Carbon and Nitrogen Accumulation and Decomposition from Coarse Woody Debris in a Naturally Regenerated Korean Red Pine (Pinus densiflora S. et Z.) Forest
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Forest
2.2. Tree Morality, CWD Volume and Mass
2.3. Decomposition of CWD and Needle Litter
2.4. Carbon and N Concentrations
2.5. Statistical Analysis
3. Results
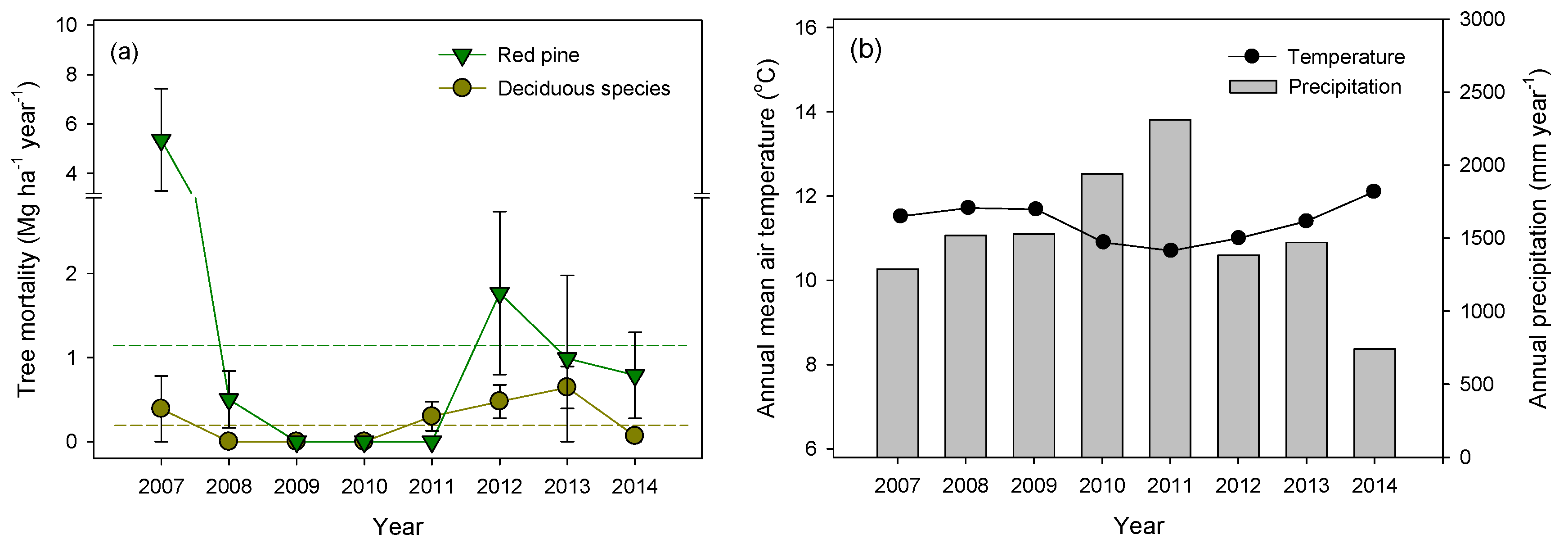
3.1. Tree Mortality and CWD Input
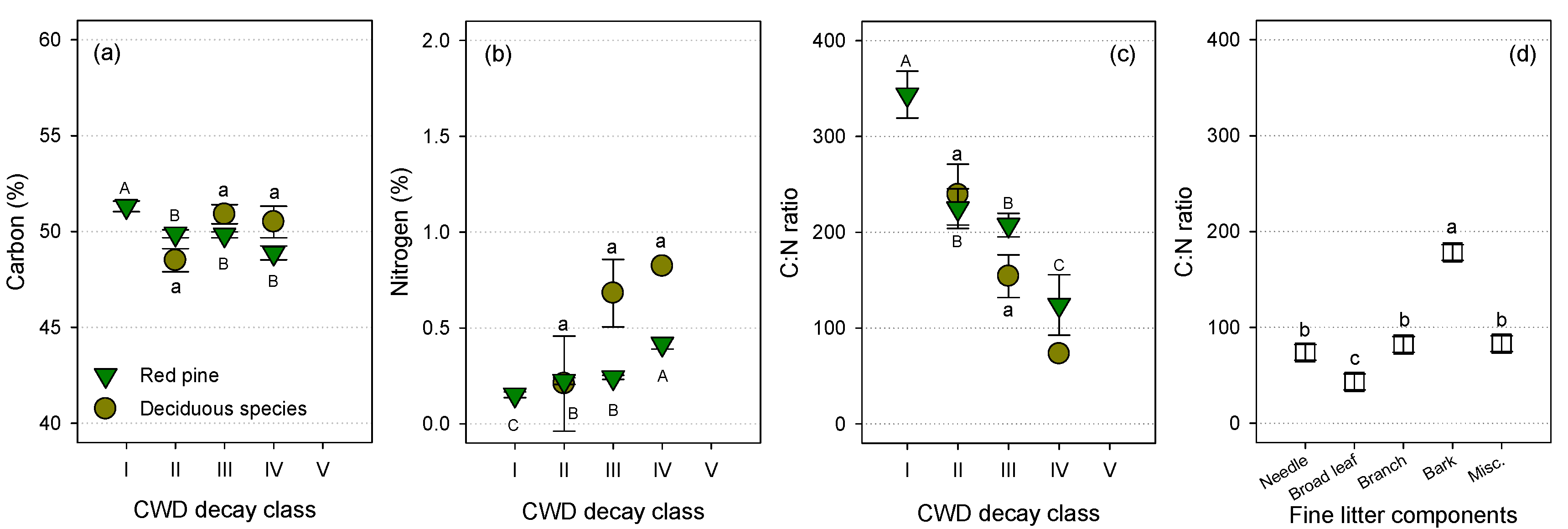
3.2. Carbon and N Concentrations
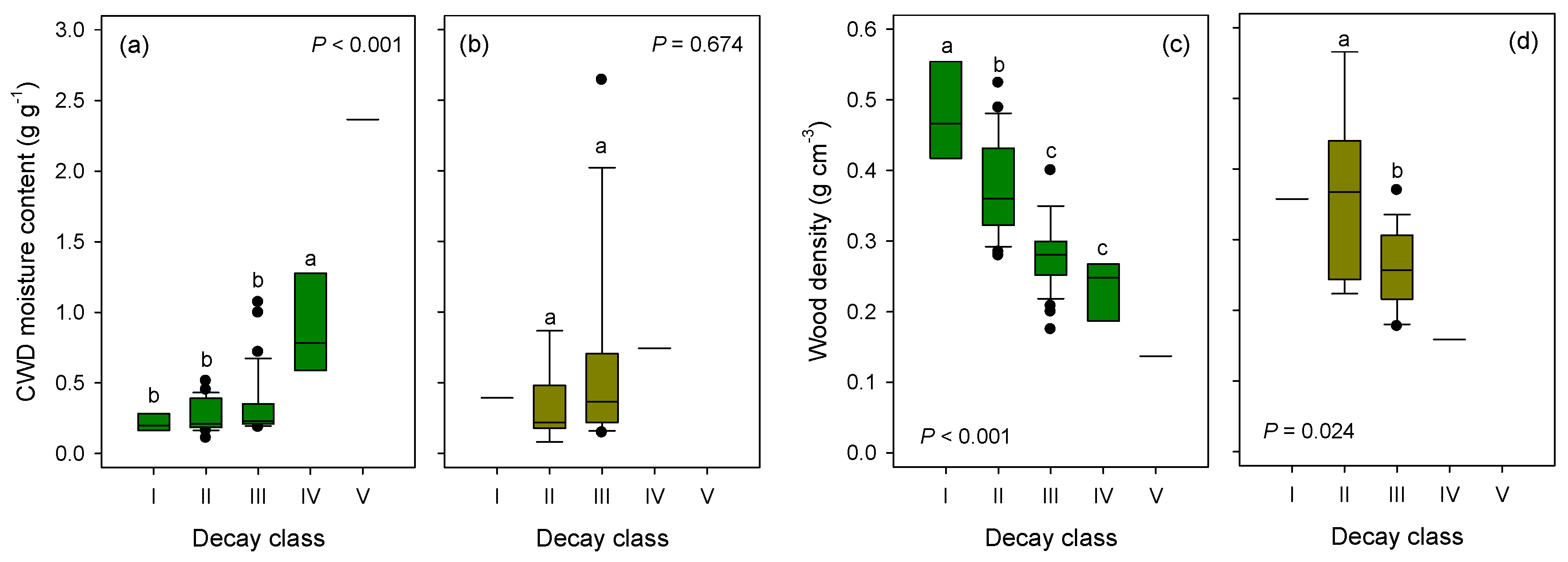
3.3. Distribution, C and N Contents of CWD
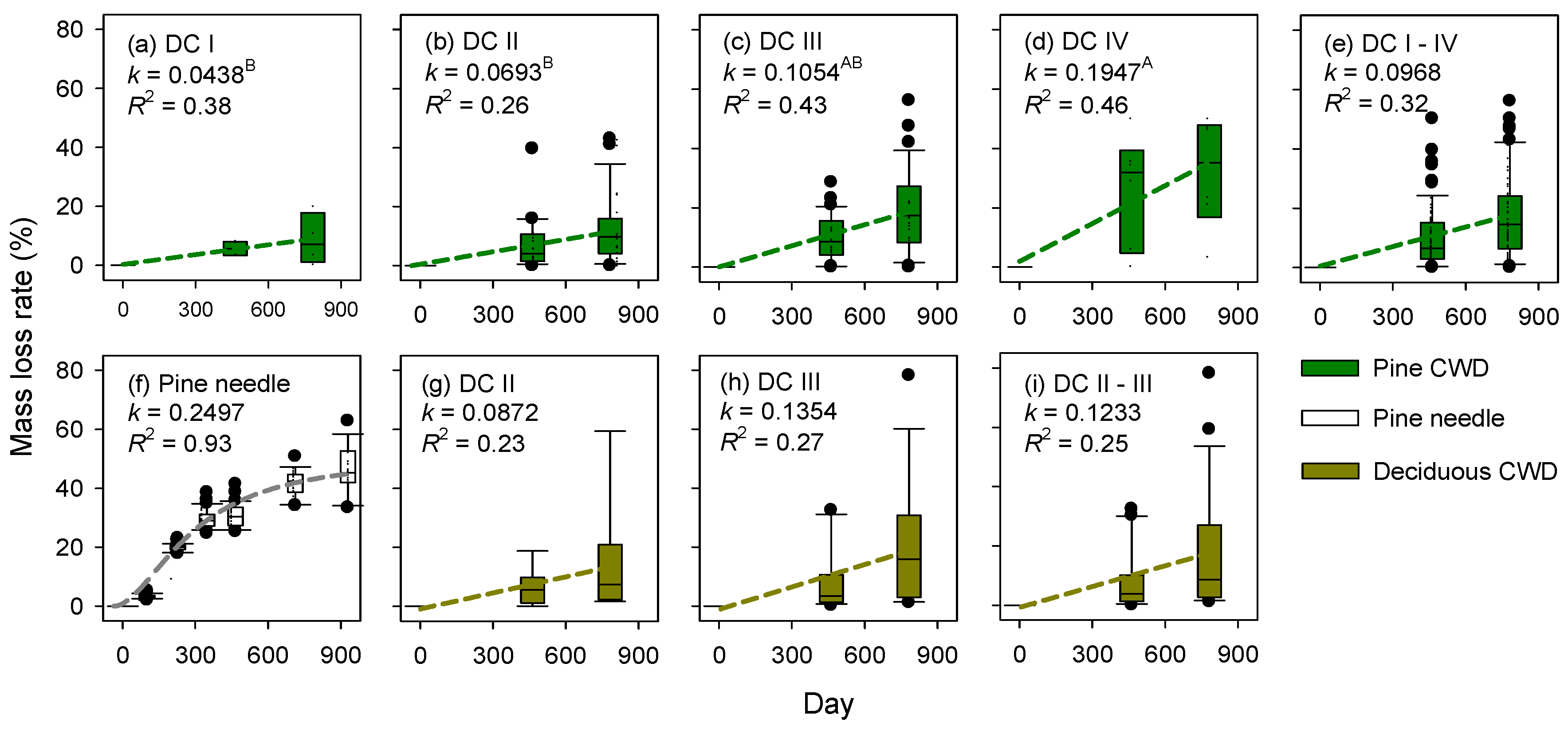
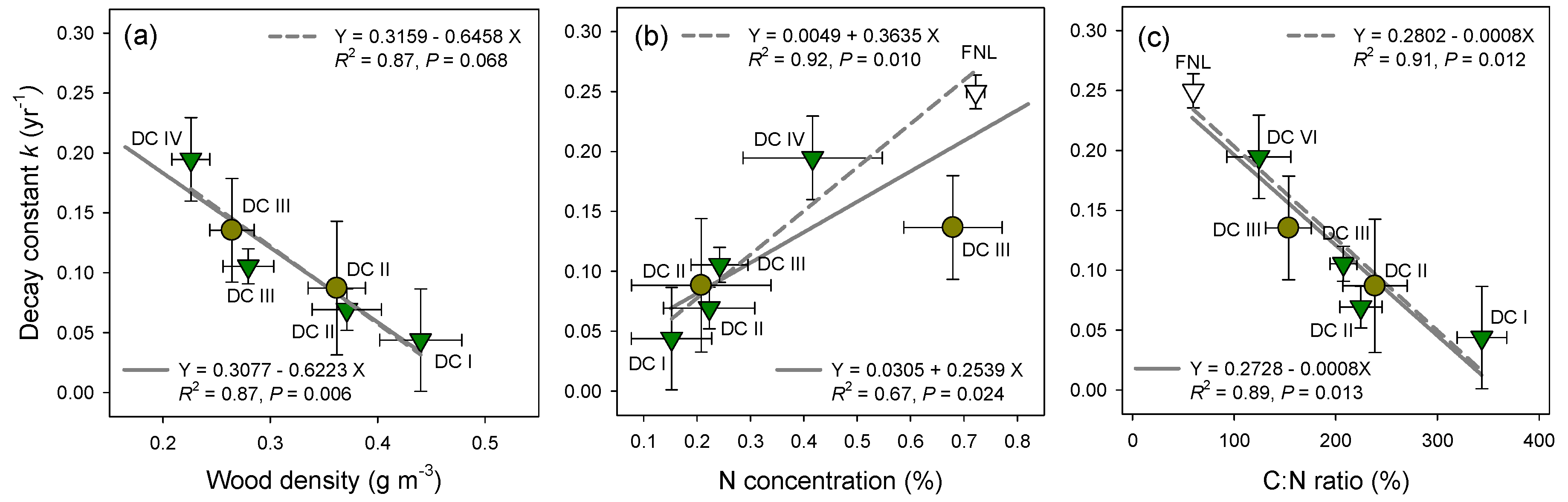
3.4. Mass Loss Rates of CWD and Needle Litter
4. Discussion
4.1. Variations in N Concentration of CWD
4.2. C and N Contents of CWD
4.3. Decomposition of Fine Litterfall and CWD
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Harmon, M.E.; Franklin, J.F.; Swanson, F.J.; Sollins, P.; Gregory, S.V.; Lattin, J.D.; Anderson, N.H.; Cline, S.P.; Aumen, N.G.; Sedell, J.R.; et al. Ecology of coarse woody debris in temperate ecosystems. Adv. Ecol. Res. 1986, 15, 133–302. [Google Scholar]
