Lessons from Research for Sustainable Development and Conservation in Borneo
Abstract
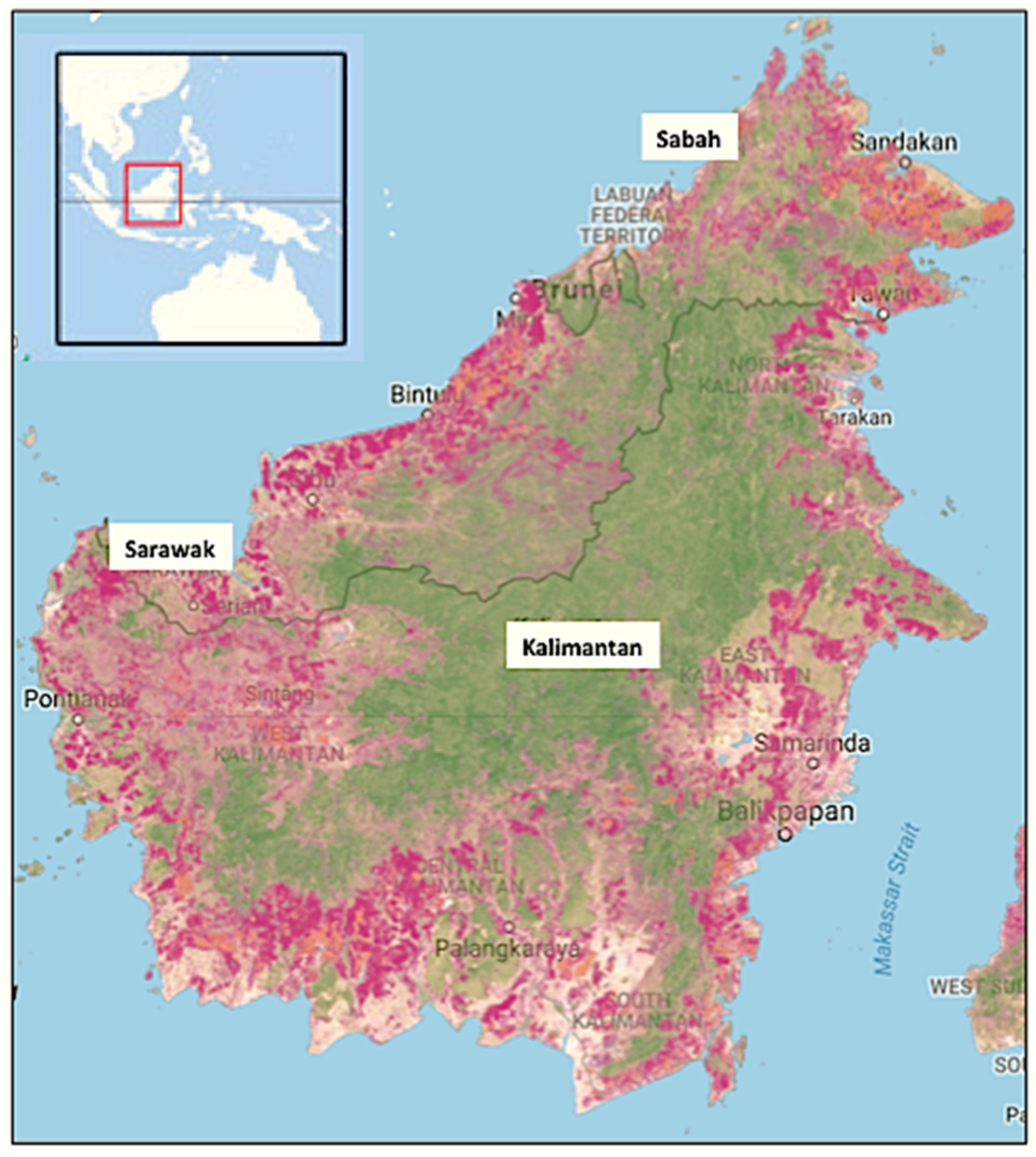
:1. Introduction
2. Lessons for Conservation
2.1. Save Logged Forests
2.2. Combat Poaching in Protected Areas
2.3. Maintain Forest Connectivity
2.4. Limit Infrastructure Expansion in Environmentally Sensitive Habitats
2.5. Include Climate Change in Conservation Planning
2.6. Promote Research as a Tool for Conservation
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Butler, R. A Desperate Effort to Save the Rainforests of Borneo; Yale Environment 360: New Haven, CT, USA, 2012. [Google Scholar]
- Laurance, W.F. In Imperiled Forests of Borneo, A Rich Tropical Eden Endures; Yale Environment 360: New Haven, CT, USA, 2013. [Google Scholar]
- Laurance, W.F. Forest destruction in tropical Asia. Curr. Sci. 2007, 93, 1544–1550. [Google Scholar]
- Koh, L.P.; Jukka Miettinen, J.; Liew, S.C.; Ghazoul, J. Remotely sensed evidence of tropical peatland conversion to oil palm. Proc. Nat. Acad. Sci. USA 2011, 108, 5127–5132. [Google Scholar] [CrossRef] [PubMed]
- Gaveau, D.L.A.; Sheil, D.; Husnayaen; Salim, M.A.; Arjasakusuma, S.; Ancrenaz, M.; Pacheco, P.; Meijaard, E. Rapid conversions and avoided deforestation: Examining four decades of industrial plantation expansion in Borneo. Scientif. Rep. 2016, 6, 32017. [Google Scholar] [CrossRef] [PubMed]
- Meijaard, E.; Sheil, D. Is wildlife research useful for wildlife conservation in the tropics? A review for Borneo with global implications. Biodiv. Conserv. 2007, 16, 3053–3065. [Google Scholar] [CrossRef]
