Fertilization Response, Light Use, and Growth Efficiency in Eucalyptus Plantations across Soil and Climate Gradients in Brazil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description and Location
2.2. Growth Determination and Fertilization Response
2.3. Leaf Area Index, Light Use Efficiency, and Growth Efficiency
2.4. Statistical Analyses
3. Results
3.1. Productivity Plots
3.2. Fertilization Response (FR)
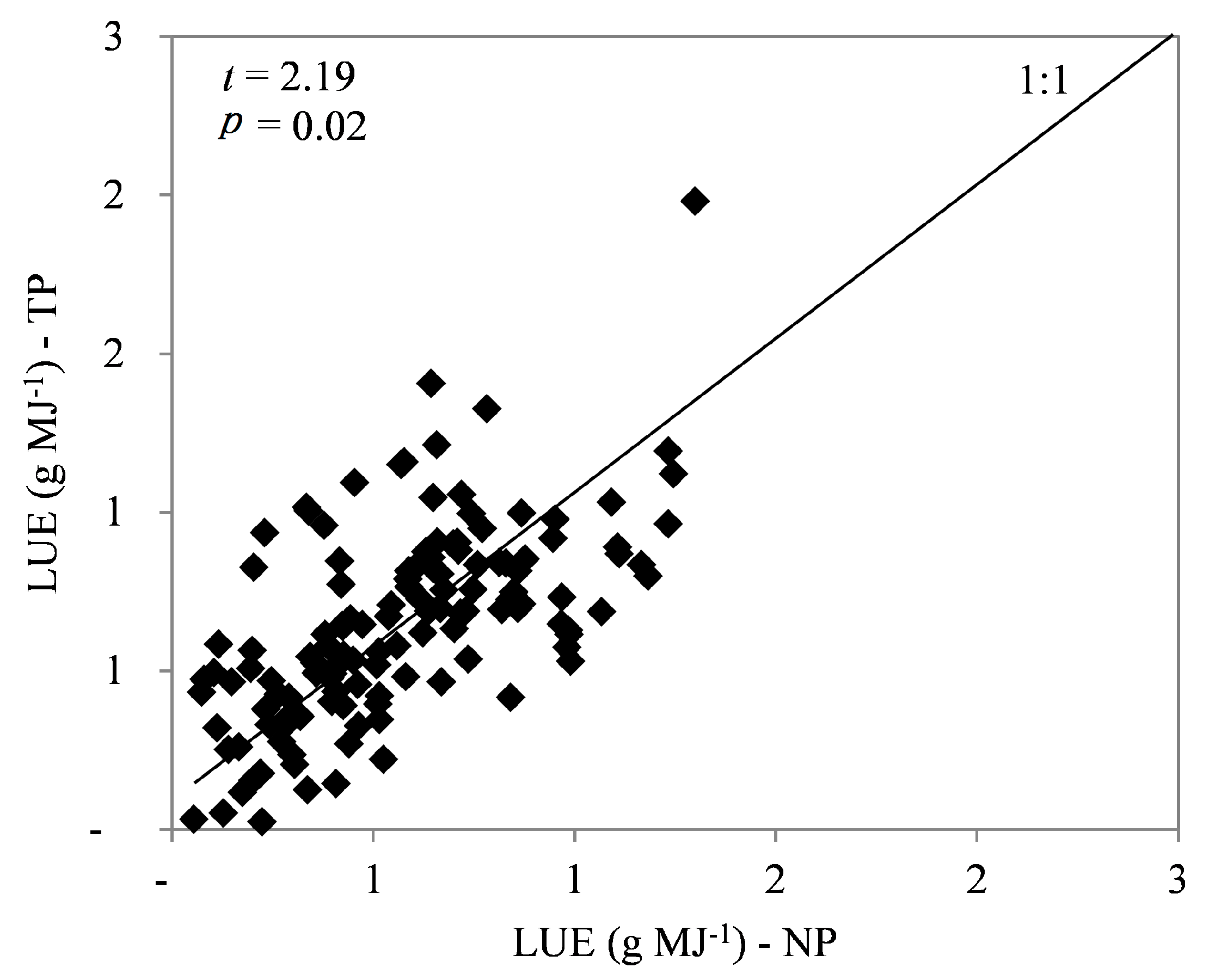
3.3. Leaf Area Index, Light Use Efficiency, and Growth Efficiency
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lungo, A.D.; Ball, J.; Carle, J. Global Planted Forests Thematic Study: Results and Analysis; Planted Forests and Trees Working Paper; The Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2006. [Google Scholar]
- Associação Brasileira Dos Produtores De Florestas Plantadas. Anuário Estatístico ABRAF 2014: Base Year 2013; Associação Brasileira Dos Produtores De Florestas Plantadas: Brasília, Brazil, 2014; p. 74. [Google Scholar]
- IBÁ. Indústria Brasileira de Árvores. Available online: http://www.iba.org/shared/iba_2014_pt.pdf (accessed on 10 June 2015).
