The Fusarium Circinatum Gene Fcrho1, Encoding a Putative Rho1 GTPase, Is Involved in Vegetative Growth but Dispensable for Pathogenic Development
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Material and Growth Conditions
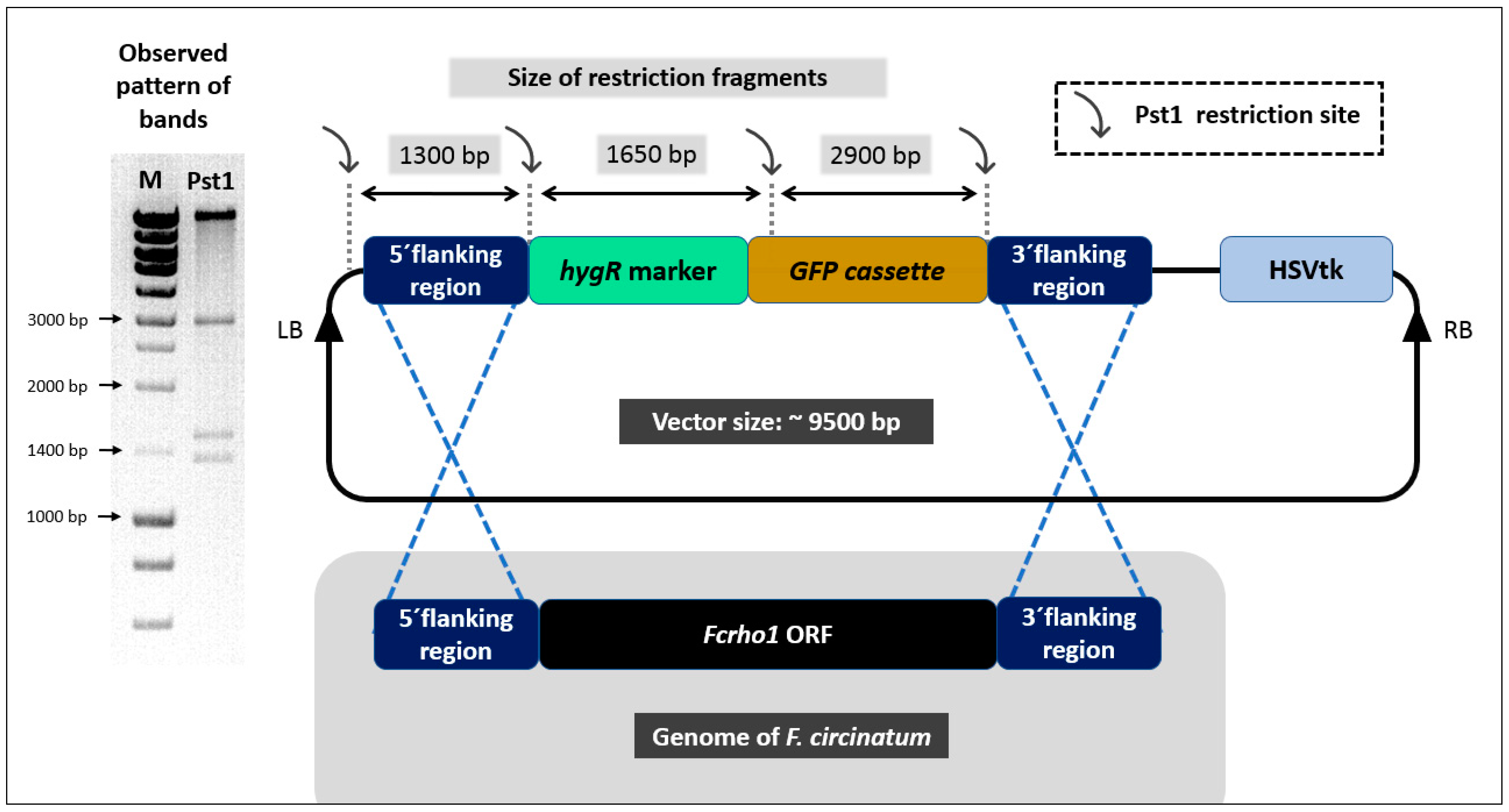
2.2. Generation of Fcrho1 Deletion Construct
2.3. Transformation of F. circinatum with Fcrho1 Deletion Construct
