The Role of Provenance for the Projected Growth of Juvenile European Beech under Climate Change
Abstract
1. Introduction
2. Materials and Methods
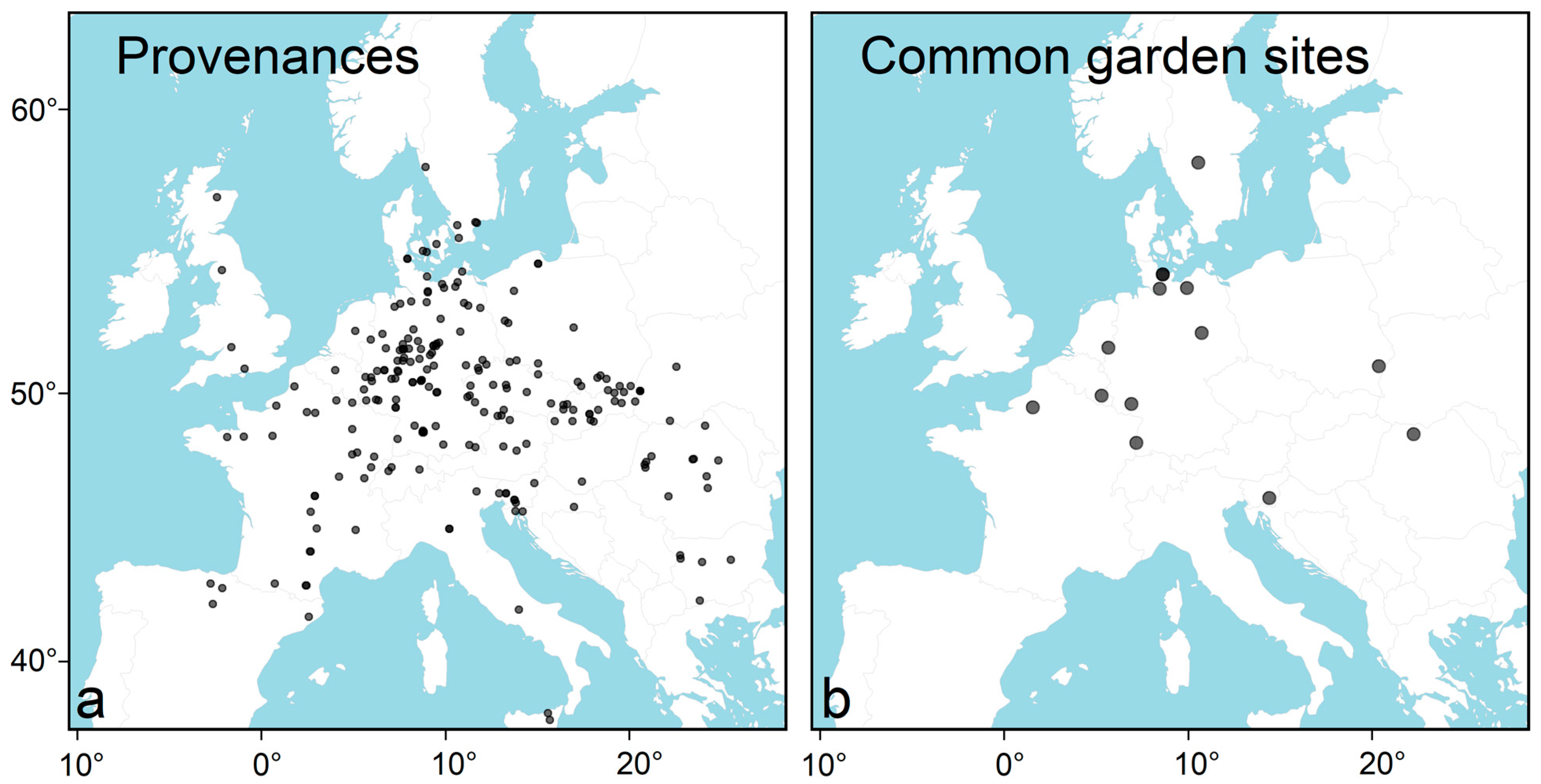
2.1. Phenotypic Data
2.2. Climatic Data
2.3. Model Development
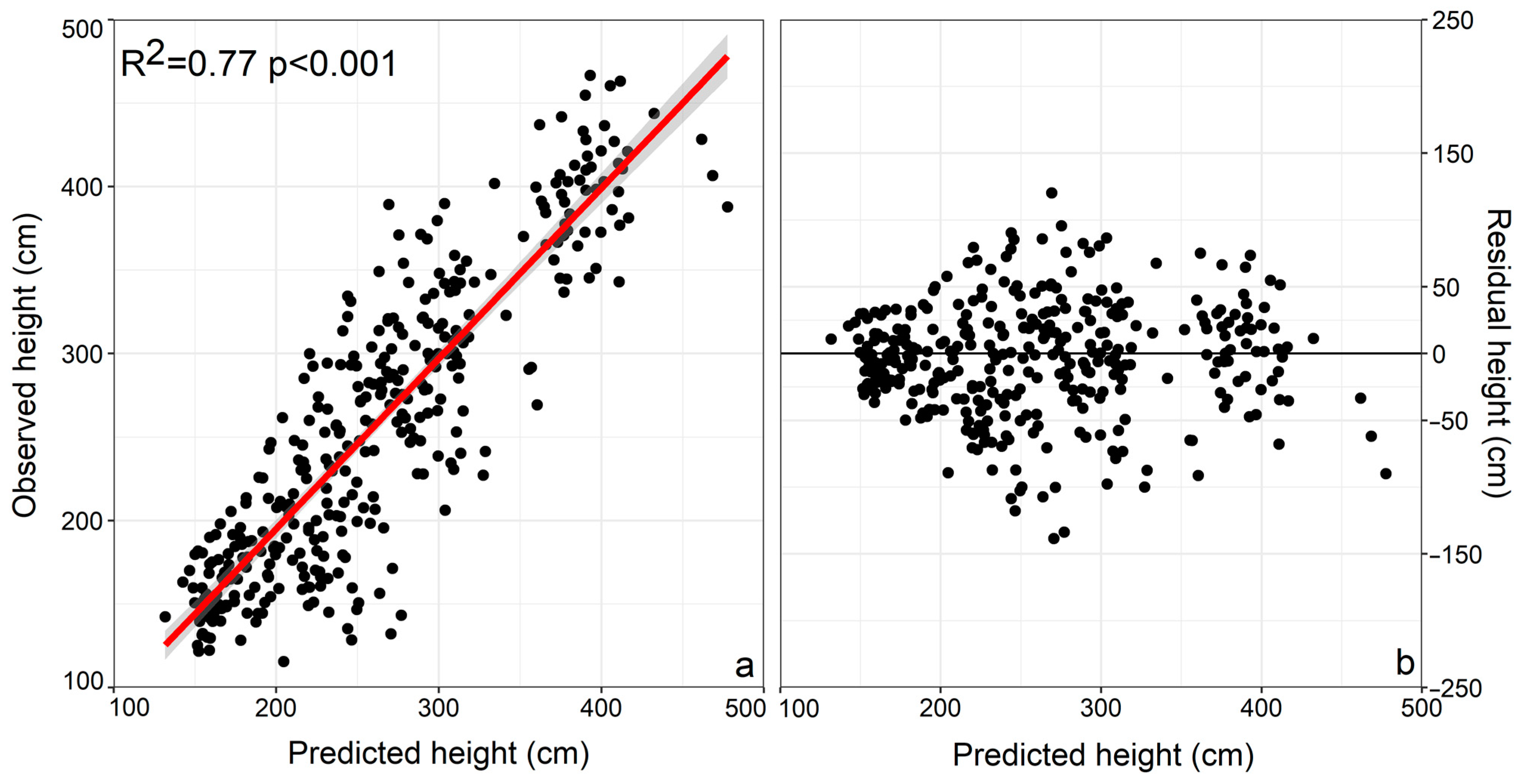
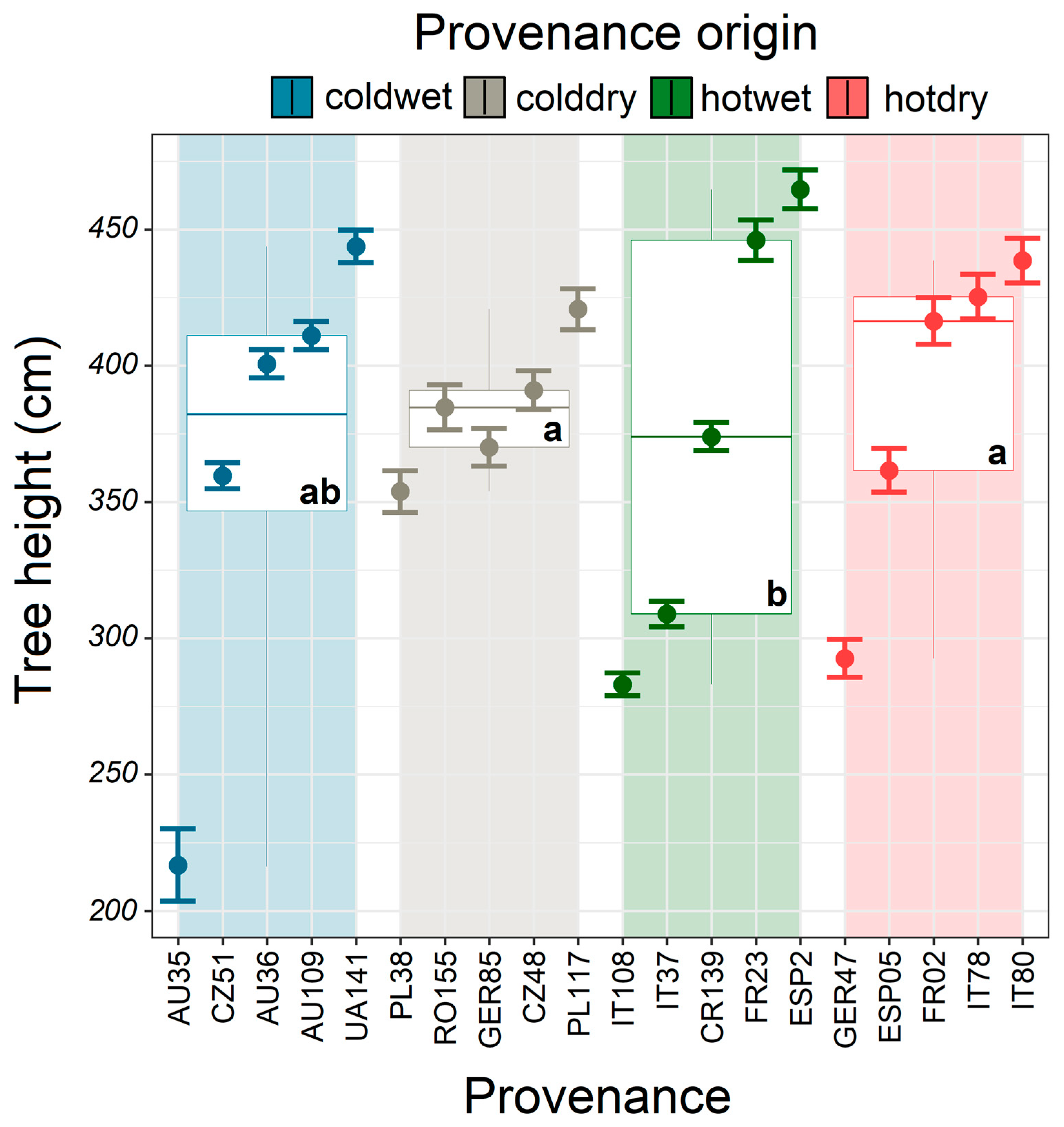
2.4. Statistical Analysis
3. Results
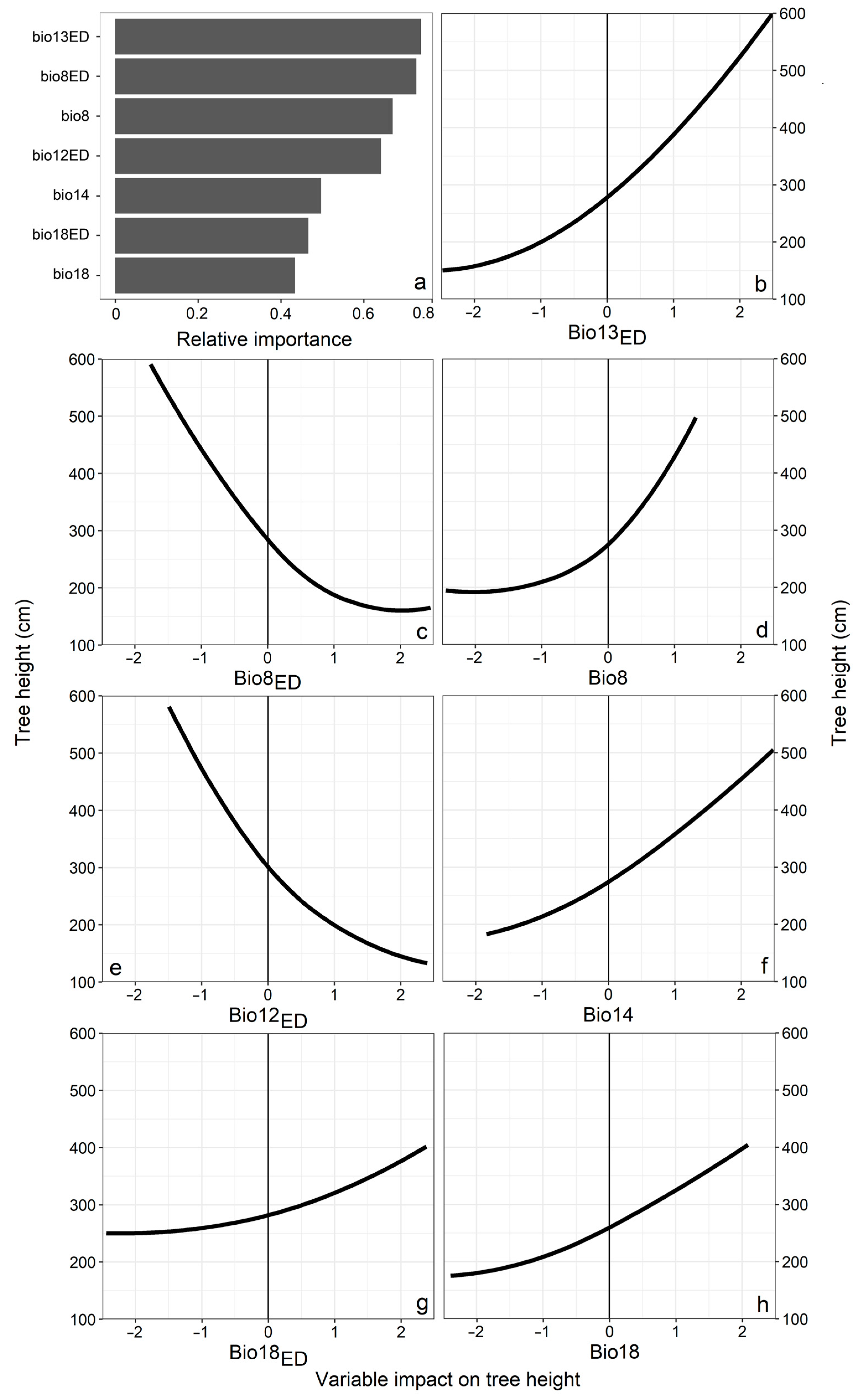
3.1. Climatic Variable Importance in Model
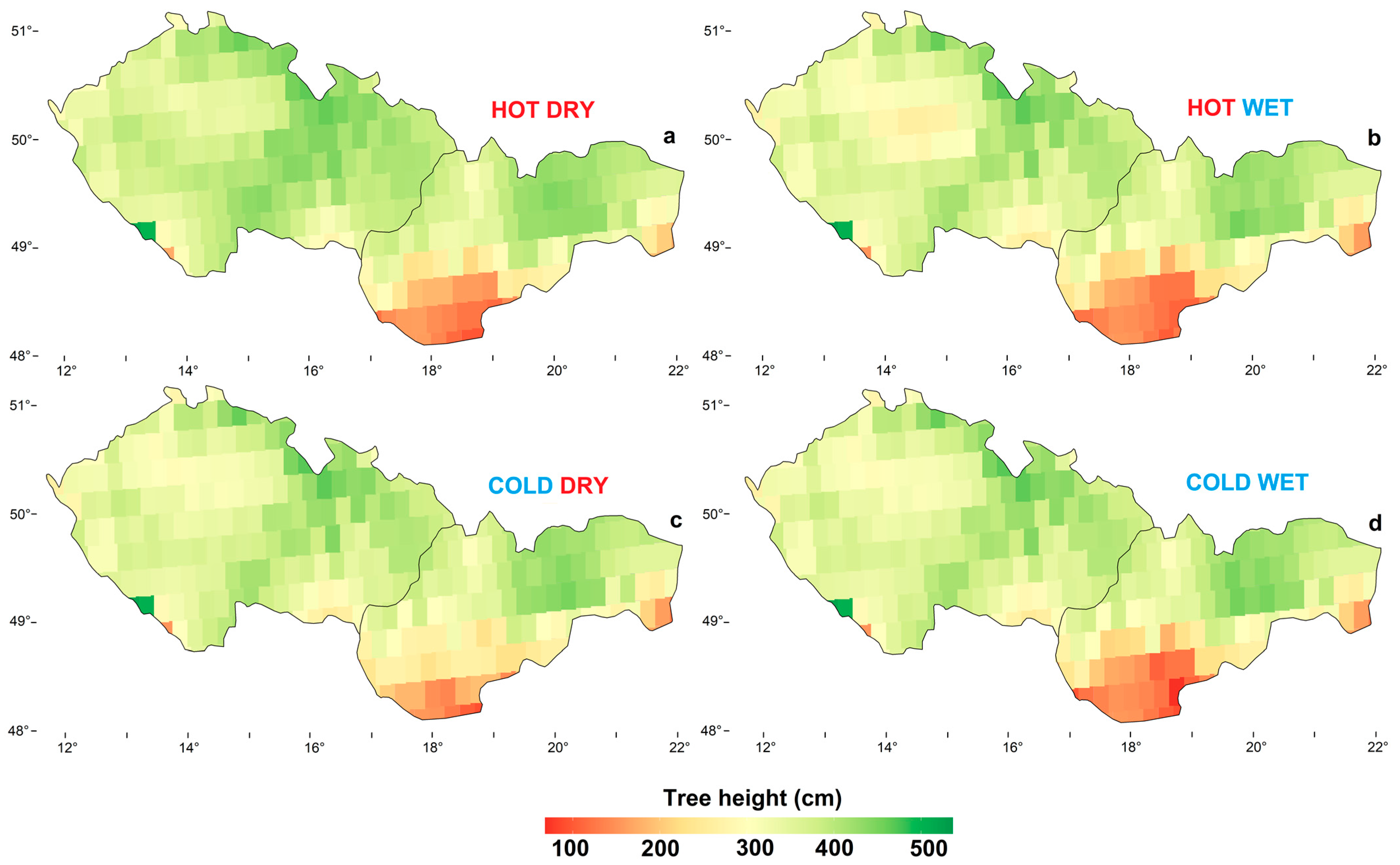
3.2. Model Climatic Limits and Provenance Grouping
3.3. Spatial Height Growth Predictions
4. Discussion
4.1. Ecodistance as a Predictor of Provenance Response
4.2. Limitations of the Study
4.3. High Phenotypic Plasticity of European Beech
4.4. Drought and Temperature Limited Growth
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dyderski, M.K.; Paź, S.; Frelich, L.E.; Jagodziński, A.M. How much does climate change threaten European forest tree species distributions? Glob. Chang. Biol. 2018, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- Schuldt, B.; Buras, A.; Arend, M.; Vitasse, Y.; Beierkuhnlein, C.; Damm, A.; Gharun, M.; Grams, T.E.E.; Hauck, M.; Hajek, P.; et al. A first assessment of the impact of the extreme 2018 summer drought on Central European forests. Basic Appl. Ecol. 2020, 45, 86–103. [Google Scholar] [CrossRef]
- Bandh, S.A.; Shafi, S.; Peerzada, M.; Rehman, T.; Bashir, S.; Wani, S.A.; Dar, R. Multidimensional analysis of global climate change: A review. Environ. Sci. Pollut. Res. 2021, 28, 24872–24888. [Google Scholar] [CrossRef] [PubMed]
- Szatniewska, J.; Zavadilova, I.; Nezval, O.; Krejza, J.; Petrik, P.; Čater, M.; Stojanović, M. Species-specific growth and transpiration response to changing environmental conditions in floodplain forest. For. Ecol. Manag. 2022, 516, 120248. [Google Scholar] [CrossRef]
- Gessler, A.; Keitel, C.; Kreuzwieser, J.; Matyssek, R.; Seiler, W.; Rennenberg, H. Potential risks for European beech (Fagus sylvatica L.) in a changing climate. Trees 2006, 21, 1–11. [Google Scholar] [CrossRef]
- Etzold, S.; Ziemińska, K.; Rohner, B.; Bottero, A.; Bose, A.K.; Ruehr, N.K.; Zingg, A.; Rigling, A. One Century of Forest Monitoring Data in Switzerland Reveals Species- and Site-Specific Trends of Climate-Induced Tree Mortality. Front. Plant Sci. 2019, 10, 307. [Google Scholar] [CrossRef]
- Pretzsch, H.; del Río, M.; Grote, R.; Klemmt, H.-J.; Ordóñez, C.; Oviedo, F.B. Tracing Drought Effects from the Tree to the Stand Growth in Temperate and Mediterranean Forests: Insights and Consequences for Forest Ecology and Management. Eur. J. Forest. Res. 2022, 141, 727–751. [Google Scholar] [CrossRef]