- Zen, S. Influência da Matocompetição em Plantios de Eucalyptus grandis, Série técnica. IPEF 1987, 4, 25–35. [Google Scholar]
- Sgarbi, F. Produtividade do Eucalyptus sp. em Função do Estado Nutricional e da Fertilidade do Solo em Diferentes Regiões do Estado de São Paulo. Ph.D. Thesis, Superior School of Agriculture Luiz de Queiroz, University of Sao Paulo, Piracicaba, Brazil, 2002; p. 101. [Google Scholar]
- Ferreira, J.M.A.; Stape, J.L. Productivity gains by fertilization in Eucalyptus urophylla clonal plantations across gradients in site and stand conditions. South. For. 2009, 71, 253–258. [Google Scholar]
- Laclau, J.P.; Ranger, J.; Gonçalves, J.L.M.; Maquère, V.; Krusche, A.V.; M’bou, A.T.; Nouvellon, Y.; Saint-Ándré, L.; Bouillet, J.-P.; de Cassia Piccolo, M.; et al. Biogeochemical cycles of nutrients in tropical Eucalyptus plantations Main features shown by intensive monitoring in Congo and Brazil. For. Ecol. Manag. 2010, 259, 1771–1785. [Google Scholar] [CrossRef]
- Stape, J.L.; Binkley, D.; Jacob, W.S.; Takahashi, E.N. A twin-plot approach to determine nutrient limitation and potential productivity in Eucalyptus plantations as landscapes scales in Brazil. For. Ecol. Manag. 2006, 223, 358–362. [Google Scholar] [CrossRef]
- Hakamada, R.E.; Stape, J.L.; Lemos, C.C.Z.; Almeida, A.A.; Silva, L.F. Uso do inventário florestal e da uniformidade entre árvores como ferramenta de monitoramento da qualidade silvicultural em plantios clonais de eucalipto. Sci. For. 2015, 43, 27–36. [Google Scholar]
- Binkley, D.; Stape, J.L.; Ryan, M.G. Thinking about efficiency of resource use in forests. For. Ecol. Manag. 2004, 193, 5–16. [Google Scholar] [CrossRef]
- Stape, J.L.; Binkley, D.; Ryan, M.G. Eucalyptus production and the supply, use and efficiency of use of water, light and nitrogen across a geographic gradient in Brazil. For. Ecol. Manag. 2004, 193, 17–31. [Google Scholar] [CrossRef]
