Guiding Conservation for Mountain Tree Species in Lebanon
Abstract
:1. Introduction
2. Species and Methods
2.1. Studied Species
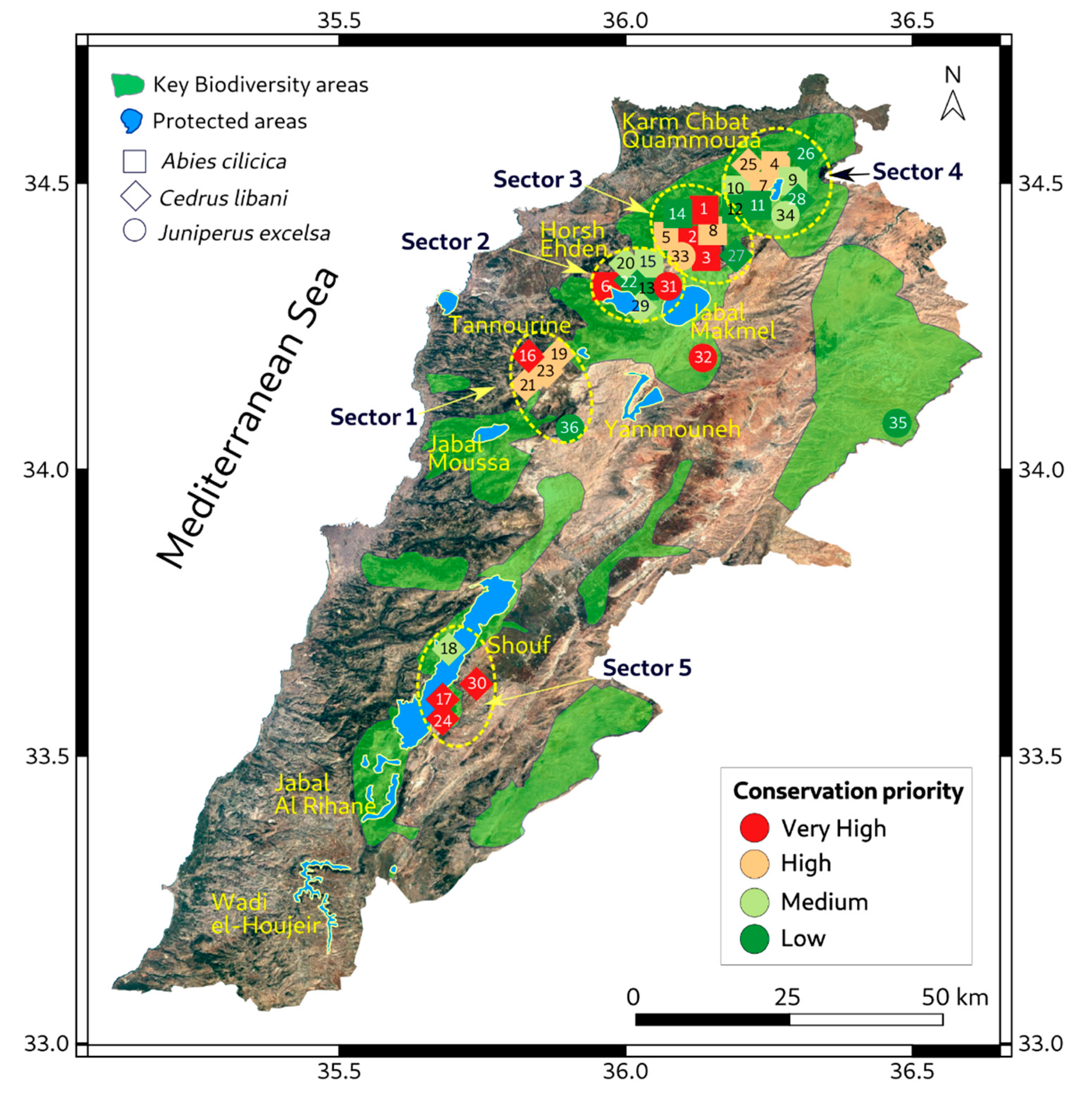
2.2. Conservation Index
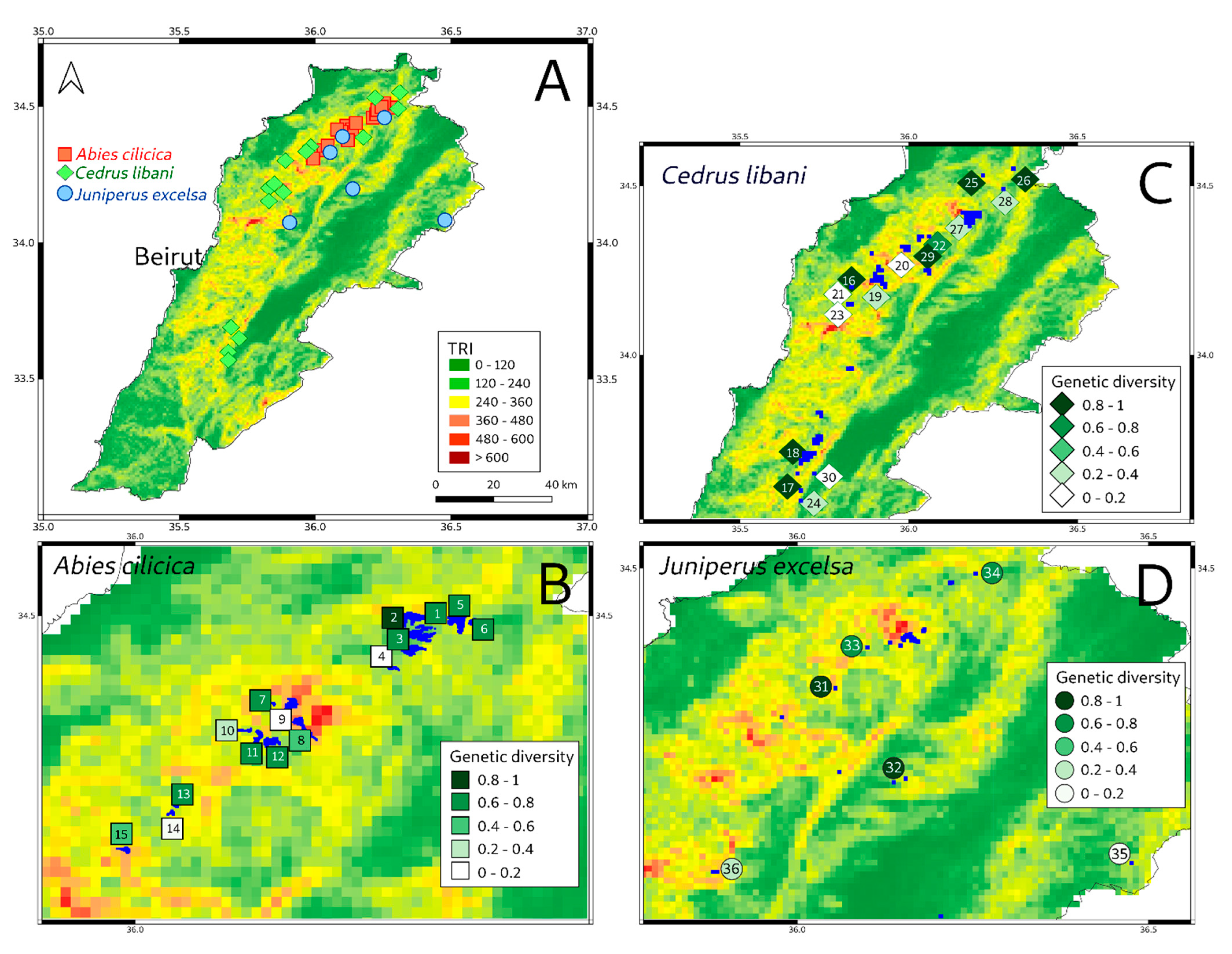

| Pop | Spot | Species | Site Name | Longitude | Latitude | Gd | Tr | Di | Ci |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 3 | A | Jaïroun | 36.12559 | 34.42586 | 0.71 | 431 | 103 | 2.97 |
| 2 | 3 | A | Kfarbnine | 36.10355 | 34.40200 | 0.75 | 347 | 95 | 2.75 |
| 3 | 3 | A | Kfarbnine | 36.10797 | 34.39806 | 0.74 | 320 | 96 | 2.47 |
| 6 | 2 | A | Ehden | 35.99223 | 34.30729 | 0.34 | 364 | 55 | 2.24 |
| 5 | 3 | A | Kfarbnine | 36.09566 | 34.40533 | 0.56 | 347 | 94 | 2.05 |
| 4 | 4 | A | Kobayat | 36.26463 | 34.49820 | 1.00 | 288 | 151 | 1.91 |
| 7 | 4 | A | Qammouaa | 36.24337 | 34.49640 | 0.80 | 303 | 147 | 1.66 |
| 8 | 3 | A | Wadi Jhanam | 36.13486 | 34.41423 | 0.68 | 232 | 106 | 1.49 |
| 9 | 4 | A | Hermel | 36.27785 | 34.49399 | 0.72 | 288 | 153 | 1.35 |
| 10 | 4 | A | Qammouaa | 36.22776 | 34.48551 | 0.80 | 176 | 142 | 1.00 |
| 15 | 2 | A | Bqaa_Safrine | 36.03345 | 34.34325 | 0.10 | 584 | 69 | 0.85 |
| 13 | 2 | A | Karm El Mohr | 36.03026 | 34.33841 | 0.22 | 232 | 68 | 0.76 |
| 11 | 4 | A | Qammouaa | 36.22445 | 34.47012 | 0.67 | 148 | 141 | 0.71 |
| 12 | 4 | A | Qammouaa | 36.21097 | 34.45737 | 0.52 | 154 | 137 | 0.58 |
| 14 | 3 | A | Qarsita | 36.11414 | 34.42744 | 0.11 | 382 | 102 | 0.41 |
| 16 | 1 | C | Bcharre/Hadeth el Jebbe | 35.83333 | 34.20000 | 1.00 | 388 | 13 | 29.85 |
| 17 | 5 | C | Barouk | 35.68333 | 33.60000 | 0.99 | 360 | 23 | 15.47 |
| 24 | 5 | C | Chouf | 35.68333 | 33.56667 | 0.67 | 292 | 23 | 8.52 |
| 30 | 5 | C | Ain Zhalta | 35.71667 | 33.65000 | 0.57 | 318 | 26 | 6.91 |
| 21 | 1 | C | Jaj | 35.82884 | 34.14986 | 0.11 | 245 | 10 | 2.62 |
