Abstract
Tropical forests are critical for ecosystem functioning and are the major provider of global ecosystem services. The combined effects of climate change and anthropogenic disturbances in the tropics, however, have caused a mass decline in major ecosystem services in recent decades. Nevertheless, most previous studies focused entirely on these disturbances’ effects on forest ecosystem services without taking into consideration the combined effects of climate change and disturbances on key ecosystem services’ indicators. This is more pronounced in Bangladesh, which is one of the countries most adversely affected by global climate change. We investigated anthropogenic disturbances’ effects on regulating ecosystem services under different rainfall regimes in Khadimnagar National Park (KNP) and Satchari National Park (SNP), two major forested, protected areas in Northeast Bangladesh. We considered twelve indicators for five aboveground ecosystem services (i.e., primary production, carbon storage, biodiversity, wildlife habitat, and timber production) and four indicators for four belowground ecosystem services (i.e., organic carbon storage, soil aggregate stability, soil physical support, and soil water regulation) in our study. The effects of disturbances on the indicators of four aboveground ecosystem services (i.e., primary production, tree carbon storage, biodiversity conservation, and timber production) and one belowground ecosystem service (soil physical support) differed in terms of rainfall regimes. Disturbances negatively influenced indicators of primary production (p = 0.03, r = −0.28), tree carbon storage (p = 0.02, r = −0.29), and timber production (p = 0.002, r = −0.39), and positively impacted soil organic carbon storage (p = 0.04, r = 0.27) only under a medium-rainfall regime. Interestingly, a significant positive impact of disturbances on soil physical support (p = 0.001, r = 0.39) was observed under heavy rainfall in our study. In addition, disturbances were significantly associated with biodiversity conservation (p < 0.05) and wildlife habitat quality (p < 0.05) under both rainfall classes. Our findings provide critical insights into the synergistic role of anthropogenic perturbations in regulating ecosystem services with rainfall pattern as a key climatic parameter, which can be useful in restoring large tracts of degraded tropical forests in the United Nations (UN) Decade on Ecosystem Restoration.
1. Introduction
Tropical forests are a major ecosystem services provider through a wide range of critical ecosystem functions [1,2]. One-fourth of Earth’s terrestrial carbon (C) is stored in aboveground biomass (AGB) in tropical forests, which is also one of the major forest ecosystem services with implications for climate change mitigation [3,4]. The increasing frequency of anthropogenic disturbances in the tropics, along with global climate change, has significantly altered forest health and its capacity to provide key ecosystem functions and services [3,5]. A limited number of studies have assessed the effects of disturbances [6] and climate change [3] on tropical forest ecosystem services. The combined effects of these two major factors have rarely been studied. Our study, therefore, aims to assess the combined effects of disturbances and climatic patterns on tropical forest regulating ecosystem services in Bangladesh, with a focus on rainfall regimes.
Most tropical forests historically have supported low-income forest-resource-dependent communities by providing fodder, firewood, and timber [7,8]. Disturbing small portions of forested areas or the removal of plant biomass is known as an ‘anthropogenic disturbance’ [9]. The effects of such disturbances on the aboveground and belowground biodiversity of tropical forests and the provision of ecosystem services have been investigated globally in recent years [10,11]. For anthropogenic disturbances, we refer to the following: decreases in plant population and densities [12], illegal tree felling/removal in forested areas and disrupted forest regeneration, the decrease in tree species richness and tree abundance [13], alterations in forest structure, the composition of tree species [12], and finally the decrease in tree functional diversity [10].
Global ongoing climate change also alters tropical forest biodiversity and health through imposing strong environmental filters within this unique ecosystem by climate extremes [14,15]. Tropical forests will face warmer temperatures by 2100 [16], leading to extreme drought events which can reduce overall forest ecosystem services by lessening net primary production [17]. Future climate change can also alter annual population growth [18] and enhance the chance of species elimination [19]. In fact, less precipitation can reduce trees’ natural regeneration and survival rate in dry forests, hypothetically changing the structure and composition of tropical ecosystems to new rigid states, such as shrubland or grassland [20]. Changing rainfall regimes also have a significant impact on tropical forests’ soil ecosystem services [21].
There has been growing evidence that anthropogenic disturbances and climate change can have complex (sometimes synergistic) effects on biological diversity [22]. It is likely that hot and arid environments are at a higher risk [23]. Yet, to our knowledge, the isolated and interactive effects of climate (mostly the degree of local rainfall) and anthropogenic disturbances on tropical forest ecosystem services have not fully been explored. To address this research gap, we examined two protected areas in Northeast Bangladesh, located in the most diverse and wettest region of the country [24,25]. The region has historically been subject to anthropogenic disturbances and is highly prone to global warming [11,26,27,28]. Our study assessed the combined effects of local rainfall regimes and anthropogenic disturbances on both the above and belowground ecosystem services in the area. We believe our study will improve our understanding of the separate and combined effects of rainfall patterns and anthropogenic disturbances on major forest ecosystem services, thus allowing us to predict (and possibly mitigate) forthcoming changes in forest vegetation and to restore this biodiverse forest ecosystem in the recent UN Decade on Ecosystem Restoration [29].
