davies, R.J. Sheet mulching as an aid to broadleaved tree establishment II. Comparison of various sizes of black polythene mulch and herbicide treated spot. Forestry 1988, 61, 107–124. [Google Scholar] [CrossRef]
- Truax, B.; Gagnon, D. Effects of straw and black plastic mulching on the initial growth and nutrition of butternut, white ash and bur oak. For. Ecol. Manag. 1993, 57, 17–27. [Google Scholar] [CrossRef]
- Lambert, F.; Truax, B.; Gagnon, D.; Chevrier, N. Growth and N nutrition, monitored by enzyme assays, in a hardwood plantation: Effects of mulching materials and glyphosate application. For. Ecol. Manag. 1994, 70, 231–244. [Google Scholar] [CrossRef]
- Environment and Climate Change Canada. Canadian Climate Normals 1981–2010. Montréal/Pierre Elliott Trudeau Intl A, Québec. Available online: http://climate.weather.gc.ca/climate_normals/ (accessed on 22 January 2013).
- Environment and Climate Change Canada. Monthly data report for 2010, 2011, 2012. Montréal/Pierre Elliott Trudeau Intl A, Québec. Available online: http://climate.weather.gc.ca/index_f.html (accessed on 22 January 2013).
- Institut de recherche et de développement en agroenvironnement (IRDA). Études pédologiques. Available online: http://www.irda.qc.ca/fr/outils-et-services/informations-sur-les-sols/etudes-pedologiques/ (accessed on 12 december 2012).
- Cornelissen, J.H.C.; Lavorel, S.; Garnier, E.; Díaz, S.; Buchmann, N.; Gurvich, D.E.; Reich, P.B.; Ter Steege, H.; Morgan, H.D.; van der Heijden, M.G.A.; et al. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture. The PLANTS Database. Available online: http://plants.usda.gov (accessed on 11 February 2013).
- Burns, R.M.; Honkala, B.H. Silvics of North America: 1. Conifers; 2. Hardwoods; U.S. Department of Agriculture, Forest Service: Washington, DC, USA, 1990; Volume 2, p. 877.
- Niinemets, Ü.; Valladares, F. Tolerance to shade, drought, and waterlogging of temperate Northern Hemisphere trees and shrubs. Ecol. Monogr. 2006, 76, 521–547. [Google Scholar] [CrossRef]
- Messier, C.; Puttonen, P. Spatial and temporal variation in the light environment of developing scots pine stands: The basis for a quick and efficient method of characterizing light. Can. J. For. Res. 1995, 25, 343–354. [Google Scholar] [CrossRef]
- Bergman, M.; Iason, G.R.; Hester, A.J. Feeding patterns by roe deer and rabbits on pine, willow and birch in relation to spatial arrangement. Oikos 2005, 109, 513–520. [Google Scholar] [CrossRef]
- Bucyanayandi, J.D.; Bergeron, J.M.; Menard, H. Preference of meadow voles (Microtus pennsylvanicus) for conifer seedlings: Chemical components and nutritional quality of bark of damaged and undamaged trees. J. Chem. Ecol. 1990, 16, 2569–2579. [Google Scholar] [CrossRef] [PubMed]
- Bertness, M.D.; Callaway, R. Positive interactions in communities. Trends Ecol. Evol. 1994, 9, 191–193. [Google Scholar] [CrossRef]
- Holmgren, M.; Scheffer, M.; Huston, M.A. The interplay of facilitation and competition in plant communities. Ecology 1997, 78, 1966–1975. [Google Scholar] [CrossRef]
- Callaway, R.M.; Walker, L.R. Competition and facilitation: A synthetic approach to interactions in plant communities. Ecology 1997, 78, 1958–1965. [Google Scholar] [CrossRef]
- Strong, W.L.; Roi, G.L. Root-system morphology of common boreal forest trees in Alberta, Canada. Can. J. For. Res. 1983, 13, 1164–1173. [Google Scholar] [CrossRef]
- Tobner, C.M.; Paquette, A.; Messier, C. Interspecific coordination and intraspecific plasticity of fine root traits in North American temperate tree species. Front. Plant Sci. 2013, 4, 242. [Google Scholar] [CrossRef] [PubMed]
