Abstract
Abiotic stresses could have complex and diverse effects on the growth and development of forest trees. In this review, we summarized the responses of morphological, physiological and anatomical traits in woody plants to abiotic stresses, including drought, flood, extreme temperature, salinity, heavy metal, microplastics and combined stresses, especially from the xylem perspective. Under most abiotic stress, xylem hydraulic conductivity decreases, which is associated with leaf stomatal regulation and the inhibition of aquaporin (AQP) activity. Meanwhile, woody plants regulate the size and morphology of their roots and leaves to balance water absorption and transpiration. The anatomical traits are also altered, such as denser leaf stomata, narrower conduits and thicker cell walls. In addition, different stresses have unique effects, such as flood-induced adventitious roots and aeration tissues, forest fire-induced irreversible xylem damage, low temperature-induced tissue freezing, salt stress-induced hinderance of ion absorption and heavy metal-induced biological toxicity. Under stresses of drought, flooding and heavy metals, woody plants’ growth may occasionally be promoted. The effects of combined stress on the physiological, morphological and anatomical traits of woody plants are not simply additive, with the related mechanism to be further studied, especially in natural or near-natural conditions.
1. Introduction
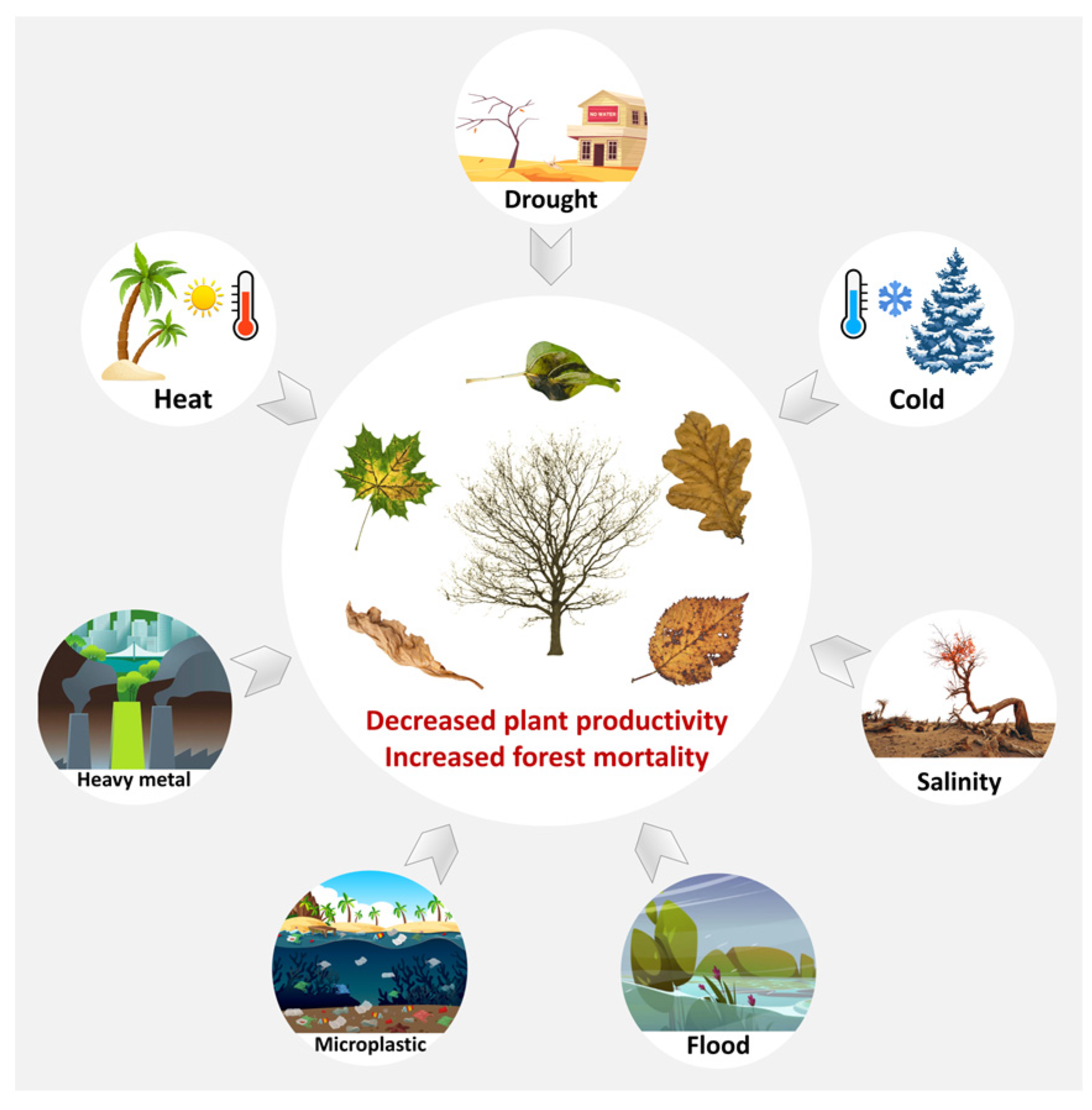
Forest trees provide great economic and ecological values for human beings. However, with increasing abiotic stresses such as drought, flood, extreme temperature, salinity and heavy metals, forest tree growth and development is greatly challenged [1] (Figure 1). For instance, the frequency and intensity of extreme temperature has been on the rise, leading to more severe drought conditions and large-scale tree mortality [2]. Extreme rainfall and the resulting floods are becoming more frequent, which is one of the main factors reducing tree productivity [3]. Global warming may increase the frequency and severity of heatwaves and the risk of wildfires, reducing tree survival and productivity [4,5]. Extreme cold events lead to cell membrane damage, cell dehydration and tissue freezing of trees, especially for tropical and subtropical tree species that are not cold tolerant [6]. Salinization damages soil fertility and causes osmotic stress and ion toxicity in trees [7]. In addition, mineral extraction, overuse of fertilizers and pesticides, and emissions from industrial activities all contribute to heavy metal contamination of the soil [8], which has persistent toxicity in woody plants, hindering their nutrient absorption, causing developmental defects, abnormal growth and even death [9].
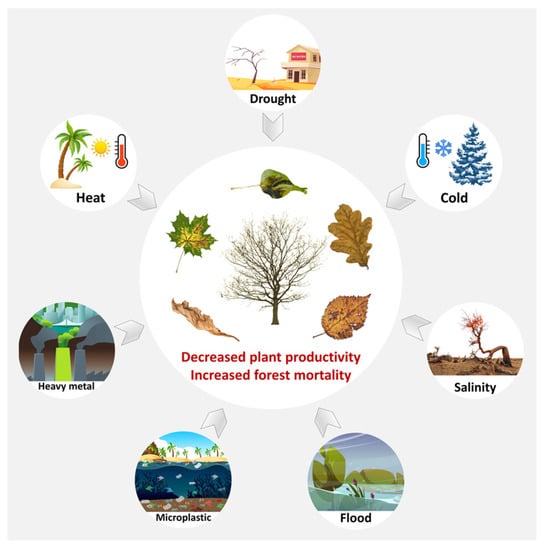
Figure 1.
Under different abiotic stresses, forest trees may show decreased productivity and increased mortality.
Abiotic stresses usually result in changes in water availability in the environment and impact the water transport function of woody plants. Water in woody plants is transported through the xylem conduit by a negative pressure gradient generated by leaf transpiration [10]. Leaf stomatal regulation plays a critical role under abiotic stress conditions, as it determines the balance between water loss (transpiration) and carbon gain (photosynthesis) [11]. In addition, woody plants have developed complex signaling networks to sense different environmental stresses and plant hormone regulation plays an essential role in plants’ responses to abiotic stresses [12]. For instance, abscisic acid (ABA) promotes stomatal closure under abiotic stress, and its interaction with other hormones enables plants to develop complex and efficient stomatal regulation mechanisms [13].
As the stress continues, woody plants adjust their morphological and anatomical traits. For example, as soil water availability decreases, they increase the length, density and depth of the roots to absorb water and nutrients from the soil more efficiently [14]. Leaf morphology also plays an important role in woody plants’ water transport and response to abiotic stress. For example, a thicker cuticle and epidermis lead to lower water permeability, which can help plants adapt to different water conditions [15]. In addition, woody plants can change the size, number and thickness of their leaves to adapt to environments with different moisture, temperature and nutrients [16,17,18]. Woody plants can also adjust their xylem anatomical structure to regulate hydraulic function in response to abiotic stresses. For instance, they can construct narrower conduits with thicker walls to enhance the water transport safety [19].
Xylem, the tissue responsible for long-distance water transport from roots to leaves, is a complex multicellular network consisting of tracheary elements, parenchyma cells, fibers and other cell types [10,20]. These cells play roles in solute transport, nutrient storage and mechanical support. The xylem conducting tissue in gymnosperm species consists primarily of tracheids, while that in angiosperm species consists mainly of vessels [21]. Tracheids and vessel elements are elongated and inanimate cells with lignified secondary walls. Water moves between neighboring conduits through pit pairs formed by uneven thickening of the secondary walls [22]. Vessels are more efficient in water transport than tracheids as they are wider and interconnected with perforation plates, facilitating water transport. Xylem parenchyma cells, including axial and ray parenchyma cells, are responsible for storing and transporting nutrients and water [23].
In this paper, we summarized previous work on the responses of physiological, morphological and anatomical traits of woody plants to various environmental stresses by a literature investigation. Specifically, we explored the impact of some stresses on xylem hydraulic traits and the associated changes in plant water transport. In addition, we presented our recent work on the effects of heavy metals and the combined stress of drought and salt on the physiological and anatomical traits of woody plants.
2. Response of Woody Plants to a Single Abiotic Stress
2.1. Drought
In forest ecosystems, tree mortality and drought were positively correlated [24] and forest ecosystems with low tree population densities have higher drought resistance and resilience [25]. Functional traits explain the varied responses of trees to drought stress and are valuable for understanding drought-induced tree mortality patterns [24]. Xylem is the main channel for long-distance water transport in trees, in which the xylem sap ascent is driven in a metastable state and could be interrupted by air bubbles in the xylem network, i.e., embolism occurs [10,26,27]. Under drought conditions, external air bubbles from embolized conduits could spread between neighboring conduits via pits, which could break the continuous water column, resulting in prohibited water transport, xylem hydraulic failure and eventually tree mortality [26,28].
2.1.1. Physiological Responses of Plants under Drought
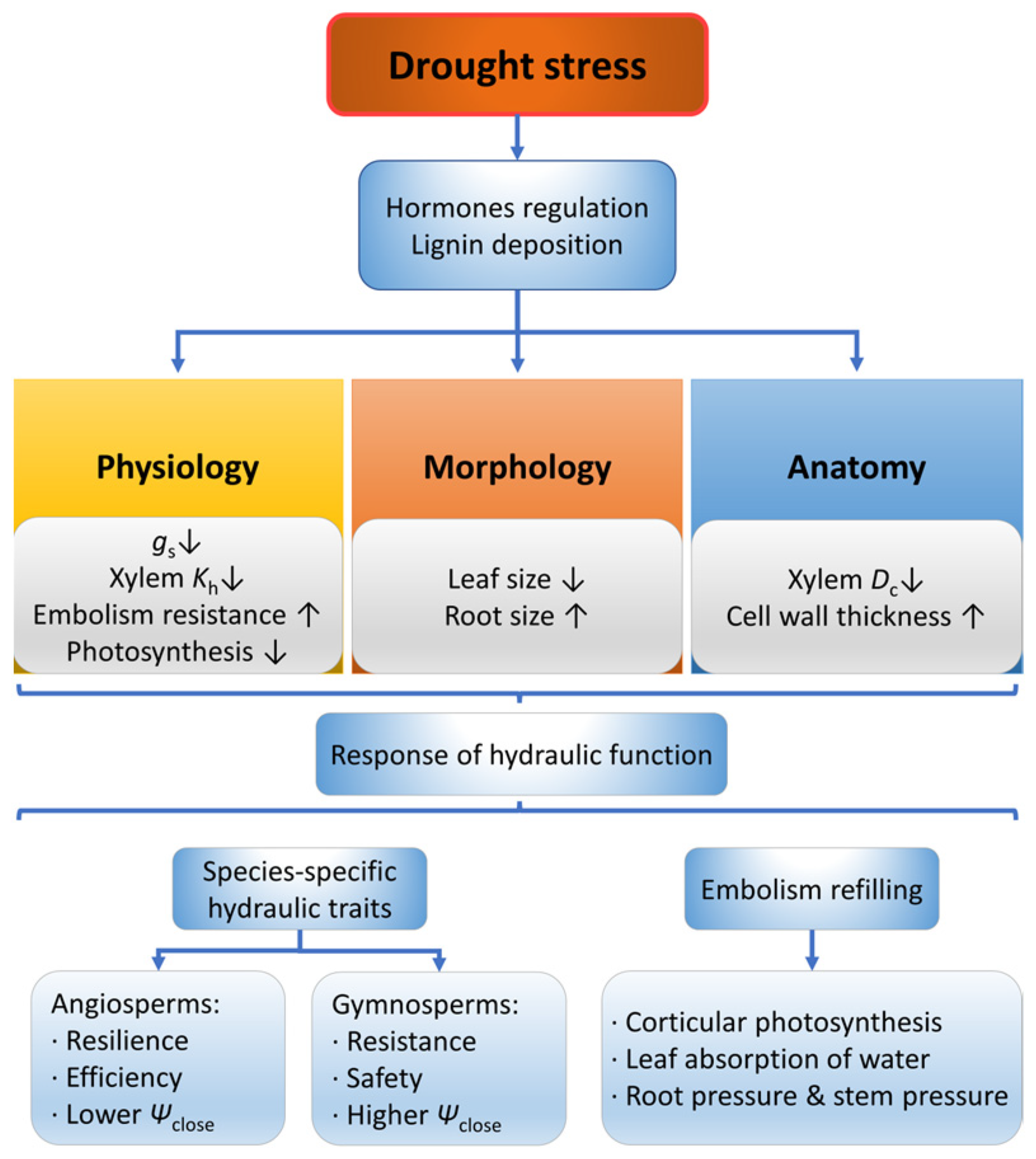
Woody plants exhibit responses to drought stress from multiple perspectives (Figure 2), among which their physiological characteristics rapidly respond to drought stress within a short term [29]. Specifically, leaf physiological characteristics such as xylem water potential, stomatal conductance [30] and transpiration rate may decrease under drought conditions [31]. In addition, photosynthesis is inhibited as the amount and activity of related enzymes is decreased [32]. As the aggravation of drought level increases, the percentage loss of xylem hydraulic conductivity (PLC) of the xylem also increases [33,34], and the decrease of xylem hydraulic conductivity may occur in different plant organs or tissues under different drought stress levels. Specifically, under mild drought stress, the hydraulic decline in the leaves is mainly due to the decreased hydraulic conductivity of outside-xylem tissue, while only under severe drought would the substantial xylem embolism occur in the xylem of leaves [35]. However, under mild drought stress, the resistance to water transport of riparian trees was mainly from the twig xylem, while under severe drought, the resistance was primarily from the root xylem [36].
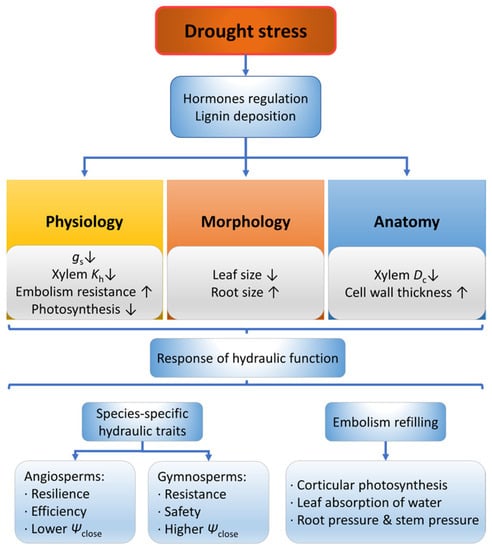
Figure 2.
Responses of plants in morphology, anatomy and physiology under drought stress. Dc, conduit diameter; Kh, hydraulic conductivity; gs, stomatal conductance; Ψclose, water potential at stomatal closure. The up arrow indicates an increase, and the down arrow indicates a decrease.
Gymnosperms and angiosperms demonstrate different response patterns between resistance (sensitivity to drought) and resilience (recovery after drought) [37]. Gymnosperms have stronger drought resistance and a wider hydraulic safety margin [37,38,39] and tend to close stomata early to reduce transpiration [40,41], demonstrating a much safer hydraulic function. In contrast, angiosperms have stronger drought recovery rate after rewatering [37,38] and tend to maintain leaf transpiration and photosynthesis to assimilate carbon [37,40], hence a more efficient hydraulic function. Angiosperms do not show irreversible damage before 88% to 98.6% loss of stem conductivity [34,42], while gymnosperms’ lethal threshold is between 50% and 80% loss of hydraulic conductivity in most species reported [34,43,44].
Leaf stomatal regulation is an effective mechanism for trees to cope with drought stress and avoid hydraulic failure. The stomatal regulation of trees under drought showed a wide continuum of isohydric and anisohydric behavior at the inter- and intra-specific levels [45,46]. Isohydric species dramatically decrease their stomatal conductance within a small range of leaf water potential under drought to prevent further xylem embolism, while anisohydric species try to keep their stomata open at a lower water potential to maintain the photosynthetic rate [47,48,49]. The involvement of ABA is essential for the strategy of stomatal regulation under drought stress. On the one hand, the expression of ABA synthesis-controlling enzymes in leaves increases, causing the increase of ABA content in leaves and the release of anions and K+ by guard cells, resulting in decreased turgor of guard cells and stomatal closure [50]. On the other hand, the water stress induces ABA to interact with phytohormones such as jasmonic acid and ethylene to stimulate stomatal closure, which inhibits CO2 assimilation and transpiration [51].
2.1.2. Morphological and Anatomical Responses of Plants under Drought
Under long-term drought stress, in addition to the physiological changes, a series of morphological and anatomical changes would occur in woody plants, such as small leaves [52,53], deep and large roots [54,55] and a decreased increment of tree height and basal area [56,57]. Narrower conduits with thickened cell walls are usually constructed under drought stress [33,58,59], which show lower hydraulic efficiency [60] but higher embolism resistance [61,62]. The increase in drought severity is significantly linked to the decrease in conduit diameter and the thickening of the conduit cell wall [63,64,65] and even leads to conduit deformation [66]. However, Pinus edulis was reported to construct low-cost, more efficient, but less safe xylem structures under drought stress [67]. In addition, changes in conduit density and wood density under drought stress are species specific [68,69,70], trees with high wood density had higher survival rates during past drought events [24,71], demonstrating that higher wood density could be a positive response under drought conditions. Under drought stress, the leaf anatomy of trees is also altered. With the increase of drought severity, the thickness of palisade tissue and leaf tissue structural tightness increase and the intercellular air space decreases [72,73]. Moreover, the mesophyll cells may be deformed under severe drought [74].
Hormone regulation is one of the mechanisms that causes morphological and anatomical changes in woody plants [75,76]. For example, under drought stress, ABA content in Populus roots increases, promoting the indoleacetic acid transport in root tips and increasing root growth [55]. In addition, the decrease of auxin content in the xylem under drought may reduce meristems activity and inhibit the expansion of tracheary elements, thus reducing the tree’s conduit diameter [62,77,78].
