Maintenance of K+/Na+ Balance in the Roots of Nitraria sibirica Pall. in Response to NaCl Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Hydroponic Culture
2.2. NaCl Treatment
2.3. Ion Content Analysis
2.4. Flux Measurements with NMT
2.5. Data Analysis
3. Results
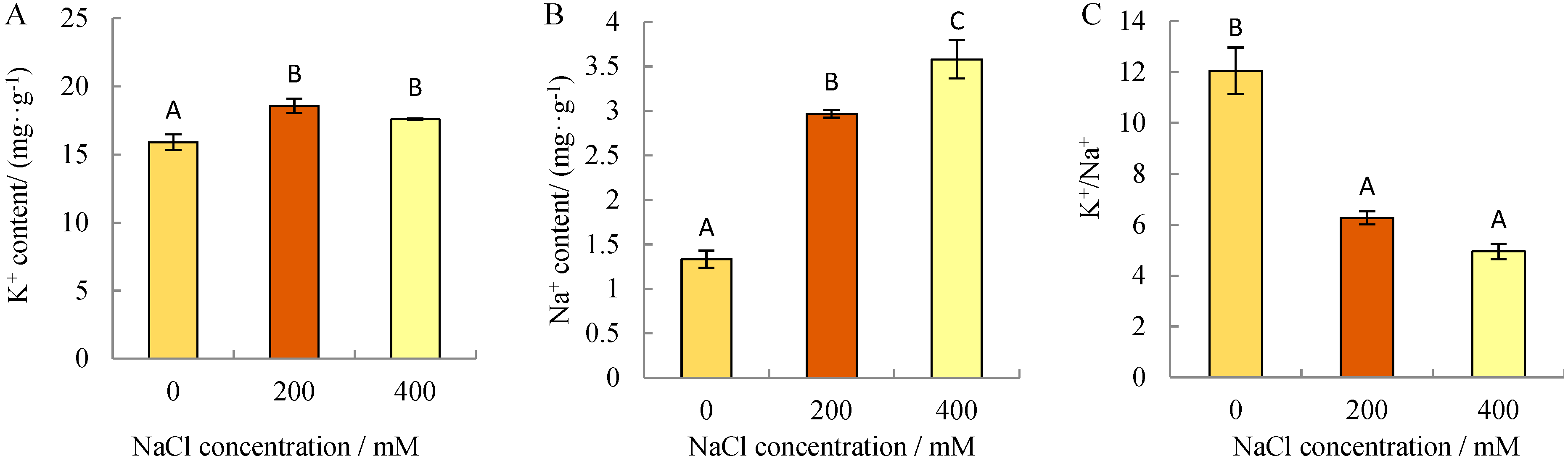
3.1. Variations of Na+ and K+ in N. sibirica Roots under NaCl Stress
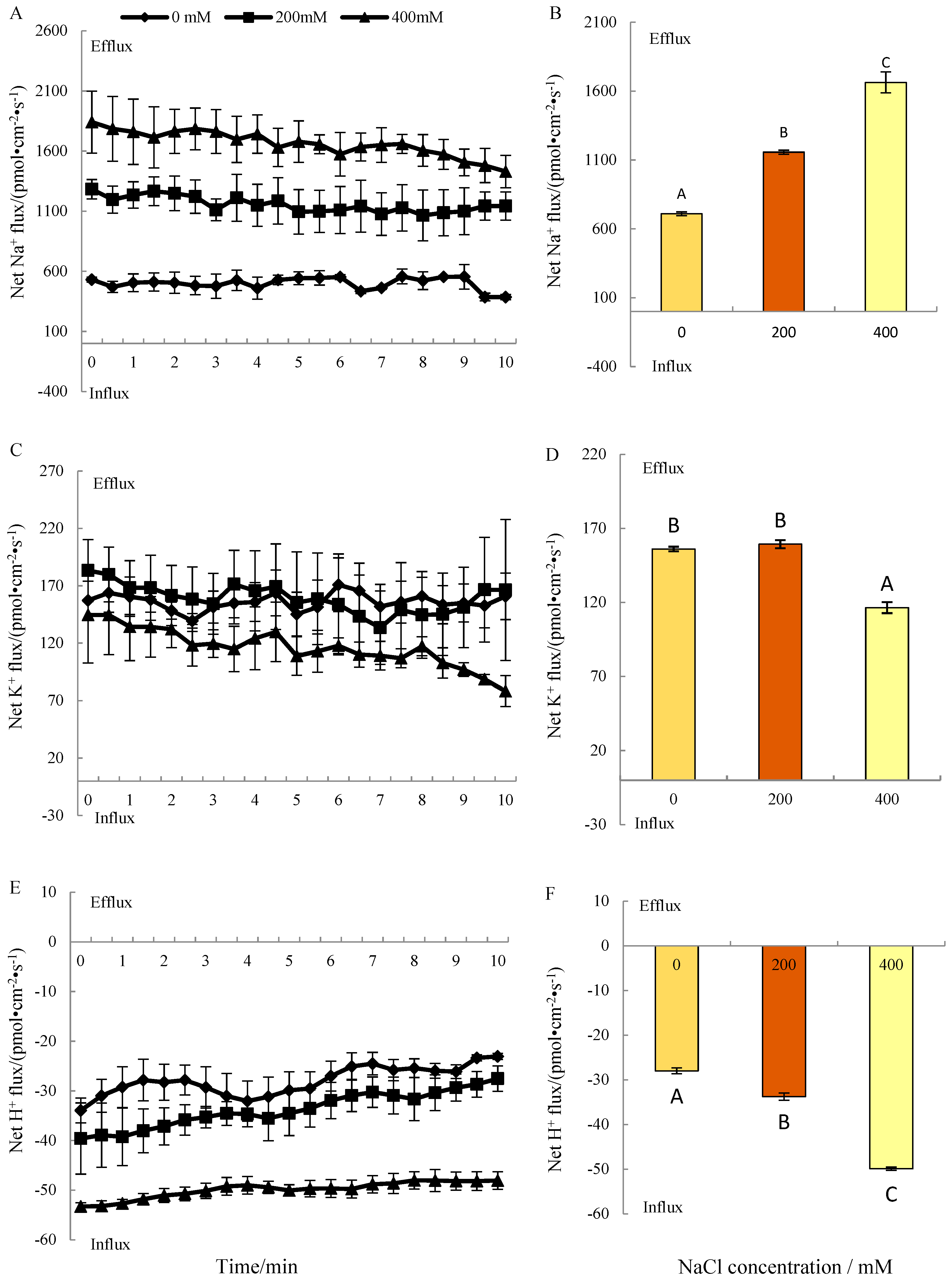
3.2. Steady Ion Fluxes under Salt Treatment
3.2.1. Na+ Fluxes
3.2.2. K+ Fluxes
3.2.3. H+ Fluxes
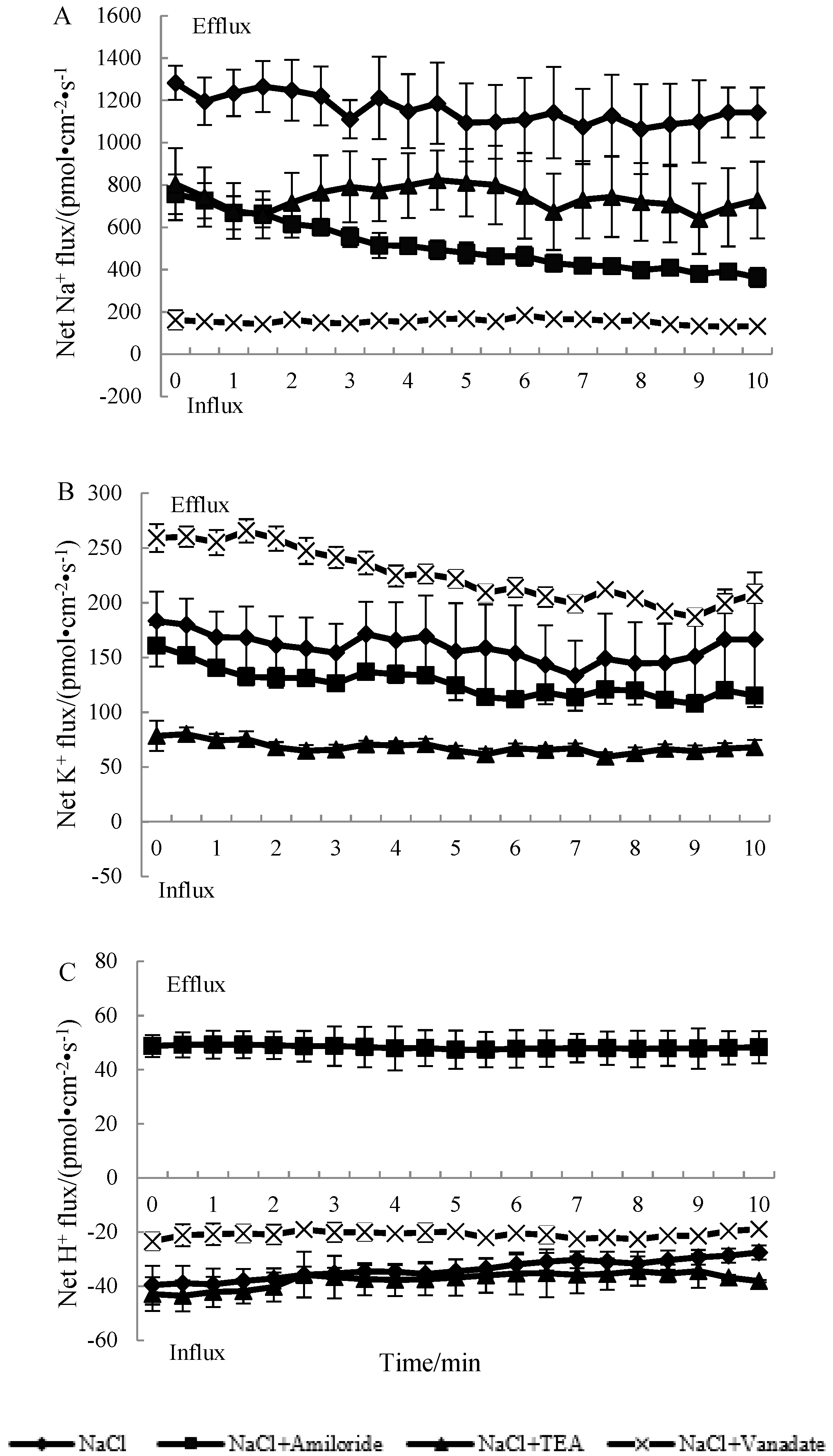
3.3. The Effects of Ion Transport Inhibitors on the Ion Fluxes of N. sibirica Seedling Roots under NaCl Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Niu, X.; Pardo, J.M. Ion Homeostasis in NaCl Stress Environments. Plant Physiol. 1995, 109, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Maathuis, F.J.M.; Amtmann, A. K+ Nutrition and Na+ Toxicity: The Basis of Cellular K+/Na+ Ratios. Ann. Bot. 1999, 84, 123–133. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.-K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S. Ionic and osmotic components of salt stress specifically modulate net ion fluxes from bean leaf mesophyll. Plant Cell Environ. 2000, 23, 825–837. [Google Scholar] [CrossRef]
- Chen, Z.-H.; Zhou, M.-X.; Newman, I.A.; Mendham, N.J.; Zhang, G.-P.; Shabala, S. Potassium and sodium relations in salinised barley tissues as a basis of differential salt tolerance. Funct. Plant Biol. 2007, 34, 150–162. [Google Scholar] [CrossRef]
- Tester, M.; Davenport, R. Na+ tolerance and Na+ transport in higher plants. Ann. Bot. 2003, 91, 503–527. [Google Scholar] [CrossRef] [PubMed]
- Volkovs, V.; Wang, B.; Dominy, P.; Fricke, W.; Amtmann, A. Thellungiella halophila, a salt-tolerant relative of Arabidopsis thaliana, possesses effective mechanisms to discriminate between potassium and sodium. Plant Cell Environ. 2004, 27, 1–14. [Google Scholar] [CrossRef]
