The Effect of Re-Planting Trees on Soil Microbial Communities in a Wildfire-Induced Subalpine Grassland
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site and Soil Sampling
2.2. Biochemical Assay
2.3. Statistical Analysis
3. Results
3.1. Soil Properties and Microbial Biomass
3.2. Soil Enzyme Activities
3.3. PLFA Biomarkers
3.4. Principal Component Analysis
3.5. DGGE Analysis
3.6. Correlation of Soil Properties and Microbial Communities
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smithwick, E.A.H.; Naithani, K.J.; Balser, T.C.; Romme, W.H.; Turner, M.G. Post-fire spatial patterns of soil nitrogen mineralization and microbial abundance. PLoS ONE 2012, 7, e50597. [Google Scholar] [CrossRef] [PubMed]
- Pourreza, M.; Hosseini, S.M.; Sinegani, A.A.S.; Matinizadeh, M.; Dick, W.A. Soil microbial activity in response to fire severity in Zagros oak (Quercus brantii Lindl.) forests, Iran, after one year. Geoderma 2014, 213, 95–102. [Google Scholar] [CrossRef]
- Dickens, S.J.M.; Allen, E.B. Soil nitrogen cycling is resilient to invasive annuals following restoration of coastal sage scrub. J. Arid Environ. 2014, 110, 12–18. [Google Scholar] [CrossRef]
- Imberger, K.T.; Chiu, C.Y. Spatial change of soil fungal and bacterial biomass from a sub-alpine coniferous forest to grassland in a humid, sub-tropical region. Biol. Fertil. Soils 2001, 33, 105–110. [Google Scholar] [CrossRef]
- Tavşanoğlu, Ç.; Gürkan, B. Long-term post-fire dynamics of co-occurring woody species in Pinus brutia forests: The role of regeneration mode. Plant Ecol. 2014, 215, 355–365. [Google Scholar] [CrossRef]
- Siles, J.A.; Rachid, C.T.C.C.; Sampedro, I.; García-Romera, I.; Tidje, J.M. Microbial diversity of a Mediterranean soil and its changes after biotransformed dry olive residue amendment. PLoS ONE 2014, 9, e103035. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Zhu, B. Diversity and abundance of soil fauna as influenced by long-term fertilization in cropland of purple soil, China. Soil Tillage Res. 2015, 146, 39–46. [Google Scholar] [CrossRef]
- Bender, S.F.; Heijden, M.G. Soil biota enhance agricultural sustainability by improving crop yield, nutrient uptake and reducing nitrogen leaching losses. J. Appl. Ecol. 2015, 52, 228–239. [Google Scholar] [CrossRef]
- Hodge, A.; Storer, K. Arbuscular mycorrhiza and nitrogen: Implications for individual plants through to ecosystems. Plant Soil 2015, 386, 1–19. [Google Scholar] [CrossRef]
