Abstract
The occurrence of short-interval, severe wildfires are increasing drastically at a global scale, and appear as a novel phenomenon in areas where fire historically returns in large time lapses. In forest ecosystems, these events induce drastic changes in population dynamics, which could dramatically impact species diversity. Here, we studied the effect on diversity of recent short-interval, severe wildfires (SISF), which occurred in rapid succession in the summers of 2002 and 2015 in Chilean Northern Patagonian Araucaria–Nothofagus forests. We analyzed the diversity of deadwood-dependent (i.e., saproxylic) and fire-sensitive beetles as biological indicators across four conditions: 2002-burned areas, 2015-burned areas, SISF areas (i.e., burned in 2002 and again in 2015), and unburned areas. Saproxylic beetles were collected using window traps in 2017 to 2019 summer seasons. To investigate the mechanisms underpinning the fire-related disturbance of the assemblage, we evaluated the effects of post-fire habitat quality (e.g., dead wood decomposition) and quantity (e.g., burned dead wood volume and tree density) on the abundances and species richness of the entire assemblage and also multiple trophic groups. Compared with the unburned condition, SISF drastically reduced species richness, evenness, and Shannon’s diversity and altered the composition of the saproxylic beetle assemblages. The between-condition variation in composition was accounted for by a species replacement (turnover) between SISF and 2015-burned areas, but both species replacement and extinction (nestedness) between SISF and unburned areas. Dead wood decomposition and tree density were the variables with the strongest effects on the abundance and species richness of the entire saproxylic beetle assemblage and most trophic groups. These results suggest that SISF, through degraded habitat quality (dead wood decomposition) and quantity (arboreal density), have detrimental impacts on diversity and population dynamics of saproxylic beetle assemblages. Therefore, habitat loss is a central mechanism underpinning fire-related biodiversity loss in these forest ecosystems.
1. Introduction
Climate change and human activities have induced significant alterations in wildfire dynamics at the global scale. In particular, the occurrence of high-severity events in forest ecosystems has steeply increased over the last few decades [1,2,3,4]. Thus, the time interval between consecutive fires (“fire return”) is becoming shorter across different forest ecosystems [5,6]. Such short-interval, severe wildfires (hereafter referred to as SISF) constitute a compound disturbance in reburned areas [7,8,9]. Fire returns are crucial for the population dynamics in many forest ecosystems [10]. However, the response capacity of these ecosystems has been largely exceeded under the current global warming scenario [11,12]. Additionally, wildfires are becoming more frequent in areas where large fires have been historically less frequent, resulting in an increased risk of habitat loss [12,13]. Therefore, SISF have recently become a focus of interest for ecology due to their additive effects on forest ecosystems [8,12,14,15,16].
Depending on the frequency, severity, and extent of fire events, plants [8,12] and animals [17,18,19] can respond differentially in forest ecosystems. For instance, in conifer-dominated forests, two overlapping severe wildfires (<15 y interval) had a negative impact on legacies, reducing the dead wood mass [7], but neutral effect on bird communities [20]. Relatively frequent low- or moderate-severity fires are beneficial for many fire-dependent plant species [8,21]; recurrent high-severity fires, however, have been shown to reduce plant diversity [22,23].
There is an ample recognition that SISF can result in either state changes or alternative stable states in temperate and tropical forest ecosystems [24,25,26,27]. A recent rise in wildfires linked to a warmer climate is transforming the structure, composition, and function of temperate forest ecosystems. For instance, high-severity fires have been transforming Nothofagus pumilio forests into shrublands in Patagonia, Argentina [23]; the Eucalyptus forests into non-arboreal forest stages in Victoria, Australia [28]; and the conifer-dominated forests into shrub/hardwood chaparral in Oregon, USA [29]. Therefore, SISF have become a novel phenomenon due to the increase of dry and warmer conditions and human activities [30,31]. Empirical studies of animal communities’ response to novel fire regimens are still scarce, and elucidating the underlying mechanisms of the resulting diversity changes is critical to develop or improve management and conservation practices.
In this study, we evaluate the effect of SISF on beetle diversity in temperate northwest Patagonia Andean forests. These landscapes represent the last habitat refugia for native and red-listed Araucaria araucana Molina K. Kosh trees [32,33] in the Southern Cone of America, and provide an unique habitat for rare and endangered animal and plant species. For this purpose, we used saproxylic beetles (deadwood-dependent species [34]) as biological indicators of the post-fire diversity in Araucaria–Nothofagus forests [35]. These beetles are a proxy frequently used in many ecological studies relative to management practices [36], forest restorations [37], and ecosystem service assessment [38,39], among others. Moreover, saproxylic beetles are highly sensitive to wildfire impacts in different forest types [35,40,41,42], and are key for post-fire restoration due to biogeochemical facilitation [43]. In addition, Araucaria’s beetle assemblages are characterized by the presence of endemic [44,45], endangered [46,47], and rare species [48,49], being a group with special risk of diversity loss due to wildfires in northern Patagonian forests.
In post-fire forests, most of the saproxylic community changes (diversity and assemblage composition) are produced by the alteration of the quality (e.g., decaying stages and species type) and quantity (e.g., dead wood volume and arboreal density) of dead wood [41,50]. Therefore, the compounded effect (i.e., additive losses of deadwood volume) induced by SISF on dead wood accumulation [7] could be a critical factor to persistence of many saproxylic species [41]. Moreover, in Andean Araucaria–Nothofagus forests, the high-severity fire has a negative impact on saproxylic beetles assemblages [35], and on the recruitment of fire-resistant trees [51]. Thus, SISF may produce significant impact on saproxylic species as a result of losses of dead wood volume and habitat modifications [7,8,12].
We tested four hypotheses relative to the diversity of saproxylic beetle assemblages affected by short-interval severe wildfires: (1) SISF have a negative effect on saproxylic beetles diversity and induce changes in beetle assemblages; (2) severe reburn-related diversity decline will impact the spatial variation in community composition (beta-diversity), increasing local species turnover (species replacement) over space; (3) the quality and quantity of wood legacy will show significant impacts on saproxylic beetle communities; (4) finally, changes in species richness, species compositions, and abundances of saproxylic beetles will be reflected in the abundances of the trophic guilds. All these predictions are based on the premise that short-interval, high-severity wildfires are infrequent phenomena in Andean Araucaria forests [52,53,54], and therefore most species in these communities are not well-adapted to this short-interval return of fire.
2. Materials and Methods
2.1. Study Area
Fieldwork was conducted in two national parks in the Andean range in the Araucanía region, dominated by Araucaria araucana (Molina) K. Koch (araucaria or pehuén) and Nothofagus-type species.
Tolhuaca National Park (38°12′ S, 71°41′ W) (henceforth: TNP) has distinct forest associations along the altitudinal gradient. From 900 to 1300 m asl, secondary and old-growth forests composed mainly of Nothofagus dombeyi (Mirb.) Oerst., N. nervosa (Poepp. & Endl.) Oerst., and N. pumilio (Poepp. & Endl.) Krasser are found. Over 1300 m asl, old-growth A. araucana, N. dombeyi, and N. pumilio are the dominant plant associations. In the subalpine bottom valley, sparse Araucaria trees are associated with N. antarctica, conforming an open forest woodland. Along this altitudinal gradient, Chusquea culeou E. Desv. forms dense bushes between 2 and 4 m high, together with other shrub species [33].
China Muerta National Reserve (38°44′ S, 71°31′ W) (henceforth: CMNR) covers an area of 12,825 ha along an altitudinal range between 800 and 1850 m asl. The main forest associations are dominated by Araucaria–Nothofagus forests and mixed Nothofagus forests. Similar to in TNP, C. culeou, Maytenus disticha (Hook. f.) Urb., and Gaultheria poepigii DC. are common shrub species in the undergrowth [33,51]. In the past, these forests have been affected by extensive livestock ranching and the extraction of non-wood products [31,55].
2.2. Fire Regimes in TNP and CMNRs Parks
Tree-ring-based reconstructions of fire regimes indicate that Araucaria–Nothofagus forests have been shaped by mixed-severity fires that span a continuum of low-severity surface fires to high-severity crown fires [56,57]. Comparisons between the Native American period and Euro–Chilean settlement show that fires have become more frequent as a result of the colonization process after 1880 [56,57]. Subsequently, the protection status and better fire prevention granted to these forested areas resulted in a lower occurrence of fires, especially after the 1950s [56,57]. However, in the last two decades, TNP has been burned by two extensive (>60% of the protected area burned) high-severity wildfires in a short time span, i.e., in 2002 and 2015 [52,54,58]. The 2002 and 2015 fire events were caused by a natural and human source, respectively, with an important overlap between their burned areas. On the other hand, the CMNR suffered a mainly high-severity fire in the summer of 2015 that affected an estimated forest area of 3765 ha [59].
This study was conducted in four different conditions of burned and unburned Araucaria–Nothofagus forests: (a) forests burned in 2002, (b) forests burned in 2015, (c) forests reburned (burned in 2002 and reburned in 2015), and (d) unburned forests (Figure 1). Our measurements were established in forests with partially or totally burned undergrowth, and with an estimated mortality of >90% of trees, defined as high-severity burned forests [59,60].
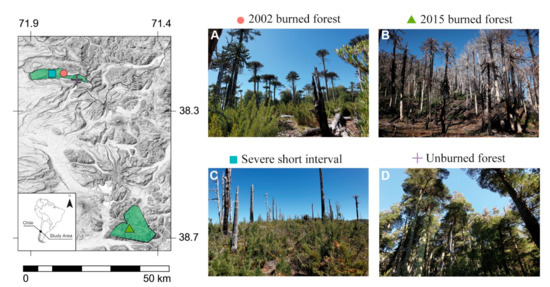
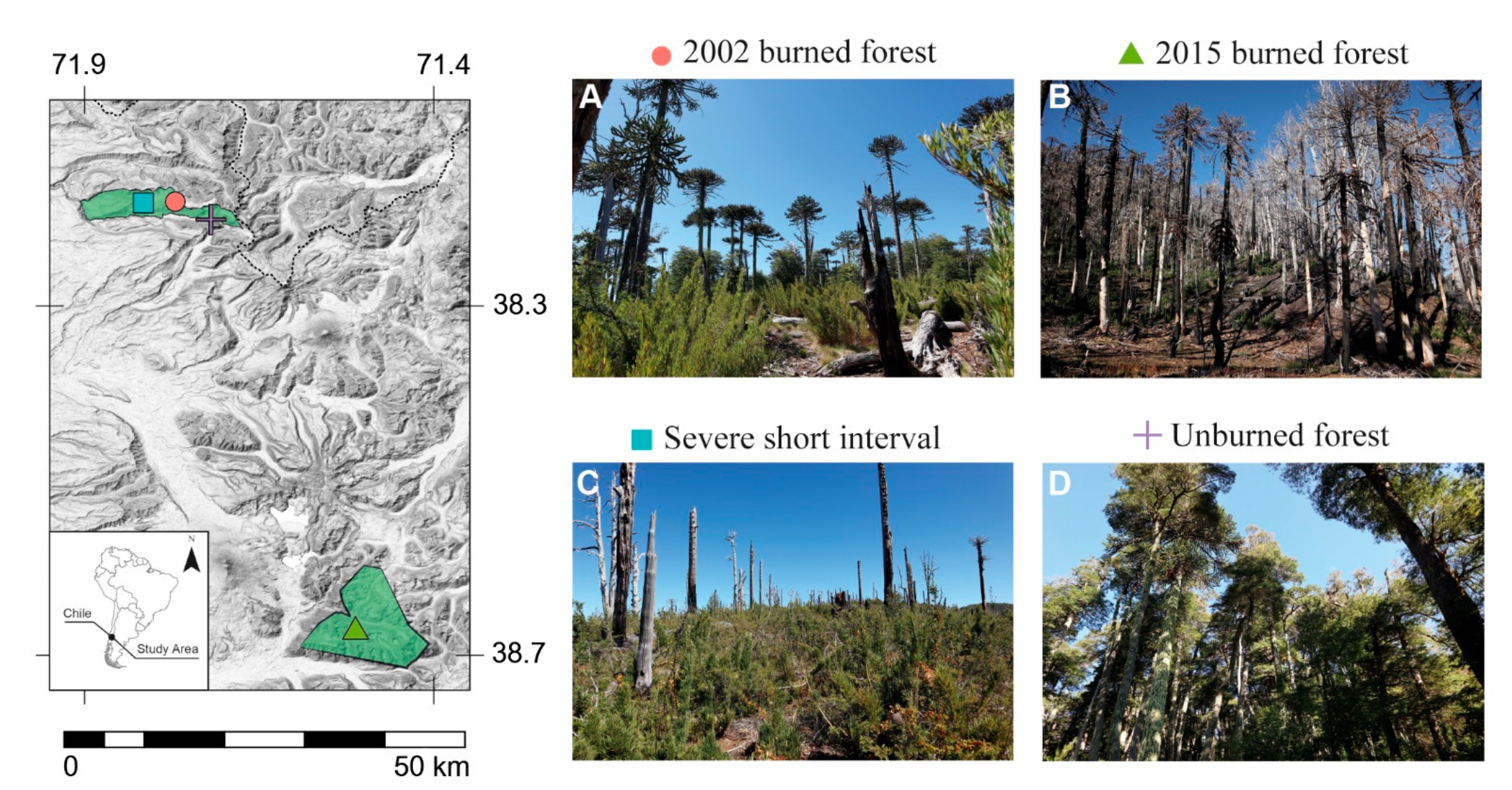
Figure 1.
