Understanding Soil Respiration Dynamics in Temperate Forests in Northwestern Mexico
Abstract
1. Introduction
2. Materials and Methods
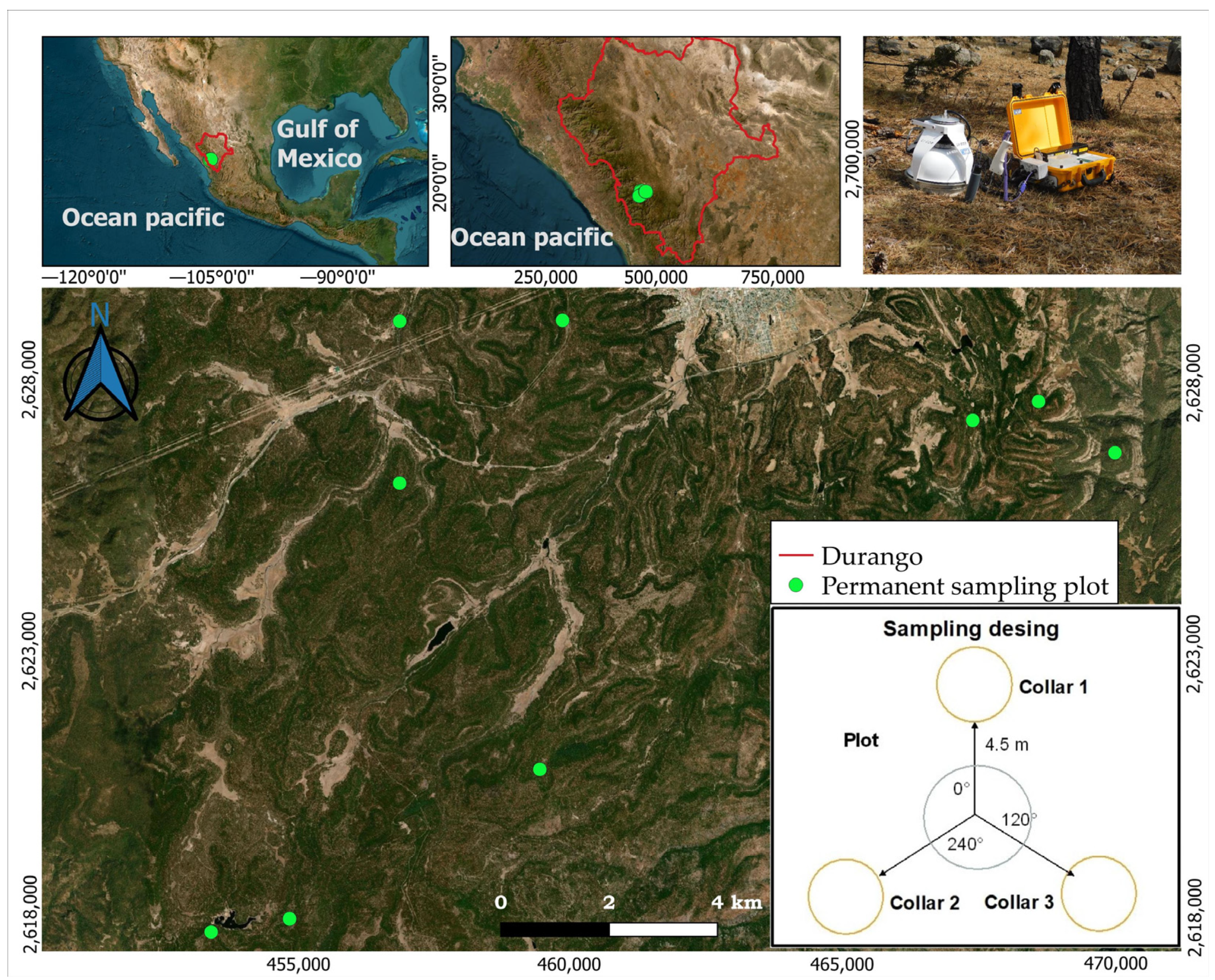
2.1. Site Description
2.2. Inventory Data and Sampling Design
2.3. Soil CO2 Flux Measurements
2.4. Diversity Metrics and Aboveground Biomass Estimations
2.5. Statistical Analyses
3. Results
3.1. Diversity Metrics and Aboveground Biomass
3.2. Monthly CO2 Efflux
3.3. Soil Respiration by Forest Type
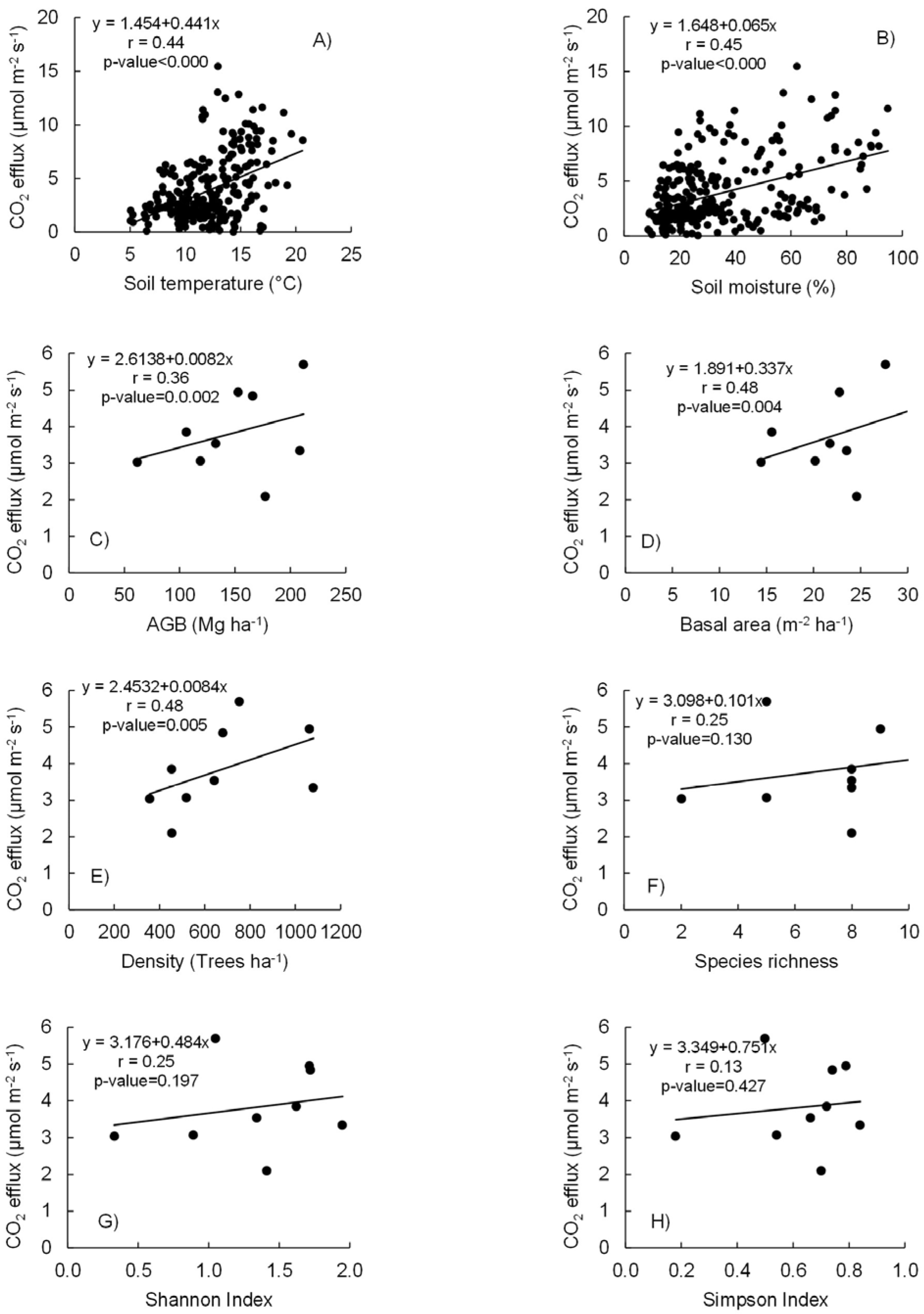
3.4. Relationships of Soil CO2 Fluxes with Forest Diversity Metrics, Soil Temperature, Soil Moisture, and Forest Stand Variables
3.5. Model for Estimating Soil Respiration
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lal, R. Sequestration of atmospheric CO2 in global carbon pools. Energy Environ. Sci. 2008, 1, 86–100. [Google Scholar] [CrossRef]
- Nielsen, U.N.; Ayres, E.; Wall, D.H.; Bardgett, R.D. Soil biodiversity and carbon cycling: A review and synthesis of studies examining diversity-function relationships. Eur. J. Soil Sci. 2011, 62, 105–116. [Google Scholar] [CrossRef]
- Darenova, E.; Čater, M. Effect of spatial scale and harvest on heterogeneity of forest floor CO2 efflux in a sessile oak forest. CATENA 2020, 188, 104455. [Google Scholar] [CrossRef]
- Yan, W.D.; Xu, W.M.; Chen, X.Y.; Tian, D.L.; Peng, Y.Y.; Zhen, W.; Zhang, C.; Xu, J. Soil CO2 flux in different types of forests under a subtropical microclimatic environment. Pedosphere 2014, 24, 243–250. [Google Scholar] [CrossRef]