- Jia, G.M.; Cao, J.; Wang, C.; Wang, G. Microbial biomass and nutrients in soil at the different stages of secondary forest succession in Ziwulin, northwest China. For. Ecol. Manag. 2005, 217, 117–125. [Google Scholar] [CrossRef]
- Hedo, J.; Lucas-Borja, M.; Wic, C.; Andrés-Abellán, M.; Las Heras, J.D. Soil microbiological properties and enzymatic activities of long-term post-fire recovery in dry and semiarid Aleppo pine (Pinus halepensis M.) forest stands. Solid Earth 2015, 6, 243–252. [Google Scholar] [CrossRef]
- Wang, C.; Wang, G.; Wang, Y.; Rafique, R.; Ma, L.; Hu, L.; Luo, Y. Fire alters vegetation and soil microbial community in alpine meadow. Land Degrad. Dev. 2016, 27, 1379–1390. [Google Scholar] [CrossRef]
- Isobe, K.; Otsuka, S.; Sudiana, I.; Nurkanto, A.; Senoo, K. Community composition of soil bacteria nearly a decade after a fire in a tropical rainforest in East Kalimantan, Indonesia. J. Gen. Appl. Microbiol. 2009, 55, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.T.; Lin, C.P.; Chaw, S.M.; Whitman, W.B.; Coleman, D.C.; Chiu, C.Y. Bacterial community of very wet and acidic subalpine forest and fire-induced grassland soils. Plant Soil 2010, 332, 417–427. [Google Scholar] [CrossRef]
- Frostegård, Å.; Tunlid, A.; Bååth, E. Use and misuse of PLFA measurements in soils. Soil Biol. Biochem. 2011, 43, 1621–1625. [Google Scholar] [CrossRef]
- Farrell, M.; Griffith, G.W.; Hobbs, P.J.; Perkins, W.T.; Jones, D.L. Microbial diversity and activity are increased by compost amendment of metal-contaminated soil. FEMS Microbiol. Ecol. 2010, 71, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, Y.; Bao, Z.; Kurose, D.; Okada, H.; Takemoto, S.; Sawada, A.; Nagase, H.; Takano, M.; Murakami, H.; Koitabashi, M.; et al. Community structure, diversity, and species dominance of bacteria, fungi, and nematodes from naturally and conventionally farmed soil: A case study on Japanese apple orchards. Org. Agric. 2015, 5, 11–28. [Google Scholar] [CrossRef]
- Chiu, C.Y.; Chen, T.H.; Imberger, K.; Tian, G. Particle size fractionation of fungal and bacterial biomass in subalpine grassland and forest soils. Geoderma 2006, 130, 265–271. [Google Scholar] [CrossRef]
- McLean, E.O. Soil pH and lime requirement. In Methods of Soil Analysis, Part 2 Chemical and Microbiological Properties; Page, A.L., Ed.; ASA: Madison, WI, USA, 1982; pp. 199–224. [Google Scholar]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Wu, J.; Joergensen, R.G.; Pommerening, B.; Chaussod, R.; Brookes, P.C. Measurement of soil microbial biomass C by fumigation-extraction-an automated procedure. Soil Biol. Biochem. 1990, 22, 1167–1169. [Google Scholar] [CrossRef]