- Laiho, R.; Prescott, C.E. Decay and nutrient dynamics of coarse woody debris in northern coniferous forests: A synthesis. Can. J. For. Res. 2004, 34, 763–777. [Google Scholar] [CrossRef]
- Woodall, C.W.; Walters, B.F.; Oswalt, S.N.; Domke, G.M.; Toney, C.; Gray, A.N. Biomass and carbon attributes of downed woody materials in forests of the United States. For. Ecol. Manag. 2013, 305, 48–59. [Google Scholar] [CrossRef]
- Russell, M.B.; Fraver, S.; Aakala, T.; Gove, J.H.; Woodall, C.W.; D’Amato, A.W.; Ducey, M.J. Quantifying carbon stores and decomposition in dead wood: A review. For. Ecol. Manag. 2015, 350, 107–128. [Google Scholar] [CrossRef]
- Magnússon, R.Í.; Tietema, A.; Cornelissen, J.H.C.; Hefting, M.M.; Kalbitz, K. Sequestration of carbon from coarse woody debris in forest soils. For. Ecol. Manag. 2016, 377, 1–15. [Google Scholar] [CrossRef]
- Liski, J.; Nissinen, A.; Erhard, M.; Taskinen, O. Climatic effects on litter decomposition from arctic tundra to tropical rainforest. Glob. Chang. Biol. 2003, 9, 575–584. [Google Scholar] [CrossRef]