- Nilsson, U.; Allen, H.L. Short- and long-term effects of site preparation, fertilization and vegetation control on growth and stand development of planted loblolly pine. For. Ecol. Manag. 2003, 175, 367–377. [Google Scholar] [CrossRef]
- Otto, M.S.G.; Hubbard, R.M.; Binkley, D.; Stape, J.L. Dominant clonal Eucalyptus grandis x urophylla trees use water more efficiently. For. Ecol. Manag. 2014, 328, 117–121. [Google Scholar] [CrossRef]
- Laclau, J.P.; Almeida, J.C.R.; Gonçalves, J.L.M. Influence of nitrogen and potassium fertilization on leaf lifespan and allocation of above-ground growth in Eucalyptus plantation. Tree Physiol. 2009, 29, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Barros, N.F.; Novais, R.F.; Neves, J.C.L.; Leal, P.G.L. Fertilizing eucalypt plantations on the Brazilian savannah soils. S. Afr. For. J. 1992, 160, 7–12. [Google Scholar]
- Gonçalves, J.L.M.; Barros, N.F.; Nambiar, E.K.S.; Novais, R.F. Soil and stand management for short-rotation plantations. In Management of Soil, Water, and Nutrients in Tropical Plantation Forests; Nambiar, E.K.S., Brown, A., Eds.; ACIAR Monograph 43; Australian Centre for International Agricultural Research: Canberra, Australia, 1997; pp. 379–418. [Google Scholar]
- Gonçalves, J.L.M.; Stape, J.L.; Laclau, J.-P.; Bouillet, J.-P.; RANGER, J. Assessing the effects of early silvicultural managements on long-term site productivity of fast-growing Eucalypt plantations: The Brazilian experience. South. For. 2008, 70, 105–118. [Google Scholar] [CrossRef]
- Van Raij, B.; Andrade, J.C.; Cantarella, H.; Quaggio, J.A. Análise Química para Avaliação da Fertilidade de Solos Tropicais; Instituto Agronômico de Campinas: Campinas, Brazil, 2001; p. 285. [Google Scholar]