- Finderup, N.T.; Rand, R.N.; Michelsen, A. Increased CO2 efflux due to long-term experimental summer warming and litter input in subarctic tundra—CO2 fluxes at snowmelt, in growing season, fall and winter. Plant Soil 2019, 444, 365–382. [Google Scholar] [CrossRef]
- Chase, J.M.; Leibold, M.A. Spatial scale dictates the productivity-biodiversity relationship. Nature 2002, 416, 427–430. [Google Scholar] [CrossRef]
- Bohdalková, E.; Toszogyova, A.; Šímová, I.; Storch, D. Universality in biodiversity patterns: Variation in species–temperature and species–productivity relationships reveal a prominent role of productivity in diversity gradients. Ecography 2021, 44, 1366–1378. [Google Scholar] [CrossRef]
- Van Ruijven, J.; Berendse, F. Positive effects of plant species diversity on productivity in the absence of legumes. Ecol. Lett. 2003, 6, 170–175. [Google Scholar] [CrossRef]
- Lellei-Kovács, E.; Kovács-Láng, E.; Botta-Dukát, Z.; Kalapos, T.; Emmett, B.; Beier, C. Thresholds and interactive effects of soil moisture on the temperature response of soil respiration. Eur. J. Soil Biol. 2011, 47, 247–255. [Google Scholar] [CrossRef]
- Smith, K.; Ball, T.; Conen, F.; Dobbie, K.; Massheder, J.; Rey, A. Exchange of greenhouse gases between soil and atmosphere: Interactions of soil physical factors and biological processes. Eur. J. Soil Sci. 2003, 54, 779–791. [Google Scholar] [CrossRef]
- Luo, Y.; Zhou, X. Soil Respiration and the Environment; Elsevier: Berkeley, CA, USA, 2006. [Google Scholar] [CrossRef]