- Amato, M.; Ladd, J.N. Assay for microbial biomass based on ninhydrin-reactive nitrogen in extracts of fumigated soils. Soil Biol Biochem. 1988, 20, 107–114. [Google Scholar] [CrossRef]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 8, 199–202. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Schinner, F.; von Mersi, W. Xylanase-, CM-cellulase- and invertase activity in soil: An improved method. Soil Biol. Biochem. 1990, 22, 511–515. [Google Scholar] [CrossRef]
- Frostegård, A.; Tunlid, A.; Baath, E. Phospholipid fatty acid composition, biomass, and activity of microbial communities from two soil types exposed to different heavy metals. Appl. Environ. Microbiol. 1993, 59, 3605–3617. [Google Scholar] [PubMed]
- Chang, E.H.; Chen, C.T.; Chen, T.H.; Chiu, C.Y. Soil microbial communities and activities in sand dunes of subtropical coastal forests. Appl. Soil Ecol. 2011, 49, 256–262. [Google Scholar] [CrossRef]
- Zogg, G.P.; Zak, D.R.; Ringleberg, D.B.; MacDonald, N.W.; Pregitzer, K.S.; White, D.C. Compositional and functional shifts in microbial communities due to soil warming. Soil Sci. Soc. Am. J. 1997, 61, 475–481. [Google Scholar] [CrossRef]
- Zelles, L. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterization of microbial communities in soil: A review. Biol. Fertil. Soils 1999, 29, 111–129. [Google Scholar] [CrossRef]
- Nübel, U.; Engelen, B.; Felske, A.; Snaidr, J.; Wieshuber, A.; Amann, R.I.; Ludwig, W.; Backhaus, H. Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. J. Bacteriol. 1996, 178, 5636–5643. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, J.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Suzuki, C.; Nagaoka, K.; Shimada, A.; Takenaka, M. Bacterial communities are more dependent on soil type than fertilizer type, but the reverse is true for fungal communities. Soil Sci. Plant Nutr. 2009, 55, 80–90. [Google Scholar] [CrossRef]
- Umemura, M.; Takenaka, C. Changes in chemical characteristics of surface soils in hinoki cypress (Chamaecyparis obtusa) forests induced by the invasion of exotic Moso bamboo (Phyllostachys pubescens) in central Japan. Plant Species Biol. 2015, 30, 72–79. [Google Scholar] [CrossRef]
