Fires of the Last Millennium Led to Landscapes Dominated by Early Successional Species in Québec’s Clay Belt Boreal Forest, Canada
Abstract
:1. Introduction
2. Materials and Methods
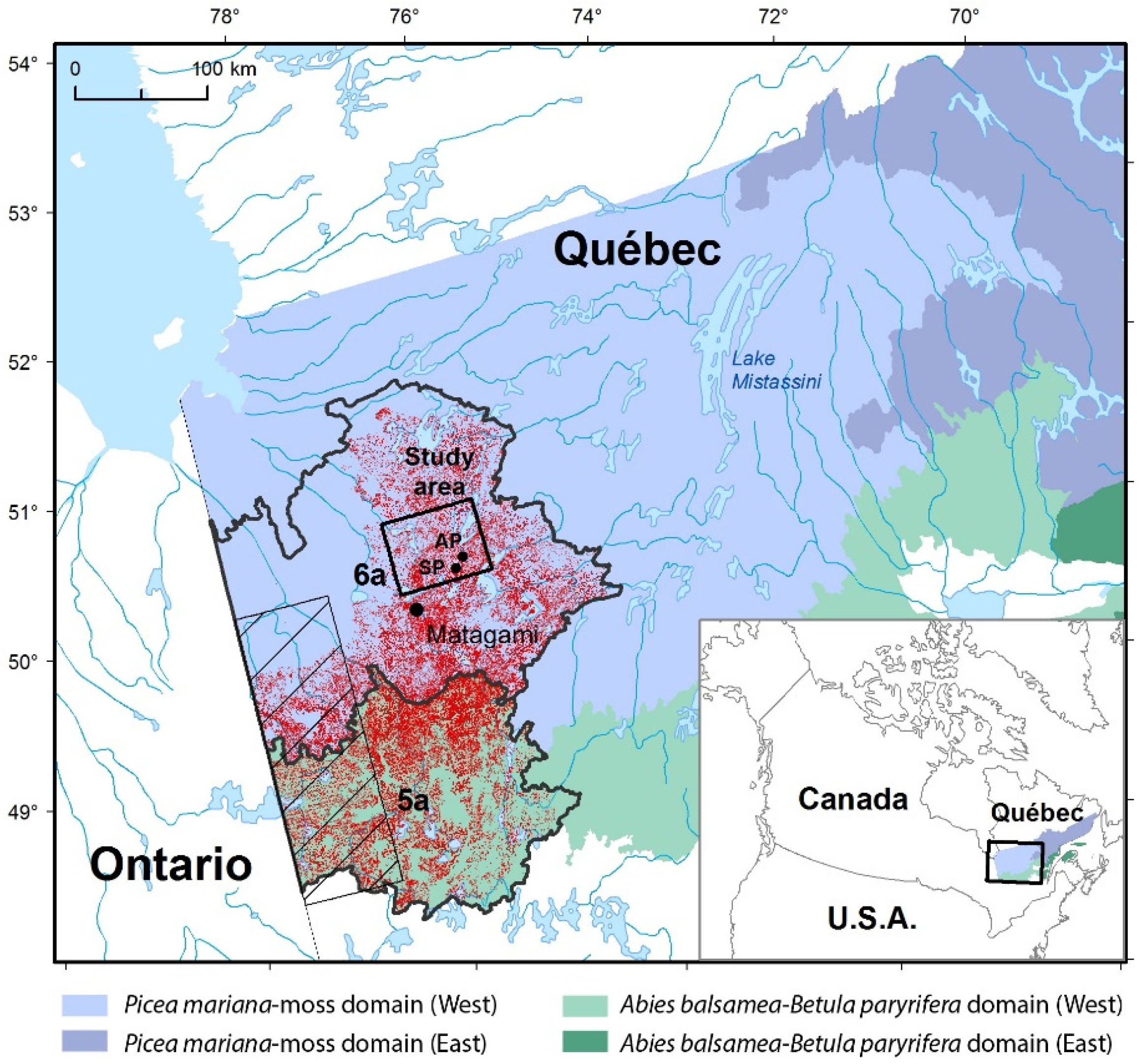
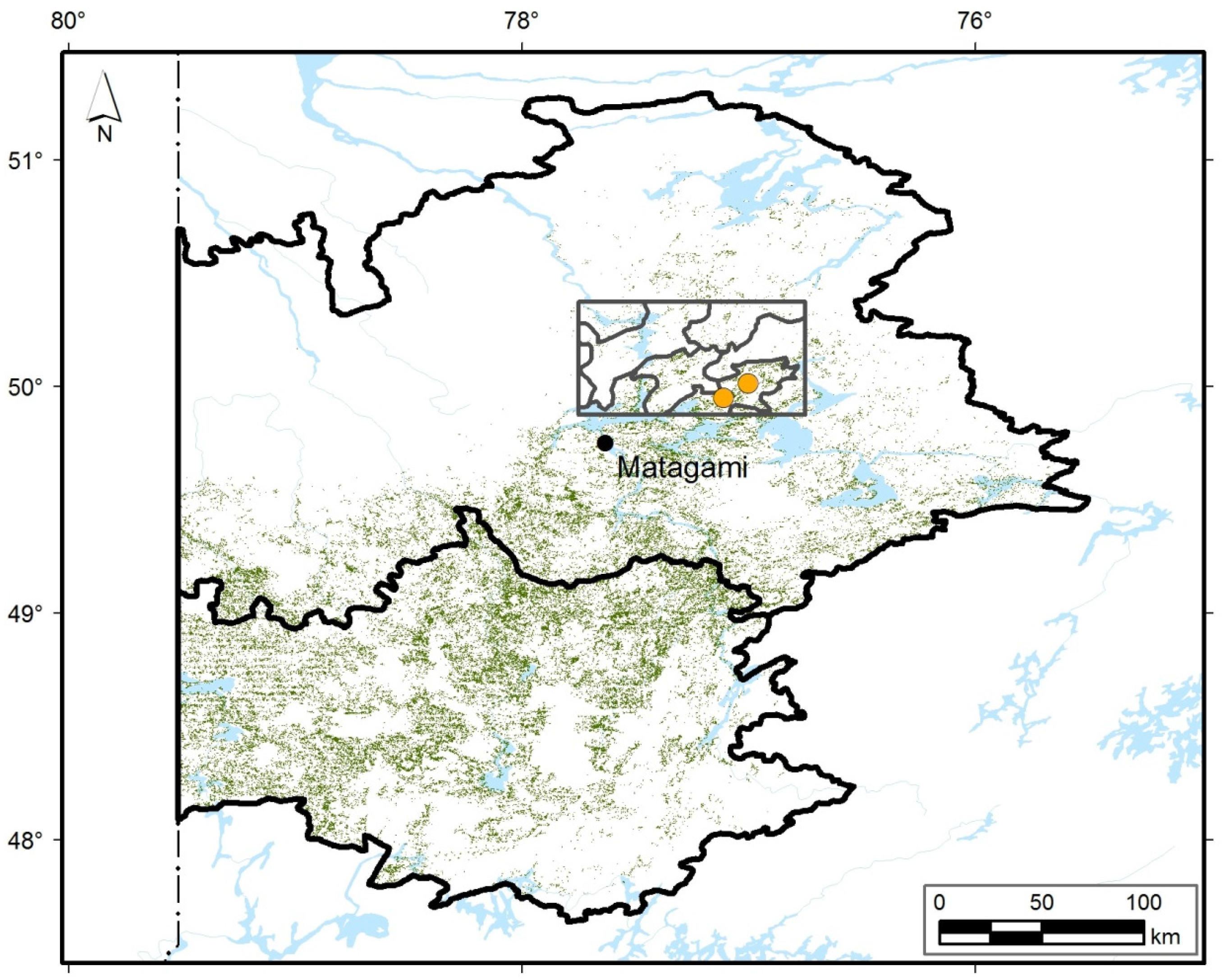
2.1. Study Area
2.2. Contemporary Forest Landscapes
2.3. Study Sites and Sampling
2.4. Characterization of the Contemporary Forest Surrounding the Peatlands
2.5. Stratigraphy and Chronology
2.6. Macrocharcoal Analysis
2.7. Reconstruction of Vegetation History
3. Results
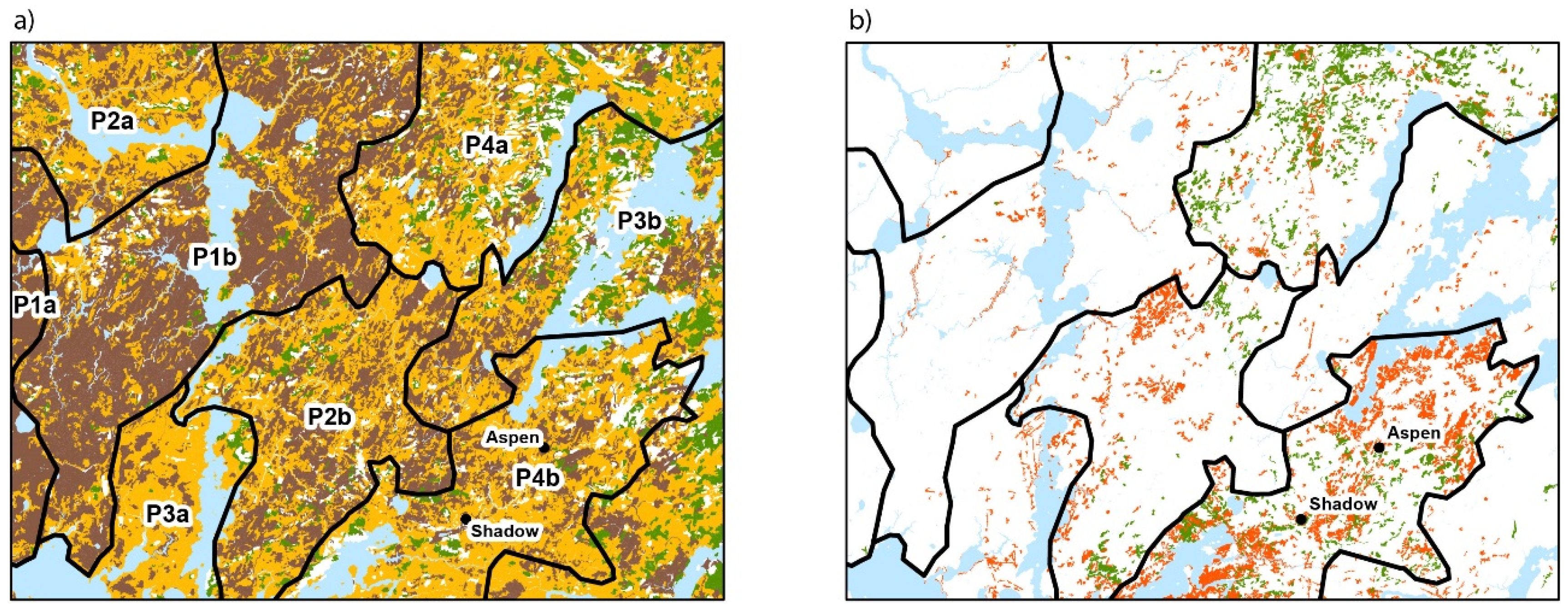
3.1. Contemporary Forest Landscapes of the Study Area
3.2. Contemporary Forest Surrounding the Two Peatlands Studied
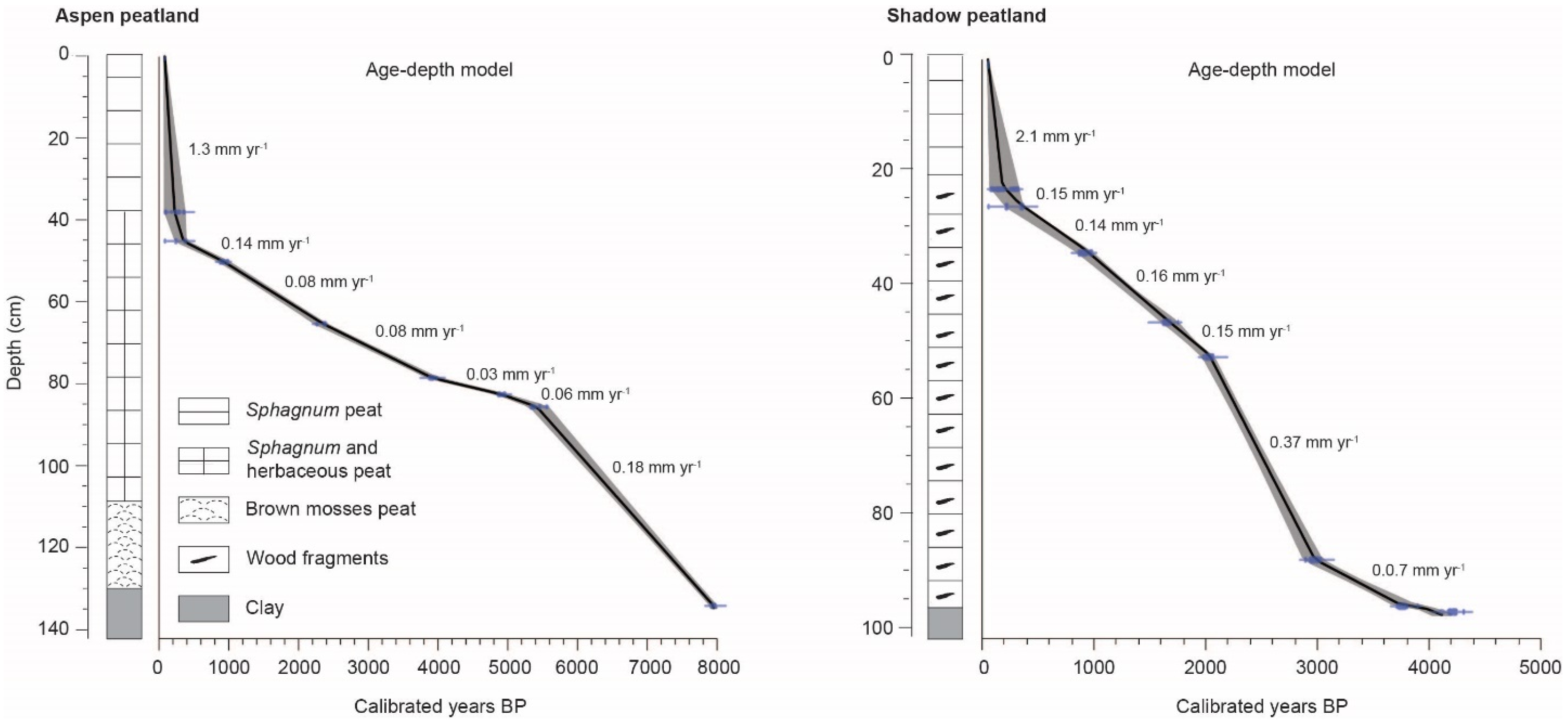
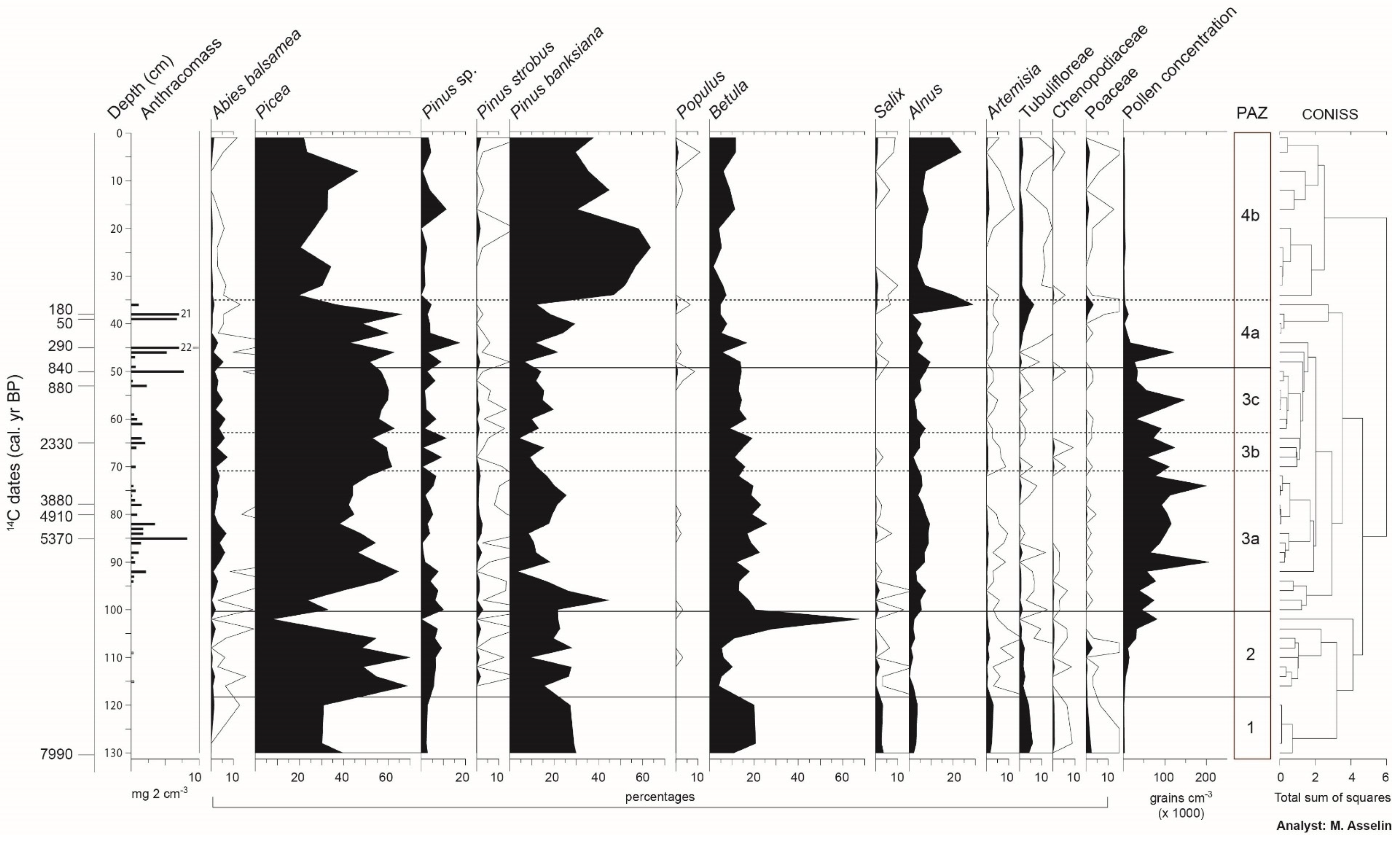
3.3. Stratigraphy and Chronology of Sedimentary Cores
3.4. Reconstruction of the Fire History
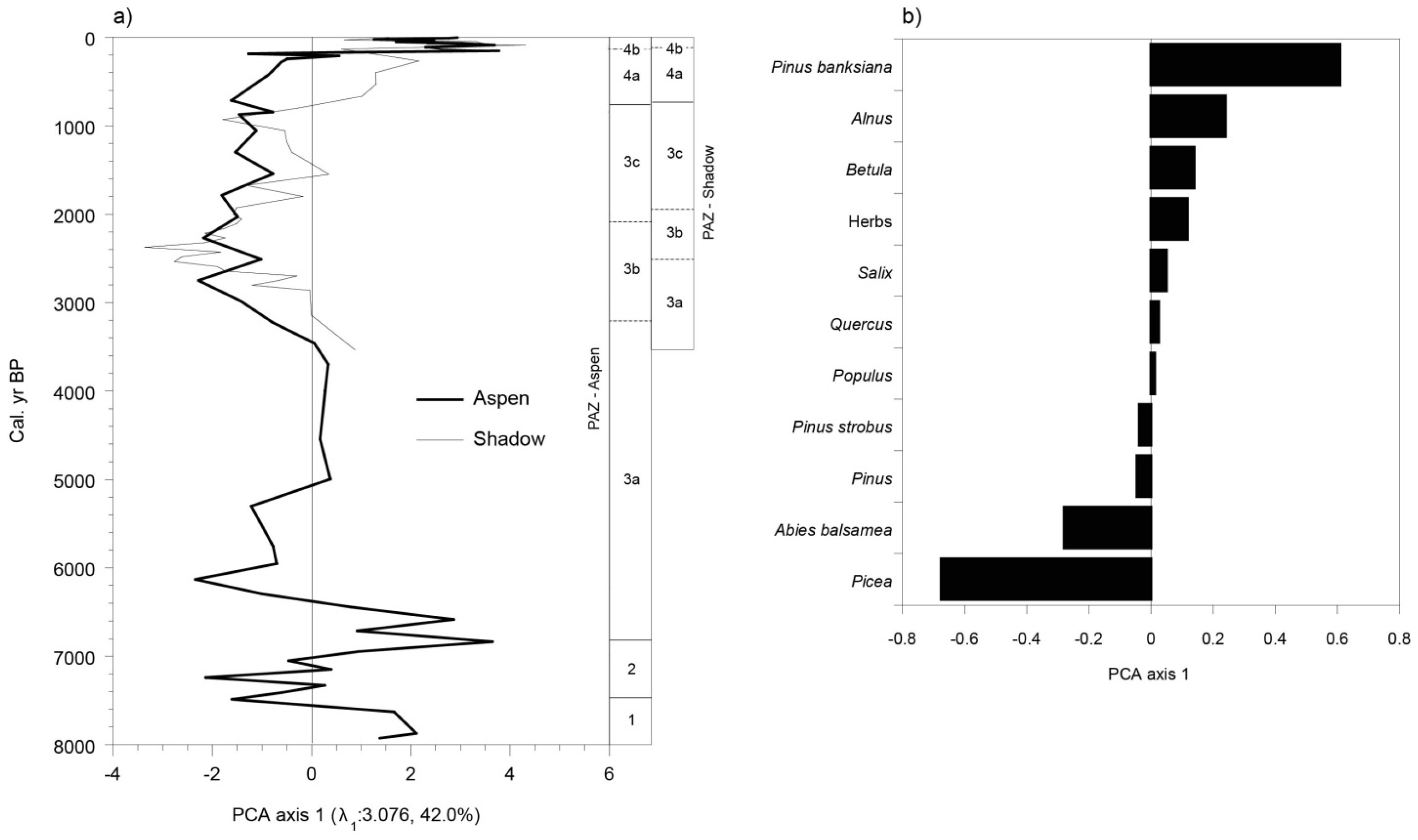
3.5. Vegetation History
4. Discussion
4.1. The Afforestation Phase (ca. 8000 to ca. 6800 BP)
4.2. A Landscape Dominated by Late and Early Successional Forest Stands (ca. 6800 to ca. 2500 BP)
4.3. Landscapes Dominated by Picea mariana and Abies balsamea (ca. 2500 to ca. 2100 BP)
4.4. Increase of Early Successional Species (ca. 2100 to ca. 800 BP)