- Hlásny, T.; Mátyás, C.; Seidl, R.; Kulla, L.; Merganičová, K.; Trombik, J.; Dobor, L.; Barcza, Z.; Konôpka, B. Climate change increases the drought risk in Central European forests: What are the options for adaptation? For. J. 2014, 60, 5–18. [Google Scholar] [CrossRef]
- Rita, A.; Camarero, J.J.; Nolè, A.; Borghetti, M.; Brunetti, M.; Pergola, N.; Serio, C.; Vicente-Serrano, S.M.; Tramutoli, V.; Ripullone, F. The impact of drought spells on forests depends on site conditions: The case of 2017 summer heat wave in southern Europe. Glob. Chang. Biol. 2019, 26, 851–863. [Google Scholar] [CrossRef]
- Vitasse, Y.; Bottero, A.; Cailleret, M.; Bigler, C.; Fonti, P.; Gessler, A.; Lévesque, M.; Rohner, B.; Weber, P.; Rigling, A.; et al. Contrasting resistance and resilience to extreme drought and late spring frost in five major European tree species. Glob. Chang. Biol. 2019, 25, 3781–3792. [Google Scholar] [CrossRef]
- Leuschner, C. Drought response of European beech (Fagus sylvatica L.)—A review. Perspect. Plant Ecol. Evol. Syst. 2020, 47, 125576. [Google Scholar] [CrossRef]
- Petrík, P.; Zavadilová, I.; Šigut, L.; Kowalska, N.; Petek-Petrik, A.; Szatniewska, J.; Jocher, G.; Pavelka, M. Impact of Environmental Conditions and Seasonality on Ecosystem Transpiration and Evapotranspiration Partitioning (T/ET Ratio) of Pure European Beech Forest. Water 2022, 14, 3015. [Google Scholar] [CrossRef]
- Bigler, C.; Vitasse, Y. Premature leaf discoloration of European deciduous trees is caused by drought and heat in late spring and cold spells in early fall. Agric. For. Meteorol. 2021, 307, 108492. [Google Scholar] [CrossRef]
- Húdoková, H.; Petrik, P.; Petek-Petrik, A.; Konôpková, A.; Leštianska, A.; Střelcová, K.; Kmeť, J.; Kurjak, D. Heat-stress response of photosystem II in five ecologically important tree species of European temperate forests. Biologia 2022, 77, 671–680. [Google Scholar] [CrossRef]
- Kunert, N.; Hajek, P. Shade-tolerant temperate broad-leaved trees are more sensitive to thermal stress than light-demanding species during a moderate heatwave. Trees For. People 2022, 9, 100282. [Google Scholar] [CrossRef]
- Sangüesa-Barreda, G.; Di Filippo, A.; Piovesan, G.; Rozas, V.; Di Fiore, L.; García-Hidalgo, M.; García-Cervigón, A.I.; Muñoz-Garachana, D.; Baliva, M.; Olano, J.M. Warmer springs have increased the frequency and extension of late-frost defoliations in southern European beech forests. Sci. Total Environ. 2021, 775, 145860. [Google Scholar] [CrossRef]