- Yu, X.; Liu, X.; Zhao, Z.; Liu, J.; Zhang, S. Effect of Monospecific and Mixed Sea-Buckthorn (Hippophae rhamnoides) Plantations on the Structure and Activity of Soil Microbial Communities. PLoS ONE 2015, 10, e0117505. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Ingram, L.J.; Vance, G.F.; Stahl, P.D. Soil processes and microbial community structures in 45- and 135-year-old lodgepole pine stands. Can. J. For. Res. 2009, 39, 2263–2271. [Google Scholar] [CrossRef]
- Campbell, C.D.; Cameron, C.M.; Bastias, B.A.; Chen, C.R.; Cairney, J.W.G. Long term repeated burning in a wet sclerophyll forest reduces fungal and bacterial biomass and responses to carbon substrates. Soil Biol. Biochem. 2008, 40, 2246–2252. [Google Scholar] [CrossRef]
- Saleem, M. Microbiome Community Ecology: Fundamentals and Applications; Springer International Publishing: New York, NY, USA, 2015. [Google Scholar]
- Hamman, S.T.; Burke, I.C.; Stromberger, M.E. Relationships between microbial community structure and soil environmental conditions in a recently burn system. Soil Biol. Biochem. 2007, 39, 1703–1711. [Google Scholar] [CrossRef]
- Hebel, C.L.; Smith, J.E.; Cromack, J.K. Invasive plant species and soil microbial response to wildfire burn severity in the Cascade Range of Oregon. Appl. Soil Ecol. 2009, 42, 150–159. [Google Scholar] [CrossRef]
- Malmström, A.; Persson, T.; Ahlström, K.; Gongalsky, K.B.; Bengtsson, J. Dynamics of soil meso- and macrofauna during a 5-year period after clear-cut burning in a boreal forest. Appl. Soil Ecol. 2009, 43, 61–74. [Google Scholar] [CrossRef]
- Pascual, J.A.; Hernandez, T.; Ayuso, M.; Garcia, C. Changes in the microbial activity of arid soils amended with urban organic wastes. Biol. Fertil. Soils 1997, 24, 429–434. [Google Scholar] [CrossRef]
- Dangi, S.R.; Stahl, P.D.; Pendall, E.; Cleary, M.B.; Buyer, J.S. Recovery of soil microbial community structure after fire in a Sagebrush-grassland ecosystem. Land Degrad. Dev. 2010, 21, 423–432. [Google Scholar] [CrossRef]
- Grandy, A.S.; Strickland, M.S.; Lauber, C.L.; Bradford, M.A.; Fierer, N. The influence of microbial communities, management, and soil texture on soil organic matter chemistry. Geoderma 2009, 150, 278–286. [Google Scholar] [CrossRef]
- Mandal, A.; Patra, A.K.; Singh, D.; Swarup, A.; Masto, R.E. Effect of long-term application of manure and fertilizer on biological and biochemical activities in soil during crop development stages. Bioresour. Technol. 2007, 98, 3585–3592. [Google Scholar] [CrossRef] [PubMed]
- Chodak, M.; Niklinska, M. The effect of different tree species on the chemical and microbial properties of the reclaimed mine soils. Biol. Fertil. Soils 2010, 46, 555–566. [Google Scholar] [CrossRef]