- Adachi, M.; Ito, A.; Yonemura, S.; Takeuchi, W. Estimation of global soil respiration by accounting for land-use changes derived from remote sensing data. J. Environ. Manag. 2017, 200, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Acosta, M.; Darenova, E.; Krupková, L.; Pavelka, M. Seasonal and inter-annual variability of soil CO2 efflux in a Norway spruce forest over an eight-year study. Agric. For. Meteorol. 2018, 256–257, 93–103. [Google Scholar] [CrossRef]
- De Carlo, N.D.; Oelbermann, M.; Gordon, A.M. Carbon dioxide emissions: Spatiotemporal variation in a young and mature riparian forest. Ecol. Eng. 2019, 138, 353–361. [Google Scholar] [CrossRef]
- Jílková, V. Soil respiration in temperate forests is increased by a shift from coniferous to deciduous trees but not by an increase in temperature. Appl. Soil Ecol. 2020, 154, 103635. [Google Scholar] [CrossRef]
- Merino, A.; Edeso, J.M.; González, M.J.; Marauri, P. Soil properties in a hilly area following different harvesting management practices. For. Ecol. Manag. 1997, 103, 235–246. [Google Scholar] [CrossRef]
- Challenger, A. Use and Conservation of Mexico’s Terrestrial Ecosystems: Past, Present and Future; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad/Instituto de Biología, UNAM-Agrupación Sierra Madre: Mexico City, Mexico, 1998. [Google Scholar]
- García, E. Modificaciones al Régimen de Clasificación Climática de Köppen; Instituto de Geografía, UNAM: Mexico City, Mexico, 1988. [Google Scholar]
- Instituto Nacional de Estadística Geográfica e Informática (INEGI). Uso del Suelo y Vegetación Escala 1:250 000 Serie V, Información Vectorial; INEGI: Mexico City, Mexico, 2012.