| 23 | 1 | C | Bcharre | 35.83333 | 34.20000 | 0.11 | 307 | 13 | 2.53 |
| 25 | 4 | C | Akkar/Quammoua | 36.21667 | 34.53333 | 0.98 | 303 | 138 | 2.14 |
| 19 | 1 | C | Tannourine | 35.88660 | 34.20318 | 0.11 | 353 | 21 | 1.90 |
| 18 | 5 | C | Arz El Chouf | 35.69391 | 33.68814 | 0.14 | 302 | 23 | 1.88 |
| 20 | 2 | C | Jord Njas | 36.03081 | 34.34111 | 0.14 | 350 | 68 | 0.73 |
| 29 | 2 | C | Ehden | 35.99223 | 34.30729 | 0.10 | 387 | 55 | 0.70 |
| 22 | 2 | C | Karm El Mohr | 36.03026 | 34.33841 | 0.13 | 350 | 68 | 0.69 |
| 27 | 3 | C | Jabal Illy | 36.16951 | 34.39775 | 0.11 | 584 | 120 | 0.56 |
| 26 | 4 | C | Kharm Chbat | 36.31379 | 34.55227 | 0.15 | 265 | 162 | 0.24 |
| 28 | 4 | C | Hermel | 36.27785 | 34.49399 | 0.11 | 288 | 153 | 0.22 |
| 31 | 2 | J | Wadi El Njass | 36.05444 | 34.33028 | 0.68 | 350 | 79 | 3.00 |
| 32 | 3 | J | Donniyeh | 36.10000 | 34.38806 | 0.34 | 584 | 93 | 2.15 |
| 33 | 6 | J | Barqa | 36.13750 | 34.19667 | 0.83 | 282 | 114 | 2.06 |
| 34 | 4 | J | Qammouaa | 36.25389 | 34.49278 | 0.80 | 302 | 149 | 1.63 |
| 35 | 6 | J | Aarsal | 36.47611 | 34.08250 | 1.00 | 202 | 212 | 0.95 |
| 36 | 1 | J | Afqa | 35.90550 | 34.07361 | 0.10 | 361 | 45 | 0.80 |
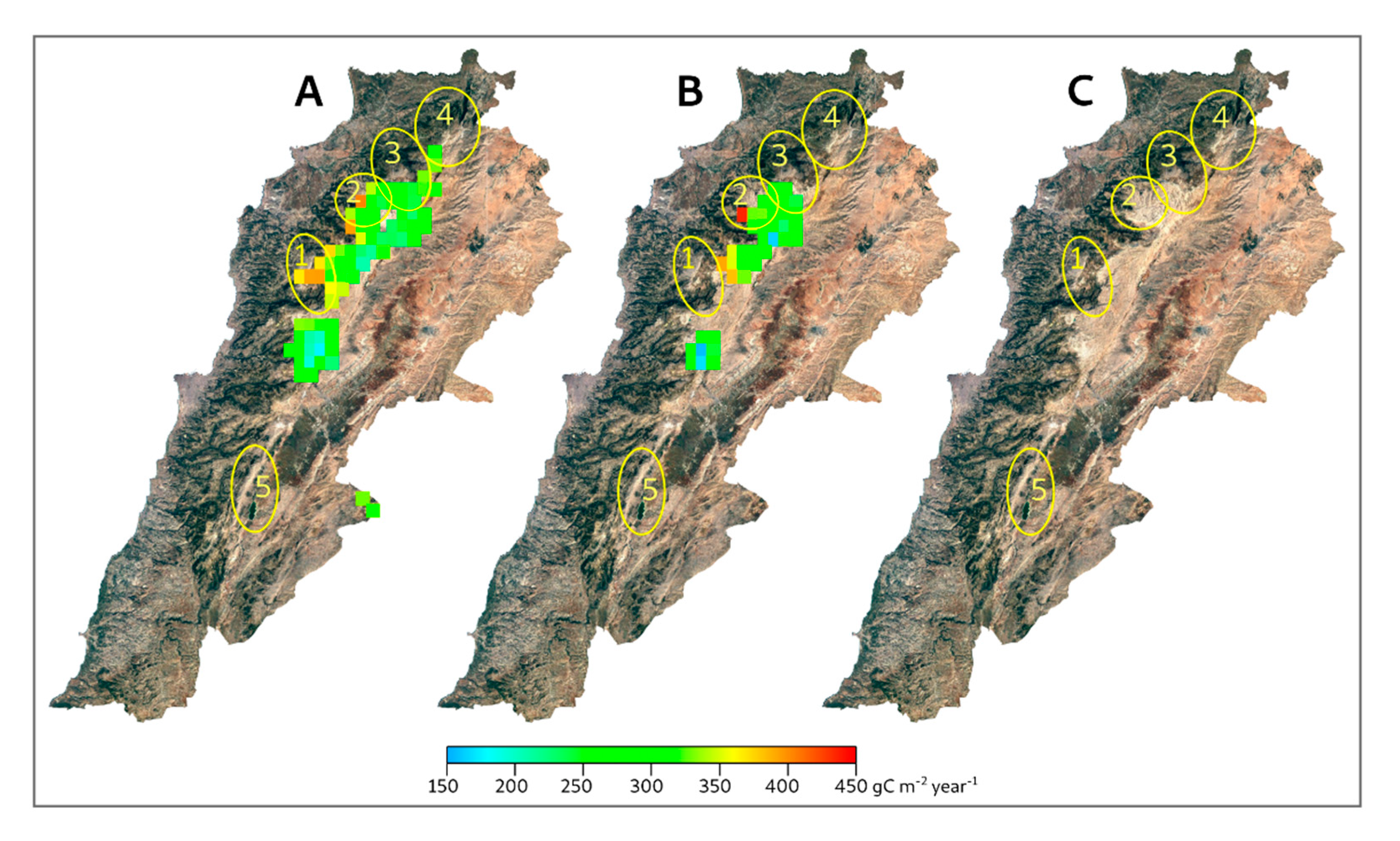
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mikesell, M.W. The Deforestation of Mount Lebanon. Geogr. Rev. 1969, 59, 1–28. [Google Scholar] [CrossRef]
- Khuri, S.; Shmoury, M.; Baalbaki, R.; Maunder, M.; Talhouk, S. Conservation of the Cedrus libani populations in Lebanon: History, current status and experimental application of somatic embryogenesis. Biodivers. Conserv. 2000, 9, 1261–1273. [Google Scholar] [CrossRef]
- Jomaa, I.; Auda, Y.; Khater, C. Contribution of the Charaterization of Forest Fragmentation on the Eastern Flank of Mount Lebanon Over 33 years. Leban. Sci. J. 2007, 8, 59–74. [Google Scholar]