- Buitrago, M.; Paquette, A.; Thiffault, N.; Bélanger, N.; Messier, C. Early performance of planted hybrid larch: Effects of mechanical site preparation and planting depth. New For. 2014, 46, 319–337. [Google Scholar] [CrossRef]
- Knops, J.M.; Tilman, D. Dynamics of soil nitrogen and carbon accumulation for 61 years after agricultural abandonment. Ecology 2000, 81, 88–98. [Google Scholar] [CrossRef]
- Reynolds, H.L.; Hartley, A.E.; Vogelsang, K.M.; Bever, J.D.; Schultz, P.A. Arbuscular mycorrhizal fungi do not enhance nitrogen acquisition and growth of old-field perennials under low nitrogen supply in glasshouse culture. New Phytol. 2005, 167, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Connell, J.H.; Slatyer, R.O. Mechanisms of succession in natural communities and their role in community stability and organization. Am. Nat. 1977, 111, 1119–1144. [Google Scholar] [CrossRef]
- Gebbers, R.; Adamchuk, V.I. Precision agriculture and food security. Science 2010, 327, 828–831. [Google Scholar] [CrossRef] [PubMed]
- Laliberté, E.; Cogliastro, A.; Bouchard, A. Spatiotemporal patterns in seedling emergence and early growth of two oak species direct-seeded on abandoned pastureland. Ann. For. Sci. 2008, 65, 407. [Google Scholar] [CrossRef]
- St-Denis, A.; Messier, C.; Kneeshaw, D. Seed size, the only factor positively affecting direct seeding success in an abandoned field in Quebec, Canada. Forests 2013, 4, 500–516. [Google Scholar] [CrossRef]
- Martelletti, S.; Lingua, E.; Meloni, F.; Freppaz, M.; Motta, R.; Nosenzo, A.; Marzano, R. Microsite manipulation in lowland oak forest restoration result in indirect effects on acorn predation. For. Ecol. Manag. 2018, 411, 27–34. [Google Scholar] [CrossRef]
| Common Name | Scientific Name | Mycorrhizal Association | Successional Status | Growth Rate | Shade Tolerance | Initial Mean Height (cm) | Initial Mean Diameter (mm) |
|---|---|---|---|---|---|---|---|
| Tamarack | Larix laricina (Du Roi) K. Koch | EM | Pioneer | Rapid | 1 | 38.7 | 6.8 |
| Red pine | Pinusresinosa Aiton | EM | Pioneer | Rapid | 1.9 | 25.9 | 5.9 |
| Paper birch | Betula papyrifera Marsh. | EM | Pioneer | Rapid | 1.5 | 30.1 | 4.0 |
| Red ash | Fraxinus pennsylvanica Marsh. | AM | Pioneer | Rapid | 3.1 | 33.8 | 4.8 |
| Red maple | Acer rubrum L. | AM | Pioneer | Rapid | 3.4 | 62.3 | 6.9 |
| Northern red oak | Quercus rubra L. | EM | Non-pioneer | Moderate | 2.8 | 41.8 | 5.7 |
| Yellow birch | Betula alleghaniensis Britt. | EM | Non-pioneer | Slow | 3.2 | 37.0 | 3.8 |
| Sugar maple | Acer saccharum Marsh. | AM | Non-pioneer | Slow | 4.8 | 53.0 | 5.6 |
| Species | Factor | Fall 2010 | Spring 2011 | Fall 2011 | Spring 2012 | Fall 2012 | Spring 2015 |
|---|---|---|---|---|---|---|---|
| Tamarack | Protection | ||||||
| Vegetation | Facilitation * | Facilitation * | Facilitation * | Facilitation * | Facilitation ** | Facilitation ** | |
| Red pine | Protection | ||||||
| Vegetation | |||||||
| Paper birch | Protection | Predation in VG* | Predation ** | Predation ** | |||
| Vegetation | M < BS = VG * | Competition *** | |||||
| Red ash | Protection | ||||||
| Vegetation | |||||||
| Red maple | Protection | Predation ** | |||||
| Vegetation | Competition ** | ||||||
| Red oak | Protection | Predation ** | Predation ** | Predation ** | |||
| Vegetation | Competition ** | Competition ** | Competition *** | Competition *** | Competition *** | ||
| Yellow birch | Protection | Predation *** | Predation *** | Predation *** | Predation *** | ||