- Borken, W.; Xu, Y.J.; Davidson, E.A.; Beese, F. Site and temporal variation of soil respiration in European beech, Norway spruce, and Scots pine forests. Glob. Change Biol. 2002, 8, 1205–1216. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Vargas-Larreta, B.; López, S.C.; Corral-Rivas, J.J.; López, M.J.; Aguirre, C.C.; Álvarez-González, J.G. Allometric equations for estimating biomass and carbon stocks in the temperate forests of North-Western Mexico. Forests 2017, 8, 269. [Google Scholar] [CrossRef]
- Hibbard, K.A.; Law, B.E.; Reichstein, M.; Sulzman, J. An analysis of soil respiration across northern hemisphere temperate ecosystems. Biogeochemistry 2005, 73, 29–70. [Google Scholar] [CrossRef]
- Adachi, M.; Bekku, Y.; Konuma, A.; Kadir, W.; Okuda, T.; Koizumi, H. Required sample size for estimating soil respiration rates in large areas of two tropical forests and of two types of plantations in Malaysia. For. Ecol. Manag. 2005, 210, 455–459. [Google Scholar] [CrossRef]
- Se-Hoon, J.; Dong-Joo, K.; Chan-Woo, P.; Seong-Deog, K. Appropriate sampling points and frequency of CO2 measurements for soil respiration analysis in a pine (Pinus densiflora) forest. Anim. Cells Syst. 2015, 19, 332–338. [Google Scholar] [CrossRef]
- Pantani, O.L.; Fioravanti, F.; Stefanini, F.M.; Berni, R.; Certini, G. Accounting for soil respiration variability—Case study in a Mediterranean pine-dominated forest. Sci. Rep. 2020, 10, 1787. [Google Scholar] [CrossRef] [PubMed]
- Barba, J.; Yuste, J.C.; Martínez-Vilalta, J.; Lloret, F. Drought-induced tree species replacement is reflected in the spatial variability of soil respiration in a mixed Mediterranean forest. For. Ecol. Manag. 2013, 306, 79–87. [Google Scholar] [CrossRef]
- Yáñez-Díaz, M.I.; Cantú-Silva, I.; González-Rodríguez, H.; Marmolejo-Monsiváis, J.G.; Jurado, E.; Gómez-Meza, M.V. Soil respiration in four land use systems. Rev. Mex. Cienc. For. 2017, 8, 123–149. [Google Scholar]
- Čater, M.; Darenova, E.; Simončič, P. Harvesting intensity and tree species affect soil respiration in uneven-aged Dinaric Forest stands. For. Ecol. Manag. 2021, 480, 118638. [Google Scholar] [CrossRef]
- Zhang, Y.; Zou, J.; Meng, D.; Dang, S.; Zhou, J.; Osborne, B.; Ren, Y.; Liang, T.; Yu, K. Effect of soil microorganisms and labile C availability on soil respiration in response to litter inputs in forest ecosystems: A metaanalysis. Ecol. Evol. 2020, 10, 13602–13612. [Google Scholar] [CrossRef]