- Jomaa, I.; Auda, Y.; Saleh, B.A.; Hamzé, M.; Safi, S. Landscape spatial dynamics over 38 years under natural and anthropogenic pressures in Mount Lebanon. Landsc. Urban Plan. 2008, 87, 67–75. [Google Scholar] [CrossRef]
- Haroutunian, G.; Chojnacky, D.C.; El Riachy, R.; Chojnacky, C.C. Reducing Reforestation Costs in Lebanon: Adaptive Field Trials. Forests 2017, 8, 169. [Google Scholar] [CrossRef] [Green Version]
- Jomaa, I.; Khater, C. Mapping Glitches of Juniper Forests in Lebanon under Natural Conditions and Anthropogenic Activities. Open J. For. 2019, 9, 168181. [Google Scholar] [CrossRef] [Green Version]
- Gardner, M.; Knees, S. Abies Cilicica. The IUCN Red List of Threatened Species 2013: E.T42275A2968944. 2013. Available online: https://doi.org/10.2305/IUCN.UK.2013-1.RLTS.T42275A2968944.en (accessed on 18 January 2022).
- Evans, J.P. 21st century climate change in the Middle East. Clim. Chang. 2008, 92, 417–432. [Google Scholar] [CrossRef]
- Gao, X.; Pal, J.S.; Giorgi, F. Projected changes in mean and extreme precipitation over the Mediterranean region from a high resolution double nested RCM simulation. Geophys. Res. Lett. 2006, 33, 14. [Google Scholar] [CrossRef]
- Hajar, L.; François, L.; Khater, C.; Jomaa, I.; Déqué, M.; Cheddadi, R. Cedrus libani (A. Rich) distribution in Lebanon: Past, present and future. C. R. Biol. 2010, 333, 622–630. [Google Scholar] [CrossRef]
- MOA. National Action Programme to Combat Desertification; Ministry of Agriculture: Beyrout, Lebanon, 2003; 198p.
- MOE/UNDP. Climate Change Vulnerability and Adaptation: Climate Risks, Vulnerability & Adaptation Assessment; Ministry of Environment: Beyrouth, Lebanon, 2011; 44p.
- Dobrowski, S.Z. A climatic basis for microrefugia: The influence of terrain on climate. Glob. Chang. Biol. 2011, 17, 1022–1035. [Google Scholar] [CrossRef]