2. Materials and Methods
2.1. The Study Area
The present study was conducted in two forested, protected areas, namely Khadimnagar National Park (KNP) and Satchari National Park (SNP). These two forested, protected areas are located in the northeastern region of Bangladesh (Figure 1). Ecologically, both areas lie in tropical evergreen and semi-evergreen biogeographic zone. The Forest Department declared KNP as a national park in 2009 and SNP in 2005 [24]. The total areas of the KNP and SNP are 679 ha and 243 ha, respectively [30]. Geographically, the KNP lies between 24°56′ and 24°58′ N latitude and 91°55′ and 91°59′ E longitude. The SNP lies between 24°5′ and 24°10′ N latitude and 91°25′ and 91°30′ E longitude. The maximum and minimum mean temperatures of KNP are 30.7 °C and 18.9 °C, whereas the maximum and minimum mean temperatures of SNP are 34.8 °C and 9.6 °C, respectively. The total annual precipitation is 3931 mm/year−1 with July being the wettest month, having an average of about 1250 mm of rain [11]. In SNP, the average annual rainfall is 4162 mm [1]. The soil of Khadimnagar hills is slightly fertile compared to that of the SNP. The soil textures of both forested areas are sandy loams [11].
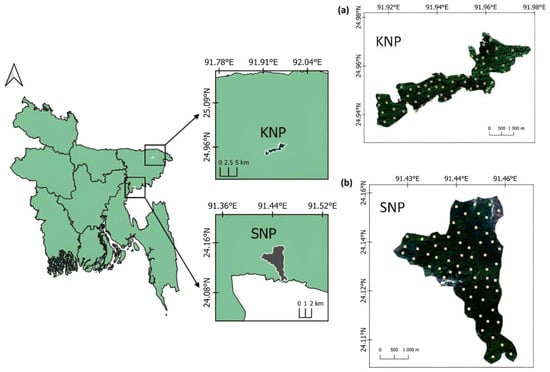
Figure 1.
Location map of two studied protected areas (a) Khadimnagar National Park (KNP) and (b) Satchari National Park (SNP) in Northeast Bangladesh. The yellow dots indicate the plot location within each of the study areas.
2.2. Sampling Design and Plot Selection
From each study area, 60 main plots were selected for tree data collection. The plot size was 20 × 20 m2. Within each main plot, 3 sampling sub-plots of 2 × 2 m2 were chosen randomly for shrub and herb species. Altogether, data were collected from 120 main plots and 180 sub-plots. Main plots were randomly generated through ArcGIS Desktop (Version 10.5.1, Redlands, California, USA) geoprocessing tool with surrounding areas. When a randomly generated plot location was unreachable, plots were carefully chosen from the adjacent forested areas with similar biophysical conditions. The boundaries of each of the main plots and sub-plots were pegged and marked before data collection.
2.3. Indicators of Forest Ecosystem Services
We measured effects of disturbances on nine forest ecosystem services based on 16 response parameters (Table 1).

Table 1.
List of responses associated with key ecosystem services in our study based on previous research.
2.4. Assessment of Disturbance and Ecosystem Services
2.4.1. Field Data Collection
Field data collection was conducted from August to October 2019. For each site, the general characteristics of each plot were recorded. Geographic coordinates and elevation were measured for topographical factors in the center of plots with a hand-held GPS (Garmin Ltd., Schaffhausen, Switzerland). Tree, shrub, and herb coverage was collected at the plot level. A mobile device with the application Compass (Xiaomi Compass 9.2.8, Beijing, China) was used to measure slopes (degree) [37]. Non-destructive approach was followed to accumulate tree biomass data. According to a previous study [27], all the stems with a diameter at breast height (DBH) ≥ 5 cm were considered trees, identified by species in the plot, and selected for height and DBH measurements. Diameter tape was used to measure over bark stem diameter and rangefinder was used for tree height calculation (LTI TruPulse 200, USA). The stem diameters of all stems were measured separately. Around 100 gm of tree biomass from each plant part, such as leaves, branches, twigs, bark, and roots, was collected at the species level for laboratory analysis. For dry weight determination and organic carbon analysis, the contents were put in plastic bags and the samples were carried to the laboratory. The tree canopy openness, number of cut stems, shrub and herb canopy openness, and bare ground percentage of plots were recorded as indicators of disturbances in each plot. Data were collected by a densitometer at the center of each plot to measure the tree canopy coverage. The effect of the severity of disturbances (harvesting and canopy openness due to removal of trees with stem diameter ≥ 5 cm) on carbon stocks was evaluated. In each sub-plots of shrubs and herbs, number of each species and individuals was counted to use in the calculation of species richness and abundance. Five soil samples were taken from each corner and the center of the plot to reduce any site homogeneity. Each of the samples was taken at 10 cm of depth using a core soil sampler. Then five soil samples from each plot were thoroughly mixed up together in the field and marked as one sample used to measure soil variables [38]. Then the samples were taken to the laboratory for further analysis.
2.4.2. Anthropogenic Disturbance Index
The magnitude of disturbance in the field was measured using the following formula [37]:
where DI = the disturbance index, CS (%) = percentage of cut stems, i.e., trees illegally felled and/or removed (number of cut stem/total stem of the plot), TCO = tree canopy openness, SHCO = shrub and herb canopy openness, and BG = the bare ground percentage of the plot [11].
The DI measures a comprehensive bioassay that incorporates all sorts of disturbance effects throughout recent history, such as windthrow, collection of fuelwood, illicit felling, logging, or forest fires.
2.4.3. Estimation of Tree Biomass and Carbon Stock
A non-destructive method developed by Brown et al. [39] was used to access the aboveground biomass (AGB) of trees. Previous studies suggested that this method is the most appropriate method for tropical forests [37,40,41]. Belowground biomass (BGB) was accessed by considering 15% of the AGB. Total biomass of each individual tree was calculated by adding AGB and BGB. Biomass of all species was added to obtain the plot-wise total biomass [37].