- Dashtban, M.; Schraft, H.; Qin, W. Fungal bioconversion of lignocellulosic residues; Opportunities & Perspectives. Int. J. Biol. Sci. 2009, 5, 578–595. [Google Scholar] [PubMed]
- Ali, M.A.; Louche, J.; Legname, E.; Duchemin, M.; Plassard, C. Pinus pinaster seedlings and their fungal symbionts show high plasticity in phosphorus acquisition in acidic soils. Tree Physiol. 2009, 29, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Grierson, P.F.; Adams, M.A. Plant species affect acid phosphatase, ergosterol and microbial P in a Jarrah (Eucalyptus marginata Donn ex Sm.) forest in south-western Australia. Soil Biol. Biochem. 2000, 32, 1817–1827. [Google Scholar] [CrossRef]
- Vega, J.A.; Fontúrbel, T.; Merino, A.; Fernández, C.; Ferreiro, A.; Jiménez, E. Testing the ability of visual indicators of soil burn severity to reflect changes in soil chemical and microbial properties in pine forests and shrubland. Plant Soil 2013, 369, 73–91. [Google Scholar] [CrossRef]
- Scott, N.A.; Binkley, D. Foliage litter quality and annual net N mineralization: Comparison across North American forest sites. Oecologia 1997, 111, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Zornoza, R.; Guerrero, C.; Mataix-Solera, J.; Scow, K.M.; Arcenegui, V.; Mataix-Beneyto, J. Changes in soil microbial community structure following the abandonment of agricultural terraces in mountainous areas of Eastern Spain. Appl. Soil Ecol. 2009, 42, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.H.; Anacker, B.L.; Strauss, S.Y.; Burke, D.J. Soil microbial community variation correlates most strongly with plant species identity, followed by soil chemistry, spatial location and plant genus. AoB Plants 2015, 7, plv030. [Google Scholar] [CrossRef] [PubMed]
- Dilly, O.; Bartsch, S.; Rosenbrock, P.; Buscot, F.; Munch, J.C. Shifts in physiological capabilities of the microbiota during the decomposition of leaf litter in a black alder (Alnus glutinosa (Gaertn.) L.) forest. Soil Biol. Biochem. 2001, 33, 921–930. [Google Scholar] [CrossRef]
- Djukic, I.; Zehetner, F.; Mentler, A.; Gerzabek, M.H. Microbial community composition and activity in different Alpine vegetation zones. Soil Biol. Biochem. 2010, 42, 155–161. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis; Academic Press: San Diego, CA, USA, 1997. [Google Scholar]
- Allison, V.J.; Miller, R.M.; Jastrow, J.D.; Matamala, R.; Zak, D.R. Changes in soil microbial community structure in a tallgrass prairie chronosequence. Soil Sci. Soc. Am. J. 2005, 69, 1412–1421. [Google Scholar] [CrossRef]
