Elemental Stoichiometry of Tropical Peatland Trees: Implications for Adaptation and Carbon Sequestration †
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling
2.3. Sample Preparation and Extraction
2.4. Nutrient Analyses
2.5. Data Analysis
3. Results
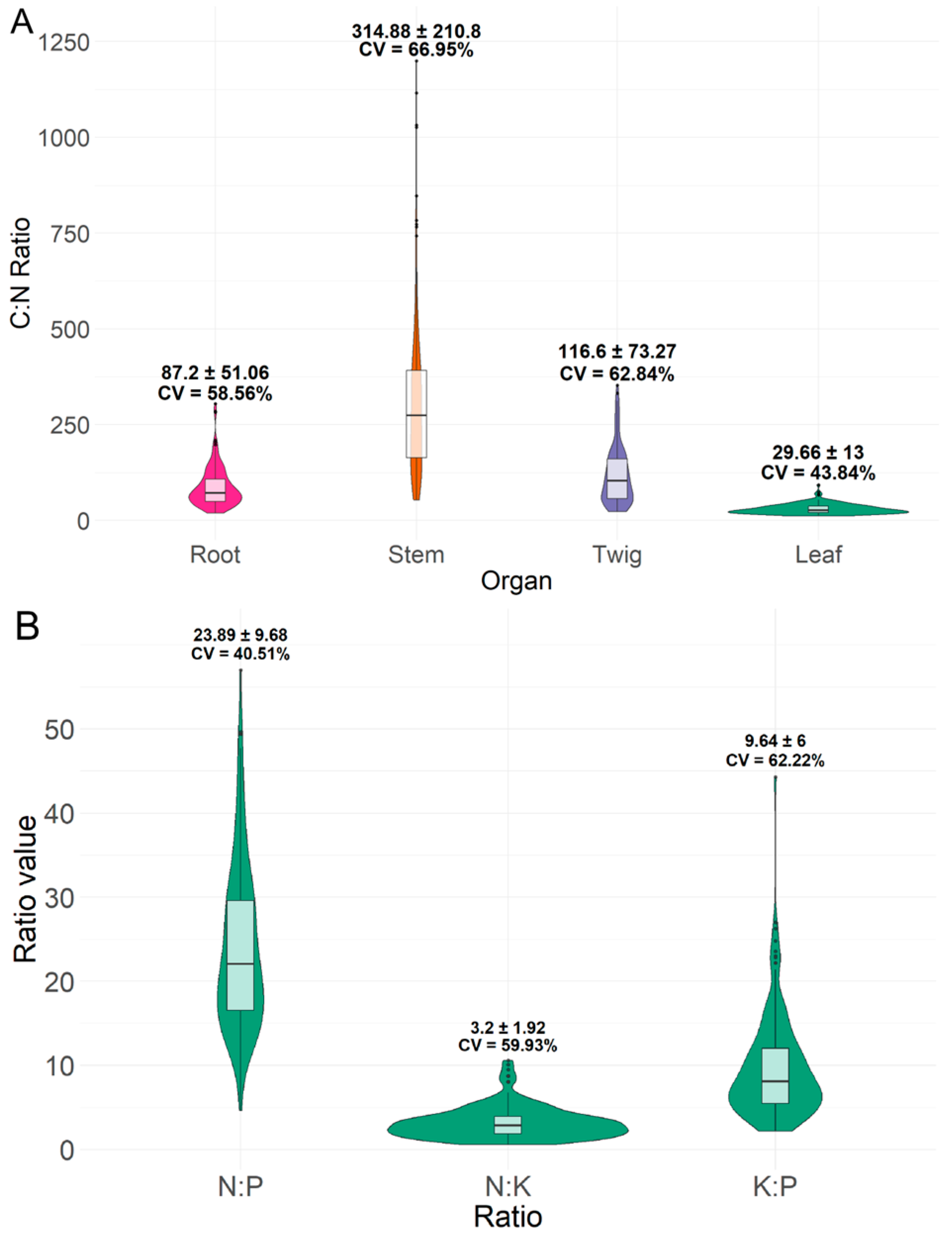
3.1. C:N Ratios in Plant Organs and N:P:K Stoichiometry in Leaves
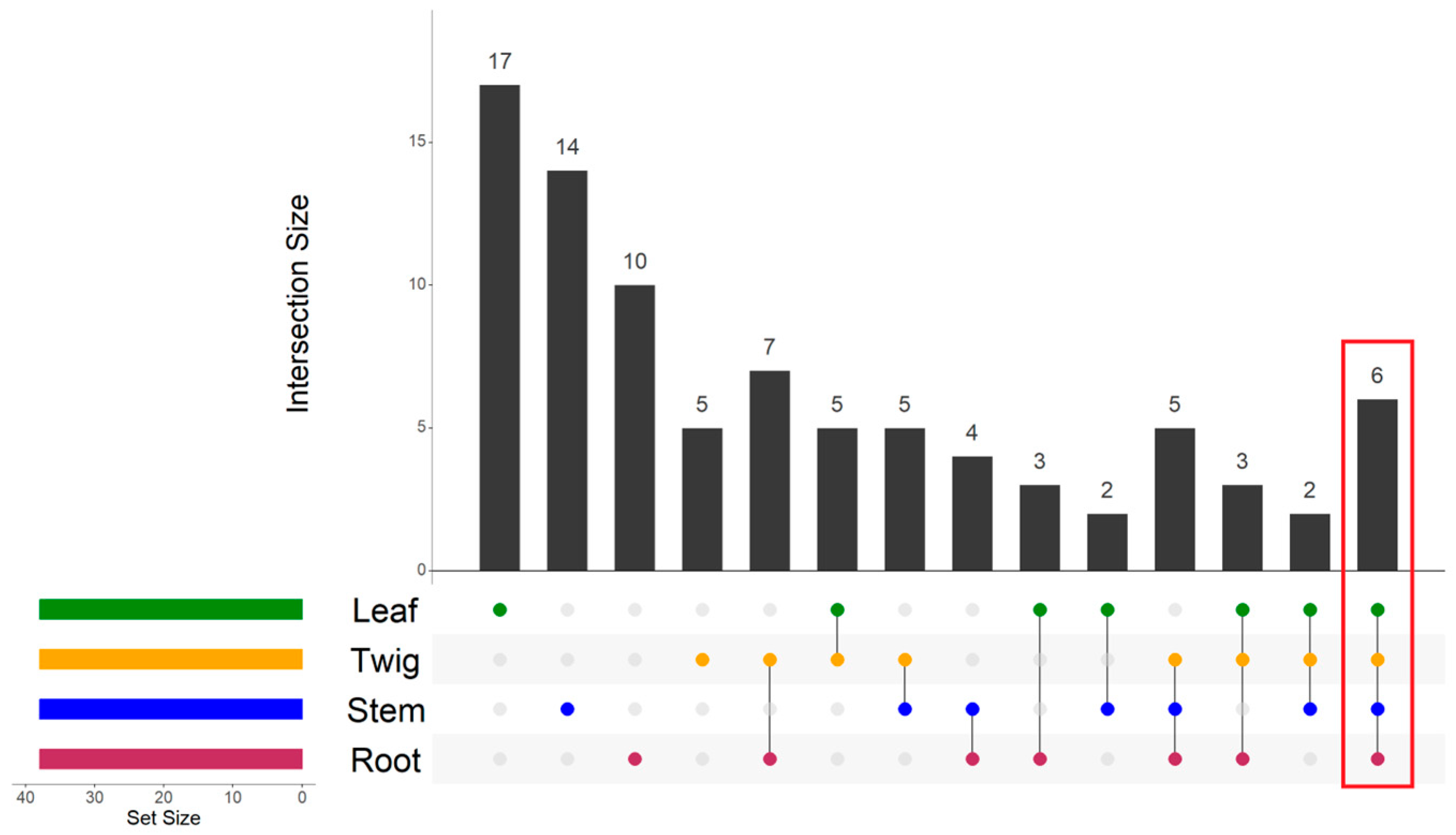
3.2. Hierarchical Clustering Based on C:N Ratios Across Four Organs
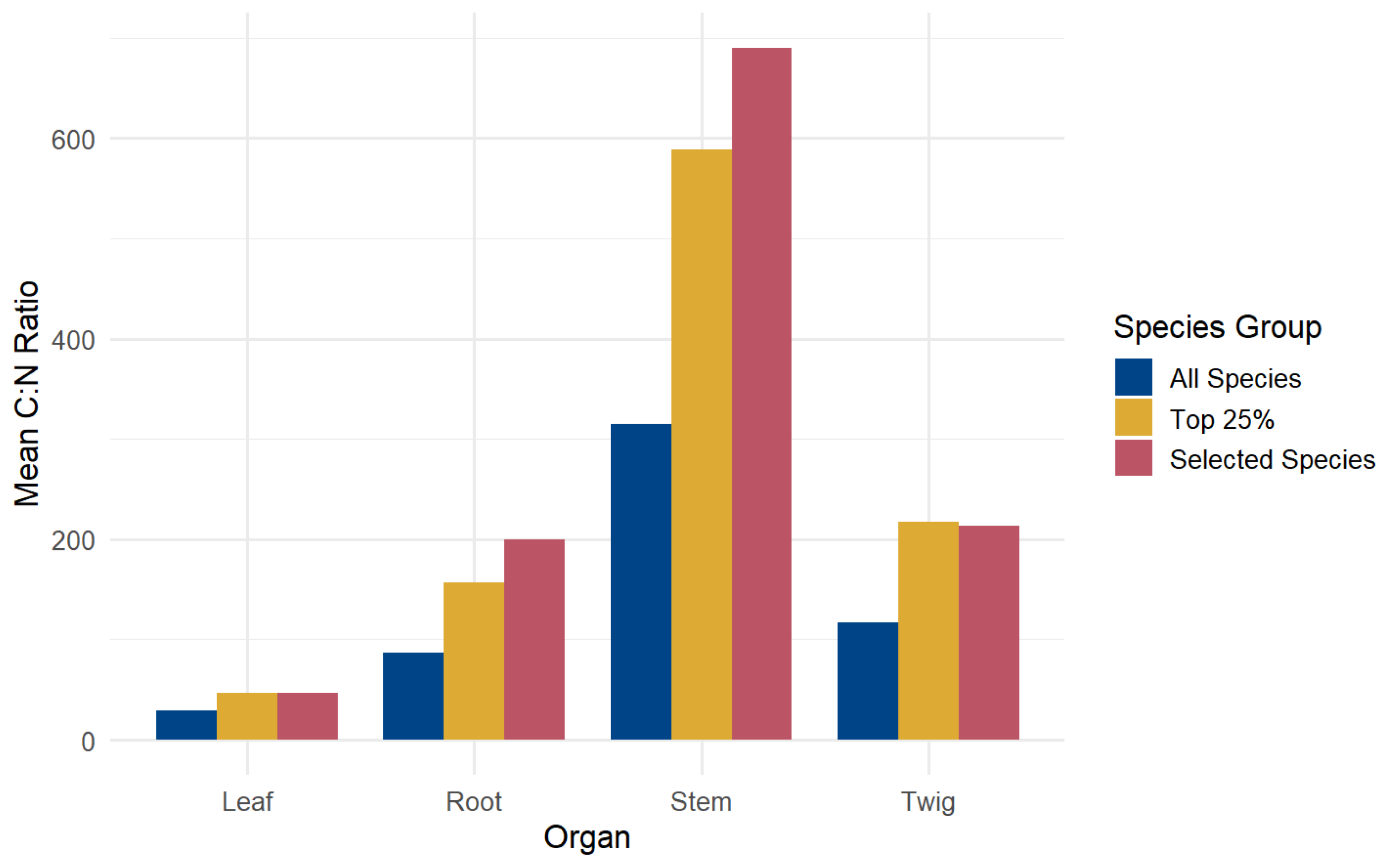
3.3. Species with the Highest C:N Ratios
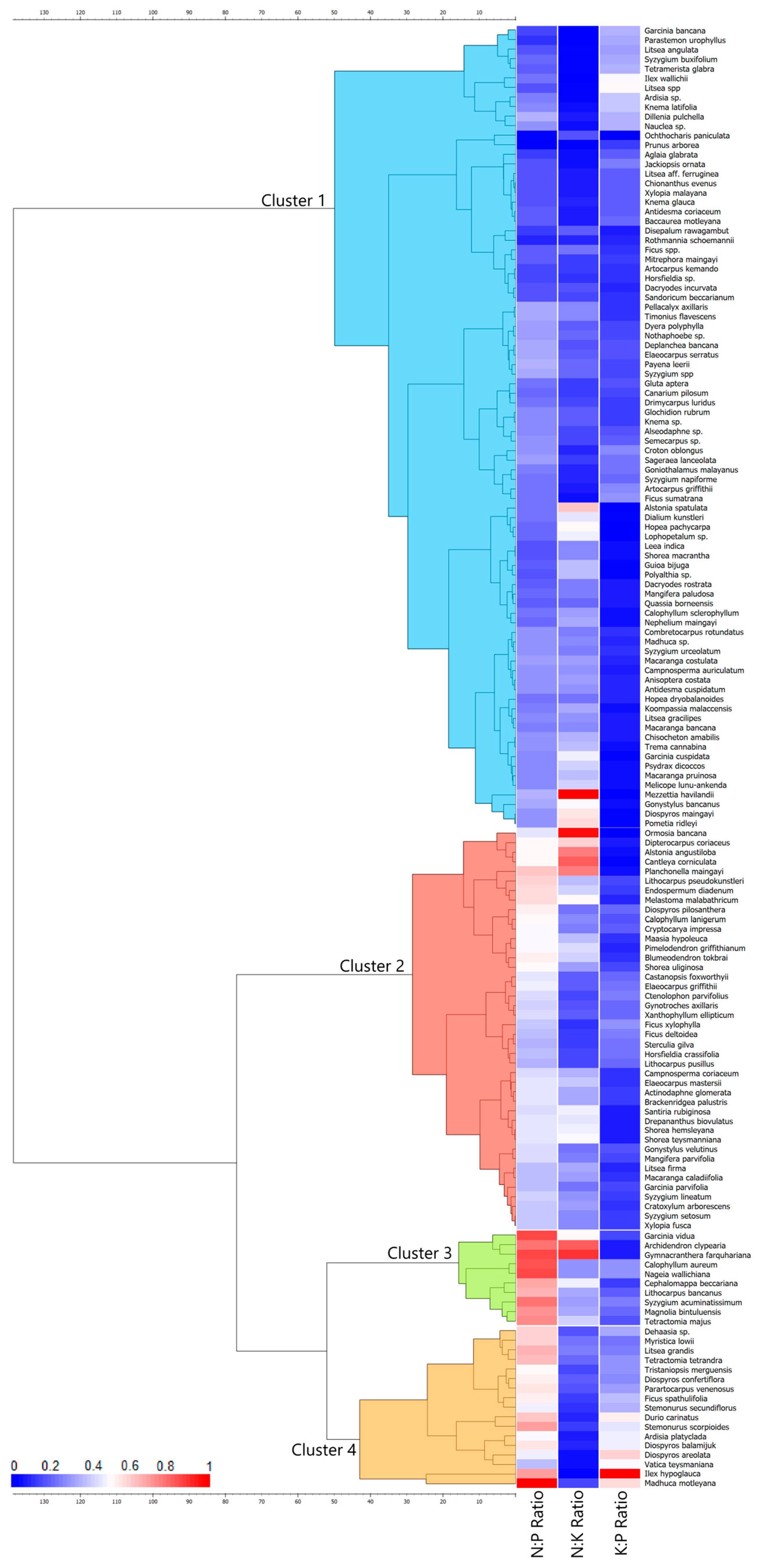
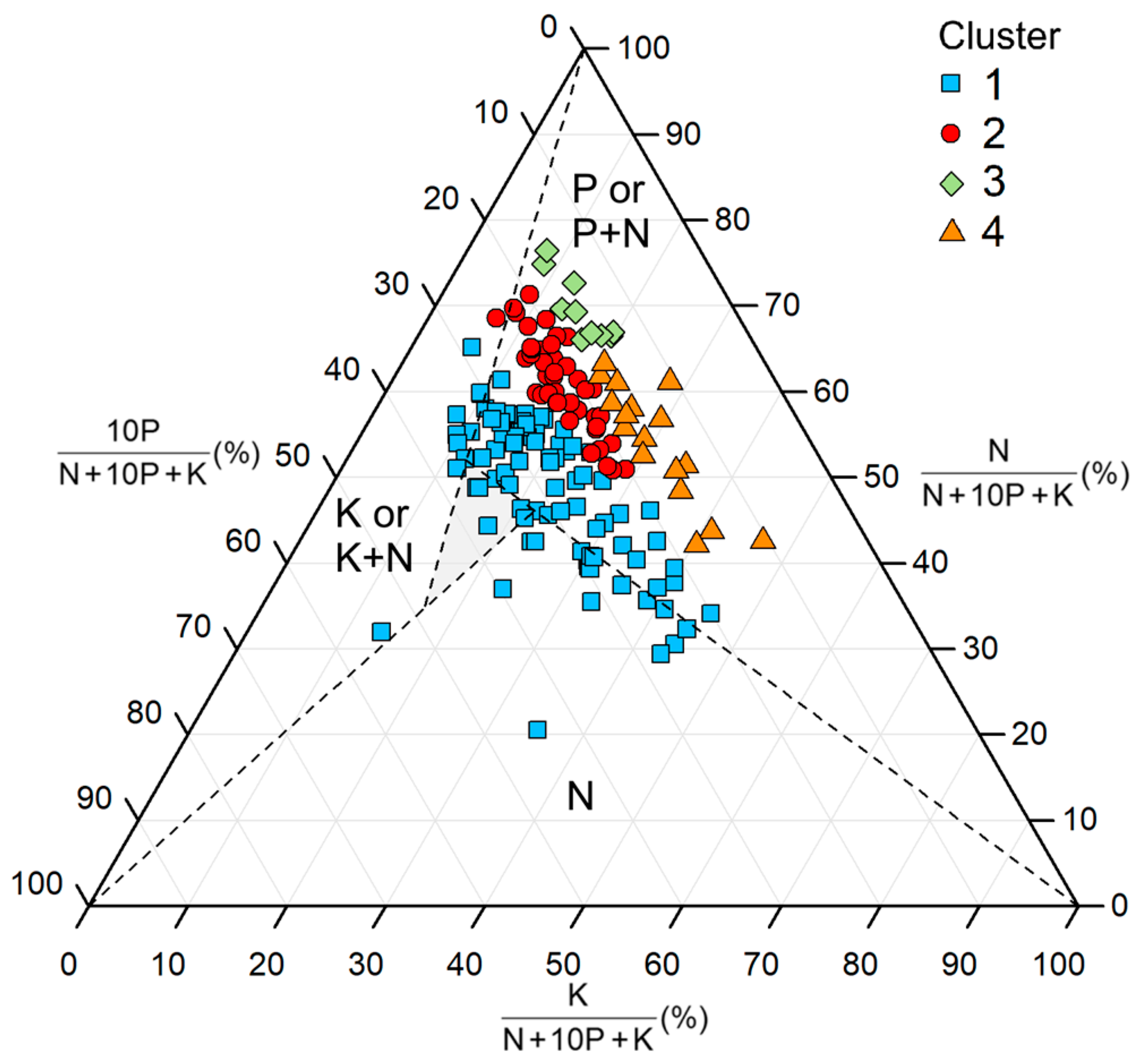
3.4. Hierarchical Clustering Based on Leaves N:P:K Stoichiometry
3.5. Nutrient Deficiencies Based on N:P:K Stoichiometry
4. Discussion
4.1. Organ C:N Ratios and Implications for Ecosystem and Carbon Sequestration
4.2. Leaf N:P:K Stoichiometry and Nutrient Deficiency in Tropical Peatland Trees
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Page, S.E.; Rieley, J.O.; Banks, C.J. Global and Regional Importance of the Tropical Peatland Carbon Pool. Glob. Change Biol. 2011, 17, 798–818. [Google Scholar] [CrossRef]
- Xu, J.; Morris, P.J.; Liu, J.; Holden, J. PEATMAP: Refining Estimates of Global Peatland Distribution Based on a Meta-Analysis. CATENA 2018, 160, 134–140. [Google Scholar] [CrossRef]
- Girkin, N.T.; Cooper, H.V.; Ledger, M.J.; O’Reilly, P.; Thornton, S.A.; Åkesson, C.M.; Cole, L.E.S.; Hapsari, K.A.; Hawthorne, D.; Roucoux, K.H. Tropical Peatlands in the Anthropocene: The Present and the Future. Anthropocene 2022, 40, 100354. [Google Scholar] [CrossRef]
- Anda, M.; Ritung, S.; Suryani, E.; Hikmat, M.; Yatno, E.; Mulyani, A.; Subandiono, R.E. Revisiting Tropical Peatlands in Indonesia: Semi-Detailed Mapping, Extent and Depth Distribution Assessment. Geoderma 2021, 402, 115235. [Google Scholar] [CrossRef]
