Elucidating SNP-Based Population Structure and Genetic Diversity of Bruguiera gymnorhiza (L.) Savigny in Thailand
Abstract
1. Introduction
2. Materials and Methods
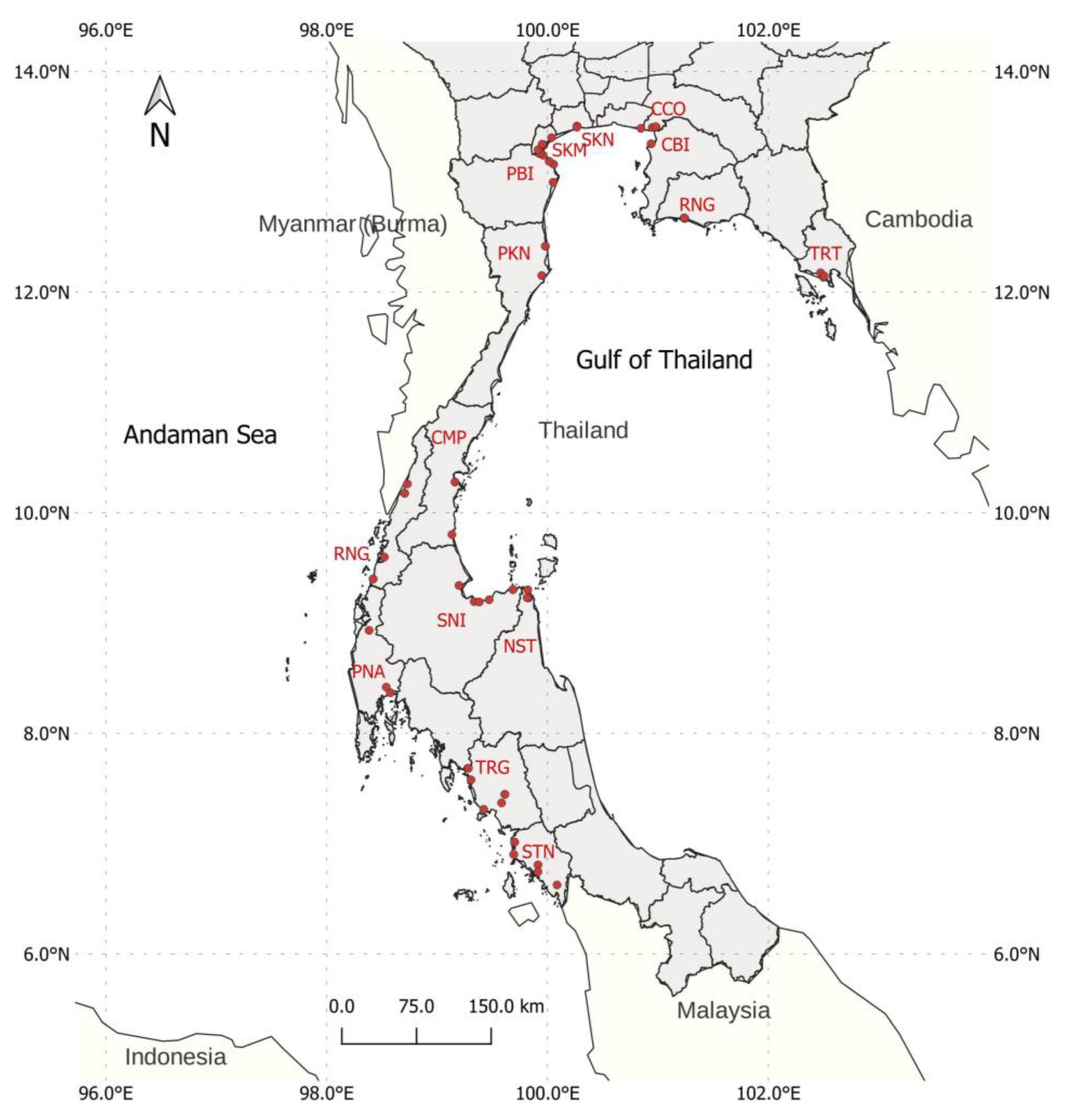
2.1. Sample Collection and DNA Extraction
2.2. RAD-Seq Library Construction and RAD-Seq Sequencing
2.3. SNP Identification and LD Pruning
2.4. Phylogenetic Analysis
2.5. Population Genetic Structure Analysis
2.6. Genetic Diversity Analysis
3. Results
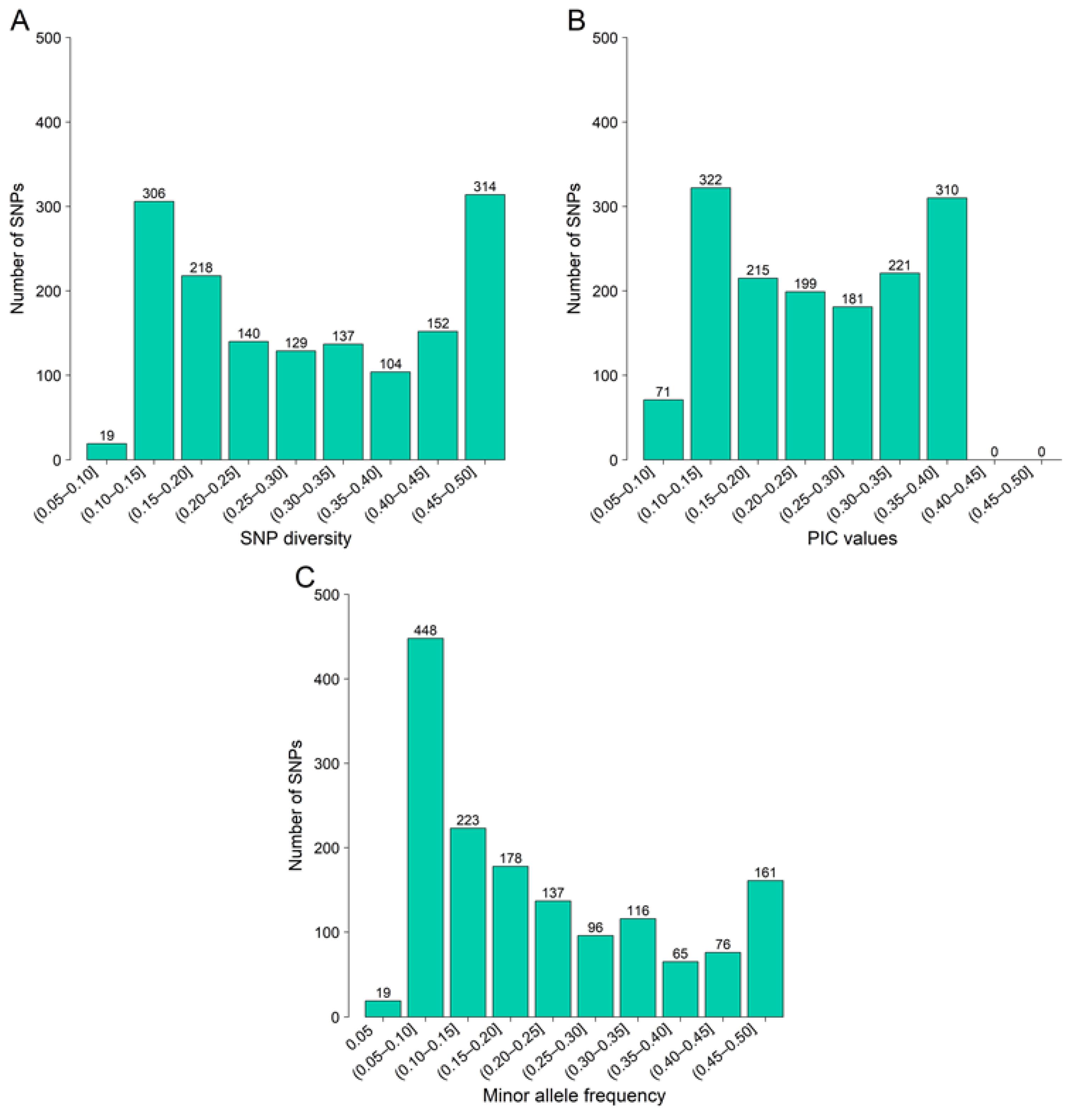
3.1. SNP Identification and Characterization
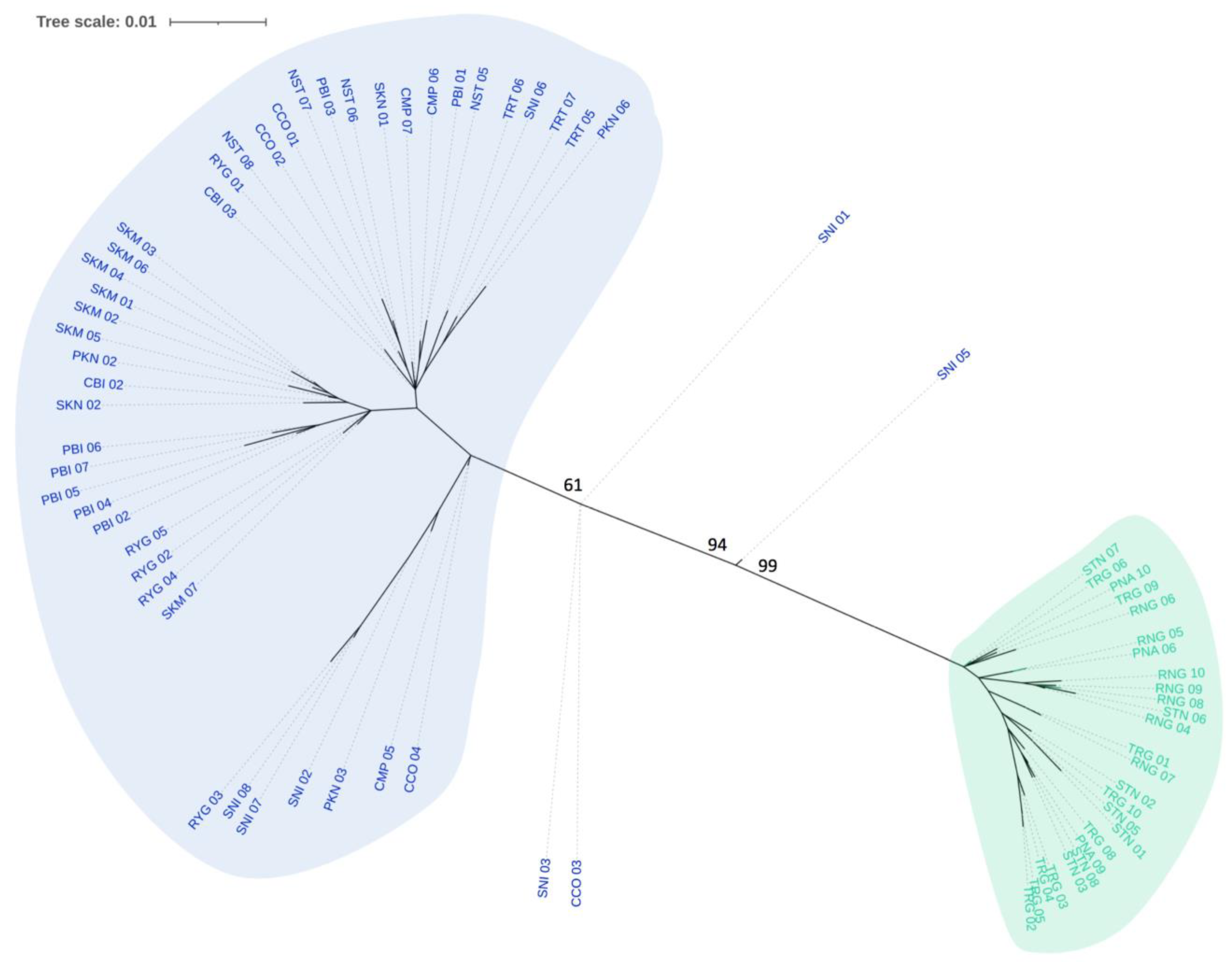
3.2. Phylogenetic Tree
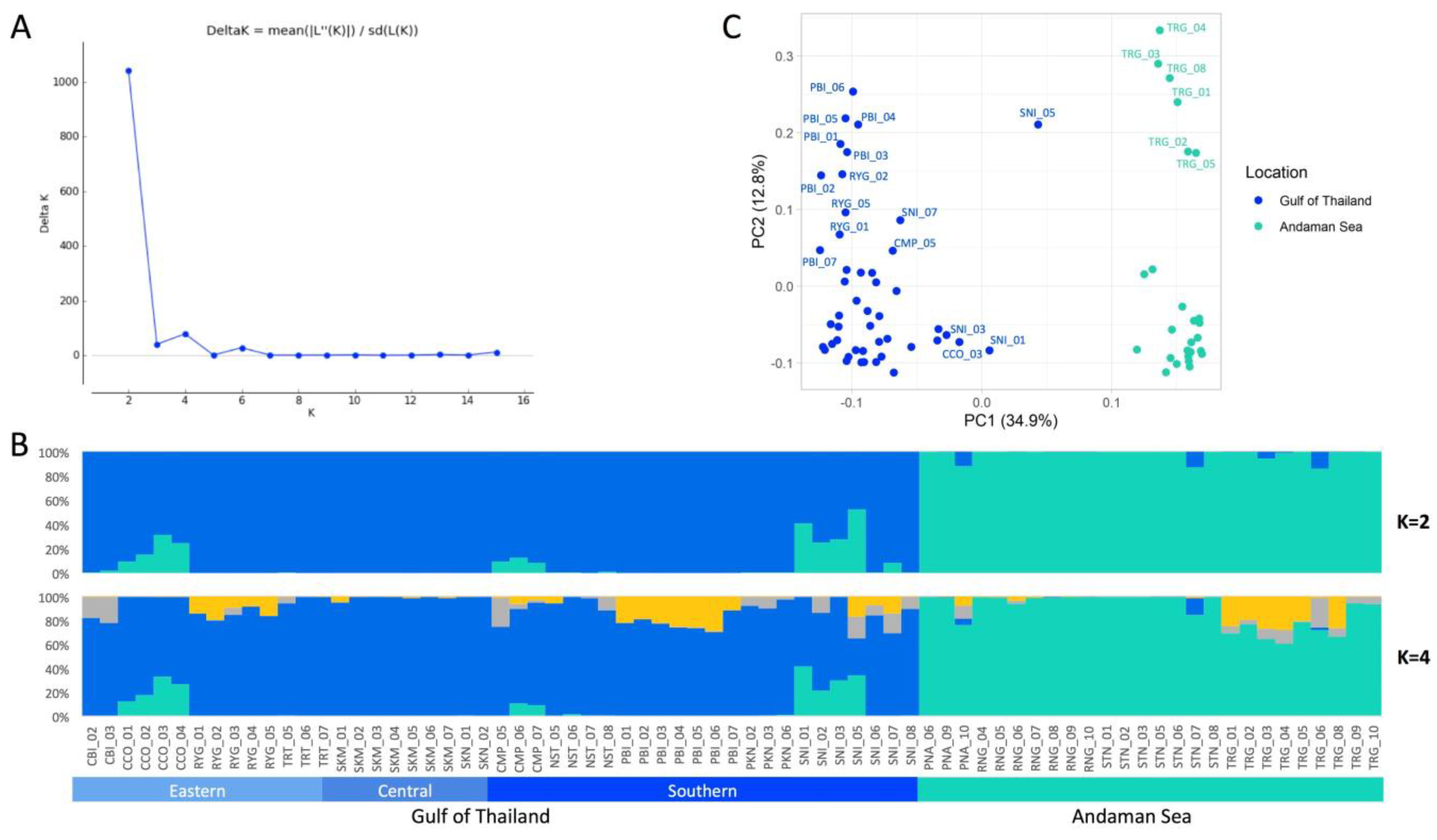
3.3. Population Genetic Structure and PCA
3.4. Genetic Diversity and Differentiation
4. Discussion
4.1. Genetic Relationship and Genetic Structure
4.2. Genetic Diversity and Genetic Differentiation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allen, J.A.; Duke, N.C. Bruguiera gymnorrhiza (large-leafed mangrove). In Species Profiles for Pacific Island Agroforestry; Elevitch, C.R., Ed.; Permanent Agriculture Resources (PAR): Holualoa, HI, USA, 2006; ISBN 0-9702544-5-8. [Google Scholar]
- Lee, S.Y.; Primavera, J.H.; Dahdouh-Guebas, F.; Mckee, K.; Bosire, J.O.; Cannicci, S.; Diele, K.; Fromard, F.; Koedam, N.; Marchand, C.; et al. Ecological role and services of tropical mangrove ecosystems: A reassessment. Glob. Ecol. Biogeogr. 2014, 23, 726–743. [Google Scholar] [CrossRef]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- Alongi, D.M. Present state and future of the world’s mangrove forests. Environ. Conserv. 2002, 29, 331–349. [Google Scholar] [CrossRef]
- Giri, C.; Ochieng, E.; Tieszen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Bunting, P.; Rosenqvist, A.; Hilarides, L.; Lucas, R.M.; Thomas, N.; Tadono, T.; Worthington, T.A.; Spalding, M.; Murray, N.J. Global mangrove extent change 1996–2020: Global mangrove watch version 3.0. Remote Sens. 2022, 14, 3657. [Google Scholar] [CrossRef]
- Plathong, S.; Plathong, J. Past and present threats on mangrove ecosystem in Peninsular Thailand. In Coastal Biodiversity in Mangrove Ecosystems: Paper Presented in UNU-INWEH-UNESCO International Training Course, Held at Centre of Advanced Studies; Annamalai University: Chidambaram, India, 2004; pp. 1–13. [Google Scholar]
- Boyd, C.E.; Davis, R.P.; McNevin, A.A. Perspectives on the mangrove conundrum, land use, and benefits of yield intensification in farmed shrimp production: A review. J. World Aquac. Soc. 2022, 53, 8–46. [Google Scholar] [CrossRef]