Selected sampling areas in Araucaria–Nothofagus forests from northern Patagonian Chilean landscape. (A) Unburned areas; (B) short-interval, severe areas (reburned forest); (C) 2015-burned areas (recently burned forest); and (D) 2002-burned areas (old-burned forest).
2.3. Sampling Method and Field Design
We randomly selected six 0.05 ha plots in each of the unburned, 2002-burned, 2015-burned, and SISF conditions. To reduced unwanted variability and potential confounding effects, the sampling plots were selected so that they all maintained similar environmental characteristics, defined as Araucaria–Nothofagus forests type with A. araucana, N. nervosa, and N. pumilio as the dominant species, and N. dombeyi in lesser proportion; between 1200 and 1500 m asl; understory with presence of C. culeou and Gaultheria sp. as dominant shrubs.
In each sampling plot (24 in total), we collected adults of saproxylic beetles using tree-trunk flight-interception traps (henceforth: Wts), one in each sampling plot, consisting of transparent cross vanes (55 cm long, 40 cm width) lying over a funnel and a collection container with preserving glycerol liquid (water, salt, and 5% glycerin). The Wts clearly responded to the immediate surroundings of the trap (i.e., the saproxylic habitat). Thus, traps located on tree trunks recorded a different species assemblage than those collected by freely hanging traps, baited tube, pitfalls, and malaise, among other traps [61,62]. Therefore, Wts have been widely used in many forests to collect active flying and flightless saproxylic beetles associated with a wide range of woody resources, such as decaying aerial branches, bark, or tree hollows, and also to deadwood on the ground [62,63,64,65]. In order to minimize the probability to capture non-saproxylic insects, we installed Wts at 1.5–2 m above the ground on dead Araucaria trees with >40 cm DBH. Each trap was separated by at least ~150 m to avoid interference effects between the traps [61]. Wts were monitored monthly from November 2017 to March 2018 and from December 2018 to April 2019. However, it was not possible to collect insects in all months for all sampling plots because occasionally Wts were affected by tree falls (in TNP and CMRN), wind gusts (in TNP), or destroyed by cattle activity (in CMRN). Thus, we obtained 79 replications; 28 in 2002-burned areas, 21 in control areas, 15 in 2015-burned areas, and 15 in SISF areas.
Saproxylic beetles were identified at the lowest possible taxonomical level, i.e., family, tribe, genus, species, or morphospecies. For this purpose, we used the available taxonomical literature for Chilean species with emphasis in Araucaria forest-inhabiting beetles [26,44,45,48,66,67,68,69,70,71,72]. Additionally, we compared the caught insects with the material stored in the Museo Nacional de Historia Natural (M. Elgueta: Santiago, Chile), and Colección Entomológica Ernesto Krahmer (C. Montalva: Valdivia, Chile). Moreover, to resolve the delimitation of cryptic species, we consulted with the specialist in each group (e.g., M. Elgueta and G. Arriagada for multiples families, A. Lüer for Ptinidae, and L. Kirkendall for Scolytinae).
2.4. Ecological Variables
The variety of forest conditions generated by fire was represented by a wide range of continuous and categorical environmental variables, measured both at the stand and tree scale. To estimate the stand variables, we established a circular plot of 500 m2 (~0.05 ha) around the Wts in each sampling plot. This area is sufficiently large to avoid unrealistically high estimates, but small enough to capture small-scale variation [73,74].
At stand level we evaluated the following:
- -
- Arboreal density (arboreal_D, continuous). We registered all standing dead and living trees with diameter ≥5 cm in each selected plot. The data were extrapolated to hectare.
- -
- Dead wood volume (dead_vol, continuous). In each sampling plot, we registered all woody debris on the ground (logs) with diameter ≥10 cm, and all the dead tree branches with stem diameter ≥10 cm and length ≥2 m. For each log or branch registered, we recorded total length (m), and diameter (cm) at the base and top. Additionally, we recorded the diameter at breast height (dbh) and total height of each standing dead and living tree with diameter ≥5. To estimate the wood volume, a cylindrical shape was assumed for each piece. Finally, the data was extrapolated to hectare (see Supplementary Material S1).
At tree trap level (Wts) we recorded the following:
- -
- Diameter (dbh_WT, continuous). The dbh of each tree where the traps were installed.
- -
- Decomposition stage (dec_WT: five categories 0–4, categorical). Decomposition stages were recorded based on the adapted method used by Carmona et al. [75] (Table 1).
 Table 1. Qualitative classification system of decay classes.
Table 1. Qualitative classification system of decay classes. - -
- Burned area (burn_WT: five categories 0–4; categorical). We estimated the burned area (i.e., carbonized area) of each trap tree, considering: 0, unburned; 1, ≥1 to 25%; 2, ≥26 to 50%; 3, ≥51 to 75%; 4, ≥76% to 100% burned area.
- -
- Bark cover (bark_WT: presence or absence, categorical). We consider two categories of bark cover: present, when the trunk is covered by bark on at least 75% of its total surface; absent, when the bark is absent from at least 75% of its total surface.
2.5. Data Analysis
First, we evaluated the effects of fire condition on saproxylic species richness and composition. For this, we used sampled-size-based rarefaction–extrapolation analysis [76] to estimate the differences in species richness between beetle assemblages on different fire conditions. For the analysis of differences in diversity, we used iNEXT R package to compute the Hill’s numbers, corresponding to q = 0, species richness; q = 1, exponential Shannon entropy; and q = 2, inverse of Simpson [76,77]. Based on species frequency (i.e., the number of samples in which a species occurred, regardless of abundance within a sample), this method allowed us to avoid biases caused by different sampling efforts [78]. Additionally, we used PERMANOVA [79] to assess the differences in community composition between fire conditions. Comparisons were performed using the adonis function in the vegan R package, based on Bray–Curtis dissimilarity matrices and 999 random permutations. For this purpose, data was log10(n + 1) transformed to avoid the effect of the most abundant species. To visualize the differences in composition of beetle assemblages, we performed an agglomerative clustering, using the metaNMDS function in the vegan package, and plotted with the ggplot2 R package.
Secondly, we analyzed the changes in beta-diversity between post-fire saproxylic assemblages. Beta-diversity is the differences in species composition between local communities [80]. Thus, beta-diversity patterns may arise from species replacement (turnover) or from species loss/gain related to species richness variation (nestedness-resultant), and each component can have distinct underlying processes (i.e., turnover and nestedness [81]). This allowed us to observe the effect of the different fire conditions on the turnover and persistence/removal of saproxylic species in post-fire forests [82]. The patterns of species turnover and nestedness-resultant components were examined using the beta-diversity partitioning method [81]. We used the function beta.pair of the betapart R package to calculate dissimilarities [83], as average values derived from pairwise comparisons are different from those values calculated using multiple-site comparisons. For this purpose, we used Sørensen dissimilarity (βsør) to calculate pairwise measurements, which was partitioned into turnover (βsim) and nestedness (βnes) dissimilarity related to fire conditions.
Third, we tested the differences induced by the fire conditions on community abundances and number of species of trophic guilds. We grouped the species into trophic guilds based on larval life history and the biological information available in the published literature [44,47,48,68,69,71]. Thus, three trophic guilds were used: (1) mycophagous (fungivorous, including dead, rotting, and living fungus); (2) xylophagous (detritivores of dead wood); and (3) zoophagous (predators). The significance of observed differences in abundances and number of species was tested using one-way ANOVA tests (α = 0.05) and Tukey’s HSD post hoc pairwise comparisons in the aov function of the stats package and plotted with the ggplot2 R package. For abundances and number of species data, logarithm and square root formulas were applied, respectively.
Finally, generalized linear models (GLMs) were used to evaluate the effect of standardized stand and tree variables on species assemblages. We estimated the effects of habitat variables on diversity as total number of species, q = 1 values, and abundances, while the trophic responses were estimated using abundances and total number of species in each group. With this, we also aimed to assess whether the beetle community and the trophic guilds respond to the habitat post-fire conditions. For this analysis, dead wood volumes were log-transformed, and arboreal density was rescaled to avoid inconsistencies in the data analysis. We used the bias-corrected Akaike information criterion (AICc [84]) to identify the models with the strongest empirical support from a set of candidate models. Environmental data were assumed to be Poisson or Gaussian distributed, and thus, modeled with a log- and identity-link function, respectively. We conducted an automated model selection procedure using all possible combinations of predictor variables from a global model using the dredge function included in the MuMIn R package. The models were selected using ΔAICc < 4 criterion, and the maximum number of factors involved in each model was chosen based on the number of sampling replications (e.g., equal to 2). Afterwards, we calculated a pseudo R2 value (McFadden’s pseudo R2) for each selected models using the RsqGLM function in the modEvA R package [85,86]. McFadden’s pseudo R2 values generally above 0.2 are considered an “excellent fit” [87]. Additionally, we displayed the effect size of the explanatory variables by plotting the estimate parameters of the best performing model, using plot_model function in sjPlots R package [88]. For plot_model function, we used the decomposition stage I (Decaying_I) as mechanism of comparison between all the other conditions of dead wood decaying. All analyses were performed in R v.3.6.3 (R Core Team, 2020).
3. Results
3.1. Saproxylic Beetle Catch
In the 79 samplings across the burned, reburned, and unburned Araucaria–Nothofagus forest, we collected a total of 7957 individuals of saproxylic beetles belonging to 42 families. In addition, we registered a total of 253 species/morpho-species, where the most abundant species were Araucarius minor Kuschel, 1966 (Curculionidae; 3315 individuals), and Enicmus transversithorax Dajoz 1967 (Latridiidae; 1185 individuals (see Supplementary Material S2).