- Hooijer, A.; Page, S.; Canadell, J.G.; Silvius, M.; Kwadijk, J.; Wösten, H.; Jauhiainen, J. Current and Future CO2 Emissions from Drained Peatlands in Southeast Asia. Biogeosciences 2010, 7, 1505–1514. [Google Scholar] [CrossRef]
- Uda, S.K.; Hein, L.; Sumarga, E. Towards Sustainable Management of Indonesian Tropical Peatlands. Wetl. Ecol. Manag. 2017, 25, 683–701. [Google Scholar] [CrossRef]
- Noon, M.L.; Goldstein, A.; Ledezma, J.C.; Roehrdanz, P.R.; Cook-Patton, S.C.; Spawn-Lee, S.A.; Wright, T.M.; Gonzalez-Roglich, M.; Hole, D.G.; Rockström, J.; et al. Mapping the Irrecoverable Carbon in Earth’s Ecosystems. Nat. Sustain. 2021, 5, 37–46. [Google Scholar] [CrossRef]
- Warren, M.; Hergoualc’h, K.; Kauffman, J.B.; Murdiyarso, D.; Kolka, R. An Appraisal of Indonesia’s Immense Peat Carbon Stock Using National Peatland Maps: Uncertainties and Potential Losses from Conversion. Carbon Balance Manag. 2017, 12, 12. [Google Scholar] [CrossRef]
- Ribeiro, K.; Pacheco, F.S.; Ferreira, J.W.; De Sousa-Neto, E.R.; Hastie, A.; Krieger Filho, G.C.; Alvalá, P.C.; Forti, M.C.; Ometto, J.P. Tropical Peatlands and Their Contribution to the Global Carbon Cycle and Climate Change. Glob. Change Biol. 2021, 27, 489–505. [Google Scholar] [CrossRef]
- Page, S.E.; Baird, A.J. Peatlands and Global Change: Response and Resilience. Annu. Rev. Environ. Resour. 2016, 41, 35–57. [Google Scholar] [CrossRef]
- Vesala, R.; Kiheri, H.; Hobbie, E.A.; Van Dijk, N.; Dise, N.; Larmola, T. Atmospheric Nitrogen Enrichment Changes Nutrient Stoichiometry and Reduces Fungal N Supply to Peatland Ericoid Mycorrhizal Shrubs. Sci. Total Environ. 2021, 794, 148737. [Google Scholar] [CrossRef]