- Shabala, S.; Cuin, T.A. Potassium transport and plant salt tolerance. Physiol. Plant. 2008, 133, 651–669. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Chen, S.-L.; Dai, S.-X.; Wang, R.-G.; Li, N.-Y.; Shen, X.; Zhou, X.-Y.; Lu, C.-F.; Zheng, X.-J.; Hu, Z.-M.; et al. Ion flux profiles and plant ion homeostasis control under salt stress. Plant Signal. Behav. 2009, 4, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-H.; Pottosin, I.I.; Cuin, T.A.; Fuglsang, A.T.; Tester, M.; Jha, D.; Zepedajazo, I.; Zhou, M.-X.; Palmgren, M.G.; Newman, I.A.; et al. Root plasma membrane transporters controlling K+/Na+ homeostasis in salt-stressed barley. Plant Physiol. 2007, 145, 1714–1725. [Google Scholar] [CrossRef] [PubMed]
- Cuin, T.A.; Zhou, M.; Parsons, D.; Shabala, S. Genetic behaviour of physiological traits conferring cytosolic K+/Na+ homeostasis in wheat. Plant Biol. 2012, 14, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Apse, M.P.; Aharon, G.S.; Snedden, W.A.; Blumwald, E. Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 1999, 285, 1256–1258. [Google Scholar] [CrossRef] [PubMed]
- Ohta, M.; Hayashi, Y.; Nakashima, A.; Hamada, A.; Tanaka, A.; Nakamura, T.; Hayakawa, T. Introduction of a Na+/H+ antiporter gene from Atriplex gmelini confers salt tolerance to rice. FEBS Lett. 2002, 532, 279–282. [Google Scholar] [CrossRef]
- Parks, G.E.; Dietrich, M.A.; Schumaker, K.S. Increased vacuolar Na+/H+ exchange activity in Salicornia bigelovii Torr. in response to NaCl. J. Exp. Bot. 2002, 53, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, A.; Nakamura, A.; Tagiri, A.; Tanaka, H.; Miyao, A.; Hirochika, H.; Tanaka, Y. Function, intracellular localization and the importance in salt tolerance of a vacuolar Na+/H+ antiporter from rice. Plant Cell Physiol. 2004, 45, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wang, R.-H.; Yang, X.-Y.; Zhu, J.-F.; Liu, Z.-X.; Ni, J.-W.; Zhang, H.-X. Isolation and expression analysis of a vacuolar membrane Na+/H+ antiporter gene NtNHX1 from Nitraria tangutorum. Sci. Silvae Sin. 2014, 50, 38–44. (In Chinese) [Google Scholar]
- Wang, L.; Ma, Y.-K.; Li, N.-N.; Zhang, W.-B.; Mao, H.-P.; Lin, X.-F. Isolation and characterization of a tonoplast Na+/H+ antiporter from the halophyte Nitraria sibirica. Biol. Plant. 2016, 60, 113–122. [Google Scholar] [CrossRef]