Enzymes and phenolic compounds produced under drought could also significantly affect cell wall structure and the xylem structure. For instance, enzymes such as phenylalanine ammonia-lyase and caffeoyl-CoA 3-O-methyltransferase play a vital role in the synthesis of lignin, and the related genes can be up-regulated under drought stress [79]. The deposition of lignin may lead to the thickening of the secondary wall, which increases the strength of the cell wall and prevents it from collapsing under drought [22,80,81]. In addition, under drought stress, phenolic compounds can bind to cell walls to make them hydrophobic, hard and compact, which allows cells to maintain turgor at low water potentials and prevents water loss [82,83,84].
2.1.3. Xylem Hydraulic Refilling
After experiencing drought–rewatering cycles, trees can rapidly recover their water potential within 3 days, while leaf gas exchange may take several weeks to recover fully [85]. ABA has been implicated as a key factor causing delayed stomatal opening after rehydration [86], and recent studies have also indicated that a decrease in ethylene emission rate can result in slow stomatal opening [87]. After rewatering, xylem embolism refilling occurs in some species, while some species do not show embolism refilling [88]. For example, xylem embolism refilling was found in the isolated branches of Salix matsudan through corticular photosynthesis and bark water uptake [89]. However, neither F. ornus nor Olea europaea can restore xylem embolism by water absorption of bark, indicating that the mechanism of embolism repair may be species specific [90]. In addition, the excised twigs of Avicennia marina showed xylem embolism refilling by leaf absorption of atmospheric water [91].
Positive xylem pressure including root and stem pressure could result in the xylem embolism refilling. The development of root pressure may be attributed to the active transport of salts into the xylem conducting system, leading to more negative osmotic potential within the xylem, ultimately resulting in the entry of water into the xylem vessels [92]. However, the embolism refilling capacity generated by root pressure is limited. Not all tree species are capable of generating root pressure and root pressure can only effectively refill embolized vessels within a few meters above the root system [93]. Alternatively, stem pressure, which is generated by the osmotic gradient formed by the release of soluble sugars from xylem parenchyma cells into the vessels, shows a lower refilling efficiency than root pressure [26]. For instance, the deep roots of Juglans regia have a stronger embolism refilling ability than branches, as the former mainly depends on root pressure to refill the embolized vessels, while the latter mainly depends on stem pressure produced by non-structural carbohydrates (NSCs) [94]. The involvement of the parenchyma, which are responsible for transporting salts, NSCs and water into the xylem, may play a vital role in generating root and stem pressure [95,96]. For instance, Carpinus betulus, a species with root pressure has abundant xylem parenchyma cells in its root xylem [97] The species that have been observed to show embolism refilling are mainly angiosperms with a greater proportion of parenchyma [88]. Moreover, species grown in areas with low precipitation tend to have a higher proportion of parenchyma to prevent xylem embolism formation or facilitate embolism refilling [23], demonstrating a crucial response to drought.
Although a significant number of species have been shown to perform xylem embolism refilling by a destructive hydraulic method [88], at present, xylem embolism refilling has only been observed in a few species by non-destructive Micro-CT technology, such as Populus tremula × Populus alba, F. ornus, Vitis vinifera and Zea mays [98,99,100]. In contrast, no embolism refillig has been reported in some other tree species using the same technique, such as Eucalyptus calmudulensis, E. grandis, E. saligna and Quercus palustris [101]. The shortage of Micro-CT is low image resolution, which makes it difficult to differentiate between water droplets and tyloses. In addition, even when droplets are present in embolized vessels, the water column within may still be discontinuous, leading to the potential overestimation of embolism refilling levels based solely on cross-sectional Micro-CT images [101]. More studies based on non-destructive techniques are needed to confirm whether embolism refilling can be performed in plants of different life forms, and the mechanisms of embolism refilling need to be further elucidated.
2.2. Flood
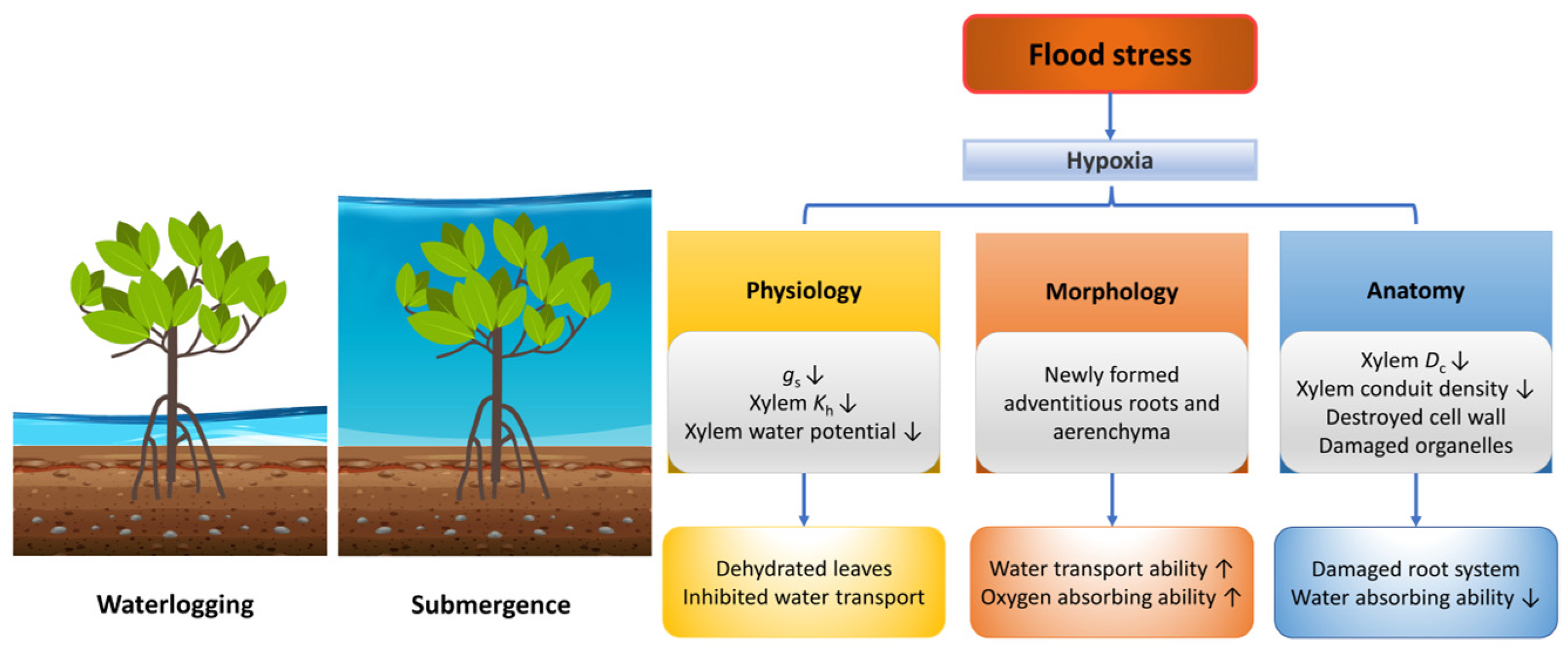
Woody plants grown near lakes, coastal or riparian zones often experience flood stress, which includes waterlogging and submergence depending on water levels (Figure 3). Flood stress reduces forest productivity, species richness, stand density, above-ground biomass, tree height and tree basal area [102,103,104].
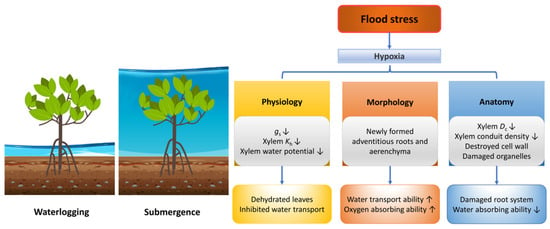
Figure 3.
Responses of plants in morphology, anatomy and physiology under flood stress. Dc, conduit diameter; gs, stomatal conductance; Kh, hydraulic conductivity. The up arrow indicates an increase, and the down arrow indicates a decrease.
2.2.1. Physiological Responses of Plants under Flood Stress
Flooding puts a severe selective pressure on woody plants, such as the shortage of oxygen and carbon dioxide [105], hence inhibiting their respiration and photosynthetic abilities, leading to inhibited plant growth, especially for flood-intolerant species [106,107,108,109]. Flooding-induced hypoxia leads to leaf physiological changes such as leaf stomatal closure and decreased transpiration rate, which may be related to the decline of photosynthetic assimilates transportation ability in the root phloem [110]. Specifically, leaf ABA concentration increases with short-term flooding [111], initiating H2O2 production by the plasma membrane NADPH oxidase [112], which activates Ca2+ channels and leads to the increase of Ca2+ level in guard cells, thus inhibiting inward K+ channels and leading to leaf stomatal closure [113]. Meanwhile, flood stress destroys root function and makes it difficult for trees to absorb water, leading to the dehydration of tree leaves [114]; therefore, plants close their stomata quickly to avoid leaf wilting [115]. In Pisum sativum subjected to flood stress, ABA is transported from old leaves to new leaves to protect young leaves [116].
During flood stress, reduced xylem water potential has also been reported [114,117]. In addition, flood stress inhibits the xylem hydraulic function of woody plants. For example, the xylem sap flow of mangrove was reduced due to early spring flooding [118], and the PLCs of Ulmus laevis and U. minor were four times higher than that of the controlled group after 46 days of flood stress [107]. Flood stress reduces the pH value of root sap in Carrizo citrange, thus inhibiting the activity of aquaporins (AQPs) and decreasing xylem hydraulic conductivity [119,120].
2.2.2. Morphological and Anatomical Responses of Plants under Flood Stress
Trees respond to flooding stress through morphological and anatomical changes [121]. Morphologically, impaired photosynthesis usually results in the suppression of plant growth. For example, root and leaf growth of two Quercus species decreased after 90 days of flooding stress and lateral roots could only initiate and elongate in the soil surface layer [122]. The height of S. viminalis increased under flood stress [123], while bud height and basal diameter of Ormosia arborea were both negatively affected [124], revealing species-specific effects of flooding on plant morphology. In addition, flooding increases ethylene biosynthesis, leading to lower ABA levels and increased sensitivity to GA, ultimately promoting plant growth [125]. For example, stem elongation of mangroves was facilitated and the biomass of A. marina was promoted under flood stress [126]. The biomass of flood-tolerant species Senna reticulata under flood stress was higher than that under non-flood conditions and its root–shoot ratio also increased [127].
Induced adventitious roots or aerenchyma by ethylene and auxin signals could maintain woody plants’ aerobic respiration and water absorption [128,129,130]. For instance, Larix laricina, a flooding-tolerant tree species, could produce adventitious roots with a high xylem hydraulic conductivity under flooding conditions, with almost the same stomatal conductance, net photosynthetic rate, and aboveground water potential as the unflooded samples [131]. Adventitious roots can transport more oxygen and express AQPs more stably than normal roots, which may reduce the effects of flood stress on trees [108,132]. For instance, flooding inhibited the root water absorption of Campsiandra laurifolia in the initial stage; however, as the acclimation process continued, the aeration of adventitious roots was improved and their hydraulic conductivity gradually recovered [106].
Anatomically, wetland plants usually have distinct sclerenchyma, which protects the root system’s structural integrity [133,134]. Flooding could also alter the xylem structure, such as decreasing the xylem conduit diameter and conduit density of trees [121,135]. The adventitious roots produced by L. laricina under flood stress have fewer secondary tissues, poorly developed endodermis and small-diameter tracheids than control roots, and the cortex of flooded adventitious roots was filled with more starch grains than control roots, which eventually increased its flood tolerance [131]. Although woody plants have evolved various strategies to adapt to flood stress, ultrastructural damage to them may also occur after long-term flooding. After 15 days of waterlogging stress, not only the palisade cells of poplar leaves were deformed, but the parenchyma cells of roots were also dissolved, and the organelles were destroyed, resulting in root dysfunction [136]. Flooding may also influence the structure of plant cell walls, inducing the degradation of cell wall polysaccharides, decreased pectin content, reduced lignification and cell wall thinning [84].
2.3. Extreme Temperature
2.3.1. Heat
Heat stress, often combined with drought, is one of the main triggers of forest mortality due to climate change [137]. High temperature stress can lead to the migration of forests to higher elevations, decrease forest water storage capacity, reduce tree basal areas and increase the risk of forest fires [138,139,140,141]. Forest fires can lead to a decrease in overall forest canopy height, a decrease in tree biomass, a decrease in forest basal area and can have persistent effects on forest health and structure [142,143].
Physiological Responses of Plants under Heat Stress
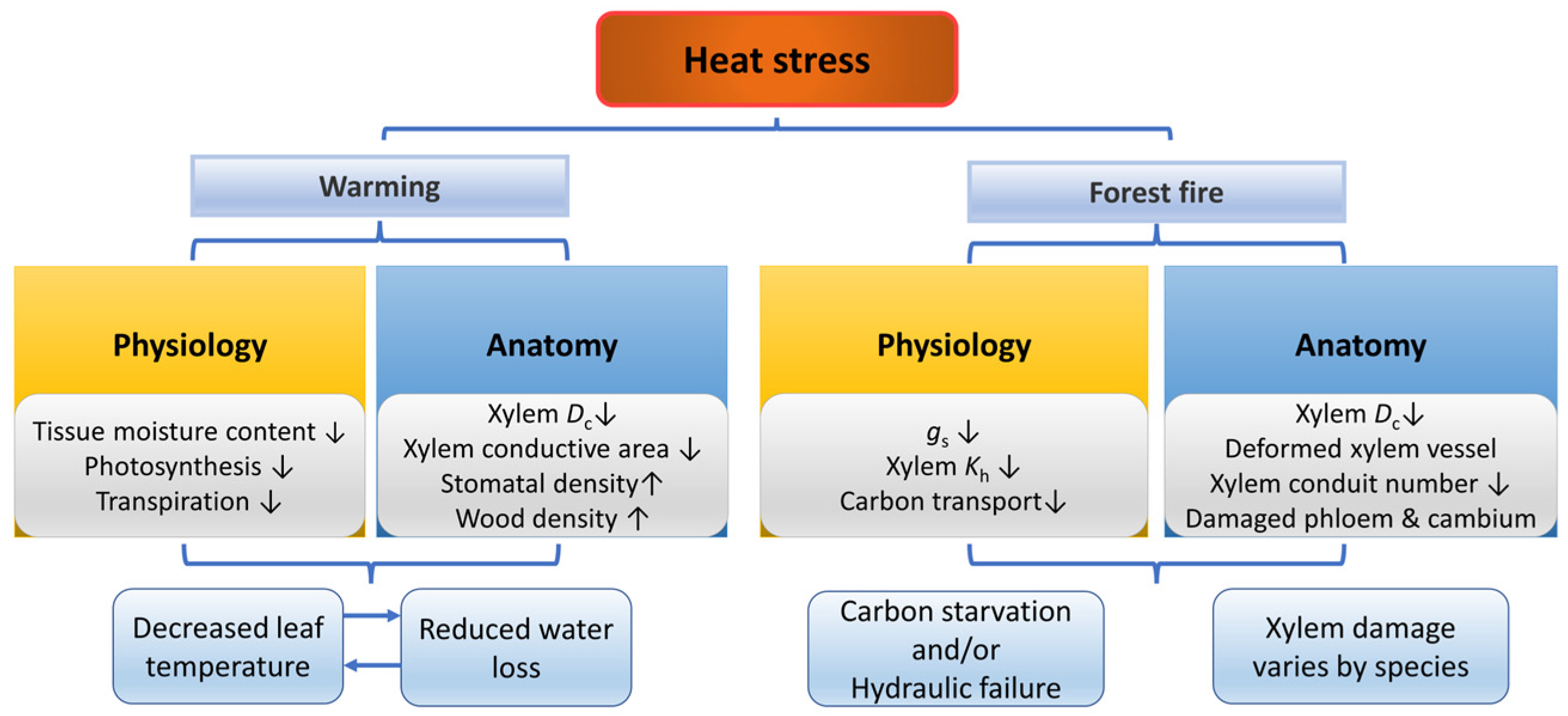
Trees show the highest growth and development rate under the optimum temperature and extreme temperatures may inhibit trees’ growth and development [144] (Figure 4). High temperatures can lead to tissue dehydration and an imbalance between photosynthesis and respiration. Once the leaf temperature exceeds the maximum temperature for growth, the physiological activities of trees decline due to the inactivation of enzymes [144]. Increasing stomatal conductance is a key mechanism to lower leaf temperature and restore optimal photosynthesis, but it also enhances transpiration and increases the sensitivity to water deficit, leading to more severe functional damage and delayed hydraulic recovery processes [145,146,147]. However, tree species with conservative water-use strategies, such as Pseudotsuga menziesii, can reduce this effect [148]. Therefore, the decline of leaf water potential and hydraulic conductivity is usually observed under high-temperature stress [146,149]. Moreover, tree leaves show increased thermo-tolerance during and after heatwaves [150].
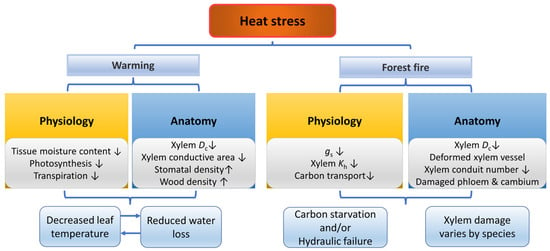
Figure 4.
Responses of plants in physiology and anatomy under heat stress and forest fire. The up arrow indicates an increase, and the down arrow indicates a decrease.
Forest fires are significant disturbances that forest trees experience in natural environments. Even if the tissue damage caused by fires does not immediately kill the trees, it can impair their carbon and water relations [151]. The mechanisms underlying trees’ impaired function and potential mortality after forest fires may involve carbon starvation and hydraulic failure [152]. On the one hand, fire damage restricts the transfer of carbon to roots, therefore inhibiting the physiological functions of roots and finally leading to carbon starvation [151,153]. On the other hand, heating destroys the xylem water transport function, leading to decreased xylem water potential and hydraulic conductivity, xylem embolism, stomatal closure, inhibition of photosynthesis and growth and finally resulting in hydraulic failure [152,154].
Morphological and Anatomical Responses of Plants under Heat Stress
Heat stress can lead to reduced tree biomass, reduced leaf area, leaf abscission and the negative effects of heatwaves on growth are more severe than increased temperature [155,156,157]. Trees increase the leaf thickness to improve heat tolerance or build reflective leaves, as well as change leaf angles to reduce heat radiation loads [158,159]. The growth rate of fine roots also decreases under heat stress [160]. In addition, the xylem conductive area of Q. pubescens and Fagus sylvatica was significantly reduced, even when soil moisture was adequate [129]. As the temperature rises, water viscosity decreases, resulting in reduced conduit lumen area and increased wood density in Eucalyptus camaldulensis cultured at different temperatures for several weeks [161]. In addition, under high-temperature, both the tracheid lumen diameter, cell wall thickness and the wood density of Picea mariana remained stable in the central portion of the tree rings, rather than changing gradually from earlywood to latewood as in the control plants [70].
Fire can induce phloem and cambium necrosis and damage the xylem structure, such as deformed xylem conduits due to softening of hemicellulose and lignin in the cell wall. For instance, Nothofagus pumilio showed a decrease in the vessel number and vessel lumen diameter near the burned wounds after exposure to fire [162]. In contrast, fire-damaged Q. pubescens showed increased xylem radial growth compared to those that were undamaged [154]. Surprisingly, even though the 100% phloem and cambium of Pinus ponderosa were destroyed by fire, the function of xylem water transport did not change and xylem deformation was not observed [153].
