The Potential Distribution of Tree Species in Three Periods of Time under a Climate Change Scenario
Abstract
1. Introduction
2. Materials and Methods
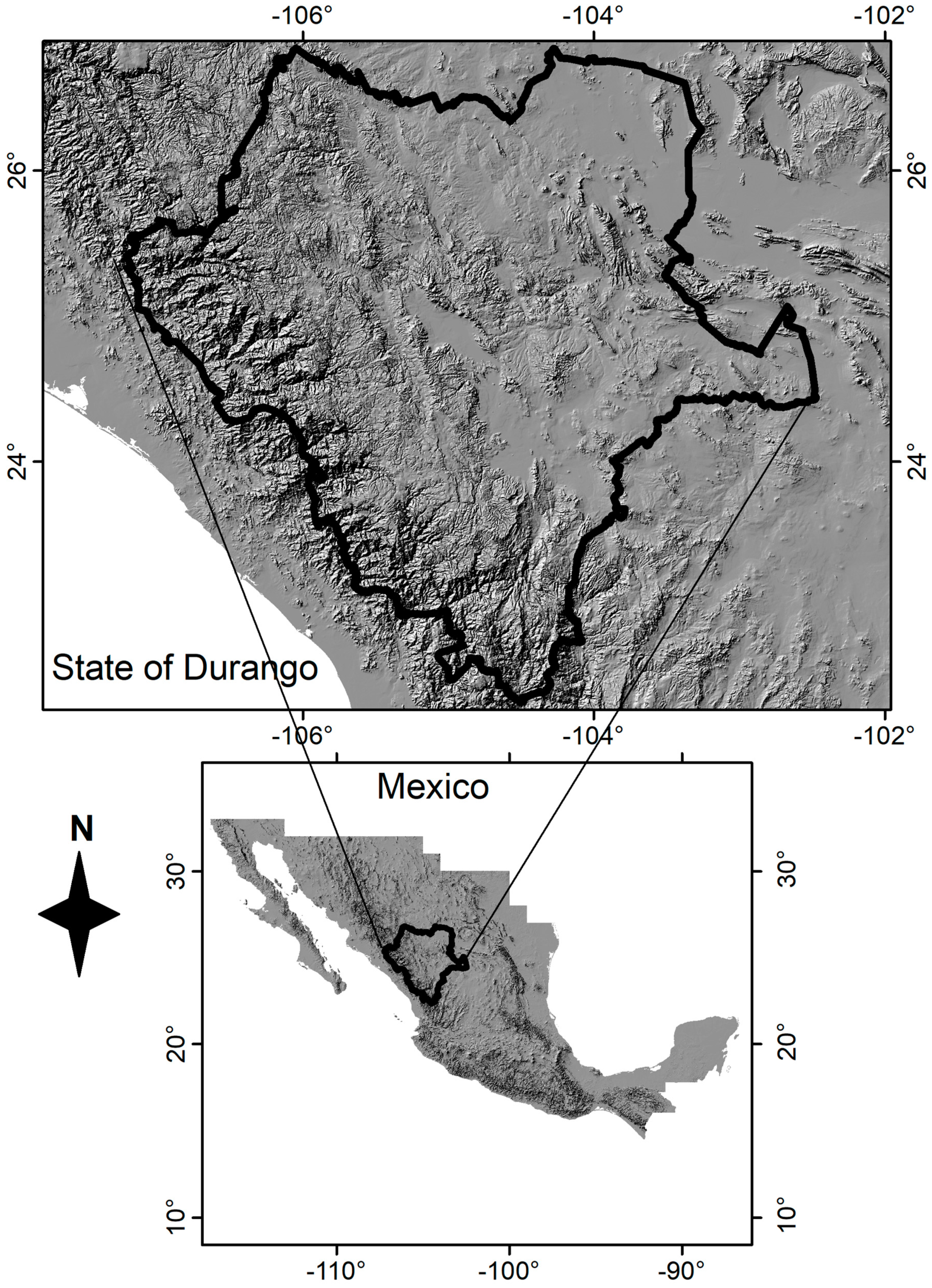
2.1. Study Region
2.2. Obtaining Data
2.3. Distribution Modeling
2.4. Model Evaluation
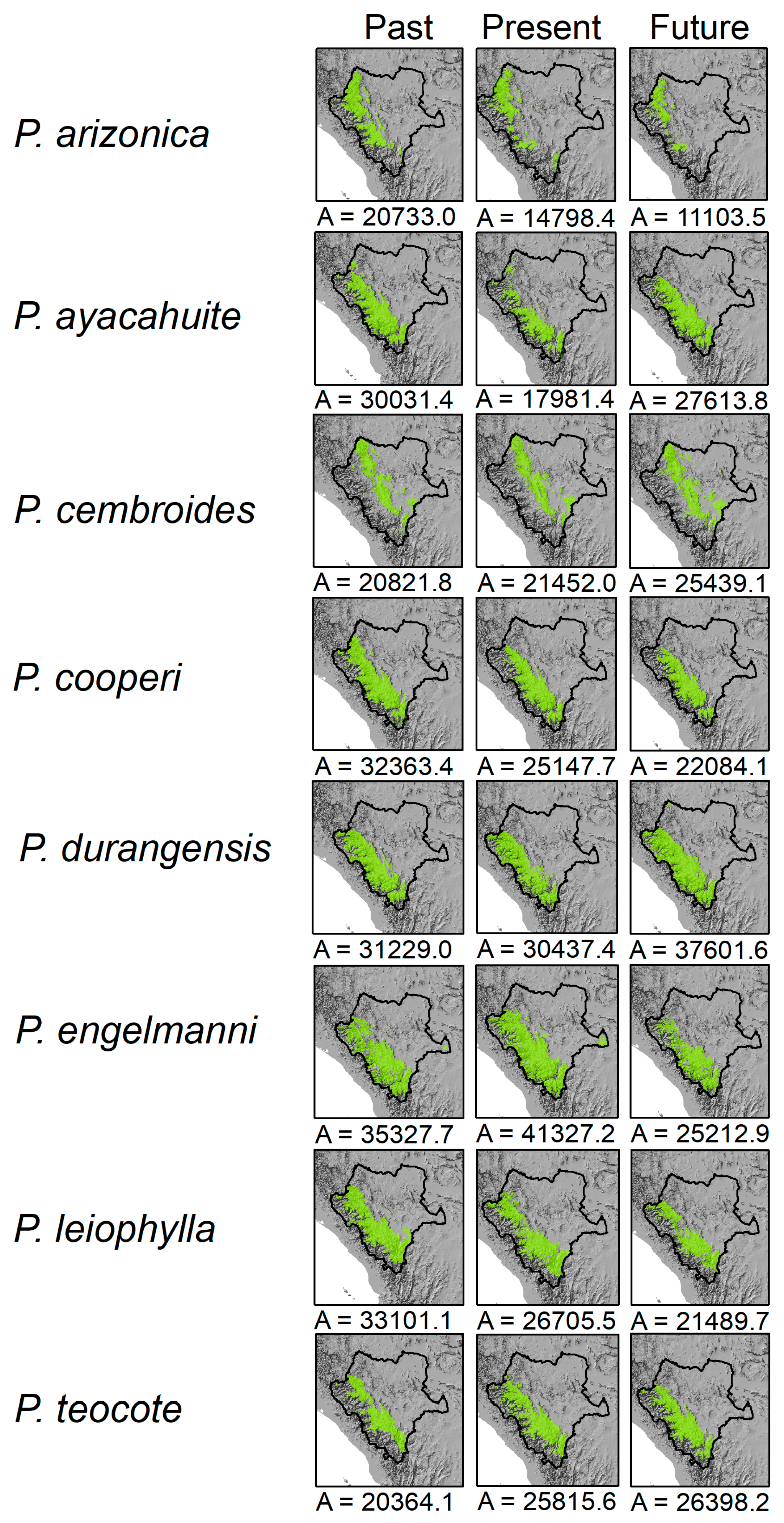
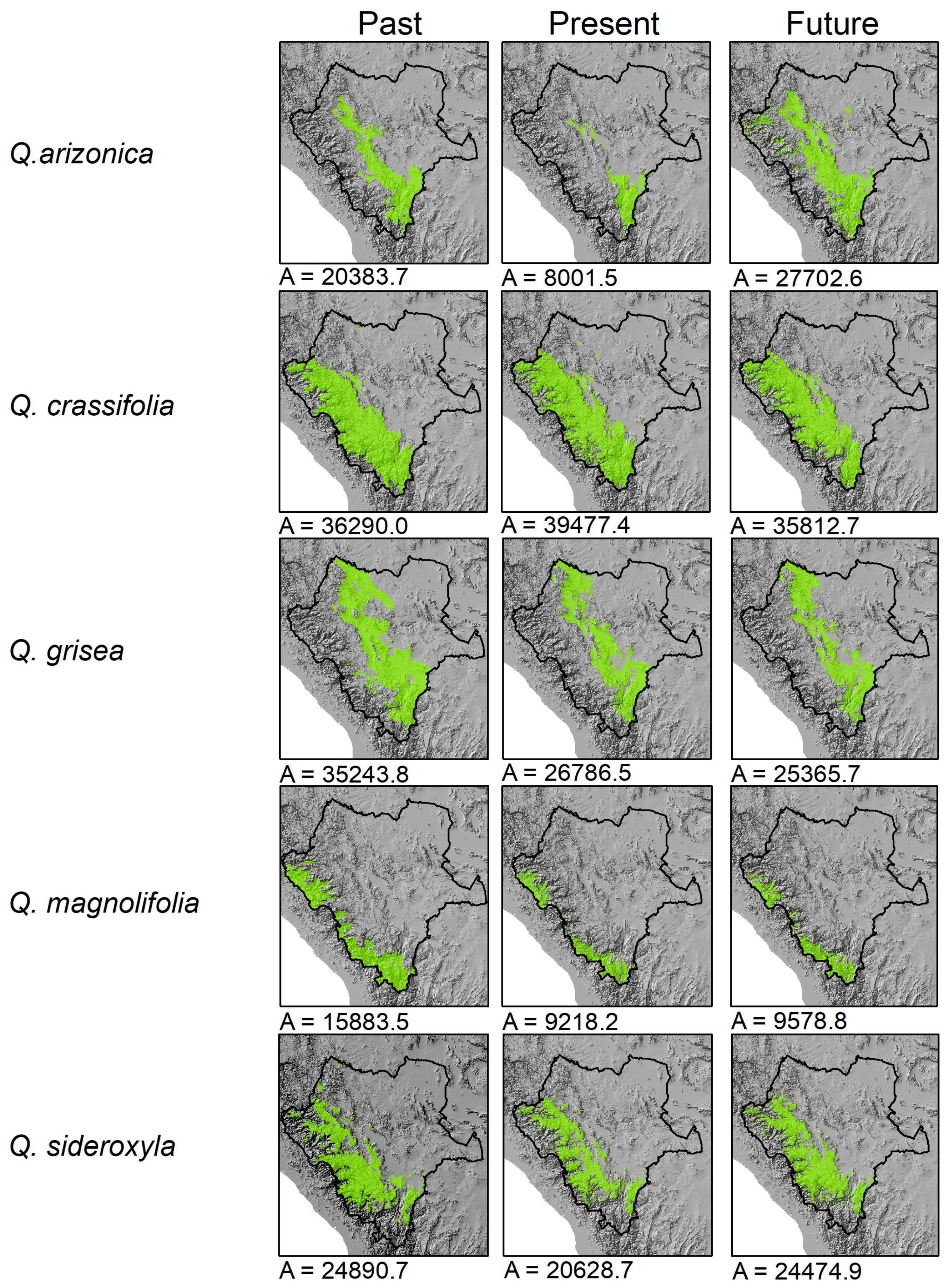
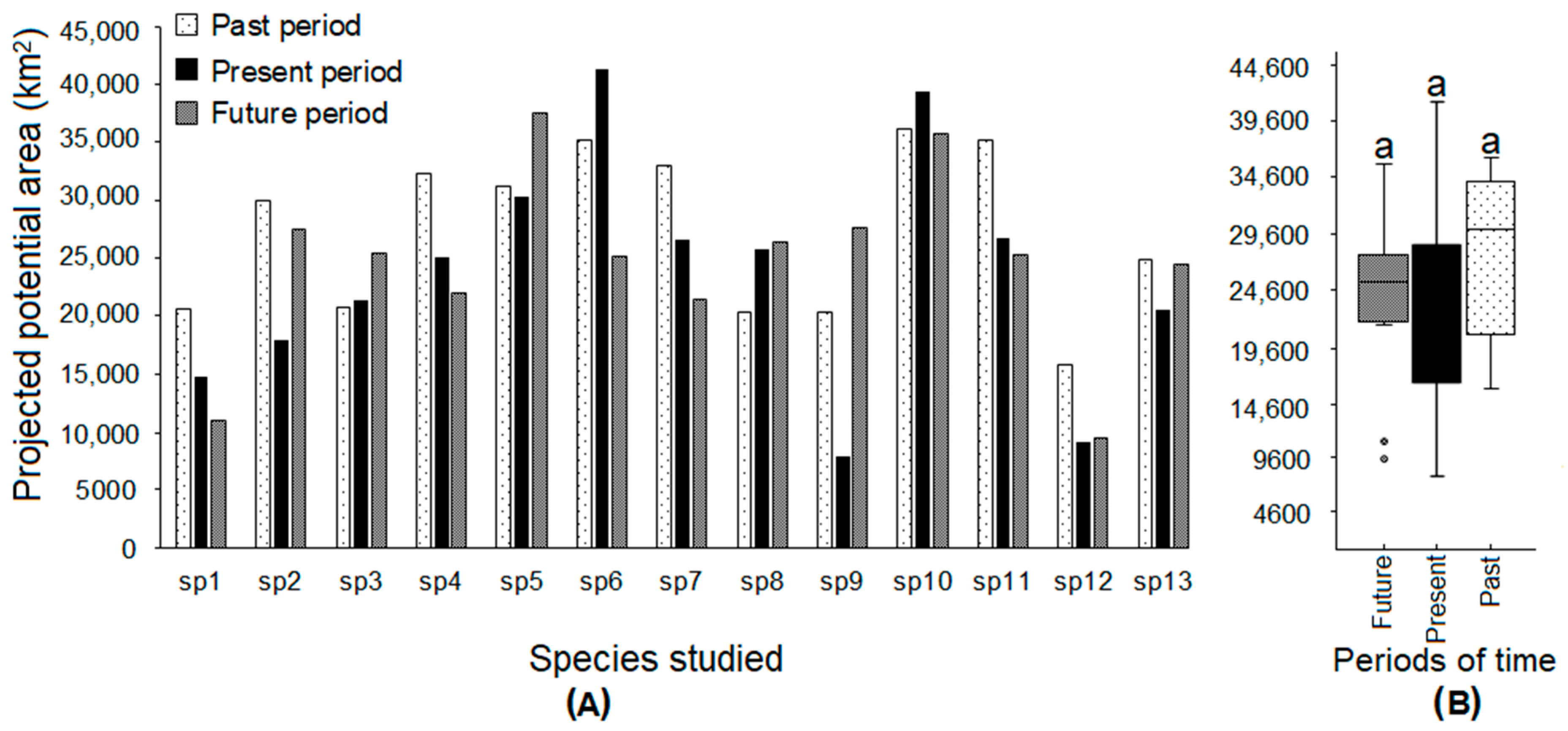
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Soberón, J.; Nakamura, M. Niches and distributional areas: Concepts, methods, and assumptions. Proc. Natl. Acad. Sci. USA 2009, 106, 19644–19650. [Google Scholar] [CrossRef] [PubMed]
- Crowther, T.W.; Glick, H.B.; Covey, K.R.; Bettigole, C.; Maynard, D.S.; Thomas, S.M.; Tuanmu, M.N. Mapping tree density at a global scale. Nature 2015, 525, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Perosa, M.; Rojas, J.F.; Villagra, P.E.; Tognelli, M.F.; Carrara, R.; Alvarez, J.A. Distribución potencial de los bosques de Prosopis flexuosa en la Provincia Biogeográfica del Monte (Argentina). Ecol. Austral. 2014, 24, 238–248. [Google Scholar]
