Testing New Provenances of Eucalyptus polybractea: A Eucalypt Oil Mallee Adapted to Semi-Arid Environments
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection of Seedlots for Trial
2.2. Trial Establishment and Design
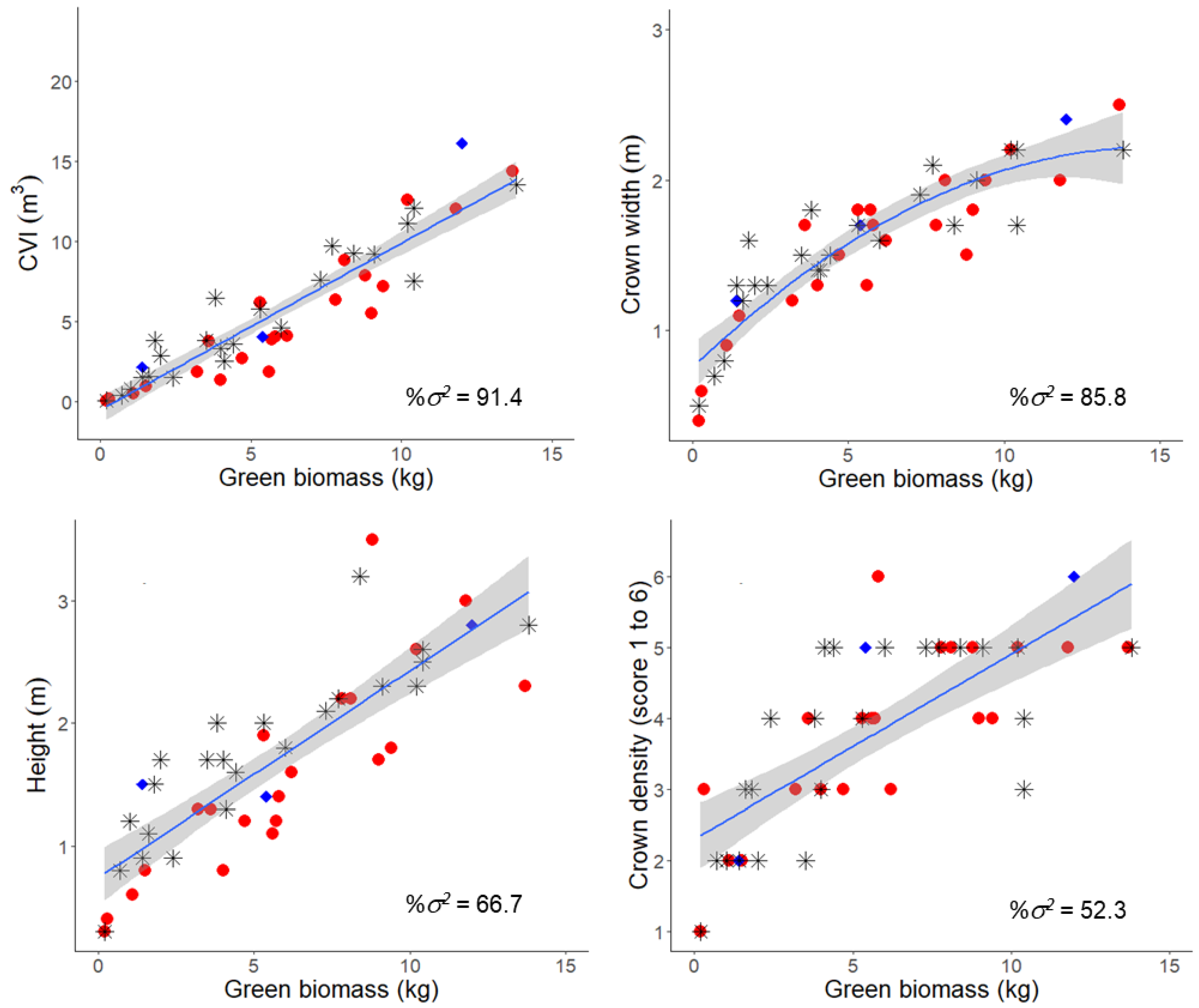
2.3. Growth Traits
2.4. Foliar Oil Characteristics
2.5. Data Analysis
3. Results
3.1. Survival and Growth
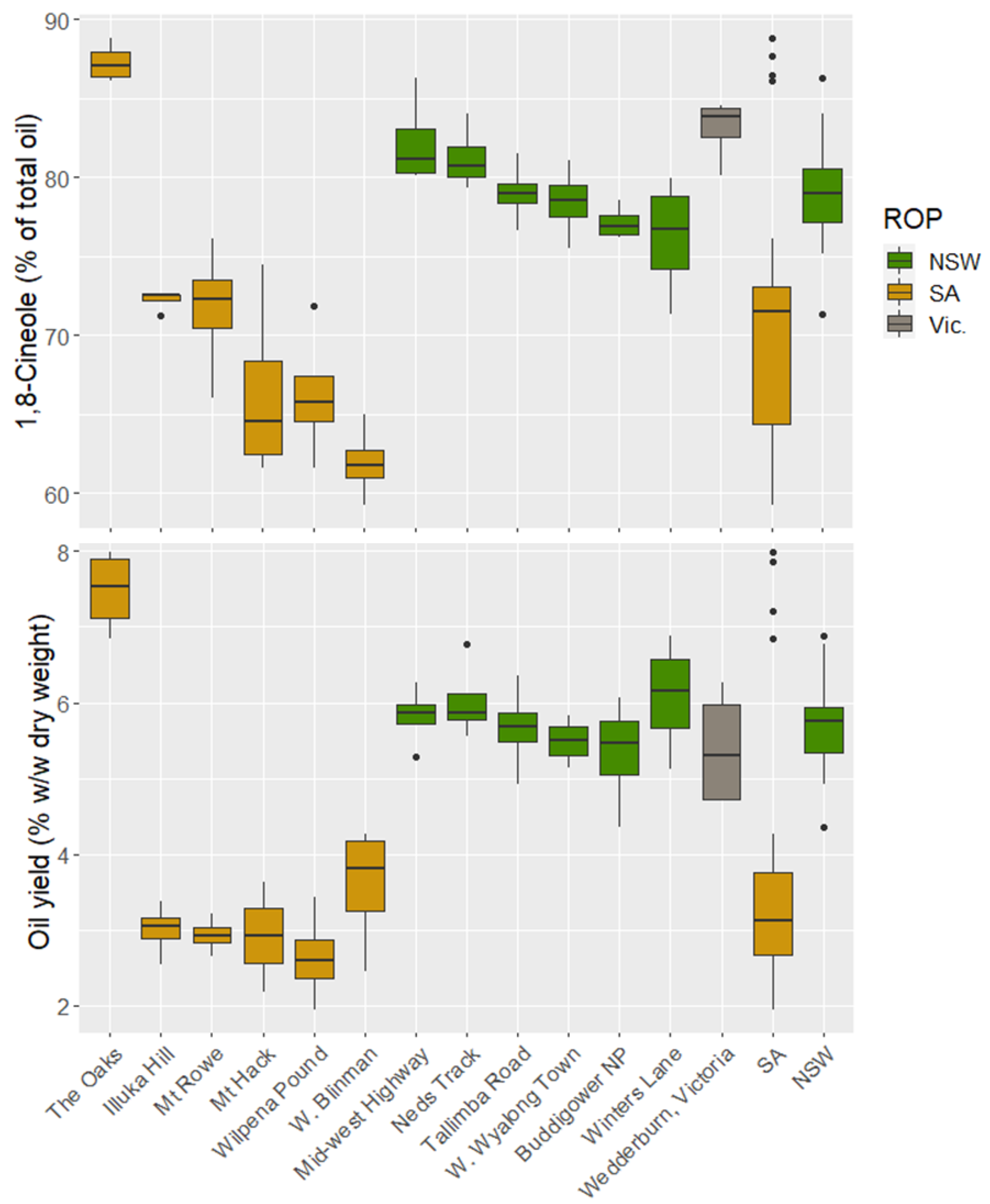
3.2. Oil Characteristics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nicolle, D. Native Eucalypts of South Australia; Dean Nicolle: Adelaide, Australia, 2013; p. 237. [Google Scholar]
- Slee, A.V.; Brooker, M.I.H.; Duffy, S.M.; West, J.G. EUCLID Eucalypts of Australia, 4th ed.; Identic Pty Ltd.: Brisbane, Australia, 2020. [Google Scholar]
- Beerling, J.; Meakins, S.; Small, L. Eucalyptus oil products: Formulations and legislation. In Eucalyptus: The Genus Eucalyptus. Medicinal and Aromatic Plants—Industrial Profiles; Coppen, J.J.W., Ed.; Taylor and Francis: London, UK; New York, NY, USA, 2002; pp. 345–364. [Google Scholar]
- Abbott, P.; Abbott, T. Eucalyptus Oil: Australia’s Natural Wonder; Felton, Grimwade and Bosisto’s Pty Ltd.: Melbourne, Australia, 2013; p. 134. [Google Scholar]
- BP. British Pharmacopoeia. Ph. Eur. 10.0 Update; The Stationery Office: London, UK, 2020.