- Peng, Y.; Thomas, S.C.; Tian, D. Forest management and soil respiration: Implications for carbon sequestration. Environ. Rev. 2008, 16, 93–111. [Google Scholar] [CrossRef]
- Wang, C.; Yu, G.; Zhon, G.; Yan, J.; Zhang, L.; Wang, X.; Tang, X.; Sun, X. CO2 flux elevations over the evergreen coniferous and broadleaved mixed forest in Ginghushan, China. Sci. China Ser. D Earth Sci. 2006, 49 (Suppl. S2), 127–138. [Google Scholar] [CrossRef]
- Wertin, T.M.; Belnap, J.; Reed, S.C. Experimental warming in a dryland community reduced plant photosynthesis and soil CO2 efflux although the relationship between the fluxes remained unchanged. Funct. Ecol. 2017, 31, 297–305. [Google Scholar] [CrossRef]
- Kim, D.G.; Mu, S.; Kang, S.; Lee, D. Factors controlling soil CO2 effluxes and the effects of rewetting on effluxes in adjacent deciduous, coniferous, and mixed forests in Korea. Soil Biol. Biochem. 2010, 42, 576–585. [Google Scholar] [CrossRef]
- Yan, T.; Qu, T.; Song, H.; Sun, Z.; Zeng, H.; Peng, S. Ectomycorrhizal fungi respiration quantification and drivers in three differently-aged larch plantations. Agric. For. Meteorol. 2019, 265, 245–251. [Google Scholar] [CrossRef]
- Tilman, D.; Knops, J.; Wedin, D.; Reich, P.; Ritchie, M.; Siemann, E. The Influence of functional diversity and composition on ecosystem processes. Science 1997, 277, 1300–1302. [Google Scholar] [CrossRef]
- Bréchet, L.; Ponton, S.; Roy, J.; Freycon, V.; Coûteaux, M.; Bonal, D.; Epron, D. Do tree species characteristics influence soil respiration in tropical forests? A test based on 16 tree species planted in monospecific plots. Plant Soil 2009, 319, 235–246. [Google Scholar] [CrossRef]
- Hirota, M.; Zhang, P.; Gu, S.; Shen, H.; Kuriyama, T.; Li, Y.; Tang, Y. Small-scale variation in ecosystem CO2 fluxes in an alpine meadow depends on plant biomass and species richness. J. Plant Res. 2010, 123, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Gessner, M.O.; Swan, C.M.; Dang, C.K.; McKie, B.G.; Bardgett, R.D.; Wall, D.H.; Hättenschwiler, S. Diversity meets decomposition. Trends Ecol. Evol. 2010, 25, 372–380. [Google Scholar] [CrossRef]
- Li, Q.; Tian, Y.; Zhan, X.; Xu, X.; Wang, H.; Kuzyakov, Y. Labile carbon and nitrogen additions affect soil organic matter decomposition more strongly than temperature. Appl. Soil Ecol. 2017, 114, 152–160. [Google Scholar] [CrossRef]