- Valencia, B.G.; Matthews-Bird, F.; Urrego, D.H.; Williams, J.; Gosling, W.D.; Bush, M. Andean microrefugia: Testing the Holocene to predict the Anthropocene. New Phytol. 2016, 212, 510–522. [Google Scholar] [CrossRef]
- Keppel, G.; Van Niel, K.P.; Wardell-Johnson, G.W.; Yates, C.J.; Byrne, M.; Mucina, L.; Schut, A.G.T.; Hopper, S.D.; Franklin, S.E. Refugia: Identifying and understanding safe havens for biodiversity under climate change. Glob. Ecol. Biogeogr. 2012, 21, 393–404. [Google Scholar] [CrossRef]
- Bennett, K.D.; Provan, J. What do we mean by ‘refugia’? Quat. Sci. Rev. 2008, 27, 2449–2455. [Google Scholar] [CrossRef]
- Hajar, L.; Khater, C.; Cheddadi, R. Vegetation changes during the late Pleistocene and Holocene in Lebanon: A pollen record from the Bekaa Valley. Holocene 2008, 18, 1089–1099. [Google Scholar] [CrossRef]
- Pearson, R.G. Climate change and the migration capacity of species. Trends Ecol. Evol. 2006, 21, 111–113. [Google Scholar] [CrossRef]
- Hannah, L.; Flint, L.; Syphard, A.; Moritz, M.A.; Buckley, L.B.; McCullough, I.M. Fine-grain modeling of species’ response to climate change: Holdouts, stepping-stones, and microrefugia. Trends Ecol. Evol. 2014, 29, 390–397. [Google Scholar] [CrossRef]
- Keppel, G.; Mokany, K.; Wardell-Johnson, G.W.; Phillips, B.; Welbergen, J.; Reside, A. The capacity of refugia for conservation planning under climate change. Front. Ecol. Environ. 2015, 13, 106–112. [Google Scholar] [CrossRef]
- Rull, V. Microrefugia. J. Biogeogr. 2009, 36, 481–484. [Google Scholar] [CrossRef]
- Barrows, C.W.; Ramirez, A.R.; Sweet, L.C.; Morelli, T.L.; Millar, C.I.; Frakes, N.; Rodgers, J.; Mahalovich, M.F. Validating climate-change refugia: Empirical bottom-up approaches to support management actions. Front. Ecol. Environ. 2020, 18, 298–306. [Google Scholar] [CrossRef]