- Martínez-Antúnez, P.; Hernández-Díaz, J.C.; Wehenkel, C.; López-Sánchez, C.A. Estimación de la densidad de especies de coníferas a partir de variables ambientales. Madera Bosques 2015, 21, 23–33. [Google Scholar] [CrossRef]
- Sánchez-Cordero, V.; Cirelli, V.; Murguíal, M.; Sarkar, S. Place prioritization for biodiversity representation using species ecological niche modelling. Biodivers. Inform. 2005, 2, 11–23. [Google Scholar] [CrossRef]
- Rodríguez, F.J.; Pereda, M.E. La dinámica espacial de los ecosistemas del estado de Durango. Ra Ximhai 2012, 8, 91–96. [Google Scholar]
- Calderón-Aguilera, L.E.; Rivera-Monroy, V.H.; Porter-Bolland, L.; Martínez-Yrízar, A.; Ladah, L.B.; Martínez-Ramos, M.; Alcocer, J.; Santiago-Pérez, A.L.; Hernandez-Arana, H.A.; Reyes-Gómez, V.M.; et al. An assessment of natural and human disturbance effects on Mexican ecosystems: Current trends and research gaps. Biodivers. Conserv. 2012, 21, 589–617. [Google Scholar] [CrossRef]
- Galicia, L.; Potvin, C.; Messier, C. Maintaining the high diversity of pine and oak species in Mexican temperate forests: A new management approach combining functional zoning and ecosystem adaptability. Can. J. For. Res. 2015, 45, 1358–1368. [Google Scholar] [CrossRef]