- Laurance, W.F.; Useche, D.C.; Rendeiro, J.; Kalka, M.; Bradshaw, C.J.A.; Sloan, S.P.; Laurance, S.G.; Campbell, M.; Abernethy, K.; Alvarez, P.; et al. Averting biodiversity collapse in tropical forest protected areas. Nature 2012, 489, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Putz, F.E.; Zuidema, P.A.; Synnott, T.; Peña-Claros, M.; Pinard, M.A.; Sheil, D.; Vanclay, J.K.; Sist, P.; Gourlet-Fleury, S.; Griscom, B.; et al. Sustaining conservation values in selectively logged tropical forests: The attained and the attainable. Cons. Lett. 2012, 5, 296–303. [Google Scholar] [CrossRef]
- Lynam, A.J.; Porter, L.; Campos-Arceiz, A. Introduction: The challenge of conservation in changing tropical Southeast Asia. Conserv. Biol. 2016, 30, 931–932. [Google Scholar] [CrossRef] [PubMed]
- Gibson, L.; Lee, T.M.; Koh, L.P.; Brook, B.W.; Gardner, T.; Barlow, J.; Peres, C.; Bradshaw, C.; Laurance, W.F.; Lovejoy, T.E.; et al. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 2011, 478, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.P.; Laurance, W.F. Biodiversity despite selective logging. Science 2013, 339, 646–647. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F.; Edwards, D.P. Saving logged tropical forests. Front. Ecol. Environ. 2014, 12, 147. [Google Scholar] [CrossRef]
- Wright, S.J.; Stonger, K.; Beckman, N.; Corlett, R.; Dirzo, R.; Muller-Landau, H.; Nuñez-Ituri, G.; Peres, C.; Wang, B. The plight of large animals in tropical forests and the consequences for plant regeneration. Biotropica 2007, 39, 289–291. [Google Scholar] [CrossRef]
- Harrison, R.D.; Sreekar, R.; Brodie, J.F.; Brook, S.; Luskin, M.; O’Kelly, H.; Rao, M.; Scheffers, B.; Velho, N. Impacts of hunting on tropical forests of Southeast Asia. Conserv. Biol. 2016, 30, 972–981. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.D. Emptying the forest: Hunting and the extirpation of wildlife from tropical nature reserves. BioScience 2011, 61, 919–924. [Google Scholar] [CrossRef]
- Borneo Futures. Borneo Futures: Science For Change. Available online: http://www.borneofutures.org/about.html (accessed on 10 December 2016).