- Shabala, L.; Cuin, T.A.; Newman, I.A.; Shabala, S. Salinity-induced ion flux patterns from the excised roots of Arabidopsis sos mutants. Planta 2005, 222, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.-Z.; Ishitani, M.; Cheolsoo, K.; Zhu, J.-K. The Arabidopsis thaliana salt tolerance gene SOS1 encodes a putative Na+/H+ antiporter. Proc. Natl. Acad. Sci. USA 2000, 97, 6896–6901. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Quintero, F.J.; Pardo, J.M.; Zhu, J.-K. The putative plasma membrane Na+/H+ antiporter SOS1 controls long-distance Na+ transport in plants. Plant Cell 2002, 14, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-J.; Ding, L.; Zhu, J.-K. SOS1, a genetic locus essential for salt tolerance and potassium acquisition. Plant Cell 1996, 8, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.-L.; Zhang, H.-R.; He, L.-M.; Wang, Y.-C. Isolation and expression analysis of a plasma membrane Na+/H+ antiporter gene NtNHX1 from Nitraria tangutorum. Acta Prataculturae Sin. 2013, 22, 179–186. (In Chinese) [Google Scholar]
- Wang, L.; Yang, Y.-L.; Li, K.-W.; Liu, J.-M.; Bao, Y.; Wang, J.-Y. Advance of plant plasma membrance H+-ATPase in response to salt stress. J. Northwest Norm. Univ. (Nat. Sci.) 2006, 42, 72–77. (In Chinese) [Google Scholar]
- Maathuis, F.J.M.; Sanders, D. Sodium uptake in Arabidopsis roots is regulated by cyclic nucleotides. Plant Physiol. 2001, 127, 1617–1625. [Google Scholar] [CrossRef] [PubMed]
- Demidchik, V.; Tester, M. Sodium fluxes through nonselective cation channels in the plasma membrane of protoplasts from Arabidopsis roots. Plant Physiol. 2002, 128, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Demidchik, V.; Davenport, R.J.; Tester, M. Nonselective cation channels in plants. Annu. Rev. Plant Biol. 2002, 53, 67–107. [Google Scholar] [CrossRef] [PubMed]
- Maathuis, F.J. The role of monovalent cation transporters in plant responses to salinity. J. Exp. Bot. 2006, 57, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Horie, T.; Hauser, F.; Schroeder, J.I. HKT transporter-mediated salinity resistance mechanisms in Arabidopsis and monocot crop plants. Trends Plant Sci. 2009, 14, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-L.; Flowers, T.J.; Wang, S.-M. Differentiation of low-affinity Na+ uptake pathways and kinetics of the effects of K+ on Na+ uptake in the halophyte Suaeda maritima. Precis. Eng. 2013, 24, 70–76. [Google Scholar] [CrossRef]