- Omar, M.S.; Ifandi, E.; Sukri, R.S.; Kalaitzidis, S.; Christanis, K.; Lai, D.T.C.; Bashir, S.; Tsikouras, B. Peatlands in Southeast Asia: A Comprehensive Geological Review. Earth-Sci. Rev. 2022, 232, 104149. [Google Scholar] [CrossRef]
- Kayranli, B.; Scholz, M.; Mustafa, A.; Hedmark, Å. Carbon Storage and Fluxes within Freshwater Wetlands: A Critical Review. Wetlands 2010, 30, 111–124. [Google Scholar] [CrossRef]
- Alonso-Serra, J. Carbon Sequestration: Counterintuitive Feedback of Plant Growth. Quant. Plant Biol. 2021, 2, e11. [Google Scholar] [CrossRef]
- Dommain, R.; Cobb, A.R.; Joosten, H.; Glaser, P.H.; Chua, A.F.L.; Gandois, L.; Kai, F.; Noren, A.; Salim, K.A.; Su’ut, N.S.H.; et al. Forest Dynamics and Tip-up Pools Drive Pulses of High Carbon Accumulation Rates in a Tropical Peat Dome in Borneo (Southeast Asia). J. Geophys. Res. Biogeosci. 2015, 120, 617–640. [Google Scholar] [CrossRef]
- Yule, C.M.; Gomez, L.N. Leaf Litter Decomposition in a Tropical Peat Swamp Forest in Peninsular Malaysia. Wetl. Ecol. Manag. 2009, 17, 231–241. [Google Scholar] [CrossRef]
- Lampela, M.; Jauhiainen, J.; Sarkkola, S.; Vasander, H. Promising Native Tree Species for Reforestation of Degraded Tropical Peatlands. For. Ecol. Manag. 2017, 394, 52–63. [Google Scholar] [CrossRef]
- Kissinger, K.; Pitri, R.M.N.; Nasrulloh, A.V. Implementation of Plant Selection Based-On Plant Growth on Revegetation of Peatland in South Kalimantan. Int. J. Environ. Agric. Biotechnol. 2022, 7, 137–142. [Google Scholar] [CrossRef]
- Anjani, R.; Siahaan, H. Initial Growth of Four Endemic Species in Degraded Peatland Revegetation in Ogan Komering Ilir, South Sumatra. IOP Conf. Ser. Earth Environ. Sci. 2023, 1201, 012056. [Google Scholar] [CrossRef]