- Moore, T.R.; Trofymow, J.A.; Prescott, C.E.; Fyles, J.; Titus, B.D.; CIDET Working Group. Patterns of carbon, nitrogen and phosphorus dynamics in decomposing foliar litter in Canadian forests. Ecosystems 2006, 9, 46–62. [Google Scholar] [CrossRef]
- Zhang, D.; Hui, D.; Luo, Y.; Zhou, G. Rates of litter decomposition in terrestrial ecosystems: Global patterns and controlling factors. J. Plant Ecol. 2008, 1, 85–93. [Google Scholar] [CrossRef]
- Prescott, C.E. Litter decomposition: What controls it and how can we alter it to sequester more carbon in forest soils? Biogeochemistry 2010, 101, 133–149. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.C.; Allison, S.D.; Bauhus, J.; Eggleton, P.; Preston, C.M.; Scarff, F.; Weedon, J.T.; Wirth, C.; Zanne, A.E. Plant traits and wood fates across the globe: Rotted, burned, or consumed? Glob. Chang. Biol. 2009, 15, 2431–2449. [Google Scholar] [CrossRef]
- Brovkin, V.; van Bodegom, P.M.; Kleinen, T.; Wirth, C.; Cornwell, W.K.; Cornelissen, J.H.C.; Kattge, J. Plant-driven variation in decomposition rates improves projections of global litter stock distribution. Biogeosciences 2012, 9, 565–576. [Google Scholar] [CrossRef]
- Herrero, C.; Krankina, O.; Monleon, V.J.; Bravo, F. Amount of distribution of coarse woody debris in pine ecosystems of north-western Spain, Russia and the United States. iForest 2014, 7, 53–60. [Google Scholar] [CrossRef]
- Holub, S.M.; Spears, D.H.; Lajtha, K. A reanalysis of nutrient dynamics in coniferous coarse woody debris. Can. J. For. Res. 2001, 31, 1894–1902. [Google Scholar] [CrossRef]
- Sturtevant, B.R.; Bissonette, J.A.; Long, J.N.; Roberts, D.W. Coarse woody debris as a function of age, stand structure, and disturbance in boreal Newfoundland. Ecol. Appl. 1997, 7, 702–712. [Google Scholar] [CrossRef]
- Siitonen, J. Forest management, coarse woody debris and saproxylic organisms: Fennoscandian boreal forests as an example. Ecol. Bull. 2001, 49, 11–41. [Google Scholar]
- Korea Forest Service. Statistical Yearbook of Forestry; Korea Forest Service: Daejeon, Korea, 2016.