| Vegetation | Competition ** | Competition ** | Competition ** | Competition *** | Competition *** | ||
| Sugar maple | Protection | Predation in VG * | |||||
| Vegetation | Facilitation * | Facilitation if Protection * M < BS = VG if No Protection * | M < BS = VG * | M < BS = VG ** | |||
| Species | Mortality Due to Predation (2010–2012) | % Dead Seedlings (Fall 2012) | % dead Seedlings (Spring 2015) | ||||
|---|---|---|---|---|---|---|---|
| Bare Soil | Mulch Mat | Herbaceous Vegetation | Total | Unprotected | Protected | ||
| Tamarack | 0 | 0 | 0 | 0 | 6.8 | 5.8 | 15.2 |
| Red pine | 0 | 0 | 0 | 0 | 0.5 | 4.2 | 15.1 |
| Paper birch | 1.1 a | 8.9 b | 4.7 c | 14.7 | 20.4 a | 9.4 b | 37.7 |
| Red ash | 0 | 2.1 | 0 | 2.1 | 2.1 | 0.5 | 3.1 |
| Red maple | 0 | 0.5 | 1.6 | 2.1 | 4.2 | 1.6 | 11.5 |
| Red oak | 0 a | 2.6 b | 4.2 b | 6.8 | 13.6 a | 5.2 b | 33.2 |
| Yellow birch | 0.5 a | 10.1 b | 6.4 c | 17.0 | 27.5 a | 12.7 b | 60.8 |
| Sugar maple | 0 a | 3.7 b | 2.1 b | 5.8 | 12.1 | 7.9 | 33.5 |
| Species | Leaf Nitrogen (N) | Superficial Bare Roots | ||||
|---|---|---|---|---|---|---|
| BS | M | VG | BS | M | VG | |
| Tamarack | 1.91 | 1.46 | 1.43 | 11.6 x | 1.7 y | 2.3 y |
| Red pine | 1.02 | 1.07 | 0.97 | 12.8 x | 1.1 y | 2.7 z |
| Red ash | 2.30 a | 1.43 b | 1.62 b | 8.1 x | 0 y | 3.8 z |
| Paper birch | 2.28 | 1.84 | 1.93 | 3.4 | 0.7 | 0.7 |
| Red maple | 1.68 a | 1.16 b | 1.23 b | 10.4 x | 0.6 y | 2.7 y |
| Yellow birch | 1.95 | 1.86 | 1.79 | 6.1 x | 0 y | 0 y |
| Red oak | 1.68 a | 1.11 b | 1.24 ab | 9.2 x | 0.6 y | 1.8 y |
| Sugar maple | 1.68 a | 1.13 b | 1.22 b | 1.8 | 0 | 2.4 |
| Species | Adjusted R2 | Soil Water Content | Herb Height | Grasses | Solidago spp. | Asteraceae spp. | Apiaceae spp. | Fabaceae spp. | |
|---|---|---|---|---|---|---|---|---|---|
| Tamarack | RGRHeight | 0.30 | 0.03 * (+) | 0.09 (+) | 0.09 (−) | ||||
| RGRDiameter | 0.28 | 0.01 * (+) | 0.08 (+) | 0.03 * (+) | |||||
| Red pine | RGRHeight | 0.39 | 0.06 (+) | ||||||
| RGRDiameter | 0.14 | 0.02 * (+) | |||||||
| Paper birch | RGRHeight | −0.07 | |||||||
| RGRDiameter | −0.13 | ||||||||
| Red ash | RGRHeight | 0.11 | |||||||
| RGRDiameter | 0.20 | 0.02 * (−) | |||||||
| Red maple | RGRHeight | 0.36 | 0.004 **(+) | 0.04 * (−) | |||||
| RGRDiameter | 0.31 | 0.02 * (+) | 0.09 (−) | ||||||
| Red oak | RGRHeight | 0.40 | 0.04 * (−) | 0.02 * (−) | |||||
| RGRDiameter | −0.05 | ||||||||
| Yellow birch | RGRHeight | 0.03 | |||||||
| RGRDiameter | −0.26 | ||||||||
| Sugar maple | RGRHeight | −0.16 | |||||||
| RGRDiameter | −0.11 | ||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
St-Denis, A.; Kneeshaw, D.; Messier, C. Effect of Predation, Competition, and Facilitation on Tree Survival and Growth in Abandoned Fields: Towards Precision Restoration. Forests 2018, 9, 692. https://doi.org/10.3390/f9110692
St-Denis A, Kneeshaw D, Messier C. Effect of Predation, Competition, and Facilitation on Tree Survival and Growth in Abandoned Fields: Towards Precision Restoration. Forests. 2018; 9(11):692. https://doi.org/10.3390/f9110692
Chicago/Turabian StyleSt-Denis, Annick, Daniel Kneeshaw, and Christian Messier. 2018. "Effect of Predation, Competition, and Facilitation on Tree Survival and Growth in Abandoned Fields: Towards Precision Restoration" Forests 9, no. 11: 692. https://doi.org/10.3390/f9110692
APA StyleSt-Denis, A., Kneeshaw, D., & Messier, C. (2018). Effect of Predation, Competition, and Facilitation on Tree Survival and Growth in Abandoned Fields: Towards Precision Restoration. Forests, 9(11), 692. https://doi.org/10.3390/f9110692