- Gonçalves, J.L.M.; Mendes, K.C.F.S.; Sasaki, C.M. Mineralização de nitrogênio em ecossistemas florestais naturais e implantados do estado de São Paulo. Rev. Bras. Ciênci. Solo 2001, 25, 601–616. [Google Scholar] [CrossRef]
- Gonçalves, J.L.M.; Alvares, C.A.; Higa, A.R.; Silva, L.D.; Alfenas, A.C.; Stahl, J.; Ferraz, S.F.B.; Lima, W.P.; Brancalion, P.H.S.; Hubner, A.; et al. Integrating genetic and silvicultural strategies to minimize abiotic and biotic constraints in Brazilian Eucalypt plantations. For. Ecol. Manag. 2013, 301, 6–27. [Google Scholar] [CrossRef]
- Pulito, A.P.; Leonardo de Moraes Gonçalves, J.; Smethurst, P.J.; Junior, J.C.A.; Alcarde Alvares, C.; Rocha, J.H.T.; Hübner, A.; de Moraes, L.F.; Miranda, A.C.; Kamogawa, M.Y.; et al. Available Nitrogen and Responses to Nitrogen Fertilizer in Brazilian Eucalypt Plantations on Soils of Contrasting Texture. Forests 2015, 6, 973–991. [Google Scholar] [CrossRef]
- Sentelhas, P.C.; Pereira, A.R.; Marin, F.R.; Angelocci, L.R.; Alfonsi, R.R.; Caramori, P.H.; Swart, S. Balanços Hídricos Climatológicos de 500 Localidades Brasileiras. Piracicaba: Núcleo de Monitoramento Agroclimático. Disponível em. 2001. Available online: http://www.esalq.usp.br/departamentos/lce/nurma.htm (accessed on 20 May 2008).
- Donagemma, G.K.; de Campos, D.V.B.; Calderano, S.B.; Teixeira, W.G.; Viana, J.H.M. Manual de Métodos de Análise de Solo, 2nd ed.; Centro Nacional de Pesquisa de Solos: Rio de Janeiro, Brazil, 1997; p. 212. [Google Scholar]
- Schumacher, F.X.; Hall, F.S. Logarithmic expression of timber-tree volume. J. Agric. Res. 1993, 47, 719–734. [Google Scholar]
- Barrichelo, L.E.G.; Foelkel, C.E.B.; Brasil, M.A.M. Métodos para determinação da densidade básica de coníferas e folhosas. IPEF 1971, 2, 65–74. [Google Scholar]