- Tomlinson, P.B.; Primack, R.B.; Bunt, J.S. Preliminary observations on floral biology in mangrove Rhizophoraceae. Biotropica 1979, 11, 256. [Google Scholar] [CrossRef]
- Zhu, Z.; Chen, J.; Zheng, H.L. Physiological and proteomic characterization of salt tolerance in a mangrove plant, Bruguiera gymnorrhiza (L.) Lam. Tree Physiol. 2012, 32, 1378–1388. [Google Scholar] [CrossRef]
- Haq, M.; Sani, W.; Hossain, A.B.M.S.; Taha, R.M.; Monneruzzaman, K.M. Total phenolic contents, antioxidant and antimicrobial activities of Bruguiera gymnorrhiza. J. Med. Plants Res. 2011, 5, 4112–4118. [Google Scholar] [CrossRef]
- Bibi, S.N.; Fawzi, M.M.; Gokhan, Z.; Rajesh, J.; Nadeem, N.; Kannan, R.R.R.; Albuquerque, R.D.D.G.; Pandian, S.K. Ethnopharmacology, phytochemistry, and global distribution of mangroves—A comprehensive review. Mar. Drugs 2019, 17, 40231. [Google Scholar] [CrossRef]
- Karim, M.A.; Islam, M.A.; Islam, M.M.; Rahman, M.S.; Sultana, S.; Biswas, S.; Hosen, M.J.; Mazumder, K.; Rahman, M.M.; Hasan, M.N. Evaluation of antioxidant, anti-hemolytic, cytotoxic effects and anti-bacterial activity of selected mangrove plants (Bruguiera gymnorrhiza and Heritiera littoralis) in Bangladesh. Clin. Phytosci. 2020, 6, 8. [Google Scholar] [CrossRef]
- Ge, J.P.; Cai, B.; Ping, W.; Song, G.; Ling, H.; Lin, P. Mating system and population genetic structure of Bruguiera gymnorrhiza (Rhizophoraceae), a viviparous mangrove species in China. J. Exp. Mar. Biol. Ecol. 2005, 326, 48–55. [Google Scholar] [CrossRef]
- Kondo, K.; Nakamura, T.; Piyakarnchana, T.; Mechvichai, W. Pollination in Bruguiera gymnorrhiza in Miyara river, Ishigaki island, Japan and Phangnga, Thailand. Plant Species Biol. 1991, 6, 105–109. [Google Scholar] [CrossRef]
- Sheue, C.-R.; Yong, J.W.H.; Yang, Y.-P. The Bruguiera (Rhizophoraceae) species in the mangroves of Singapore, especially on the new record and the rediscovery. Taiwania 2005, 50, 251–260. [Google Scholar]
- Ragavan, P.; Saxena, M.; Saxena, A.; Mohan, P.M.; Sachithanandam, V.; Coomar, T. Floral composition and taxonomy of mangroves of Andaman and Nicobar Islands. Indian J. Geo-Mar. Sci. 2014, 43, 1031–1044. [Google Scholar]
- Allen, J.A.; Krauss, K.W.; Duke, N.C.; Herbst, D.R.; Bjorkman, O.; Shih, C. Bruguiera species in Hawai’i: Systematic considerations and ecological implications. Pac. Sci. 2000, 54, 331–343. [Google Scholar]
- Shearman, J.R.; Naktang, C.; Sonthirod, C.; Kongkachana, W.; U-thoomporn, S.; Jomchai, N.; Maknual, C.; Yamprasai, S.; Promchoo, W.; Ruang-areerate, P.; et al. Assembly of a hybrid mangrove, Bruguiera hainesii, and its two ancestral contributors, Bruguiera cylindrica and Bruguiera gymnorhiza. Genomics 2022, 114, 110382. [Google Scholar] [CrossRef]