- Yang, A.R.; Son, Y.; Noh, N.J.; Lee, S.K.; Jo, W.; Son, J.A.; Kim, C.; Bae, S.W.; Lee, S.T.; Kim, H.S.; et al. Effect of thinning on carbon storage in soil, forest floor and coarse woody debris of Pinus densiflora stands with different stand ages in Gangwon-do, central Korea. For. Sci. Technol. 2011, 7, 30–37. [Google Scholar]
- Shin, S.C. Pine wilt disease in Korea. In Pine Wilt Disease; Zhao, K., Futai, K., Sutherland, J.R., Takeuchi, Y., Eds.; Springer: Tokyo, Japan, 2008; pp. 26–32. [Google Scholar]
- Kwon, T.S.; Shin, J.H.; Lim, J.H.; Kim, Y.K.; Lee, E.J. Management of pine wilt disease in Korea through preventative silvicultural control. For. Ecol. Manag. 2011, 261, 562–569. [Google Scholar] [CrossRef]
- Fukasawa, Y. The geographical gradient of pine log decomposition in Japan. For. Ecol. Manag. 2015, 349, 29–35. [Google Scholar] [CrossRef]
- Kim, C.; Yoo, B.O.; Jung, S.Y.; Lee, K.S. Allometric equations to assess biomass, carbon and nitrogen content of black pine and red pine trees in southern Korea. iForest 2017, 10, 483–490. [Google Scholar] [CrossRef]
- Noh, N.J.; Kim, C.; Bae, S.W.; Lee, W.K.; Yoon, T.K.; Muraoka, H.; Son, Y. Carbon and nitrogen dynamics in a Pinus densiflora forest with low and high stand densities. J. Plant Ecol. 2013, 6, 368–379. [Google Scholar] [CrossRef]
- Harmon, M.E.; Fasth, B.; Woodall, C.W.; Sexton, J. Carbon concentration of standing and downed woody detritus: Effects of tree taxa, decay class, position, and tissue type. For. Ecol. Manag. 2013, 291, 259–267. [Google Scholar] [CrossRef]
- Klockow, P.A.; D’Amato, A.W.; Bradford, J.B.; Fraver, S. Nutrient concentrations in coarse and fine woody debris of Populus tremuloides Michx.-dominated forest, northern Minnesota, USA. Silva Fenn. 2014, 48, 962. [Google Scholar] [CrossRef]
- Ganjegunte, G.K.; Condron, L.M.; Clinton, P.W.; Davis, M.R.; Mahieu, N. Decomposition and nutrient release from radiate pine (Pinus radiate) coarse woody debris. For. Ecol. Manag. 2004, 187, 197–211. [Google Scholar] [CrossRef]
- Mobley, M.L.; de Richter, D.B.; Heine, P.R. Accumulation and decay of woody detritus in a humid subtropical secondary pine forest. Can. J. For. Res. 2013, 43, 109–118. [Google Scholar] [CrossRef]
- Jomura, M.; Akashi, Y.; Itoh, H.; Yuki, R.; Sakai, Y. Biotic and abiotic factors controlling respiration rates of above- and belowground woody debris of Fagus crenata and Quercus crispula in Japan. PLoS ONE 2015, 10, e0145113. [Google Scholar] [CrossRef] [PubMed]
- Yoon, T.K.; Noh, N.J.; Han, S.; Lee, J.; Son, Y. Soil moisture effects on leaf litter decomposition and soil carbon efflux in wetland and upland forests. Soil Sci. Soc. Am. J. 2014, 78, 1804–1816. [Google Scholar] [CrossRef]
- Yang, Y.; Luo, Y. Carbon: Nitrogen stoichiometry in forest ecosystems during stand development. Glob. Ecol. Biogeogr. 2011, 20, 354–361. [Google Scholar]
- Mackensen, J.; Bauhus, J. Density loss and respiration rates in coarse woody debris of Pinus radiate, Eucalyptus regnans and Eucalyptus maculate. Soil Biol. Biochem. 2003, 35, 177–186. [Google Scholar] [CrossRef]
