Tree Height-Diameter Relationships in the Alpine Treeline Ecotone Compared with Those in Closed Forests on Changbai Mountain, Northeastern China
Abstract
:1. Introduction
2. Methods
2.1. Study Area and Climate
2.2. Site Description and Field Investigation
2.3. Data Analysis
3. Results
3.1. Height and Diameter Growth of Treeline Birch
3.2. Stature Change in Treeline Birch from 2006 to 2013
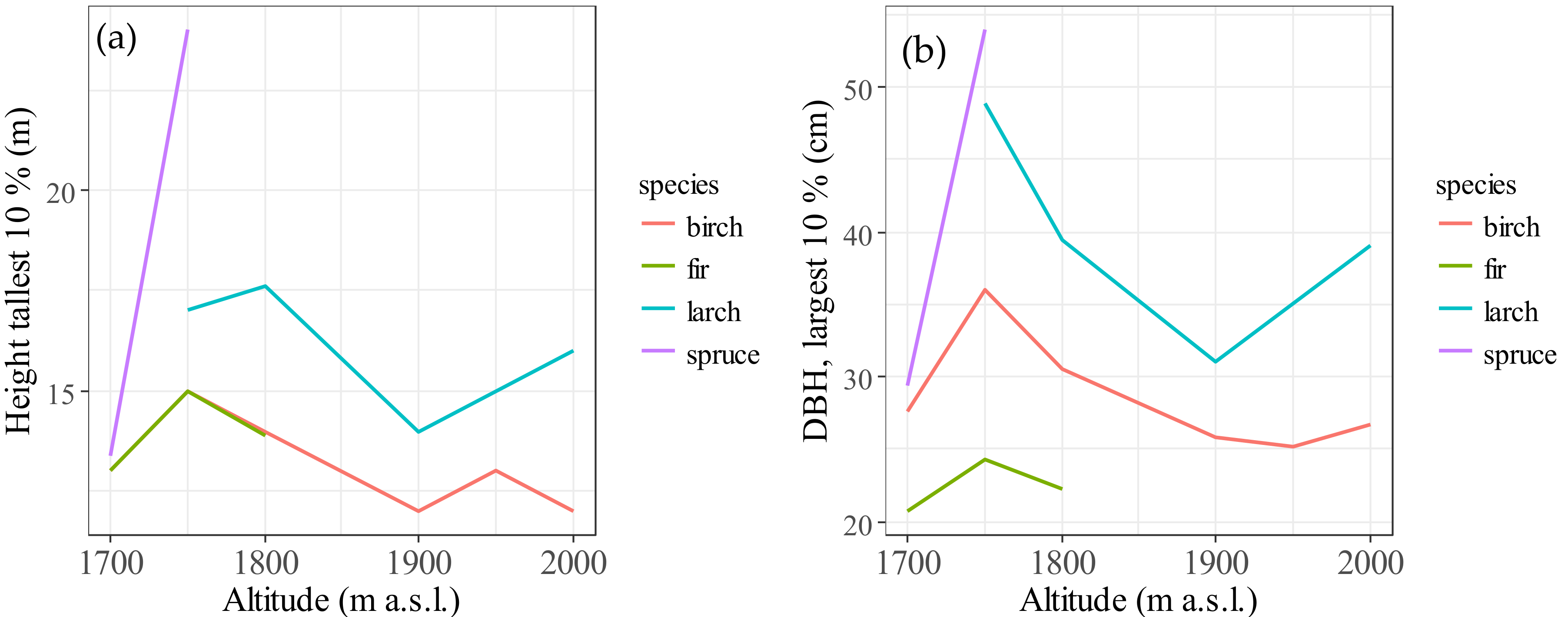
3.3. Change in Stature of Birch Along Altitudinal Gradients
3.4. Allometric Height-Diameter Growth for Species at Different Altitudes
4. Discussion
4.1. Stature of Birch Growing Along an Altitudinal Gradient
4.2. Species-Specific Height-Diameter Relationship
4.3. Stature Change of Birch at the Treeline over Time
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holtmeier, F.K. Mountain Timberlines. Ecology, Patchiness, and Dynamics; Springer: Berlin, Germany, 2009. [Google Scholar]
- Körner, C. Alpine Treelines; Springer: Basel, Switzerland, 2012. [Google Scholar]
- Körner, C.; Paulsen, J. A world-wide study of high altitude treeline temperatures. J. Biogeogr. 2004, 31, 713–732. [Google Scholar] [CrossRef]
- Körner, C. A re-assessment of high elevation treeline positions and their explanation. Oecologia 1998, 115, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Carbon limitation in trees. J. Ecol. 2003, 91, 4–17. [Google Scholar] [CrossRef]
- Saltre, F.; Bentaleb, I.; Favier, C.; Jolly, D. The role of temperature on treeline migration for an eastern african mountain during the last glacial maximum. Clim. Chang. 2013, 118, 901–918. [Google Scholar] [CrossRef]
- Devi, N.; Hagedorn, F.; Moiseev, P.; Bugmann, H.; Shiyatov, S.; Mazepa, V.; Rigling, A. Expanding forests and changing growth forms of siberian larch at the polar urals treeline during the 20th century. Glob. Chang. Biol. 2008, 14, 1581–1591. [Google Scholar] [CrossRef]