- Ruang-areerate, P.; Kongkachana, W.; Naktang, C.; Sonthirod, C.; Narong, N.; Jomchai, N.; Maprasop, P.; Maknual, C.; Phormsin, N.; Shearman, J.R.; et al. Complete chloroplast genome sequences of five Bruguiera species (Rhizophoraceae): Comparative analysis and phylogenetic relationships. PeerJ 2021, 9, e12268. [Google Scholar] [CrossRef]
- He, Z.; Feng, X.; Chen, Q.; Li, L.; Li, S.; Han, K.; Guo, Z.; Wang, J.; Liu, M.; Shi, C.; et al. Evolution of coastal forests based on a full set of mangrove genomes. Nat. Ecol. Evol. 2022, 6, 738–749. [Google Scholar] [CrossRef]
- Shi, W.; Song, W.; Chen, Z.; Cai, H.; Gong, Q.; Liu, J.; Shi, C.; Wang, S. Comparative chloroplast genome analyses of diverse Phoebe (Lauraceae) species endemic to China provide insight into their phylogeographical origin. PeerJ 2023, 11, e14573. [Google Scholar] [CrossRef]
- Ruang-areerate, P.; Yoocha, T.; Kongkachana, W.; Phetchawang, P.; Maknual, C.; Meepol, W.; Jiumjamrassil, D.; Pootakham, W.; Tangphatsornruang, S. Comparative analysis and phylogenetic relationships of Ceriops species (Rhizophoraceae) and Avicennia lanata (Acanthaceae): Insight into the chloroplast genome evolution between middle and seaward zones of mangrove forests. Biology 2022, 11, 383. [Google Scholar] [CrossRef] [PubMed]
- Pootakham, W.; Naktang, C.; Sonthirod, C.; Kongkachana, W.; Yoocha, T.; Jomchai, N.; Maknual, C.; Chumriang, P.; Pravinvongvuthi, T.; Tangphatsornruang, S. De Novo reference assembly of the upriver orange mangrove (Bruguiera sexangula) genome. Genome Biol. Evol. 2022, 14, evac025. [Google Scholar] [CrossRef] [PubMed]
- Abeysinghe, P.D.; Triest, L.; Greef, B.D.; Koedam, N.; Hettiarachi, S. Genetic and geographic variation of the mangrove tree Bruguiera in Sri Lanka. Aquat. Bot. 2000, 67, 131–141. [Google Scholar] [CrossRef]
- Mondini, L.; Noorani, A.; Pagnotta, M.A. Assessing plant genetic diversity by molecular tools. Diversity 2009, 1, 19–35. [Google Scholar] [CrossRef]
- Govindaraj, M.; Vetriventhan, M.; Srinivasan, M. Importance of genetic diversity assessment in crop plants and its recent advances: An overview of its analytical perspectives. Genet. Res. Int. 2015, 2015, 431487. [Google Scholar] [CrossRef]
- Nadeem, M.A.; Nawaz, M.A.; Shahid, M.Q.; Doğan, Y.; Comertpay, G.; Yıldız, M.; Hatipoğlu, R.; Ahmad, F.; Alsaleh, A.; Labhane, N.; et al. DNA molecular markers in plant breeding: Current status and recent advancements in genomic selection and genome editing. Biotechnol. Biotechnol. Equip. 2018, 32, 261–285. [Google Scholar] [CrossRef]