3.2. Diversity Patterns
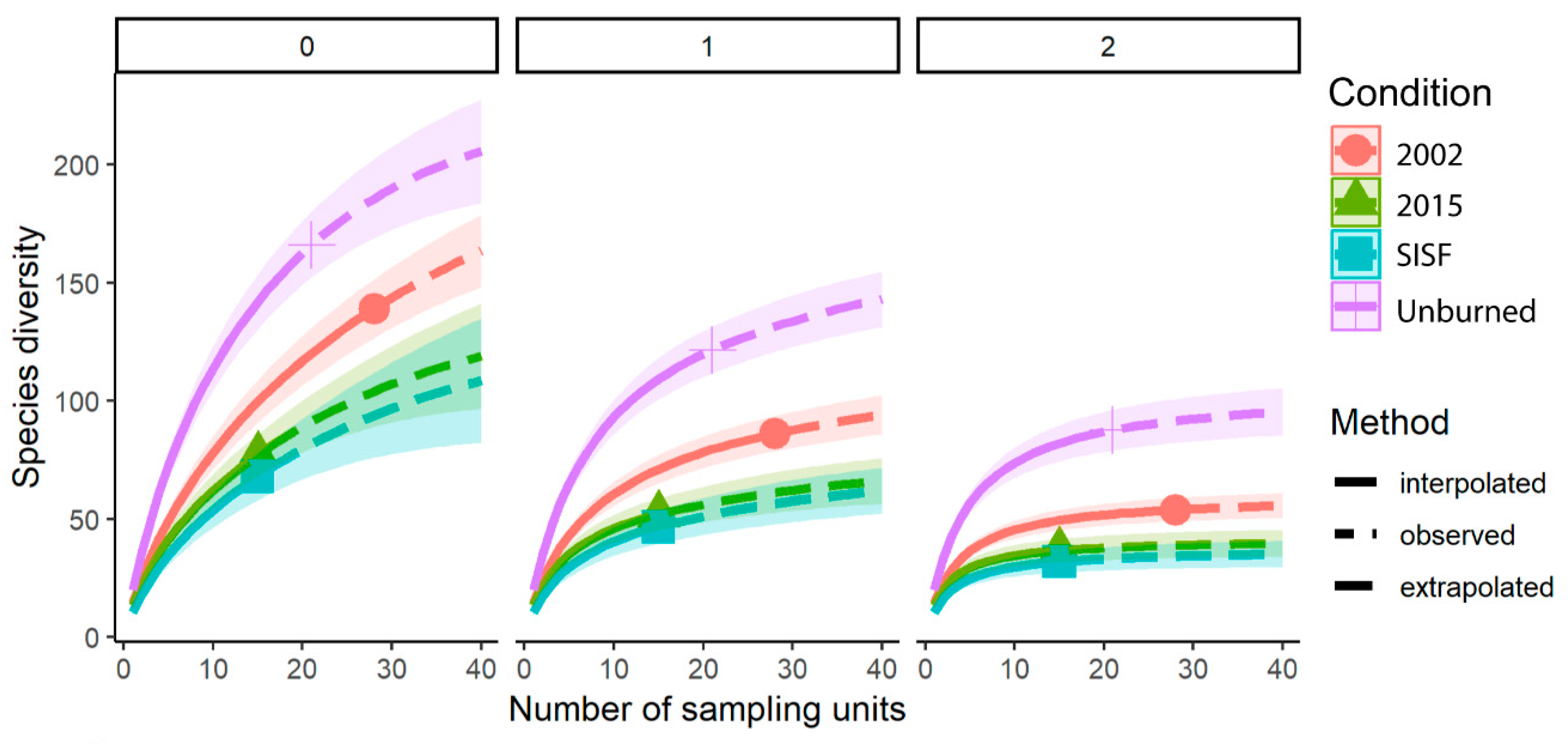
Rarefaction and extrapolation curves, based on Hill series analysis (q = 0, 1, 2), showed a significantly higher species richness in unburned forests compared to different burned conditions. In addition, this analysis demonstrated that both recently burned (2015) and reburned (2002–2015) forests harbored less diverse communities than previously burned forests (2002). No differences in species richness were observed between SISF and 2015 beetle assemblages (Figure 2). All patterns were consistent across the three diversity indexes (Figure 2).
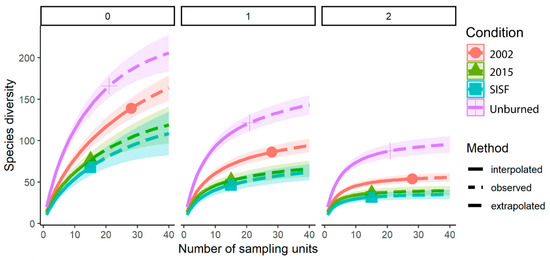
Figure 2.
Sample-based rarefaction (solid lines) and extrapolation (dotted lines) of saproxylic beetles collected in unburned areas (plus); short-interval, severe areas (square); 2015-burned areas (triangle); 2002-burned areas (dots), with 95% unconditional confidence intervals (transparent shading). Species diversity correspond to q = 0, species richness; q = 1, exponential of Shannon’s entropy index; q = 2, inverse of Simpson’s index. Solid symbols represent the total number of samples recovered in field work.
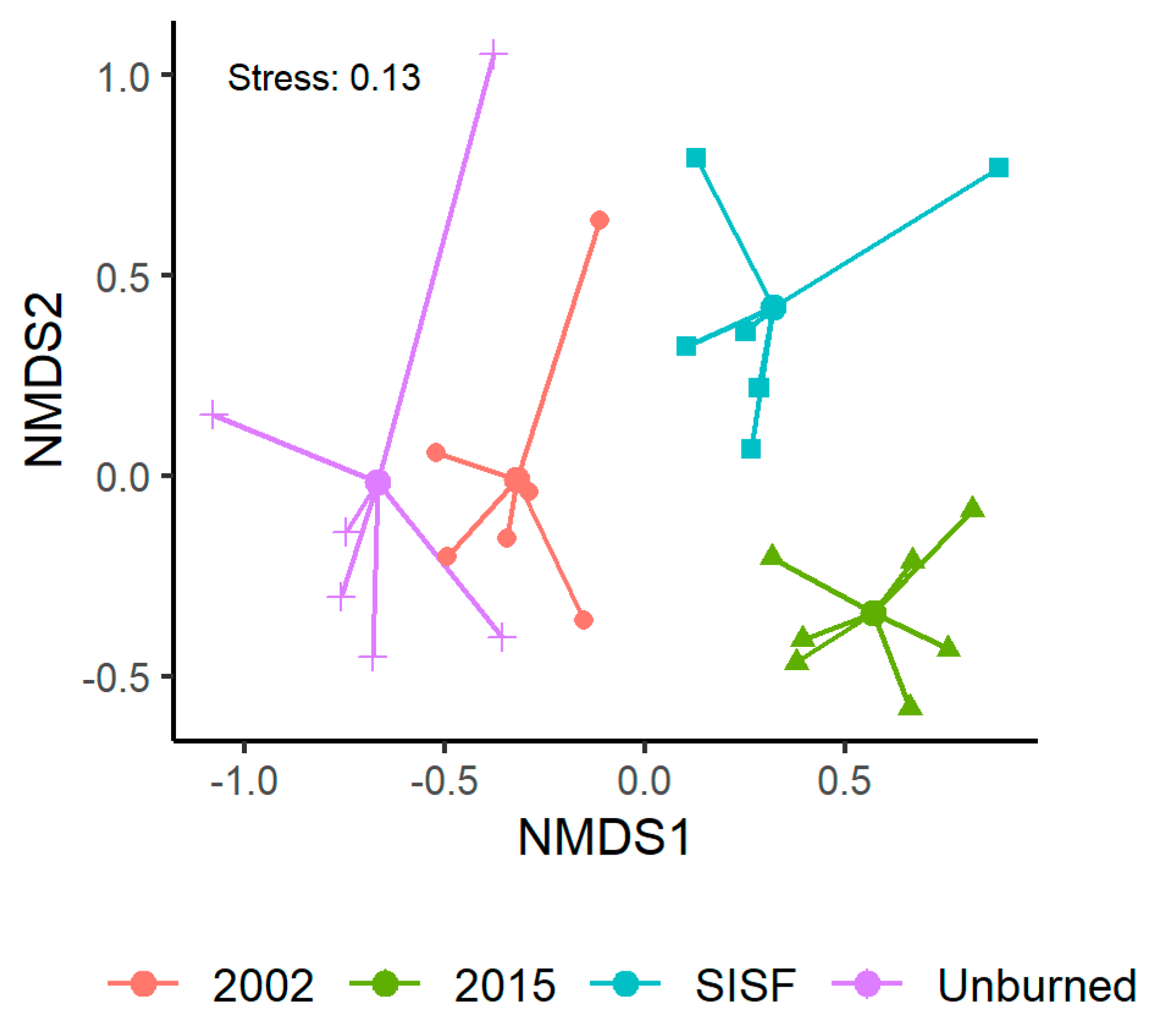
Saproxylic assemblage composition significantly varied among fire conditions (Table 2: PERMANOVA, pseudo F(3,21) = 4.19, p-value < 0.01), and NMDS ordinations revealed substantial site clustering according to the fire conditions of the forests (Figure 3, stress = 0.13).

Table 2.
Pairwise comparison of assemblage compositions between sampling locations. PERMANOVA tests are df 3, 21 = 4.19, p-value < 0.01.
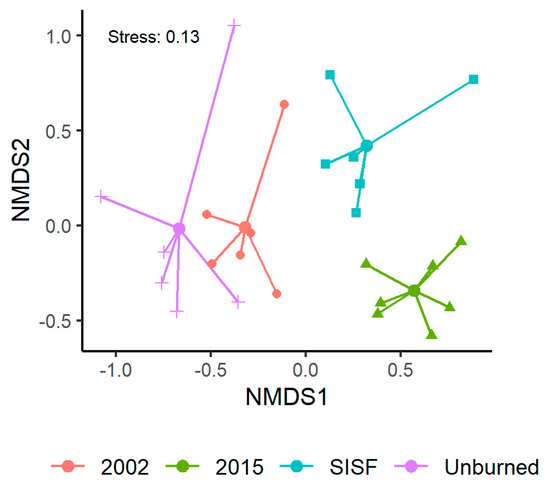
Figure 3.
Non-metric multidimensional scaling (NMDS) visualization of species composition between fire conditions.
3.3. Partition of Beta-Diversity
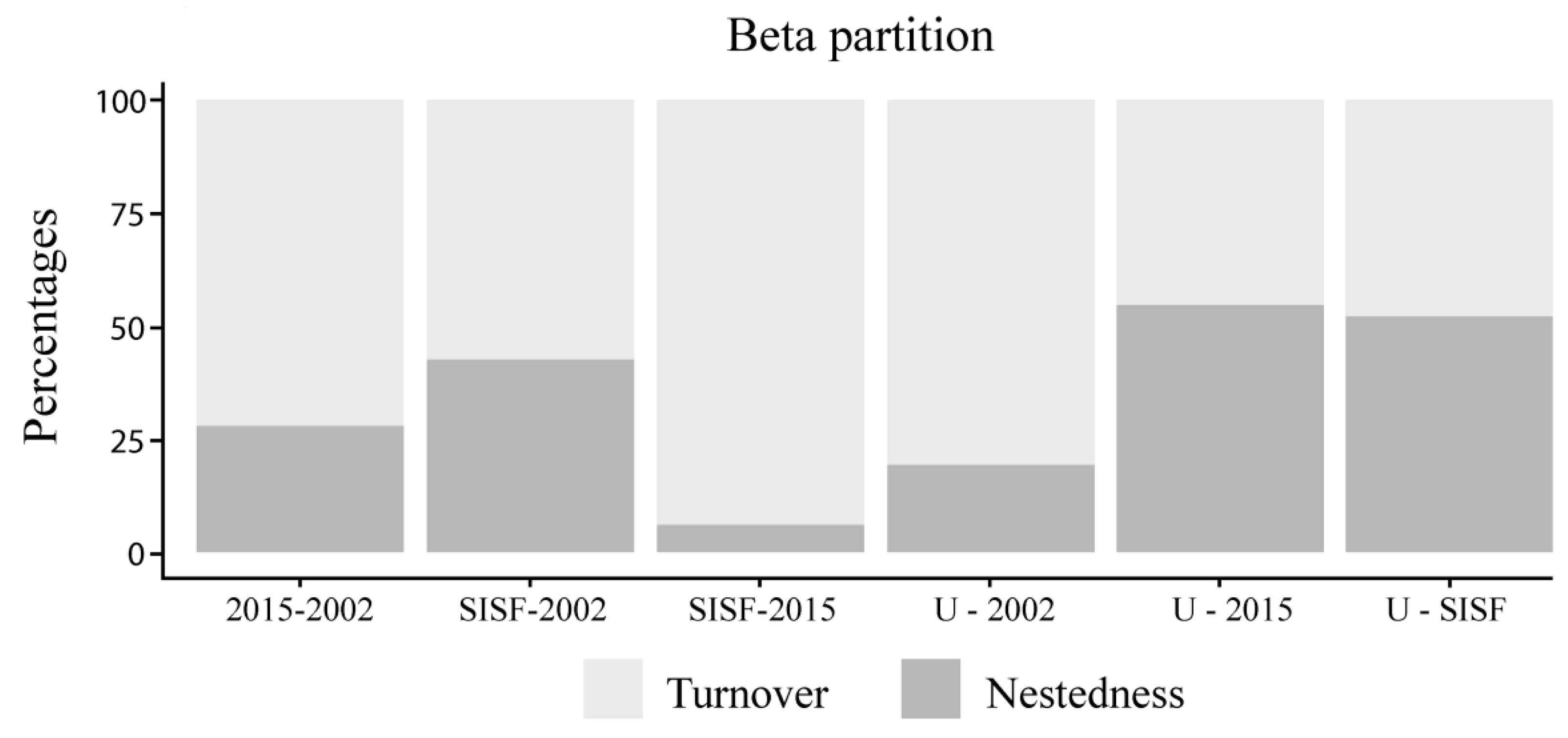
Species composition was most dissimilar between SISF and unburned assemblages (βSØR = 0.52). Therefore, dissimilarity due to nestedness (βnes = 0.27) was high between these conditions. In contrast, lower dissimilarity was found between 2002 and unburned assemblages (βsør = 0.32), and the majority of this dissimilarity was due to turnover (βsim = 0.25). Most of the total dissimilarity between SISF and 2015-burned assemblages (βsør = 0.48) was due to turnover (βsim = 0.45), while a smaller proportion was due to nestedness (βnes = 0.03) (Figure 4).