4.5. More Severe Fires Favored the Development of Landscapes Dominated by Early Successional Species (from 800 BP to the Present-Day)
4.6. Contemporary Forest Dynamics
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- White, P.S. Pattern, process, and natural disturbance in vegetation. Bot. Rev. 1979, 45, 229–299. [Google Scholar] [CrossRef]
- Grondin, P.; Gauthier, S.; Borcard, D.; Bergeron, Y.; Noël, J. A new approach to ecological land classification for the Canadian boreal forest that integrates disturbances. Landsc. Ecol. 2014, 29, 1–16. [Google Scholar] [CrossRef]
- Pojar, J.; Klinka, J.; Meidinger, D.V. Biogeoclimatic ecosystem classification in British-Columbia. For. Ecol. Manag. 1987, 22, 119–154. [Google Scholar] [CrossRef]
- Saucier, J.P.; Grondin, P.; Robitaille, A.; Gosselin, J.; Morneau, C.; Richard, P.J.H.; Brisson, J.; Sirois, L.; Leduc, A.; Morin, H.; et al. Écologie Forestière. In Manuel de Foresterie, 2nd ed.; Éditions MultiMondes: Québec, QC, Canada, 2009; pp. 165–316. [Google Scholar]
- Rowe, J.S.; Scotter, G.W. Fire in the boreal forest. Quat. Res. 1973, 3, 444–464. [Google Scholar] [CrossRef]
- Zackrisson, O. Influence of forest fires on the North Swedish boreal forest. Oikos 1977, 29, 22–32. [Google Scholar] [CrossRef]