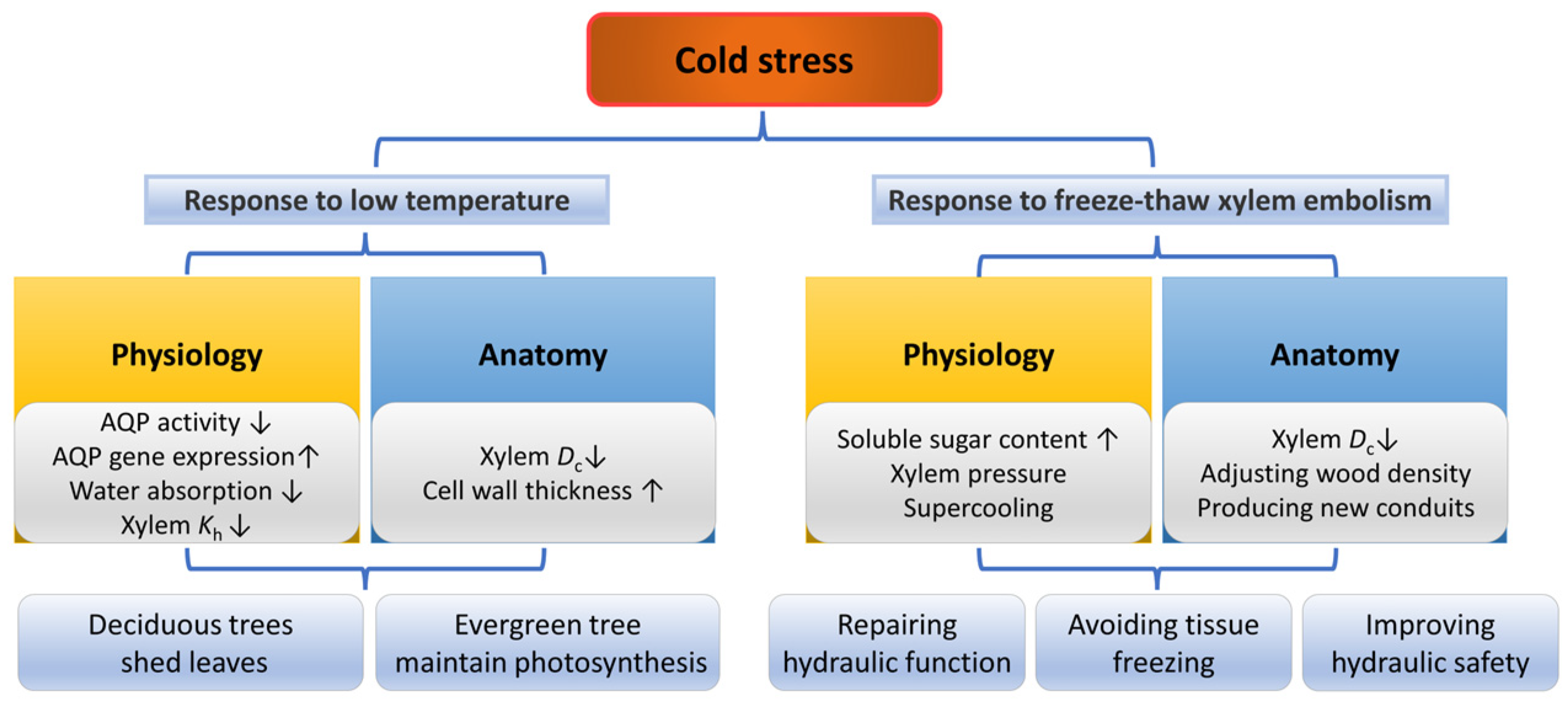
2.3.2. Cold
The low temperature in the winter is an important abiotic stress for high-altitude, high-latitude boreal and temperate forests [163,164]. Freezes and ice storms can result in decreased leaf area index, stand density and tree basal areas, and ultimately reduce the forest productivity [165,166,167]. The sensitivity of different tree species to cold stress varies, therefore, repeated freezes may alter the forest community structure [167,168].
Physiological Responses of Plants under Cold Stress
Cold stress can directly inhibit metabolic responses by inhibiting the activity of AQPs in plant roots, thereby reducing the woody plant’s ability to absorb water, lowering hydraulic conductivity and causing cell dehydration [169,170,171]. Under cold but not below-freezing temperatures, some cold-tolerant species could recover hydraulic conductivity gradually over time, which may be related to the upregulation of AQP gene expression and the increased contribution of AQPs [172]. Moreover, evergreen trees close their stomata in winter to reduce water loss, as low temperatures may cause soil freezing and water supply difficulties [63].
When the temperature is reduced to the freezing point of the xylem sap, freeze–thaw cycles in the xylem sap could induce an embolism [173,174] and seriously affect the xylem water transport [175,176,177]. When the water in the xylem freezes, gases that do not dissolve in ice form bubbles in the conduits [178]. After thawing, some gas bubbles dissolve again in the water [179], but some expand [151]. After repeated freeze–thaw cycles, these gases in the conduits collide several times and eventually trigger xylem embolism [180]. Conduit diameter was considered to be an important anatomical trait affecting vulnerability to freeze–thaw embolism, as larger bubbles tended to form in a wide conduit [181]. However, recent studies using non-invasive techniques do not seem to support this hypothesis [182,183].
Trees can resist or avoid freeze –thaw embolisms by physiological responses (Figure 5). Some trees can produce root and stem pressure to refill freeze–thaw-induced xylem embolism [184,185,186], which is produced from the soluble sugars from the phloem [89,187] to prevent tissue freezing [188]. In addition, some trees deal with cold stress by supercooling, a strategy preventing tissue freezing by lowering the freezing point of the cellular liquid below the ambient temperature [189,190]. Moreover, closing the stomata at night may reduce the possibility of ice nucleating on the leaf surface and entering the plant [191]. In contrast, Olea europaea increased its stomatal conductance in winter, allowing xylem embolism and continuous water loss to occur, which decreases its ice nucleation temperature and in turn increased its supercooling capacity, enabling it to avoid further damage caused by freezing [192]. In addition, cells with highly viscous contents due to severe dehydration will “vitrify” the remaining water at low temperatures instead of freezing, which may also increase the ability of the trees to survive in severe cold conditions [191,193].
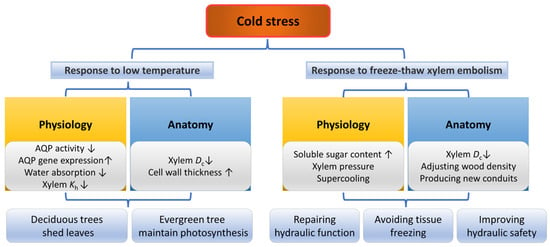
Figure 5.
Responses of plants in physiology and anatomy under cold stress and freeze–thaw induced xylem embolism. The up arrow indicates an increase, and the down arrow indicates a decrease.
The hydraulic function of trees during the subsequent growing season is affected after repeated freezing–thaw-induced xylem embolism events, referred to as “frost fatigue” [194]. For instance, the resistance of xylem embolism fatigue in Acer mono was the highest in winter and the lowest in summer, which may be due to changes in the mechanical properties of the pit membrane [177]. However, freeze–thaw fatigue was not observed in Picea abies, proving that the presence or absence of frost fatigue may be species specific [195]. Therefore, freeze–thaw fatigue may be species dependent and its mechanism remains further investigated.
Morphological and Anatomical Responses of Plants under Cold Stress
In winter, deciduous tree species senesce their leaves and redistribute the stored nutrients in the leaves to other organs (roots or reproductive tissues) to escape the damage of low-temperature stress on the leaves [196]. This process may be associated with the massive accumulation of ABA and jasmonate (JA) [197]. Cold stress also affects xylem anatomy. For example, the xylem secondary cell walls of E. gundal were thickened by lignification under low temperatures [198], and the tracheid lumen diameter of Pinus. pinaster was decreased while its tracheid wall thickness was increased [199]. The vessel hydraulic diameter and relative vessel lumen area of F. sylvatica decreased with decreasing temperature [170]. Interestingly, species with wider vessels may have a later defoliation time, which may be related to the developmental and physiological links between leaves and wood or a larger safety efficiency trade-off [200].
Adjusting the anatomical structure is also an effective way for trees to resist freeze–thaw embolism. For angiosperms, the construction of narrower vessels is a direct way to deal with cold stress and freeze–thaw-induced xylem embolism [62,201], and narrower vessels were associated with higher freeze–thaw embolism resistance [202]. Therefore, diffuse-porous species have a higher xylem embolism resistance than ring-porous species due to relatively narrower vessels [203,204]. Gymnosperms with generally narrower conduits are considered to have higher embolism resistance, although there is no difference in embolism resistance between gymnosperms and angiosperms with the same conduit diameter [63]. However, for species that can produce root and stem pressure, the resistance to freeze–thaw-induced xylem embolism may be independent of conduit diameter [175]. Interestingly, the lower the temperature, the lower the wood density of gymnosperms, while angiosperms show the opposite [205]. This may be due to the fact that gymnosperms have higher resilience to freeze–thaw embolism and competitive pressures in winter result in higher growth rates [205]. In addition, under low temperatures, the embolized vessels of some angiosperms (especially ring-porous species) may become dysfunctional and new conduits formed in the following spring could restore water transport function [174,206,207].
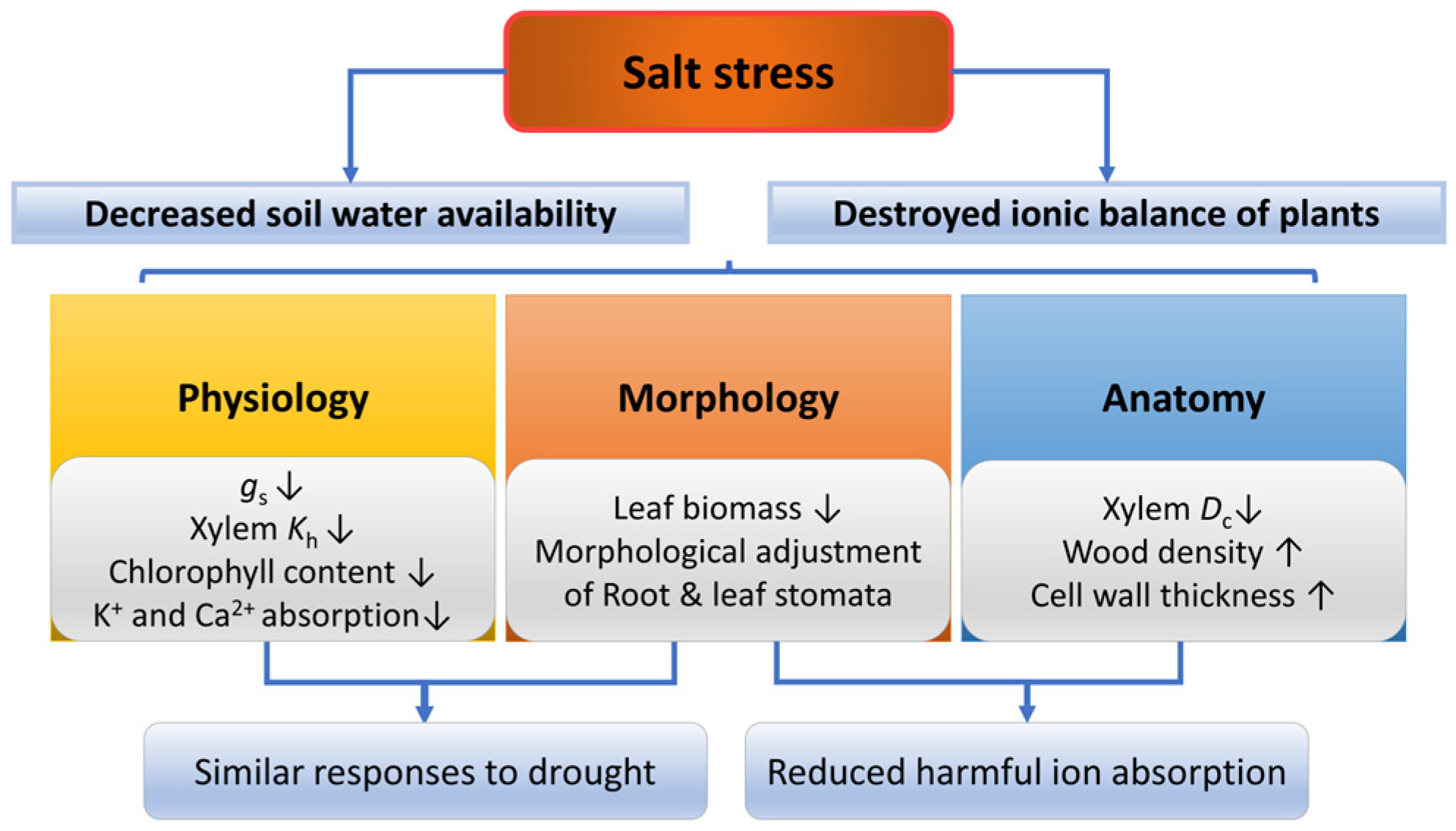
2.4. Salinity
Increased salinity leads to changes in forest structure, reducing tree height, basal area, leaf area index and creating less productive ecosystems dominated by dwarf species [208,209]. Under salt stress, woody plants regulate physiological traits in the short term and morphological as well as anatomical traits in the long term [210]. There are two factors by which salinity limits plant growth (Figure 6), the first is reduced water availability caused by a decrease in soil osmotic potential, and the second is destroyed ionic balance caused by the accumulation of the Na+ and Cl− [211]. Low soil water availability associated with high salinity causes trees to exhibit drought-like symptoms, as mentioned previously. Specifically, salt stress can destroy ion balance in plant cells, thus inducing osmotic stress, and the stress will be aggravated when the pH is high [212]. High concentrations of ions, especially Na+ and Cl−, may be toxic to plants and hinder the absorption of other ions (predominantly Ca2+) [213].
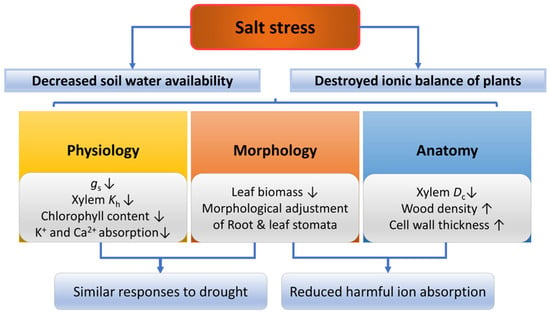
Figure 6.
Responses of plants in morphology, anatomy and physiology under salt stress. gs, stomatal conductance; Kh, hydraulic conductivity; Dc, conduit diameter. The up arrow indicates an increase and the down arrow indicates a decrease.
2.4.1. Physiological Responses of Plants under Salt Stress
Under salt stress, a high concentration of Na+ in leaves could decrease stomatal conductance by reducing K+ and Ca2+ uptake, thus preventing water loss [214,215]. In addition, ABA interacts with gibberellic acids (GAs), which leads to stomatal closure and decreases the flow rate of toxic ions to limit the accumulation of these ions in the shoot [216,217]. Moreover, xylem hydraulic conductivity and water potential is decreased under salt stress [218,219], while the turgor loss point is increased [220]. The response of physiological traits under salt stress varies with different species or varieties. For example, the salt-intolerant mangrove species Laguncularia racemosa is more susceptible to xylem embolism under salt stress than the salt-tolerant mangrove species Rhizophora mangle [221]. Under salt stress, Lycium chinense Miller has a lower PLC and lower content of NSC in all organs, while the PLC of Tamarix chinensis and the content of NSC in the shoot is higher under salt stress [222].
2.4.2. Morphological and Anatomical Responses of Plants under Salt Stress
Salt stress alters woody plants’ morphology and anatomy [217]. Salt stress can significantly reduce the leaf biomass, alter the stomatal area and density, and may cause damage to mesophyll cells [223,224,225]. In addition, salt stress can significantly reduce salt-sensitive plants’ root biomass [226,227,228], while salt-tolerant plants are generally unaffected or build denser root systems [227,229,230]. Under salt stress, xylem anatomical traits were changed, such as decreased conduit diameter [183] and increased wood density [231]. On the one hand, the decrease of K+ and Ca2+ absorption induced by osmotic stress may lead to a decline in radial growth of the xylem and hence decreased xylem conduit diameter [232]. On the other hand, salt stress could reduce the auxin concentration, affecting cambium activity and hence xylem growth and differentiation [233]. For example, with decreased concentration of free indole-3-acetic acid (IAA) due to salt stress, two Populus species showed decreased vessel lumen diameter and thickened vessel wall, although salt-tolerant Populus euphratica was slightly affected compared with the other species [234].
However, high auxin concentrations could lead to higher vessel density and lower vessel size [235]. In addition, salt stress results in a highly lignified cell wall of the woody plants’ root system due to changes in hormone regulation (such as ABA and ethylene) and the enzymes in the lignin synthesis pathway [84,236,237], which increases cell wall thickness and mechanical strength and plays an essential role in protecting cell membrane integrity and improving salt tolerance [238].
2.5. Heavy Metal
Heavy metal pollution is worldwide, insidious, persistent and irreversible, and can accumulate in the food chain, posing a great threat to the health of living beings [239,240]. Heavy metal pollution leads to lower forest species richness and reduces tree regeneration [241]. Heavy metals such as Cu, Zn and Mn are essential elements for trees [242], while heavy metals such as Cd, Hg, As, Pb and Cr are always biologically toxic [243], which leads to the reduction in leaf photosynthesis and transpiration rate [244]. In addition, the toxicity of heavy metals to plant roots includes accelerating the aging of root cells, affecting cell division, thus reducing the absorption of nitrate and other nutrients by roots [245,246]. However, Populus alba watered with wastewater containing heavy metals (Cu, Pb, As and Ni) had a higher biomass and more leaves than those irrigated with drinking water, which may be due to the higher nutrient content in wastewater [247].
2.5.1. Heavy Metal Absorption and Physiological Responses of Plants
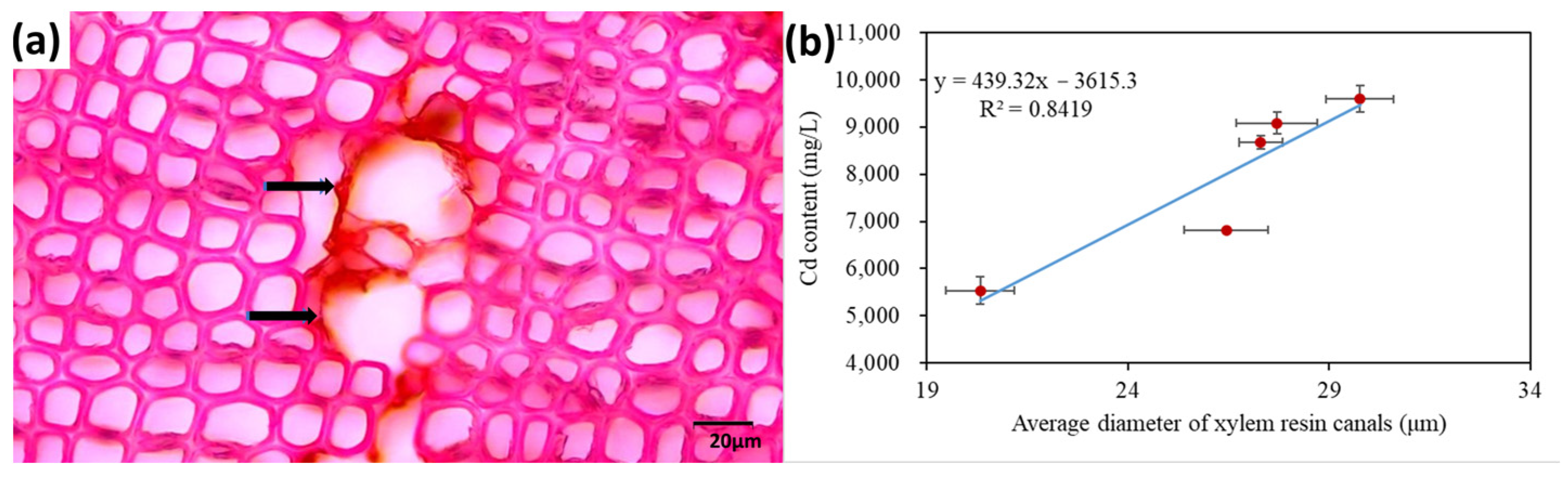
Plant roots absorb heavy metals mainly in two ways. Firstly, the root exudates, which have an acidic carboxyl group, could bind heavy metals and thus increase the availability of heavy metals [248]. Secondly, heavy metal ions could enter the plant through the cell membrane of root hair and move to the root epidermal cells through symplastic transport [249]. Heavy metals are transported from root to shoot through the xylem and phloem [250,251]. The average diameter of the resin canals in the root xylem of five Pinaceae species has a significant positive relationship with the root Cd concentration (Figure 7) [252], demonstrating that xylem anatomical traits could be useful indicators for evaluating trees’ heavy metal accumulation ability.