- Arend, M.; Link, R.M.; Zahnd, C.; Hoch, G.; Schuldt, B.; Kahmen, A. Lack of hydraulic recovery as a cause of post-drought foliage reduction and canopy decline in European beech. New Phytol. 2022, 234, 1195–1205. [Google Scholar] [CrossRef]
- Sperlich, D.; Nadal-Sala, D.; Gracia, C.; Kreuzwieser, J.; Hanewinkel, M.; Yousefpour, R. Gains or Losses in Forest Productivity under Climate Change? The Uncertainty of CO2 Fertilization and Climate Effects. Climate 2020, 8, 141. [Google Scholar] [CrossRef]
- Huang, W.; Fonti, P.; Larsen, J.B.; Ræbild, A.; Callesen, I.; Pedersen, N.B.; Hansen, J.K. Projecting tree-growth responses into future climate: A study case from a Danish-wide common garden. Agric. For. Meteorol. 2017, 247, 240–251. [Google Scholar] [CrossRef]
- del Castillo, E.M.; Zang, C.S.; Buras, A.; Hacket-Pain, A.; Esper, J.; Serrano-Notivoli, R.; Hartl, C.; Weigel, R.; Klesse, S.; de Dios, V.R.; et al. Climate-change-driven growth decline of European beech forests. Commun. Biol. 2022, 5, 163. [Google Scholar] [CrossRef]
- Arend, M.; Sever, K.; Pflug, E.; Gessler, A.; Schaub, M. Seasonal photosynthetic response of European beech to severe summer drought: Limitation, recovery and post-drought stimulation. Agric. For. Meteorol. 2016, 220, 83–89. [Google Scholar] [CrossRef]
- Brunn, M.; Hafner, B.D.; Zwetsloot, M.J.; Weikl, F.; Pritsch, K.; Hikino, K.; Ruehr, N.K.; Sayer, E.J.; Bauerle, T.L. Carbon allocation to root exudates is maintained in mature temperate tree species under drought. New Phytol. 2022, 235, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Kolář, T.; Čermák, P.; Trnka, M.; Žid, T.; Rybníček, M. Temporal changes in the climate sensitivity of Norway spruce and European beech along an elevation gradient in Central Europe. Agric. For. Meteorol. 2017, 239, 24–33. [Google Scholar] [CrossRef]
- Šimůnek, V.; Vacek, Z.; Vacek, S.; Králíček, I.; Vančura, K. Growth Variability of European Beech (Fagus sylvatica L.) Natural Forests: Dendroclimatic Study from Krkonoše National Park. Cent. Eur. For. J. 2019, 65, 92–102. [Google Scholar] [CrossRef]
- Bosela, M.; Štefančík, I.; Petráš, R.; Vacek, S. The effects of climate warming on the growth of European beech forests depend critically on thinning strategy and site productivity. Agric. For. Meteorol. 2016, 222, 21–31. [Google Scholar] [CrossRef]