- Peng, Y.; Schmidt, I.K.; Zheng, H.; Heděnec, P.; Bachega, L.R.; Yue, K.; Wu, F.; Vesterdal, L. Tree Species Effects on Topsoil Carbon Stock and Concentration Are Mediated by Tree Species Type, Mycorrhizal Association, and N-Fixing Ability at the Global Scale. For. Ecol. Manag. 2020, 478, 118510. [Google Scholar] [CrossRef]
- Fernández-Guisuraga, J.M.; Calvo, L.; Marcos, E. Plant Species Diversity and Abundance of Functional Groups Influence Net Nitrogen Mineralization along Productivity Gradients in Montane Calluna-dominated Heathlands. Plants People Planet 2025, 7, 1388–1402. [Google Scholar] [CrossRef]
- Straková, P.; Penttilä, T.; Laine, J.; Laiho, R. Disentangling Direct and Indirect Effects of Water Table Drawdown on Above- and Belowground Plant Litter Decomposition: Consequences for Accumulation of Organic Matter in Boreal Peatlands. Glob. Change Biol. 2012, 18, 322–335. [Google Scholar] [CrossRef]
- Sterner, R.W.; Elser, J.J. Ecological Stoichiometry: The Biology of Elements from Molecules to the Biosphere; Princeton University Press: Princeton, NJ, USA, 2002. [Google Scholar]
- Lynch, M.J.; Mulvaney, M.J.; Hodges, S.C.; Thompson, T.L.; Thomason, W.E. Decomposition, Nitrogen and Carbon Mineralization from Food and Cover Crop Residues in the Central Plateau of Haiti. SpringerPlus 2016, 5, 973. [Google Scholar] [CrossRef]
- Hapsari, K.A.; Biagioni, S.; Jennerjahn, T.C.; Reimer, P.M.; Saad, A.; Achnopha, Y.; Sabiham, S.; Behling, H. Environmental Dynamics and Carbon Accumulation Rate of a Tropical Peatland in Central Sumatra, Indonesia. Quat. Sci. Rev. 2017, 169, 173–187. [Google Scholar] [CrossRef]
- Jílková, V.; Straková, P.; Frouz, J. Foliage C:N Ratio, Stage of Organic Matter Decomposition and Interaction with Soil Affect Microbial Respiration and Its Response to C and N Addition More than C:N Changes during Decomposition. Appl. Soil Ecol. 2020, 152, 103568. [Google Scholar] [CrossRef]