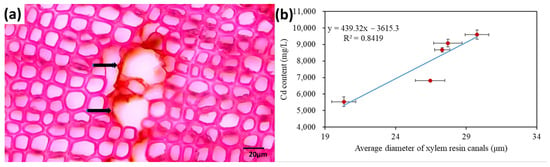
Figure 7.
(a) Cross-section of the stem xylem of Pinus thunbergii. The arrows point to the resin canals. (b) Unpublished data showed the relationship between the average diameter of resin canals in the root xylem of five tree species (Pinus bungeana, Pinus massoniana, Pinus tabuliformis, Pinus armandii and Cedrus deodara and root Cd concentration (n = 3). Horizontal and vertical error bars represent ±standard deviation (SD).
Under heavy metal stress, the leaf stomatal conductance and transpiration rate decreases, where ABA plays a vital role in this process [253]. For instance, the induction of NADPH oxidase by Cd and Cu could have been mediated by ABA, which has been shown to enhance NADPH oxidase activity and hence H2O2 accumulation and propagation in guard cells, inducing stomatal closure to prevent water loss and translocation of heavy metals from the belowground to the aboveground [254,255]. Woody plants under heavy metal stress often exhibited symptoms similar to those under drought stress [256,257], such as decreased xylem hydraulic conductivity, which could be due to xylem embolism formation and spread and decrease of AQP activity [258,259]. Reduced xylem embolism resistance was also observed in A. rubrum under heavy metal stress [260].
2.5.2. Morphological and Anatomical Responses of Plants under Heavy Metal Stress
Under heavy metal stress, woody plants’ morphological and anatomical traits change to reduce the absorption and translocation of heavy metals. For example, reduced root length and total root area, thickened endodermis and exodermis [261] and lignified pericycle and endodermis were observed [262,263]. Heavy metal stress also affects leaf morphological and anatomical structure, such as reduced leaf area and thickness [262,264,265] and decreased stomatal density [256,266]. Anatomically, changes in xylem structure were reported (Table 1), such as a decreased xylem conduit area and conduit lumen diameter [267,268]. The xylem tracheids of Pisum sylvestris grown on tailings became smaller [269]; the root xylem structure of A. rubrum was damaged under Cr stress [260]. Under Cd, Pb and Zn stress, F. sylvatica showed a decrease in mean ring width and vessel radial diameter, an increase in vessel density and vessel grouping index, while conductivity per xylem area were not significantly affected [270]. We also found the tracheid lumen diameter of Pinus thunbergii and Platycladus orientalis reduced significantly under Cd stress [271]. These changes lead to decreased root water absorption, decreased xylem hydraulic conductivity and reduced heavy metal uptake [262,263,272]. In addition, the responses of plant functional traits under heavy metal stress vary with different plant organs. For instance, A. rubrum’s root conduit density increased under heavy metal stress while its stem conduit density decreased [256]. To resist heavy metals, non-hyperaccumulator plants adopt exclusion strategies such as keeping metal ions in the cell wall or enhancing tolerance to harmful metal ions by metal chelators [273]. Cell wall polysaccharides are essential in heavy metal accumulation and the protection of protoplasts [274]. Under heavy metal stress, woody plants increase the cell wall thickness, which increases the mechanical strength of the cell wall and enables more heavy metals to accumulate in the cell wall to avoid entering the protoplasts [274,275,276].

Table 1.
The effects of different heavy metals on the plant morphological and anatomical traits. Dc, conduit diameter; Ds, leaf stomatal density. The up arrow indicates an increase, and the down arrow indicates a decrease.
3. Response of Woody Plants to Combined Abiotic Stresses
In most studies, researchers focus on the effects of single abiotic stress on woody plants; however, in the natural environment, most stresses occur as combined stresses that become more frequent in the future [283,284]. The effects of combined stresses on woody plants may be either synergistic or opposite compared with that of the single stress. As the response of woody plants to combined stresses cannot be inferred from a single stress, more attention has been paid to the effects and mechanism of combined stresses on them [284].
Drought often occurs alongside stresses such as heat, heavy metals and salt, which could lead to unique plant responses. For instance, isohydric trees close their stomata under single stress, such as drought and heat, while under the combined stress of drought and heat, they usually open their stomata to decrease leaf temperature. However, the increased stomatal conductance also leads to more severe water loss, which makes them more likely to reach the death threshold [285]. Different from isohydric trees, species of Citrus close their stomata under the combined stress of drought and heat [286,287]. However, even if the stomata are closed, trees lose water by cuticular transpiration [149,288]. Under combined drought and heat stress, the cuticular conductance increases sharply above the cuticular phase change temperature, leading to stronger cuticular transpiration and further xylem embolism [288].
Heavy metal ions could aggravate the degree of drought stress in an additive way, making trees more vulnerable to xylem embolism [276]. However, under the combined stress of heavy metals and drought, the root conduit density of A. rubrum decreased while it increased under the single stress of drought or heavy metal [256], which indicated that drought and heavy metal had an interaction. Under combined drought and salt stress, leaf photosynthetic traits such as the chlorophyll content reduced; however, proline, total soluble protein and sugar levels increased [289,290]. The combined stress of drought and soil salinity may induce increased ABA concentration and weakened plant metabolism, triggering earlier leaf senescence and less nutrient retranslation [291]. In addition, under combined stress of drought and salt, wood density and xylem embolism resistance of Taxodium distichum (L.) Rich. increased while hydraulic conductivity decreased [231]. We also found that the combination of drought and salt stress caused more severe embolisms in Platycladus orientalis than either single stress (Li et al., unpublished data [97]).
In addition to drought, a wide variety of combined stresses exist in nature that may affect the physiological, morphological and anatomical traits of woody plants. The interaction between different combinations of stress may not be simply additive. Under the combined stress of nitrogen deficiency and Cd, the antioxidant defense capacity of Populus × euramericana was reduced, resulting in lower Cd tolerance, inhibition of xylem development and reduced Cd accumulation capacity [292]. Under combined Cd and salt stress, the addition of NaCl increased the transport of Cd from root to shoot, but also slowed the reduction in the relative water content caused by Cd stress [293]. In addition, salinity reduced root biomass of Populus deltoides more severely than Cd stress, but under the combined stress of the two, the inhibition of root growth was not significantly different compared with salt stress alone [294]. Under combined drought and salt stress, the total biomass and total leaf area of Populus tomentosa decreased and the proportion of fine roots to total root biomass increased, which were more severe under combined stress than any single stress [295]. Under single stress of heat or CO2, the tracheid walls of Pinus sylvestris thickened; however, under the combined stress of heat and CO2, the tracheid walls of Pinus sylvestris thinned [296]. In summary, the response of woody plants under combined stresses could be different from any single stress and the intrinsic mechanisms are open to investigation.
4. Conclusions
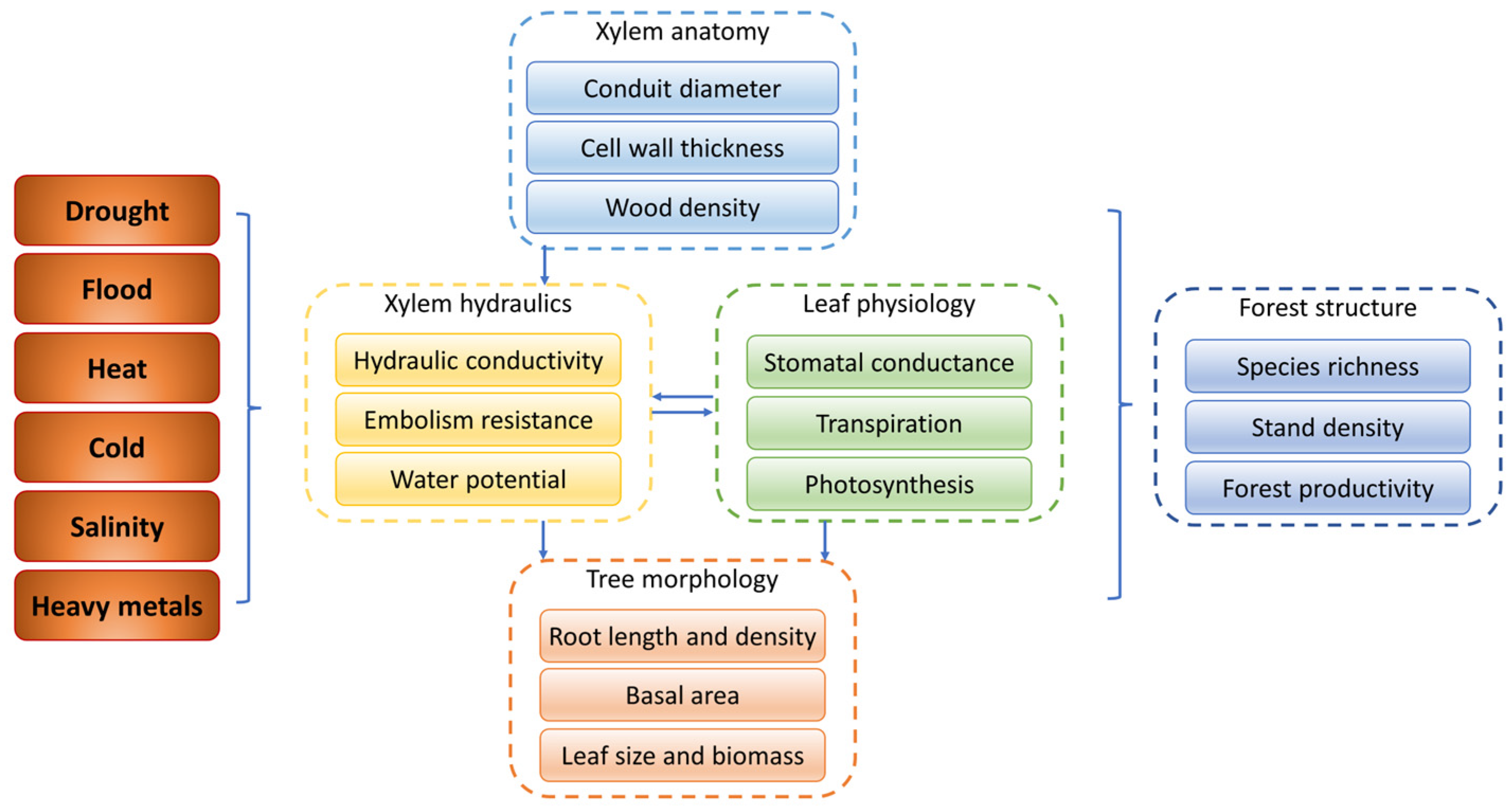
In this paper, the responses of woody plants to most abiotic stresses are mainly discussed from physiological, morphological and anatomical traits (Figure 8). Under most abiotic stress, leaf photosynthetic indicators decline, and xylem hydraulic conductivity decreases, with AQP activity inhibition. Stomatal regulation serves as the primary defense mechanism of woody plants against abiotic stresses, with stomata being the fastest to respond to such stresses. Most abiotic stresses result in limited water uptake by woody plants from the soil, prompting woody plants to close their stomata to reduce water loss, albeit at the cost of impeding leaf carbon assimilation. Morphologically, under drought stress, woody plants tend to increase the length and depth of their roots to enhance their water uptake efficiency. Under flooding stress, adventitious roots and aerenchyma facilitate greater oxygen availability for the woody plants. However, under other detrimental stresses such as forest fires, salinity and heavy metal toxicity, root growth in woody plants will be suppressed. The morphological adjustment of leaves is closely related to photosynthesis, respiration and transpiration, and when necessary, some species lose their leaves to escape abiotic stress. Moreover, most abiotic stresses lead to narrower conduits and thickened, lignified conduit walls, which enhances the embolism resistance and reduces hydraulic conductivity. Although different abiotic stresses may show similar effects on the anatomical traits of woody plants, they also have unique physiological effects. Under hypoxia caused by flooding stress, woody plants develop adventitious roots and aeration tissues to maintain gas exchange and nutrient transport. Forest fires can cause permanent damage to the tree and eventually result in tree mortality due to hydraulic failure and carbon starvation. Trees respond to freeze–thaw embolisms due to low-temperature stress through supercooling and embolism refilling in the following year. Under salt and heavy metal stress, woody plants build thicker cell walls and narrower conduits to reduce the efficiency of transporting harmful substances to the aboveground parts. Under stresses of drought, flooding and heavy metals, plant growth may occasionally be promoted. In addition to the various abiotic stresses mentioned in this review, microplastics, a new type of pollutant, have been shown to enter the root of Betula pendula, Q. petraeus and Picea abies, and may be transported through the xylem or in the apoplast of the lignified root epidermis [297,298]. Although xylem has been reported as an important pathway for microplastic transport in corps [299,300], whether it applies to woody plants is unknown. Woody plants are exposed to a higher risk of microplastic stress than crops and have a higher potential uptake capacity [297]. As a persistent pollutant, the interaction of microplastics with tree xylem needs further study.
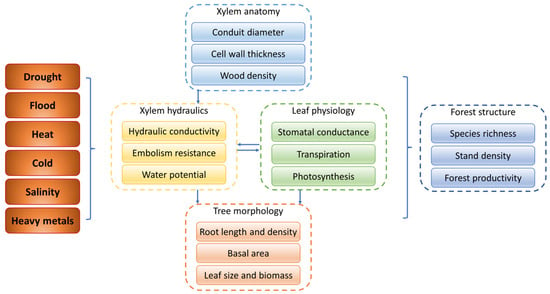
Figure 8.
The responses of physiological, morphological and anatomical traits to abiotic stresses. Arrows indicate interactions between the traits.
Although the responses of physiological, morphological and anatomical traits under abiotic stress have been widely studied, the molecular mechanisms of xylem development and adaptation to abiotic stress are still unclear. For example, the diameter of vessels or tracheids in trees may vary under abiotic stress, but which genes and pathways regulate this variation? How does xylem tissue respond to different abiotic stress signals? The interactions and trade-offs between the effects of multiple abiotic stresses on woody plants also deserve further investigation.
Author Contributions
Writing—original draft preparation, S.L. (Shan Li), S.L. (Sen Lu) and J.W.; data curation, S.L. (Sen Lu), J.W., P.L. and X.W.; writing—review and editing, S.L. (Shan Li), S.L. (Sen Lu), Z.C., Y.Z., J.D. and J.G.; supervision, S.L. (Shan Li), Z.C., Y.Z., J.D. and J.G.; conceptualization and funding acquisition, S.L. (Shan Li). All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China, grant number 32001291, the Talent Project of Shaanxi University of Science and Technology, grant number 126022037, the Shaanxi Province Science and Technology Innovation Team, grant number 2022TD-09 and the Key Industrial Chain Project of Shaanxi Province, grant number 2022ZDLNY02-02.
Data Availability Statement
Data sharing is not applicable.