- Schleppi, P.; Conedera, M.; Sedivy, I.; Thimonier, A. Correcting non-linearity and slope effects in the estimation of the leaf area index of forests from hemispherical photographs. Agric. For. Meteorol. 2007, 144, 236–242. [Google Scholar] [CrossRef]
- Giunti Neto, C.J.; Stape, J.L.; Silva, S.R.; Hakamada, R.E. Calibração do Uso de Fotos Hemisféricas, Do LAI—2000 e Do Ceptômetro para Estimativas de Índice de Área Foliar em Plantações de Eucalyptus; Technical Report; Superior School of Agriculture “Luiz de Queiroz”, University of Sao Paulo: Piracicaba, Brazil, 2007; p. 61. [Google Scholar]
- Waring, R.H.; Thies, W.G.; Muscato, D. Stem growth per unit of leaf area: A measure of tree vigor. For. Sci. 1980, 26, 112–117. [Google Scholar]
- Landsberg, J.J.; Gower, S.T. Applications of Physiological Ecology to Forest Management; Academic Press: Waltham, MA, USA, 1997; p. 354. [Google Scholar]
- Almeida, A.C.; Landsberg, J.J.; Sands, P.J. Parametrization of 3-PG model for fast growing Eucalyptus grandis plantations. For. Ecol. Manag. 2004, 193, 179–195. [Google Scholar] [CrossRef]
- Larcher, W. Ecofisiologia Vegetal; RiMa: São Carlos, Brazil, 2000; p. 531. [Google Scholar]
- Sinclair, T.R. Mineral nutrition and plant growth response to climate change. J. Exp. Bot. 1992, 43, 1141–1146. [Google Scholar] [CrossRef]
- Lana, M.C.; Neves, J.C.L. Capacidade de suprimento de potássio em solos sob reflorestamento com eucalipto do estado de São Paulo. Rev. Árvore 1994, 18, 115–122. [Google Scholar]
- Gava, J.L.; Gonçalves, J.L.M. Soil atributes and wood quality for pulp production in plantations of Eucalyptus grandis clone. Sci. Agric. 2008, 63, 306–313. [Google Scholar]
- Albaugh, T.J.; Allen, H.L.; Dougherty, P.M.; Kress, L.W.; King, J.S. Leaf area and above- and belowground growth responses of loblolly pine to nutrient and water additions. For. Sci. 1998, 44, 1–12. [Google Scholar]