- Coppen, J.J.W. Eucalyptus: The Genus Eucalyptus; Taylor and Francis: London, UK; New York, NY, USA, 2002. [Google Scholar]
- Doran, J.C. Genetic improvement of eucalypts: With special reference to oil-bearing species. In Eucalyptus: The Genus Eucalyptus. Medicinal and Aromatic Plants—Industrial Profiles; Coppen, J.J.W., Ed.; Taylor and Francis: London, UK; New York, NY, USA, 2002; pp. 75–101. [Google Scholar]
- Barton, A.F.M.; Cotterill, P.P.; Brooker, M.I.H. Short note-heritability of cineole yield in Eucalyptus kochii. Silvae Genet. 1991, 40, 37–38. [Google Scholar]
- Spencer, B.; Mazanec, R.; Gibberd, M.; Zerihun, A. Sapling and coppice biomass heritabilities and potential gains from Eucalyptus polybractea progeny trials. Tree Genet. Genomes 2021, 17, 15. [Google Scholar] [CrossRef]
- Mazanec, R.A.; Grayling, P.M.; Doran, J.; Spencer, B.; Turnbull, P. Genetic parameters and potential gains from breeding for biomass and cineole production in three-year-old Eucalyptus polybractea progeny trials. Aust. For. 2021, 84, 13–24. [Google Scholar] [CrossRef]
- Doran, J.; Bush, D.; Davis, R. Breeding to improve foliar oil yields in Eucalyptus polybractea. In Proceedings of the Forest Genetics for Productivity, Rotorua, New Zealand, 14–28 March 2016; p. 41. [Google Scholar]
- Goodger, J.Q.D.; Connelly, C.A.; Woodrow, I.E. Examination of the consistency of plant traits driving oil yield and quality in short-rotation coppice cultivation of Eucalyptus polybractea. For. Ecol. Manag. 2007, 250, 196–205. [Google Scholar] [CrossRef]
- Paul, K.I.; Roxburgh, S.H.; de Ligt, R.; Ritson, P.; Brooksbank, K.; Peck, A.; Wildy, D.T.; Mendham, D.; Bennett, R.; Bartle, J.; et al. Estimating temporal changes in carbon sequestration in plantings of mallee eucalypts: Modelling improvements. For. Ecol. Manag. 2015, 335, 166–175. [Google Scholar] [CrossRef]
- Satori, D.; Tovar, C.; Faruk, A.; Hammond Hunt, E.; Muller, G.; Cockel, C.; Kühn, N.; Leitch, I.; Lulekal, E.; Pereira, L.; et al. Prioritising crop wild relatives to enhance agricultural resilience in sub-Saharan Africa under climate change. Plants People Planet 2021, 4, 269–282. [Google Scholar] [CrossRef]
- Ortiz, R.; Sayre, K.D.; Govaerts, B.; Gupta, R.; Subbarao, G.V.; Ban, T.; Hodson, D.; Dixon, J.M.; Iván Ortiz-Monasterio, J.; Reynolds, M. Climate change: Can wheat beat the heat? Agric. Ecosyst. Environ. 2008, 126, 46–58. [Google Scholar] [CrossRef]
- Cortés, A.J.; López-Hernández, F. Harnessing Crop Wild Diversity for Climate Change Adaptation. Genes 2021, 12, 783. [Google Scholar] [CrossRef] [PubMed]
- Prober, S.; Byrne, M.; McLean, E.; Steane, D.; Potts, B.; Vaillancourt, R.; Stock, W. Climate-adjusted provenancing: A strategy for climate-resilient ecological restoration. Front. Ecol. Evol. 2015, 3, 65. [Google Scholar] [CrossRef] [Green Version]
- Fady, B.; Rihm, G. Arboretums, common gardens and forest tree resilience. New For. 2022, 53, 603–606. [Google Scholar] [CrossRef]
- CSIRO and Bureau of Meteorology. Climate Change in Australia Information for Australia’s Natural Resource Management Regions: Technical Report; CSIRO and Bureaa of Meteorology: Melbourne, Australia, 2015; p. 216.