- Fischer, M.C.; Rellstab, C.; Leuzinger, M.; Roumet, M.; Gugerli, F.; Shimizu, K.K.; Holderegger, R.; Widmer, A. Estimating genomic diversity and population differentiation—An empirical comparison of microsatellite and SNP variation in Arabidopsis halleri. BMC Genom. 2017, 18, 69. [Google Scholar] [CrossRef]
- Zimmerman, S.J.; Aldridge, C.L.; Oyler-McCance, S.J. An empirical comparison of population genetic analyses using microsatellite and SNP data for a species of conservation concern. BMC Genom. 2020, 21, 382. [Google Scholar] [CrossRef]
- He, Z.; Li, X.; Yang, M.; Wang, X.; Zhong, C.; Duke, N.C.; Wu, C.I.; Shi, S. Speciation with gene flow via cycles of isolation and migration: Insights from multiple mangrove taxa. Natl. Sci. Rev. 2019, 6, 275–288. [Google Scholar] [CrossRef]
- Pootakham, W.; Sonthirod, C.; Naktang, C.; Kongkachana, W.; Sangsrakru, D.; U-thoomporn, S.; Maknual, C.; Meepol, W.; Promchoo, W.; Maprasop, P.; et al. A chromosome-scale reference genome assembly of yellow mangrove (Bruguiera parviflora) reveals a whole genome duplication event associated with the Rhizophoraceae lineage. Mol. Ecol. Resour. 2022, 22, 1939–1953. [Google Scholar] [CrossRef]
- Ruang-areerate, P.; Naktang, C.; Kongkachana, W.; Sangsrakru, D.; Narong, N.; Maknual, C.; Pravinvongvuthi, T.; Promchoo, W.; Yamprasai, S.; Tangphatsornruang, S.; et al. Assessment of the genetic diversity and population structure of Rhizophora apiculata Blume (Rhizophoraceae) in Thailand. Biology 2022, 11, 1449. [Google Scholar] [CrossRef] [PubMed]
- Khanbo, S.; Kongkachana, W.; Jomchai, N.; Charoensri, S.; Maknual, C.; Maprasop, P.; Phormsin, N.; Tangphatsornruang, S.; Pootakham, W. Genetic diversity and population structure of Bruguiera cylindrica along coastal areas in Thailand. Aquat. Bot. 2022, 183, 103575. [Google Scholar] [CrossRef]
- Takeuchi, T.; Sugaya, T.; Kanazashi, A.; Yoshimaru, H.; Katsuta, M. Genetic diversity of Kandelia candel and Bruguiera gymnorrhiza in the Southwest Islands, Japan. J. For. Res. 2001, 6, 157–162. [Google Scholar] [CrossRef]
- Minobe, S.; Fukui, S.; Saiki, R.; Kajita, T.; Changtragoon, S.; Ab Shukor, N.A.; Latiff, A.; Ramesh, B.R.; Koizumi, O.; Yamazaki, T. Highly differentiated population structure of a mangrove species, Bruguiera gymnorhiza (Rhizophoraceae) revealed by one nuclear GapCp and one chloroplast intergenic spacer trnF-trnL. Conserv. Genet. 2010, 11, 301–310. [Google Scholar] [CrossRef]