- Hagedorn, F.; Shiyatov, S.G.; Mazepa, V.S.; Devi, N.M.; Grigor’ev, A.A.; Bartysh, A.A.; Fomin, V.V.; Kapralov, D.S.; Terent’ev, M.; Bugman, H.; et al. Treeline advances along the urals mountain range—Driven by improved winter conditions? Glob. Chang. Biol. 2014, 20, 3530–3543. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Yu, D.P.; Zhou, L.; Zhou, W.M.; Wu, Z.J.; Guo, Y.; Bao, Y.; Meng, Y.Y.; Dai, L.M. Spatial patterns of a treeline Betula ermanii cham. Population on the north slope of Changbai mountain. Acta Ecol. Sin. 2015, 35, 116–124. [Google Scholar]
- Körner, C. Climatic treelines: Conventions, global patterns, causes. Erdkunde 2007, 61, 316–324. [Google Scholar] [CrossRef]
- Scherrer, D.; Korner, C. Infra-red thermometry of alpine landscapes challenges climatic warming projections. Glob. Chang. Biol. 2010, 16, 2602–2613. [Google Scholar] [CrossRef]
- Fajardo, A.; Piper, F.I. An experimental approach to explain the southern andes elevational treeline. Am. J. Bot. 2014, 101, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Gamache, I.; Payette, S. Height growth response of tree line black spruce to recent climate warming across the forest-tundra of eastern Canada. J. Ecol. 2004, 92, 835–845. [Google Scholar] [CrossRef]
- Li, M.H.; Yang, J.; Kräuchi, N. Growth responses ofpicea abiesandlarix deciduato elevation in subalpine areas of Tyrol, Austria. Can. J. For. Res. 2003, 33, 653–662. [Google Scholar] [CrossRef]
- Li, M.H.; Yang, J. Effects of microsite on growth of pinus cembra in the subalpine zone of the Austrian Alps. Ann. For. Sci. 2004, 61, 319–325. [Google Scholar] [CrossRef]
- Wang, X.; Fang, J.; Tang, Z.; Zhu, B. Climatic control of primary forest structure and DBH–height allometry in Northeast China. For. Ecol. Manag. 2006, 234, 264–274. [Google Scholar] [CrossRef]
- Körner, C. When it gets cold, plant size matters—A comment on tree line. J. Veg. Sci. 2016, 27, 6–7. [Google Scholar] [CrossRef]