2.4. Confirmation of Deletion Mutants
2.5. Analysis of Hyphal Growth and Conidiation
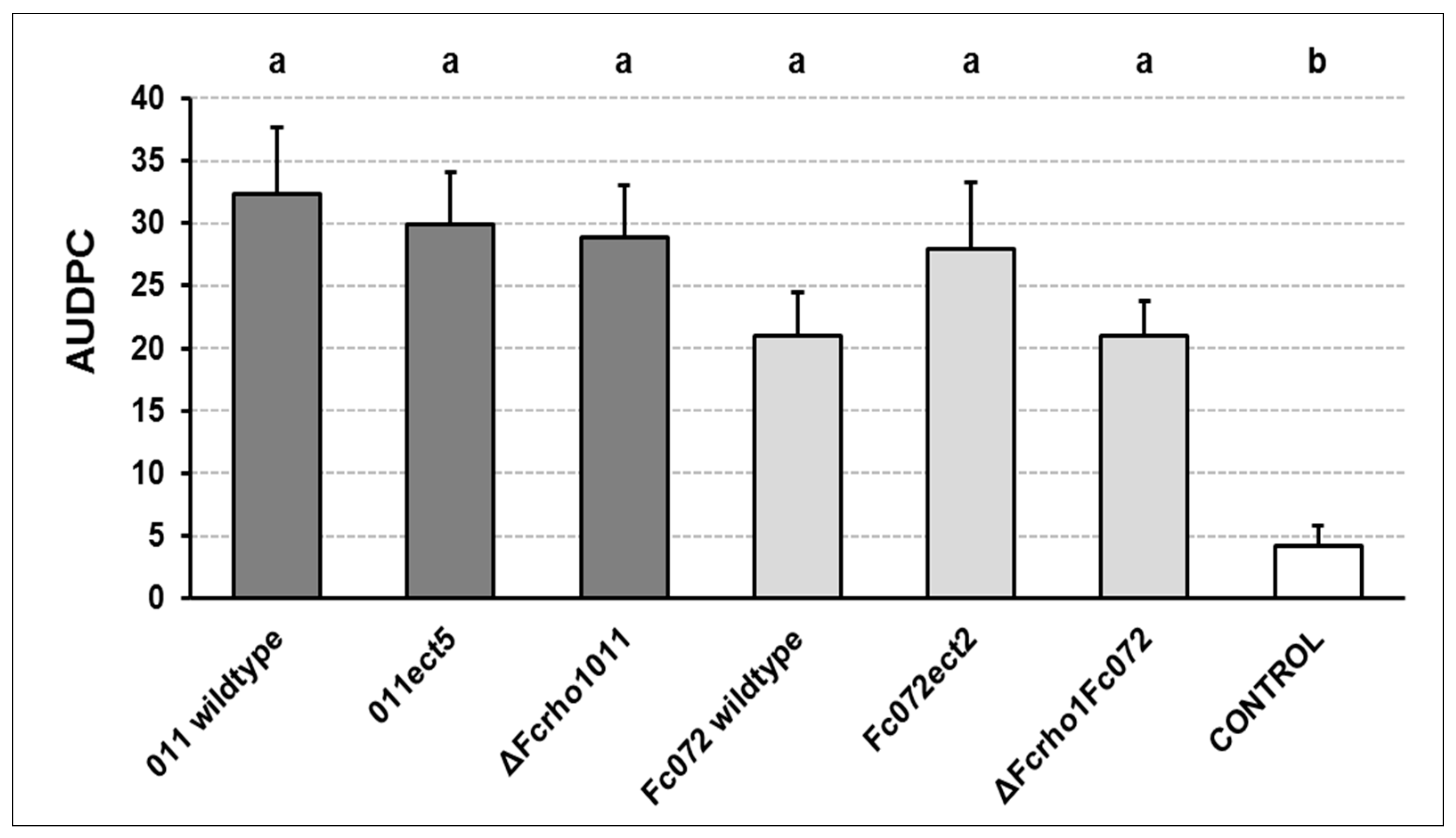
2.6. Pathogenicity Assay
2.7. Statistical Analysis
3. Results and Discussion
3.1. Production of Deletion Mutants Lacking Fcrho1 Open Reading Frame
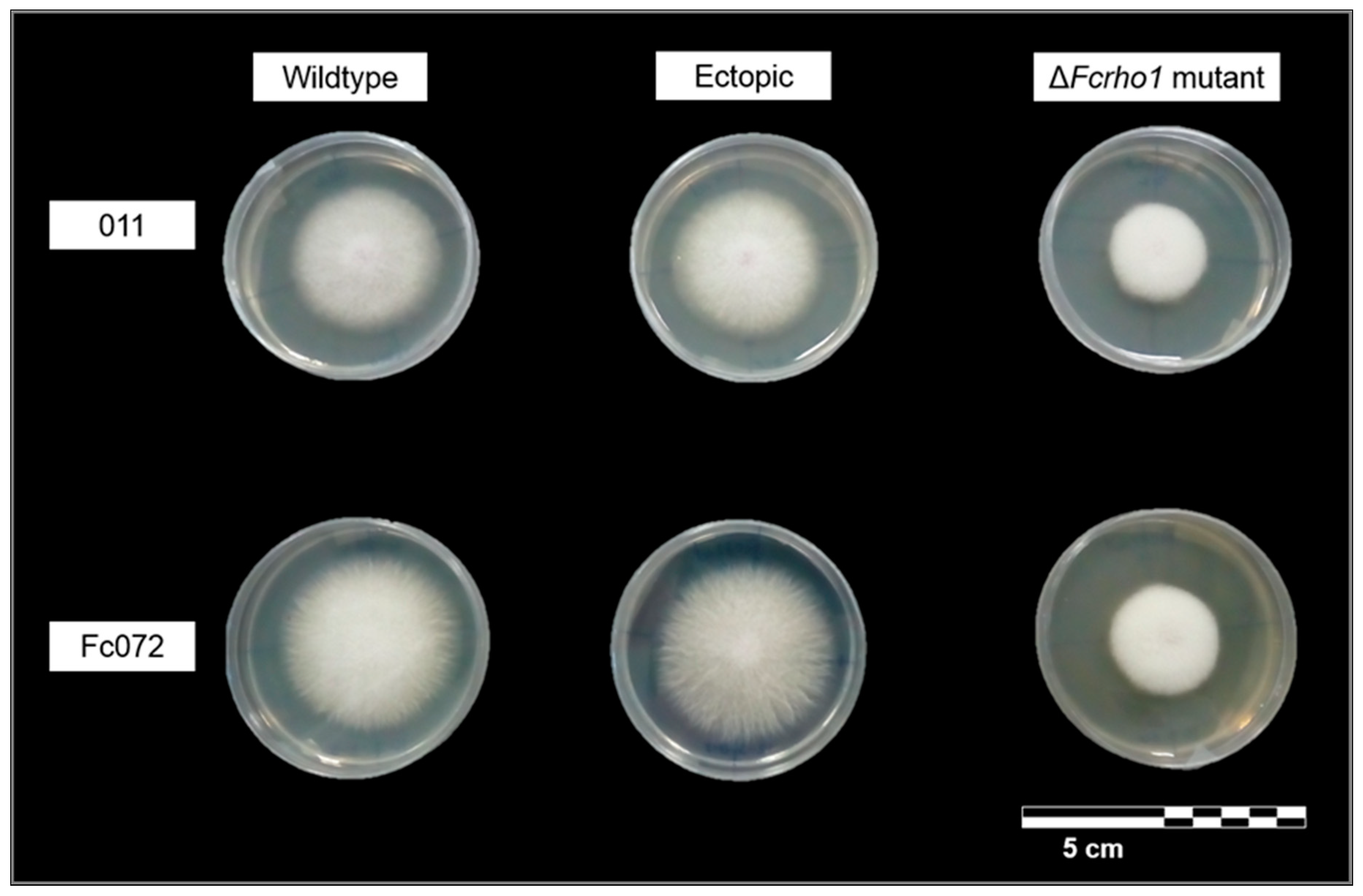
3.2. Phenotypic Characterization of Deletion Mutants
4. Conclusions
- A modified version of the OSCAR protocol for generating a deletion construction in a single cloning step was successfully used in F. circinatum. This method, therefore, proved to be suitable for gene modifications in this phytopathogen. Furthermore, OSCAR was proved to be a useful methodology for the future functional characterization of pathogenicity-related genes in the PPC fungus.