- Thuiller, W.; Albert, C.; Araújo, M.B.; Berry, P.M.; Cabeza, M.; Guisan, A.; Sykes, M.T. Predicting global change impacts on plant species’ distributions: Future challenges. Perspect. Plant. Ecol. 2008, 9, 137–152. [Google Scholar] [CrossRef]
- Sykes, M.T.; Prentice, I.C.; Cramer, W. A bioclimatic model for the potential distributions of north European tree species under present and future climates. J. Biogeogr. 1996, 23, 203–233. [Google Scholar]
- Kearney, M.; Porter, W.P. Mechanistic niche modelling: Combining physiological and spatial data to predict species’ ranges. Ecol. Lett. 2009, 12, 334–350. [Google Scholar] [CrossRef] [PubMed]
- Hanson, H.C.; Churchill, E.D. The Plant Community; Reinhold Publishing Corp.: New York, NY, USA, 1961; pp. 1–218. [Google Scholar]
- Chapman, S.B. Methods in Plant Ecology; Blackwell Scientific: Oxford, UK, 1976; pp. 1–580. [Google Scholar]
- Guisan, A.; Thuiller, W. Predicting species distribution: Offering more than simple habitat models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Anderson, R.P.; Peterson, A.T.; Gómez-Laverde, M. Using