- Bergeron, Y.; Gauthier, S.; Flannigan, M.; Kafka, V. Fire regimes at the transition between mixedwood and coniferous boreal forest in northwestern Québec. Ecology 2004, 85, 1916–1932. [Google Scholar] [CrossRef]
- Lynch, J.A.; Hollis, J.L.; Hu, F.S. Climatic and landscape control of the boreal forest fire regime: Holocene records from Alaska. J. Ecol. 2004, 92, 477–489. [Google Scholar] [CrossRef]
- Cyr, D.; Bergeron, Y.; Gauthier, S.; Larouche, A.C. Are the old growth forests of the Clay Belt part of a fire-regulated mosaic? Can. J. For. Res. 2005, 35, 65–73. [Google Scholar] [CrossRef]
- Ali, A.A.; Carcaillet, C.; Bergeron, Y. Long-term fire frequency variability in the eastern Canadian boreal forest: The influences of climate vs. local factors. Glob. Chang. Biol. 2009, 15, 1230–1241. [Google Scholar] [CrossRef]
- Mansuy, N.; Gauthier, S.; Robitaille, A.; Bergeron, Y. The effects of surficial deposit–drainage combinations on spatial variations of fire cycles in the boreal forest of eastern Canada. Int. J. Wildland Fire 2010, 19, 1083–1098. [Google Scholar] [CrossRef]