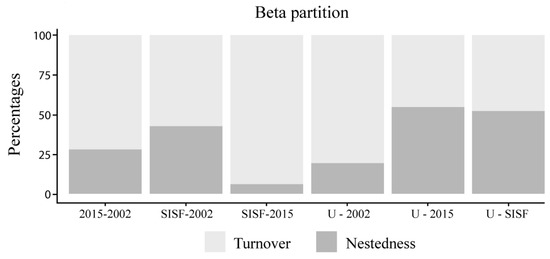
Figure 4.
Bar plot of the beta-diversity partition of the saproxylic beetle assemblages in Araucaria–Nothofagus forests. The analyzed conditions were 2002-burned areas (2002), 2015-burned areas (2015), reburned areas in 2002 and again in 2015 (SISF), and unburned forests (U).
3.4. Trophic Guilds Response
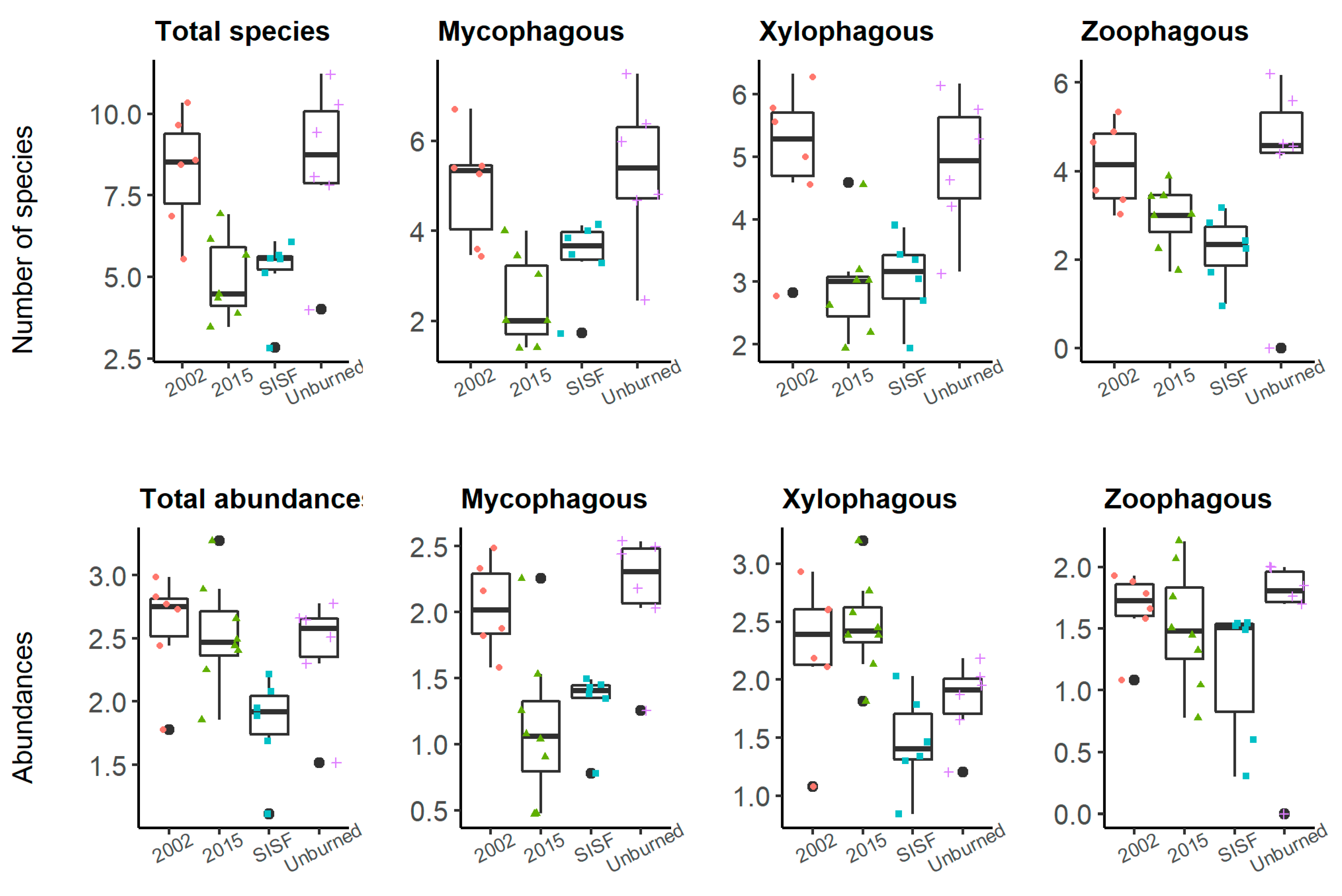
The number of species of the mycophagous group was higher in unburned and old-burned forests (2002) than recently burned forests (2015 and SISF, Figure 5), but no statistical differences were observed between unburned and SISF areas (ANOVA, p-value = 0.073). The number of xylophagous species was lower in both recently burned forests compared to the other burned and unburned conditions, and no differences were detected between SISF and 2015, and 2002 and unburned conditions. The number of species of the zoophagous groups did not vary across the burned conditions (Table 3 and Figure 5). Additionally, the abundances for each trophic guild were xylophagous: 6060, mycophagous: 2435, and zoophagous: 1256. ANOVA showed significant differences when we compared abundances of trophic guilds regarding the fire conditions (p-value < 0.01), except for the abundance of zoophagous beetles (p-value = 0.463). For mycophagous group, Tukey tests showed that unburned forest had significant higher abundances comparing with 2015 (p-value = 0.002) and SISF (p-value = 0.018) conditions, and no difference with 2002 abundances (p-value = 0.972). Comparisons of abundances in mycophagous group between burned conditions showed that 2002 assemblages were significantly more abundant compared with 2015 (p-value = 0.006) and SISF (p-value = 0.047), and no significant differences were detected between 2015 and SISF (p-value = 0.873). Abundances of xylophagous species varied significantly among conditions (ANOVA: p-value < 0.01). Tukey’s comparisons showed that SISF xylophagous abundances were significantly lower than 2002 (p-value = 0.035) and 2015 (p-value < 0.01) burned forests, and no significant differences in abundances were observed between unburned and SISF. Therefore, recently burned assemblages had a higher abundance but fewer number of species than old burned and unburned assemblages (Table 3 and Figure 5). In contrast, SISF areas harbored lower abundances and species richness, comparing between old burned and unburned conditions.
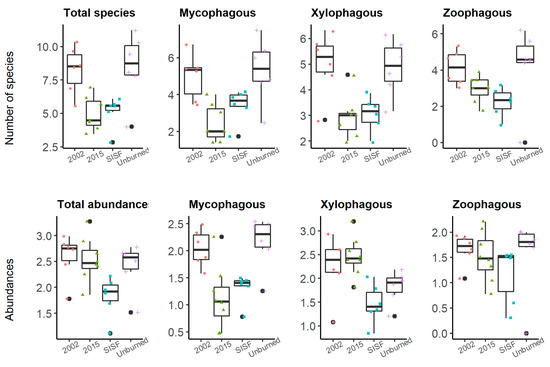
Figure 5.
Box plot of the square-root-transformed number of individuals in trophic guilds in unburned areas (plus); short-interval, severe areas (square); 2015-burned areas (triangle); 2002-burned areas (dots) of the Araucaria–Nothofagus Chilean forests.

Table 3.
Differences (p-value) of the trophic guilds abundances between sampling locations using Tukey HSD tests comparisons. Bold font refers to statistically significant Tukey tests.