- Salahuddin, R.B.; Razaq, M.; Li, J.; Khan, F.; Jie, Z. Root order-based traits of Manchurian walnut & larch and their plasticity under interspecific competition. Sci. Rep. 2018, 8, 9815. [Google Scholar] [CrossRef]
- Kammer, A.; Schmidt, M.W.; Hagedorn, F. Decomposition pathways of 13C-depleted leaf litter in forest soils of the Swiss Jura. Biogeochemistry 2012, 108, 395–411. [Google Scholar] [CrossRef][Green Version]
- Shaver, G.R.; Johnson, L.C.; Cades, D.H.; Murray, G.; Laundre, J.A.; Rastetter, E.B.; Nadelhoffer, K.J.; Giblin, A.E. Biomass and CO2 flux in wet sedge tundras: Responses to nutrients, temperature, and light. Ecol. Monogr. 1998, 68, 75–97. [Google Scholar] [CrossRef]
- Reth, S.; Reichstein, M.; Falge, E. The effect of soil water content, soil temperature, soil pH-value and the root mass on soil CO2 efflux—A modified model. Plant Soil 2005, 268, 21–33. [Google Scholar] [CrossRef]
- Wei, W.; Weile, C.; Shaopeng, W. Forest soil respiration and its heterotrophic and autotrophic components: Global patterns and responses to temperature and precipitation. Soil Biol. Biochem. 2010, 42, 1236–1244. [Google Scholar] [CrossRef]
- Cantú-Silva, I.; González-Rodríguez, H.; Gómez-Meza, M.V. CO2 efflux in vertisol under different land use systems. Trop. Subtrop. Agroecosystems 2010, 12, 389–403. [Google Scholar]
- Sugasti, L.; Eng, B.; Pinzón, R. Medición continúa de flujo de CO2 en suelo en una parcela de bosque tropical en Isla Barro Colorado, Canal de Panamá. In Proceedings of the 16th LACCEI International Multi-Conference for Engineering, Education, and Technology: Innovation in Education and Inclusion, Lima, Peru, 19–21 July 2018. [Google Scholar] [CrossRef]
- Li, Y.L.; Otieno, D.; Owen, K.; Zhang, Y.; Tenhunen, J.; Rao, X.Q.; Lin, Y.B. Temporal variability in soil CO2 emission in an orchard forest ecosystem. Pedosphere 2008, 18, 273–283. [Google Scholar] [CrossRef]
- Zhang, P.; Tang, Y.; Hirota, M.; Yamamoto, A.; Mariko, S. Use of a regression method to partition sources of ecosystem respiration in an alpine meadow. Soil Biol. Biochem. 2009, 41, 663–670. [Google Scholar] [CrossRef]