For the estimation of biomass carbon, fresh weight and oven-dried weight (48 h at 65 °C) of the collected plant samples were calculated. One (1.00) gm of oven-dried grind sample was taken in pre-weighted crucibles. The crucibles with the samples were placed in the furnace for one hour at 550 °C. Then the crucibles cooled down gradually inside the furnace. Then the weight of the crucibles with ash was measured. Afterward, the percentage of organic carbon was computed by Alamgir and Al-Amin [42].
Here, W1 = crucible weight; W2 = litter weight plus crucible weight; W3 = ash weight plus crucible weight; C = organic carbon; and “0.58” = considering 58% carbon in ash-free litter material.
Tree biomass carbon was calculated by multiplying AGB with the organic carbon percentage of that particular species. Plot-wise total AGB carbon was determined by adding the AGB carbon of all trees. Plot-specific biomass carbon stocks were also calculated and then converted to tons per hectare.
2.4.4. Soil Sample Analysis and Organic Carbon Estimation
Soil bulk density and soil moisture content were used as indicators of soil physical support and soil water regulation, respectively [21]. Relevant soil analysis was performed to quantify the bulk density (gm/cm3) and moisture content (in percent) of soil. Oven-drying method at 105 °C was used to measure soil bulk density in gm/cm3 [43]. Soil moisture content was also measured using the oven-drying method at 105 °C [44]. Soil aggregate stability was indicated by the soil sand % in this study [21]. Hydrometer method was used to access the soil sand % [45]. The corresponding carbon content of soils was calculated using a conversion coefficient of 0.58 multiplied by the soil organic matter [46]. Loss of ignition procedure was used to measure soil organic matter content of our study areas [21].
2.5. Statistical Analysis
All data were analyzed using R (Version 3.6.1, Vienna, Austria) and figures were produced using the packages “ggplot2” [47], “corrplot” for correlogram [48], “nlme” for analysis of variance, and raster package for bio-variable extraction. Before statistical analysis, data were transformed if conditions of normality were not met or to improve the homogeneity of variance. The relationships between disturbances and ecosystem services under two precipitation regimes were determined using regression analysis. All statistical significances were decided at 5% significance level.
3. Results
3.1. Aboveground Ecosystem Services
3.1.1. Primary Production
Tree biomass (t/ha) was used as an indicator of primary production in our study (Table 1). The disturbances had significant effects on primary production (p = 0.02, Table 2). Disturbances were negatively correlated with (p = 0.03, r = −0.28) the primary production of the study area under a medium amount of rainfall (Figure 2a). However, the association between disturbances and primary production was not significant (p = 0.3, r = −0.14) under a high-rainfall regime in our study (Figure 2a).

Table 2.
p value and slope extent of precipitation and disturbance effects on ecosystem services under different rainfall regimes in our study sites in Northeast Bangladesh. Significant codes: 0.0001 = ‘***’; 0.001 = ‘**’; 0.05 = ‘*’.
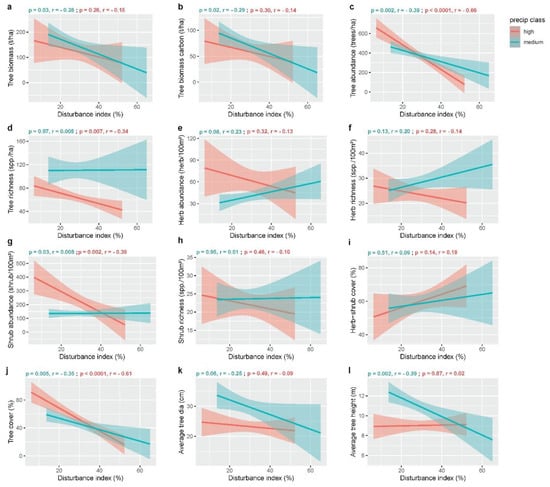
Figure 2.
Relationships between disturbance and aboveground ecosystem services under high- and medium-rainfall regimes in our study sites in Northeast Bangladesh.
3.1.2. Tree Carbon Storage
Tree carbon storage was represented by tree biomass carbon (t/ha) in our study (Table 1). Disturbances had significant effects on tree carbon storage (p = 0.02, Table 2). Aboveground carbon storage significantly decreased (p = 0.02, r = −0.29) along the disturbance gradient under a medium amount of rainfall, but it was not significantly (p = 0.3, r = −0.14) affected in the high-rainfall regime (Figure 2b).