niche-based GIS modeling to test geographic predictions of competitive exclusion and competitive release in South American pocket mice. Oikos 2002, 98, 3–16. [Google Scholar] [CrossRef]
- Suárez-Mota, M.E.; Villaseñor, J.L.; López-Mata, L. La región del Bajío, México y la conservación de su diversidad florística. Rev. Mex. Biod. 2015, 86, 799–808. [Google Scholar] [CrossRef]
- Suárez-Mota, M.E.; Ortiz, E.; Villaseñor, J.L.; Espinosa-Garcia, F.J. Ecological niche modeling of invasive plant species according to invasion status and management needs: The case of Chromolaena odorata (Asteraceae) in South Africa. Pol. J. Ecol. 2016, 64, 369–383. [Google Scholar] [CrossRef]
- Townsend, P.; Klusa, D.A. New distributional modelling approaches for gap analysis. Animal Conservation. Zool. Soc. Lond. 2003, 6, 47–54. [Google Scholar] [CrossRef]
- Leal-Nares, O.; Mendoza, M.E.; Pérez-Salicrup, D.; Geneletti, D.; López-Granados, E.; Carranza, E. Distribución potencial del Pinus martinezii: Un modelo espacial basado en conocimiento ecológico y análisis multicriterio. Rev. Mex. Biodivers. 2012, 83, 1152–1170. [Google Scholar] [CrossRef]