- Kosugi, Y.; Mitani, T.; Itoh, M.; Noguchi, S.; Tani, M.; Matsuo, N.; Takanashi, S.; Ohkubo, S.; Nik, A.R. Spatial and temporal variation in soil respiration in a Southeast Asian tropical rainforest. Agric. For. Meteorol. 2007, 147, 35–47. [Google Scholar] [CrossRef]
- Darenova, E.; Pavelka, M.; Macalkova, L. Spatial heterogeneity of CO2 efflux and optimization of the number of measurement positions. Eur. J. Soil Biol. 2016, 75, 123–134. [Google Scholar] [CrossRef]
- Tscharntke, T.; Clough, Y.; Bhagwat, S.A.; Buchori, D.; Faust, H.; Hertel, D.; Hölscher, D.; Juhrbandt, J.; Kessler, M.; Perfecto, I.; et al. Multifunctional shade-tree management in tropical agroforestry landscapes—A review. J. Appl. Ecol. 2011, 48, 619–629. [Google Scholar] [CrossRef]
- Chen, X.Y.; Eamus, D.; Hutley, L.B. Seasonal patterns of soil carbon dioxide efflux from a wet-dry tropical savanna of northern Australia. Aust. J. Bot. 2002, 50, 43–51. [Google Scholar] [CrossRef]
- Wang, C.K.; Yang, J.Y.; Zhang, Q.Z. Soil respiration in six temperate forests in China. Glob. Chang. Biol. 2006, 12, 2103–2114. [Google Scholar] [CrossRef]
- Tian, D.L.; Peng, Y.Y.; Yan, W.D.; Fang, X.; Kang, W.X.; Wang, G.J.; Chen, X.Y. Effects of thinning and litter fall removal on fine root production and soil organic carbon content in Masson pine plantations. Pedosphere 2010, 20, 486–493. [Google Scholar] [CrossRef]
- Reichstein, M.; Rey, A.; Freibauer, A.; Tenhunen, J.; Valentini, R.; Banza, J.; Casals, P.; Cheng, Y.; Grünzweig, J.; Irvine, J.; et al. Modeling temporal and large-scale spatial variability of soil respiration from soil water availability, temperature and vegetation productivity indices. Glob. Biogeochem. Cycles 2003, 17, 1104. [Google Scholar] [CrossRef]
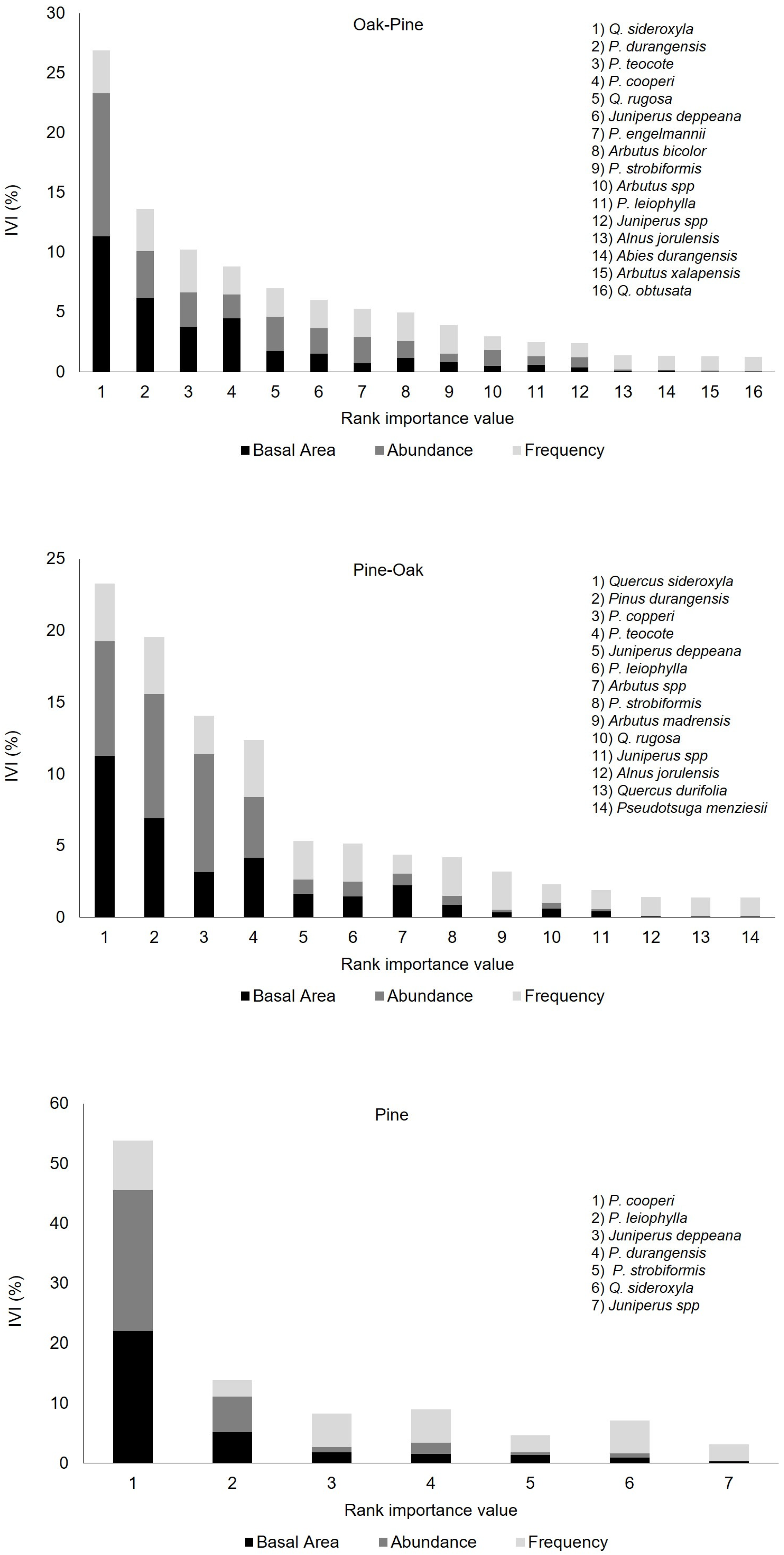

| Forest Type | Dq (cm) | DBH (cm) | BA (m2 ha−1) | Density (Stems ha−1) | GS (m3 ha−1) | AGB (Mg ha−1) | Genera | Species | Elevation (m.a.s.l) |
|---|---|---|---|---|---|---|---|---|---|
| Pine | 22.1 | 19.1 | 20.63 | 543 | 191.1 | 154.1 | 3 | 7 | 2707 |
| Pine–oak | 20.2 | 18.1 | 23.01 | 719 | 203.5 | 130.7 | 6 | 14 | 2690 |
| Oak–pine | 20.7 | 17.8 | 24.91 | 737 | 202.6 | 160.1 | 7 | 16 | 2687 |
| Month | Soil CO2 Efflux (µmol m−2 s−1) † | Soil Temperature (°C) | Soil Moisture (%) |
|---|---|---|---|
| January | 2.81 ± 1.24 a | 8.81 ± 1.28 | 59.2 ± 10.9 |
| February | 2.49 ± 1.46 ab | 9.05 ± 1.98 | 29.9 ± 13.7 |
| March | 2.46 ± 1.42 ab | 11.6 ± 1.95 | 20.5 ± 5.66 |