3.1.3. Biodiversity Conservation
We considered tree abundance (tree/ha), tree species richness (spp./ha), herb abundance (herbs/100 m2), herb species richness (spp./100 m2), shrub abundance (shrubs/100 m2), and shrub species richness (spp./100 m2) as indicators of biodiversity conservation, one of the major regulating ecosystem services (Table 1). The interactive effects of disturbances and rainfall class were significant on tree abundance (p = 0.008, Table 2). Disturbances had a significant negative correlation with tree abundance under both rainfall classes (p = 0.002, r = −0.39 under a medium amount of rainfall; p < 0.0001, r = −0.66 under a high amount of rainfall), though the correlation was stronger in the high-rainfall class (Figure 2c). Rainfall class had significant effects on tree richness (p < 0.0001), herb abundance (p = 0.009), and herb richness (p = 0.01, Table 2). Disturbances had a significant negative correlation (p = 0.007, r = −0.34) with tree richness in the high-rainfall class, whereas no significant correlation (p = 0.97, r = 0.005) was found in the medium-rainfall class (Figure 2d). Herb and shrub communities are also good indicators of forest biodiversity. Herb abundance (p = 0.08, r = 0.23 in the medium-rainfall class; p = 0.32, r = −0.13 in the high-rainfall class) and richness (p = 0.13, r = 0.20 in the medium-rainfall class; p = 0.28, r = −0.14 in the high-rainfall class) had no significant correlation with disturbances in any rainfall class in our study (Figure 2e,f). The combined effects of disturbances and rainfall were significant on shrub abundance (p = 0.003, Table 2). Shrub abundance was also significantly correlated with a disturbance gradient in both rainfall classes (p = 0.03, r = 0.008 for the medium-rainfall class and p = 0.002, r = −0.39 for the high-rainfall class, Figure 2g).
3.1.4. Wildlife Habitat
Vegetation coverage was the main indicator of wildlife habitat provision, another important forest ecosystem service in our study (Table 1). We did not find any significant correlation between herb–shrub coverage and disturbance gradients in the high- (p = 0.5, r = 0.09) or medium (p = 0.1, r = 0.19)-rainfall classes in the study sites (Figure 2i). On the contrary, the combined effects of disturbances and rainfall were significant on tree canopy coverage (p = 0.04, Table 2). Moreover, tree canopy coverage had a significant negative association with disturbance gradients in both medium- (p = 0.005, r = −0.35) and high (p < 0.0001, r = −0.61)-rainfall classes (Figure 2j).
3.1.5. Timber Production
Average tree height (m) and average tree diameter at breast height (cm) were considered as indicators for timber production in our study (Table 1). The correlation analysis showed that the disturbances had no significant association with the average tree diameter under the high- (p = 0.06, r = −0.25) or medium (p = 0.49, r = −0.09)-rainfall classes (Figure 2k). However, the interactive effects of disturbances and rainfall class were significant on tree height (p = 0.01, Table 2). The average tree height showed a significant negative correlation with a disturbance gradient in the medium-rainfall class (p = 0.002, r = −0.39), whereas no significant (p = 0.87, r = 0.02) relation was found in the high-rainfall class (Figure 2k,I).
3.2. Belowground Ecosystem Services
3.2.1. Soil Organic Carbon Storage
Soil organic carbon (%) was the indicator of soil organic carbon storage in our study (Table 1). Rainfall class had significant effects on soil organic carbon storage (p < 0.0001, Table 2). Belowground organic carbon storage was positively correlated (r = 0.27) with disturbance gradients and it was significant (p = 0.04) in the medium-rainfall class. On the other hand, the correlation between disturbances and belowground carbon storage was not significant (p = 0.47, r = −0.10) in our study in the high-rainfall class (Figure 3a).
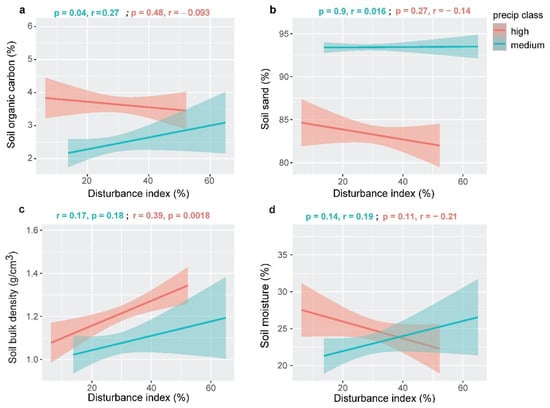
Figure 3.
Relationships between disturbance and belowground ecosystem services under high- and medium-rainfall regimes in our study sites in Northeast Bangladesh.
3.2.2. Soil Aggregate Stability
Soil aggregate stability is an important belowground soil ecosystem function that shows the ability of soil aggregates to resist degradation when exposed to external forces. We considered soil sand (%) as an indicator of soil aggregate stability (Table 1). A higher sand percentage lowers the aggregate stability of the soil. No significant correlation was observed between the disturbance gradient and soil sand (%) in the medium- (p = 0.9, r = 0.02) and high (p = 0.27, r = −0.14)-rainfall classes (Figure 3b) in our study.
3.2.3. Soil Physical Support
Soil physical support was indicated by soil bulk density (g/cm3) in our study (Table 1). A higher bulk density ensures stable sedimented soil in belowground forest ecosystems. Rainfall class had significant effects on soil bulk density (p <0.0001, Table 2). We observed a significant positive (p = 0.002, r = 0.39) correlation between disturbances and soil bulk density in the high-rainfall class, whereas no significant correlation (p = 0.18, r = 0.17) was observed in the medium-rainfall class (Figure 3c).
3.2.4. Soil Water Regulation
Soil moisture content (%) was the main indicator of soil water regulation in our study (Table 1). The combined effects of disturbances and rainfall were significant on soil moisture (p = 0.02, Table 2). Although the post hoc analysis showed no significant association between disturbances and soil moisture in any rainfall class, there was a positive correlation under a medium amount of rainfall and a negative correlation under a high amount of rainfall between disturbances and soil moisture in our study (Figure 3d).