- Genries, A.; Finsinger, W.; Asnong, H.; Bergeron, Y.; Carcaillet, C.; Garneau, M.; Hély, C.; Ali, A.A. Local versus regional processes: Can soil characteristics overcome climate and fire regimes by modifying vegetation trajectories? J. Quat. Sci. 2012, 27, 745–756. [Google Scholar] [CrossRef]
- Kuosmanen, N.; Fang, K.; Bradshaw, R.H.W.; Clear, J.L.; Seppa, H. Role of forest fires in Holocene stand-scale dynamics in the unmanaged taiga forest of northwestern Russia. Holocene 2014, 24, 503–514. [Google Scholar] [CrossRef]
- Senici, D.; Chen, H.Y.H.; Bergeron, Y.; Ali, A.A. The effects of forest fuel connectivity on spatiotemporal dynamics of Holocene fire regimes in the central boreal forest of North America. J. Quat. Sci. 2015, 30, 365–375. [Google Scholar] [CrossRef]
- Remy, C.C.; Lavoie, M.; Girardin, M.; Hély, C.; Bergeron, Y.; Grondin, P.; Oris, F.; Asselin, H.; Ali, A.A. Wildfire size alters long-term vegetation trajectories in boreal forests of eastern North America. J. Biogeogr. In press.
- Landres, P.B.; Morgan, P.; Swanson, F.J. Overview of the use of natural variability concepts in managing ecological systems. Ecol. Appl. 1999, 9, 1179–1188. [Google Scholar]
- Kuuluvainen, T. Natural variability of forests as a reference for restoring and managing biological diversity in boreal Fennoscandia. Silva Fenn. 2002, 36, 97–125. [Google Scholar] [CrossRef]