- Takahashi, K.; Tokumitsu, Y.; Yasue, K. Climatic factors affecting the tree-ring width of Betula ermanii at the timberline on mount norikura, central Japan. Ecol. Res. 2005, 20, 445–451. [Google Scholar] [CrossRef]
- Yu, D.; Wang, Q.; Liu, J.; Zhou, W.; Qi, L.; Wang, X.; Zhou, L.; Dai, L. Formation mechanisms of the alpine erman’s birch (Betula ermanii) treeline on Changbai mountain in Northeast China. Trees 2014, 28, 935–947. [Google Scholar] [CrossRef]
- Yu, D.; Wang, G.G.; Dai, L.; Wang, Q. Dendroclimatic analysis of Betula ermanii forests at their upper limit of distribution in Changbai mountain, Northeast China. For. Ecol. Manag. 2007, 240, 105–113. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, X.; Gao, L.; Jiang, Q. Climatic response of Betula ermanii tree-ring growth along an altitudinal gradient on the northern slope of the Changbai mountains. Chin. J. Appl. Environ. Biol. 2013, 19, 929. [Google Scholar] [CrossRef]
- Zong, S.W.; Wu, Z.F.; Du, H.B. Study on climate change in alpine tundra of the Changbai mountain in growing season in recent 52 years. Arid Zone Res. 2013, 30, 41–49. [Google Scholar]
- Muggeo, V.M.R. Estimating regression models with unknown break-points. Stat. Med. 2003, 22, 3055–3071. [Google Scholar] [CrossRef] [PubMed]
- Crawley, M.J. The R Book; John Wiley & Sons: Chichester, UK, 2007. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Paulsen, J.; Weber, U.M.; Korner, C. Tree growth near treeline: Abrupt or gradual reduction with altitude? Arct. Antarct. Alp. Res. 2000, 32, 14–20. [Google Scholar] [CrossRef]