- Urashi, C.; Teshima, K.M.; Minobe, S.; Koizumi, O.; Inomata, N. Inferences of evolutionary history of a widely distributed mangrove species, Bruguiera gymnorrhiza, in the Indo-West Pacific region. Ecol. Evol. 2013, 3, 2251–2261. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, N.; Nandy, P.; Sengupta, C.; Das, S. RAPD and ISSR marker mediated genetic polymorphism of two mangroves Bruguiera gymnorrhiza and Heritiera fomes from Indian Sundarbans in relation to their sustainability. Physiol. Mol. Biol. Plants 2015, 21, 375–384. [Google Scholar] [CrossRef]
- Geng, Q.; Wang, Z.; Tao, J.; Kimura, M.K.; Liu, H.; Hogetsu, T.; Lian, C. Ocean currents drove genetic structure of seven dominant mangrove species along the coastlines of southern China. Front. Genet. 2021, 12, 615911. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Lian, C.; Kameyama, N.; Hogetsu, T. Analyses of genetic population structure of two ecologically important mangrove tree species, Bruguiera gymnorrhiza and Kandelia obovata from different river basins of Iriomote Island of the Ryukyu Archipelago, Japan. Tree Genet. Genomes 2012, 8, 1247–1260. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Davey, J.L.; Blaxter, M.W. RADseq: Next-generation population genetics. Brief. Funct. Genom. 2010, 9, 416–423. [Google Scholar] [CrossRef]
- Baird, N.A.; Etter, P.D.; Atwood, T.S.; Currey, M.C.; Shiver, A.L.; Lewis, Z.A.; Selker, E.U.; Cresko, W.A.; Johnson, E.A. Rapid SNP discovery and genetic mapping using sequenced RAD markers. PLoS ONE 2008, 3, e3376. [Google Scholar] [CrossRef] [PubMed]
- Etter, P.D.; Bassham, S.; Hohenlohe, P.A.; Johnson, E.A.; Cresko, W.A. SNP discovery and genotyping for evolutionary genetics using RAD sequencing. Methods Mol. Biol. 2011, 772, 157–178. [Google Scholar] [CrossRef] [PubMed]
- Andrews, K.R.; Good, J.M.; Miller, M.R.; Luikart, G.; Hohenlohe, P.A. Harnessing the power of RADseq for ecological and evolutionary genomics. Nat. Rev. Genet. 2016, 17, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- McKenna, A.; Hanna, M.; Banks, E.; Sivachenko, A.; Cibulskis, K.; Kernytsky, A.; Garimella, K.; Altshuler, D.; Gabriel, S.; Daly, M.; et al. The genome analysis Toolkit: A MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2009, 20, 1297–1303. [Google Scholar] [CrossRef]
- Li, H. A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 2011, 27, 2987–2993. [Google Scholar] [CrossRef]
- Danecek, P.; Auton, A.; Abecasis, G.; Albers, C.A.; Banks, E.; DePristo, M.A.; Handsaker, R.E.; Lunter, G.; Marth, G.T.; Sherry, S.T.; et al. The variant call format and VCFtools. Bioinformatics 2011, 27, 2156–2158. [Google Scholar] [CrossRef]