- Fourcade, Y.; Engler, J.O.; Rödder, D.; Secondi, J. Mapping species distributions with MAXENT using a geographically biased sample of presence data: A performance assessment of methods for correcting sampling bias. PLoS ONE 2014, 9, e97122. [Google Scholar] [CrossRef] [PubMed]
- Chefaoui, R.M.; Hortal, J.; Lobo, J.M. Potential distribution modelling, niche characterization and conservation status assessment using GIS tools: A case study of Iberian Copris species. Biol. Conserv. 2005, 122, 327–338. [Google Scholar] [CrossRef]
- Rzedowski, J. Vegetación de México; Limusa: México, D.F., Mexico, 1978; pp. 1–432. [Google Scholar]
- Silva-Flores, R.; Pérez-Verdín, G.; Wehenkel, C. Patterns of tree species diversity in relation to climatic factors on the Sierra Madre Occidental, Mexico. PLoS ONE 2014, 9, 105034. [Google Scholar] [CrossRef] [PubMed]
- CONAFOR (Comisión Nacional Forestal). Manual and Procedures for Field Sampling—National Forest and Soil Inventory. 2009. Available online: http://www.snieg.mx/contenidos/espanol/iin/Acuerdo_3_X/Manual_y_Procedimientos_para_el_Muestreo_de_Campo_INFyS_2004–2009.pdf (accessed on 17 November 2017).