3.5. Influence of Environmental Variables on Diversity Patterns
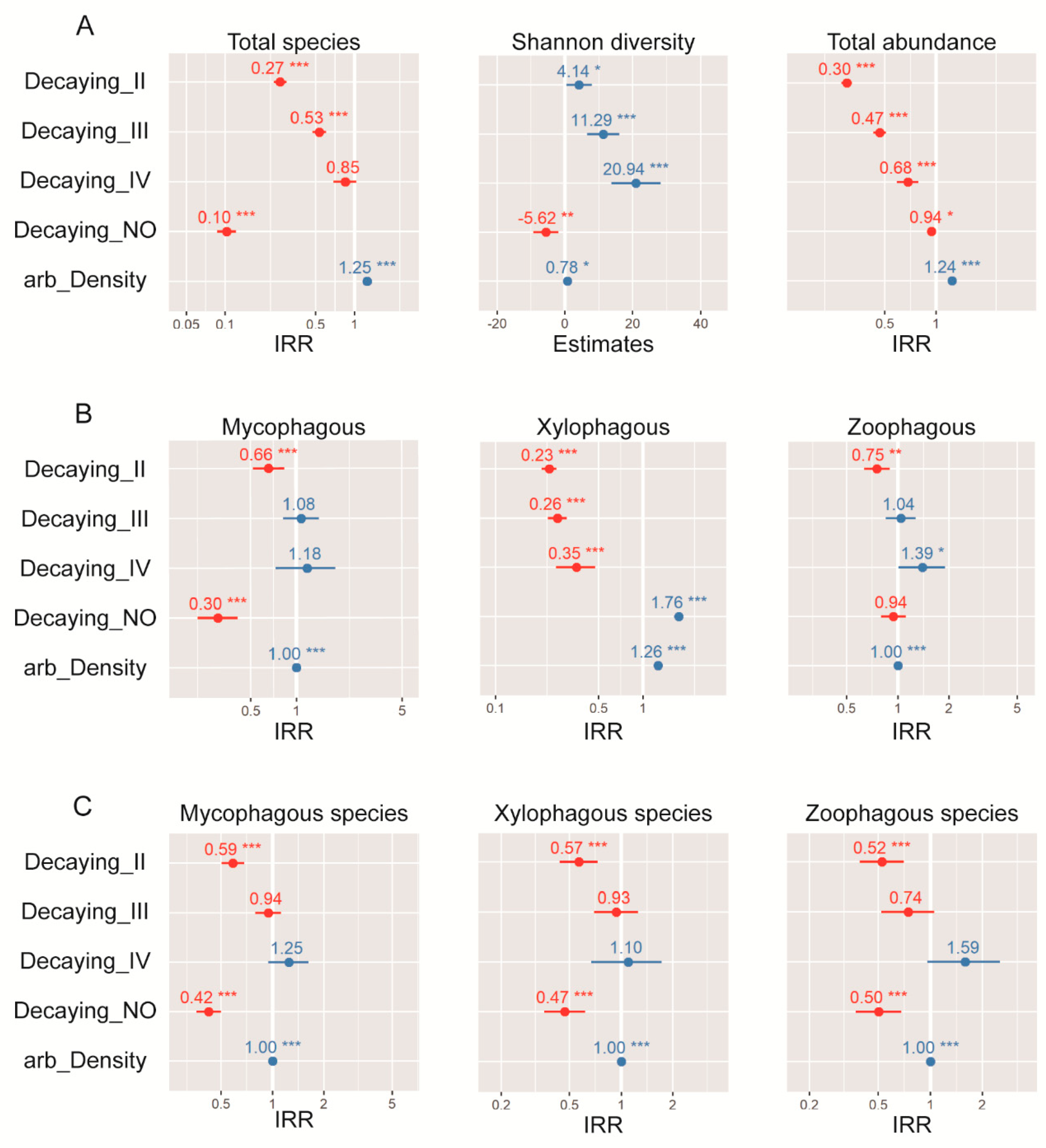
The model with the strongest empirical support (ΔAICc < 4) indicated that arboreal density and decaying stages were the variables that most affected both diversity and abundances of the saproxylic beetle assemblages (Table 4). Thus, species richness, exponential Shannon entropy (q = 1), and the abundance of the whole assemblage increased with greater arboreal density, and the decaying stages of wood showed a bimodal effect on beetles (e.g., panel 1: row 1 in Figure 6). The early decaying stages appeared related to higher abundances but lower species richness. In contrast, the advanced decaying stages promoted higher species richness but lower abundance of beetles. Regarding the trophic guilds abundances, xylophagous and mycophagous also were affected by arboreal density and decaying stages. High arboreal density increased the abundances of both groups, and undecomposed trees promoted high abundance of xylophagous but less abundance of mycophagous. Abundance of zoophagous was explained by two slightly different models: arboreal density and decaying stages, and total volume in each plot and arboreal density. In the first model (“arboreal density + decaying”: Akaike weight = 0.53, pseudo R2 = 0.22), the abundances increased with the advanced decaying stages and decreased in early decaying stages of trees. The second model (“arboreal density + volume of CWD”: Akaike weight = 0.47, pseudo R2 = 0.10) suggested that high volume of dead wood at stand level negatively affected the abundances of this group; however, the low pseudo R2 value indicated a low performance of this model. In both models, the arboreal density increased the abundances of the zoophagous group (Table 3 and Figure 6). Regarding trophic guilds diversity (i.e., number of species), all groups were less diverse in early decaying stages, while high arboreal density promotes higher number of species. Only the mycophagous species richness showed two types of response models, corresponding to “arboreal density + decaying stage” (Akaike weight = 0.78, pseudo R2 = 0.30), and “arboreal density + burn severity” (Akaike weight = 0.22, pseudo R2 = 0.33) (Table 3, Figure 6).

Table 4.
The best-supported (ΔAIC < 4) generalized linear models (GLM) testing for the effects of habitat variable on the species richness and abundances of total community, and the different trophic guilds number of species and abundances of saproxylic beetles.
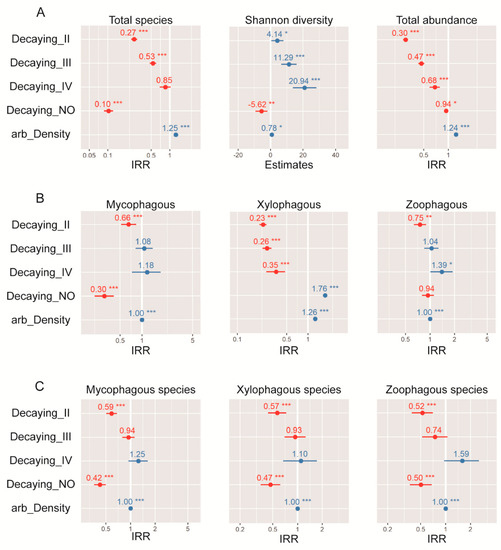
Figure 6.
GLM results for (A) total community, trophic guilds (B) abundances, and (C) diversity of saproxylic beetles in unburned and burned conditions. For significance of incidence rate ratios (IRR) and estimate, the SE bar must not cross the 0.0 line. The further away from 0, the larger the effect; red and blue colors represent positive and negative effects, respectably. Asterisks (*) refer to significant estimate (* <0.05, ** <0.01, *** <0.001). Fits of the models are represented in Table 3.
4. Discussion
4.1. Impacts of Post-Fire Habitat Conditions on Saproxylic Beetles Diversity
We found that abundances and species diversity were higher in plots with higher tree density. These results could be explained by higher habitat heterogeneity, providing for variables such as volume of dead wood and number of dead trees [89]. In contrast, less dense forests provide a less suitable habitat for saproxylic beetles for several reasons. First, losses of arboreal density are the result of the consumption of dead wood [7], including fine and aerial biomass that eliminate a substantial portion of habitat for saproxylic species. The combustion of biomass in severe wildfires also mostly affects Nothofagus sp. trees (see Supplementary Material S1) because these species are less resistant (e.g., thin bark) to fire [54]. Therefore, the specialist species associated with Nothofagus sp. could be severely affected compared with species associated with Araucaria. Second, losses of arboreal density are related to reduction of canopy cover that could drastically dry microclimatic conditions on the ground, exposing saproxylic species to unfavorable conditions. Finally, the carbonization of dead wood is remarkable in SISF areas, where most reburned and dead trees were bark-free, resulting in a major carbonization that could inhibit the presence of many species [90]. For instance, the species belonging to the genera Araucarius, Hylurgonotus, and Sinophloeus are mostly xylophagous in sub-cortical trunks and/or branches of Araucaria trees [44]; therefore, these species could have been excluded from bark-free trees. On the other hand, at a tree level, the advanced decomposition stages induced a bimodal response in assemblages of saproxylic beetles in recently burned Araucaria–Nothofagus forests, resulting in a benefit for some groups while damage for others. The quality of the habitat substratum in terms of decayed wood and species type are important to many beetle species [41,91,92], but also the presence of tree bark for early stages of saproxylic turnover [50]. In this way, decay stage is a key attribute of dead wood in native forests [91,93]. Thus, we concluded that in severely reburned forests (SISF), habitat amount and quality reduced the persistence for saproxylic beetles, as subsequent fire consumed a large portion of dead wood habitat, including most of the fine woody debris on the ground.
4.2. Saproxylic Diversity Pattern Induced by Burned Conditions
At the community level, the diversity of saproxylic beetles was affected mostly by the arboreal density and decaying stages (Table 3, Figure 2 and Figure 6A). Thus, in old burned (2002) and unburned sites, the diversity was consistent with post-fire advance stage (circa two decades after fire occurrence) where the decomposition of dead wood and higher arboreal density permits the persistence of a high number of species and abundances (fungivorous and detritivores in high-decay dead wood [50]). Moreover, no statistically significant difference of diversity was found between recently burned assemblages and SISF. This result was unexpected, because SISF was assumed to reduce the habitat habitability to saproxylic species, which should have reduced beetle diversity. However, the assemblage composition analysis showed different beetle communities (Figure 3), and β-diversity patterns indicated high turnover of species with very low abundances of some trophic guilds. These patterns in SISF and recently burned forests (i.e., equal species richness but different species compositions) could be explained by compensatory dynamics that allow for apparently stable community-level properties (i.e., species richness), but unstable species-level properties (i.e., composition) [94]. Thus, we suggest that in advanced successional stages, the 2015-burned forests could permit the persistence of more species due to the high amount of habitat (e.g., arboreal density) compared with SISF areas. Additionally, the partition of β-diversity (βSIM + βNES) indicated that changes in assemblage composition was principally due to the increase of turnover in all conditions, except when we compared unburned and 2015 assemblages, where nestedness component was slightly higher (Figure 4).
These results are consistent with the effect described in many different post-disturbance assemblages, particularly in post-fire plant [95] and animal community responses [82]. Moreover, SISF increased the contribution of turnover components of β-diversity with respect to unburned and other recently burned forests (Figure 4; ~94% and ~6% for turnover and nestedness, respectively). Turnover is caused by the replacement of species from one plot to another, and the nestedness-related component is determined by species gains or losses and can arise from processes such as selective extinction or colonization [81]. Hence, the higher contribution of turnover to SISF indicates that these assemblages are species-poor plots, and not a subset of assemblages of species-rich plots (unburned forests) [81,83]. Thus, we concluded that in SISF forests, the potential process underlying the drastic increase in species turnover is due to sustaining distinct communities compared with other burned and unburned conditions. The habitat transformations induced by SISF (i.e., dense forests into less dense forests or shrublands [28,96]) indicate that the distinct and poor species pool observed in SISF areas can be the result of habitat transitions of dense forest into shrubs stages [97,98]. However, the ecological consequences of this change in species composition are still unknown, and a functional approach is needed. Additionally, long-term monitoring is required to elucidate if the habitat transition is an ephemeral condition that will be modified by the recolonization of the forest, or the shrub cover (stratum) will be a permanent condition, preventing the re-establishment of the pool of species that are pre-existing to the SISF.
4.3. Trophic Guild Responses to Burned Conditions
Trophic guild indicates the position of an organism in the food web, and the abundance of some trophic guilds are determined by the quality of resources at the habitat level, which influences individual fitness in saproxylic beetles species [99]. Therefore, burned conditions result in changes in abundances and species richness of many saproxylic groups due to alterations in the quality and quantity of dead wood [21,41,100]. In our study, the most abundant trophic guild was the xylophagous (Figure 5), which were especially abundant in recently burned forests (2015: 4515 individuals). In the case of recently burned forests, our results are consistent with similar studies, where the xylophagous group and sub-cortical species rapidly start the colonization in early stages of succession [50,101]. Additionally, the diversity of this group decreased on these recently dead trees, and the higher number of species was related to highly decaying trees (Figure 6B,C).
We observed contrasting performances of abundances and diversity induced by the 2015-burned condition; while the abundances were higher, the number of species was lower in these conditions. This suggests that in the 2015 assemblage, few species with high abundances dominated, resulting in the reduction of the evenness of these communities [102]. Moreover, the abundances of the xylophagous group were markedly lower in SISF regarding 2015-burned areas, and no differences with unburned areas were detected (Table 2). These results indicate that the SISF reduces accessibility to these early colonizers due to losses of habitat availability (i.e., losses of arboreal density and bark cover) and high carbonization of the dead wood [90]. Moreover, no “fire-beetle” species has been reported in Araucaria–Nothofagus forests, resulting in a less fire-adapted assemblage; however, we do not dismiss the possibility that some beetle species (e.g., Araucarius spp. and Hylurgonotus spp.) have evolved under a selective fire pressure.
On the other hand, the higher abundances and diversity of mycophagous in unburned and old-burned forests is due to highly decayed trees (Figure 6B,C), because this habitat condition supports high abundances of fungus that support the feeding and reproduction of immature and adults of saproxylic beetles, respectively [103,104,105]. Additionally, this group has shown a response to habitat changes in Chilean forests, such as clear-cutting practices [106] or wildfires [35]. Collectively, these results reinforce the pivotal role of dead wood’s decay stage in structuring insect communities in native forests [91,93]. Finally, the not-significant differences in abundances and number of species of zoophagous group, and the effects of habitat conditions on this group, could be explained by the increases in the xylophagous species that serve as food resource for predator beetles. Thus, in the presence of burned conditions, the zoophagous species use an alternative food resource, stabilizing the trophic dynamics.