Acknowledgments
We thank Yann Salmon for suggestions on manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhang, H.; Zhu, J.; Gong, Z.; Zhu, J.-K. Abiotic stress responses in plants. Nat. Rev. Genet. 2022, 23, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Stott, P. How climate change affects extreme weather events. Science 2016, 352, 1517–1518. [Google Scholar] [CrossRef]
- Salvatierra, A.; Toro, G.; Mateluna, P.; Opazo, I.; Ortiz, M.; Pimentel, P. Keep calm and survive: Adaptation strategies to energy crisis in fruit trees under root hypoxia. Plants 2020, 9, 1108. [Google Scholar] [CrossRef]
- Jagadish, S.V.K.; Way, D.A.; Sharkey, T.D. Plant heat stress: Concepts directing future research. Plant Cell Environ. 2021, 44, 1992–2005. [Google Scholar] [CrossRef] [PubMed]
- Bowd, E.J.; Blair, D.P.; Lindenmayer, D.B. Prior disturbance legacy effects on plant recovery post-high-severity wildfire. Ecosphere 2021, 12, e03480. [Google Scholar] [CrossRef]
- Knight, M.R.; Knight, H. Low-temperature perception leading to gene expression and cold tolerance in higher plants. New Phytol. 2012, 195, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Ilangumaran, G.; Smith, D.L. Plant growth promoting rhizobacteria in amelioration of salinity stress: A systems biology perspective. Front. Plant Sci. 2017, 8, 1768. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Ilahi, I. Environmental chemistry and ecotoxicology of hazardous heavy metals: Environmental persistence, toxicity, and bioaccumulation. J. Chem. 2019, 2019, 6730305. [Google Scholar] [CrossRef]
- Ghuge, S.A.; Nikalje, G.C.; Kadam, U.S.; Suprasanna, P.; Hong, J.C. Comprehensive mechanisms of heavy metal toxicity in plants, detoxification, and remediation. J. Hazard. Mater. 2023, 450, 131039. [Google Scholar] [CrossRef]
- Zimmermann, M.H. Xylem Structure and the Ascent of Sap; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Li, S.; Tan, T.; Fan, Y.; Raza, M.A.; Wang, Z.; Wang, B.; Zhang, J.; Tan, X.; Chen, P.; Shafiq, I.; et al. Responses of leaf stomatal and mesophyll conductance to abiotic stress factors. J. Integr. Agric. 2022, 21, 2787–2804. [Google Scholar] [CrossRef]
- Devireddy, A.R.; Zandalinas, S.I.; Fichman, Y.; Mittler, R. Integration of reactive oxygen species and hormone signaling during abiotic stress. Plant J. 2021, 105, 459–476. [Google Scholar] [CrossRef]
- Peng, P.; Li, R.; Chen, Z.-H.; Wang, Y. Stomata at the crossroad of molecular interaction between biotic and abiotic stress responses in plants. Front. Plant Sci. 2022, 13, 1031891. [Google Scholar] [CrossRef] [PubMed]
- Karlova, R.; Boer, D.; Hayes, S.; Testerink, C. Root plasticity under abiotic stress. Plant Physiol. 2021, 187, 1057–1070. [Google Scholar] [CrossRef] [PubMed]
- Gutschick, V.P. Biotic and abiotic consequences of differences in leaf structure. New Phytol. 1999, 143, 3–18. [Google Scholar] [CrossRef]
- Hu, W.; Lu, Z.; Meng, F.; Li, X.; Cong, R.; Ren, T.; Sharkey, T.D.; Lu, J. The reduction in leaf area precedes that in photosynthesis under potassium deficiency: The importance of leaf anatomy. New Phytol. 2020, 227, 1749–1763. [Google Scholar] [CrossRef]
- Zhou, H.; Zhou, G.; He, Q.; Zhou, L.; Ji, Y.; Zhou, M. Environmental explanation of maize specific leaf area under varying water stress regimes. Environ. Exp. Bot. 2020, 171, 103932. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, L.; Qi, D. Variation in leaf traits at different altitudes reflects the adaptive strategy of plants to environmental changes. Ecol. Evol. 2020, 10, 8166–8175. [Google Scholar] [CrossRef]
- Ahmad, P.; Prasad, M.N.V. Abiotic Stress Responses in Plants: Metabolism, Productivity and Sustainability; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Zheng, J.; Zhao, X.; Morris, H.; Jansen, S. Phylogeny best explains latitudinal patterns of xylem tissue fractions for woody angiosperm species across China. Front. Plant Sci. 2019, 10, 556. [Google Scholar] [CrossRef]
- Choat, B.; Cobb, A.R.; Jansen, S. Structure and function of bordered pits: New discoveries and impacts on whole-plant hydraulic function. New Phytol. 2008, 177, 608–626. [Google Scholar] [CrossRef]
- Evert, R.F. Esau’s Plant Anatomy: Meristems, Cells, and Tissues of the Plant Body: Their Structure, Function, and Development; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Morris, H.; Plavcová, L.; Cvecko, P.; Fichtler, E.; Gillingham, M.A.F.; Martínez-Cabrera, H.I.; McGlinn, D.J.; Wheeler, E.; Zheng, J.; Ziemińska, K.; et al. A global analysis of parenchyma tissue fractions in secondary xylem of seed plants. New Phytol. 2015, 209, 1553–1565. [Google Scholar] [CrossRef]
- Greenwood, S.; Ruiz-Benito, P.; Martinez-Vilalta, J.; Lloret, F.; Kitzberger, T.; Allen, C.D.; Fensham, R.; Laughlin, D.C.; Kattge, J.; Bonisch, G.; et al. Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area. Ecol. Lett. 2017, 20, 539–553. [Google Scholar] [CrossRef]
- Bottero, A.; D’Amato, A.W.; Palik, B.J.; Bradford, J.B.; Fraver, S.; Battaglia, M.A.; Asherin, L.A.; Bugmann, H. Density-dependent vulnerability of forest ecosystems to drought. J. Appl. Ecol. 2017, 54, 1605–1614. [Google Scholar] [CrossRef]
- Nardini, A.; Savi, T.; Trifilò, P.; Lo Gullo, M.A. Drought stress and the recovery from xylem embolism in woody plants. In Progress in Botany Vol. 79; Progress in Botany; Springer: Berlin/Heidelberg, Germany, 2017; pp. 197–231. [Google Scholar]
- Schenk, H.J.; Steppe, K.; Jansen, S. Nanobubbles: A new paradigm for air-seeding in xylem. Trends Plant Sci. 2015, 20, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, J.; Yin, Y.; Li, X.; Deng, L.; Jiang, X.; Chen, Z.; Li, Y. Investigating effects of bordered pit membrane morphology and properties on plant xylem hydraulic functions-A case study from 3d reconstruction and microflow modelling of pit membranes in angiosperm xylem. Plants 2020, 9, 231. [Google Scholar] [CrossRef] [PubMed]
- Puglielli, G.; Catoni, R.; Spoletini, A.; Varone, L.; Gratani, L. Short-term physiological plasticity: Trade-off between drought and recovery responses in three Mediterranean cistus species. Ecol. Evol. 2017, 7, 10880–10889. [Google Scholar] [CrossRef]
- Martin-StPaul, N.; Delzon, S.; Cochard, H. Plant resistance to drought depends on timely stomatal closure. Ecol. Lett. 2017, 20, 1437–1447. [Google Scholar] [CrossRef]
- Seleiman, M.F.; Al-Suhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; Abdul-Wajid, H.H.; Battaglia, M.L. Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef]
- Ramachandra Reddy, A.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Li, S.; Li, X.; Wang, J.; Chen, Z.; Lu, S.; Wan, X.; Sun, H.; Wang, L.; Delzon, S.; Cochard, H. Hydraulic traits are coupled with plant anatomical traits under drought-rewatering cycles in Ginkgo biloba L. Tree Physiol. 2021, 42, 1216–1227. [Google Scholar] [CrossRef]
- Mantova, M.; Menezes-Silva, P.E.; Badel, E.; Cochard, H.; Torres-Ruiz, J.M. The interplay of hydraulic failure and cell vitality explains tree capacity to recover from drought. Physiol. Plant 2021, 172, 247–257. [Google Scholar] [CrossRef]
- Scoffoni, C.; Albuquerque, C.; Brodersen, C.R.; Townes, S.V.; John, G.P.; Bartlett, M.K.; Buckley, T.N.; McElrone, A.J.; Sack, L. Outside-xylem vulnerability, not xylem embolism, controls leaf hydraulic decline during dehydration. Plant Physiol. 2017, 173, 1197–1210. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, Y.; Li, W.; Ayup, M. Xylem hydraulic conductivity and embolism in riparian plants and their responses to drought stress in desert of Northwest China. Ecohydrology 2013, 6, 984–993. [Google Scholar] [CrossRef]
- Li, X.; Piao, S.; Wang, K.; Wang, X.; Wang, T.; Ciais, P.; Chen, A.; Lian, X.; Peng, S.; Penuelas, J. Temporal trade-off between gymnosperm resistance and resilience increases forest sensitivity to extreme drought. Nat. Ecol. Evol. 2020, 4, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Choat, B.; Jansen, S.; Brodribb, T.J.; Cochard, H.; Delzon, S.; Bhaskar, R.; Bucci, S.J.; Feild, T.S.; Gleason, S.M.; Hacke, U.G.; et al. Global convergence in the vulnerability of forests to drought. Nature 2012, 491, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Bauerle, T.L. A global analysis of plant recovery performance from water stress. Oikos 2017, 126, 1377–1388. [Google Scholar] [CrossRef]
- Anderegg, L.D.; HilleRisLambers, J. Drought stress limits the geographic ranges of two tree species via different physiological mechanisms. Glob. Chang. Biol. 2016, 22, 1029–1045. [Google Scholar] [CrossRef]
- Carnicer, J.; Barbeta, A.; Sperlich, D.; Coll, M.; Peñuelas, J. Contrasting trait syndromes in angiosperms and conifers are associated with different responses of tree growth to temperature on a large scale. Front. Plant Sci. 2013, 4, 409. [Google Scholar] [CrossRef]
- Urli, M.; Porte, A.J.; Cochard, H.; Guengant, Y.; Burlett, R.; Delzon, S. Xylem embolism threshold for catastrophic hydraulic failure in angiosperm trees. Tree Physiol. 2013, 33, 672–683. [Google Scholar] [CrossRef]
- Brodribb, T.J.; Cochard, H. Hydraulic failure defines the recovery and point of death in water-stressed conifers. Plant Physiol. 2009, 149, 575–584. [Google Scholar] [CrossRef]
- Hammond, W.M.; Yu, K.; Wilson, L.A.; Will, R.E.; Anderegg, W.R.L.; Adams, H.D. Dead or dying? Quantifying the point of no return from hydraulic failure in drought-induced tree mortality. New Phytol. 2019, 223, 1834–1843. [Google Scholar] [CrossRef]
- Stocker, O. Die Abhängigkeit der Transpiration von den Umweltfaktoren; Springer: Berlin/Heidelberg, Germany, 1956. [Google Scholar]
- Tardieu, F.; Simonneau, T. Variability among species of stomatal control under fluctuating soil water status and evaporative demand: Modelling isohydric and anisohydric behaviours. J. Exp. Bot. 1998, 49, 419–432. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, Y.; Peng, Q.; Li, K.; Mohammat, A.; Han, W. Nitrogen and phosphorus resorption of desert plants with various degree of propensity to salt in response to drought and saline stress. Ecol. Indic. 2021, 125, 107488. [Google Scholar] [CrossRef]
- Sade, N.; Gebremedhin, A.; Moshelion, M. Risk-taking plants: Anisohydric behavior as a stress-resistance trait. Plant Signal Behav. 2012, 7, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Klein, T. The variability of stomatal sensitivity to leaf water potential across tree species indicates a continuum between isohydric and anisohydric behaviours. Funct. Ecol. 2014, 28, 1313–1320. [Google Scholar] [CrossRef]
- McAdam, S.A.; Brodribb, T.J. The evolution of mechanisms driving the stomatal response to vapor pressure deficit. Plant Physiol. 2015, 167, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Daszkowska-Golec, A.; Szarejko, I. Open or close the gate-stomata action under the control of phytohormones in drought stress conditions. Front. Plant Sci. 2013, 4, 138. [Google Scholar] [CrossRef]
- Ennajeh, M.; Vadel, A.; Cochard, H.; Khemira, H. Comparative impacts of water stress on the leaf anatomy of a drought-resistant and a drought-sensitive olive cultivar. J. Hortic. Sci. Biotechnol. 2010, 85, 289–294. [Google Scholar] [CrossRef]
- Farooq, M.; Hussain, M.; Wahid, A.; Siddique, K. Drought stress in plants: An overview. In Plant Responses to Drought Stress; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 1–33. [Google Scholar]
- Strock, C.F.; Burridge, J.D.; Niemiec, M.D.; Brown, K.M.; Lynch, J.P. Root metaxylem and architecture phenotypes integrate to regulate water use under drought stress. Plant Cell Environ. 2021, 44, 49–67. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Y.; Wang, X.; Han, X.; An, Y.; Lin, S.; Shen, C.; Wen, J.; Liu, C.; Yin, W.; et al. Root-specific NF-Y family transcription factor, PdNF-YB21, positively regulates root growth and drought resistance by abscisic acid-mediated indoylacetic acid transport in Populus. New Phytol. 2020, 227, 407–426. [Google Scholar] [CrossRef]
- Rais, A.; van de Kuilen, J.-W.G.; Pretzsch, H. Growth reaction patterns of tree height, diameter, and volume of Douglas-fir (Pseudotsuga menziesii [Mirb.] Franco) under acute drought stress in Southern Germany. Eur. J. For. Res. 2014, 133, 1043–1056. [Google Scholar] [CrossRef]
- Yang, B.; Shan, L.; Ma, J.; Xie, T.; Yang, J.; Wei, C. Response of growth and root morphological characteristics of Reaumuria soongorica seedlings to drought-rehydration. Arid. Zone Res. 2021, 38, 469–478. [Google Scholar]
- Sasani, N.; Paques, L.E.; Boulanger, G.; Singh, A.P.; Gierlinger, N.; Rosner, S.; Brendel, O. Physiological and anatomical responses to drought stress differ between two larch species and their hybrid. Trees 2021, 35, 1467–1484. [Google Scholar] [CrossRef]
- Thangthong, N.; Jogloy, S.; Jongrungklang, N.; Kvien, C.K.; Dodd, I.C.; Vorasoot, N. Changes in root xylem anatomy of peanut genotypes with different drought resistance levels under early-season drought. J. Agron. Crop Sci. 2021, 207, 803–813. [Google Scholar] [CrossRef]
- Jing, L.; Yubao, G.; Zhirong, Z.; Zenglu, G. Hydraulic architecture of three Caragana species and its relationship with environmental factors in different habitats of the Inner Mongolian Plateau, China. Acta Ecol. Sin. 2007, 27, 837–845. [Google Scholar] [CrossRef]
- Cai, J.; Tyree, M.T. The impact of vessel size on vulnerability curves: Data and models for within-species variability in saplings of aspen, Populus tremuloides Michx. Plant Cell Environ. 2010, 33, 1059–1069. [Google Scholar] [CrossRef]
- Hacke, U.G.; Spicer, R.; Schreiber, S.G.; Plavcová, L. An ecophysiological and developmental perspective on variation in vessel diameter. Plant Cell Environ. 2017, 40, 831–845. [Google Scholar] [CrossRef] [PubMed]
- Hacke, U.G.; Lachenbruch, B.; Pittermann, J.; Mayr, S.; Domec, J.-C.; Schulte, P.J. The hydraulic architecture of conifers. In Functional and Ecological Xylem Anatomy; Springer: Berlin/Heidelberg, Germany, 2015; pp. 39–75. [Google Scholar]
- Li, L.; Li, Z.; Jing, C.; Han, Z.; Yongqin, C.; Zaimin, J. Hydraulic characteristics and embolism repair of Populus alba × P. glandulosa after drought stress and rehydration. J. Beijing For. Univ. 2021, 43, 22–30. [Google Scholar]
- Montwé, D.; Spiecker, H.; Hamann, A. An experimentally controlled extreme drought in a Norway spruce forest reveals fast hydraulic response and subsequent recovery of growth rates. Trees 2014, 28, 891–900. [Google Scholar] [CrossRef]
- Bouche, P.S.; Delzon, S.; Choat, B.; Badel, E.; Brodribb, T.J.; Burlett, R.; Cochard, H.; Charra-Vaskou, K.; Lavigne, B.; Li, S. Are needles of Pinus pinaster more vulnerable to xylem embolism than branches? New insights from X-ray computed tomography. Plant Cell Environ. 2016, 39, 860–870. [Google Scholar] [CrossRef]
- Guerin, M.; von Arx, G.; Martin-Benito, D.; Andreu-Hayles, L.; Griffin, K.L.; McDowell, N.G.; Pockman, W.; Gentine, P. Distinct xylem responses to acute vs prolonged drought in pine trees. Tree Physiol. 2020, 40, 605–620. [Google Scholar] [CrossRef]
- Jupa, R.; Krabickova, D.; Plichta, R.; Mayr, S.; Gloser, V. Do angiosperm tree species adjust intervessel lateral contact in response to soil drought? Physiol. Plant 2021, 172, 2048–2058. [Google Scholar] [CrossRef]
- Soro, A.; Lenz, P.; Roussel, J.R.; Larochelle, F.; Bousquet, J.; Achim, A. The phenotypic and genetic effects of drought-induced stress on apical growth, ring width, wood density and biomass in white spruce seedlings. New For. 2022, 54, 789–811. [Google Scholar] [CrossRef]
- Balducci, L.; Deslauriers, A.; Giovannelli, A.; Beaulieu, M.; Delzon, S.; Rossi, S.; Rathgeber, C.B.K. How do drought and warming influence survival and wood traits of Picea mariana saplings? J. Exp. Bot. 2015, 66, 377–389. [Google Scholar] [CrossRef]
- Rosner, S.; Světlík, J.; Andreassen, K.; Børja, I.; Dalsgaard, L.; Evans, R.; Karlsson, B.; Tollefsrud, M.M.; Solberg, S. Wood density as a screening trait for drought sensitivity in Norway spruce. Can. J. For. Res. 2014, 44, 154–161. [Google Scholar] [CrossRef]
- Zou, J.; Hu, W.; Li, Y.; Zhu, H.; He, J.; Wang, Y.; Meng, Y.; Chen, B.; Zhao, W.; Wang, S.; et al. Leaf anatomical alterations reduce cotton’s mesophyll conductance under dynamic drought stress conditions. Plant J. 2022, 111, 391–405. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, Y.; Su, M.X.; Han, Z.J.; Shi, J.Y. Evaluation of salt and drought tolerances of Populus talassica × Populus euphratica seedlings using leaf anatomical structures and physiological processes. Pak. J. Bot. 2023, 55, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Cao, L. Eco-Physiology Responses of Camellia oleifera to the Drought Stress. Master’s Thesis, Chinese Academy of Forestry, Beijing, China, 2017. [Google Scholar]
- Dickison, W.C. Integrative Plant Anatomy; Academic Press: Cambridge, MA, USA, 2000. [Google Scholar]
- Little, C.; Savidge, R. The role of plant growth regulators in forest tree cambial growth. In Hormonal Control of Tree Growth; Springer: Berlin/Heidelberg, Germany, 1987; pp. 137–169. [Google Scholar]
- Abe, H.; Nakai, T.; Utsumi, Y.; Kagawa, A. Temporal water deficit and wood formation in Cryptomeria japonica. Tree Physiol. 2003, 23, 859–863. [Google Scholar] [CrossRef]
- Popko, J.; Hansch, R.; Mendel, R.R.; Polle, A.; Teichmann, T. The role of abscisic acid and auxin in the response of poplar to abiotic stress. Plant Biol. 2010, 12, 242–258. [Google Scholar] [CrossRef]
- Zhao, D.; Luan, Y.; Shi, W.; Zhang, X.; Meng, J.; Tao, J. A Paeonia ostii caffeoyl-CoA O-methyltransferase confers drought stress tolerance by promoting lignin synthesis and ROS scavenging. Plant Sci. 2021, 303, 110765. [Google Scholar] [CrossRef]
- Donaldson, L.A. Abnormal lignin distribution in wood from severely drought stressed Pinus radiata trees. IAWA J. 2002, 23, 161–178. [Google Scholar] [CrossRef]
- Niu, Z.; Li, G.; Hu, H.; Lv, J.; Zheng, Q.; Liu, J.; Wan, D. A gene that underwent adaptive evolution, LAC2 (LACCASE), in Populus euphratica improves drought tolerance by improving water transport capacity. Hortic. Res. 2021, 8, 88. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Linker, R.; Gepstein, S.; Tanimoto, E.; Yamamoto, R.; Neumann, P.M. Progressive inhibition by water deficit of cell wall extensibility and growth along the elongation zone of maize roots is related to increased lignin metabolism and progressive stelar accumulation of wall phenolics. Plant Physiol. 2006, 140, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Hura, T.; Hura, K.; Dziurka, K.; Ostrowska, A.; Bączek-Kwinta, R.; Grzesiak, M. An increase in the content of cell wall-bound phenolics correlates with the productivity of triticale under soil drought. J. Plant Physiol. 2012, 169, 1728–1736. [Google Scholar] [CrossRef]
- Le Gall, H.; Philippe, F.; Domon, J.M.; Gillet, F.; Pelloux, J.; Rayon, C. Cell wall metabolism in response to abiotic stress. Plants 2015, 4, 112–166. [Google Scholar] [CrossRef] [PubMed]
- Creek, D.; Blackman, C.J.; Brodribb, T.J.; Choat, B.; Tissue, D.T. Coordination between leaf, stem, and root hydraulics and gas exchange in three arid-zone angiosperms during severe drought and recovery. Plant Cell Environ. 2018, 41, 2869–2881. [Google Scholar] [CrossRef]
- Hasan, M.M.; Gong, L.; Nie, Z.-F.; Li, F.-P.; Ahammed, G.J.; Fang, X.-W. ABA-induced stomatal movements in vascular plants during dehydration and rehydration. Environ. Exp. Bot. 2021, 186, 104436. [Google Scholar] [CrossRef]
- Bi, M.H.; Jiang, C.; Brodribb, T.; Yang, Y.J.; Yao, G.Q.; Jiang, H.; Fang, X.W. Ethylene constrains stomatal reopening in Fraxinus chinensis post moderate drought. Tree Physiol. 2023, 43, 883–892. [Google Scholar] [CrossRef]
- Klein, T.; Zeppel, M.J.; Anderegg, W.R.; Bloemen, J.; De Kauwe, M.G.; Hudson, P.; Ruehr, N.K.; Powell, T.L.; von Arx, G.; Nardini, A. Xylem embolism refilling and resilience against drought-induced mortality in woody plants: Processes and trade-offs. Ecol. Res. 2018, 33, 839–855. [Google Scholar] [CrossRef]
- Liu, J.; Gu, L.; Yu, Y.; Huang, P.; Wu, Z.; Zhang, Q.; Qian, Y.; Wan, X.; Sun, Z. Corticular photosynthesis drives bark water uptake to refill embolized vessels in dehydrated branches of Salix matsudana. Plant Cell Environ. 2019, 42, 2584–2596. [Google Scholar] [CrossRef]
- Tomasella, M.; Natale, S.; Petruzzellis, F.; Di Bert, S.; D’Amico, L.; Tromba, G.; Nardini, A. No evidence for light-induced embolism repair in cut stems of drought-resistant mediterranean species under soaking. Plants 2022, 11, 307. [Google Scholar] [CrossRef]
- Fuenzalida, T.I.; Blacker, M.J.; Turner, M.; Sheppard, A.; Ball, M.C. Foliar water uptake enables embolism removal in excised twigs of Avicennia marina. New Phytol. 2023, 237, 1136–1145. [Google Scholar] [CrossRef]
- Singh, S. Root pressure: Getting to the root of pressure. In Progress in Botany 77; Progress in Botany; Springer: Berlin/Heidelberg, Germany, 2016; pp. 105–150. [Google Scholar]
- Ewers, F.W.; Cochard, H.; Tyree, M.T. A survey of root pressures in vines of a tropical lowland forest. Oecologia 1997, 110, 191–196. [Google Scholar] [CrossRef]
- Wang, L.; Dai, Y.; Zhang, J.; Meng, P.; Wan, X. Xylem structure and hydraulic characteristics of deep roots, shallow roots and branches of walnut under seasonal drought. BMC Plant Biol. 2022, 22, 440. [Google Scholar] [CrossRef] [PubMed]
- Wegner, L.H. Root pressure and beyond: Energetically uphill water transport into xylem vessels? J. Exp. Bot. 2014, 65, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Trifilò, P.; Kiorapostolou, N.; Petruzzellis, F.; Vitti, S.; Petit, G.; Gullo, M.A.L.; Nardini, A.; Casolo, V. Hydraulic recovery from xylem embolism in excised branches of twelve woody species: Relationships with parenchyma cells and non-structural carbohydrates. Plant Physiol. Biochem. 2019, 139, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; (Institute for Systematic Botany and Ecology, Ulm University, Ulm, Germany). Unpublished work. 2015.