- Dubois, J.; Cheptou, P.-O. Effects of fragmentation on plant adaptation to urban environments. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160038. [Google Scholar] [CrossRef] [Green Version]
- Leimu, R.; Fischer, M. A Meta-Analysis of Local Adaptation in Plants. PLoS ONE 2008, 3, e4010. [Google Scholar] [CrossRef] [Green Version]
- Young, A.; Boyle, T.; Brown, T. The population genetic consequences of habitat fragmentation for plants. Trends Ecol. Evol. 1996, 11, 413–418. [Google Scholar] [CrossRef]
- Petit, R.J.; Hampe, A. Some Evolutionary Consequences of Being a Tree. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 187–214. [Google Scholar] [CrossRef] [Green Version]
- Davis, M.B.; Shaw, R.G. Range Shifts and Adaptive Responses to Quaternary Climate Change. Science 2001, 292, 673–679. [Google Scholar] [CrossRef] [Green Version]
- Christmas, M.J.; Breed, M.F.; Lowe, A.J. Constraints to and conservation implications for climate change adaptation in plants. Conserv. Genet. 2016, 17, 305–320. [Google Scholar] [CrossRef] [Green Version]
- Watson, J.; Dudley, N.; Segan, D.B.; Hockings, M. The performance and potential of protected areas. Nature 2014, 515, 67–73. [Google Scholar] [CrossRef]
- Albrich, K.; Rammer, W.; Seidl, R. Climate change causes critical transitions and irreversible alterations of mountain forests. Glob. Chang. Biol. 2020, 26, 4013–4027. [Google Scholar] [CrossRef] [PubMed]
- Morelli, T.L.; Barrows, C.W.; Ramirez, A.R.; Cartwright, J.M.; Ackerly, D.D.; Eaves, T.D.; Ebersole, J.L.; Krawchuk, M.A.; Letcher, B.H.; Mahalovich, M.F.; et al. Climate-change refugia: Biodiversity in the slow lane. Front. Ecol. Environ. 2020, 18, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Michalak, J.L.; Stralberg, D.; Cartwright, J.M.; Lawler, J.J. Combining physical and species-based approaches improves refugia identification. Front. Ecol. Environ. 2020, 18, 254–260. [Google Scholar] [CrossRef]