- Parkash, V.; Aggarwal, A.; Sharma, S.; Sharma, D. Effect of endophytic mycorrhizae and fungal bioagent on the development and growth of Eucalyptus saligna seedlings. Bull. Natl. Inst. Ecol. 2005, 15, 127–131. [Google Scholar]
- Olsson, P.A.; Baath, E.; Jakobsen, I.; Soderstrom, B. The use of phospholipid and neutral lipid fatty acids to estimate biomass of arbuscular mycorrhizal fungi in soil. Mycol. Res. 1995, 99, 623–629. [Google Scholar] [CrossRef]
- Nichols, P.; Stulp, B.K.; Jones, J.G.; White, D.C. Comparison of fatty acid content and DNA homology of the filamentous gliding bacteria Vitreoscilla, Flexibacter, Filibacter. Arch. Microbiol. 1986, 146, 1–6. [Google Scholar] [CrossRef]
- Jonasson, S.; Michelsen, A.; Schmidt, I.K. Coupling of nutrient cycling and C dynamics in the Arctic, integration of soil microbial and plant processes. Appl. Soil Ecol. 1999, 11, 135–146. [Google Scholar] [CrossRef]
- Thoms, C.; Gattinger, A.; Jacob, M.; Thomas, F.M.; Gleixner, G. Direct and indirect effects of tree diversity drive soil microbial diversity in temperate deciduous forest. Soil Biol. Boichem. 2010, 42, 1558–1565. [Google Scholar] [CrossRef]
- Baldrian, P. Wood-inhabiting ligninolytic basidiomycetes in soils: Ecology and constraints for applicability in bioremediation. Fungal Ecol. 2008, 1, 4–12. [Google Scholar] [CrossRef]
- Bååth, E.; Frostegård, A.; Pennanen, T.; Fritze, H. Microbial community structure and pH response in relation to soil organic matter quality in wood-ash fertilized, clear-cut or burned coniferous forest soils. Soil Biol. Biochem. 1995, 27, 229–240. [Google Scholar] [CrossRef]
- Sterner, R.W.; Elsner, J.J. Ecological stoichiometry. In The Biology of Elements from Molecules to the Biosphere; Princeton University Press: Princeton, NJ, USA, 2002. [Google Scholar]
- Ingham, E.R.; Coleman, D.C.; Moore, J.C. An analysis of food-web structure and function in a shortgrass prairie, a mountain meadow, and a lodgepole pine forest. Biol. Fertil. Soils 1989, 8, 29–37. [Google Scholar] [CrossRef]
- Personeni, E.; Loiseau, P. How does the nature of living and dead roots affect the residence time of carbon in the root litter continuum? Plant Soil 2004, 267, 129–141. [Google Scholar] [CrossRef]
- Atlas, R.M.; Bartha, R. Microbial Ecology: Fundamentals and Applications; Benjamin/Cummings: Redwood City, CA, USA, 1998. [Google Scholar]