- Antúnez, P.; Wehenkel, C.; López-Sánchez, C.A.; Hernández-Díaz, J.C. The role of climatic variables for estimating probability of abundance of tree species. Pol. J. Ecol. 2017, 65, 324–338. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Clim. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. Worldclim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Cuervo-Robayo, A.; Téllez-Valdés, O.; Gómez, M.A.; Venegas-Barrera, C.S.; Manjarrez, J.F.; Mártinez-Meyer, E. An update of high-resolution monthly climate surface for Mexico. Int. J. Climatol. 2013, 34, 2427–3437. [Google Scholar] [CrossRef]
- Cruz-Cárdenas, G.; López-Mata, L.; Villaseñor, J.L.; Ortiz, E. Potential species distribution modeling and the use of principal component analysis as predictor variables. Rev. Mex. Biodiver. 2014, 85, 189–199. [Google Scholar] [CrossRef]
- Nakicenovic, N.; Swart, R. IPCC: Special Report on Emissions Scenarios; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Elith, J.; Graham, J.C.H.; Anderson, P.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Peterson, A.T.; Soberón, J.; Pearson, R.G.; Anderson, R.; Martínez-Meyer, E.; Nakamura, M.; Araujo, M. Ecological Niches and Geographic Distributions; Princeton University Press: Princeton, NJ, USA, 2011; pp. 1–314. [Google Scholar]
- Phillips, S.J.; Dudik, M. Modeling of species distributions with MaxEnt: New extensions and a comprehensive evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Phillips, S.J. Transferability, sample selection bias and background data in presenceonly modelling: A response to Peterson et al. (2007). Ecography 2008, 31, 272–278. [Google Scholar] [CrossRef]
- Pearson, R.G.; Raxworthy, C.J.; Nakamura, M.; Townsend Peterson, A. Predicting species distributions from small numbers of occurrence records: A test case using cryptic geckos in Madagascar. J. Biogeogr. 2007, 34, 102–117. [Google Scholar] [CrossRef]
- Jolliffe, I.T. Principal component analysis and factor analysis. In Principal Component Analysis; Springer: New York, NY, USA, 1986; pp. 115–128. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. 2018. Available online: https://www.r-project.org/ (accessed on 1 January 2017).
- Kuhn, G.M. The phi coefficient as an index of ear differences in dichotic listening. Cortex 1973, 9, 450–457. [Google Scholar] [CrossRef]
- González-Elizondo, M.S.; González-Elizondo, M.; González, L.R.; Enríquez, I.L.; Rentería, F.R.; Flores, J.T. Ecosystems and diversity of the Sierra Madre Occidental. In Merging Science and Management in a Rapidly Changing World: Biodiversity and Management of the Madrean Archipelago III and 7th Conference on Research and Resource Management in the Southwestern Deserts, Tucson, AZ, USA, 1–5 May 2012; Gottfried, G.J., Ffolliott, P.F., Gebow, B.S., Eskew, L.G., Collins, L.C., Eds.; Proceedings RMRS-P-67; US Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2013; pp. 204–211. [Google Scholar]
- Toledo, V.M.; Ordóñez, M. The biodiversity scenario of Mexico: A review of terrestrial habitats. In Biological Diversity of Mexico. Origins and Distribution; Ramamoorthy, T.P., Bye, R., Lot, A., Eds.; Oxford University Press: New York, NY, USA, 1993; pp. 757–777. [Google Scholar]
- Challenger, A. Utilización y Conservación de los Ecosistemas Terrestres de México: Pasado, Presente y futuro; Conabio-Instituto de Biología, UNAM-Agrupación Sierra Madre SC. Distrito Federal: Tlalpan, Mexico, 1998. [Google Scholar]