- Koerselman, W.; Meuleman, A.F.M. The Vegetation N:P Ratio: A New Tool to Detect the Nature of Nutrient Limitation. J. Appl. Ecol. 1996, 33, 1441–1450. [Google Scholar] [CrossRef]
- Güsewell, S.; Koerselman, W. Variation in Nitrogen and Phosphorus Concentrations of Wetland Plants. Perspect. Plant Ecol. Evol. Syst. 2002, 5, 37–61. [Google Scholar] [CrossRef]
- Venterink, H.O.; Wassen, M.J.; Verkroost, A.W.M.; De Ruiter, P.C. Species Richness–Productivity Patterns Differ Between N-, P-, and K-Limited Wetlands. Ecology 2003, 84, 2191–2199. [Google Scholar] [CrossRef]
- Brearley, F.Q.; Mansur, M. Nutrient Stoichiometry of Nepenthes Species from a Bornean Peat Swamp Forest. Carniv. Plant Newsl. 2012, 41, 105–108. [Google Scholar] [CrossRef]
- Vourlitis, G.L.; De Almeida Lobo, F.; Lawrence, S.; Holt, K.; Zappia, A.; Pinto, O.B.; De Souza Nogueira, J. Nutrient Resorption in Tropical Savanna Forests and Woodlands of Central Brazil. Plant Ecol. 2014, 215, 963–975. [Google Scholar] [CrossRef]
- Yanbuaban, M.; Nuyim, T.; Matsubara, T.; Watanabe, T.; Osaki, M. Nutritional Ecology of Plants Grown in a Tropical Peat Swamp. Tropics 2007, 16, 31–39. [Google Scholar] [CrossRef]
- Wang, M.; Moore, T.R. Carbon, Nitrogen, Phosphorus, and Potassium Stoichiometry in an Ombrotrophic Peatland Reflects Plant Functional Type. Ecosystems 2014, 17, 673–684. [Google Scholar] [CrossRef]
- Balai Penelitian Tanah. Analisis Kimia Tanah, Tanaman, Air, Dan Pupuk; Balai Penelitian Tanah, Badan Penelitian dan Pengembangan Pertanian, Departemen Pertanian: Bogor, Indonesia, 2005. [Google Scholar]
- ISO/IEC 17025:2017; General Requirements for the Competence of Testing and Calibration Laboratories. International Organization for Standardization (ISO): Geneva, Switzerland, 2017.