- Yuan, J.; Hou, L.; Wei, X.; Shang, Z.; Cheng, F.; Zhang, S. Decay and nutrient dynamics of coarse woody debris in the Qinling Mountains, China. PLoS ONE 2017, 12, e0175203. [Google Scholar] [CrossRef] [PubMed]
- Harmon, M.E.; Sexton, J. Guidelines for Measurements of Woody Detritus in Forest Ecosystem; US Long-Term Ecological Research Network Office, University of Washington: Seattle, WA, USA, 1996. [Google Scholar]
- Kim, R.H.; Son, Y.; Lim, J.H.; Lee, I.K.; Seo, K.W.; Koo, J.W.; Noh, N.J.; Ryu, S.-R.; Hong, S.K.; Im, B.S. Coarse woody debris mass and nutrients in forest ecosystems of Korea. Ecol. Res. 2006, 21, 819–827. [Google Scholar] [CrossRef]
- Sollins, P. Input and decay of coarse woody debris in coniferous stands in western Oregon and Washington. Can. J. For. Res. 1982, 12, 18–28. [Google Scholar] [CrossRef]
- Sollins, P.; Cline, S.P.; Verhoeven, T.; Sachs, D.; Spycher, G. Patterns of log decay in old-growth Douglas-fir forests. Can. J. For. Res. 1987, 17, 1585–1595. [Google Scholar] [CrossRef]
- Whitmore, T.C. Tropical Rain Forests of the Far East; Oxford University Press: Oxford, UK, 1984. [Google Scholar]
- Bradford, M.A.; Tordoff, G.M.; Eggers, T.; Jones, T.H.; Newington, J.E. Microbiota, fauna, and mesh size interactions in litter decomposition. Oikos 2002, 99, 317–323. [Google Scholar] [CrossRef]
- Berg, B.; Laskowski, R. Litter decomposition: A guide to carbon and nutrient turnover. Adv. Ecol. Res. 2006, 38, 20–71. [Google Scholar]
- Hinrichs, C.; Boiler, C. JMP Essentials: An Illustrated Step-by-Step Guide for New Users, 2nd ed.; SAS Institute Inc.: Cary, NC, USA, 2014. [Google Scholar]
- Laiho, R; Prescott, C.E. The contribution of coarse woody debris to carbon, nitrogen, and phosphorus cycles in three Rocky Mountain coniferous forests. Can. J. For. Res. 1999, 29, 1592–1603. [Google Scholar] [CrossRef]
- Wei, X.; Kimmins, J.P.; Peel, K.; Steen, O. Mass nutrients in woody debris in harvested and wildfire-killed lodgepole pine forests in the central interior of British Columbia. Can. J. For. Res. 1997, 27, 148–155. [Google Scholar] [CrossRef]
- Yim, J.S.; Kim, R.H.; Lee, S.J.; Son, Y. Dead wood inventory and assessment in South Korea. In Pushing Boundaries: New Directions in Inventory Techniques and Applications; Forest Inventory and Analysis Symposium: Portland, OR, USA, 8–10 December 2015; PNW-GTR-931; US Department of Argiculture: Portland, OR, USA, 2015; pp. 134–138. [Google Scholar]
- Ohtsuka, T.; Negishi, M.; Sugita, K.; Iimura, Y.; Hirota, M. Carbon cycling and sequestration in a Japanese red pine (Pinus densiflora) forest on lava flow of Mt. Fuji. Ecol. Res. 2013, 28, 855–867. [Google Scholar] [CrossRef]
- Maguire, D.A. Branch mortality and potential litterfall from Douglas-fir trees in stands of varying density. For. Ecol. Manag. 1994, 70, 41–53. [Google Scholar] [CrossRef]