- Lischer, H.E.L.; Excoffier, L. PGDSpider: An automated data conversion tool for connecting population genetics and genomics programs. Bioinformatics 2012, 28, 298–299. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive tree of life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021, 49, W293–W296. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef] [PubMed]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; VonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Jakobsson, M.; Rosenberg, N.A. CLUMPP: A cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 2009, 23, 1801–1806. [Google Scholar] [CrossRef] [PubMed]
- Wickham, H. ggplot2 Elegant Graphics for Data Analysis, 2nd ed.; Gentleman, R., Hornik, K., Parmigiani, G., Eds.; Springer Nature: Houston, TX, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Liu, K.; Muse, S.V. PowerMaker: An integrated analysis environment for genetic maker analysis. Bioinformatics 2005, 21, 2128–2129. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenALEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Ge, X.J.; Sun, M. Population genetic structure of Ceriops tagal (Rhizophoraceae) in Thailand and China. Wetl. Ecol. Manag. 2001, 9, 203–209. [Google Scholar] [CrossRef]
- Guo, Z.; Huang, Y.; Chen, Y.; Duke, N.C.; Zhong, C.; Shi, S. Genetic discontinuities in a dominant mangrove Rhizophora apiculata (Rhizophoraceae) in the Indo-Malesian region. J. Biogeogr. 2016, 43, 1856–1868. [Google Scholar] [CrossRef]
- Yang, Y.; Li, J.; Yang, S.; Li, X.; Fang, L.; Zhong, C.; Duke, N.C.; Zhou, R.; Shi, S. Effects of Pleistocene sea-level fluctuations on mangrove population dynamics: A lesson from Sonneratia alba. BMC Evol. Biol. 2017, 17, 22. [Google Scholar] [CrossRef] [PubMed]
- Wee, A.K.S.; Teo, J.X.H.; Chua, J.L.; Takayama, K.; Asakawa, T.; Meenakshisundaram, S.H.; Onrizal; Adjie, B.; Ardli, E.R.; Sungkaew, S.; et al. Vicariance and oceanic barriers drive contemporary genetic structure of widespread mangrove species Sonneratia alba J. Sm in the Indo-West Pacific. Forests 2017, 8, 483. [Google Scholar] [CrossRef]
- Triest, L.; Satyanarayana, B.; Delange, O.; Sarker, K.K.; Sierens, T.; Dahdouh-Guebas, F. Barrier to gene flow of grey mangrove Avicennia marina populations in the Malay Peninsula as revealed from nuclear microsatellites and chloroplast haplotypes. Front. Conserv. Sci. 2021, 2, 727819. [Google Scholar] [CrossRef]