- The GTPase encoding gene Fcrho1 played an important role in regulating the vegetative growth of F. circinatum in a generalist solid medium. This gene seems to be involved in some aspects of vegetative growth such as hyphal tip elongation and aerial mycelium formation.
- Fcrho1 is not essential for developing some functions in the lifecycle of F. circinatum, being deletion mutants able to grow, sporulate and colonize P. radiata seedlings. However, the high susceptibility of this pine species could also participate in the observed virulent development of mutant strains. In consequence, further research is required to clarify whether a lower growth rate in mutant strains could cause attenuated virulence in less susceptible hosts.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Culture Media and Buffers Used in This Article
- Induction medium: 1% v/v K buffer, 2% v/v M-N solution, 1% v/v CaCl2 (1% w/v), 1% v/v FeSO4 (0.01% w/v), 0.5% v/v spore elements (0.1% w/v of ZnSO4·7H2O, H3BO3, CuSO4·5H2O and MnSO4·4H2O), 0.25% v/v NH4NO3 (20% w/v), 1% v/v glycerol (50% v/v), 4% v/v MES (1 M) and 0.5% w/v glucose (2 M). Adjust the medium at pH = 6.
- LB: 10% w/v triptone, 5% w/v yeast extract and 10% w/v NaCl (99% w/v).
- Minimum medium: 0.1% v/v K buffer (20% w/v K2HPO4 and 14.5% w/v KH2PO4; pH = 7), 2% M-N solution (3% w/v MgSO4·7H2O and 1.5% w/v NaCl), 0.01% v/v CaCl2 (10% w/v), 1% v/v FeSO4 (0.01% w/v), 0.5% v/v spore elements, 2.5% v/v NH4NO3 (20% w/v), 0.1% w/v glucose (2 M).
- PDA: 3.9% w/v potato dextrose agar.
- PDB: 2.8% w/v potato dextrose broth.
- Selection medium: 3.9% w/v potato dextrose agar, 0.05% v/v hygromycin B, 0.2% v/v cefotaxim, 0.1% v/v moxalactum and 0.06% w/v streptomycin.
- TENS: 1% v/v NaOH (10 N), 5% v/v SDS (10% v/v), 1% Tris base (1 M; pH = 8) and 0.25% v/v EDTA (0.5 M; pH = 8).
- YEPS: 1% w/v yeast extract, 2% w/v bacto-peptone and 2% w/v sucrose.