- Kourtev, P.S.; Ehrenfeld, J.G.; Haggblom, M. Experimental analysis of the effect of exotic and native plant species on the structure and function of soil microbial communities. Soil Biol. Biochem. 2003, 35, 895–905. [Google Scholar] [CrossRef]
- Chang, E.H.; Tian, G.; Chiu, C.Y. Soil microbial communities in natural and managed cloud montane forests. Forests 2017, 8, 33. [Google Scholar] [CrossRef]
- Chang, E.H.; Chiu, C.Y. Changes in soil microbial community structure and activity in a cedar plantation invaded by moso bamboo. Appl. Soil Ecol. 2015, 91, 1–7. [Google Scholar] [CrossRef]
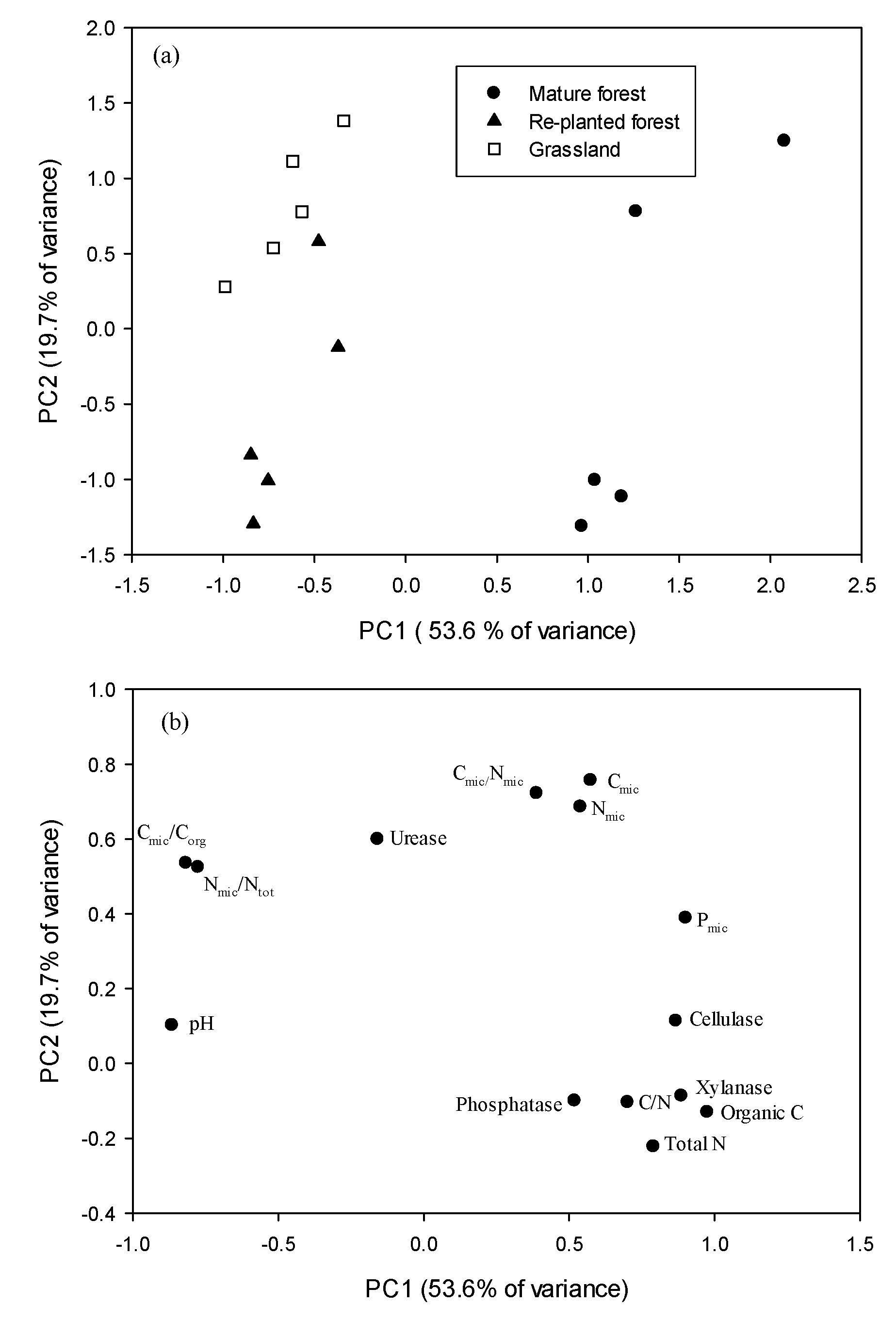
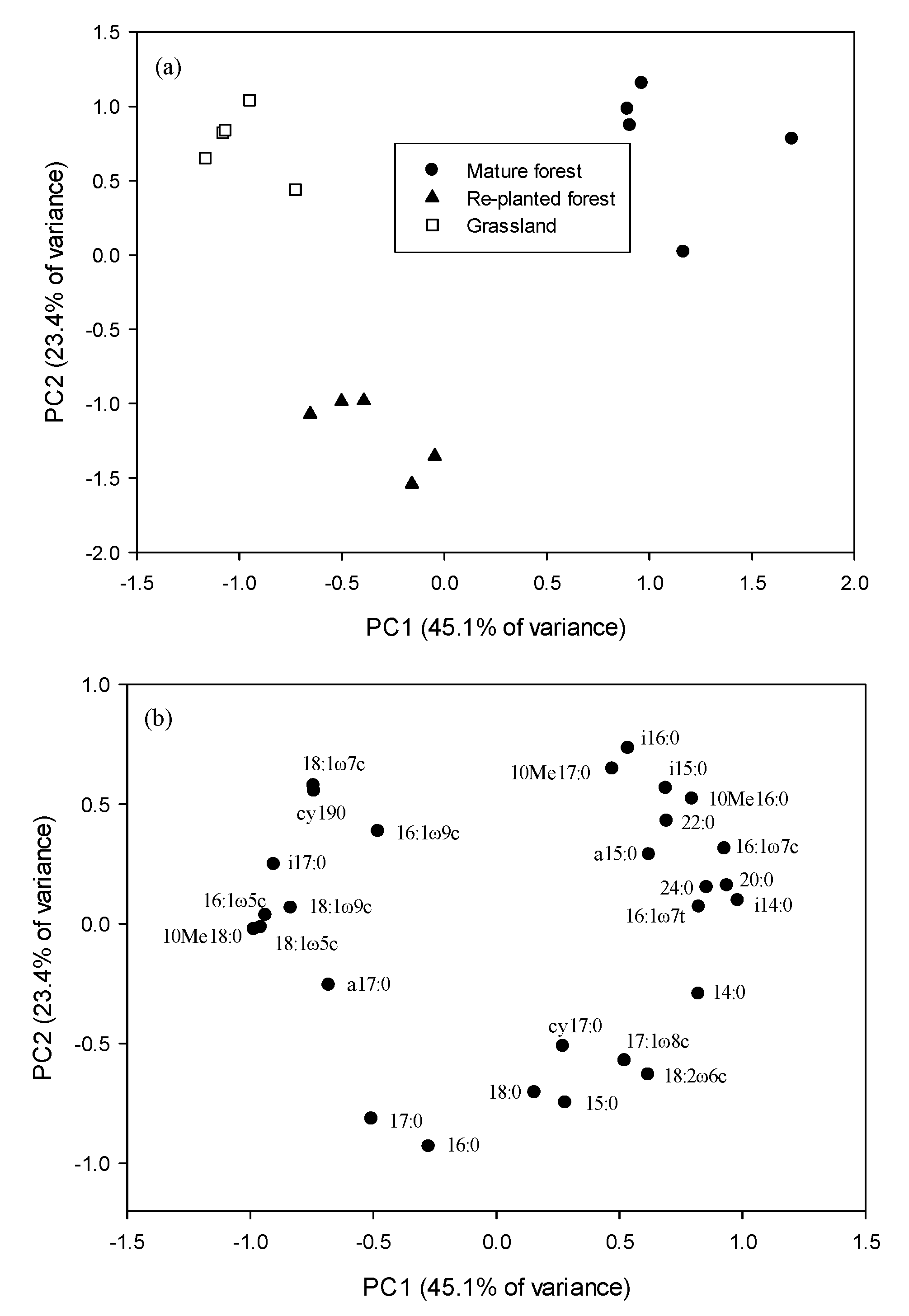
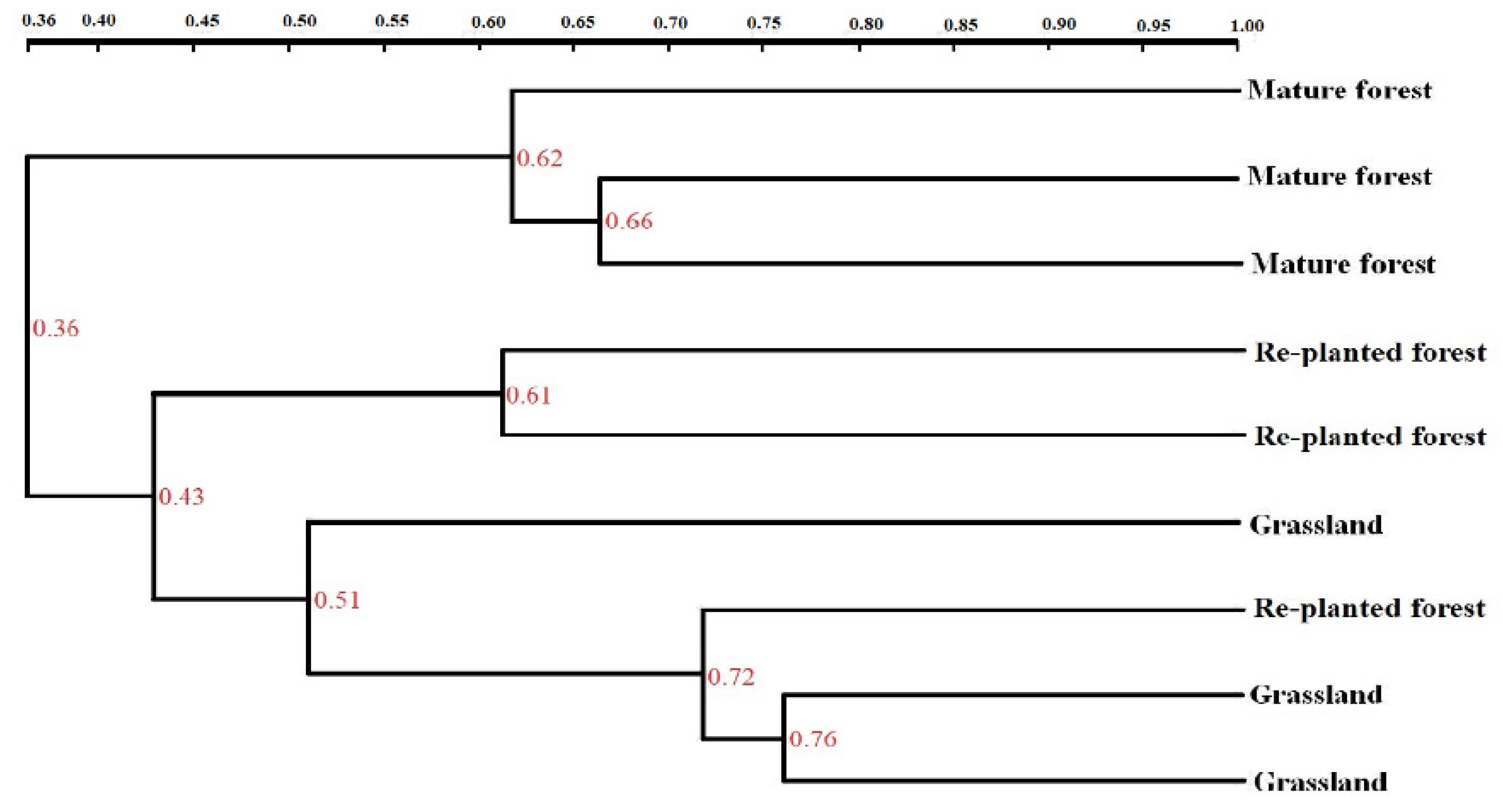

| Vegetation | pH | Organic C (g kg−1) | Total N (g kg−1) | Total P (mg kg−1) | Cmic (mg kg−1) | Nmic (mg kg−1) | Cmic/Corg (%) | Nmic/Ntot (%) |
|---|---|---|---|---|---|---|---|---|
| Grassland | 4.14 a | 106 c | 7.93 c | 606 a | 5505 a | 587 a | 5.19 a | 7.43 a |
| Re-planted forest | 4.20 a | 131 b | 9.82 b | 574 a | 4792 a | 557 a | 3.65 b | 5.67 b |
| Mature forest | 3.56 b | 472 a | 25.0 a | 650 a | 6165 a | 648 a | 1.30 c | 2.88 c |
| Vegetation | Cellulase (mg Glucose g−1 d−1) | Xylanase (mg Glucose g−1 d−1) | Urease (mmole NH4+-N g−1 h−1 ) | Acid Phosphatase (μg Nitrophenol g−1 h−1) |
|---|---|---|---|---|
| Grassland | 2.786 b | 5.252 b | 77.89 a | 1919 b |
| Re-planted forest | 0.867 c | 5.167 b | 17.52 c | 2189 b |
| Mature forest | 5.608 a | 10.98 a | 41.90 b | 2897 a |
| Vegetation | Total PLFA (nmol g−1 soil) | Bacteria | Fungi | VAM Fungi | Actinobacteria | G+ Bacteria | G− Bacteria | G+/G− | Fungi/Bacteria |
|---|---|---|---|---|---|---|---|---|---|
| Grassland | 389 c | 202 b | 18.3 b | 14.7 c | 31.6 b | 43.7 b | 91.1 b | 0.461 c | 0.091 b |
| Re-planted forest | 508 b | 223 b | 37.4 a | 18.6 b | 37.4 b | 51.7 b | 94.8 b | 0.568 b | 0.168 a |
| Mature forest | 789 a | 371 a | 52.3 a | 22.5 a | 53.7 a | 99.2 a | 131 a | 0.755 a | 0.187 a |
| Cellulase | Xylanase | Urease | Phosphatase | Total PLFA | Bacteria | Gram-Positive Bacteria (G+) | Gram-Negative Bacteria (G−) | G+/G− | Fungi | VAM Fungi | Bacteria/Fungi | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | 0.790 *** | 0.849 *** | −0.216 | 0.432 | 0.897 *** | 0.867 *** | 0.876 *** | 0.732 ** | 0.903 *** | 0.753 ** | 0.776 ** | 0.517 * |
| N | 0.569 * | 0.719 ** | −0.158 | 0.269 | 0.650 ** | 0.564 * | 0.598 * | 0.390 | 0.762 ** | 0.633 * | 0.505 | 0.529 * |
| P | 0.163 | 0.160 | 0.134 | 0.114 | 0.379 | 0.359 | 0.341 | 0.329 | 0.292 | 0.378 | 0.294 | 0.213 |