- Bush, D.; Harwood, C.; Pinkard, E. Species for changing climates–Australian dryland forestry opportunities. Aust. For. 2018, 81, 102–115. [Google Scholar] [CrossRef]
- Weiss, E.A. Essential Oil Crops; CAB International: New York, NY, USA, 1997; 600p. [Google Scholar]
- Kainer, D.; Bush, D.; Foley, W.J.; Külheim, C. Assessment of a non-destructive method to predict oil yield in Eucalyptus polybractea (blue mallee). Ind. Crops Prod. 2017, 102, 32–44. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Mazanec, R.A.; Grayling, P.M.; Doran, J.; Spencer, B.; Neumann, C. Provenance variation, genetic parameters and potential gains from selection for biomass and cineole production in three-year-old Eucalyptus loxophleba subsp. gratiae progeny trials. Aust. For. 2020, 83, 75–90. [Google Scholar] [CrossRef]
- Mazanec, R.A.; Grayling, P.M.; Spencer, B.; Doran, J.; Neumann, C. Provenance variation, genetic parameters and potential gains from breeding for biomass and cineole production in three-year-old Eucalyptus loxophleba subsp. lissophloia progeny trials. Aust. For. 2017, 80, 34–42. [Google Scholar] [CrossRef]
- Pont, D.; Dungey, H.S.; Suontama, M.; Stovold, G.T. Spatial Models With Inter-Tree Competition From Airborne Laser Scanning Improve Estimates of Genetic Variance. Front. Plant Sci. 2021, 11, 596315. [Google Scholar] [CrossRef] [PubMed]
- Kotivuori, E.; Kukkonen, M.; Mehtätalo, L.; Maltamo, M.; Korhonen, L.; Packalen, P. Forest inventories for small areas using drone imagery without in-situ field measurements. Remote Sens. Environ. 2020, 237, 111404. [Google Scholar] [CrossRef]
- Ximenes, F. Forestry, bioenergy and climate—A way forward in Australia. Aust. For. 2021, 84, 1–3. [Google Scholar] [CrossRef]
- Shepherd, M.; Bartle, J.; Lee, D.J.; Brawner, J.; Bush, D.; Turnbull, P.; Macdonell, P.; Brown, T.R.; Simmons, B.; Henry, R. Eucalypts as a biofuel feedstock. Biofuels 2011, 2, 639–657. [Google Scholar] [CrossRef] [Green Version]

| Region-Of-Provenance and Provenance | ATSC Seedlot Number | No. of Families in Provenance | Latitude (Decimal Degrees) | Longitude (Decimal Degrees) | Altitude (m above Sea Level) | MAR (mm/ Year) | MAT (°C) |
|---|---|---|---|---|---|---|---|
| South Australia-Modified BoM Koeppen climate is Grassland, warm (persistently dry) | |||||||
| W of Blinman, Flinders Ranges | 21,336 | 2 | −31.1033 | 138.6674 | 630 | 330 | 16 |
| ENE Mt Rowe, Vulkathuna-Gammon Ranges | 21,337 | 13 | −30.5200 | 138.9828 | 750 | 350 | 15 |
| Mt Hack, Warraweena Station | 21,338 | 15 | −30.7917 | 138.7947 | 700 | 320 | 15 |
| Illuka Hill, Flinders Ranges | 21,340 | 5 | −31.6006 | 138.6317 | 610 | 360 | 14 |
| The Oaks, SW of Hawker | 21,342 | 9 | −31.9464 | 138.3231 | 500 | 310 | 16 |