References
- Wingfield, M.J.; Hammerbacher, A.; Ganley, R.J.; Steenkamp, E.T.; Gordon, T.R.; Wingfield, B.D.; Coutinho, T.A. Pitch canker caused by Fusarium circinatum—A growing threat to pine plantations and forests worldwide. Australas. Plant Pathol. 2008, 37, 319–334. [Google Scholar] [CrossRef]
- Bezos, D.; Martínez-Álvarez, P.; Fernández, M.; Diez, J.J. Epidemiology and management of Pine Pitch Canker Disease in Europe—A review. Balt. For. 2017, 23, 279–293. [Google Scholar]
- Muñoz-Adalia, E.J.; Fernández, M.; Wingfield, B.D.; Diez, J.J. In silico annotation of five candidate genes associated with pathogenicity in Fusarium circinatum. For. Pathol. 2018, 48. [Google Scholar] [CrossRef]
- Wingfield, B.D.; Steenkamp, E.T.; Santana, Q.C.; Coetzee, M.P.A.; Bam, S.; Barnes, I.; Beukes, C.W.; Chan, W.Y.; de Vos, L.; Fourie, G.; et al. First fungal genome sequence from Africa: A preliminary analysis. S. Afr. J. Sci. 2012, 108, 1–9. [Google Scholar] [CrossRef]
- Araujo-Palomares, C.L.; Richthammer, C.; Seiler, S.; Castro-Longoria, E. Functional characterization and cellular dynamics of the CDC-42-RAC-CDC-24 module in Neurospora crassa. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Iden, S.; Collard, J.G. Crosstalk between small GTPases and polarity proteins in cell polarization. Nat. Rev. Mol. Cell Biol. 2008, 9, 846–859. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zheng, W.; Zheng, S.; Zhang, D.; Sang, W.; Chen, X.; Li, G.; Lu, G.; Wang, Z. Rac1 is required for pathogenicity and Chm1-dependent conidiogenesis in rice fungal pathogen Magnaporthe grisea. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Zhao, Z.; Chen, J.; Liu, W.; Ke, H.; Zhou, J.; Lu, G.; Darvill, A.G.; Albersheim, P.; Wu, S.; et al. A Cdc42 ortholog is required for penetration and virulence of Magnaporthe grisea. Fungal Genet. Biol. 2009, 46, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Rolke, Y.; Tudzynski, P. The small GTPase Rac and the p21-activated kinase Cla4 in Claviceps purpurea: Interaction and impact on polarity, development and pathogenicity. Mol. Microbiol. 2008, 68, 405–423. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Rocha, A.L.; Roncero, M.I.G.; López-Ramirez, A.; Mariné, M.; Guarro, J.; Martínez-Cadena, G.; Di Pietro, A. Rho1 has distinct functions in morphogenesis, cell wall biosynthesis and virulence of Fusarium oxysporum. Cell. Microbiol. 2008, 10, 1339–1351. [Google Scholar] [CrossRef] [PubMed]
- Bluhm, B.H.; Zhao, X.; Flaherty, J.E.; Xu, J.-R.; Dunkle, L.D. RAS2 Regulates Growth and Pathogenesis in Fusarium graminearum. Mol. Plant-Microbe Interact. 2007, 20, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, Y.; Wang, J.; Zhai, Z.; Zhang, L.; Zheng, W.; Zheng, W.; Yu, W.; Zhou, J.; Lu, G.; et al. Functional characterization of Rho family small GTPases in Fusarium graminearum. Fungal Genet. Biol. 2013, 61, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Vainio, E.J.; Martínez-Álvarez, P.; Bezos, D.; Hantula, J.; Diez, J.J. Fusarium circinatum isolates from northern Spain are commonly infected by three distinct mitoviruses. Arch. Virol. 2015, 160, 2093–2098. [Google Scholar] [CrossRef] [PubMed]