- Carroll, C.; Roberts, D.R.; Michalak, J.; Lawler, J.J.; Nielsen, S.E.; Stralberg, D.; Hamann, A.; McRae, B.H.; Wang, T. Scale-dependent complementarity of climatic velocity and environmental diversity for identifying priority areas for conservation under climate change. Glob. Chang. Biol. 2017, 23, 4508–4520. [Google Scholar] [CrossRef] [PubMed]
- Dagher-Kharrat, M.B.; El Zein, H.; Rouhan, G. Setting conservation priorities for Lebanese flora—Identification of important plant areas. J. Nat. Conserv. 2018, 43, 85–94. [Google Scholar] [CrossRef]
- Hannah, L.; Midgley, G.; Andelman, S.; Araújo, M.B.; Hughes, G.; Martinez-Meyer, E.; Pearson, R.; Williams, P. Protected area needs in a changing climate. Front. Ecol. Environ. 2007, 5, 131–138. [Google Scholar] [CrossRef]
- Lawler, J.J.; Rinnan, D.S.; Michalak, J.L.; Withey, J.C.; Randels, C.R.; Possingham, H.P. Planning for climate change through additions to a national protected area network: Implications for cost and configuration. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190117. [Google Scholar] [CrossRef] [Green Version]
- Wolf, H. TEMPERATE ECOSYSTEMS | Spruces, Firs and Larches. Encycl. For. Sci. 2004, 1449–1458. [Google Scholar] [CrossRef]
- Farjon, A. Pinaceae. Drawings and Descriptions of the Genera. In Koeltz Scientific Books; Koeltz Botanical Books: Königstein, Germany, 1990; ISBN 3-87429-298-3. [Google Scholar]
- Miller, R.O. Ecology and Managemnet of white Cedar. In Proceedings of the Workshop on Northern White Cedar in Michigan; Department of Forestry, Michigan State University: Escanaba, MI, USA, 1990; pp. 1–14. Available online: https://www.canr.msu.edu/uploads/files/Research_Center/FBIC/Northern_White_Cedar/Ecology_and_Mgmt_of_Northern_White_Cedar.pdf (accessed on 5 December 1990).