- Secchi, F.; Pagliarani, C.; Cavalletto, S.; Petruzzellis, F.; Tonel, G.; Savi, T.; Tromba, G.; Obertino, M.M.; Lovisolo, C.; Nardini, A.; et al. Chemical inhibition of xylem cellular activity impedes the removal of drought-induced embolisms in poplar stems—New insights from micro-CT analysis. New Phytol. 2021, 229, 820–830. [Google Scholar] [CrossRef]
- Brodersen, C.R.; Knipfer, T.; McElrone, A.J. In vivo visualization of the final stages of xylem vessel refilling in grapevine (Vitis vinifera) stems. New Phytol. 2018, 217, 117–126. [Google Scholar] [CrossRef]
- Ryu, J.; Hwang, B.G.; Lee, S.J. In vivo dynamic analysis of water refilling in embolized xylem vessels of intact Zea mays leaves. Ann. Bot. 2016, 118, 1033–1042. [Google Scholar] [CrossRef]
- Choat, B.; Nolf, M.; Lopez, R.; Peters, J.M.R.; Carins-Murphy, M.R.; Creek, D.; Brodribb, T.J. Non-invasive imaging shows no evidence of embolism repair after drought in tree species of two genera. Tree Physiol. 2019, 39, 113–121. [Google Scholar] [CrossRef]
- Hawes, J.E.; Peres, C.A.; Riley, L.B.; Hess, L.L. Landscape-scale variation in structure and biomass of Amazonian seasonally flooded and unflooded forests. For. Ecol. Manag. 2012, 281, 163–176. [Google Scholar] [CrossRef]
- Allen, S.T.; Keim, R.F.; Dean, T.J. Contrasting effects of flooding on tree growth and stand density determine aboveground production, in baldcypress forests. For. Ecol. Manag. 2019, 432, 345–355. [Google Scholar] [CrossRef]
- Araújo, F.D.C.; Tng, D.Y.P.; Apgaua, D.M.G.; Morel, J.D.; Pereira, D.G.S.; Santos, P.F.; Santos, R.M.d.; Collins, B. Flooding regime drives tree community structure in Neotropical dry forests. J. Veg. Sci. 2019, 30, 1195–1205. [Google Scholar] [CrossRef]
- Jackson, M.B.; Ishizawa, K.; Ito, O. Evolution and mechanisms of plant tolerance to flooding stress. Ann. Bot. 2009, 103, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Herrera, A.; Tezara, W.; Rengifo, E.; Flores, S. Changes with seasonal flooding in sap flow of the tropical flood-tolerant tree species, Campsiandra laurifolia. Trees 2008, 22, 551–558. [Google Scholar] [CrossRef]
- Li, M.; López, R.; Venturas, M.; Pita, P.; Gordaliza, G.G.; Gil, L.; Rodríguez-Calcerrada, J. Greater resistance to flooding of seedlings of Ulmus laevis than Ulmus minor is related to the maintenance of a more positive carbon balance. Trees 2015, 29, 835–848. [Google Scholar] [CrossRef]
- Tan, X.; Zwiazek, J.J. Stable expression of aquaporins and hypoxia-responsive genes in adventitious roots are linked to maintaining hydraulic conductance in tobacco (Nicotiana tabacum) exposed to root hypoxia. PLoS ONE 2019, 14, e0212059. [Google Scholar] [CrossRef]
- Wang, J.; Sun, H.; Sheng, J.; Jin, S.; Zhou, F.; Hu, Z.; Diao, Y. Transcriptome, physiological and biochemical analysis of Triarrhena sacchariflora in response to flooding stress. BMC Genetics 2019, 20, 88. [Google Scholar] [CrossRef]
- Ferner, E.; Rennenberg, H.; Kreuzwieser, J. Effect of flooding on C metabolism of flood-tolerant (Quercus robur) and non-tolerant (Fagus sylvatica) tree species. Tree Physiol. 2012, 32, 135–145. [Google Scholar] [CrossRef]
- Habibi, F.; Liu, T.; Shahid, M.A.; Schaffer, B.; Sarkhosh, A. Physiological, biochemical, and molecular responses of fruit trees to root zone hypoxia. Environ. Exp. Bot. 2023, 206, 105179. [Google Scholar] [CrossRef]
- Kwak, J.M.; Mori, I.C.; Pei, Z.-M.; Leonhardt, N.; Torres, M.A.; Dangl, J.L.; Bloom, R.E.; Bodde, S.; Jones, J.D.; Schroeder, J.I. NADPH oxidase AtrbohD and AtrbohF genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J. 2003, 22, 2623–2633. [Google Scholar] [CrossRef]
- Bashar, K.; Tareq, M.; Amin, M.; Honi, U.; Tahjib-Ul-Arif, M.; Sadat, M.; Hossen, Q. Phytohormone-mediated stomatal response, escape and quiescence strategies in plants under flooding stress. Agronomy 2019, 9, 43. [Google Scholar] [CrossRef]
- Nicolás, E.; Torrecillas, A.; Dell’Amico, J.; Alarcón, J.J. The effect of short-term flooding on the sap flow, gas exchange and hydraulic conductivity of young apricot trees. Trees 2005, 19, 51–57. [Google Scholar] [CrossRef]
- Ashraf, M.A. Waterlogging stress in plants: A review. Afr. J. Agric. Res. 2012, 7, 1976–1981. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X. Can early wilting of old leaves account for much of the ABA accumulation in flooded pea plants? J. Exp. Bot. 1994, 45, 1335–1342. [Google Scholar] [CrossRef]
- Domingo, R.; Pérez-Pastor, A.; Ruiz-Sánchez, M.C. Physiological responses of apricot plants grafted on two different rootstocks to flooding conditions. J. Plant Physiol. 2002, 159, 725–732. [Google Scholar] [CrossRef]
- Krauss, K.W.; Young, P.J.; Chambers, J.L.; Doyle, T.W.; Twilley, R.R. Sap flow characteristics of neotropical mangroves in flooded and drained soils. Tree Physiol. 2007, 27, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Steppe, K.; Cochard, H.; Lacointe, A.; Améglio, T. Could rapid diameter changes be facilitated by a variable hydraulic conductance? Plant Cell Environ. 2012, 35, 150–157. [Google Scholar] [CrossRef]
- Rodríguez-Gamir, J.; Ancillo, G.; González-Mas, M.C.; Primo-Millo, E.; Iglesias, D.J.; Forner-Giner, M.A. Root signalling and modulation of stomatal closure in flooded citrus seedlings. Plant Physiol. Biochem. 2011, 49, 636–645. [Google Scholar] [CrossRef]
- Bhusal, N.; Kim, H.S.; Han, S.-G.; Yoon, T.-M. Photosynthetic traits and plant–water relations of two apple cultivars grown as bi-leader trees under long-term waterlogging conditions. Environ. Exp. Bot. 2020, 176, 104111. [Google Scholar] [CrossRef]
- Gérard, B.; Alaoui-Sossé, B.; Badot, P.-M. Flooding effects on starch partitioning during early growth of two oak species. Trees 2009, 23, 373–380. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, S.; Li, J.; Li, Z.; Li, H.; Mao, J.; Zhai, F.; Liu, J.; Sun, Z. Gender-specific responses of Salix viminalis roots in morphology and physiology subjected to flooding. Flora 2023, 303, 152296. [Google Scholar] [CrossRef]
- Bogarín, M.R.A.; Reis, L.K.; Laura, V.A.; Pott, A.; Szabo, J.K.; Garcia, L.C. Morphological and phenological strategies for flooding tolerance in Cerrado and Pantanal trees: Implications for restoration under new legislation. Restor. Ecol. 2022, 31, e13660. [Google Scholar] [CrossRef]
- Zhou, W.; Chen, F.; Meng, Y.; Chandrasekaran, U.; Luo, X.; Yang, W.; Shu, K. Plant waterlogging/flooding stress responses: From seed germination to maturation. Plant Physiol. Biochem. 2020, 148, 228–236. [Google Scholar] [CrossRef]
- He, B.; Lai, T.; Fan, H.; Wang, W.; Zheng, H. Comparison of flooding-tolerance in four mangrove species in a diurnal tidal zone in the Beibu Gulf. Estuar. Coast. Shelf Sci. 2007, 74, 254–262. [Google Scholar] [CrossRef]
- Parolin, P. Senna reticulata, a pioneer tree from Amazonian várzea floodplains. Bot. Rev. 2001, 67, 239–254. [Google Scholar] [CrossRef]
- Kim, Y.; Shahzad, R.; Lee, I.-J. Regulation of flood stress in plants. In Plant Life Under Changing Environment; Academic Press: Cambridge, MA, USA, 2020; pp. 157–173. [Google Scholar]
- Oliveira, A.S.d.; Ferreira, C.S.; Graciano-Ribeiro, D.; Franco, A.C. Anatomical and morphological modifications in response to flooding by six Cerrado tree species. Acta Bot. Bras. 2015, 29, 478–488. [Google Scholar] [CrossRef]
- Steffens, B.; Rasmussen, A. The physiology of adventitious roots. Plant Physiol. 2016, 170, 603–617. [Google Scholar] [CrossRef]
- Calvo-Polanco, M.; Señorans, J.; Zwiazek, J.J. Role of adventitious roots in water relations of tamarack (Larix laricina) seedlings exposed to flooding. BMC Plant Biol. 2012, 12, 99. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Xu, H.; Khan, S.; Equiza, M.A.; Lee, S.H.; Vaziriyeganeh, M.; Zwiazek, J.J. Plant water transport and aquaporins in oxygen-deprived environments. J. Plant Physiol. 2018, 227, 20–30. [Google Scholar] [CrossRef]
- Vasellati, V.; Oesterheld, M.; Medan, D.; Loreti, J. Effects of flooding and drought on the anatomy of Paspalum dilatatum. Ann. Bot. 2001, 88, 355–360. [Google Scholar] [CrossRef]
- Yamauchi, T.; Noshita, K.; Tsutsumi, N. Climate-smart crops: Key root anatomical traits that confer flooding tolerance. Breed. Sci. 2021, 71, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Parvin, D.; Rashid, P.; Karmoker, J. Anatomical responses of jute (Corchorus capsularis L. cv. D-154) to waterlogging. Dhaka Univ. J. Biol. Sci. 2018, 27, 213–219. [Google Scholar] [CrossRef]
- Peng, Y.; Zhou, Z.; Tong, R.; Hu, X.; Du, K. Anatomy and ultrastructure adaptations to soil flooding of two full-sib poplar clones differing in flood-tolerance. Flora 2017, 233, 90–98. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Sun, J.; Yu, X.; Wang, H.; Jia, G.; Zhao, Y.; Tu, Z.; Deng, W.; Jia, J.; Chen, J. Effects of forest structure on hydrological processes in China. J. Hydrol. 2018, 561, 187–199. [Google Scholar] [CrossRef]
- Parente, J.; Pereira, M.G.; Amraoui, M.; Fischer, E.M. Extreme weather conditions: The role of an heat wave on wildfires in Portugal. In Advances in Forest Fire Research 2018; Coimbra University Press: Coimbra, Portugal, 2018; pp. 1200–1204. [Google Scholar]
- Bradford, J.B.; Bell, D.M. A window of opportunity for climate-change adaptation: Easing tree mortality by reducing forest basal area. Front. Ecol. Environ. 2016, 15, 11–17. [Google Scholar] [CrossRef]
- Peñuelas, J.; Ogaya, R.; Boada, M.; Jump, A.S. Migration, invasion and decline: Changes in recruitment and forest structure in a warming-linked shift of European beech forest in Catalonia (NE Spain). Ecography 2007, 30, 829–837. [Google Scholar] [CrossRef]
- Stoddard, M.T.; Huffman, D.W.; Fulé, P.Z.; Crouse, J.E.; Sánchez Meador, A.J. Forest structure and regeneration responses 15 years after wildfire in a ponderosa pine and mixed-conifer ecotone, Arizona, USA. Fire Ecol. 2018, 14, 12. [Google Scholar] [CrossRef]
- Reilly, S.; Clark, M.L.; Bentley, L.P.; Matley, C.; Piazza, E.; Oliveras Menor, I. The Potential of Multispectral Imagery and 3D Point Clouds from Unoccupied Aerial Systems (UAS) for Monitoring Forest Structure and the Impacts of Wildfire in Mediterranean-Climate Forests. Remote Sens. 2021, 13, 3810. [Google Scholar] [CrossRef]
- Pareek, A.; Sopory, S.K.; Bohnert, H. Abiotic Stress Adaptation in Plants; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Rehschuh, R.; Ruehr, N.K. Unrevealing water and carbon relations during and after heat and hot drought stress in Pinus sylvestris. Tree Phisiol. 2022, 42, 1532–1548. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, R.; Xu, X.; Fowler, J.C.; Miller, T.E.X.; Dong, T. Effect of summer warming on growth, photosynthesis and water status in female and male Populus cathayana: Implications for sex-specific drought and heat tolerances. Tree Physiol. 2020, 40, 1178–1191. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Xuan, A.; Bu, C.; Liu, X.; Zhang, D. Identification of a transcriptional regulatory module that reduces leaf temperature in poplar under heat stress. Tree Physiol. 2020, 40, 1108–1125. [Google Scholar] [CrossRef]
- Ruehr, N.K.; Gast, A.; Weber, C.; Daub, B.; Arneth, A. Water availability as dominant control of heat stress responses in two contrasting tree species. Tree Physiol. 2016, 36, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Schönbeck, L.C.; Schuler, P.; Lehmann, M.M.; Mas, E.; Mekarni, L.; Pivovaroff, A.L.; Turberg, P.; Grossiord, C. Increasing temperature and vapour pressure deficit lead to hydraulic damages in the absence of soil drought. Plant Cell Environ. 2022, 45, 3275–3289. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.E.; Tjoelker, M.G.; Varhammar, A.; Medlyn, B.E.; Reich, P.B.; Leigh, A.; Pfautsch, S.; Blackman, C.J.; Lopez, R.; Aspinwall, M.J.; et al. Trees tolerate an extreme heatwave via sustained transpirational cooling and increased leaf thermal tolerance. Glob. Chang. Biol. 2018, 24, 2390–2402. [Google Scholar] [CrossRef]
- Bar, A.; Michaletz, S.T.; Mayr, S. Fire effects on tree physiology. New Phytol. 2019, 223, 1728–1741. [Google Scholar] [CrossRef] [PubMed]
- Michaletz, S.T. Xylem dysfunction in fires: Towards a hydraulic theory of plant responses to multiple disturbance stressors. New Phytol. 2018, 217, 1391–1393. [Google Scholar] [CrossRef]
- Partelli-Feltrin, R.; Smith, A.M.S.; Adams, H.D.; Thompson, R.A.; Kolden, C.A.; Yedinak, K.M.; Johnson, D.M. Death from hunger or thirst? Phloem death, rather than xylem hydraulic failure, as a driver of fire-induced conifer mortality. New Phytol. 2022, 237, 1154–1163. [Google Scholar] [CrossRef]
- Gricar, J.; Hafner, P.; Lavric, M.; Ferlan, M.; Ogrinc, N.; Krajnc, B.; Eler, K.; Vodnik, D. Post-fire effects on development of leaves and secondary vascular tissues in Quercus pubescens. Tree Physiol. 2020, 40, 796–809. [Google Scholar] [CrossRef]
- Bauweraerts, I.; Ameye, M.; Wertin, T.M.; McGuire, M.A.; Teskey, R.O.; Steppe, K. Water availability is the decisive factor for the growth of two tree species in the occurrence of consecutive heat waves. Agr. For. Meteorol. 2014, 189–190, 19–29. [Google Scholar] [CrossRef]
- Filewod, B.; Thomas, S.C. Impacts of a spring heat wave on canopy processes in a northern hardwood forest. Glob. Chang. Biol. 2014, 20, 360–371. [Google Scholar] [CrossRef]
- Teskey, R.; Wertin, T.; Bauweraerts, I.; Ameye, M.; McGuire, M.A.; Steppe, K. Responses of tree species to heat waves and extreme heat events. Plant Cell Environ. 2015, 38, 1699–1712. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, L.; Wittemann, M.; Mujawamariya, M.; Manishimwe, A.; Zibera, E.; Ntirugulirwa, B.; Ract, C.; Manzi, O.J.L.; Andersson, M.X.; Spetea, C.; et al. Handling the heat—Photosynthetic thermal stress in tropical trees. New Phytol. 2022, 233, 236–250. [Google Scholar] [CrossRef] [PubMed]
- Leigh, A.; Sevanto, S.; Ball, M.C.; Close, J.D.; Ellsworth, D.S.; Knight, C.A.; Nicotra, A.B.; Vogel, S. Do thick leaves avoid thermal damage in critically low wind speeds? New Phytol. 2012, 194, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Mackay, D.S.; Savoy, P.R.; Grossiord, C.; Tai, X.; Pleban, J.R.; Wang, D.R.; McDowell, N.G.; Adams, H.D.; Sperry, J.S. Conifers depend on established roots during drought: Results from a coupled model of carbon allocation and hydraulics. New Phytol. 2020, 225, 679–692. [Google Scholar] [CrossRef]
- Thomas, D.S.; Montagu, K.D.; Conroy, J.P. Changes in wood density of Eucalyptus camaldulensis due to temperature—The physiological link between water viscosity and wood anatomy. For. Ecol. Manag. 2004, 193, 157–165. [Google Scholar] [CrossRef]
- Mundo, I.A.; González, C.V.; Stoffel, M.; Ballesteros-Cánovas, J.A.; Villalba, R. Fire damage to cambium affects localized xylem anatomy and hydraulics: The case of Nothofagus pumilio in Patagonia. Am. J. Bot. 2019, 106, 1536–1544. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. The cold range limit of trees. Trends Ecol. Evol. 2021, 36, 979–989. [Google Scholar] [CrossRef]
- Reich, P.B.; Sendall, K.M.; Stefanski, A.; Wei, X.; Rich, R.L.; Montgomery, R.A. Boreal and temperate trees show strong acclimation of respiration to warming. Nature 2016, 531, 633–636. [Google Scholar] [CrossRef]
- Carroll, C.J.W.; Martin, P.H.; Knapp, A.K.; Ocheltree, T.W. Temperature induced shifts in leaf water relations and growth efficiency indicate climate change may limit aspen growth in the Colorado Rockies. Environ. Exp. Bot. 2019, 159, 132–137. [Google Scholar] [CrossRef]
- Lafon, C.W. Forest disturbance by ice storms in Quercus forests of the southern Appalachian Mountains, USA. Ecoscience 2006, 13, 30–43. [Google Scholar] [CrossRef]
- Rubio-Cuadrado, A.; Gomez, C.; Rodriguez-Calcerrada, J.; Perea, R.; Gordaliza, G.G.; Julio Camarero, J.; Montes, F.; Gil, L. Differential response of oak and beech to late frost damage: An integrated analysis from organ to forest. Agr. For. Meteorol. 2021, 297, 108243. [Google Scholar] [CrossRef]
- Girardin, M.P.; Guo, J.; Gervais, D.; Metsaranta, J.; Campbell, E.M.; Arsenault, A.; Isaac-Renton, M.; Hogg, E.H. Cold-season freeze frequency is a pervasive driver of subcontinental forest growth. Proc. Natl. Acad. Sci. USA 2022, 119, e2117464119. [Google Scholar] [CrossRef] [PubMed]
- Chinnusamy, V.; Zhu, J.; Zhu, J.K. Cold stress regulation of gene expression in plants. Trends Plant Sci. 2007, 12, 444–451. [Google Scholar] [CrossRef]
- Vicente, E.; Didion-Gency, M.; Morcillo, L.; Morin, X.; Vilagrosa, A.; Grossiord, C. Aridity and cold temperatures drive divergent adjustments of European beech xylem anatomy, hydraulics and leaf physiological traits. Tree Physiol. 2022, 42, 1720–1735. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, S.; Arzac, A.; Cooper, D.J.; Jin, Y.; Yuan, D.; Zhu, Y.; Zhang, X.; Li, Z.; Zhang, Y.; et al. Different response of earlywood vessel features of Fraxinus mandshurica to rapid warming in warm-dry and cold-wet areas. Agric. For. Meteorol. 2021, 307, 108523. [Google Scholar] [CrossRef]
- Gambetta, G.A.; Knipfer, T.; Fricke, W.; McElrone, A.J. Aquaporins and root water uptake. In Plant Aquaporins; Signaling and Communication in Plants; Springer: Berlin/Heidelberg, Germany, 2017; pp. 133–153. [Google Scholar]
- Maruta, E.; Yazaki, K.; Ogasa, M.Y.; Taneda, H. Pit aspiration causes an apparent loss of xylem hydraulic conductivity in a subalpine fir (Abies mariesii Mast.) overwintering at the alpine timberline. Tree Physiol. 2021, 42, 1228–1238. [Google Scholar] [CrossRef]
- Sperry, J.S.; Sullivan, J.E. Xylem embolism in response to freeze-thaw cycles and water stress in ring-porous, diffuse-porous, and conifer species. Plant Physiol. 1992, 100, 605–613. [Google Scholar] [CrossRef]
- Niu, C.; Meinzer, F.C.; Hao, G.; Watling, J. Divergence in strategies for coping with winter embolism among co-occurring temperate tree species: The role of positive xylem pressure, wood type and tree stature. Funct. Ecol. 2017, 31, 1550–1560. [Google Scholar] [CrossRef]