4. Discussion
4.1. Aboveground Ecosystem Services
4.1.1. Primary Production
A significant decrease in primary production was observed with an increasing disturbance gradient in our study under a medium amount of rainfall. However, this decrease was not significant under a high amount of precipitation. Previous studies reported a decrease in rainfall and water availability and an increase in the physiological stress of plants, limiting biomass production and fitness, potentially leading to the death of stress-sensitive species [49]. Yan et al. [50] reported an increase in tree biomass with increased precipitation, which might be the reason why there were limited effects of disturbances on primary production in the present study.
4.1.2. Tree Carbon Storage
In our study, tree carbon storage significantly decreased with the increasing disturbance gradient under a medium-rainfall regime, whereas disturbances had no significant impact on tree carbon storage in high-rainfall areas. A significant decrease in tree biomass lowers the tree carbon storage under less rainfall, which was also observed by Fekete et al. [51] in their study.
4.1.3. Biodiversity
Our study observed a combined association of disturbances and rainfall class with tree abundance. Greater disturbances decreased the tree abundance in both rainfall classes and mostly in the high-rainfall class. We also found disturbances significantly lower the tree richness in the high-rainfall class, but this correlation was not significant in medium-rainfall areas. The previous study showed that anthropogenic disturbances reduced the tree species richness [52]. We observed this negative effect of disturbances on tree species abundance and richness under heavy rainfall. We found no significant changes in herb richness and abundance with a disturbance gradient in both the high- and medium-precipitation classes. However, the combined effects of disturbances and rainfall class were significant on shrub abundance. In the high-rainfall class, disturbances significantly lowered the shrub abundance, but in the medium-precipitation region, this correlation was almost linear.
4.1.4. Wildlife Habitat
The interaction effects of rainfall and disturbance class were significant on tree canopy coverage in our study. We observed disturbances lower the canopy coverage in high-rainfall areas rather than medium-rainfall regions. Pfeifer et al. [53] reported the canopy coverage changes under different precipitation patterns with the disturbances fully dependent on the forest’s adaptation capability under a deficit of water. Moreover, we found no interaction between herb and shrub coverage with the disturbance gradients in both medium- and high-rainfall regions. Due to the high restoring capacity of tropical understories, vegetation disturbances cannot alter their coverage significantly [54].
4.1.5. Timber Production
We observed the interaction of rainfall class and disturbances had a marginal correlation with average tree height, and in a specific trend analysis, we observed that highly disturbed areas contained species with a significantly lower average height in medium-rainfall regions, whereas no interaction was observed in high-rainfall areas. Disturbances are one of the main drivers of reduced tree density, diversity, and ecosystem production [52,55]. The present study revealed that disturbances can reduce tree height under a medium-precipitation regime. However, no significant interaction was observed in our study between the average diameter of trees and the disturbance gradients in both rainfall classes.
4.2. Belowground Ecosystem Services
4.2.1. Organic Carbon Storage
We observed a significant rise in soil organic carbon with disturbances in the medium-precipitation region, whereas in the high-rainfall region, this correlation was not significant. This is because anthropogenic disturbance can increase the illegal logging of tree species, which ultimately increases the tree debris and soil organic matter on the forest floor [38,52]. However, this positive correlation between disturbances and soil organic carbon was not observed under heavy rainfall because a higher amount of precipitation might decrease the soil organic carbon content on the forest floor due to a higher incidence of leaching or washout [56].
4.2.2. Soil Aggregate Stability
Disturbances had no significant correlation with soil aggregate stability in our study in both rainfall classes. The larger portion of sand content was deposited in our study sites due to the presence of numerous streams in the rainy season, which might be the main driving factor of soil aggregate stability, more so than the disturbance gradients.
4.2.3. Soil Physical Support
We found a significant positive association between disturbance and soil bulk density in our study in the medium-rainfall class. Our study’s findings are in accordance with those of a previous study which reported an increase in soil bulk density with forest disturbances. However, under the high-rainfall class, no significant correlation was evident in our study, as also reported by Rab et al. [57].
4.2.4. Soil Water Regulation
The interaction of disturbances and rainfall class had a significant effect on soil water regulation. However, disturbances had no significant association with soil water regulation function in both the medium- and high-rainfall classes in our study.
5. Conclusions
The combined effects of climate change and anthropogenic disturbances have caused a substantial decline in the supply of ecosystem services from tropical forested regions in recent decades. Bangladesh is one of the countries most adversely affected by climate change with a major variability in rainfall patterns in recent decades. Our study found that disturbances negatively influenced indicators of primary production, tree carbon storage, and timber production, and may positively influence soil organic carbon storage under different rainfall regimes. There are, however, some indicators that we used in our study as response factors that could also be used as causal factors (e.g., soil fertility), and thus our results need careful interpretation. We believe our study will contribute to our understanding of the combined effects of rainfall regimes and disturbances on forest ecosystem services with potential implications for forest ecosystem restoration.
Author Contributions
Conceptualization, F.S., M.A.S.A.-K. and S.A.M.; methodology, M.A.S.A.-K., S.A.M.; formal analysis, F.S. and M.R.K.; investigation, F.S. and M.R.K.; resources, M.A.S.A.-K.; data curation, F.S. and M.R.K.; writing—original draft preparation, F.S. and M.R.K.; writing—review and editing, M.A.S.A.-K. and S.A.M.; supervision, M.A.S.A.-K.; project administration, M.A.S.A.-K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Relevant data used in this study will be made available upon request.