- Paz, Z.; García-Pedrajas, M.D.; Andrews, D.L.; Klosterman, S.J.; Baeza-Montañez, L.; Gold, S.E. One Step Construction of Agrobacterium-Recombination-ready-plasmids (OSCAR), an efficient and robust tool for ATMT based gene deletion construction in fungi. Fungal Genet. Biol. 2011, 48, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Gold, S.E.; Paz, Z.; García-Pedrajas, M.D.; Glenn, A. Rapid Deletion Production in Fungi via Agrobacterium Mediated Transformation of OSCAR Deletion Constructs. J. Vis. Exp. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [PubMed]
- García-Pedrajas, M.D.; Paz, Z.; Andrews, D.L.; Baeza-Montañez, L.; Gold, S.E. Rapid Deletion Plasmid Construction Methods for Protoplast and Agrobacterium-based Fungal Transformation Systems. In Laboratory Protocols in Fungal Biology: Current Methods in Fungal Biology; Gupta, V.K., Tuohy, M.G., Eds.; Springer: Berlin, Germany, 2013; pp. 375–394. [Google Scholar]
- Khang, C.H.; Park, S.Y.; Lee, Y.H.; Kang, S. A dual selection based, targeted gene replacement tool for Magnaporthe grisea and Fusarium oxysporum. Fungal Genet. Biol. 2005, 42, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Sarmiento-Villamil, J.L.; Prieto, P.; Klosterman, S.J.; García-Pedrajas, M.D. Characterization of two homeodomain transcription factors with critical but distinct roles in virulence in the vascular pathogen Verticillium dahliae. Mol. Plant Pathol. 2018, 19, 986–1004. [Google Scholar] [CrossRef] [PubMed]
- Hellens, R.; Mullineaux, P.; Klee, H. A guide to Agrobacterium binary Ti vectors. Trends Plant Sci. 2000, 5, 446–451. [Google Scholar] [CrossRef]
- Vainio, E.J.; Korhonen, K.; Hantula, J. Genetic variation in Phlebiopsis gigantea as detected with random amplified microsatellite (RAMS) markers. Mycol. Res. 1998, 102, 187–192. [Google Scholar] [CrossRef]
- Correll, J.C.; Gordon, T.R.; McCain, A.H.; Fox, J.W.; Koehler, C.S.; Wood, D.L.; Schultz, M.E. Pitch Canker Disease in California–Pathogenicity, Distribution, and Canker Development on Monterey Pine (Pinus radiata). Plant Dis. 1991, 75, 676–682. [Google Scholar] [CrossRef]
- Martínez-Álvarez, P.; Pando, V.; Diez, J.J. Alternative species to replace Monterey pine plantations affected by pitch canker caused by Fusarium circinatum in northern Spain. Plant Pathol. 2014, 63, 1086–1094. [Google Scholar] [CrossRef]
- Muñoz-Adalia, E.J.; Flores-Pacheco, J.A.; Martínez-Álvarez, P.; Martín-García, J.; Fernández, M.; Diez, J.J. Effect of mycoviruses on the virulence of Fusarium circinatum and laccase activity. Physiol. Mol. Plant Pathol. 2016, 94, 8–15. [Google Scholar] [CrossRef]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing Professional: Ames, IA, USA, 2006; p. 388. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Development Core Team: Vienna, Austria, 2015; Volume 55, pp. 275–286. [Google Scholar]
- Signorell, A.; Aho, K.; Anderegg, N.; Aragon, T.; Arppe, A.; Baddeley, A.; Bolker, B.; Caeiro, F.; Champely, S.; Chessel, D.; et al. DescTools: Tools for Descriptive Statistics. 2015. Available online: https://cran.r-project.org/web/packages/DescTools/index.html (accessed on 20 April 2018).
- Flores-Pacheco, J.A.; Muñoz-Adalia, E.J.; Martínez-Álvarez, P.; Pando, V.; Diez, J.J.; Martín-García, J. Effect of mycoviruses on growth, spore germination and pathogenicity of the fungus Fusarium circinatum. For. Syst. 2017, 26. [Google Scholar] [CrossRef]