- Gauthier, S.; Vaillancourt, M.A.; Leduc, A.; de Grandpré, L.; Kneeshaw, D.; Morin, H.; Drapeau, P.; Bergeron, Y. Aménagement Ecosystémique en Forêt Boréale; Presses de l’Université du Québec: Montréal, QC, Canada, 2008. [Google Scholar]
- Conedera, M.; Tinner, W.; Neff, C.; Meurer, M.; Dickens, A.F.; Krebs, P. Reconstructing past fire regimes: Methods, applications, and relevance to fire management and conservation. Quat. Sci. Rev. 2009, 28, 555–576. [Google Scholar] [CrossRef]
- Cyr, D.; Gauthier, S.; Bergeron, Y.; Carcaillet, C. Forest management is driving the eastern North American boreal forest outside its natural range of variability. Front. Ecol. Environ. 2009, 7, 519–524. [Google Scholar] [CrossRef]
- Cogbill, C.V. Dynamics of the boreal forests of the Laurentian Highlands, Canada. Can. J. For. Res. 1985, 15, 252–261. [Google Scholar] [CrossRef]
- Blouin, J.; Berger, J.-P. Guide de reconnaissance des types écologiques de la région écologique 6a-Plaine du lac Matagami et 6b-Plaine de la baie de Rupert; ministère des Ressources naturelles et de la Faune, Direction des inventaires forestiers, Division de la classification écologique et productivité des stations: Québec, QC, Canada, 2005.