- Wright, J. Pollen dispersion of some forest trees. In Station Paper 46; United States Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: Upper Darby, PA, USA, 1952; p. 42. [Google Scholar]
- Poska, A.; Pidek, I.A. Pollen dispersal and deposition characteristics of Abies alba, Fagus sylvatica and Pinus sylvestris, Roztocze region (SE Poland). Veg. Hist. Archaeobot. 2010, 19, 91–101. [Google Scholar] [CrossRef]
- Awad, L.; Fady, B.; Khater, C.; Roig, A.; Cheddadi, R. Genetic Structure and Diversity of the Endangered Fir Tree of Lebanon (Abies cilicica Carr.): Implications for Conservation. PLoS ONE 2014, 9, e90086. [Google Scholar] [CrossRef] [Green Version]
- Dagher-Kharrat, M.B.; Mariette, S.; Lefèvre, F.; Fady, B.; March, G.G.-D.; Plomion, C.; Savouré, A. Geographical diversity and genetic relationships among Cedrus species estimated by AFLP. Tree Genet. Genomes 2006, 3, 275–285. [Google Scholar] [CrossRef]
- Douaihy, B.; Restoux, G.; Machon, N.; Bou Dagher-kharrat, M. Ecological characterization of the Juniperus excelsa stands in Lebanon. Ecol. Mediterr. 2013, 39, 169–180. [Google Scholar] [CrossRef]
- Fady, B.; Lefèvre, F.; Vendramin, G.G.; Ambert, A.; Régnier, C.; Bariteau, M. Genetic consequences of past climate and human impact on eastern Mediterranean Cedrus libani forests. Implications for their conservation. Conserv. Genet. 2008, 9, 85–95. [Google Scholar] [CrossRef]
- Semaan, M.T.; Dodd, R.S. Genetic variability and structure of the remnant natural populations of Cedrus libani (Pinaceae) of Lebanon. Tree Genet. Genomes 2008, 4, 757–766. [Google Scholar] [CrossRef]
- El Hasnaoui, Y.; Mhammdi, N.; Bajolle, L.; Nourelbait, M.; Bouimetarhan, I.; Cheddadi, R. Locating North African microrefugia for mountain tree species from landscape ruggedness and fossil records. J. Afr. Earth Sci. 2020, 172, 103996. [Google Scholar] [CrossRef]
- Kübler, D.; Hildebrandt, P.; Günter, S.; Stimm, B.; Weber, M.; Mosandl, R.; Muñoz, J.; Cabrera, O.; Zeilinger, J.; Silva, B. Assessing the importance of topographic variables for the spatial distribution of tree species in a tropical mountain forest. Erdkunde 2015, 70, 19–47. [Google Scholar] [CrossRef]
- Lembrechts, J.J.; Hoogen, J.; Aalto, J.; Ashcroft, M.B.; De Frenne, P.; Kemppinen, J.; Kopecký, M.; Luoto, M.; Maclean, I.M.D.; Crowther, T.W.; et al. Global maps of soil temperature. Glob. Chang. Biol. 2021, 28, 3110–3144. [Google Scholar] [CrossRef]
- Zhang, Y.; Schaap, M.G.; Zha, Y. A high-resolution global map of soil hydraulic properties produced by a hierarchical parameteri-zation of a physically based water retention model. Water Resour. Res. 2018, 54, 9774–9790. [Google Scholar] [CrossRef] [Green Version]
- USGS EROS Archive-Digital Elevation-Global 30 Arc-Second Elevation (GTOPO30). Available online: https://doi.org/10.5066/F7DF6PQS (accessed on 11 July 2018). [CrossRef]
- Riley, S. A terrain ruggedness Index that quantifies topographic heterogeneity. Int. J. Soil Sci. 1999, 5, 23–27. [Google Scholar]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. 2018. Available online: http://qgis.osgeo.org (accessed on 11 March 2022).