- Guisan, A.; Zimmermann, N.E. Predictive habitat distribution models in ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- Barve, N.; Barve, V.; Jiménez-Valverde, A.; Lira-Noriega, A.; Maher, S.P.; Peterson, A.T.; Villalobos, F. The crucial role of the accessible area in ecological niche modeling and species distribution modeling. Ecol. Model. 2011, 222, 1810–1819. [Google Scholar] [CrossRef]
- Loiselle, B.A.; Howell, C.A.; Graham, C.H.; Goerck, J.M.; Brooks, T.; Smith, K.G.; Williams, P.H. Avoiding pitfalls of using species distribution models in conservation planning. Conserv. Biol. 2003, 17, 1591–1600. [Google Scholar] [CrossRef]
- Araújo, M.B.; Pearson, R.G.; Thuiller, W.; Erhard, M. Validation of species–climate impact models under climate change. Glob. Chang. Biol. 2005, 11, 1504–1513. [Google Scholar] [CrossRef]
- Carneiro, L.R.D.A.; Lima, A.P.; Machado, R.B.; Magnusson, W.E. Limitations to the use of species-distribution models for environmental-impact assessments in the Amazon. PLoS ONE 2016, 11, e0146543. [Google Scholar] [CrossRef] [PubMed]
- Kadmon, R.; Farber, O.; Danin, A. A systematic analysis of factors affecting the performance of climatic envelope models. Ecol. Appl. 2003, 13, 853–867. [Google Scholar] [CrossRef]
- Ward, G.; Hastie, T.; Barry, S.; Elith, J.; Leathwick, J.R. Presence-only data and the EM algorithm. Biometrics 2009, 65, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Walker, P.A.; Cocks, K.D. HABITAT: A procedure for modelling a disjoint environmental envelope for a plant or animal species. Glob. Ecol. Biogeogr. Lett. 1991, 1, 108–118. [Google Scholar] [CrossRef]
- Hunter, P. The human impact on biological diversity. How species adapt to urban challenges sheds light on evolution and provides clues about conservation. EMBO Rep. 2007, 8, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Antúnez, P.; Hernández-Díaz, J.C.; Wehenkel, C.; Clark-Tapia, R. Generalized models: An application to identify environmental variables that significantly affect the abundance of three tree species. Forests 2017, 8, 59. [Google Scholar] [CrossRef]
- Ackerly, D. Canopy gaps to climate change -extreme events, ecology and evolution. New Phytol. 2003, 160, 2–4. [Google Scholar] [CrossRef]
- Ackerly, D.D. Community assembly, niche conservatism, and adaptive evolution in changing environments. Int. J. Plant. Sci. 2003, 164, 165–184. [Google Scholar] [CrossRef]
- Eiserhardt, W.L.; Svenning, J.C.; Borchsenius, F.; Kristiansen, T.; Balslev, H. Separating environmental and geographical determinants of phylogenetic community structure in Amazonian palms (Arecaceae). Bot. J. Linn. Soc. 2013, 171, 244–259. [Google Scholar] [CrossRef]
- Davis, M.B.; Shaw, R.G.; Etterson, J.R. Evolutionary responses to changing climate. Ecology 2005, 86, 1704–1714. [Google Scholar] [CrossRef]
- Davis, M.B.; Shaw, R.G. Range shifts and adaptive responses to quaternary climate change. Science 2001, 292, 673–679. [Google Scholar] [CrossRef] [PubMed]
| Species | AUC Values | ||
|---|---|---|---|
| Past | Present | Future | |
| Pinus arizonica Engelm. var. stormiae Martínez | 0.960 | 0.961 | 0.962 |
| Pinusstrobiformis Engelm | 0.927 | 0.945 | 0.945 |
| Pinus cembroides Zucc | 0.948 | 0.944 | 0.950 |
| Pinus cooperi C.E.Blanco | 0.961 | 0.965 | 0.966 |
| Pinus durangensis Martínez | 0.915 | 0.909 | 0.908 |
| Pinus engelmanni Carr | 0.926 | 0.929 | 0.908 |
| Pinus leiophylla Schiede ex Schltdl. et Cham. | 0.954 | 0.937 | 0.943 |
| Pinus teocote Schiede ex Schltdl. et Cham. | 0.951 | 0.942 | 0.947 |
| Quercus arizonica Sarg | 0.791 | 0.784 | 0.794 |