4.4. Implication of SISF for the Diversity and Conservation of Saproxylic Beetles and Forest Management
Management practices are crucial to promote more diverse and healthier communities in severely disturbed ecosystems. For example, salvage logging (i.e., removing dead or dying trees after a forest disturbance) is the most common practice to maintain a minimum threshold of diversity and to decrease the economic losses induced by wildfires [39,107]. Nonetheless, these practices have been linked to decreases of diversity (e.g., species richness) due to reduction of habitat heterogeneity [108,109]. On the other hand, unmanaged post-fire forests also represent a risk of SISF occurrence due to the massive accumulation of dead wood that serves as fuel [8]. Therefore, the election of adequate practices for these post-fire forests is a complex issue that must be based on multiple criteria, including at least the creation of habitat qualities to promote highly diverse communities and prevent the occurrence of new fire events. Our main results suggest that a satisfactory management practice should be oriented to emulate the dead wood quality of the unburned forests, maintaining higher decomposed trees and promoting denser forests. Thus, replacing recently burned (i.e., most carbonized) dead trees with highly decayed and unburned trees, and repopulating with new living native trees, is needed to improve post-fire habitat conditions. Surplus of large-sized dead and burned trees could be used for complementary management practices such as log erosion barriers on steep slopes [110]. In this way, this method improves the quality of dead wood, and avoids the losses of arboreal density and dead wood volume related to traditional salvage logging practices.
5. Conclusions
This work provides novel evidence of the negative effects of short-interval, severe wildfires on forests that are not well-adapted to resist these disturbances. In addition, we provide new insights into the fire community ecology of northern Patagonian Araucaria–Nothofagus forest. The mains findings of this study show that fire conditions induced differentiated patterns in alpha (i.e., species richness and abundances) and beta diversity (i.e., patterns of nestedness/turnover), and changes in the assemblage composition of the saproxylic beetles. The saproxylic beetles were significantly less abundant and diverse in SISF areas, particularly xylophagous species, while the most diverse assemblages were observed in unburned areas. Overall, the patterns of high abundance and low species richness in burned forests were consistent with habitat conditions, where high arboreal density and advanced decaying stages supported the persistence of high diversity in these assemblages. In contrast, losses of arboreal density resulted in a substantially lower diversity and abundances, and the early decaying stages of dead wood promoted higher abundances of the saproxylic beetles but less diverse assemblages.
We noted that the successional process of saproxylic beetles was abruptly disturbed by the reburn. Thus, in old burned areas (2002), the assemblages tend to be more similar in composition compared with unburned areas, while SISF areas were more dissimilar. These data also indicate that the poor habitat conditions resulting from short-interval high-severity wildfires induce an alternative stage in saproxylic beetle succession. We suggest that alternative stages could be the consequence of overcoming the resilience of the community through the elimination and turnover of species. Finally, to avoid the negative long-term effects of SISF, we propose a novel management practice based on replacing the most severely burned trees with habitat-rich trees (decayed) for saproxylic beetles.
There are numerous future directions for this work. For instance, long-term research is needed to evaluate the “successional direction” of these alternative communities; at species scale, we need to establish the mechanisms that underpin the fitness responses in burned conditions; at community scale, we need to evaluate the functional trait responses to fire in order to gauge the vulnerability of these assemblages. This research agenda could improve the fundamental knowledge needed to establish strategies to mitigate the impact of these severe fire events, which are likely to recur as a result of climate change.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/f13030441/s1, Supplementary Material S1: Supplementary data of dead wood volume recorded in study areas, Supplementary Material S2: Systematic list of the taxa recorded in this study.
Author Contributions
Conceptualization, F.T. (Francisco Tello); Methodology, F.T. (Francisco Tello), M.E.G., E.M., A.G.-L. and N.V.; Fieldwork, F.T. (Francisco Tello) and F.T. (Fernanda Torres); Validation, F.T. (Francisco Tello), M.E.G., E.M. and N.V.; Formal analysis, F.T. (Francisco Tello), N.V. and A.G.-L.; Data curation, F.T. (Francisco Tello) and A.G.-L.; Writing—original draft preparation, F.T. (Francisco Tello), N.V., A.G.-L., A.L., E.M., F.T. (Fernanda Torres) and M.E.G.; Visualization, F.T. (Francisco Tello) and F.T. (Fernanda Torres); Supervision, M.E.G., N.V., A.G.-L., A.L. and E.M.; Funding acquisition, F.T. (Francisco Tello), M.E.G. and A.L. All authors have read and agreed to the published version of the manuscript.
Funding
F.T. (Francisco Tello) was financially supported by CONICYT doctoral scholarship no. 21171980. F.T. (Francisco Tello), M.E.G., and A.L. were financially supported by ANID/FONDAP center no. 15110009 (CR2). M.E.G. was financially supported by FONDECYT grant no. 1201528. N.V. was financially supported by FONDECYT grants no. 1190529, no. 1181300, and FONDAP center no. 15150003 (IDEAL).
Institutional Review Board Statement
This study was authorized by the Department of Protected Wildlife Areas, CONAF, Chile (ID = 593919).
Informed Consent Statement
Not applicable.
Data Availability Statement
Additional information will be provided by the Corresponding Author by email.
Acknowledgments
We would like to express sincere thanks to Mario Elgueta, Alfredo Lüer, and Gerardo Arriagada for his taxonomical assistance. We thank Francisco Beels, Javier Aravena, Emma Gleeman, and Julio Fritz for field assistance. Additionally, we thank the CONAF rangers, especially Alvaro Marín, for fieldwork support. Finally, we thank the Scientific Editor and anonymous reviewers that provided helpful recommendations and comments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Westerling, A.L. Warming and Earlier Spring Increase Western U.S. Forest Wildfire Activity. Science 2006, 313, 940–943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jolly, W.M.; Cochrane, M.A.; Freeborn, P.H.; Holden, Z.A.; Brown, T.J.; Williamson, G.J.; Bowman, D.M.J.S. Climate-Induced Variations in Global Wildfire Danger from 1979 to 2013. Nat. Commun. 2015, 6, 7537. [Google Scholar] [CrossRef] [PubMed]
- Littell, J.S.; Peterson, D.L.; Riley, K.L.; Liu, Y.; Luce, C.H. A Review of the Relationships between Drought and Forest Fire in the United States. Glob. Chang. Biol. 2016, 22, 2353–2369. [Google Scholar] [CrossRef]
- Turco, M.; Von Hardenberg, J.; AghaKouchak, A.; Llasat, M.C.; Provenzale, A.; Trigo, R.M. On the Key Role of Droughts in the Dynamics of Summer Fires in Mediterranean Europe. Sci. Rep. 2017, 7, 81. [Google Scholar] [CrossRef] [Green Version]
- Dennison, P.E.; Brewer, S.C.; Arnold, J.D.; Moritz, M.A. Large Wildfire Trends in the Western United States, 1984–2011. Geophys. Res. Lett. 2014, 41, 2928–2933. [Google Scholar] [CrossRef]
- He, T.; Lamont, B.B.; Pausas, J.G. Fire as a Key Driver of Earth’s Biodiversity. Biol. Rev. 2019, 94, 1983–2010. [Google Scholar] [CrossRef]
- Donato, D.C.; Fontaine, J.B.; Campbell, J.L. Burning the Legacy? Influence of Wildfire Reburn on Dead Wood Dynamics in a Temperate Conifer Forest. Ecosphere 2016, 7, e01341. [Google Scholar] [CrossRef] [Green Version]
- Harvey, B.J.; Donato, D.C.; Turner, M.G. Burn Me Twice, Shame on Who? Interactions between Successive Forest Fires across a Temperate Mountain Region. Ecology 2016, 97, 2272–2282. [Google Scholar] [CrossRef] [PubMed]
- Paine, R.T.; Tegner, M.J.; Johnson, E.A. Compounded Perturbations Yield Ecological Surprises. Ecosystems 1998, 1, 535–545. [Google Scholar] [CrossRef]
- Turner, M.G. Disturbance and Landscape Dynamics in a Changing World. Ecology 2010, 91, 2833–2849. [Google Scholar] [CrossRef] [Green Version]
- Millar, C.I.; Stephenson, N.L. Temperate Forest Health in an Era of Emerging Megadisturbance. Science 2015, 349, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.G.; Braziunas, K.H.; Hansen, W.D.; Harvey, B.J. Short-Interval Severe Fire Erodes the Resilience of Subalpine Lodgepole Pine Forests. Proc. Natl. Acad. Sci. USA 2019, 116, 11319–11328. [Google Scholar] [CrossRef] [Green Version]
- Bowman, D.M.J.S.; Balch, J.K.; Artaxo, P.; Bond, W.J.; Carlson, J.M.; Cochrane, M.A.; D’Antonio, C.M.; Defries, R.S.; Doyle, J.C.; Harrison, S.P.; et al. Fire in the Earth System. Science 2009, 324, 481–484. [Google Scholar] [CrossRef]
- Donato, D.C.; Fontaine, J.B.; Robinson, W.D.; Kauffman, J.B.; Law, B.E. Vegetation Response to a Short Interval between High-Severity Wildfires in a Mixed-Evergreen Forest. J. Ecol. 2009, 97, 142–154. [Google Scholar] [CrossRef] [Green Version]
- Fairman, T.A.; Bennett, L.T.; Nitschke, C.R. Short-Interval Wildfires Increase Likelihood of Resprouting Failure in Fire-Tolerant Trees. J. Environ. Manag. 2019, 231, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Meng, R.; Dennison, P.E.; D’Antonio, C.M.; Moritz, M.A. Remote Sensing Analysis of Vegetation Recovery Following Short-Interval Fires in Southern California Shrublands. PLoS ONE 2014, 9, e110637. [Google Scholar] [CrossRef] [Green Version]
- Beale, C.M.; Courtney Mustaphi, C.J.; Morrison, T.A.; Archibald, S.; Anderson, T.M.; Dobson, A.P.; Donaldson, J.E.; Hempson, G.P.; Probert, J.; Parr, C.L. Pyrodiversity Interacts with Rainfall to Increase Bird and Mammal Richness in African Savannas. Ecol. Lett. 2018, 21, 557–567. [Google Scholar] [CrossRef] [Green Version]
- Carbone, L.M.; Tavella, J.; Pausas, J.G.; Aguilar, R. A Global Synthesis of Fire Effects on Pollinators. Glob. Ecol. Biogeogr. 2019, 28, 1487–1498. [Google Scholar] [CrossRef]