- Demidchik, V.; Maathuis, F.J. Physiological roles of nonselective cation channels in plants: From salt stress to signalling and development. New Phytol. 2007, 175, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Cuin, T.A.; Betts, S.A.; Chalmandrier, R.; Shabala, S. A root’s ability to retain K+ correlates with salt tolerance in wheat. J. Exp. Bot. 2008, 59, 2697–2706. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Chen, S.-L.; Dai, S.-X.; Wang, R.-G.; Li, N.-Y.; Xin, S.; Zhou, X.-Y.; Lu, C.-F.; Zheng, X.-J.; Hu, Z.-X.; et al. NaCl-induced alternations of cellular and tissue ion fluxes in roots of salt-resistant and salt-sensitive poplar species. Plant Physiol. 2009, 149, 1141–1153. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.-W.; Wu, X.; Zhang, H.-X.; Liu, T.; Zhang, L. Comparative analysis of salt tolerance of three Nitraria species. For. Res. 2012, 25, 48–53. (In Chinese) [Google Scholar]
- Li, Q.-H.; Wang, S.-X.; Xu, J.; Ren, W.-J.; Zhao, Y.-M. Comprehensive evaluation on salt tolerance of different desert shrubs in Ulan Buh Desert regions. Pratacultural Sci. 2012, 28, 103–108. (In Chinese) [Google Scholar]
- Yang, S.; Zhang, H.-X.; Liu, T. Effect of salt stress on osmotic adjustment substances in plants. For. Res. 2012, 25, 269–277. (In Chinese) [Google Scholar]
- Zou, J.-H.; Xu, S.-Y.; Miao, J.-W.; Zhang, J. Salt tolerance test of Nitraria. Shangdong For. Sci. Technol. 2005, 1, 19–20. (In Chinese) [Google Scholar]
- Zhang, G.-L. Effects of iso-osmotic salt and water stresses on growth and ionic absorption and distribution in Nitraria sibirica seedlings. Agric. Res. Arid Areas 2013, 31, 114–118. (In Chinese) [Google Scholar]
- Rina, S.; Guilin, C. Effect of exogenous spermidine on antioxidant enzyme system in leaves of Nitraria sibirica Pall. Seedlings under salt stress. Acta Bot. Boreali Occident. Sin. 2013, 33, 352–356. [Google Scholar]
- Cheng, T.-L.; Li, H.-Y.; Wu, H.-W.; Liu, X.-Z.; Wu, X.; Yang, S.; Zhang, H.-X.; Yang, X.-Y. Comparison on osmotica accumulation of different salt-tolerant plants under salt stress. For. Res. 2015, 28, 826–832. (In Chinese) [Google Scholar]
- Wu, X.; Ni, J.; Zhang, H.; Liu, T.; Zhang, L. Effects of salt stress on osmotic adjustment substances in three species of Nitraria. J. Northeast For. Univ. 2012, 40, 44–47. (In Chinese) [Google Scholar]