- Frelich, L.E.; Reich, P.B. Spatial patterns and succession in a Minnesota southern-boreal forest. Ecol. Monogr. 1995, 65, 325–346. [Google Scholar] [CrossRef]
- Swain, A.M. Environmental changes during the past 2000 years in north-central Wisconsin: Analysis of pollen charcoal and seeds from varved lake sediments. Quat. Res. 1978, 10, 55–68. [Google Scholar] [CrossRef]
- Frégeau, M.; Payette, S.; Grondin, P. Fire history of the central boreal forest in eastern North America reveals stability since the mid-Holocene. Holocene 2015, 25, 1912–1922. [Google Scholar] [CrossRef]
- Blarquez, O.; Ali, A.A.; Girardin, M.P.; Grondin, P.; Fréchette, B.; Bergeron, Y.; Hély, C. Regional paleofire regimes affected by non-uniform climate, vegetation and human drivers. Sci. Rep. 2015, 5, 13356. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; Blarquez, O.; Girardin, M.P.; Hély, C.; Tinquaut, F.; El-Guellab, A.; Valsecchi, V.; Terrier, A.; Bremond, L.; Genries, A.; et al. Control of the multimillenial wildfire size in boreal North America by spring climatic conditions. Proc. Natl. Acad. Sci. USA 2012, 109, 20966–20970. [Google Scholar] [CrossRef] [PubMed]
- El-Guellab, A.; Asselin, H.; Gauthier, S.; Bergeron, Y.; Ali, A.A. Holocene variations of wildfire occurrence as a guide for sustainable management of the northeastern Canadian boreal forest. For. Ecosyst. 2015, 2, 1–7. [Google Scholar] [CrossRef]
- Viereck, L.A. Wildfire in the taiga of Alaska. Quat. Res. 1973, 3, 465–495. [Google Scholar] [CrossRef]
- Viau, A.E.; Gajewski, K. Reconstructing millennial-scale, regional paleoclimates of boreal Canada during the Holocene. J. Clim. 2009, 22, 316–330. [Google Scholar] [CrossRef]
- Hély, C.; Girardin, M.P.; Ali, A.A.; Carcaillet, C.; Brewer, S.; Bergeron, Y. Eastern boreal North American wildfire risk of the past 7000 years: A model-data comparison. Geophys. Res. Lett. 2010, 37, L14709. [Google Scholar] [CrossRef]
- Girardin, M.P.; Ali, A.A.; Carcaillet, C.; Gauthier, S.; Hély, C.; Le Goff, H.; Terrier, A.; Bergeron, Y. Fire in managed forests of eastern Canada: Risks and options. For. Ecol. Manag. 2013, 294, 238–249. [Google Scholar] [CrossRef]
- Girardin, M.P.; Ali, A.A.; Carcaillet, C.; Blarquez, O.; Hély, C.; Terrier, A.; Genries, A.; Bergeron, Y. Vegetation limits the impact of a warm climate on boreal wildfires. New Phytol. 2013, 199, 1001–1011. [Google Scholar] [CrossRef] [PubMed]
- Oris, F.; Asselin, H.; Finsinger, W.; Hély, C.; Blarquez, O.; Ferland, M.E.; Bergeron, Y.; Ali, A.A. Long-term fire history in northern Québec: Implications for the northern limit of commercial forests. J. Appl. Ecol. 2014, 51, 675–683. [Google Scholar] [CrossRef]
- Fréchette, B.; Richard, P.J.H.; Grondin, P.; Lavoie, M.; Larouche, A.C. Histoire Postglaciaire de la Végétation et du Climat des Pessières et des Sapinières de l’Ouest du Québec; Direction des Inventaires Forestiers, Gouvernement du Québec: Québec, QC, Canada, 2016.
- Environment Canada. National Climate Data and Information Archive. Canadian Climate Normals 1971–2000. 2013. Available online: http://www.climate.weatheroffice.ec.gc.ca (accessed on 17 September 2015). [Google Scholar]
- Carcaillet, C.; Bergeron, Y.; Richard, P.J.H.; Fréchette, B.; Gauthier, S.; Prairie, Y.T. Change of fire frequency in the eastern Canadian boreal forests during the Holocene: Does vegetation composition or climate trigger the fire regime? J. Ecol. 2001, 89, 930–946. [Google Scholar] [CrossRef]
- Carcaillet, C.; Richard, P.J.H.; Bergeron, Y.; Fréchette, B.; Ali, A.A. Resilience of the boreal forest in response to Holocene fire-frequency changes assessed by pollen diversity and population dynamics. Int. J. Wildland Fire 2010, 19, 1026–1039. [Google Scholar] [CrossRef]