- Körner, C. Treelines will be understood once the functional difference between a tree and a shrub is. Ambio 2012, 41, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Antonucci, S.; Rossi, S.; Deslauriers, A.; Lombardi, F.; Marchetti, M.; Tognetti, R. Synchronisms and correlations of spring phenology between apical and lateral meristems in two boreal conifers. Tree Physiol. 2015, 35, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.Z.; Bao, X.C.; Li, C.G. Structural characteristics of certain dominant species on the northern slope of Changbai mountain in Jilin province. J. Plant Ecol. 1964, 2, 207–225. [Google Scholar]
- Martínez, A.J.; López-Portillo, J. Allometry of prosopis glandulosa var. Torreyana along a topographic gradient in the Chihuahuan desert. J. Veg. Sci. 2003, 14, 111–120. [Google Scholar]
- Ryan, M.G.; Phillips, N.; Bond, B.J. The hydraulic limitation hypothesis revisited. Plant Cell Environ. 2006, 29, 367–381. [Google Scholar] [CrossRef] [PubMed]
- Rais, A.; van de Kuilen, J.W.G.; Pretzsch, H. Growth reaction patterns of tree height, diameter, and volume of douglas-fir (Pseudotsuga menziesii (Mirb.) Franco) under acute drought stress in southern germany. Eur. J. For. Res. 2014, 133, 1043–1056. [Google Scholar]
- Callaway, R.M.; Delucia, E.H.; Schlesinger, W.H. Biomass allocation of montane and desert ponderosa pine—An analog for response to climate-change. Ecology 1994, 75, 1474–1481. [Google Scholar] [CrossRef]
- Hulshof, C.M.; Swenson, N.G.; Weiser, M.D. Tree height-diameter allometry across the United States. Ecol. Evol. 2015, 5, 1193–1204. [Google Scholar] [CrossRef] [PubMed]
- Culmsee, H.; Christoph, L.; Gerald, M.; Ramadhanil, P. Forest aboveground biomass along an elevational transect in Sulawesi, Indonesia, and the role of fagaceae in tropical montane rain forests. J. Biogeogr. 2010, 37, 960–974. [Google Scholar] [CrossRef]
- Chi, C.-H.; Ryan, W.M.; Chung-Te, C.; Chengyang, Z.; Zhijie, Y.; Jyh-Min, C.; Teng-Chiu, L. Typhoon disturbance mediates elevational patterns of forest structure, but not species diversity, in humid monsoon Asia. Ecosystems 2015, 18, 1410–1423. [Google Scholar] [CrossRef]