- Tang, X.-Q.; Li, H.-Y.; Yang, X.-Y.; Liu, Z.-X.; Zhang, H.-X. Effect of short-time stress on distribution and balance of Na+ and K+ in Nitraria sibirica Pall. seedlings. For. Res. 2017, 30, 1022–1027. (In Chinese) [Google Scholar]
- Li, H.-Y.; Tang, X.-Q.; Yang, X.-Y.; Wu, H.-W.; Zhang, H.-X. Effects of NaCl stress on mineral element contents in Nitraria sibirica seedlings. Plant Physiol. J. 2017, 53, 2125–2136. (In Chinese) [Google Scholar]
- Liu, Z.-X.; Zhu, J.-F.; Yang, X.-Y.; Wu, H.-W.; Wei, Q.; Wei, H.-R.; Zhang, H.-X. Growth performance, organ-level ionic relations and organic osmoregulation of Elaeagnus angustifoliain response to salt stress. PLoS ONE 2018, 13, e0191552. [Google Scholar]
- Xing, J.-H.; Pan, D.-Z.; Tan, F.-L.; Chen, W. Effects of NaCl stress on the osmotic substance contents in Kandelia candel roots. Ecol. Environ. Sci 2017, 26, 1856–1871. (In Chinese) [Google Scholar]
- Kong, X.-Q.; Zhen, L.; Dong, H.-Z.; Eneji, A.E.; Li, W.-J. Effects of non-uniform root zone salinity on water use, Na+ recirculation, and Na+ and H+ flux in cotton. J. Exp. Bot. 2012, 63, 2105–2116. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Dai, S.-X.; Wang, R.-G.; Chen, S.-L.; Li, N.-Y.; Zhou, X.-Y.; Lu, C.-F.; Shen, X.; Zheng, X.-J.; Hu, Z.-M.; et al. Calcium mediates root K+/Na+ homeostasis in poplar species differing in salt tolerance. Tree Physiol. 2009, 29, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-L.; Shi, H.-Z. Physiological and molecular mechanisms of plant salt tolerance. Photosynth. Res. 2013, 115, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, H.-X.; Liu, T.; Wu, H.-W.; Ni, J.-W.; Chen, Q.-X. Study on Ion Metabolism Characteristics of Elaeagnus angustifolia Seedlings under NaCl Stress. For. Res. 2016, 29, 140–146. (In Chinese) [Google Scholar]
- Wang, X.-D.; Wang, C.; Ma, Z.-H.; Hou, R.-F.; Gao, Q.; Chen, Q. Effect of short term salt stress on the absorption of K+ and accumulation of Na+, K+ in seedlings of different wheat varieties. Acta Ecol. Sin. 2011, 31, 2822–2830. (In Chinese) [Google Scholar]