- Griffiths, A.D.; Garnett, S.T.; Brook, B.W. Fire Frequency Matters More than Fire Size: Testing the Pyrodiversity-Biodiversity Paradigm for at-Risk Small Mammals in an Australian Tropical Savanna. Biol. Conserv. 2015, 186, 337–346. [Google Scholar] [CrossRef]
- Fontaine, J.B.; Donato, D.C.; Robinson, W.D.; Law, B.E.; Kauffman, J.B. Bird Communities Following High-Severity Fire: Response to Single and Repeat Fires in a Mixed-Evergreen Forest, Oregon, USA. For. Ecol. Manag. 2009, 257, 1496–1504. [Google Scholar] [CrossRef] [Green Version]
- Moretti, M.; Obrist, M.K.; Duelli, P. Arthropod Biodiversity after Forest Fires: Winners and Losers in the Winter Fire Regime of the Southern Alps. Ecography 2004, 27, 173–186. [Google Scholar] [CrossRef]
- Coop, J.D.; Parks, S.A.; Mcclernan, S.R.; Holsinger, L.M. Influences of Prior Wildfires on Vegetation Response to Subsequent Fire in a Reburned Southwestern Landscape. Ecol. Appl. 2016, 26, 346–354. [Google Scholar] [CrossRef]
- Paritsis, J.; Veblen, T.T.; Holz, A. Positive Fire Feedbacks Contribute to Shifts from Nothofagus pumilio Forests to Fire-Prone Shrublands in Patagonia. J. Veg. Sci. 2015, 26, 89–101. [Google Scholar] [CrossRef]
- Cochrane, M.A.; Alencar, A.; Schulze, M.D.; Souza, C.M.; Nepstad, D.C.; Lefebvre, P.; Davidson, E.A. Positive Feedbacks in the Fire Dynamic of Closed Canopy Tropical Forests. Science 1999, 284, 1832–1835. [Google Scholar] [CrossRef]
- Fletcher, M.-S.; Wood, S.W.; Haberle, S.G. A Fire-Driven Shift from Forest to Non-Forest: Evidence for Alternative Stable States? Ecology 2014, 95, 2504–2513. [Google Scholar] [CrossRef] [Green Version]
- Kitzberger, T.; Perry, G.; Paritsis, J.; Gowda, J.; Tepley, A.; Holz, A.; Veblen, T. Fire–Vegetation Feedbacks and Alternative States: Common Mechanisms of Temperate Forest Vulnerability to Fire in Southern South America and New Zealand. N. Z. J. Bot. 2016, 54, 247–272. [Google Scholar] [CrossRef] [Green Version]
- Steel, Z.L.; Foster, D.; Coppoletta, M.; Lydersen, J.M.; Stephens, S.L.; Paudel, A.; Markwith, S.H.; Merriam, K.; Collins, B.M. Ecological Resilience and Vegetation Transition in the Face of Two Successive Large Wildfires. J. Ecol. 2021, 109, 3340–3355. [Google Scholar] [CrossRef]
- Fairman, T.A.; Nitschke, C.R.; Bennett, L.T. Too Much, Too Soon? A Review of the Effects of Increasing Wildfire Frequency on Tree Mortality and Regeneration in Temperate Eucalypt Forests. Int. J. Wildl. Fire 2016, 25, 831. [Google Scholar] [CrossRef]
- Fidelis, A.; Alvarado, S.; Barradas, A.; Pivello, V. The Year 2017: Megafires and Management in the Cerrado. Fire 2018, 1, 49. [Google Scholar] [CrossRef] [Green Version]
- McWethy, D.B.; Pauchard, A.; García, R.A.; Holz, A.; González, M.E.; Veblen, T.T.; Stahl, J.; Currey, B. Landscape Drivers of Recent Fire Activity (2001–2017) in South-Central Chile. PLoS ONE 2018, 13, e0201195. [Google Scholar] [CrossRef]
- Moreno-Gonzalez, R.; Giesecke, T.; Fontana, S.L. The Impact of Recent Land-Use Change in the Araucaria araucana Forest in Northern Patagonia. Holocene 2020, 30, 1101–1114. [Google Scholar] [CrossRef]
- Premoli, A.; Quiroga, P.; Gardner, M. Araucaria araucana: Monkey Puzzle Tree. IUCN Red List. Threat. Species 2013, 8235, 1–2. [Google Scholar] [CrossRef]
- Luebert, F.; Pliscoff, P. Sinopsis Bioclimática y Vegetacional de Chile, 3rd ed.; Editorial Universitaria: Santiago, Chile, 2019; ISBN 978-956-11-2651-0. [Google Scholar]
- Speight, M.C.D. Saproxylic Invertebrates and Their Conservation. Nat. Environ. 1989, 42, 1–79. [Google Scholar]
- Tello, F.; González, M.E.; Valdivia, N.; Torres, F.; Lara, A.; García-López, A. Diversity Loss and Changes in Saproxylic Beetle Assemblages Following a High-Severity Fire in Araucaria–Nothofagus Forests. J. Insect Conserv. 2020, 24, 585–601. [Google Scholar] [CrossRef]
- Lindhe, A.; Lindelöw, Å.; Åsenblad, N. Saproxylic Beetles in Standing Dead Wood Density in Relation to Substrate Sun-Exposure and Diameter. Biodivers. Conserv. 2005, 14, 3033–3053. [Google Scholar] [CrossRef]
- Djupström, L.B.; Weslien, J.; Ten Hoopen, J.; Schroeder, L.M. Restoration of Habitats for a Threatened Saproxylic Beetle Species in a Boreal Landscape by Retaining Dead Wood on Clear-Cuts. Biol. Conserv. 2012, 155, 44–49. [Google Scholar] [CrossRef]
- Fierro, A.; Vergara, P.M. A Native Long Horned Beetle Promotes the Saproxylic Diversity in Exotic Plantations of Monterrey Pine. Ecol. Indic. 2019, 96, 532–539. [Google Scholar] [CrossRef]
- Müller, J.; Bütler, R. A Review of Habitat Thresholds for Dead Wood: A Baseline for Management Recommendations in European Forests. Eur. J. For. Res. 2010, 129, 981–992. [Google Scholar] [CrossRef]
- Boulanger, Y.; Sirois, L.; Hébert, C. Fire Severity as a Determinant Factor of the Decomposition Rate of Fire-Killed Black Spruce in the Northern Boreal Forest. Can. J. For. Res. 2011, 41, 370–379. [Google Scholar] [CrossRef]
- Hjältén, J.; Dynesius, M.; Hekkala, A.; Karlsson-Tiselius, A.; Löfroth, T.; Mugerwa-Pettersson, R. Saproxylic Insects and Fire. In Zoological Monographs; Ulyshen, M., Ed.; Springer: Cham, Switzerland, 2018; pp. 669–691. ISBN 9783319759371. [Google Scholar]
- Moretti, M.; De Cáceres, M.; Pradella, C.; Obrist, M.K.; Wermelinger, B.; Legendre, P.; Duelli, P. Fire-Induced Taxonomic and Functional Changes in Saproxylic Beetle Communities in Fire Sensitive Regions. Ecography 2010, 33, 760–771. [Google Scholar] [CrossRef]
- Cobb, T.P.; Hannam, K.D.; Kishchuk, B.E.; Langor, D.W.; Quideau, S.A.; Spence, J.R. Wood-Feeding Beetles and Soil Nutrient Cycling in Burned Forests: Implications of Post-Fire Salvage Logging. Agric. For. Entomol. 2010, 12, 9–18. [Google Scholar] [CrossRef]
- Kuschel, G. La Fauna Curculiónica (Coleoptera: Curculionoidae) de La Araucaria araucana. Rev. Chil. Entomol. 2000, 27, 41–51. [Google Scholar]
- Rühm, V.W. Riisselkafer (Araucariini, Cossoninae, Col.) Mit Einer an Der Araucaria araucana (Mol.) Koch in Chile. Zool. Staatsinst. Mus. Univ. Hambg. 1977, 84, 283–295. [Google Scholar]
- Paulsen, M.J.; Smith, A. Revision of the Genus Chiasognathus Stephens of Southern South America with the Description of a New Species (Coleoptera, Lucanidae, Lucaninae, Chiasognathini). Zookeys 2010, 43, 33–63. [Google Scholar] [CrossRef]
- Paulsen, M.J. The Stag Beetles of Southern South America (Coleoptera: Lucanidae). Bull. Univ. Neb. State Mus. 2010, 24, 148. [Google Scholar]
- Arias, E.T.; Richardson, B.J.; Elgueta, M. The Canopy Beetle Faunas of Gondwanan Element Trees in Chilean Temperate Rain Forests. J. Biogeogr. 2008, 35, 914–925. [Google Scholar] [CrossRef]
- Tello, F. New Record of Hilophyllus martinezi Paulsen and Mondaca, 2006 (Coleoptera: Lucanidae: Aesalinae) in Southern Chile. Coleopt. Bull. 2020, 74, 424. [Google Scholar] [CrossRef]
- Boulanger, Y.; Sirois, L. Postfire Succession of Saproxylic Arthropods, with Emphasis on Coleoptera, in the North Boreal Forest of Quebec. Environ. Entomol. 2007, 36, 128–141. [Google Scholar] [CrossRef]
- Fuentes-Ramírez, A.; Arroyo-Vargas, P.; Del Fierro, A.; Pérez, F. Post-Fire Response of Araucaria araucana (Molina) K. Koch: Assessment of Vegetative Resprouting, Seed Production and Germination. Gayana Bot. 2019, 76, 119–122. [Google Scholar] [CrossRef] [Green Version]
- González, M.E.; Lara, A. Large Fires in the Andean Araucaria Forests: When a Natural Ecological Process Becomes a Threat. Oryx 2015, 49, 394. [Google Scholar] [CrossRef] [Green Version]
- Veblen, T.; Kitzberger, T.; Villalba, R. Nuevos paradigmas en ecología y su influencia sobre el conocimiento de la dinámica de los bosques del sur de Argentina y Chile. In Ecología y Manejo de Bosques de Argentina; Arturi, M., Frangi, J., Goya, J., Eds.; Editorial de la Universidad Nacional de La Plata: La Plata, Argentina, 2004; pp. 1–48. [Google Scholar]
- González, M.E.; Muñoz, A.A.; González-Reyes, Á.; Christie, D.A.; Sibold, J. Fire History in Andean Araucaria–Nothofagus Forests: Coupled Influences of Past Human Land-Use and Climate on Fire Regimes in North-West Patagonia. Int. J. Wildl. Fire 2020, 29, 649. [Google Scholar] [CrossRef]
- Zamorano-Elgueta, C.; Cayuela, L.; González-Espinosa, M.; Lara, A.; Parra-Vázquez, M.R. Impacts of Cattle on the South American Temperate Forests: Challenges for the Conservation of the Endangered Monkey Puzzle Tree (Araucaria araucana) in Chile. Biol. Conserv. 2012, 152, 110–118. [Google Scholar] [CrossRef]
- González, M.E.; Veblen, T.T.; Sibold, J.S. Fire History of Araucaria-Nothofagus Forests in Villarrica National Park, Chile. J. Biogeogr. 2005, 32, 1187–1202. [Google Scholar] [CrossRef]
- Mundo, I.A.; Kitzberger, T.; Roig Juñent, F.A.; Villalba, R.; Barrera, M.D. Fire History in the Araucaria araucana Forests of Argentina: Human and Climate Influences. Int. J. Wildl. Fire 2013, 22, 194–206. [Google Scholar] [CrossRef] [Green Version]
- Assal, T.J.; González, M.E.; Sibold, J.S. Burn Severity Controls on Postfire Araucaria-Nothofagus Regeneration in the Andean Cordillera. J. Biogeogr. 2018, 45, 2483–2494. [Google Scholar] [CrossRef] [Green Version]
- Urrutia-Estrada, J.; Fuentes-Ramírez, A.; Hauenstein, E. Diferencias En La Composición Florística En Bosques de Araucaria-Nothofagus Afectados Por Distintas Severidades de Fuego. Gayana Bot. 2018, 75, 625–638. [Google Scholar] [CrossRef] [Green Version]
- González, M.E.; Veblen, T.T.; Sibold, J.S. Influence of Fire Severity on Stand Development of Araucaria araucana-Nothofagus pumilio Stands in the Andean Cordillera of South-Central Chile. Austral Ecol. 2010, 35, 597–615. [Google Scholar] [CrossRef]
- Sverdrup-Thygeson, A.; Birkemoe, T. What Window Traps Can Tell Us: Effect of Placement, Forest Openness and Beetle Reproduction in Retention Trees. J. Insect Conserv. 2009, 13, 183–191. [Google Scholar] [CrossRef]
- Quinto, J.; Marcos-García, M.d.l.Á.; Brustel, H.; Galante, E.; Micó, E. Effectiveness of Three Sampling Methods to Survey Saproxylic Beetle Assemblages in Mediterranean Woodland. J. Insect Conserv. 2013, 17, 765–776. [Google Scholar] [CrossRef]
- Bouget, C.; Brustel, H.; Brin, A.; Noblecourt, T. Sampling Saproxylic Beetles with Window Flight Traps: Methodological Insights. Rev. D’ecologie Terre Vie 2008, 63, 21–32. [Google Scholar]