- Zanne, A.E.; Tank, D.C.; Cornwell, W.K.; Eastman, J.M.; Smith, S.A.; FitzJohn, R.G.; McGlinn, D.J.; O’Meara, B.C.; Moles, A.T.; Reich, P.B.; et al. Three keys to the radiation of angiosperms into freezing environments. Nature 2014, 506, 89–92. [Google Scholar] [CrossRef]
- Zhang, W.; Feng, F.; Tyree, M.T. Seasonality of cavitation and frost fatigue in Acer mono Maxim. Plant Cell Environ. 2018, 41, 1278–1286. [Google Scholar] [CrossRef]
- Lintunen, A.; Lindfors, L.; Kolari, P.; Juurola, E.; Nikinmaa, E.; Holtta, T. Bursts of CO2 released during freezing offer a new perspective on avoidance of winter embolism in trees. Ann. Bot. 2014, 114, 1711–1718. [Google Scholar] [CrossRef]
- Hacke, U.G.; Sperry, J.S. Functional and ecological xylem anatomy. In Perspectives in Plant Ecology, Evolution and Systematics; Elsevier: Amsterdam, The Netherlands, 2001; Volume 4, pp. 97–115. [Google Scholar]
- Mayr, S.; Sperry, J.S. Freeze-thaw-induced embolism in Pinus contorta: Centrifuge experiments validate the ‘thaw-expansion hypothesis’ but conflict with ultrasonic emission data. New Phytol. 2010, 185, 1016–1024. [Google Scholar] [CrossRef]
- Pittermann, J.; Sperry, J.S. Analysis of freeze-thaw embolism in conifers. The interaction between cavitation pressure and tracheid size. Plant Physiol. 2006, 140, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Lintunen, A.; Salmon, Y.; Hölttä, T.; Suhonen, H. Inspection of gas bubbles in frozen Betula pendula xylem withmicro-CT: Conduit size, water status and bark permeability affect bubble characteristics. Physiol. Plant 2022, 174, e13749. [Google Scholar] [CrossRef]
- Charra-Vaskou, K.; Lintunen, A.; Améglio, T.; Badel, E.; Cochard, H.; Mayr, S.; Salmon, Y.; Suhonen, H.; van Rooij, M.; Charrier, G. Xylem embolism and bubble formation during freezing suggest complex dynamics of pressure-tension in Betula pendula stems. J. Exp. Bot. 2022, erad275. [Google Scholar]
- Lens, F.; Tixier, A.; Cochard, H.; Sperry, J.S.; Jansen, S.; Herbette, S. Embolism resistance as a key mechanism to understand adaptive plant strategies. Curr. Opin. Plant Biol. 2013, 16, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Ogasa, M.Y.; Taneda, H.; Ooeda, H.; Ohtsuka, A.; Maruta, E. Repair of severe winter xylem embolism supports summer water transport and carbon gain in flagged crowns of the subalpine conifer Abies veitchii. Tree Physiol. 2019, 39, 1725–1735. [Google Scholar] [CrossRef]
- Sperry, J.S.; Nichols, K.L.; Sullivan, J.E.; Eastlack, S.E. Xylem embolism in ring-porous, diffuse-porous, and coniferous trees of northern Utah and interior Alaska. Ecology 1994, 75, 1736–1752. [Google Scholar] [CrossRef]
- Nardini, A.; Lo Gullo, M.A.; Salleo, S. Refilling embolized xylem conduits: Is it a matter of phloem unloading? Plant Sci. 2011, 180, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Bowling, D.R.; Logan, B.A.; Hufkens, K.; Aubrecht, D.M.; Richardson, A.D.; Burns, S.P.; Anderegg, W.R.; Blanken, P.D.; Eiriksson, D.P. Limitations to winter and spring photosynthesis of a Rocky Mountain subalpine forest. Agric. For. Meteorol. 2018, 252, 241–255. [Google Scholar] [CrossRef]
- Morris, H.; Plavcova, L.; Gorai, M.; Klepsch, M.M.; Kotowska, M.; Jochen Schenk, H.; Jansen, S. Vessel-associated cells in angiosperm xylem: Highly specialized living cells at the symplast-apoplast boundary. Am. J. Bot. 2018, 105, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, D.; Willick, I.R.; Kasuga, J.; Livingston III, D.P. Responses of the plant cell wall to sub-zero temperatures: A brief update. Plant Cell Physiol. 2021, 62, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Pearce, R. Plant freezing and damage. Ann. Bot. 2001, 87, 417–424. [Google Scholar] [CrossRef]
- Arias, N.S.; Scholz, F.G.; Goldstein, G.; Bucci, S.J. The cost of avoiding freezing in stems: Trade-off between xylem resistance to cavitation and supercooling capacity in woody plants. Tree Physiol. 2017, 37, 1251–1262. [Google Scholar] [CrossRef]
- Mayr, S.; Ameglio, T. Freezing stress in tree xylem. Prog. Bot. 2016, 77, 381–414. [Google Scholar] [CrossRef]
- Feng, F.; Ding, F.; Tyree, M.T. Investigations concerning cavitation and frost fatigue in clonal 84K poplar using high-resolution cavitron measurements. Plant Physiol. 2015, 168, 144–155. [Google Scholar] [CrossRef]
- Mayr, S.; Schmid, P.; Beikircher, B.; Feng, F.; Badel, E. Die hard: Timberline conifers survive annual winter embolism. New Phytol. 2020, 226, 13–20. [Google Scholar] [CrossRef]
- Caselles, V.; Casadesus, A.; Munne-Bosch, S. A dual role for abscisic acid integrating the cold stress response at the whole-plant level in Iris pseudacorus L. growing in a natural wetland. Front. Plant Sci. 2021, 12, 722525. [Google Scholar] [CrossRef]
- Hu, Y.; Jiang, Y.; Han, X.; Wang, H.; Pan, J.; Yu, D. Jasmonate regulates leaf senescence and tolerance to cold stress: Crosstalk with other phytohormones. J. Exp. Bot. 2017, 68, 1361–1369. [Google Scholar] [CrossRef]
- Ployet, R.; Soler, M.; Carocha, V.; Ladouce, N.; Alves, A.; Rodrigues, J.C.; Harvengt, L.; Marque, C.; Teulieres, C.; Grima-Pettenati, J.; et al. Long cold exposure induces transcriptional and biochemical remodelling of xylem secondary cell wall in Eucalyptus. Tree Physiol. 2018, 38, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.; Nabais, C.; Vieira, J.; Rossi, S.; Campelo, F. Plastic response of tracheids in Pinus pinaster in a water-limited environment: Adjusting lumen size instead of wall thickness. PLoS ONE 2015, 10, e0136305. [Google Scholar] [CrossRef]
- Savage, J.A.; Kiecker, T.; McMann, N.; Park, D.; Rothendler, M.; Mosher, K. Leaf out time correlates with wood anatomy across large geographic scales and within local communities. New Phytol. 2022, 235, 953–964. [Google Scholar] [CrossRef]
- Davis, S.D.; Sperry, J.S.; Hacke, U.G. The relationship between xylem conduit diameter and cavitation caused by freezing. Am. J. Bot. 1999, 86, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Choat, B.; Medek, D.E.; Stuart, S.A.; Pasquet-Kok, J.; Egerton, J.J.; Salari, H.; Sack, L.; Ball, M.C. Xylem traits mediate a trade-off between resistance to freeze–thaw-induced embolism and photosynthetic capacity in overwintering evergreens. New Phytol. 2011, 191, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Wang, L.; Wan, X. Frost fatigue and its spring recovery of xylem conduits in ring-porous, diffuse-porous, and coniferous species in situ. Plant Physiol. Biochem. 2020, 146, 177–186. [Google Scholar] [CrossRef]
- Tedla, B.; Dang, Q.-L.; Inoue, S. Freeze-thaw events delay spring budburst and leaf expansion while longer photoperiods have opposite effect under different [CO2] in white birch: Advance it under elevated but delay it under ambient [CO2]. Environ. Exp. Bot. 2020, 173, 103982. [Google Scholar] [CrossRef]
- Clough, B.J.; Curzon, M.T.; Domke, G.M.; Russell, M.B.; Woodall, C.W. Climate-driven trends in stem wood density of tree species in the eastern United States: Ecological impact and implications for national forest carbon assessments. Glob. Ecol. Biogeogr. 2017, 26, 1153–1164. [Google Scholar] [CrossRef]
- Cochard, H.; Tyree, M.T. Xylem dysfunction in Quercus: Vessel sizes, tyloses, cavitation and seasonal changes in embolism. Tree Physiol. 1990, 6, 393–407. [Google Scholar] [CrossRef]
- Jansen, S.; Schuldt, B.; Choat, B. Current controversies and challenges in applying plant hydraulic techniques: International Workshop on Plant Hydraulic Techniques, Ulm University, Germany, September 2014. New Phytol 2015, 205, 961–964. [Google Scholar] [CrossRef]
- Ahmed, S.; Sarker, S.K.; Friess, D.A.; Kamruzzam, M.; Jacobs, M.; Islam, M.A.; Alam, M.A.; Suvo, M.J.; Sani, M.N.H.; Dey, T.; et al. Salinity reduces site quality and mangrove forest functions. From monitoring to understanding. Sci. Total Environ. 2022, 853, 158662. [Google Scholar] [CrossRef] [PubMed]
- Perri, S.; Detto, M.; Porporato, A.; Molini, A. Salinity-induced limits to mangrove canopy height. Glob. Ecol. Biogeogr. 2023, 32, 1561–1574. [Google Scholar] [CrossRef]
- Qie, Y.; Jiang, L.; Lv, G.; Yang, X.; Wang, H.; Teng, D. Response of plant leaf functional traits to soil aridity and salinity in temperate desert ecosystem. Ecol. Environ. Sci. 2018, 27, 2000–2010. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef]
- Guo, R.; Yang, Z.; Li, F.; Yan, C.; Zhong, X.; Liu, Q.; Xia, X.; Li, H.; Zhao, L. Comparative metabolic responses and adaptive strategies of wheat (Triticum aestivum) to salt and alkali stress. BMC Plant Biol. 2015, 15, 170. [Google Scholar] [CrossRef] [PubMed]
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant Physiological Ecology; Springer: Berlin/Heidelberg, Germany, 2008; Volume 2. [Google Scholar]
- Liao, Q.; Gu, S.; Kang, S.; Du, T.; Tong, L.; Wood, J.D.; Ding, R. Mild water and salt stress improve water use efficiency by decreasing stomatal conductance via osmotic adjustment in field maize. Sci. Total Environ. 2022, 805, 150364. [Google Scholar] [CrossRef]
- Tavakkoli, E.; Fatehi, F.; Coventry, S.; Rengasamy, P.; McDonald, G.K. Additive effects of Na+ and Cl– ions on barley growth under salinity stress. J. Exp. Bot. 2011, 62, 2189–2203. [Google Scholar] [CrossRef]
- Vysotskaya, L.; Hedley, P.E.; Sharipova, G.; Veselov, D.; Kudoyarova, G.; Morris, J.; Jones, H.G. Effect of salinity on water relations of wild barley plants differing in salt tolerance. AoB Plants 2010, 2010, plq006. [Google Scholar] [CrossRef]
- Acosta-Motos, J.; Ortuño, M.; Bernal-Vicente, A.; Diaz-Vivancos, P.; Sanchez-Blanco, M.; Hernandez, J. Plant responses to salt stress: Adaptive mechanisms. Agronomy 2017, 7, 18. [Google Scholar] [CrossRef]
- Navarro, A.; Bañon, S.; Olmos, E.; Sánchez-Blanco, M.d.J. Effects of sodium chloride on water potential components, hydraulic conductivity, gas exchange and leaf ultrastructure of Arbutus unedo plants. Plant Sci. 2007, 172, 473–480. [Google Scholar] [CrossRef]
- Baraldi, R.; Przybysz, A.; Facini, O.; Pierdonà, L.; Carriero, G.; Bertazza, G.; Neri, L. Impact of drought and salinity on sweetgum tree (Liquidambar styraciflua L.): Understanding tree ecophysiological responses in the urban context. Forests 2019, 10, 1032. [Google Scholar] [CrossRef]
- Zhang, H.; Li, X.; Wang, W.; Pivovaroff, A.L.; Li, W.; Zhang, P.; Ward, N.D.; Myers-Pigg, A.; Adams, H.D.; Leff, R.; et al. Seawater exposure causes hydraulic damage in dying Sitka-spruce trees. Plant Physiol. 2021, 187, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Ewers, F.W.; Lopez-Portillo, J.; Angeles, G.; Fisher, J.B. Hydraulic conductivity and embolism in the mangrove tree Laguncularia racemosa. Tree Physiol. 2004, 24, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, N.; Wang, H.; Ren, J.; Yao, Y. Hydraulic characteristics and carbon metabolism of Lycium chinense Miller and Tamarix chinensis Lour.under saline-alkali stress. Plant Sci. J. 2017, 35, 865–873. [Google Scholar] [CrossRef]
- Feng, S.; Ren, L.; Sun, H.; Qiao, K.; Liu, S.; Zhou, A. Morphological and physiological responses of two willow species from different habitats to salt stress. Sci. Rep. 2020, 10, 18228. [Google Scholar] [CrossRef]
- Regni, L.; Del Pino, A.M.; Mousavi, S.; Palmerini, C.A.; Baldoni, L.; Mariotti, R.; Mairech, H.; Gardi, T.; D’Amato, R.; Proietti, P. Behavior of four olive cultivars during salt stress. Front. Plant Sci. 2019, 10, 867. [Google Scholar] [CrossRef]
- Abbruzzese, G.; Beritognolo, I.; Muleo, R.; Piazzai, M.; Sabatti, M.; Mugnozza, G.S.; Kuzminsky, E. Leaf morphological plasticity and stomatal conductance in three Populus alba L. genotypes subjected to salt stress. Environ. Exp. Bot. 2009, 66, 381–388. [Google Scholar] [CrossRef]
- Álvarez, S.; Sanchez-Blanco, M. Long-term effect of salinity on plant quality, water relations, photosynthetic parameters and ion distribution in Callistemon citrinus. Plant Biol. 2014, 16, 757–764. [Google Scholar] [CrossRef]
- Cao, B.; Li, N.; Xu, K. Crosstalk of phenylpropanoid biosynthesis with hormone signaling in Chinese cabbage is key to counteracting salt stress. Environ. Exp. Bot. 2020, 179, 104209. [Google Scholar] [CrossRef]
- Gómez-Bellot, M.J.; Alvarez, S.; Castillo, M.; Bañón, S.; Ortuño, M.F.; Sánchez-Blanco, M.J. Water relations, nutrient content and developmental responses of Euonymus plants irrigated with water of different degrees of salinity and quality. J. Plant Res. 2013, 126, 567–576. [Google Scholar] [CrossRef]
- Franco, J.; Bañón, S.; Vicente, M.; Miralles, J.; Martínez-Sánchez, J. Root development in horticultural plants grown under abiotic stress conditions–a review. J. Hortic. Sci. Biotechnol. 2011, 86, 543–556. [Google Scholar] [CrossRef]
- Croser, C.; Renault, S.; Franklin, J.; Zwiazek, J. The effect of salinity on the emergence and seedling growth of Picea mariana, Picea glauca, and Pinus banksiana. Environ. Pollut. 2001, 115, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Stiller, V. Soil salinity and drought alter wood density and vulnerability to xylem cavitation of baldcypress (Taxodium distichum (L.) Rich.) seedlings. Environ. Exp. Bot. 2009, 67, 164–171. [Google Scholar] [CrossRef]
- Escalante-Perez, M.; Lautner, S.; Nehls, U.; Selle, A.; Teuber, M.; Schnitzler, J.P.; Teichmann, T.; Fayyaz, P.; Hartung, W.; Polle, A.; et al. Salt stress affects xylem differentiation of grey poplar (Populus x canescens). Planta 2009, 229, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Eckert, C.; Sharmin, S.; Kogel, A.; Yu, D.; Kins, L.; Strijkstra, G.-J.; Polle, A. What makes the wood? Exploring the molecular mechanisms of xylem acclimation in hardwoods to an ever-changing environment. Forests 2019, 10, 358. [Google Scholar] [CrossRef]
- Junghans, U.; Polle, A.; Düchting, P.; Weiler, E.; Kuhlman, B.; Gruber, F.; Teichmann, T. Adaptation to high salinity in poplar involves changes in xylem anatomy and auxin physiology. Plant Cell Environ. 2006, 29, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Aloni, R. The induction of vascular tissues by auxin. In Plant Hormones; Springer: Berlin/Heidelberg, Germany, 2010; pp. 485–518. [Google Scholar]
- Hori, C.; Yu, X.; Mortimer, J.C.; Sano, R.; Matsumoto, T.; Kikuchi, J.; Demura, T.; Ohtani, M. Impact of abiotic stress on the regulation of cell wall biosynthesis in Populus trichocarpa. Plant Biotechnol. 2020, 37, 273–283. [Google Scholar] [CrossRef]
- Janz, D.; Lautner, S.; Wildhagen, H.; Behnke, K.; Schnitzler, J.P.; Rennenberg, H.; Fromm, J.; Polle, A. Salt stress induces the formation of a novel type of ‘pressure wood’in two Populus species. New Phytol. 2012, 194, 129–141. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, W.; Long, S.; Zhao, C. Maintenance of cell wall integrity under high salinity. Int. J. Mol. Sci. 2021, 22, 3260. [Google Scholar] [CrossRef]
- Kankia, H.; Abdulhamid, Y. Determination of accumulated heavy metals in benthic invertebrates found in Ajiwa Dam, Katsina State, Northern Nigeria. Arch. Appl. Sci. Res. 2014, 6, 80–87. [Google Scholar]
- Sarwar, N.; Imran, M.; Shaheen, M.R.; Ishaque, W.; Kamran, M.A.; Matloob, A.; Rehim, A.; Hussain, S. Phytoremediation strategies for soils contaminated with heavy metals: Modifications and future perspectives. Chemosphere 2017, 171, 710–721. [Google Scholar] [CrossRef]
- Trammell, T.L.E.; Schneid, B.P.; Carreiro, M.M. Forest soils adjacent to urban interstates: Soil physical and chemical properties, heavy metals, disturbance legacies, and relationships with woody vegetation. Urban. Ecosyst. 2011, 14, 525–552. [Google Scholar] [CrossRef]
- Barker, A.V.; Pilbeam, D.J. Handbook of Plant Nutrition; CRC press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. In Molecular, Clinical and Environmental Toxicology. Experientia Supplementum; Springer: Berlin/Heidelberg, Germany, 2012; pp. 133–164. [Google Scholar] [CrossRef]
- Ke, W.; Wang, W.; Yang, Y. The characteristic of some micrometal elements accumulation in elsholtizia spledens population at tongllushan copper mine area of Daye in Hubei province. Environ. Sci. Technol. 2000, S1, 33–35. [Google Scholar] [CrossRef]
- Ghori, N.-H.; Ghori, T.; Hayat, M.; Imadi, S.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Zhang, J.; Shu, W. Mechanisms of heavy metal cadmium tolerance in plants. J. Plant Physiol. Mol. Biol. 2006, 32, 1–8. [Google Scholar]
- Houda, Z.; Bejaoui, Z.; Albouchi, A.; Gupta, D.K.; Corpas, F.J. Comparative study of plant growth of two poplar tree species irrigated with treated wastewater, with particular reference to accumulation of heavy metals (Cd, Pb, As, and Ni). Environ. Monit. Assess. 2016, 188, 99. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Luo, Y.; Xing, X.; Christie, P. EDTA-enhanced phytoremediation of heavy metal contaminated soil with Indian mustard and associated potential leaching risk. Agric. Ecosyst. Environ. 2004, 102, 307–318. [Google Scholar] [CrossRef]
- Wang, X.; Wang, W.; Yang, L.; Jin, L.; Song, Y.; Jiang, S.; Qin, L. Transport pathways of cadmium (Cd) and its regulatory mechanisms in plant. Acta Ecol. Sin. 2015, 35, 7921–7929. [Google Scholar]
- Álvarez-Fernández, A.; Díaz-Benito, P.; Abadía, A.; López-Millán, A.-F.; Abadía, J. Metal species involved in long distance metal transport in plants. Front. Plant Sci. 2014, 5, 105. [Google Scholar] [CrossRef]
- Page, V.; Feller, U. Heavy metals in crop plants: Transport and redistribution processes on the whole plant level. Agronomy 2015, 5, 447–463. [Google Scholar] [CrossRef]
- Li, S.; (Department of Environmental Science and Ecology, School of Environmental Science and Engineering, Shaanxi University of Science and Technology, Xi’an, China); Wang, X.; (Department of Environmental Science and Ecology, School of Environmental Science and Engineering, Shaanxi University of Science and Technology, Xi’an, China). Unpublished work. 2022.