| April | 2.28 ± 1.47 ab | 12.7 ± 1.72 | 22.1 ± 9.69 |
| May | 4.15 ± 2.57 ac | 13.8 ± 2.16 | 22.8 ± 10.1 |
| June | 7.97 ± 2.94 d | 15.1 ± 1.85 | 54.8 ± 23.1 |
| July | 8.79 ± 1.81 de | 16.1 ± 1.80 | 68.4 ± 25.8 |
| August | 8.07 ± 1.47 def | 14.2 ± 2.21 | 66.4 ± 16.5 |
| October | 7.64 ± 1.94 dfg | 12.7 ± 1.70 | 65.7 ± 18.2 |
| November | 6.38 ± 1.74 fgh | 10.7 ± 1.43 | 64.4 ± 16.6 |
| December | 5.53 ± 1.86 h | 9.8 ± 0.82 | 51.7 ± 11.5 |
| Forest Type | Soil CO2 Efflux (µmol m−2 s−1) † | Soil Temperature (°C) | Soil Moisture (%) |
|---|---|---|---|
| Pine | 5.03 ± 2.96 a | 12.2 ± 2.71 a | 42.4 ± 23.9 a |
| Pine–oak | 4.81 ± 2.83 a | 11.9 ± 2.82 a | 45.2 ± 24.9 a |
| Oak–pine | 5.08 ± 3.40 a | 11.7 ± 3.27 a | 43.7 ± 22.3 a |
| Cross-Validation | ||||||
|---|---|---|---|---|---|---|
| Parameter | Estimate | Std. Err. | t(262) | p-Value | RMSE | R2 |
| b1 | 0.1030 | 0.0075 | 13.6992 | <0.0000 | 2.483 | 0.483 |
| b2 | 0.0166 | 0.0011 | 16.5048 | <0.0000 | ||
| b3 | 0.0599 | 0.0190 | 3.1613 | <0.0000 | ||
| b4 | −1.2549 | 0.2572 | −4.8799 | <0.0000 | ||
| b5 | −0.0328 | 0.0055 | −5.9732 | <0.0000 | ||
| b6 | 0.0039 | 0.0007 | 5.2337 | <0.0000 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Rivas, J.A.; Vargas-Larreta, B.; López-Martínez, J.O.; Aguirre-Calderón, C.G.; Hernández, F.J.; Ángeles-Pérez, G. Understanding Soil Respiration Dynamics in Temperate Forests in Northwestern Mexico. Forests 2023, 14, 1763. https://doi.org/10.3390/f14091763
Martínez-Rivas JA, Vargas-Larreta B, López-Martínez JO, Aguirre-Calderón CG, Hernández FJ, Ángeles-Pérez G. Understanding Soil Respiration Dynamics in Temperate Forests in Northwestern Mexico. Forests. 2023; 14(9):1763. https://doi.org/10.3390/f14091763
Chicago/Turabian StyleMartínez-Rivas, José Alexis, Benedicto Vargas-Larreta, Jorge Omar López-Martínez, Cristóbal Gerardo Aguirre-Calderón, Francisco Javier Hernández, and Gregorio Ángeles-Pérez. 2023. "Understanding Soil Respiration Dynamics in Temperate Forests in Northwestern Mexico" Forests 14, no. 9: 1763. https://doi.org/10.3390/f14091763
APA StyleMartínez-Rivas, J. A., Vargas-Larreta, B., López-Martínez, J. O., Aguirre-Calderón, C. G., Hernández, F. J., & Ángeles-Pérez, G. (2023). Understanding Soil Respiration Dynamics in Temperate Forests in Northwestern Mexico. Forests, 14(9), 1763. https://doi.org/10.3390/f14091763