| Wilpena Pound, Flinders Ranges | 21,343 | 10 | −31.5431 | 138.5667 | 586 | 395 | 16 |
| Victoria-Modified BoM Koeppen climate is Temperate, no dry season (hot summer) | |||||||
| Wedderburn | 19,361 | 7 | −36.3333 | 143.6000 | 180 | 510 | 14 |
| New South Wales-Modified BoM Koeppen climate is Temperate, no dry season (hot summer) | |||||||
| Mid-Western Hwy | 19,920, 21,060 | 6 | −33.8928 | 147.0722 | 310 | 470 | 16 |
| West Wyalong township | 19,649, 21,059, 21,060 | 8 | −33.9237 | 147.2047 | 260 | 460 | 16 |
| Tallimba Rd | 19,649, 19,920, 21,059, 21,060, 21,365 | 9 | −33.9589 | 147.0453 | 290 | 470 | 16 |
| Neds Track | 19,920, 20,680, 21,060 | 10 | −34.0011 | 147.1594 | 280 | 460 | 16 |
| Winters Lane | 19,649, 21,060, 21,365 | 7 | −34.0244 | 147.1508 | 265 | 460 | 16 |
| Buddigower NP | 19,649, 21,060 | 8 | −34.0061 | 147.0822 | 270 | 450 | 16 |
| TOTAL | 109 | ||||||
| Provenance | CVI (m3) | Oil Yield w/w (g/100 g Dry Leaf) | %Cineole | |||
|---|---|---|---|---|---|---|
| Mean | %CVp | Mean | %CVp | Mean | %CVp | |
| South Australia | ||||||
| Illuka Hill | 4.8 | 71.8 | 3.0 | 11.5 | 72.2 | 0.9 |
| Mt Hack | 4.5 | 86.6 | 2.9 | 21.4 | 66.3 | 8.8 |
| Mt Rowe | 4.4 | 101.1 | 2.9 | 7.9 | 71.7 | 5.9 |
| The Oaks | 4.7 | 73.3 | 7.5 | 7.3 | 87.3 | 1.4 |
| W Blinman | 3.4 | 105.1 | 3.6 | 23.0 | 61.9 | 3.8 |
| Wilpena Pound | 5.1 | 80.9 | 2.6 | 23.1 | 66.2 | 6.4 |
| Victoria | ||||||
| Wedderburn | 6.7 | 72.3 | 5.4 | 14.8 | 83.1 | 2.5 |
| New South Wales | ||||||
| Buddigower NP | 3.8 | 88.6 | 5.3 | 13.6 | 77.1 | 1.4 |
| Mid-Western Highway | 5.9 | 64.7 | 5.8 | 6.9 | 82.2 | 3.5 |
| Neds Lane | 5.2 | 64.3 | 6.0 | 8.8 | 81.2 | 2.5 |
| Winters Lane | 6.0 | 71.0 | 6.1 | 12.6 | 76.2 | 5.0 |
| Talimba Rd | 5.3 | 67.9 | 5.7 | 10.4 | 79.0 | 2.5 |
| West Wyalong Town | 5.6 | 61.6 | 5.5 | 5.5 | 78.4 | 3.0 |
| Average SED * | 0.47 | 0.42 | 1.9 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bush, D.; Spencer, D.; Doran, J.; Davis, R. Testing New Provenances of Eucalyptus polybractea: A Eucalypt Oil Mallee Adapted to Semi-Arid Environments. Forests 2022, 13, 1109. https://doi.org/10.3390/f13071109
Bush D, Spencer D, Doran J, Davis R. Testing New Provenances of Eucalyptus polybractea: A Eucalypt Oil Mallee Adapted to Semi-Arid Environments. Forests. 2022; 13(7):1109. https://doi.org/10.3390/f13071109
Chicago/Turabian StyleBush, David, David Spencer, John Doran, and Richard Davis. 2022. "Testing New Provenances of Eucalyptus polybractea: A Eucalypt Oil Mallee Adapted to Semi-Arid Environments" Forests 13, no. 7: 1109. https://doi.org/10.3390/f13071109
APA StyleBush, D., Spencer, D., Doran, J., & Davis, R. (2022). Testing New Provenances of Eucalyptus polybractea: A Eucalypt Oil Mallee Adapted to Semi-Arid Environments. Forests, 13(7), 1109. https://doi.org/10.3390/f13071109