- Kumar, S.; Shah, S.H.; Vimala, Y.; Jatav, H.S.; Ahmad, P.; Chen, Y.; Siddique, K.H.M. Abscisic acid: Metabolism, transport, crosstalk with other plant growth regulators, and its role in heavy metal stress mitigation. Front. Plant Sci. 2022, 13, 972856. [Google Scholar] [CrossRef] [PubMed]
- Sridharamurthy, M.; Kovach, A.; Zhao, Y.; Zhu, J.-K.; Xu, H.E.; Swaminathan, K.; Melcher, K. H2O2 inhibits ABA-signaling protein phosphatase HAB1. PLoS ONE 2014, 9, e113643. [Google Scholar] [CrossRef] [PubMed]
- Jentschke, G.; Godbold, D. Metal toxicity and ectomycorrhizas. Physiol. Plant 2000, 109, 107–116. [Google Scholar] [CrossRef]
- de Silva, N.D.; Cholewa, E.; Ryser, P. Effects of combined drought and heavy metal stresses on xylem structure and hydraulic conductivity in red maple (Acer rubrum L.). J. Exp. Bot. 2012, 63, 5957–5966. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tan, X.; Zhou, Y.; Liu, N. Effects of a heavy metal (cadmium) on the responses of subtropical coastal tree species to drought stress. Environ. Sci. Pollut. Res. 2022, 30, 12682–12694. [Google Scholar] [CrossRef]
- Przedpelska-Wasowicz, E.M.; Wierzbicka, M. Gating of aquaporins by heavy metals in Allium cepa L. epidermal cells. Protoplasma 2011, 248, 663–671. [Google Scholar] [CrossRef]
- Rucińska-Sobkowiak, R.; Nowaczyk, G.; Krzesłowska, M.; Rabęda, I.; Jurga, S. Water status and water diffusion transport in lupine roots exposed to lead. Environ. Exp. Bot. 2013, 87, 100–109. [Google Scholar] [CrossRef]
- Tanentzap, F.M.; Ryser, P. Decreased resistance to embolism in red maple (Acer rubrum L.) saplings within a heavy metal contaminated region. Environ. Exp. Bot. 2015, 109, 40–44. [Google Scholar] [CrossRef]
- Staňová, A.; Ďurišová, E.; Banásová, V.; Gurinová, E.; Nadubinská, M.; Kenderešová, L.; Ovečka, M.; Čiamporová, M. Root system morphology and primary root anatomy in natural non-metallicolous and metallicolous populations of three Arabidopsis species differing in heavy metal tolerance. Biologia 2012, 67, 505–516. [Google Scholar] [CrossRef]
- Gomes, M.P.; Nogueira, M.d.O.G.; Castro, E.M.d.; Soares, Â.M. Ecophysiological and anatomical changes due to uptake and accumulation of heavy metal in Brachiaria decumbens. Sci. Agric. 2011, 68, 566–573. [Google Scholar] [CrossRef]
- Li, J.; Yu, J.; Du, D.; Liu, J.; Lu, H.; Yan, C. Analysis of anatomical changes and cadmium distribution in Aegiceras corniculatum (L.) Blanco roots under cadmium stress. Mar. Pollut. Bull. 2019, 149, 110536. [Google Scholar] [CrossRef] [PubMed]
- Bazihizina, N.; Taiti, C.; Marti, L.; Rodrigo-Moreno, A.; Spinelli, F.; Giordano, C.; Caparrotta, S.; Gori, M.; Azzarello, E.; Mancuso, S. Zn2+-induced changes at the root level account for the increased tolerance of acclimated tobacco plants. J. Exp. Bot. 2014, 65, 4931–4942. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.A.; Vassileva, V.; Petrov, P.; Popova, L.P. Cadmium-induced structural disturbances in Pisum sativum leaves are alleviated by nitric oxide. Turkish J. Bot. 2013, 37, 698–707. [Google Scholar] [CrossRef]
- Sagardoy, R.; Vázquez, S.; Florez-Sarasa, I.; Albacete, A.; Ribas-Carbó, M.; Flexas, J.; Abadía, J.; Morales, F. Stomatal and mesophyll conductances to CO2 are the main limitations to photosynthesis in sugar beet (Beta vulgaris) plants grown with excess zinc. New Phytol. 2010, 187, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Bora, M.S.; Sarma, K.P. Anatomical and ultrastructural alterations in Ceratopteris pteridoides under cadmium stress: A mechanism of cadmium tolerance. Ecotoxicol. Environ. Saf. 2021, 218, 112285. [Google Scholar] [CrossRef]
- Vaculik, M.; Konlechner, C.; Langer, I.; Adlassnig, W.; Puschenreiter, M.; Lux, A.; Hauser, M.T. Root anatomy and element distribution vary between two Salix caprea isolates with different Cd accumulation capacities. Environ. Pollut. 2012, 163, 117–126. [Google Scholar] [CrossRef]
- Górska, M.; Roszyk, E. Wood structure of Scots pine (L.) growing on flotation tailings. Folia For. Pol. 2019, 61, 112–122. [Google Scholar] [CrossRef]
- Mrak, T.; Grašič, B.; Prislan, P.; Gričar, J.; Laznik, Ž.; Voglar, G.E. Soil contamination with potentially toxic elements and root herbivory: Effects on root surface area and stem secondary xylem of young common beech (Fagus sylvatica L.). Acta Physiol. Plant 2022, 45, 23. [Google Scholar] [CrossRef]
- Li, S.; (Department of Environmental Science and Ecology, School of Environmental Science and Engineering, Shaanxi University of Science and Technology, Xi’an, China); Sun, X.; (Department of Environmental Science and Ecology, School of Environmental Science and Engineering, Shaanxi University of Science and Technology, Xi’an, China); Zhu, X.; (Department of Environmental Science and Ecology, School of Environmental Science and Engineering, Shaanxi University of Science and Technology, Xi’an, China). Unpublished work. 2021.
- Rucińska-Sobkowiak, R. Water relations in plants subjected to heavy metal stresses. Acta Physiol. Plant 2016, 38, 257. [Google Scholar] [CrossRef]
- Solanki, R.; Dhankhar, R. Biochemical changes and adaptive strategies of plants under heavy metal stress. Biologia 2011, 66, 195–204. [Google Scholar] [CrossRef]
- Jia, H.; Wang, X.; Wei, T.; Wang, M.; Liu, X.; Hua, L.; Ren, X.; Guo, J.; Li, J. Exogenous salicylic acid regulates cell wall polysaccharides synthesis and pectin methylation to reduce Cd accumulation of tomato. Ecotoxicol. Environ. Saf. 2021, 207, 111550. [Google Scholar] [CrossRef] [PubMed]
- Krzeslowska, M.; Rabeda, I.; Basinska, A.; Lewandowski, M.; Mellerowicz, E.J.; Napieralska, A.; Samardakiewicz, S.; Wozny, A. Pectinous cell wall thickenings formation—A common defense strategy of plants to cope with Pb. Environ. Pollut. 2016, 214, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Li, K.; Zheng, W.; Zhang, H.; Cao, X.; Lan, Y.; Yang, C.; Li, C. Characterization of early transcriptional responses to cadmium in the root and leaf of Cd-resistant Salix matsudana Koidz. BMC Genomics 2015, 16, 705. [Google Scholar] [CrossRef]
- da Silva Cunha, L.F.; de Oliveira, V.P.; do Nascimento, A.W.S.; da Silva, B.R.S.; Batista, B.L.; Alsahli, A.A.; Lobato, A.K.d.S. Leaf application of 24-epibrassinolide mitigates cadmium toxicity in young Eucalyptus urophylla plants by modulating leaf anatomy and gas exchange. Physiol. Plantarum 2021, 173, 67–87. [Google Scholar] [CrossRef]
- Hrkic-Ilic, Z.; Borisev, M.; Zoric, L.; Arsenov, D.; Lukovic, J. Assessment of differences in anatomical and hydraulic properties of the root and xylem of three willow (Salix L.) clones during phytostabilization after exposure to elevated cadmium. Arch. Biol. Sci. 2022, 74, 169–180. [Google Scholar] [CrossRef]
- Sruthi, P.; Puthur, J.T. Characterization of physiochemical and anatomical features associated with enhanced phytostabilization of copper in Bruguiera cylindrica (L.) Blume. Int. J. Phytorem 2019, 21, 1423–1441. [Google Scholar] [CrossRef] [PubMed]
- Marques, D.M.; Veroneze Júnior, V.; da Silva, A.B.; Mantovani, J.R.; Magalhães, P.C.; de Souza, T.C. Copper toxicity on photosynthetic responses and root morphology of Hymenaea courbaril L.(Caesalpinioideae). Water Air Soil. Pollut. 2018, 229, 138. [Google Scholar] [CrossRef]
- Ribeiro, A.T.; de Oliveira, V.P.; de Oliveira Barros Junior, U.; da Silva, B.R.S.; Batista, B.L.; da Silva Lobato, A.K. 24-Epibrassinolide mitigates nickel toxicity in young Eucalyptus urophylla ST Blake plants: Nutritional, physiological, biochemical, anatomical and morphological responses. Ann. For. Sci. 2020, 77, 5. [Google Scholar] [CrossRef]
- Iqbal, T.; Shah, S.K.; Ullah, F.; Mehmood, S.; Zeb, M.A. Analysis of deformable distortion in the architecture of leaf xylary vessel elements of Carthamus oxycantha caused by heavy metals stress using image registration. Microsc. Res. Tech. 2020, 83, 843–849. [Google Scholar] [CrossRef]
- Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Marchin, R.M.; Backes, D.; Ossola, A.; Leishman, M.R.; Tjoelker, M.G.; Ellsworth, D.S. Extreme heat increases stomatal conductance and drought-induced mortality risk in vulnerable plant species. Glob. Chang. Biol. 2022, 28, 1133–1146. [Google Scholar] [CrossRef] [PubMed]
- Zandalinas, S.I.; Rivero, R.M.; Martínez, V.; Gómez-Cadenas, A.; Arbona, V. Tolerance of citrus plants to the combination of high temperatures and drought is associated to the increase in transpiration modulated by a reduction in abscisic acid levels. BMC Plant Biol. 2016, 16, 105. [Google Scholar] [CrossRef]
- Zandalinas, S.I.; Balfagón, D.; Arbona, V.; Gómez-Cadenas, A.; Inupakutika, M.A.; Mittler, R. ABA is required for the accumulation of APX1 and MBF1c during a combination of water deficit and heat stress. J. Exp. Bot. 2016, 67, 5381–5390. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Zhou, M.; Shabala, S. How does stomatal density and residual transpiration contribute to osmotic stress tolerance? Plants 2023, 12, 494. [Google Scholar] [CrossRef] [PubMed]
- Alabdallah, N.M.; Alluqmani, S.M. The synthesis of polysaccharide crude nanoparticles extracts from taif rose petals and its effect on eggplant seedlings under drought and salt stress. J. King Saud. Univ. Sci. 2022, 34, 102055. [Google Scholar] [CrossRef]
- Zhang, M.; Luo, Y.; Yan, Z.; Chen, J.; Eziz, A.; Li, K.; Han, W. Resorptions of 10 mineral elements in leaves of desert shrubs and their contrasting responses to aridity. J. Plant Ecol. 2019, 12, 358–366. [Google Scholar] [CrossRef]
- Drenovsky, R.E.; Richards, J. Low leaf N and P resorption contributes to nutrient limitation in two desert shrubs. Plant Ecol. 2006, 183, 305–314. [Google Scholar] [CrossRef][Green Version]
- Wang, J.; Wang, H.; Chen, J.; Zhang, S.; Xu, J.; Han, X.; Feng, Y.; Chen, Y.; Zhang, X.; Dong, G.; et al. Xylem development, cadmium bioconcentration, and antioxidant defense in Populus × euramericana stems under combined conditions of nitrogen and cadmium. Environ. Exp. Bot. 2019, 164, 1–9. [Google Scholar] [CrossRef]
- Sruthi, P.; Puthur, J.T. Cadmium stress alleviation potential of Bruguiera cylindrica (L.) Blume enhances in combination with NaCl. Biorem J. 2021, 26, 89–112. [Google Scholar] [CrossRef]
- Hao, L.; Chen, L.; Zhu, P.; Zhang, J.; Zhang, D.; Xiao, J.; Xu, Z.; Zhang, L.; Liu, Y.; Li, H.; et al. Sex-specific responses of Populus deltoides to interaction of cadmium and salinity in root systems. Ecotoxicol. Environ. Saf. 2020, 195, 110437. [Google Scholar] [CrossRef]
- Lu, Y.W.; Miao, X.L.; Song, Q.Y.; Peng, S.M.; Duan, B.L. Morphological and ecophysiological plasticity in dioecious plant Populus tomentosa under drought and alkaline stresses. Photosynthetica 2018, 56, 1353–1364. [Google Scholar] [CrossRef]
- Kilpeläinen, A.; Gerendiain, A.Z.; Luostarinen, K.; Peltola, H.; Kellomäki, S. Elevated temperature and CO2 concentration effects on xylem anatomy of Scots pine. Tree Physiol. 2007, 27, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Austen, K.; MacLean, J.; Balanzategui, D.; Hölker, F. Microplastic inclusion in birch tree roots. Sci. Total Environ. 2022, 808, 152085. [Google Scholar] [CrossRef]
- Murazzi, M.E.; Cherubini, P.; Brunner, I.; Kägi, R.; Saurer, M.; Ballikaya, P.; Hagedorn, F.; Al Sid Cheikh, M.; Onandia, G.; Gessler, A. Can forest trees take up and transport nanoplastics? iForest 2022, 15, 128–132. [Google Scholar] [CrossRef]
- Wang, P.; Lombi, E.; Sun, S.; Scheckel, K.G.; Malysheva, A.; McKenna, B.A.; Menzies, N.W.; Zhao, F.-J.; Kopittke, P.M. Characterizing the uptake, accumulation and toxicity of silver sulfide nanoparticles in plants. Environ. Sci. Nano 2017, 4, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chang, X.; Zhang, J.; Wang, Y.; Zhong, R.; Wang, L.; Wei, J.; Wang, Y. Uptake and distribution of microplastics of different particle sizes in maize (Zea mays) seedling roots. Chemosphere 2023, 313, 137491. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).