| Quercus crassifolia Humb. & Bonpl. | 0.966 | 0.964 | 0.967 |
| Quercus grisea Liebm | 0.942 | 0.939 | 0.939 |
| Quercus magnolifolia Née | 0.964 | 0.967 | 0.967 |
| Quercus sideroxyla Humb. & Bonpl. | 0.879 | 0.897 | 0.898 |
| Acronyms | Description | PC1 | PC2 |
|---|---|---|---|
| bio_01 | Mean Annual Temperature (°C) | 0.34 | 0.06 |
| bio_02 | Mean Diurnal Range (Mean of monthly max. temp. min. temp.) (°C) | 0.93 | 0.07 |
| bio_03 | Isothermality (Bio_02/Bio_07) (×100) | 0.31 | −0.41 |
| bio_04 | Temperature Seasonality (standard deviation × 100) (Coefficient of Variation) | 0.98 | 0.19 |
| bio_05 | Max Temperature of Warmest Month (°C) | −0.31 | 0.13 |
| bio_06 | Min Temperature of Coldest Month (°C) | 0.72 | −0.03 |
| bio_07 | Temperature Annual Range (Bio_05–Bio_06) (°C) | 0.96 | 0.13 |
| bio_08 | Mean Temperature of Wettest Quarter (°C) | 0.19 | 0.13 |
| bio_09 | Mean Temperature of Driest Quarter (°C) | 0.44 | 0.16 |
| bio_10 | Mean Temperature of Warmest Quarter (°C) | 0.12 | 0.11 |
| bio_11 | Mean Temperature of Coldest Quarter (°C) | 0.50 | 0.027 |
| bio_12 | Annual Precipitation (mm) | 0.88 | 0.45 |
| bio_13 | Precipitation of Wettest Month (mm) | 0.88 | 0.42 |
| bio_14 | Precipitation of Driest Month (mm) | 0.22 | 0.49 |
| bio_15 | Precipitation Seasonality (Coefficient of Variation) | −0.33 | −0.24 |
| bio_16 | Precipitation of Wettest Quarter (mm) | 0.88 | 0.44 |
| bio_17 | Precipitation of Driest Quarter (mm) | 0.52 | 0.45 |
| bio_18 | Precipitation of Warmest Quarter (mm) | 0.79 | 0.42 |
| bio_19 | Precipitation of Coldest Quarter (mm) | 0.72 | 0.43 |
| Species Studied | Periods of Time | Past | Present |
|---|---|---|---|
| P. arizonica | Present | 0.75 | - |
| Future | 0.67 | 0.79 | |
| P. strobiformis | Present | 0.73 | - |
| Future | 0.75 | 0.84 | |
| P. cembroides | Present | 0.52 | - |
| Future | 0.65 | 0.46 | |
| P. cooperi | Present | 0.80 | - |
| Future | 0.78 | 0.82 | |
| P. duranguensis | Present | 0.72 | - |
| Future | 0.73 | 0.82 | |
| P. engelmanni | Present | 0.73 | - |
| Future | 0.74 | 0.84 | |
| P. leiophylla | Present | 0.67 | - |
| Future | 0.71 | 0.78 | |
| P. teocote | Present | 0.72 | - |
| Future | 0.67 | 0.81 | |
| Q. arizonica | Present | −0.04 | - |
| Future | 0.02 | 0.71 | |
| Q. crassifolia | Present | −0.04 | - |
| Future | −0.05 | 0.85 | |
| Q. grisea | Present | 0.79 | - |
| Future | 0.74 | 0.87 | |
| Q. magnolifolia | Present | 0.79 | - |
| Future | 0.74 | 0.87 | |
| Q. sideroxyla | Present | 0.75 | - |
| Future | 0.69 | 0.69 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antúnez, P.; Suárez-Mota, M.E.; Valenzuela-Encinas, C.; Ruiz-Aquino, F. The Potential Distribution of Tree Species in Three Periods of Time under a Climate Change Scenario. Forests 2018, 9, 628. https://doi.org/10.3390/f9100628
Antúnez P, Suárez-Mota ME, Valenzuela-Encinas C, Ruiz-Aquino F. The Potential Distribution of Tree Species in Three Periods of Time under a Climate Change Scenario. Forests. 2018; 9(10):628. https://doi.org/10.3390/f9100628
Chicago/Turabian StyleAntúnez, Pablo, Mario Ernesto Suárez-Mota, César Valenzuela-Encinas, and Faustino Ruiz-Aquino. 2018. "The Potential Distribution of Tree Species in Three Periods of Time under a Climate Change Scenario" Forests 9, no. 10: 628. https://doi.org/10.3390/f9100628
APA StyleAntúnez, P., Suárez-Mota, M. E., Valenzuela-Encinas, C., & Ruiz-Aquino, F. (2018). The Potential Distribution of Tree Species in Three Periods of Time under a Climate Change Scenario. Forests, 9(10), 628. https://doi.org/10.3390/f9100628