- Smethurst, P.; Baillie, C.; Cherry, M.; Holz, G. Fertilizer effects on LAI and growth of four Eucalyptus nitens plantations. For. Ecol. Manag. 2003, 176, 531–542. [Google Scholar] [CrossRef]
- Binkley, D.; Stape, J.L.; Bauerle, W.L.; Ryan, M.G. Explaining growth of individual trees: Light Interception and efficiency of light use by Eucalyptus at four sites in Brazil. For. Ecol. Manag. 2010, 259, 1695–1703. [Google Scholar] [CrossRef]
- Campoe, O.C.; Stape, J.L.; Albaugh, T.J.; Allen, H.L.; Fox, T.R.; Rubilar, R.; Binkley, D. Fertilization and irrigation effects on tree level growth, light interception and light use efficiency in Pinus taeda. For. Ecol. Manag. 2013, 288, 43–48. [Google Scholar] [CrossRef]
- Allen, C.B.; Will, R.E.; Jacobson, M.A. Production Efficiency and Radiation Use Efficiency of Four Tree Species Receiving Irrigation and Fertilization. For. Sci. 2005, 51, 556–569. [Google Scholar]
- Clearwater, M.J.; Meinzer, F.C. Relationships between hydraulic architecture and leaf photosynthetic capacity in nitrogen-fertilized Eucalyptus grandis trees. Tree Physiol. 2001, 21, 683–690. [Google Scholar] [CrossRef]
- Giardina, C.P.; Ryan, M.G.; Binkley, D.; Fownes, J.H. Primary production and carbon allocation in relation to nutrient supply in a tropical experimental forest. Glob. Change Biol. 2003, 9, 1438–1450. [Google Scholar] [CrossRef]
- Fisher, R.F.; Binkley, D. Ecology and Management of Forest Soils, 3rd ed.; Wiley: New York, NY, USA, 2000. [Google Scholar]
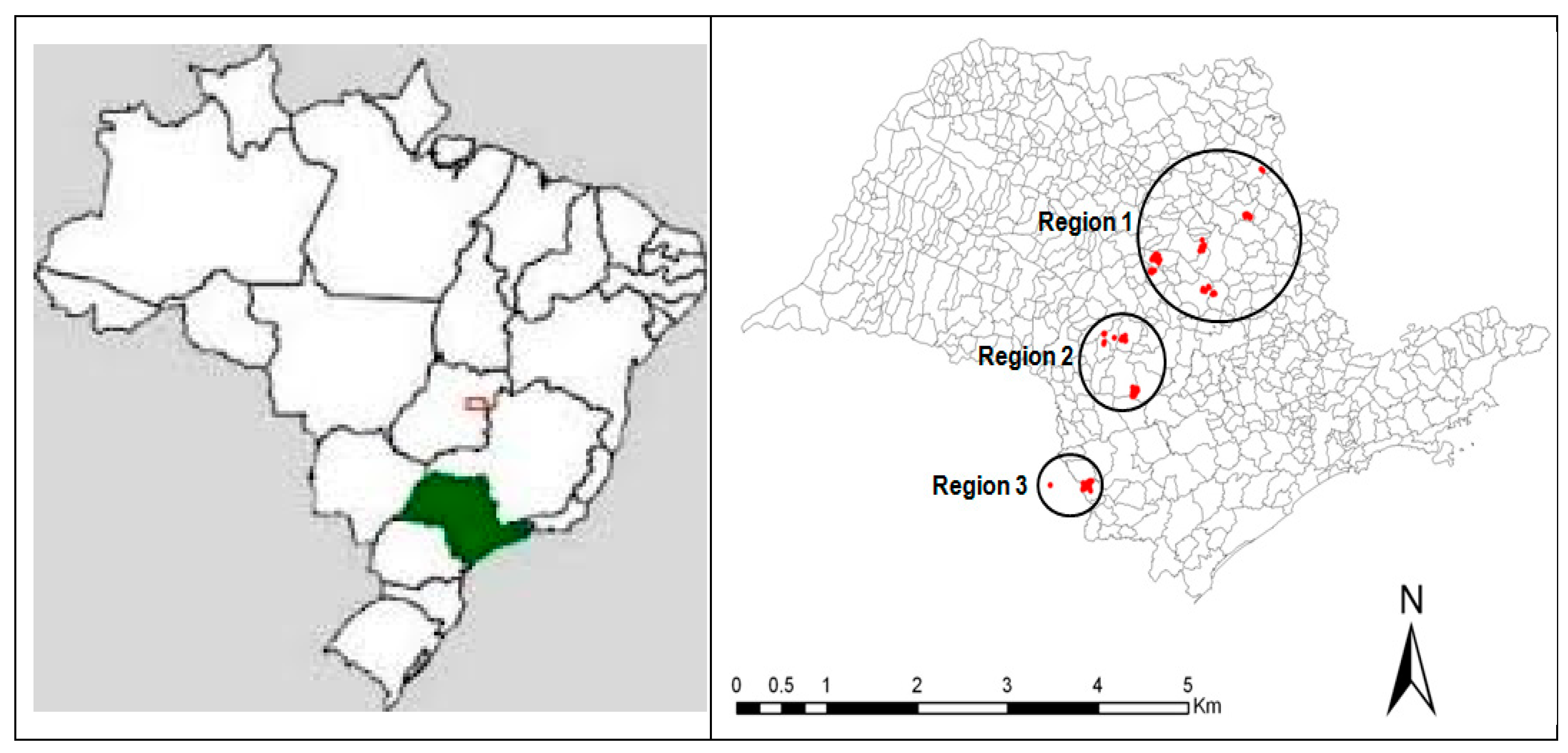
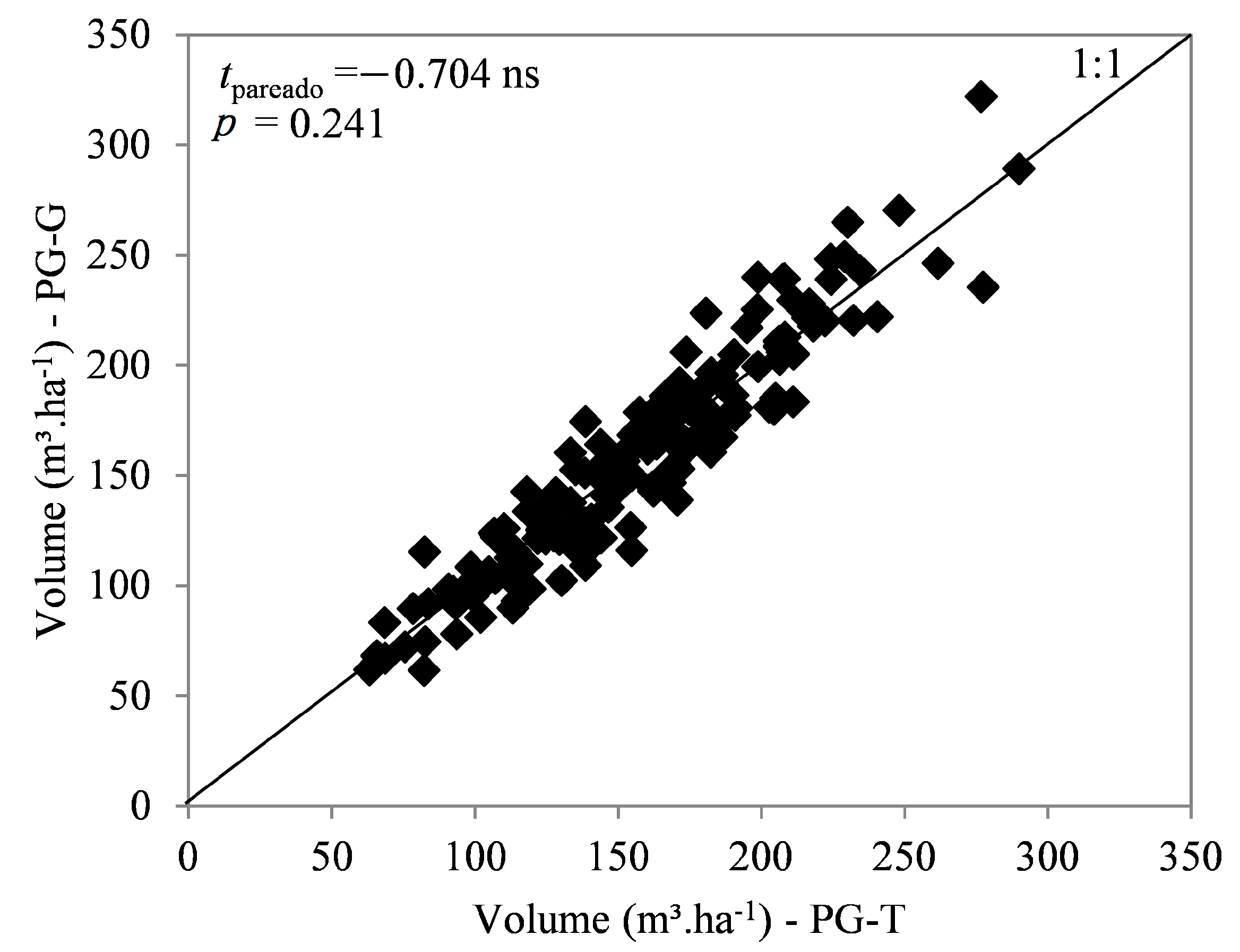
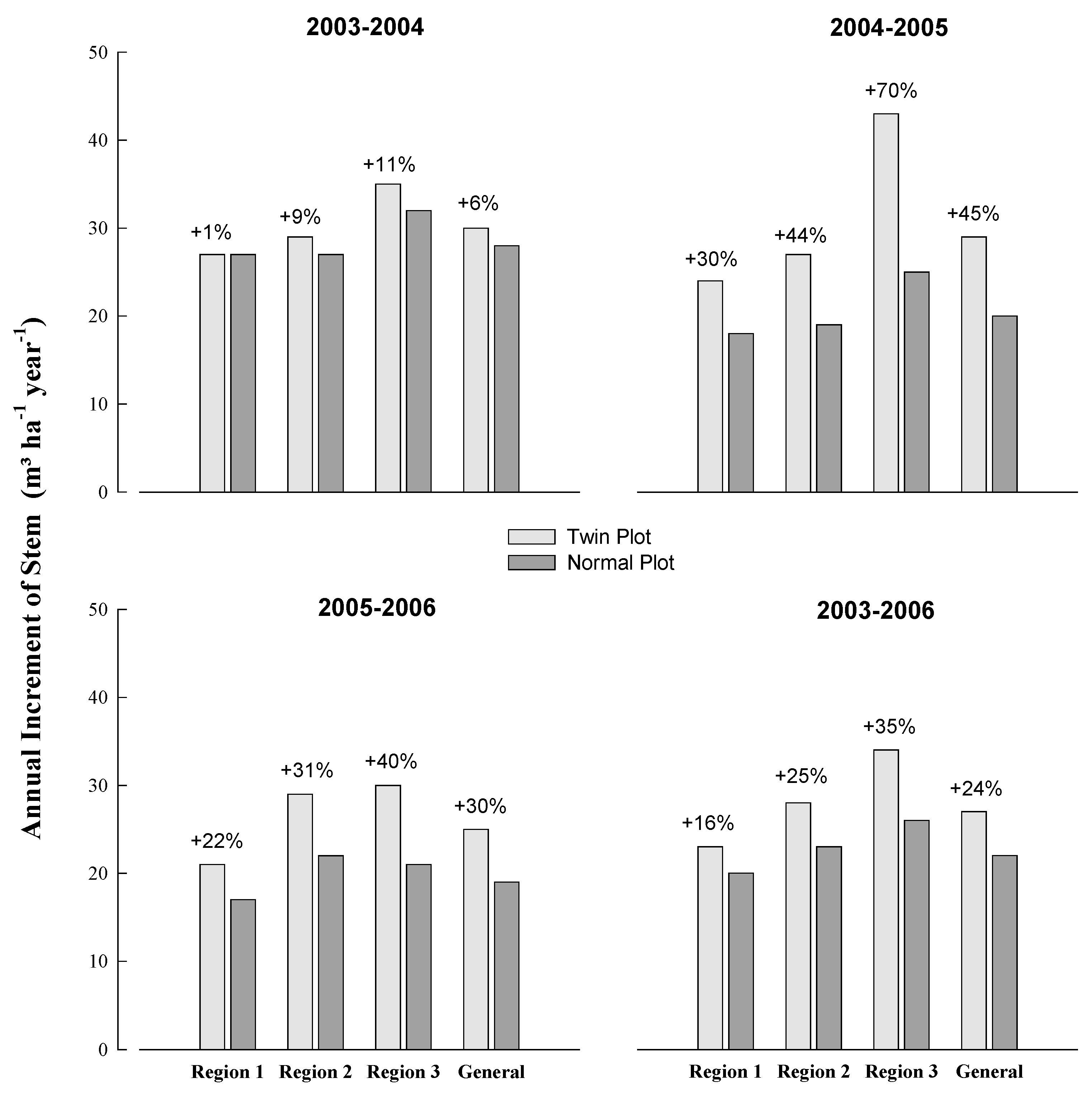

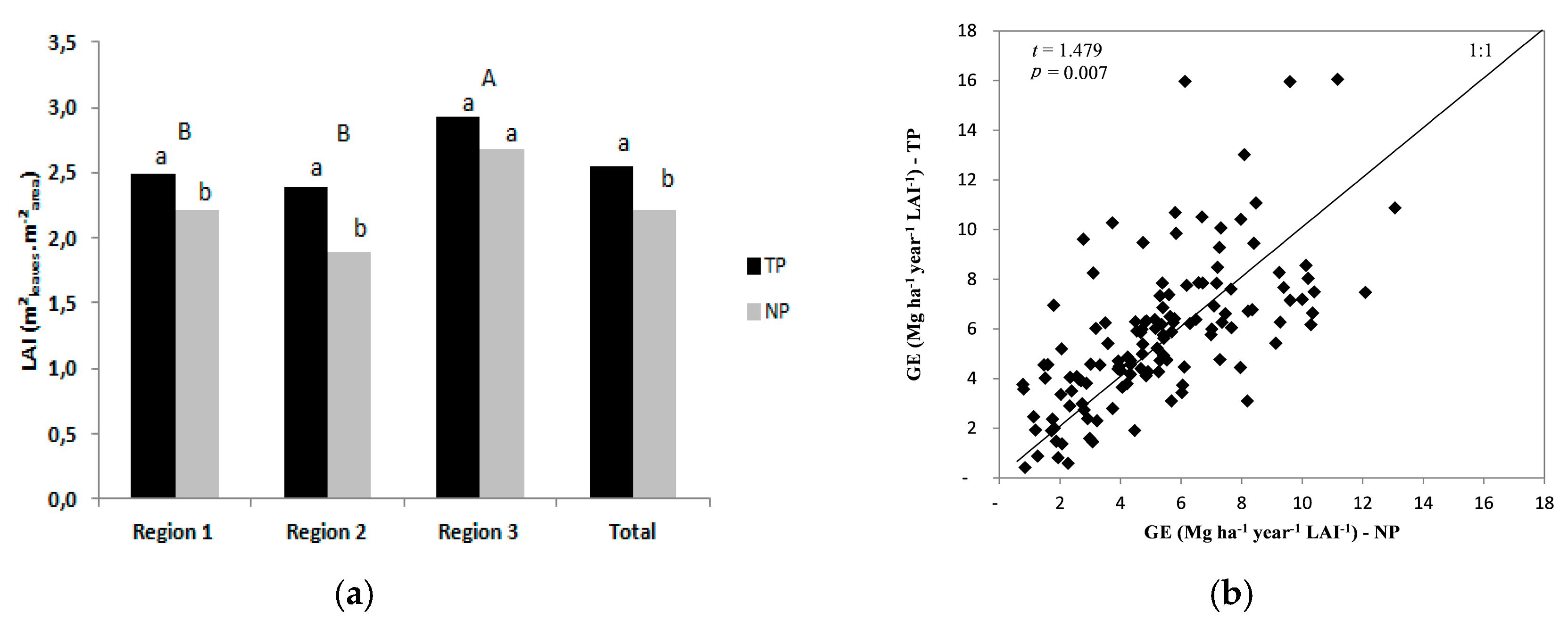
| Region | Temperature (°C) | Annual Rainfall (mm) | Annual Soil Water Deficit (mm) | Köppen Classification | ||