- Shi, H.Z.; Lee, B.-H.; Wu, S.-J.; Zhu, J.-K. Overexpression of a plasma membrane Na+/H+ antiporter gene improves salt tolerance in Arabidopsis thaliana. Nat. Biotechnol. 2003, 21, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Li, Y.-X.; Yuan, H.-J.; Hu, J.; Wei, L.; Bao, A.-K.; Zhang, J.-L.; Wang, S.-M. ZxSOS1 is essential for long-distance transport and spatial distribution of Na+ and K+ in the xerophyte Zygophyllum xanthoxylum. Plant Soil 2014, 374, 661–676. [Google Scholar] [CrossRef]
- Shabala, S.; Demidchik, V.; Shabala, L.; Cuin, T.A.; Smith, S.J.; Miller, A.J.; Davies, J.M.; Newman, I.A. Extracellular Ca2+ ameliorates NaCl-induced K+ loss from Arabidopsis root and leaf cells by controlling plasma membrane K+-permeable channels. Plant Physiol. 2006, 141, 1653–1665. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S.; Shabala, L.; Van, V.E.; Newman, I. Effect of divalent cations on ion fluxes and leaf photochemistry in salinized barley leaves. J. Exp. Bot. 2005, 56, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.-J.; Li, N.-Y.; Sun, J.; Hou, P.-C.; Jing, X.-S.; Zhu, H.-P.; Deng, S.-R.; Han, Y.-S.; Huang, X.-X.; Ma, X.-J.; et al. Exogenous hydrogen peroxide, nitric oxide and calcium mediate root ion fluxes in two non-secretor mangrove species subjected to NaCl stress. Tree Physiol. 2013, 33, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Golldack, D.; Zhao, C.; Bohnert, H.J. The expression of HAK-type K+ transporters is regulated in response to salinity stress in common ice plant. Plant Physiol. 2002, 129, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, X.; Yang, X.; Li, H.; Zhang, H. Maintenance of K+/Na+ Balance in the Roots of Nitraria sibirica Pall. in Response to NaCl Stress. Forests 2018, 9, 601. https://doi.org/10.3390/f9100601
Tang X, Yang X, Li H, Zhang H. Maintenance of K+/Na+ Balance in the Roots of Nitraria sibirica Pall. in Response to NaCl Stress. Forests. 2018; 9(10):601. https://doi.org/10.3390/f9100601
Chicago/Turabian StyleTang, Xiaoqian, Xiuyan Yang, Huanyong Li, and Huaxin Zhang. 2018. "Maintenance of K+/Na+ Balance in the Roots of Nitraria sibirica Pall. in Response to NaCl Stress" Forests 9, no. 10: 601. https://doi.org/10.3390/f9100601
APA StyleTang, X., Yang, X., Li, H., & Zhang, H. (2018). Maintenance of K+/Na+ Balance in the Roots of Nitraria sibirica Pall. in Response to NaCl Stress. Forests, 9(10), 601. https://doi.org/10.3390/f9100601