- Zhang, H.; Guo, W.; Wang, G.G.; Yu, M.; Wu, T. Effect of Environment and Genetics on Leaf N and P Stoichiometry for Quercus Acutissima across China. Eur. J. For. Res. 2016, 135, 795–802. [Google Scholar] [CrossRef]
- Zhang, J.; He, N.; Liu, C.; Xu, L.; Chen, Z.; Li, Y.; Wang, R.; Yu, G.; Sun, W.; Xiao, C.; et al. Variation and Evolution of C:N Ratio among Different Organs Enable Plants to Adapt to N-limited Environments. Glob. Change Biol. 2020, 26, 2534–2543. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, N.; Liu, C.; Yang, H.; Li, M.; Yu, G.; Wilcox, K.; Yu, Q.; He, N. C:N:P Stoichiometry in China’s Forests: From Organs to Ecosystems. Funct. Ecol. 2018, 32, 50–60. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed.; Sinauer Associates: Sunderland, MA, USA, 2015. [Google Scholar]
- Liang, X.; Liu, S.; Wang, H.; Wang, J. Variation of Carbon and Nitrogen Stoichiometry along a Chronosequence of Natural Temperate Forest in Northeastern China. J. Plant Ecol. 2018, 11, 339–350. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Photosynthetic Nitrogen Assimilation: Inter-Pathway Control and Signaling. In Photosynthetic Nitrogen Assimilation and Associated Carbon and Respiratory Metabolism; Foyer, C.H., Noctor, G., Eds.; Advances in Photosynthesis and Respiration; Springer: Dordrecht, The Netherlands, 2002; Volume 12, pp. 1–22. ISBN 978-0-7923-6336-1. [Google Scholar]
- Bauters, M.; Vercleyen, O.; Vanlauwe, B.; Six, J.; Bonyoma, B.; Badjoko, H.; Hubau, W.; Hoyt, A.; Boudin, M.; Verbeeck, H.; et al. Long-term Recovery of the Functional Community Assembly and Carbon Pools in an African Tropical Forest Succession. Biotropica 2019, 51, 319–329. [Google Scholar] [CrossRef]
- Fagundes, M.V.; Souza, A.F.; Oliveira, R.S.; Ganade, G. Functional Traits above and below Ground Allow Species with Distinct Ecological Strategies to Coexist in the Largest Seasonally Dry Tropical Forest in the Americas. Front. For. Glob. Change 2022, 5, 930099. [Google Scholar] [CrossRef]
- Moore, T.R.; Alfonso, A.; Clarkson, B.R. Plant Uptake of Organic Nitrogen in Two Peatlands. Plant Soil 2018, 433, 391–400. [Google Scholar] [CrossRef]
- Nwaishi, F.; Morison, M.; Plach, J.; Macrae, M.; Petrone, R. Carbon and Nutrient Stoichiometric Relationships in the Soil–Plant Systems of Disturbed Boreal Forest Peatlands within Athabasca Oil Sands Region, Canada. Forests 2022, 13, 865. [Google Scholar] [CrossRef]
- Hogan, J.A.; Valverde-Barrantes, O.J.; Tang, W.; Ding, Q.; Xu, H.; Baraloto, C. Evidence of Elemental Homeostasis in Fine Root and Leaf Tissues of Saplings across a Fertility Gradient in Tropical Montane Forest in Hainan, China. Plant Soil 2021, 460, 625–646. [Google Scholar] [CrossRef]
- Jiang, P.; Cao, Y.; Chen, Y. C, N, P Stoichiometric Characteristics of Tree, Shrub, Herb Leaves and Litter in Forest Community of Shaanxi Province, China. ACTA Ecol. Sin. 2023, 43, 8749–8758. [Google Scholar] [CrossRef]
- Ghimire, B.; Riley, W.J.; Koven, C.D.; Kattge, J.; Rogers, A.; Reich, P.B.; Wright, I.J. A Global Trait-based Approach to Estimate Leaf Nitrogen Functional Allocation from Observations. Ecol. Appl. 2017, 27, 1421–1434. [Google Scholar] [CrossRef]
- Dieckow, J.; Mielniczuk, J.; Knicker, H.; Bayer, C.; Dick, D.P.; Kögel-Knabner, I. Comparison of Carbon and Nitrogen Determination Methods for Samples of a Paleudult Subjected to No-till Cropping Systems. Sci. Agric. 2007, 64, 532–540. [Google Scholar] [CrossRef]
- Novaes, E.; Kirst, M.; Chiang, V.; Winter-Sederoff, H.; Sederoff, R. Lignin and Biomass: A Negative Correlation for Wood Formation and Lignin Content in Trees. Plant Physiol. 2010, 154, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; He, F.; Tian, D.; Zou, D.; Yan, Z.; Yang, Y.; Zhou, T.; Huang, K.; Shen, H.; Fang, J. Variations and Determinants of Carbon Content in Plants: A Global Synthesis. Biogeosciences 2018, 15, 693–702. [Google Scholar] [CrossRef]
- Herrera-Ramírez, D.; Hartmann, H.; Römermann, C.; Trumbore, S.; Muhr, J.; Maracahipes-Santos, L.; Brando, P.; Silvério, D.; Huang, J.; Kuhlmann, I.; et al. Anatomical Distribution of Starch in the Stemwood Influences Carbon Dynamics and Suggests Storage-growth Trade-offs in Some Tropical Trees. J. Ecol. 2023, 111, 2532–2548. [Google Scholar] [CrossRef]