- Sippola, A.L.; Siitonen, J.; Kallio, R. Amount and quality of coarse woody debris in natural and managed coniferous forests near the timberline in Finnish Lapland. Scan. J. For. Res. 1998, 13, 204–214. [Google Scholar] [CrossRef]
- Yuan, J.; Cheng, F.; Zhao, P.; Qiu, R.; Wang, L.; Zhang, S. Characteristics in coarse woody debris mediated by forest developmental stage and latest disturbances in a natural secondary forest of Pinus tabulaeformis. Acta Ecol. Sin. 2014, 34, 232–238. [Google Scholar] [CrossRef]
- Johnson, E.A.; Greene, D.F. A method for studying dead bole dynamics in Pinus contorta var. latifolia—Picea engelmannii forests. J. Veg. Sci. 1991, 2, 523–530. [Google Scholar]
- Krankina, O.N.; Harmon, M.E.; Griazkin, A.V. Nutrient stores and dynamics of woody detritus in a boreal forest: Modeling potential implications at the stand level. Can. J. For. Res. 1999, 29, 20–32. [Google Scholar] [CrossRef]
- Guo, L.B.; Bek, E.; Gifford, R.M. Woody debris in a 16-year old Pinus radiata plantation in Australia: Mass, carbon and nitrogen stocks, and turnover. For. Ecol. Manag. 2006, 228, 145–151. [Google Scholar] [CrossRef]
- Herrmann, S.; Prescott, C.E. Mass loss and nutrient dynamics of coarse woody debris in three Rocky Mountain coniferous forests: 21 year results. Can. J. For. Res. 2008, 38, 125–132. [Google Scholar] [CrossRef]
- Shorohova, E.; Kapitsa, E. Mineralization and fragmentation rates of bark attached to logs in a northern boreal forest. For. Ecol. Manag. 2014, 315, 185–190. [Google Scholar] [CrossRef]
- Yoon, T.K.; Chung, H.; Kim, R.H.; Noh, N.J.; Seo, K.W.; Lee, S.K.; Jo, W.; Son, Y. Coarse woody debris mass dynamics in temperate natural forests of Mt. Jumbong, Korea. J. Ecol. Field Biol. 2011, 34, 115–125. [Google Scholar] [CrossRef]
- Liu, W.; Schaefer, D.; Qiao, L.; Liu, X. What controls the variability of wood-decay rates? For. Ecol. Manag. 2013, 310, 623–631. [Google Scholar]
- Harmon, M.E.; Krankina, O.N.; Sexton, J. Decomposition vectors: A new approach to estimating woody detritus decomposition dynamics. Can. J. For. Res. 2000, 30, 76–84. [Google Scholar] [CrossRef]
| Species | CWD Type or Decay Class | Number (Pieces ha−1) | Number Distribution (%) | Mean Diameter (cm) | Volume (m3 ha−1) | Volume Distribution (%) |
|---|---|---|---|---|---|---|
| Red pine | Stump | 33 ± 12 | 6.1 | 9.79 ± 0.62 | 2.08 ± 0.99 | 10.1 |
| Snag | 146 ± 70 | 26.7 | 9.13 ± 0.59 | 6.25 ± 2.74 | 30.5 | |
| Log | 204 ± 69 | 37.4 | 9.56 ± 0.47 | 11.11 ± 3.96 | 54.2 | |
| Deciduous species | Stump | 46 ± 22 | 8.4 | 6.47 ± 0.45 | 0.30 ± 0.14 | 1.5 |
| Snag | 4 ± 4 | 0.8 | 6.00 | 0.06 ± 0.06 | 0.3 | |
| Log | 113± 31 | 20.6 | 5.91 ± 0.33 | 0.70 ± 0.21 | 3.4 | |
| Red pine | I | 21 ± 10 | 3.8 | 8.67 ± 1.71 | 0.97 ± 0.82 | 4.7 |
| II | 125 ± 26 | 22.9 | 8.22 ± 0.85 | 4.91 ± 2.13 | 24.0 | |
| III | 208 ± 17 | 38.2 | 9.75 ± 0.31 | 11.9 ± 1.57 | 58.1 | |
| IV | 29 ± 16 | 5.3 | 10.33 ± 0.42 | 1.64 ± 1.18 | 8.0 | |
| Deciduous species | I | 4 ± 4 | 0.8 | 6.00 | 0.05 ± 0.05 | 0.3 |
| II | 58 ± 17 | 10.7 | 6.03 ± 0.36 | 0.41 ± 0.12 | 2.0 | |
| III | 88 ± 13 | 16.0 | 5.97 ± 0.17 | 0.53 ± 0.09 | 2.6 | |