- Vincent, J.S.; Hardy, L. L’évolution et l’extension des lacs glaciaires Barlow et Ojibway en territoire québécois. Geogr. Phys. Quat. 1977, 31, 357–372. [Google Scholar] [CrossRef]
- Bergeron, Y.; Gauthier, S.; Kafka, V.; Lefort, P.; Lesieur, D. Natural fire frequency for the Eastern Canadian boreal forest: Consequences for sustainable forestry. Can. J. For. Res. 2001, 31, 384–391. [Google Scholar] [CrossRef]
- Lecomte, N.; Bergeron, Y. Successionnel pathways on different surficial deposits in the coniferous boreal forest of the Québec Clay Belt. Can. J. For. Res. 2005, 35, 1984–1995. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Bhiry, N.; Robert, É.C. Reconstruction of changes in vegetation and trophic conditions of a palsa in a permafrost peatland, subarctic Québec, Canada. Écoscience 2006, 13, 55–65. [Google Scholar] [CrossRef]
- Lavoie, M.; Filion, L.; Robert, É.C. Boreal peatland margins as repository sites of long-term natural disturbances of balsam fir/spruce forests. Quat. Res. 2009, 71, 295–306. [Google Scholar] [CrossRef]
- Stuiver, M.; Reimer, P.J. Extended 14C database and revised CALIB radiocarbon calibration program. Radiocarbon 1993, 35, 315–330. [Google Scholar] [CrossRef]
- Reimer, P.J.; Bard, E.; Bayliss, A.; Beck, J.W.; Blackwell, P.G.; Ramsey, C.B.; Buck, C.E.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. INTCAL 13 and MARINE13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 2013, 55, 1869–1887. [Google Scholar] [CrossRef]
- Blaauw, M. Methods and code for “classical” age-modelling of radiocarbon sequences. Quat. Geochron. 2010, 5, 512–518. [Google Scholar] [CrossRef]
- Hörnberg, G.; Ohlson, M.; Zackrisson, O. Stand dynamics, regeneration patterns and long-term continuity in boreal old-growth Picea abies swamp-forests. J. Veg. Sci. 1995, 6, 291–298. [Google Scholar] [CrossRef]
- Magnan, G.; Lavoie, M.; Payette, S. Impact of fire on long-term vegetation dynamics of ombrotrophic peatlands in northwestern Québec, Canada. Quat. Res. 2012, 77, 110–121. [Google Scholar] [CrossRef]
- Kasin, I.; Blanck, Y.; Storaunet, O.; Rolstad, J.; Ohlson, M. The charcoal record in peat and mineral soil across a boreal landscape and possible linkages to climate change and recent fire history. Holocene 2013, 23, 1052–1065. [Google Scholar] [CrossRef]
- Faegri, K.; Kaland, P.E.; Krzywinski, K. Textbook of Pollen Analysis, 4th ed.; John Wiley & Sons: New York, NY, USA, 1989. [Google Scholar]
- Grimm, E.C. CONISS: A FORTRAN 77 program for stratigraphically constrained cluster analysis by the method of incremental sum of squares. Comput. Geosci. 1987, 13, 13–35. [Google Scholar] [CrossRef]
- Birks, H.J.B. Quantitative Palaeoenvironmental Reconstructions. In Statistical Modelling of Quaternary Science Data; Maddy, D., Brew, J.S., Eds.; Quaternary Research Association: Cambridge, UK, 1995; pp. 161–254. [Google Scholar]
- Richard, P.J.H. Origine et dynamique postglaciaire de la forêt mixte au Québec. Rev. Paleobot. Palynol. 1993, 79, 31–68. [Google Scholar] [CrossRef]
- Comtois, P.; Payette, S. Représentation pollinique actuelle et subactuelle des peupleraies boréales au Nouveau-Québec. Geogr. Phys. Quat. 1984, 38, 123–133. [Google Scholar]
- Brossier, B.; Oris, F.; Finsinger, W.; Asselin, H.; Bergeron, Y.; Ali, A. Using tree-ring to calibrate peak detection in fire reconstruction based on sedimentary charcoal records. Holocene 2014, 24, 635–645. [Google Scholar] [CrossRef]
- Ali, A.; Asselin, H.; Larouche, A.C.; Bergeron, Y.; Carcaillet, C.; Richard, P.J.H. Changes in fire regime explain the Holocene rise and fall of Abies balsamea in the coniferous forests of western Québec, Canada. Holocene 2008, 18, 693–703. [Google Scholar] [CrossRef]