- Henn, C. These 8 Organizations Refuse to Let the Illegal Wildlife Trade Win; One Green Planet: New York, NY, USA, 2014. [Google Scholar]
- Milman, O. Melbourne Woman Takes up Fight to Stop Rhino Horn Smuggling; The Guardian: London, UK, 2014. [Google Scholar]
- Christy, B. First ever Fatwa Issued against Wildlife Trafficking; National Geographic Online: Washington, DC, USA, 2014. [Google Scholar]
- Brook, B.W.; Sodhi, N.S.; Bradshaw, C.J. Synergies among extinction drivers under global change. Trends Ecol. Evol. 2008, 23, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F.; Useche, D.C. Environmental synergisms and extinctions of tropical species. Conserv. Biol. 2009, 23, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F.; Goosem, M.; Laurance, S.G. Impacts of roads and linear clearings on tropical forests. Trends Ecol. Evol. 2009, 24, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F.; Balmford, A. A global map for road building. Nature 2013, 495, 308–309. [Google Scholar] [CrossRef] [PubMed]
- Clements, G.R.; Lynam, A.J.; Gaveau, D.; Yap, W.; Lhota, S.; Goosem, M.; Laurance, S.G.; Laurance, W.F. Where and how are roads endangering mammals in Southeast Asia’s forests? PLoS ONE 2014, 9, e115376. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F.; Clements, G.R.; Sloan, S.; O’Connell, C.; Mueller, N.D.; Goosem, M.; Venter, O.; Edwards, D.P.; Phalan, B.; Balmford, A.; et al. A global strategy for road building. Nature 2014, 513, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Brodie, J.; Post, E.; Laurance, W.F. Climate change and tropical biodiversity: A new focus. Trends Ecol. Evol. 2012, 23, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Power, S.; Delage, F.; Chung, C.; Kociuba, G.; Keay, K. Robust twenty-first-century projections of El Niño and related precipitation variability. Nature 2013, 502, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, M.A.; Laurance, W.F. Synergisms among fire, land use, and climate change in the Amazon. Ambio 2008, 37, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Curran, L.M.; Leighton, M. Vertebrate responses to spatiotemporal variation in seed production of mast-fruiting Dipterocarpaceae. Ecol. Monogr. 2008, 70, 101–128. [Google Scholar] [CrossRef]
- Kimura, K. A tropical montane forest in Borneo as a source of fruit supply for frugivorous birds. Global Environ. Res. 2003, 7, 113–122. [Google Scholar]
- Laurance, W.F. Can research help to safeguard protected areas? Trends Ecol. Evol. 2013, 28, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Laurance, W.F. Adopt a forest. Biotropica 2008, 40, 3–6. [Google Scholar] [CrossRef]
- Ewers, R.M.; Didham, R.K.; Fahrig, L.; Ferraz, G.; Hector, A.; Holt, R.D.; Kapos, V.; Reynolds, G.; Sinun, W.; Snaddon, J.L.; et al. A large-scale forest fragmentation experiment: The Stability of Altered Forest Ecosystems Project. Phil. Trans. Roy. Soc. B 2011, 366, 3292–3302. [Google Scholar] [CrossRef] [PubMed]


© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laurance, W.F. Lessons from Research for Sustainable Development and Conservation in Borneo. Forests 2016, 7, 314. https://doi.org/10.3390/f7120314
Laurance WF. Lessons from Research for Sustainable Development and Conservation in Borneo. Forests. 2016; 7(12):314. https://doi.org/10.3390/f7120314
Chicago/Turabian StyleLaurance, William F. 2016. "Lessons from Research for Sustainable Development and Conservation in Borneo" Forests 7, no. 12: 314. https://doi.org/10.3390/f7120314
APA StyleLaurance, W. F. (2016). Lessons from Research for Sustainable Development and Conservation in Borneo. Forests, 7(12), 314. https://doi.org/10.3390/f7120314