- García-López, A.; Galante, E.; Micó, E. Saproxylic Beetle Assemblage Selection as Determining Factor of Species Distributional Patterns: Implications for Conservation. J. Insect Sci. 2016, 16, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martikainen, P.; Kaila, L. Sampling Saproxylic Beetles: Lessons from a 10-Year Monitoring Study. Biol. Conserv. 2004, 120, 175–185. [Google Scholar] [CrossRef]
- Wood, S. Bark and Ambrosia Beetles of South America (Coleoptera, Scolytidae); Monte, L., Ed.; Bean Life Science Museum, Brigham Young University: Provo, UT, USA, 2007; ISBN 0-8425-2635-8. [Google Scholar]
- Elgueta, M. Estado Actual Del Conocimiento de los Coleópteros de Chile (Insecta: Coleoptera). PrIBES-2000 Proy. Para Iberoam. Entomol. Sist. 2000, 17, 145–154. [Google Scholar]
- Barriga, J.E.; Curkovic, T.; Fichet, T.; Henríquez, J.; Macaya, J. Nuevos Antecedentes de Coleopteros Xilófagos y Plantas Hospederas En Chile, Con Una Recopilación de Citas Previas. Rev. Chil. Entomol. 1993, 20, 65–91. [Google Scholar]
- Solervicens, J. Coleópteros de La Reserva Nacional Río Clarillo, en Chile Central: Taxonomía, Biología y Biogeografía; Corporación Nacional Forestal: Santiago, Chile, 2014; ISBN 9567669503. [Google Scholar]
- Ruta, R. Three New Genera of Large Marsh Beetles (Coleoptera: Scirtidae) from Valdivian Temperate Rain Forests of Southern South America. Zootaxa 2021, 5048, 451–485. [Google Scholar] [CrossRef] [PubMed]
- Lüer, A. Lista de Plantas Hospedantes de Ptinidae (Coleoptera: Bostrichoidea) de Chile. Rev. Chil. Entomol. 2020, 46, 333–344. [Google Scholar] [CrossRef]
- Sandoval, A. Artrópodos Asociados a Araucaria araucana (Molina) K. Koch en Chile; Ministerio de Agricultura, Servicio Agrícola y Ganadero: Santiago, Chile, 2017; ISBN 978-956-7987-19-1. [Google Scholar]
- Hammond, H.E.J.; Langor, D.W.; Spence, J.R. Changes in Saproxylic Beetle (Insecta: Coleoptera) Assemblages Following Wildfire and Harvest in Boreal Populus Forests. For. Ecol. Manag. 2017, 401, 319–329. [Google Scholar] [CrossRef]
- Müller, J.; Bußler, H.; Kneib, T. Saproxylic Beetle Assemblages Related to Silvicultural Management Intensity and Stand Structures in a Beech Forest in Southern Germany. J. Insect Conserv. 2008, 12, 107–124. [Google Scholar] [CrossRef]
- Carmona, M.; Armesto, J.; Aravena, J.; Cecilia, P. Coarse Woody Debris Biomass in Successional and Primary Island, Chile Temperate Forests in Chiloe. For. Ecol. Manag. 2002, 164, 265–275. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and Extrapolation with Hill Numbers: A Framework for Sampling and Estimation in Species Diversity Studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, T.C.; Ma, K.H.; Chao, A. INEXT: An R Package for Rarefaction and Extrapolation of Species Diversity (Hill Numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Colwell, R.K. Quantifying Biodiversity: Procedures and Pitfalls in the Measurment and Comparison of Species Richness. Ecol. Lett. 2001, 4, 379–391. [Google Scholar] [CrossRef] [Green Version]
- Anderson, M.J. A New Method for Non-Parametric Multivariate Analysis of Variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Whittaker, R.H. Evolution and Measurement of Species Diversity. Taxon 1972, 21, 213–251. [Google Scholar] [CrossRef] [Green Version]
- Baselga, A. Partitioning the Turnover and Nestedness Components of Beta Diversity. Glob. Ecol. Biogeogr. 2010, 19, 134–143. [Google Scholar] [CrossRef]
- Azeria, E.; Barzabal, J.; Boucher, J.; Hébert, C. Differential Effects of Post-Fire Habitat Legacies on Beta Diversity Patterns of Saproxylic Beetles in the Boreal Forest. Écosciencie 2012, 19, 316–327. [Google Scholar] [CrossRef]
- Baselga, A.; Orme, C.D.L. Betapart: An R Package for the Study of Beta Diversity. Methods Ecol. Evol. 2012, 3, 808–812. [Google Scholar] [CrossRef]
- Burnham, K.; Anderson, D. Model Selection and Multimodel Inference, 2nd ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95364-7. [Google Scholar]
- Barbosa, A.M.; Real, R.; Muñoz, A.R.; Brown, J.A. New Measures for Assessing Model Equilibrium and Prediction Mismatch in Species Distribution Models. Divers. Distrib. 2013, 19, 1333–1338. [Google Scholar] [CrossRef]
- Barbosa, A.M.; Brown, J.A.; Jimenez-Valverde, A.; Real, R. ModEvA: Model Evaluation and Analysis 2016, R Package Version 1.3.2. Available online: https://CRAN.R-project.org/package=modEvA (accessed on 4 January 2020).
- McFadden, D. Quantitative Methods for Analyzing Travel Behaviour of Individuals: Some Recent Developments. In Behavioural Travel Modelling; Hensher, D., Stopher, P., Eds.; Croom Helm: London, UK, 1978; pp. 279–318. [Google Scholar]
- Lüdecke, D. SjPlot: Data Visualization for Statistics in Social Science 2018, R Package Version 2.8.10.1. Available online: https://CRAN.R-project.org/package=sjPlot (accessed on 4 January 2020).
- Seibold, S.; Bässler, C.; Brandl, R.; Fahrig, L.; Förster, B.; Heurich, M.; Hothorn, T.; Scheipl, F.; Thorn, S.; Müller, J. An Experimental Test of the Habitat-Amount Hypothesis for Saproxylic Beetles in a Forested Region. Ecology 2017, 98, 1613–1622. [Google Scholar] [CrossRef]
- Nappi, A.; Drapeau, P.; Saint-Germain, M.; Angers, V.A. Effect of Fire Severity on Long-Term Occupancy of Burned Boreal Conifer Forests by Saproxylic Insects and Wood-Foraging Birds. Int. J. Wildl. Fire 2010, 19, 500–511. [Google Scholar] [CrossRef]
- Fierro, A.; Grez, A.A.; Vergara, P.M.; Ramírez-Hernández, A.; Micó, E. How Does the Replacement of Native Forest by Exotic Forest Plantations Affect the Diversity, Abundance and Trophic Structure of Saproxylic Beetle Assemblages? For. Ecol. Manag. 2017, 405, 246–256. [Google Scholar] [CrossRef]
- Vanderwel, M.C.; Malcolm, J.R.; Smith, S.M.; Islam, N. Insect Community Composition and Trophic Guild Structure in Decaying Logs from Eastern Canadian Pine-Dominated Forests. For. Ecol. Manag. 2006, 225, 190–199. [Google Scholar] [CrossRef]
- Stokland, J.N.; Siitonen, J.; Jonsson, B.G. Biodiversity in Dead Wood; Cambridge University Press: Cambridge, UK, 2012; ISBN 9781139025843. [Google Scholar]
- Gonzalez, A.; Loreau, M. The Causes and Consequences of Compensatory Dynamics in Ecological Communities. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 393–414. [Google Scholar] [CrossRef] [Green Version]
- Heydari, M.; Omidipour, R.; Abedi, M.; Baskin, C. Effects of Fire Disturbance on Alpha and Beta Diversity and on Beta Diversity Components of Soil Seed Banks and Aboveground Vegetation. Plant Ecol. Evol. 2017, 150, 247–256. [Google Scholar] [CrossRef]
- Pausas, J.G. Alternative Fire-Driven Vegetation States. J. Veg. Sci. 2015, 26, 4–6. [Google Scholar] [CrossRef] [Green Version]
- Azeria, E.T.; Ibarzabal, J.; Boucher, J.; Hebert, C. Towards a Better Understanding of Beta Diversity: Deconstructing Composition Patterns of Saproxylic Beetles Breeding in Recently Burnt Boreal Forest. In Research in Biodiversity-Models and Applications; InTech Open: London, UK, 2011; pp. 75–94. [Google Scholar]
- Chase, J.M.; Kraft, N.J.B.; Smith, K.G.; Vellend, M.; Inouye, B.D. Using Null Models to Disentangle Variation in Community Dissimilarity from Variation in α-Diversity. Ecosphere 2011, 2, 1–11. [Google Scholar] [CrossRef]
- Moretti, M.; Dias, A.T.C.; de Bello, F.; Altermatt, F.; Chown, S.L.; Azcárate, F.M.; Bell, J.R.; Fournier, B.; Hedde, M.; Hortal, J.; et al. Handbook of Protocols for Standardized Measurement of Terrestrial Invertebrate Functional Traits. Funct. Ecol. 2017, 31, 558–567. [Google Scholar] [CrossRef]
- Nappi, A.; Drapeau, P. Pre-Fire Forest Conditions and Fire Severity as Determinants of the Quality of Burned Forests for Deadwood-Dependent Species: The Case of the Black-Backed Woodpecker. Can. J. For. Res. 2011, 1, 994–1003. [Google Scholar] [CrossRef]
- Wikars, L.O. Dependence on Fire in Wood-Living Insects: An Experiment with Burned and Unburned Spruce and Birch Logs. J. Insect Conserv. 2002, 6, 1–12. [Google Scholar] [CrossRef]
- Avolio, M.L.; Carroll, I.T.; Collins, S.L.; Houseman, G.R.; Hallett, L.M.; Isbell, F.; Koerner, S.E.; Komatsu, K.J.; Smith, M.D.; Wilcox, K.R. A Comprehensive Approach to Analyzing Community Dynamics Using Rank Abundance Curves. Ecosphere 2019, 10, e02881. [Google Scholar] [CrossRef] [Green Version]
- Brunet, J.; Isacsson, G. Influence of Snag Characteristics on Saproxylic Beetle Assemblages in a South Swedish Beech Forest. J. Insect Conserv. 2009, 13, 515–528. [Google Scholar] [CrossRef]
- Procházka, J.; Schlaghamerský, J. Does Dead Wood Volume Affect Saproxylic Beetles in Montane Beech-Fir Forests of Central Europe? J. Insect Conserv. 2019, 23, 157–173. [Google Scholar] [CrossRef]
- Ulyshen, M.D. Wood Decomposition as Influenced by Invertebrates. Biol. Rev. 2016, 91, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Fierro, A.; Vergara, P.M.; Grez, A.A.; Alaniz, A.J.; Carvajal, M.A.; Lizama, M.; Ramírez-Hernández, A. Landscape-Scale Management of Exotic Forest Plantations: Synergy between Deadwood and Clear-Cutting Synchrony Modulates Saproxylic Beetle Diversity. Landsc. Ecol. 2020, 35, 621–638. [Google Scholar] [CrossRef]
- Heikkala, O.; Seibold, S.; Koivula, M.; Martikainen, P.; Müller, J.; Thorn, S.; Kouki, J. Retention Forestry and Prescribed Burning Result in Functionally Different Saproxylic Beetle Assemblages than Clear-Cutting. For. Ecol. Manag. 2016, 359, 51–58. [Google Scholar] [CrossRef]
- Castro, J.; Allen, C.D.; Molina-Morales, M.; Marañón-Jiménez, S.; Sánchez-Miranda, Á.; Zamora, R. Salvage Logging versus the Use of Burnt Wood as a Nurse Object to Promote Post-Fire Tree Seedling Establishment. Restor. Ecol. 2011, 19, 537–544. [Google Scholar] [CrossRef]
- Galbraith, S.M.; Cane, J.H.; Moldenke, A.R.; Rivers, J.W. Salvage Logging Reduces Wild Bee Diversity, but Not Abundance, in Severely Burned Mixed-Conifer Forest. For. Ecol. Manag. 2019, 453, 117622. [Google Scholar] [CrossRef]
- Vallejo, V.R.; Arianoutsou, M.; Moreira, F. Fire Ecology and Post-Fire Restoration Approaches in Southern European Forest Types. In Post-Fire Management and Restoration of Southern European Forests; Moreira, F., Arianoutsou, M., Corona, P., Delas Heras, J., Eds.; Springer: Dordrecht, The Netherlands, 2012; Volume 3, pp. 93–119. ISBN 1-4020-3758-9. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).