- Carvalho, F.; Brown, K.A.; Waller, M.P.; Boom, A. Leaf Traits Interact with Management and Water Table to Modulate Ecosystem Properties in Fen Peatlands. Plant Soil 2019, 441, 331–347. [Google Scholar] [CrossRef]
- Schillereff, D.N.; Chiverrell, R.C.; Sjöström, J.K.; Kylander, M.E.; Boyle, J.F.; Davies, J.A.C.; Toberman, H.; Tipping, E. Phosphorus Supply Affects Long-Term Carbon Accumulation in Mid-Latitude Ombrotrophic Peatlands. Commun. Earth Environ. 2021, 2, 241. [Google Scholar] [CrossRef]
- Byun, E.; Rezanezhad, F.; Slowinski, S.; Lam, C.; Bhusal, S.; Wright, S.; Quinton, W.L.; Webster, K.L.; Van Cappellen, P. Effects of Nitrogen and Phosphorus Amendments on CO2 and CH4 Production in Peat Soils of Scotty Creek, Northwest Territories: Potential Considerations for Wildfire and Permafrost Thaw Impacts on Peatland Carbon Exchanges. Soil 2025, 11, 309–321. [Google Scholar] [CrossRef]
- Escobar, D.; Manzoni, S.; Tapasco, J.; Vestin, P.; Belyazid, S. Evaluation of Long-Term Carbon Dynamics in a Drained Forested Peatland Using the ForSAFE-Peat Model. Biogeosciences 2025, 22, 2023–2047. [Google Scholar] [CrossRef]
- Sloan, T.J.; Ratcliffe, J.; Anderson, R.; Gehrels, W.R.; Gilbert, P.; Mauquoy, D.; Newton, A.J.; Payne, R.J.; Serafin, J.; Andersen, R. Potential for Large Losses of Carbon from Non-Native Conifer Plantations on Deep Peat over Decadal Timescales. Sci. Total Environ. 2024, 953, 175964. [Google Scholar] [CrossRef]
- Mirmanto, E. Vegetation Analyses of Sebangau Peat Swamp Forest, Central Kalimantan. Biodiversitas J. Biol. Divers. 2009, 11, 82–88. [Google Scholar] [CrossRef]
- Garsetiasih, R.; Heriyanto, N.M.; Adinugroho, W.C.; Gunawan, H.; Dharmawan, I.W.S.; Sawitri, R.; Yeny, I.; Mindawati, N.; Denny. Connectivity of Vegetation Diversity, Carbon Stock, and Peat Depth in Peatland Ecosystems. Glob. J. Environ. Sci. Manag. 2022, 8, 369. [Google Scholar] [CrossRef]
- Astiani, D.; Ekamawanti, H.A.; Ekyastuti, W.; Widiastuti, T.; Tavita, G.E.; Suntoro, M.A. Tree Species Distribution in Tropical Peatland Forest along Peat Depth Gradients: Baseline Notes for Peatland Restoration. Biodiversitas J. Biol. Divers. 2021, 22, 2571–2578. [Google Scholar] [CrossRef]
- Kettenring, K.M.; Tarsa, E.E. Need to Seed? Ecological, Genetic, and Evolutionary Keys to Seed-Based Wetland Restoration. Front. Environ. Sci. 2020, 8, 109. [Google Scholar] [CrossRef]
- Jia, X.; Wu, L.; Ren, J.; Peng, X.; Lv, H. Response of Carbon, Nitrogen, and Phosphorus in Leaves of Different Life Forms to Altitude and Soil Factors in Tianshan Wild Fruit Forest. Front. Ecol. Evol. 2024, 12, 1368185. [Google Scholar] [CrossRef]
- Marwanto, S.; Watanabe, T.; Iskandar, W.; Sabiham, S.; Funakawa, S. Effects of Seasonal Rainfall and Water Table Movement on the Soil Solution Composition of Tropical Peatland. Soil Sci. Plant Nutr. 2018, 64, 386–395. [Google Scholar] [CrossRef]
- Xing, S.; Cheng, X.; Kang, F.; Wang, J.; Yan, J.; Han, H. The Patterns of N/P/K Stoichiometry in the Quercus Wutaishanica Community among Different Life Forms and Organs and Their Responses to Environmental Factors in Northern China. Ecol. Indic. 2022, 137, 108783. [Google Scholar] [CrossRef]
- Güsewell, S. N:P Ratios in Terrestrial Plants: Variation and Functional Significance. New Phytol. 2004, 164, 243–266. [Google Scholar] [CrossRef] [PubMed]
- Mo, Q.; Zou, B.; Li, Y.; Chen, Y.; Zhang, W.; Mao, R.; Ding, Y.; Wang, J.; Lu, X.; Li, X.; et al. Response of Plant Nutrient Stoichiometry to Fertilization Varied with Plant Tissues in a Tropical Forest. Sci. Rep. 2015, 5, 14605. [Google Scholar] [CrossRef]
- Tian, D.; Yan, Z.; Niklas, K.J.; Han, W.; Kattge, J.; Reich, P.B.; Luo, Y.; Chen, Y.; Tang, Z.; Hu, H.; et al. Global Leaf Nitrogen and Phosphorus Stoichiometry and Their Scaling Exponent. Natl. Sci. Rev. 2018, 5, 728–739. [Google Scholar] [CrossRef]
- Castellanos, A.E.; Llano-Sotelo, J.M.; Machado-Encinas, L.I.; López-Piña, J.E.; Romo-Leon, J.R.; Sardans, J.; Peñuelas, J. Foliar C, N, and P Stoichiometry Characterize Successful Plant Ecological Strategies in the Sonoran Desert. Plant Ecol. 2018, 219, 775–788. [Google Scholar] [CrossRef]