- Karger, D.N.; Conrad, O.; Böhner, J.; Kawohl, T.; Kreft, H.; Soria-Auza, R.W.; Zimmermann, N.E.; Linder, H.P.; Kessler, M. Climatologies at high resolution for the earth’s land surface areas. Sci. Data 2017, 4, 170122. [Google Scholar] [CrossRef] [Green Version]
- Akhtar, N.; Brauch, J.; Ahrens, B. Climate modeling over the Mediterranean Sea: Impact of resolution and ocean coupling. Clim. Dyn. 2018, 51, 933–948. [Google Scholar] [CrossRef] [Green Version]
- Cheddadi, R.; Khater, C. Climate change since the last glacial period in Lebanon and the persistence of Mediterranean species. Quat. Sci. Rev. 2016, 150, 146–157. [Google Scholar] [CrossRef]
- Cheddadi, R.; Taberlet, P.; Boyer, F.; Coissac, E.; Rhoujjati, A.; Urbach, D.; Remy, C.; Khater, C.; Antry, S.; Aoujdad, J.; et al. Priority conservation areas for Cedrus atlantica in the Atlas Mountains, Morocco. Conserv. Sci. Pract. 2022, E12680, 1–15. [Google Scholar] [CrossRef]
- Douaihy, B.; Vendramin, G.G.; Boratyński, A.; Machon, N.; Dagher-Kharrat, M.B. High genetic diversity with moderate differentiation in Juniperus excelsa from Lebanon and the eastern Mediterranean region. AoB Plants 2011, 2011, plr003. [Google Scholar] [CrossRef] [Green Version]
- Douaihy, B.; Sobierajska, K.; Jasińska, A.K.; Boratyńska, K.; Ok, T.; Romo, A.; Machon, N.; Didukh, Y.; Dagher-Kharrat, M.B.; Boratyński, A. Morphological versus molecular markers to describe variability in Juniperus excelsa subsp. excelsa (Cupressaceae). AoB Plants 2012, 2012, pls013. [Google Scholar] [CrossRef] [Green Version]
- Loarie, S.R.; Duffy, P.B.; Hamilton, H.; Asner, G.P.; Field, C.B.; Ackerly, D.D. The velocity of climate change. Nature 2009, 462, 1052–1055. [Google Scholar] [CrossRef]
- Sattout, E.J.; Nemer, N. Managing climate change effects on relic forest ecosystems: A program for Lebanese Cedar. Biodiversity 2008, 9, 122–130. [Google Scholar] [CrossRef]
- Klausmeyer, K.R.; Shaw, M.R. Climate Change, Habitat Loss, Protected Areas and the Climate Adaptation Potential of Species in Mediterranean Ecosystems Worldwide. PLoS ONE 2009, 4, e6392. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheddadi, R.; Khater, C. Guiding Conservation for Mountain Tree Species in Lebanon. Forests 2022, 13, 711. https://doi.org/10.3390/f13050711
Cheddadi R, Khater C. Guiding Conservation for Mountain Tree Species in Lebanon. Forests. 2022; 13(5):711. https://doi.org/10.3390/f13050711
Chicago/Turabian StyleCheddadi, Rachid, and Carla Khater. 2022. "Guiding Conservation for Mountain Tree Species in Lebanon" Forests 13, no. 5: 711. https://doi.org/10.3390/f13050711
APA StyleCheddadi, R., & Khater, C. (2022). Guiding Conservation for Mountain Tree Species in Lebanon. Forests, 13(5), 711. https://doi.org/10.3390/f13050711