Acknowledgments
We acknowledge the lab and other support provided by the Department of Forestry and Environmental Science (FES), Shahjalal University of Science and Technology (SUST), Bangladesh, which have made this study possible. We also acknowledge the funding of SUST Research center (Project ID: FES/2019/2/01; FES/2020/01/03; FES/2021/2/04).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mukul, S.A.; Sohel, M.S.I.; Herbohn, J.; Inostroza, L.; König, H. Integrating ecosystem services supply potential from future land-use in protected area management: A Bangladesh case study. Ecosyst. Serv. 2017, 26, 355–364. [Google Scholar] [CrossRef]
- Ninan, K.N.; Kontoleon, A. Valuing forest ecosystem services and disservices—Case study of a protected area in India. Ecosyst. Serv. 2016, 20, 1–14. [Google Scholar] [CrossRef]
- Hofhansl, F.; Chacón-Madrigal, E.; Fuchslueger, L.; Jenking, D.; Morera-Beita, A.; Plutzar, C.; Silla, F.; Andersen, K.M.; Buchs, D.M.; Dullinger, S.; et al. Climatic and edaphic controls over tropical forest diversity and vegetation carbon storage. Sci. Rep. 2020, 10, 5066. [Google Scholar] [CrossRef] [PubMed]
- Mukul, S.A.; Halim, M.A.; Herbohn, J. Forest Carbon Stock and Fluxes: Distribution, Biogeochemical Cycles, and Measurement Techniques. In Life on Land. Encyclopedia of the UN Sustainable Development Goals; Leal Filho, W., Azul, A.M., Brandli, L., Lange Salvia, A., Wall, T., Eds.; Springer: Cham, Switzerland, 2021; pp. 376–385. [Google Scholar] [CrossRef]
- Bullock, E.L.; Woodcock, C.E.; Olofsson, P. Monitoring tropical forest degradation using spectral unmixing and Landsat time series analysis. Remote Sens. Environ. 2020, 238, 110968. [Google Scholar] [CrossRef]
- Morris, R.J. Anthropogenic impacts on tropical forest biodiversity: A network structure and ecosystem functioning perspective. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3709–3718. [Google Scholar] [CrossRef]
- Arfin-Khan, M.A.S.; Saimun, M.S.R. Forest Dependency: Status, Assessment Tools, and Influencing Factors. In Life on Land. Encyclopedia of the UN Sustainable Development Goals; Leal Filho, W., Azul, A.M., Brandli, L., Lange Salvia, A., Wall, T., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Nerfa, L.; Rhemtulla, J.M.; Zerriffi, H. Forest dependence is more than forest income: Development of a new index of forest product collection and livelihood resources. World Dev. 2020, 125, 104689. [Google Scholar] [CrossRef]
- Antongiovanni, M.; Venticinque, E.M.; Matsumoto, M.; Fonseca, C.R. Chronic anthropogenic disturbance on Caatinga dry forest fragments. J. Appl. Ecol. 2020, 57, 2064–2074. [Google Scholar] [CrossRef]
- Barlow, J.; Lennox, G.D.; Ferreira, J.; Berenguer, E.; Lees, A.C.; Nally, R.M.; Thomson, J.R.; Ferraz, S.F.B.; Louzada, J.; Oliveira, V.H.F.; et al. Anthropogenic disturbance in tropical forests can double biodiversity loss from deforestation. Nature 2016, 535, 144–147. [Google Scholar] [CrossRef]
- Karim, M.R.; Sultana, F.; Saimun, M.S.R.; Mukul, S.A.; Arfin-Khan, M.A.S. Plant diversity and local rainfall regime mediate soil ecosystem functions in tropical forests of north-east Bangladesh. Environ. Adv. 2020, 2, 100022. [Google Scholar] [CrossRef]
- de Oliveira Roque, F.; Menezes, J.F.S.; Northfield, T.; Ochoa-Quintero, J.M.; Campbell, M.J.; Laurance, W.F. Warning signals of biodiversity collapse across gradients of tropical forest loss. Sci. Rep. 2018, 8, 1622. [Google Scholar] [CrossRef]
- Ribeiroa, M.C.; Metzger, J.P.; Martensen, A.C.; Ponzoni, F.J.; Hirota, M.M. The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biol. Conserv. 2009, 142, 1141–1153. [Google Scholar] [CrossRef]
- Karim, M.R.; Mukul, S.A.; Zahira, R.B.; Saimun, M.S.R.; Arfin-Khan, M.A.S. The role of protected areas co-management in enhancing resistance and resilience of deciduous forest ecosystem to extreme climatic events in Bangladesh. J. Environ. Manag. 2023, 326, 116800. [Google Scholar] [CrossRef]
- Moro, M.F.; Silva, I.A.; Araújo, F.S.; Nic Lughadha, E.; Meagher, T.R.; Martins, F.R. The role of edaphic environment and climate in structuring phylogenetic pattern in seasonally dry tropical plant communities. PLoS ONE 2015, 10, e0119166. [Google Scholar] [CrossRef]
- Smith, M.N.; Taylor, T.C.; van Haren, J.; Rosolem, R.; Restrepo-Coupe, N.; Adams, J.; Wu, J.; de Oliveira, R.C.; da Silva, R.; de Araujo, A.C.; et al. Empirical evidence for resilience of tropical forest photosynthesis in a warmer world. Nat. Plants 2020, 6, 1225–1230. [Google Scholar] [CrossRef]
- Zhao, M.; Running, S.W. Drought-induced reduction in global terrestrial net primary production from 2000 through 2009. Science 2010, 329, 940. [Google Scholar] [CrossRef]
- Ureta, C.; Martorell, C.; Hortal, J.; Fornoni, J. Assessing extinction risks under the combined effects of climate change and human disturbance through the analysis of life-history plasticity. Perspect. Plant Ecol. Evol. Syst. 2012, 14, 393–401. [Google Scholar] [CrossRef]
- Deb, J.C.; Phinn, S.; Butt, N.; McAlpine, C.A. Climate change impacts on tropical forests: Identifying risks for tropical Asia. J. Trop. For. Sci. 2018, 30, 182–194. [Google Scholar] [CrossRef]