- Van Bloem, S.J.; Murphy, P.G.; Lugo, A.E. A link between hurricane-induced tree sprouting, high stem density and short canopy in tropical dry forest. Tree Physiol. 2007, 27, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Hiemstra, C.A.; Glen, E.L.; William, A.R. Snow redistribution by wind and interactions with vegetation at upper treeline in the medicine bow mountains, Wyoming, USA. Arct. Antarct. Alp. Res. 2002, 34, 262–273. [Google Scholar] [CrossRef]
- Brown, J.H.; West, G.B.; Enquist, B.J. Scaling in biology: Patterns and processes, causes and consequences. In Scaling in Biology; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Grime, J.P. Plant Strategies, Vegetation Processes, and Ecosystem Properties; Wiley: New York, NY, USA, 2002. [Google Scholar]
- Niklas, K.J.; Spatz, H.C. Allometric theory and the mechanical stability of large trees: Proof and conjecture. Am. J. Bot. 2006, 93, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Liu, S.R.; Berninger, F. Picea seedlings show apparent acclimation to drought with increasing altitude in the eastern himalaya. Trees Struct. Funct. 2004, 18, 277–283. [Google Scholar] [CrossRef]
- Loretta, G. Plant phenotypic plasticity in response to environmental factors. Adv. Bot. 2014, 2014, 1–17. [Google Scholar]
- Bretharte, M.S.; Shaver, G.R.; Zoerner, J.P.; Johnstone, J.F.; Wagner, J.L.; Chavez, A.S.; Gunkelman, R.F.; Lippert, S.C.; Laundre, J.A. Developmental plasticity allows betula nana to dominate tundra subjected to an altered environment. Ecology 2001, 82, 18–32. [Google Scholar] [CrossRef]
- Ellum, D.S.; Ashton, P.M.S.; Berlyn, G.P. Between and within genera comparisons of morphological plasticity for betula and acer seedlings grown under varying light conditions. In Proceedings of the New England Society of American Foresters 84th Winter Meeting, Quebec, QC, Canada, 23–26 March 2004; pp. 33–36. [Google Scholar]