|---|---|---|---|---|---|---|
| Minimum | Average | Maximum | ||||
| 1 | 16.9 | 22.0 | 24.2 | 1461 | 47 to 110 | Aw and Cwa |
| 2 | 16.5 | 21.0 | 23.8 | 1369 | 3 to 17 | Cfa |
| 3 | 13.5 | 17.0 | 20.1 | 1549 | 0 | Cfb |
| Region | N | P | K | Ca | Mg |
|---|---|---|---|---|---|
| Region 1 | 85 | 35 | 180 | 160 | 40 |
| Region 2 | 90 | 22 | 140 | 50 | 25 |
| Region 3 | 85 | 25 | 130 | 140 | 60 |
| Extra 1 | 270 | 115 | 224 | 942 | 408 |
| Region | Clay 1 | Silt 1 | Sand 1 | pH 2 | OM 3 | P-res 4 | K 4 | Ca 4 | Mg 4 | H+Al 5 | SB 6 | CEC 7 | BS 8 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| # | % | g·kg−1 | mg·kg−1 | _____________________________mmolc·kg−1____________________________ | % | ||||||||
| Soil depth 0–15 cm | |||||||||||||
| 1 | 9 | 2 | 90 | 4 | 17 | 9 | 0.4 | 9 | 1 | 44 | 10 | 55 | 16 |
| 2 | 11 | 3 | 86 | 4 | 17 | 9 | 0.4 | 5 | 1 | 54 | 6 | 60 | 11 |
| 3 | 41 | 7 | 52 | 4 | 37 | 11 | 0.7 | 2 | 2 | 103 | 4 | 107 | 5 |
| Soil depth 15–30 cm | |||||||||||||
| 1 | 9 | 2 | 89 | 4 | 14 | 10 | 0.4 | 6 | 1 | 43 | 7 | 50 | 13 |
| 2 | 11 | 3 | 86 | 4 | 15 | 10 | 0.3 | 4 | 1 | 49 | 5 | 54 | 9 |
| 3 | 42 | 8 | 50 | 4 | 34 | 10 | 0.6 | 2 | 1 | 101 | 3 | 104 | 4 |
| Region | n 1 | Average Increment | Maximum Increment | ||||||
|---|---|---|---|---|---|---|---|---|---|
| NP | TP | NP | TP | NP | TP | NP | TP | ||
| m3·ha−1·year−1 | Mg·ha−1·year−1 | m3·ha−1·year−1 | Mg·ha−1·year−1 | ||||||
| Region 1 | 75 | 38 | 41 | 18 | 19 | 52 | 57 | 24 | 26 |
| Region 2 | 53 | 45 | 51 | 21 | 23 | 71 | 78 | 31 | 34 |
| Region 3 | 33 | 54 | 62 | 25 | 28 | 70 | 82 | 31 | 36 |
| General | 161 | 44 | 49 | 20 | 22 | 71 | 82 | 31 | 36 |
| Region | Number of Plots | Fertilization Response* | |
|---|---|---|---|
| Mg·ha−1·year−1 | |||
| Region 1 | 75 | 3.8 | c |
| Region 2 | 53 | 5.6 | b |