- Kwon, M.J.; Arentshorst, M.; Roos, E.D.; Van Den Hondel, C.A.M.J.J.; Meyer, V.; Ram, A.F.J. Functional characterization of Rho GTPases in Aspergillus niger uncovers conserved and diverged roles of Rho proteins within filamentous fungi. Mol. Microbiol. 2011, 79, 1151–1167. [Google Scholar] [CrossRef] [PubMed]
- Martín-Rodrigues, N.; Espinel, S.; Sanchez-Zabala, J.; Ortíz, A.; González-Murua, C.; Duñabeitia, M.K. Spatial and temporal dynamics of the colonization of Pinus radiata by Fusarium circinatum, of conidiophora development in the pith and of traumatic resin duct formation. New Phytol. 2013, 198, 1215–1227. [Google Scholar] [CrossRef] [PubMed]
- Iturritxa, E.; Ganley, R.J.; Raposo, R.; García-Serna, I.; Mesanza, N.; Kirkpatrick, S.C.; Gordon, T.R. Resistance levels of Spanish conifers against Fusarium circinatum and Diplodia pinea. For. Pathol. 2013, 43, 488–495. [Google Scholar] [CrossRef]
| Primer Pair | Sequences (5′-3′) | Size | Region |
|---|---|---|---|
| FcirRHO1j | TATGCGTTGATGCTCTGAGG | 855 nt | Exon of Fcrho1 |
| FcirRHO2q | TCCAGCTCTCCAAGCTCTTC | ||
| HygF_Oscar | AAAGCCTGAACTCACCGCGACG | 736 nt | hygR marker |
| HygR_Oscar | AGCGCGTCTGCTGCTCCATAC | ||
| FRHOl1j-attB2r | GGGGACAGCTTTCTTGTACAAAGTGGAAAATTGCCATGGTACGAGAGGCT | 940 nt | 5′ flanking region of Fcrho1 |
| FRHOl2q-attB1r | GGGGACTGCTTTTTTGTACAAACTTGTTTAAGGTTCCATGCCCAAGAGG | ||
| FRHOr1j-attB4 | GGGGACAACTTTGTATAGAAAAGTTGTTAACATGATACCCAGCTCCATCG | 873 nt | 3′ flanking region of Fcrho1 |
| FRHOr2q-attB3 | GGGGACAACTTTGTATAATAAAGTTGTGATCCCGCTGACGTGAGTATTG |
| Isolate | Strain | G (mm2/day) | Ac (mm2) | C (Spores/mm2) | N (Germlings/mm2) |
|---|---|---|---|---|---|
| 011 | wildtype | 764.55 ± 76.04 a | 4553.73 ± 251.98 a | 525.00 ± 134.49 a | 108.33 ± 27.78 a |
| 011ect5 | 744.38 ± 41.38 a | 4390.89 ± 215.89 a | 508.33 ± 83.44 a | 83.33 ± 13.28 a | |
| ΔFcrho1011 | 331.89 ± 30.72 b | 1782.85 ± 35.79 b | 393.33 ± 73.49 a | 105.00 ± 17.62 a | |
| Fc072 | wildtype | 793.53 ± 91.55 a | 4643.27 ± 184.33 a | 353.33 ± 44.17 a | 85.00 ± 12.63 a |
| Fc072ect2 | 834.44 ± 63.68 a | 4791.97 ± 234.82 a | 370.00 ± 61.63 a | 71.66 ± 15.01 a | |
| ΔFcrho1Fc072 | 302.85 ± 50.27 b | 1824.74 ± 89.04 b | 451.66 ± 71.10 a | 73.33 ± 9.90 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Adalia, E.J.; Cañizares, M.C.; Fernández, M.; Diez, J.J.; García-Pedrajas, M.D. The Fusarium Circinatum Gene Fcrho1, Encoding a Putative Rho1 GTPase, Is Involved in Vegetative Growth but Dispensable for Pathogenic Development. Forests 2018, 9, 684. https://doi.org/10.3390/f9110684
Muñoz-Adalia EJ, Cañizares MC, Fernández M, Diez JJ, García-Pedrajas MD. The Fusarium Circinatum Gene Fcrho1, Encoding a Putative Rho1 GTPase, Is Involved in Vegetative Growth but Dispensable for Pathogenic Development. Forests. 2018; 9(11):684. https://doi.org/10.3390/f9110684
Chicago/Turabian StyleMuñoz-Adalia, E. Jordán, M. Carmen Cañizares, Mercedes Fernández, Julio J. Diez, and M. Dolores García-Pedrajas. 2018. "The Fusarium Circinatum Gene Fcrho1, Encoding a Putative Rho1 GTPase, Is Involved in Vegetative Growth but Dispensable for Pathogenic Development" Forests 9, no. 11: 684. https://doi.org/10.3390/f9110684
APA StyleMuñoz-Adalia, E. J., Cañizares, M. C., Fernández, M., Diez, J. J., & García-Pedrajas, M. D. (2018). The Fusarium Circinatum Gene Fcrho1, Encoding a Putative Rho1 GTPase, Is Involved in Vegetative Growth but Dispensable for Pathogenic Development. Forests, 9(11), 684. https://doi.org/10.3390/f9110684