- Xu, Z.W.; Wang, X.D.; Han, W.H.; Wang, G.J.; Liu, Q.J. Comparative analysis of laris olgensis and betula ermanii associated with environment on north slopes treeline of the Changbai mountains. Sci. Geogr. Sin. 2014, 34, 748–756. [Google Scholar]

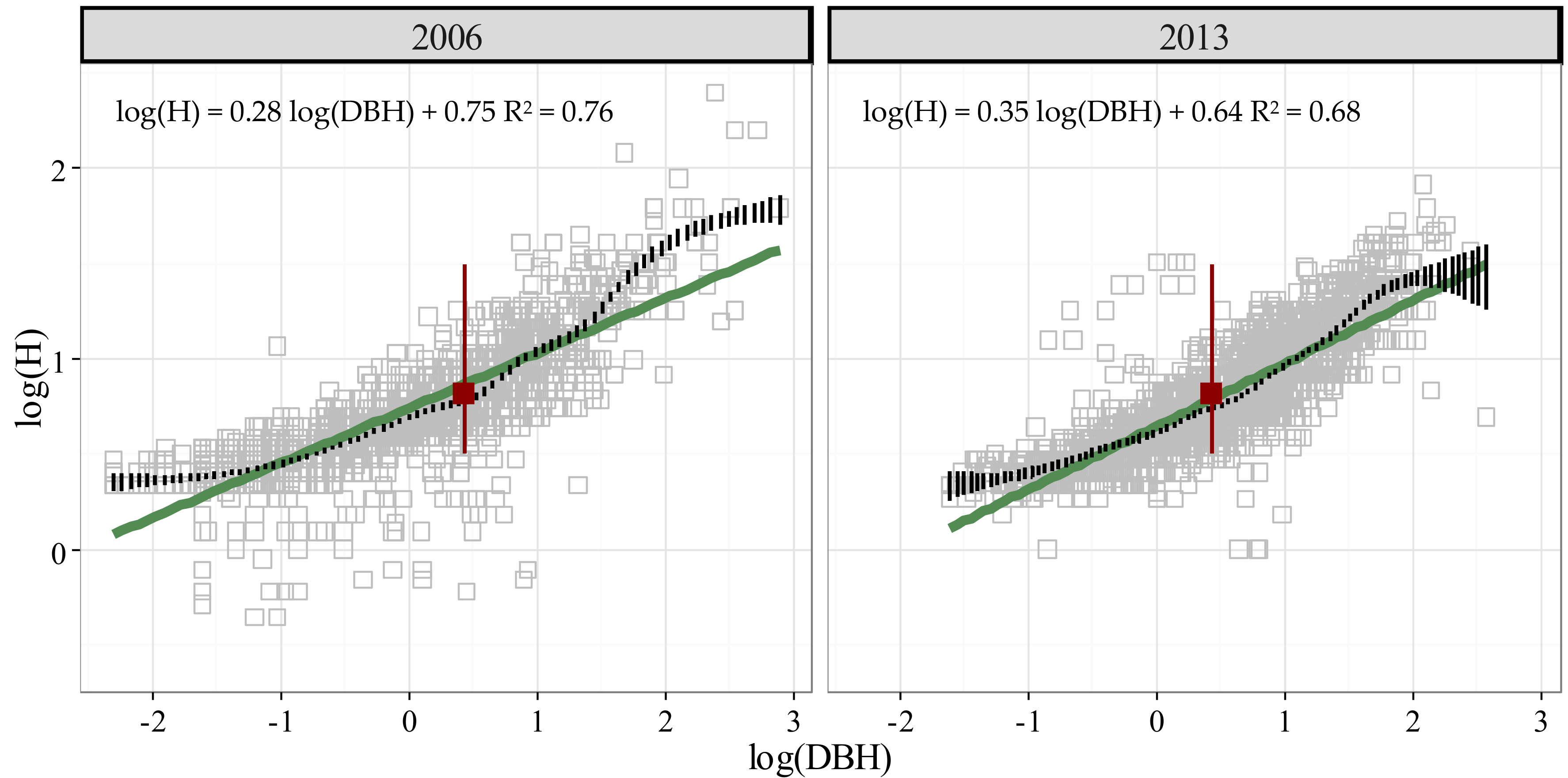
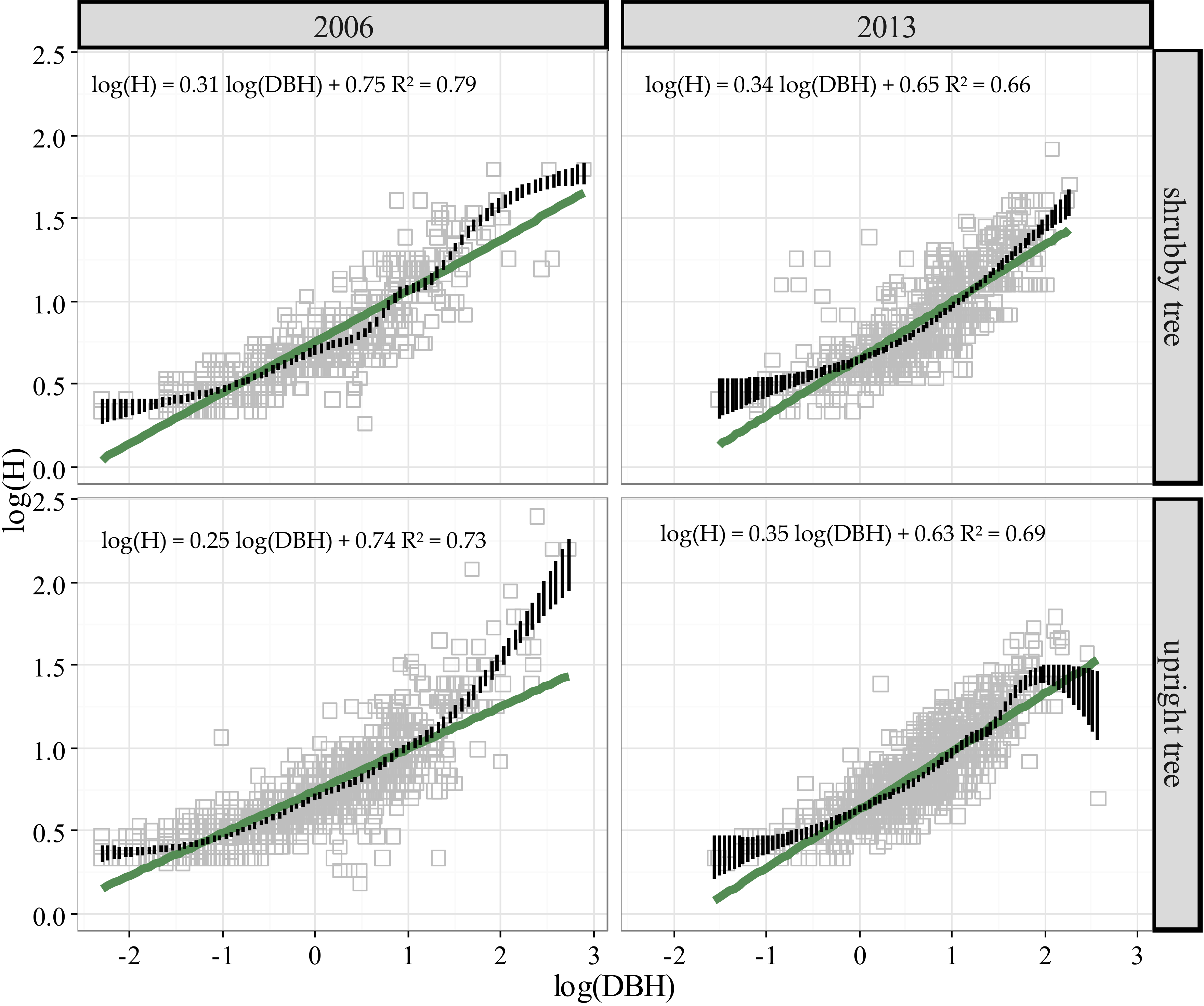
| Attributes | Tree Form Class * | |||
|---|---|---|---|---|
| Upright Tree | Shrubby Tree | Sapling | Population | |
| Number of trees | 1038 | 886 | 799 | 2723 |
| H2006 | 2.10 (0.79) | 2.38 (1.05) | 0.97 (0.23) | 1.92 (0.99) |
| H2013 | 2.44 (0.71) | 2.60 (0.81) | 1.64 (0.52) | 2.26 (0.80) |
| ΔH | 0.48 (0.44) | 0.56 (0.53) | 0.68 (0.52) | 0.56 (0.50) |
| DBH2006 | 1.3 (1.5) | 2.0 (2.8) | - | 1.6 (2.3) |
| DBH2013 | 2.4 (1.7) | 2.6 (1.8) | 1.0 (0.9) ** | 2.2 (1.7) |
| ΔDBH | 1.2 (0.8) | 1.3 (1.2) | - | 1.2 (1.0) |
| BD2006 | - | - | 1.53 (0.57) | - |
| BD2013 | - | - | 1.95 (0.64) | - |
| ΔBD | - | - | 0.59 (0.43) | - |
| Dataset | Altitude (m a.s.l.) | Species | n | Model Parameters | R2 | |
|---|---|---|---|---|---|---|
| log(a) | b | |||||
| Below treeline | 1700 | birch | 36 | 0.94 | 0.46 | 0.52 |
| fir | 165 | 0.08 | 0.81 | 0.89 | ||
| 1750 | birch | 97 | 0.65 | 0.53 | 0.63 | |
| spruce | 121 | 0.19 | 0.75 | 0.88 | ||
| larch | 29 | 0.62 | 0.57 | 0.85 | ||
| fir | 72 | 0.13 | 0.78 | 0.76 | ||
| 1800 | birch | 80 | 0.75 | 0.53 | 0.69 | |
| 1900 | birch | 85 | 0.38 | 0.65 | 0.64 | |
| larch | 47 | 0.33 | 0.67 | 0.58 | ||
| 1950 | birch | 56 | 0.60 | 0.60 | 0.43 | |
| 2000 | birch | 39 | 0.89 | 0.43 | 0.48 | |
| Treeline ** | >2000 | upright birch | 117 | 0.84 | 0.36 | 0.52 |
| shrubby birch | 248 | 0.93 | 0.30 | 0.63 | ||
| Altitude a.s.l. (m) | Species | Range of H (m) | Average H (m) | Average DBH (cm) | DBH/H | Number of Trees |
|---|---|---|---|---|---|---|
| 1700 | birch | 3–16 | 15.3 | 8.9 | 0.73 | 36 |
| spruce | 3–15 | 14.1 | 8.1 | 0.66 | 9 | |