- Pootakham, W.; Naktang, C.; Sonthirod, C.; Kongkachana, W.; Narong, N.; Sangsrakru, D.; Maknual, C.; Jiumjamrassil, D.; Chumriang, P.; Tangphatsornruang, S. Chromosome-level genome assembly of Indian mangrove (Ceriops tagal) revealed a genome-wide duplication event predating the divergence of Rhizophoraceae mangrove species. Plant Genome 2022, 15, e20217. [Google Scholar] [CrossRef]
- Duke, N.C.; Lo, E.; Sun, M. Global distribution and genetic discontinuities of mangroves—Emerging patterns in the evolution of Rhizophora. Trees 2002, 16, 65–79. [Google Scholar] [CrossRef]
- Oluwajuwon, T.V.; Attafuah, R.; Offiah, C.J.; Krabel, D. Genetic variation in tropical tree species and plantations: A review. Open J. For. 2022, 12, 350–366. [Google Scholar] [CrossRef]
- Tripiana, V.; Bourgeois, M.; Verhaegen, D.; Vigneron, P.; Bouvet, J.M. Combining microsatellites, growth, and adaptive traits for managing in situ genetic resources of Eucalyptus urophylla. Can. J. For. Res. 2007, 37, 773–785. [Google Scholar] [CrossRef]
- Senakun, C.; Changtragoon, S.; Pramual, P.; Prathepha, P. Genetic structure and diversity of Shorea obtusa (Dipterocarpaceae) in Thailand. J. Syst. Evol. 2011, 49, 120–125. [Google Scholar] [CrossRef]
- Hu, L.; Le, X.G.; Zhou, S.S.; Zhang, C.Y.; Tan, Y.H.; Ren, Q.; Meng, H.H.; Cun, Y.; Li, J. Conservation significance of the rare and endangered tree species, Trigonobalanus doichangensis (Fagaceae). Diversity 2022, 14, 666. [Google Scholar] [CrossRef]
- Azman, A.; Ng, K.K.S.; Ng, C.H.; Lee, C.T.; Tnah, L.H.; Zakaria, N.F.; Mahruji, S.; Perdan, K.; Abdul-Kadir, M.Z.; Cheng, A.; et al. Low genetic diversity indicating the threatened status of Rhizophora apiculata (Rhizophoraceae) in Malaysia: Declined evolution meets habitat destruction. Sci. Rep. 2020, 10, 19112. [Google Scholar] [CrossRef]
- Kennedy, J.P.; Garavelli, L.; Truelove, N.K.; Devlin, D.J.; Box, S.J.; Chérubin, L.M.; Feller, I.C. Contrasting genetic effects of red mangrove (Rhizophora mangle L.) range expansion along west and east Florida. J. Biogeogr. 2017, 44, 335–347. [Google Scholar] [CrossRef]
- Binks, R.M.; Byrne, M.; McMahon, K.; Pitt, G.; Murray, K.; Evans, R.D. Habitat discontinuities form strong barriers to gene flow among mangrove populations, despite the capacity for long-distance dispersal. Divers. Distrib. 2019, 25, 298–309. [Google Scholar] [CrossRef]
- Macintosh, D.J.; Ashton, E.C. A Review of Mangrove Biodiversity Conservation and Management; Centre for Tropical Ecosystems Research; University of Aarhus: Aarhus, Denmark, 2002. [Google Scholar]
- Wee, A.K.S.; Low, S.Y.; Webb, E.L. Pollen limitation affects reproductive outcome in the bird-pollinated mangrove Bruguiera gymnorrhiza (Lam.) in a highly urbanized environment. Aquat. Bot. 2014, 120, 240–243. [Google Scholar] [CrossRef]