| IV | 13 ± 9 | 2.3 | 6.00 ± 0.58 | 0.07 ± 0.06 | 0.4 | |
| Total | 546 ± 34 | 100 | 7.80 ± 0.19 | 20.50 ± 2.55 | 100 |
| Fractions, Species or Decay Class | Mass (Mg ha−1) | C Content (Mg C ha−1) | N Content (kg N ha−1) | |
|---|---|---|---|---|
| Fine litterfall (year−1) 1 | Needle | 3.35 ± 0.14 (45.8) | 1.78 ± 0.07 (46.7) | 32.49 ± 1.27 (49.5) |
| Broad leaf | 0.82 ± 0.10 (11.2) | 0.40 ± 0.05 (10.5) | 10.30 ± 1.19 (15.7) | |
| Branch | 1.31 ± 0.10 (17.9) | 0.69 ± 0.05 (18.1) | 10.83 ± 0.82 (16.5) | |
| Bark | 0.76 ± 0.06 (10.4) | 0.39 ± 0.02 (10.2) | 2.18 ± 0.12 (3.3) | |
| Miscellaneous parts 4 | 1.08 ± 0.05 (14.85) | 0.55 ± 0.03 (14.4) | 9.84 ± 0.43 (15.0) | |
| Total | 7.32 ± 0.27 (100) | 3.81 ± 0.14 (100) | 65.65 ± 2.16 (100) | |
| Forest floor litter layer 1 | 20.18 ± 0.72 | 10.09 ± 0.36 | 280.4 ± 10.1 | |
| Tree mortality (year−1) 2 | Red pine | 1.18 ± 0.38 (83.1) | 0.60 ± 0.19 (84.7) | 1.82 ± 0.01 (79.8) |
| Deciduous species | 0.24 ± 0.04 (16.9) | 0.12 ± 0.02 (16.7) | 0.46 ± 0.00 (20.2) | |
| Total | 1.42 ± 0.40 (100) | 0.72 ± 0.21 (100) | 2.28 ± 0.01 (100) | |
| CWD of red pine | I | 3.05 ± 1.77 (31.7) | 1.57 ± 0.91 (32.4) | 4.70 ± 2.72 (22.9) |
| II | 2.10 ± 0.77 (21.8) | 1.05 ± 0.38 (21.7) | 5.06 ± 1.85 (24.7) | |
| III | 3.83 ± 0.82 (39.8) | 1.92 ± 0.41 (39.7) | 9.02 ± 1.93 (44.0) | |
| IV | 0.31 ± 0.23 (3.2) | 0.15 ± 0.11 (3.1) | 0.96 ± 0.07 (4.7) | |
| CWD of deciduous species 3 | I | 0.01 ± 0.01 (0.1) | 0.01 ± 0.01 (0.2) | 0.02 ± 0.02 (0.1) |
| II | 0.08 ± 0.02 (0.8) | 0.07 ± 0.01 (1.4) | 0.32 ± 0.10 (1.6) | |
| III | 0.08 ± 0.01 (0.8) | 0.08 ± 0.02 (1.7) | 0.32 ± 0.07 (1.6) | |
| IV | 0.01 ± 0.01 (0.1) | 0.00 ± 0.00 (0.0) | 0.07 ± 0.07 (0.3) | |
| Total | 9.63 ± 1.28 (100) | 4.84 ± 0.64 (100) | 20.48 ± 2.56 (100) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noh, N.J.; Yoon, T.K.; Kim, R.-H.; Bolton, N.W.; Kim, C.; Son, Y. Carbon and Nitrogen Accumulation and Decomposition from Coarse Woody Debris in a Naturally Regenerated Korean Red Pine (Pinus densiflora S. et Z.) Forest. Forests 2017, 8, 214. https://doi.org/10.3390/f8060214
Noh NJ, Yoon TK, Kim R-H, Bolton NW, Kim C, Son Y. Carbon and Nitrogen Accumulation and Decomposition from Coarse Woody Debris in a Naturally Regenerated Korean Red Pine (Pinus densiflora S. et Z.) Forest. Forests. 2017; 8(6):214. https://doi.org/10.3390/f8060214
Chicago/Turabian StyleNoh, Nam Jin, Tae Kyung Yoon, Rae-Hyun Kim, Nicholas W. Bolton, Choonsig Kim, and Yowhan Son. 2017. "Carbon and Nitrogen Accumulation and Decomposition from Coarse Woody Debris in a Naturally Regenerated Korean Red Pine (Pinus densiflora S. et Z.) Forest" Forests 8, no. 6: 214. https://doi.org/10.3390/f8060214
APA StyleNoh, N. J., Yoon, T. K., Kim, R.-H., Bolton, N. W., Kim, C., & Son, Y. (2017). Carbon and Nitrogen Accumulation and Decomposition from Coarse Woody Debris in a Naturally Regenerated Korean Red Pine (Pinus densiflora S. et Z.) Forest. Forests, 8(6), 214. https://doi.org/10.3390/f8060214