| fir | 2–18 | 11.1 | 7.5 | 0.75 | 165 | |
| 1750 | larch | 4–17 | 35.8 | 14.1 | 0.45 | 29 |
| birch | 3–23 | 19.9 | 9.6 | 0.59 | 97 | |
| spruce | 2–29 | 27.8 | 14.2 | 0.61 | 121 | |
| fir | 2–22 | 13.3 | 8.8 | 0.70 | 72 | |
| 1800 | larch | 13–18 | 30.1 | 15.6 | 0.44 | 5 |
| birch | 3–16 | 18.1 | 9.6 | 0.63 | 80 | |
| fir | 2.5–17 | 15.5 | 8.7 | 0.62 | 32 | |
| 1900 | larch | 2–17 | 20.8 | 10.7 | 0.56 | 47 |
| birch | 3–17 | 17.1 | 9.2 | 0.57 | 85 | |
| 1950 | birch | 3–14 | 19.8 | 11.2 | 0.58 | 56 |
| 2000 | larch | 3–16 | 29.4 | 12.9 | 0.56 | 15 |
| birch | 3–13 | 16.8 | 8.2 | 0.60 | 39 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Yu, D.; Wang, S.; Lewis, B.J.; Zhou, W.; Zhou, L.; Dai, L.; Lei, J.-P.; Li, M.-H. Tree Height-Diameter Relationships in the Alpine Treeline Ecotone Compared with Those in Closed Forests on Changbai Mountain, Northeastern China. Forests 2017, 8, 132. https://doi.org/10.3390/f8040132
Wang X, Yu D, Wang S, Lewis BJ, Zhou W, Zhou L, Dai L, Lei J-P, Li M-H. Tree Height-Diameter Relationships in the Alpine Treeline Ecotone Compared with Those in Closed Forests on Changbai Mountain, Northeastern China. Forests. 2017; 8(4):132. https://doi.org/10.3390/f8040132
Chicago/Turabian StyleWang, Xiaoyu, Dapao Yu, Shoule Wang, Bernard J. Lewis, Wangming Zhou, Li Zhou, Limin Dai, Jing-Pin Lei, and Mai-He Li. 2017. "Tree Height-Diameter Relationships in the Alpine Treeline Ecotone Compared with Those in Closed Forests on Changbai Mountain, Northeastern China" Forests 8, no. 4: 132. https://doi.org/10.3390/f8040132
APA StyleWang, X., Yu, D., Wang, S., Lewis, B. J., Zhou, W., Zhou, L., Dai, L., Lei, J.-P., & Li, M.-H. (2017). Tree Height-Diameter Relationships in the Alpine Treeline Ecotone Compared with Those in Closed Forests on Changbai Mountain, Northeastern China. Forests, 8(4), 132. https://doi.org/10.3390/f8040132