| C/N | 0.682 ** | 0.641 * | −0.178 | 0.426 | 0.710 ** | 0.796 *** | 0.775 ** | 0.804 *** | 0.611 * | 0.398 | 0.733 ** | 0.116 |
| Cmic | 0.485 | 0.362 | 0.166 | 0.319 | 0.486 | 0.409 | 0.399 | 0.309 | 0.394 | 0.657 ** | 0.279 | 0.511 |
| Nmic | 0.422 | 0.363 | 0.117 | 0.197 | 0.461 | 0.393 | 0.385 | 0.300 | 0.391 | 0.592 * | 0.279 | 0.462 |
| Cmic/Corg | −0.587 * | −0.735 ** | 0.455 | −0.388 | −0.844 *** | −0.804 *** | −0.821 *** | −0.663 ** | −0.886 *** | −0.627 * | −0.820 *** | −0.458 |
| Nmic/Ntot | −0.517 * | −0.694 ** | 0.396 | −0.306 | −0.767 ** | −0.701 ** | −0.723 ** | −0.543 * | −0.828 *** | −0.619 * | −0.710 ** | −0.489 |
| Cmic/Nmic | 0.368 | 0.196 | 0.312 | 0.083 | 0.289 | 0.251 | 0.239 | 0.203 | 0.181 | 0.451 | 0.062 | 0.311 |
| Moisture | 0.440 | 0.597 * | −0.265 | 0.356 | 0.790 *** | 0.683 ** | 0.696 ** | 0.487 | 0.805 *** | 0.752 ** | 0.701 ** | 0.602 * |
| pH | −0.705 ** | −0.740 ** | 0.041 | −0.201 | −0.828 *** | −0.845 *** | −0.861 *** | −0.769 ** | −0.825 *** | −0.583 * | −0.751 ** | −0.336 |
| Cellulose | 1 | 0.886 *** | 0.062 | 0.541 * | 0.666 ** | 0.699 ** | 0.683 ** | 0.679 ** | 0.591 * | 0.580 * | 0.514 * | 0.318 |
| Xylanase | 0.886 *** | 1 | 0.035 | 0.473 | 0.651 ** | 0.676 ** | 0.683 ** | 0.602 * | 0.681 ** | 0.489 | 0.577 * | 0.265 |
| Urease | 0.062 | 0.035 | 1 | −0.557 * | −0.369 | −0.199 | −0.211 | −0.025 | −0.433 | −0.446 | −0.432 | −0.575 * |
| phosphatase | 0.541 * | 0.473 | −0.557 * | 1 | 0.394 | 0.278 | 0.260 | 0.184 | 0.365 | 0.550 * | 0.341 | 0.553 * |
| Total PLFA | 0.666 ** | 0.651 ** | −0.369 | 0.394 | 1 | 0.959 *** | 0.961 *** | 0.946 *** | 0.936 *** | 0.806 *** | 0.919 *** | 0.556 * |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, E.-H.; Tian, G.; Chiu, C.-Y. The Effect of Re-Planting Trees on Soil Microbial Communities in a Wildfire-Induced Subalpine Grassland. Forests 2017, 8, 385. https://doi.org/10.3390/f8100385
Chang E-H, Tian G, Chiu C-Y. The Effect of Re-Planting Trees on Soil Microbial Communities in a Wildfire-Induced Subalpine Grassland. Forests. 2017; 8(10):385. https://doi.org/10.3390/f8100385
Chicago/Turabian StyleChang, Ed-Haun, Guanglong Tian, and Chih-Yu Chiu. 2017. "The Effect of Re-Planting Trees on Soil Microbial Communities in a Wildfire-Induced Subalpine Grassland" Forests 8, no. 10: 385. https://doi.org/10.3390/f8100385
APA StyleChang, E.-H., Tian, G., & Chiu, C.-Y. (2017). The Effect of Re-Planting Trees on Soil Microbial Communities in a Wildfire-Induced Subalpine Grassland. Forests, 8(10), 385. https://doi.org/10.3390/f8100385