- Nassar, J.M.; Hamrick, J.L.; Fleming, T.H. Genetic variation and population structure of the mixed-mating cactus, Melocactus curvispinus (Cactaceae). Heredity 2001, 87, 69–79. [Google Scholar] [CrossRef]
- Nybom, H. Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol. Ecol. 2004, 13, 1143–1155. [Google Scholar] [CrossRef]
| Population | N | Na | Ne | I | Ho | He | PPL (%) | F |
|---|---|---|---|---|---|---|---|---|
| Gulf of Thailand | 46 | 2.510 ± 0.013 | 1.517 ± 0.008 | 0.515 ± 0.005 | 0.383 ± 0.007 | 0.311 ± 0.004 | 100.00 | −0.135 ± 0.009 |
| Andaman Sea | 27 | 2.417 ± 0.013 | 1.557 ± 0.009 | 0.529 ± 0.005 | 0.410 ± 0.008 | 0.323 ± 0.004 | 99.01 | −0.157 ± 0.010 |
| Overall | 73 | 2.463 ± 0.009 | 1.537 ± 0.006 | 0.522 ± 0.004 | 0.397 ± 0.005 | 0.317 ± 0.003 | 99.51 | −0.146 ± 0.007 |
| Source of Variation | df | Sum of Squares | Variance Components | Percentage of Variation | F-Statistics |
|---|---|---|---|---|---|
| Among populations | 1 | 2772.27 | 37.83 | 16.05 | FST = 0.16 *** |
| Within populations | 144 | 28,495.58 | 197.89 | 83.95 | |
| Total | 145 | 31,267.85 | 235.71 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruang-areerate, P.; Sonthirod, C.; Sangsrakru, D.; Waiyamitra, P.; Maknual, C.; Wanthongchai, P.; Chomriang, P.; Pootakham, W.; Tangphatsornruang, S. Elucidating SNP-Based Population Structure and Genetic Diversity of Bruguiera gymnorhiza (L.) Savigny in Thailand. Forests 2023, 14, 693. https://doi.org/10.3390/f14040693
Ruang-areerate P, Sonthirod C, Sangsrakru D, Waiyamitra P, Maknual C, Wanthongchai P, Chomriang P, Pootakham W, Tangphatsornruang S. Elucidating SNP-Based Population Structure and Genetic Diversity of Bruguiera gymnorhiza (L.) Savigny in Thailand. Forests. 2023; 14(4):693. https://doi.org/10.3390/f14040693
Chicago/Turabian StyleRuang-areerate, Panthita, Chutima Sonthirod, Duangjai Sangsrakru, Pitchaporn Waiyamitra, Chatree Maknual, Poonsri Wanthongchai, Pranom Chomriang, Wirulda Pootakham, and Sithichoke Tangphatsornruang. 2023. "Elucidating SNP-Based Population Structure and Genetic Diversity of Bruguiera gymnorhiza (L.) Savigny in Thailand" Forests 14, no. 4: 693. https://doi.org/10.3390/f14040693
APA StyleRuang-areerate, P., Sonthirod, C., Sangsrakru, D., Waiyamitra, P., Maknual, C., Wanthongchai, P., Chomriang, P., Pootakham, W., & Tangphatsornruang, S. (2023). Elucidating SNP-Based Population Structure and Genetic Diversity of Bruguiera gymnorhiza (L.) Savigny in Thailand. Forests, 14(4), 693. https://doi.org/10.3390/f14040693