- Anderson-Teixeira, K.J.; Miller, A.D.; Mohan, J.E.; Hudiburg, T.W.; Duval, B.D.; DeLucia, E.H. Altered dynamics of forest recovery under a changing climate. Glob. Chang. Biol. 2013, 19, 2001–2021. [Google Scholar] [CrossRef]
- Karim, M.R.; Halim, M.A.; Gale, N.V.; Thomas, S.C. Biochar effects on soil physiochemical properties in degraded managed ecosystems in northeastern Bangladesh. Soil Syst. 2020, 4, 69. [Google Scholar] [CrossRef]
- García-Valdés, R.; Svenning, J.C.; Zavala, M.A.; Purves, D.W.; Araújo, M.B. Evaluating the combined effects of climate and land-use change on tree species distributions. J. Appl. Ecol. 2015, 52, 902–912. [Google Scholar] [CrossRef]
- Gibb, H.; Sanders, N.J.; Dunn, R.R.; Watson, S.; Photakis, M.; Abril, S.; Andersen, A.N.; Angulo, E.; Armbrecht, I.; Arnan, X.; et al. Climate mediates the effects of disturbance on ant assemblage structure. Proc. R. Soc. B Biol. Sci. 2015, 282, 20150418. [Google Scholar] [CrossRef] [PubMed]
- Mukul, S.A.; Rashid, A.Z.M.M.; Khan, N.A. Forest protected area systems and biodiversity conservation in Bangladesh. In Protected Areas: Policies, Management and Future Directions; Mukul, S.A., Rashid, A.Z.M.M., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2017; pp. 157–177. [Google Scholar]
- Tantipisanuh, N.; Savini, T.; Cutter, P.; Gale, G.A. Biodiversity gap analysis of the protected area system of the Indo-Burma Hotspot and priorities for increasing biodiversity representation. Biol. Conserv. 2016, 195, 203–213. [Google Scholar] [CrossRef]
- Langner, A.; Miettinen, J.; Kukkonen, M.; Vancutsem, C.; Simonetti, D.; Vieilledent, G.; Verhegghen, A.; Gallego, J.; Stibig, H. Towards operational monitoring of forest canopy disturbance in evergreen rain forests: A test case in continental Southeast Asia. Remote Sens. 2018, 10, 544. [Google Scholar] [CrossRef]
- Mukul, S.A.; Saha, N. Conservation benefits of tropical multifunctional landscapes in and around a forest protected area of Bangladesh. Land 2017, 6, 2. [Google Scholar] [CrossRef]
- Corbera, E.; Hunsberger, C.; Vaddhanaphuti, C. Climate change policies, land grabbing and conflict: Perspectives from Southeast Asia. Can. J. Dev. Stud./Rev. Can. D’études Du Développement 2017, 38, 297–304. [Google Scholar] [CrossRef]
- Ota, L.; Chazdon, R.L.; Herbohn, J.; Gregorio, N.; Mukul, S.A.; Wilson, S.J. Achieving quality forest and landscape restoration in the tropics. Forests 2020, 11, 820. [Google Scholar] [CrossRef]
- Mukul, S.A.; Herbohn, J.; Rashid, A.Z.M.M.; Uddin, M.B. Comparing the effectiveness of forest law enforcement and economic incentive to prevent illegal logging in Bangladesh. Int. For. Rev. 2014, 16, 363–375. [Google Scholar] [CrossRef]
- Richmond, A.; Kaufmann, R.K.; Myneni, R.B. Valuing ecosystem services: A shadow price for net primary production. Ecol. Econ. 2007, 64, 454–462. [Google Scholar] [CrossRef]
- Fahey, T.J.; Woodbury, P.B.; Battles, J.J.; Goodale, C.L.; Hamburg, S.P.; Ollinger, S.V.; Woodall, C.W. Forest carbon storage: Ecology, management, and policy. Front. Ecol. Environ. 2010, 8, 245–252. [Google Scholar] [CrossRef]
- Gould, W. Remote sensing of vegetation, plant species richness, and regional biodiversity hotspots. Ecol. Appl. 2000, 10, 1861–1870. [Google Scholar] [CrossRef]
- Grebner, D.L.; Bettinger, P.; Siry, J.P. Wildlife habitat relationships. In Introduction to Forestry and Natural Resources; Grebner, D.L., Bettinger, P., Siry, J.P., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 125–146. [Google Scholar]
- Aishan, T.; Halik, Ü.; Betz, F.; Tiyip, T.; Ding, J.; Nuermaimaiti, Y. Stand structure and height-diameter relationship of a degraded Populus euphratica forest in the lower reaches of the Tarim River, Northwest China. J. Arid. Land 2015, 7, 544–554. [Google Scholar] [CrossRef]
- Doran, J.W.; Parkin, T.B. Quantitative Indicators of Soil Quality: A Minimum Data Set. Methods Assess. Soil Qual. 2015, 49, 25–37. [Google Scholar]
- Saimun, M.S.R.; Karim, M.R.; Sultana, F.; Arfin-Khan, M.A.S. Multiple drivers of tree and soil carbon stock in the tropical forest ecosystems of Bangladesh. Trees For. People 2021, 5, 100108. [Google Scholar] [CrossRef]
- Mukul, S.A.; Herbohn, J.; Ferraen, A.; Congdon, R. Limited role of shifting cultivation in soil carbon and nutrients recovery in regenerating tropical secondary forests. Front. Environ. Sci. 2022, 10, 1076506. [Google Scholar] [CrossRef]
- Brown, S.; Gillespie, A.; Lugo, A.E. Biomass Estimation Methods for Tropical Forests with Applications to Forest Inventory Data. For. Sci. 1989, 35, 881–902. [Google Scholar] [CrossRef]
- Alves, D.S.; Soares, J.V.S.; Amaral, E.M.K.; Mello, S.A.S.; Almeida, O.; Fernandes, S.A.; Silveira, A.M. Biomass of primary and secondary vegetation in Rondonia, western Brazilian Amazon. Glob. Chang. Biol. 1997, 3, 451–462. [Google Scholar] [CrossRef]
- FAO. Estimating Biomass and Biomass Change of Tropical Forests—A Primer; FAO: Rome, Italy, 1997; Available online: http://www.fao.org/docrep/w4095e/w4095e00.htm (accessed on 25 December 2022).