- Blarquez, O.; Girardin, M.P.; Leys, B.; Ali, A.A.; Aleman, J.C.; Bergeron, Y.; Carcaillet, C. Paleofire reconstruction based on an ensemble-member strategy applied to sedimentary charcoal. Geophysic. Res. Lett. 2013, 40, 2667–2672. [Google Scholar] [CrossRef]
- Le Goff, H.; Flannigan, M.D.; Bergeron, Y.; Girardin, M.P. Historical fire regime shift related to climate teleconnections in the Waswanipi area, central Quebec, Canada. Int. J. Wildland Fire 2007, 16, 607–618. [Google Scholar] [CrossRef]
- Bergeron, Y.; Cyr, D.; Girardin, M.; Carcaillet, C. Will climate change drive 21st century burn rates in Canadian boreal forest outside its natural variability: Collating global climate model experiment with sedimentary charcoal data. Int. J. Wildland Fire 2010, 19, 1127–1139. [Google Scholar] [CrossRef]

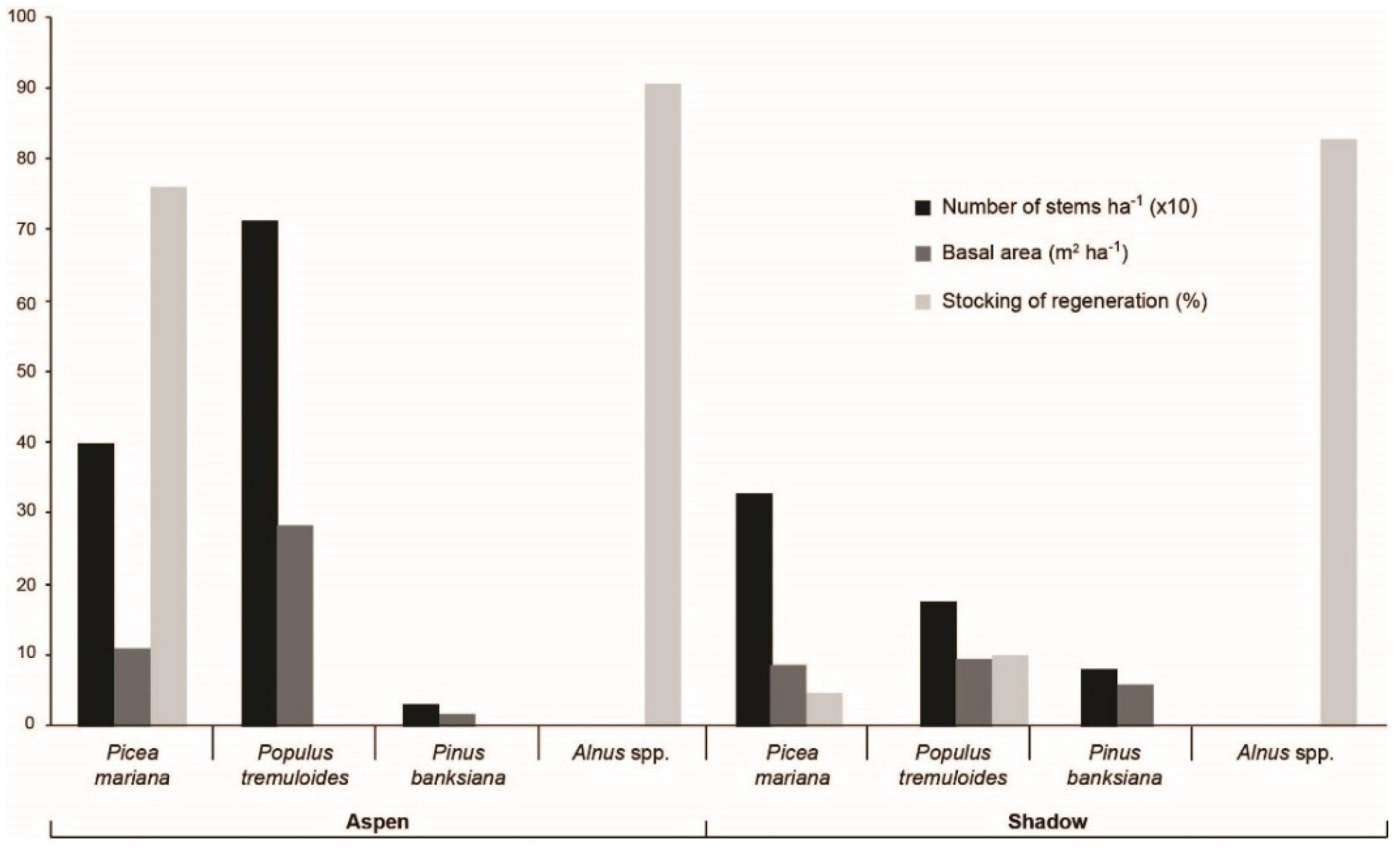

| Site and Stand Dominant Species | Picea mariana | Pinus banksiana | Abies balsamea | Betula | Populus tremuloides | Alnus |
|---|---|---|---|---|---|---|
| Aspen peatland P. tremuloides, P. banksiana, P. mariana | 43.3% | 36.2% | 0.9% | 5.4% | 0% | 10.6% |
| 10 km north of Aspen peatland P.mariana, A. balsamea | 49.4% | 23.1% | 2.1% | 9.9% | 0% | 12% |
| Site and Depth (cm) | Laboratory Number a | Material Dated | 14C Date (Years BP) | Calibrated Age Range b (Years BP) | Median c (cal. Years BP) |
|---|---|---|---|---|---|
| Aspen | |||||
| 37–38 | 145,391–4797 | Charcoal | 180 ± 30 | 136–224 | 180 |
| 38–39 | 150,957–5063 | Charcoal | 75 ±15 | 33–73 | 50 |
| 44–45 | 145,611–4828 | Charcoal | 235 ± 20 | 281–307 | 290 |
| 49–50 | 145,608–4825 | Charcoal | 955 ± 20 | 796–874 | 840 |
| 52–53 | 150,956–5062 | Charcoal | 915 ± 15 | 840–910 | 880 |
| 64–65 | 150,951–5057 | Charcoal | 2275 ± 15 | 2306–2346 | 2330 |
| 77–78 | 150,952–5058 | Charcoal | 3590 ± 20 | 3839–3929 | 3880 |
| 81–82 | 150,955–5061 | Charcoal | 4345 ± 20 | 4856–4963 | 4910 |
| 84–85 | 145,607–4824 | Charcoal | 4705 ± 20 | 5325–5409 | 5370 |
| 132–133 | 145,602–4819 | Seeds | 7185 ± 25 | 7956–8026 | 7990 |
| Shadow | |||||
| 21–22 | 145,390–4796 | Charcoal | 120 ± 30 | 51–149 | 100 |
| 24–25 | 145,609–4826 | Charcoal | 240 ± 20 | 281–308 | 300 |
| 32–33 | 145,603–4820 | Charcoal | 945 ± 20 | 796–874 | 835 |
| 41–42 | 145,612–4829 | Charcoal | 1760 ± 20 | 1608–1724 | 1670 |
| 44–45 | 145,604–4821 | Charcoal | 1680 ± 20 | 1539–1619 | 1580 |
| 45–46 | 150,954–5060 | Charcoal | 1655 ± 15 | 1524–1573 | 1550 |
| 50–51 | 145,605–4822 | Charcoal | 2010 ± 20 | 1920–1998 | 1960 |
| 85–86 | 145,606–4823 | Charcoal | 2790 ± 25 | 2842–2956 | 2900 |
| 93–94 | 145,610–4827 | Charcoal | 3425 ± 20 | 3613–3720 | 3680 |
| 94–95 | 145,582–4783 | Charcoal | 3745 ± 25 | 4068–4157 | 4110 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asselin, M.; Grondin, P.; Lavoie, M.; Fréchette, B. Fires of the Last Millennium Led to Landscapes Dominated by Early Successional Species in Québec’s Clay Belt Boreal Forest, Canada. Forests 2016, 7, 205. https://doi.org/10.3390/f7090205
Asselin M, Grondin P, Lavoie M, Fréchette B. Fires of the Last Millennium Led to Landscapes Dominated by Early Successional Species in Québec’s Clay Belt Boreal Forest, Canada. Forests. 2016; 7(9):205. https://doi.org/10.3390/f7090205
Chicago/Turabian StyleAsselin, Maxime, Pierre Grondin, Martin Lavoie, and Bianca Fréchette. 2016. "Fires of the Last Millennium Led to Landscapes Dominated by Early Successional Species in Québec’s Clay Belt Boreal Forest, Canada" Forests 7, no. 9: 205. https://doi.org/10.3390/f7090205
APA StyleAsselin, M., Grondin, P., Lavoie, M., & Fréchette, B. (2016). Fires of the Last Millennium Led to Landscapes Dominated by Early Successional Species in Québec’s Clay Belt Boreal Forest, Canada. Forests, 7(9), 205. https://doi.org/10.3390/f7090205