- Torres-Rodríguez, J.V.; Salazar-Vidal, M.N.; Chávez Montes, R.A.; Massange-Sánchez, J.A.; Gillmor, C.S.; Sawers, R.J.H. Low Nitrogen Availability Inhibits the Phosphorus Starvation Response in Maize (Zea mays Ssp. Mays L.). BMC Plant Biol. 2021, 21, 259. [Google Scholar] [CrossRef] [PubMed]
- Sjögersten, S.; Cheesman, A.W.; Lopez, O.; Turner, B.L. Biogeochemical Processes along a Nutrient Gradient in a Tropical Ombrotrophic Peatland. Biogeochemistry 2011, 104, 147–163. [Google Scholar] [CrossRef]
- Choo, L.N.L.K.; Ahmed, O.H.; Razak, N.A.; Sekot, S. Improving Nitrogen Availability and Ananas Comosus L. Merr Var. Moris Productivity in a Tropical Peat Soil Using Clinoptilolite Zeolite. Agronomy 2022, 12, 2750. [Google Scholar] [CrossRef]
- Tanjung, R.H.R.; Suharno; Rumahorbo, B.T.; Reza, M.A.; Akhmad. Characteristics of Peatland Chemicals and Their Association with the Diversity of Dominant Plants in Papua. IOP Conf. Ser. Earth Environ. Sci. 2020, 575, 012082. [Google Scholar] [CrossRef]
- Negassa, W.; Michalik, D.; Klysubun, W.; Leinweber, P. Phosphorus Speciation in Long-Term Drained and Rewetted Peatlands of Northern Germany. Soil Syst. 2020, 4, 11. [Google Scholar] [CrossRef]

| Cluster | n | Organ C:N Ratio | |||
|---|---|---|---|---|---|
| Root | Stem | Twig | Leaf | ||
| 1 | 13 | 123.20 ± 50.87 a | 833.54 ± 196.58 a | 187.01 ± 74.16 a | 32.48 ± 11.82 |
| 2 | 61 | 98.26 ± 52.59 a | 393.23 ± 85.37 b | 138.92 ± 74.59 a | 30.85 ± 11.60 |
| 3 | 79 | 73.03 ± 45.18 b | 172.08 ± 71.59 c | 57.30 ± 28.61 b | 28.61 ± 14.62 |
| Cluster | n | Leaf Nutrient Ratio | ||
|---|---|---|---|---|
| N:P | N:K | K:P | ||
| 1 | 84 | 16.85 ± 3.74 a | 2.77 ± 1.70 a | 8.31 ± 4.80 a |
| 2 | 42 | 28.33 ± 3.39 b | 4.10 ± 1.92 b | 8.05 ± 2.80 a |
| 3 | 10 | 45.00 ± 3.99 c | 5.24 ± 2.21 b | 9.71 ± 3.28 a |
| 4 | 17 | 35.26 ± 7.16 d | 1.94 ± 0.66 a | 20.16 ± 7.93 d |
| Ecosystem Type & Location | Sample | Organ C:N Ratio | Study/Reference | |||
|---|---|---|---|---|---|---|
| Root | Stem | Twig | Leaf | |||
| Tropical peatland in South Sumatra Province, Indonesia | 153 native tree species | 87.20 ± 51.06 | 314.88 ± 210.8 | 116.60 ± 73.27 | 29.66 ± 13 | This study |
| Semi-deciduous rain forest Maringa-Lopori-Wamba forest, Tshuapa province, DR Congo | Trees from five different succession stages | - | - | - | Lowest: 18.0 Highest: 19.5 ± 0.7 | Bauters et al. [42] |
| Seasonally dry tropical forest Açu National Forest, Rio Grande do Norte, Brazil | 20 native tree species | - | - | - | 22.91± 5.769 | Fagundes et al. [43] |
| 28 natural climax forests in nature reserves, China | 647 trees | 62.85 ± 1.20 | 321.36 ± 6.85 | 65.07 ± 0.76 | 24.28 ± 0.26 | Zhang et al. [37] |
| 9 typical forest ecosystems (cold-temperate to tropical zones), The north–south transect of eastern China | 803 tree, shrub, and herb species | 50.6 ± 2.6 | 369.9 ± 38.5 | 60.1 ± 1.4 | 23.6 ± 0.7 | Zhang et al. [38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ismail, M.S.; Sulistijorini, S.; Muttaqin, M.; Al Anshori, Z.; Rizaldi, M.R.; Wijedasa, L.; Moore, J.; Agusti, R.; Swarup, S.; Triadiati, T. Elemental Stoichiometry of Tropical Peatland Trees: Implications for Adaptation and Carbon Sequestration. Forests 2025, 16, 1379. https://doi.org/10.3390/f16091379
Ismail MS, Sulistijorini S, Muttaqin M, Al Anshori Z, Rizaldi MR, Wijedasa L, Moore J, Agusti R, Swarup S, Triadiati T. Elemental Stoichiometry of Tropical Peatland Trees: Implications for Adaptation and Carbon Sequestration. Forests. 2025; 16(9):1379. https://doi.org/10.3390/f16091379
Chicago/Turabian StyleIsmail, Moh Syukron, Sulistijorini Sulistijorini, Mafrikhul Muttaqin, Zakaria Al Anshori, Muhammad Rifki Rizaldi, Lahiru Wijedasa, Jared Moore, Randi Agusti, Sanjay Swarup, and Triadiati Triadiati. 2025. "Elemental Stoichiometry of Tropical Peatland Trees: Implications for Adaptation and Carbon Sequestration" Forests 16, no. 9: 1379. https://doi.org/10.3390/f16091379
APA StyleIsmail, M. S., Sulistijorini, S., Muttaqin, M., Al Anshori, Z., Rizaldi, M. R., Wijedasa, L., Moore, J., Agusti, R., Swarup, S., & Triadiati, T. (2025). Elemental Stoichiometry of Tropical Peatland Trees: Implications for Adaptation and Carbon Sequestration. Forests, 16(9), 1379. https://doi.org/10.3390/f16091379