- Alamgir, M.; Al-Amin, M. Allometric models to estimate biomass organic carbon stock in forest vegetation. J. For. Res. 2008, 19, 101–106. [Google Scholar] [CrossRef]
- Blake, G.R.; Hartge, K.H. Bulk Density. In Methods of Soil Analysis, 2nd ed.; Klute, A., Ed.; Wiley & Sons: Hoboken, NJ, USA, 1986; pp. 363–375. [Google Scholar]
- O’Kelly, B.C. Accurate determination of moisture content of organic soils using the oven drying method. Dry. Technol. 2004, 22, 1767–1776. [Google Scholar] [CrossRef]
- Bouyoucos, G.J. Hydrometer Method Improved for Making Particle Size Analyses of Soils. Agron. J. 1962, 54, 464–465. [Google Scholar] [CrossRef]
- Wang, S.; Huang, Y. Determinants of soil organic carbon sequestration and its contribution to ecosystem carbon sinks of planted forests. Glob. Chang. Biol. 2020, 26, 3163–3173. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Wei, T.; Simko, V. R Package ‘Corrplot’: Visualization of a Correlation Matrix. (Version 0.92). 2021. Available online: https://github.com/taiyun/corrplot (accessed on 3 January 2023).
- Figueiredo, K.V.; Oliveira, M.T.; Arruda, E.C.P.; Silva, B.C.F.; Santos, M.G. Changes in leaf epicuticular wax, gas exchange and biochemistry metabolism between Jatropha mollissima and Jatropha curcas under semi-arid conditions. Acta Physiol. Plant. 2015, 37, 108. [Google Scholar] [CrossRef]
- Yan, H.; Liang, C.; Li, Z.; Liu, Z.; Miao, B.; He, C.; Sheng, L. Impact of Precipitation Patterns on Biomass and Species Richness of Annuals in a Dry Steppe. PLoS ONE 2015, 10, e0125300. [Google Scholar] [CrossRef]
- Fekete, I.; Lajtha, K.; Kotroczó, Z.; Várbíró, G.; Varga, C.; Tóth, J.A.; Demeter, I.; Veperdi, G.; Berki, I. Long-term effects of climate change on carbon storage and tree species composition in a dry deciduous forest. Glob. Chang. Biol. 2017, 23, 3154–3168. [Google Scholar] [CrossRef]
- Bentsi-Enchill, F.; Damptey, F.G.; Pappoe, A.N.M.; Ekumah, B.; Akotoye, H.K. Impact of anthropogenic disturbance on tree species diversity, vegetation structure and carbon storage potential in an upland evergreen forest of Ghana, West Africa. Trees For. People 2022, 8, 100238. [Google Scholar] [CrossRef]
- Pfeifer, M.; Gonsamo, A.; Woodgate, W.; Cayuela, L.; Marshall, A.R.; Ledo, A.; Paine, T.C.; Marchant, R.; Burt, A.; Calders, K.; et al. Tropical forest canopies and their relationships with climate and disturbance: Results from a global dataset of consistent field-based measurements. For. Ecosyst. 2018, 5, 7. [Google Scholar] [CrossRef]
- Kutnar, L.; Nagel, T.A.; Kermavnar, J. Effects of Disturbance on Understory Vegetation across Slovenian Forest Ecosystems. Forests 2019, 10, 1048. [Google Scholar] [CrossRef]
- Gautam, T.P.; Mandal, T.N. Effect of disturbance on biomass, production and carbon dynamics in moist tropical forest of eastern Nepal. For. Ecosyst. 2016, 3, 11. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, D.; Liang, G.; Qiu, Q.; Liu, J.; Zhou, G.; Liu, S.; Chu, G.; Yan, J. Effects of precipitation on soil organic carbon fractions in three subtropical forests in southern China. J. Plant Ecol. 2016, 9, 10–19. [Google Scholar] [CrossRef]
- Rab, A.; Bradshaw, J.; Campbell, R.; Murphy, S. Review of Factors Affecting Disturbance, Compaction and Trafficability of Soils with Particular Reference to Timber Harvesting in the Forests of South-West Western Australia; University of Melbourne: Parkville, Australia, 2005. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).