- Bosela, M.; Lukac, M.; Castagneri, D.; Sedmák, R.; Biber, P.; Carrer, M.; Konôpka, B.; Nola, P.; Nagel, T.A.; Popa, I.; et al. Contrasting effects of environmental change on the radial growth of co-occurring beech and fir trees across Europe. Sci. Total Environ. 2017, 615, 1460–1469. [Google Scholar] [CrossRef]
- Williams, M.I.; Dumroese, R.K. Preparing for Climate Change: Forestry and Assisted Migration. J. For. 2013, 111, 287–297. [Google Scholar] [CrossRef]
- Gömöry, D.; Krajmerová, D.; Hrivnák, M.; Longauer, R. Assisted Migration vs. Close-to-Nature Forestry: What Are the Prospects for Tree Populations under Climate Change? Cent. Eur. For. J. 2020, 66, 63–70. [Google Scholar] [CrossRef]
- Lortie, C.J.; Hierro, J.L. A Synthesis of Local Adaptation to Climate through Reciprocal Common Gardens. J. Ecol. 2022, 110, 1015–1021. [Google Scholar] [CrossRef]
- Konôpková, A.; Pšidová, E.; Kurjak, D.; Stojnić, S.; Petrík, P.; Fleischer, P., Jr.; Kučerová, J.; Ježík, M.; Petek, A.; Gömöry, D.; et al. Photosynthetic Performance of Silver Fir (Abies alba) of Different Origins under Suboptimal Growing Conditions. Funct. Plant Biol. 2020, 47, 1007. [Google Scholar] [CrossRef]
- Ćelepirović, N.; Bogunović, S.; Dounavi, A.; Netzer, F.; Eiblmeier, M.; Dannenmann, M.; Rehschuh, S.; Rennenberg, H.; Ivanković, M. Phosphorus Nutrition and Water Relations of European Beech (Fagus sylvatica L.) Saplings Are Determined by Plant Origin. Forests 2022, 13, 1683. [Google Scholar] [CrossRef]
- Mátyás, C. Climatic Adaptation of Trees: Rediscovering Provenance Tests. Euphytica 1996, 92, 45–54. [Google Scholar] [CrossRef]
- Guo, X.; Khare, S.; Silvestro, R.; Huang, J.; Sylvain, J.-D.; Delagrange, S.; Rossi, S. Minimum Spring Temperatures at the Provenance Origin Drive Leaf Phenology in Sugar Maple Populations. Tree Physiol. 2020, 40, 1639–1647. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Parker, W.C.; Colombo, S.J.; Man, R.; Colombo, S. Restructuring tree provenance test data to conform to reciprocal transplant experiments for detecting local adaptation. J. Appl. Ecol. 2016, 53, 1088–1097. [Google Scholar] [CrossRef]
- Mukarram, M.; Choudhary, S.; Kurjak, D.; Petek, A.; Khan, M.M.A. Drought: Sensing, Signalling, Effects and Tolerance in Higher Plants. Physiol. Plant. 2021, 172, 1291–1300. [Google Scholar] [CrossRef]
- Robson, T.M.; Rasztovits, E.; Aphalo, P.J.; Alia, R.; Aranda, I. Flushing Phenology and Fitness of European Beech (Fagus sylvatica L.) Provenances from a Trial in La Rioja, Spain, Segregate According to Their Climate of Origin. Agric. For. Meteorol. 2013, 180, 76–85. [Google Scholar] [CrossRef]