| Region 3 | 33 | 8.2 | A |
| Variable | General | Region 1 | Region 2 | Region 3 |
|---|---|---|---|---|
| OM | 0.31 ** | −0.16 ns | 0.02 ns | 0.12 ns |
| P | −0.18 * | −0.21 * | −0.13 ns | −0.39 * |
| K | −0.09 ns | −0.32 ** | −0.48 ** | −0.24 ns |
| Ca | −0.22 ** | −0.06 ns | −0.08 ns | −0.29 ns |
| Mg | −0.14 ns | −0.35 ** | −0.42 ** | −0.39 * |
| SB | −0.24 ** | −0.11 ns | −0.16 ns | −0.45 ** |
| CEC | 0.21 ** | −0.36 ** | 0.20 ns | 0.01 ns |
| BS | −0.26 ** | −0.13 ns | −0.15 ns | −0.25 ns |
| % clay | 0.26 ** | −0.48 ** | −0.19 ns | −0.02 ns |
| % silt | 0.09 ns | −0.01 ns | −0.24 ns | −0.59 ** |
| % sand | −0.24 ** | 0.48 ** | 0.25 ns | 0.15 ns |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, R.M.L.; Hakamada, R.E.; Bazani, J.H.; Otto, M.S.G.; Stape, J.L. Fertilization Response, Light Use, and Growth Efficiency in Eucalyptus Plantations across Soil and Climate Gradients in Brazil. Forests 2016, 7, 117. https://doi.org/10.3390/f7060117
Da Silva RML, Hakamada RE, Bazani JH, Otto MSG, Stape JL. Fertilization Response, Light Use, and Growth Efficiency in Eucalyptus Plantations across Soil and Climate Gradients in Brazil. Forests. 2016; 7(6):117. https://doi.org/10.3390/f7060117
Chicago/Turabian StyleDa Silva, Renato Meulman Leite, Rodrigo Eiji Hakamada, José Henrique Bazani, Marina Shinkai Gentil Otto, and José Luiz Stape. 2016. "Fertilization Response, Light Use, and Growth Efficiency in Eucalyptus Plantations across Soil and Climate Gradients in Brazil" Forests 7, no. 6: 117. https://doi.org/10.3390/f7060117
APA StyleDa Silva, R. M. L., Hakamada, R. E., Bazani, J. H., Otto, M. S. G., & Stape, J. L. (2016). Fertilization Response, Light Use, and Growth Efficiency in Eucalyptus Plantations across Soil and Climate Gradients in Brazil. Forests, 7(6), 117. https://doi.org/10.3390/f7060117