- Gömöry, D.; Ditmarová, Ľ.; Hrivnák, M.; Jamnická, G.; Kmeť, J.; Krajmerová, D.; Kurjak, D. Differentiation in Phenological and Physiological Traits in European Beech (Fagus sylvatica L.). Eur. J. For. Res. 2015, 134, 1075–1085. [Google Scholar] [CrossRef]
- Hajek, P.; Kurjak, D.; von Wühlisch, G.; Delzon, S.; Schuldt, B. Intraspecific Variation in Wood Anatomical, Hydraulic, and Foliar Traits in Ten European Beech Provenances Differing in Growth Yield. Front. Plant Sci. 2016, 7, 791. [Google Scholar] [CrossRef]
- Knutzen, F.; Meier, I.C.; Leuschner, C. Does Reduced Precipitation Trigger Physiological and Morphological Drought Adaptations in European Beech (Fagus sylvatica L.)? Comparing Provenances across a Precipitation Gradient. Tree Physiol. 2015, 35, 949–963. [Google Scholar] [CrossRef]
- Wang, F.; Israel, D.; Ramírez-Valiente, J.-A.; Sánchez-Gómez, D.; Aranda, I.; Aphalo, P.J.; Robson, T.M. Seedlings from Marginal and Core Populations of European Beech (Fagus sylvatica L.) Respond Differently to Imposed Drought and Shade. Trees 2021, 35, 53–67. [Google Scholar] [CrossRef]
- Schuldt, B.; Knutzen, F.; Delzon, S.; Jansen, S.; Müller-Haubold, H.; Burlett, R.; Clough, Y.; Leuschner, C. How Adaptable Is the Hydraulic System of European Beech in the Face of Climate Change-related Precipitation Reduction? New Phytol. 2016, 210, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Sommer, R.J. Phenotypic Plasticity: From Theory and Genetics to Current and Future Challenges. Genetics 2020, 215, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Horváth, A.; Mátyás, C. The Decline of Vitality Caused by Increasing Drought in a Beech Provenance Trial Predicted by Juvenile Growth. SEEFOR 2016, 7, 21–28. [Google Scholar] [CrossRef][Green Version]
- Sáenz-Romero, C.; Kremer, A.; Nagy, L.; Újvári-Jármay, É.; Ducousso, A.; Kóczán-Horváth, A.; Hansen, J.K.; Mátyás, C. Common Garden Comparisons Confirm Inherited Differences in Sensitivity to Climate Change between Forest Tree Species. PeerJ 2019, 7, e6213. [Google Scholar] [CrossRef] [PubMed]
- Robson, T.M.; Garzón, M.B. Phenotypic Trait Variation Measured on European Genetic Trials of Fagus Sylvatica L. Sci. Data 2018, 5, 180149. [Google Scholar] [CrossRef] [PubMed]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Ziehn, T.; Chamberlain, M.; Lenton, A.; Law, R.; Bodman, R.; Dix, M.; Wang, Y.; Dobrohotoff, P.; Srbinovsky, J.; Stevens, L.; et al. CSIRO ACCESS-ESM1.5 Model Output Prepared for CMIP6 ScenarioMIP Ssp245. 2019. Available online: https://github.com/WCRP-CMIP/CMIP6_CVs (accessed on 19 December 2022).
- Zhang, Q.B.; Hebrda, J.H.; Zhang, Q.J.; Alfaro, R.I. Modeling Tree-Ring Growth Responses to Climatic Variables Using Artificial Neural Networks. For. Sci. 2000, 46, 229–239. [Google Scholar] [CrossRef]
- Khaki, S.; Wang, L.; Archontoulis, S.V. A CNN-RNN Framework for Crop Yield Prediction. Front. Plant Sci. 2020, 10, 1750. [Google Scholar] [CrossRef]
- Hornik, K. Approximation Capabilities of Multilayer Feedforward Networks. Neural Netw. 1991, 4, 251–257. [Google Scholar] [CrossRef]
- Montesinos López, O.A.; Montesinos López, A.; Crossa, J. Fundamentals of Artificial Neural Networks and Deep Learning. In Multivariate Statistical Machine Learning Methods for Genomic Prediction; Springer International Publishing: Cham, Switzerland, 2022; pp. 379–425. ISBN 978-3-030-89009-4. [Google Scholar]
- Pretzsch, H.; Hilmers, T.; Biber, P.; Avdagić, A.; Binder, F.; Bončina, A.; Bosela, M.; Dobor, L.; Forrester, D.I.; Lévesque, M.; et al. Evidence of Elevation-Specific Growth Changes of Spruce, Fir, and Beech in European Mixed Mountain Forests during the Last Three Centuries. Can. J. For. Res. 2020, 50, 689–703. [Google Scholar] [CrossRef]
- Hlásny, T.; Trombik, J.; Dobor, L.; Barcza, Z.; Barka, I. Future Climate of the Carpathians: Climate Change Hot-Spots and Implications for Ecosystems. Reg. Environ. Chang. 2016, 16, 1495–1506. [Google Scholar] [CrossRef]
- Čortan, D.; Nonić, M.; Šijačić-Nikolić, M. Phenotypic Plasticity of European Beech from International Provenance Trial in Serbia. In Forests of Southeast Europe Under a Changing Climate; Šijačić-Nikolić, M., Milovanović, J., Nonić, M., Eds.; Advances in Global Change Research; Springer International Publishing: Cham, Switzerland, 2019; Volume 65, pp. 333–351. ISBN 978-3-319-95266-6. [Google Scholar]
- Mátyás, C.; Berki, I.; Bidló, A.; Csóka, G.; Czimber, K.; Führer, E.; Gálos, B.; Gribovszki, Z.; Illés, G.; Hirka, A.; et al. Sustainability of Forest Cover under Climate Change on the Temperate-Continental Xeric Limits. Forests 2018, 9, 489. [Google Scholar] [CrossRef]
- Petkova, K.; Molle, E.; Konnert, M.; Knutzen, F. Comparing German and Bulgarian provenances of European beech (Fagus sylvatica L.) regarding survival, growth and ecodistance. Silva Balc. 2019, 2, 27–48. [Google Scholar] [CrossRef]
- Petrík, P.; Petek, A.; Konôpková, A.; Bosela, M.; Fleischer, P.; Frýdl, J.; Kurjak, D. Stomatal and Leaf Morphology Response of European Beech (Fagus sylvatica L.) Provenances Transferred to Contrasting Climatic Conditions. Forests 2020, 11, 1359. [Google Scholar] [CrossRef]
- Visi-Rajczi, E.; Hofmann, T.; Albert, L.; Mátyás, C. Tracing the Acclimation of European Beech (Fagus Sylvatica L.) Populations to Climatic Stress by Analyzing the Antioxidant System. iForest 2021, 14, 95–103. [Google Scholar] [CrossRef]
- Železnik, P.; Westergren, M.; Božič, G.; Eler, K.; Bajc, M.; Helmisaari, H.-S.; Horvath, A.; Kraigher, H. Root Growth Dynamics of Three Beech (Fagus sylvatica L.) Provenances. For. Ecol. Manag. 2019, 431, 35–43. [Google Scholar] [CrossRef]
- Bert, D.; Lebourgeois, F.; Ponton, S.; Musch, B.; Ducousso, A. Which Oak Provenances for the 22nd Century in Western Europe? Dendroclimatology in Common Gardens. PLoS ONE 2020, 15, e0234583. [Google Scholar] [CrossRef]
- Gömöry, D.; Longauer, R.; Hlásny, T.; Pacalaj, M.; Strmeň, S.; Krajmerová, D. Adaptation to Common Optimum in Different Populations of Norway Spruce (Picea abies Karst.). Eur. J. For. Res. 2012, 131, 401–411. [Google Scholar] [CrossRef]
- Jamnická, G.; Fleischer, P., Jr.; Konôpková, A.; Pšidová, E.; Kučerová, J.; Kurjak, D.; Živčák, M.; Ditmarová, L. Norway Spruce (Picea abies L.) Provenances Use Different Physiological Strategies to Cope with Water Deficit. Forests 2019, 10, 651. [Google Scholar] [CrossRef]
- Mátyás, C.; Bozic, G.; Gömöry, D.; Ivankovic, M.; Rasztovits, E. Juvenile Growth Response of European Beech (Fagus Sylvatica L.) to Sudden Change of Climatic Environment in SE European Trials. iForest 2009, 2, 213–220. [Google Scholar] [CrossRef]
- Martinez-Vilalta, J.; Anderegg, W.R.L.; Sapes, G.; Sala, A. Greater Focus on Water Pools May Improve Our Ability to Understand and Anticipate Drought-induced Mortality in Plants. New Phytol. 2019, 223, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Lévesque, M.; Walthert, L.; Weber, P. Soil Nutrients Influence Growth Response of Temperate Tree Species to Drought. J. Ecol. 2016, 104, 377–387. [Google Scholar] [CrossRef]
- Kreuzwieser, J.; Gessler, A. Global Climate Change and Tree Nutrition: Influence of Water Availability. Tree Physiol. 2010, 30, 1221–1234. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, E.; Miranda, B.; Sturtevant, B. Can Future CO2 Concentrations Mitigate the Negative Effects of High Temperature and Longer Droughts on Forest Growth? Forests 2018, 9, 664. [Google Scholar] [CrossRef]
- De Kauwe, M.G.; Medlyn, B.E.; Tissue, D.T. To What Extent Can Rising CO2 Ameliorate Plant Drought Stress? New Phytol. 2021, 231, 2118–2124. [Google Scholar] [CrossRef] [PubMed]
- Bussotti, F.; Pollastrini, M. Traditional and Novel Indicators of Climate Change Impacts on European Forest Trees. Forests 2017, 8, 137. [Google Scholar] [CrossRef]
- Müller, M.; Kempen, T.; Finkeldey, R.; Gailing, O. Low Population Differentiation but High Phenotypic Plasticity of European Beech in Germany. Forests 2020, 11, 1354. [Google Scholar] [CrossRef]
- Harter, D.E.V.; Nagy, L.; Backhaus, S.; Beierkuhnlein, C.; Fussi, B.; Huber, G.; Jentsch, A.; Konnert, M.; Thiel, D.; Kreyling, J. A Comparison of Genetic Diversity and Phenotypic Plasticity among European Beech (Fagus sylvatica L.) Populations from Bulgaria and Germany under Drought and Temperature Manipulation. Int. J. Plant Sci. 2015, 176, 232–244. [Google Scholar] [CrossRef]
- Frank, A.; Pluess, A.R.; Howe, G.T.; Sperisen, C.; Heiri, C. Quantitative Genetic Differentiation and Phenotypic Plasticity of European Beech in a Heterogeneous Landscape: Indications for Past Climate Adaptation. Perspect. Plant Ecol. Evol. Syst. 2017, 26, 1–13. [Google Scholar] [CrossRef]
- Kramer, K.; Ducousso, A.; Gömöry, D.; Hansen, J.K.; Ionita, L.; Liesebach, M.; Lorenţ, A.; Schüler, S.; Sulkowska, M.; de Vries, S.; et al. Chilling and Forcing Requirements for Foliage Bud Burst of European Beech (Fagus sylvatica L.) Differ between Provenances and Are Phenotypically Plastic. Agric. For. Meteorol. 2017, 2017, 172–181. [Google Scholar] [CrossRef]
- Wang, H.; Lin, S.; Dai, J.; Ge, Q. Modeling the Effect of Adaptation to Future Climate Change on Spring Phenological Trend of European Beech (Fagus sylvatica L.). Sci. Total Environ. 2022, 846, 157540. [Google Scholar] [CrossRef] [PubMed]
- Stojnić, S.; Orlović, S.; Miljković, D.; Galić, Z.; Kebert, M.; von Wuehlisch, G. Provenance Plasticity of European Beech Leaf Traits under Differing Environmental Conditions at Two Serbian Common Garden Sites. Eur. J. For. Res. 2015, 134, 1109–1125. [Google Scholar] [CrossRef]
- Vastag, E.; Kovacevic, B.; Orlovic, S.; Kesic, L.; Bojovic, M.; Stojnic, S. Leaf Stomatal Traits Variation within and among Fourteen European Beech (Fagus sylvatica L.) Provenances. Genetika 2019, 51, 937–959. [Google Scholar] [CrossRef]
- Stojnić, S.; Viscosi, V.; Marković, M.; Ivanković, M.; Orlović, S.; Tognetti, R.; Cocozza, C.; Vasić, V.; Loy, A. Spatial Patterns of Leaf Shape Variation in European Beech (Fagus sylvatica L.) Provenances. Trees 2022, 36, 497–511. [Google Scholar] [CrossRef]
- Stojnić, S.; Suchocka, M.; Benito-Garzón, M.; Torres-Ruiz, J.M.; Cochard, H.; Bolte, A.; Cocozza, C.; Cvjetković, B.; de Luis, M.; Martinez-Vilalta, J.; et al. Variation in Xylem Vulnerability to Embolism in European Beech from Geographically Marginal Populations. Tree Physiol. 2018, 38, 173–185. [Google Scholar] [CrossRef]
- Vicente, E.; Didion-Gency, M.; Morcillo, L.; Morin, X.; Vilagrosa, A.; Grossiord, C. Aridity and Cold Temperatures Drive Divergent Adjustments of European Beech Xylem Anatomy, Hydraulics and Leaf Physiological Traits. Tree Physiol. 2022, 42, 1720–1735. [Google Scholar] [CrossRef]
- Weithmann, G.; Link, R.M.; Banzragch, B.-E.; Würzberg, L.; Leuschner, C.; Schuldt, B. Soil Water Availability and Branch Age Explain Variability in Xylem Safety of European Beech in Central Europe. Oecologia 2022, 198, 629–644. [Google Scholar] [CrossRef]
- Miranda, J.C.; Calderaro, C.; Cocozza, C.; Lasserre, B.; Tognetti, R.; von Arx, G. Wood Anatomical Responses of European Beech to Elevation, Land Use Change, and Climate Variability in the Central Apennines, Italy. Front. Plant Sci. 2022, 13, 855741. [Google Scholar] [CrossRef]
- Pšidová, E.; Živčák, M.; Stojnić, S.; Orlović, S.; Gömöry, D.; Kučerová, J.; Ditmarová, Ľ.; Střelcová, K.; Brestič, M.; Kalaji, H.M. Altitude of Origin Influences the Responses of PSII Photochemistry to Heat Waves in European Beech (Fagus sylvatica L.). Environ. Exp. Bot. 2018, 152, 97–106. [Google Scholar] [CrossRef]
- Dziedek, C.; Fichtner, A.; Calvo, L.; Marcos, E.; Jansen, K.; Kunz, M.; Walmsley, D.; Von Oheimb, G.; Härdtle, W. Phenotypic Plasticity Explains Response Patterns of European Beech (Fagus sylvatica L.) Saplings to Nitrogen Fertilization and Drought Events. Forests 2017, 8, 91. [Google Scholar] [CrossRef]
- Weithmann, G.; Schuldt, B.; Link, R.M.; Heil, D.; Hoeber, S.; John, H.; Müller-Haubold, H.; Schüller, L.-M.; Schumann, K.; Leuschner, C. Leaf Trait Modification in European Beech Trees in Response to Climatic and Edaphic Drought. Plant Biol. 2021, 24, 1272–1286. [Google Scholar] [CrossRef] [PubMed]
- Dulamsuren, C.; Hauck, M.; Kopp, G.; Ruff, M.; Leuschner, C. European Beech Responds to Climate Change with Growth Decline at Lower, and Growth Increase at Higher Elevations in the Center of Its Distribution Range (SW Germany). Trees 2017, 31, 673–686. [Google Scholar] [CrossRef]
- Muffler, L.; Schmeddes, J.; Weigel, R.; Barbeta, A.; Beil, I.; Bolte, A.; Buhk, C.; Holm, S.; Klein, G.; Klisz, M.; et al. High Plasticity in Germination and Establishment Success in the Dominant Forest Tree Fagus sylvatica across Europe. Global Ecol. Biogeogr. 2021, 30, 1583–1596. [Google Scholar] [CrossRef]
- Piovesan, G.; Biondi, F.; Filippo, A.D.; Alessandrini, A.; Maugeri, M. Drought-Driven Growth Reduction in Old Beech (Fagus Sylvatica L.) Forests of the Central Apennines, Italy: Drought-driven growth decline in beech forests. Glob. Change Biol. 2008, 14, 1265–1281. [Google Scholar] [CrossRef]
- Knutzen, F.; Dulamsuren, C.; Meier, I.C.; Leuschner, C. Recent Climate Warming-Related Growth Decline Impairs European Beech in the Center of Its Distribution Range. Ecosystems 2017, 20, 1494–1511. [Google Scholar] [CrossRef]
- Aertsen, W.; Janssen, E.; Kint, V.; Bontemps, J.-D.; Van Orshoven, J.; Muys, B. Long-Term Growth Changes of Common Beech (Fagus Sylvatica L.) Are Less Pronounced on Highly Productive Sites. For. Ecol. Manag. 2014, 312, 252–259. [Google Scholar] [CrossRef]
- Zimmermann, J.; Hauck, M.; Dulamsuren, C.; Leuschner, C. Climate Warming-Related Growth Decline Affects Fagus Sylvatica, But Not Other Broad-Leaved Tree Species in Central European Mixed Forests. Ecosystems 2015, 18, 560–572. [Google Scholar] [CrossRef]
- Vacek, Z.; Prokůpková, A.; Vacek, S.; Bulušek, D.; Šimůnek, V.; Hájek, V.; Králíček, I. Mixed vs. Monospecific Mountain Forests in Response to Climate Change: Structural and Growth Perspectives of Norway Spruce and European Beech. For. Ecol. Manag. 2021, 488, 119019. [Google Scholar] [CrossRef]
- Sedmáková, D.; Sedmák, R.; Bosela, M.; Ježík, M.; Blaženec, M.; Hlásny, T.; Marušák, R. Growth-Climate Responses Indicate Shifts in the Competitive Ability of European Beech and Norway Spruce under Recent Climate Warming in East-Central Europe. Dendrochronologia 2019, 54, 37–48. [Google Scholar] [CrossRef]
- Prislan, P.; Gričar, J.; Čufar, K.; de Luis, M.; Merela, M.; Rossi, S. Growing Season and Radial Growth Predicted for Fagus Sylvatica under Climate Change. Clim. Change 2019, 153, 181–197. [Google Scholar] [CrossRef]
- Richardson, A.D.; Andy Black, T.; Ciais, P.; Delbart, N.; Friedl, M.A.; Gobron, N.; Hollinger, D.Y.; Kutsch, W.L.; Longdoz, B.; Luyssaert, S.; et al. Influence of Spring and Autumn Phenological Transitions on Forest Ecosystem Productivity. Phil. Trans. R. Soc. B 2010, 365, 3227–3246. [Google Scholar] [CrossRef] [PubMed]
- Schieber, B.; Kubov, M.; Janík, R. Effects of Climate Warming on Vegetative Phenology of the Common Beech Fagus Sylvatica in a Submontane Forest of the Western Carpathians: Two-Decade Analysis. Pol. J. Ecol. 2017, 65, 339–351. [Google Scholar] [CrossRef]
- Mura, C.; Buttò, V.; Silvestro, R.; Deslauriers, A.; Charrier, G.; Raymond, P.; Rossi, S. The Early Bud Gets the Cold: Diverging Spring Phenology Drives Exposure to Late Frost in a Picea mariana [(Mill.) BSP] Common Garden. Physiol. Plant. 2022, 174, e13798. [Google Scholar] [CrossRef] [PubMed]
- Vitali, A.; Tonelli, E.; Malandra, F.; Camarero, J.J.; Colangelo, M.; Nolè, A.; Ripullone, F.; Carrer, M.; Urbinati, C. Combining Dendroecology and Remote Sensing to Assess How Late Spring Frosts Affect European Beech Forests. EGU Gen. Assem. 2022, EGU22-5514. [Google Scholar] [CrossRef]

| Factor | Df | SumSq | MeanSq | F | p |
|---|---|---|---|---|---|
| Provenance | 130 | 71,289,815 | 548,383 | 109.66 | <0.001 |
| Site | 13 | 188,718,150 | 14,516,781 | 2902.8 | <0.001 |
| Provenance × Site | 409 | 22,573,979 | 55,193 | 11.04 | <0.001 |
| Residuals | 45,735 | 228,715,682 | 5001 |
| Bio8 | Bio14 | Bio18 | Bio8ED | Bio12ED | Bio13ED | Bio18ED | |
|---|---|---|---|---|---|---|---|
| Min | 0.1 | 12 | 78 | −16.4 | −928 | −122 | −342 |
| Max | 19.9 | 88 | 533 | 17 | 1261 | 138 | 319 |
| Unit | °C | mm | mm | °C | mm | mm | Mm |
| PV | T (°C) | P (mm) | PV | T (°C) | P (mm) |
|---|---|---|---|---|---|
| Hot–Dry | Hot–Wet | ||||
| FR02 | 10.6 | 682 | ESP02 | 9.8 | 967 |
| ESP05 | 10.5 | 633 | FR23 | 9.8 | 975 |
| GER47 | 8.8 | 556 | IT37 | 12.3 | 1251 |
| IT78 | 13.8 | 595 | IT108 | 10.9 | 1080 |
| IT80 | 8.2 | 762 | CR139 | 14.4 | 1271 |
| Cold–Dry | Cold–Wet | ||||
| PL38 | 5.9 | 624 | AU35 | 2.4 | 1524 |
| CZ48 | 4.4 | 769 | AU36 | 4 | 1184 |
| GER85 | 5.1 | 709 | CZ51 | 4.6 | 1124 |
| PL117 | 4.9 | 757 | AU39 | 3.8 | 1067 |
| RO155 | 6.5 | 583 | UA141 | 4.9 | 943 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrík, P.; Grote, R.; Gömöry, D.; Kurjak, D.; Petek-Petrik, A.; Lamarque, L.J.; Sliacka Konôpková, A.; Mukarram, M.; Debta, H.; Fleischer, P., Jr. The Role of Provenance for the Projected Growth of Juvenile European Beech under Climate Change. Forests 2023, 14, 26. https://doi.org/10.3390/f14010026
Petrík P, Grote R, Gömöry D, Kurjak D, Petek-Petrik A, Lamarque LJ, Sliacka Konôpková A, Mukarram M, Debta H, Fleischer P Jr. The Role of Provenance for the Projected Growth of Juvenile European Beech under Climate Change. Forests. 2023; 14(1):26. https://doi.org/10.3390/f14010026
Chicago/Turabian StylePetrík, Peter, Rüdiger Grote, Dušan Gömöry, Daniel Kurjak, Anja Petek-Petrik, Laurent J. Lamarque, Alena Sliacka Konôpková, Mohammad Mukarram, Harish Debta, and Peter Fleischer, Jr. 2023. "The Role of Provenance for the Projected Growth of Juvenile European Beech under Climate Change" Forests 14, no. 1: 26. https://doi.org/10.3390/f14010026
APA StylePetrík, P., Grote, R., Gömöry, D., Kurjak, D., Petek-Petrik, A., Lamarque, L. J., Sliacka Konôpková, A., Mukarram, M., Debta, H., & Fleischer, P., Jr. (2023). The Role of Provenance for the Projected Growth of Juvenile European Beech under Climate Change. Forests, 14(1), 26. https://doi.org/10.3390/f14010026









