The Impact of Biotic and Abiotic Stress Factors on Development of European Ash Tissue Cultures
Abstract
1. Introduction
2. Materials and Methods
2.1. Source and Disinfection of Plant Material, Initiation of Plant Cultures In Vitro
2.2. Plant-Fungus Dual Cultures
2.3. The Impact of Selected Heavy Metals on Development of F. excelsior Callus Cultures and Seedlings
2.4. Protein Extraction and Antioxidative Enzymes Activities Assessment
2.5. The Influence of Different Salinity Concentrations on the Physiology of Tissue Cultures In Vitro
2.6. Statistical Analyzes
3. Results
3.1. The Impact of Biotic Stress Factors on the Callus Cultures and Seedlings of F. excelsior
3.2. The Impact of Abiotic Stress Factors on the Callus Cultures and Seedlings of F. excelsior
3.2.1. Heavy Metals
3.2.2. Catalase and Peroxidase (POX) Activities
3.2.3. Salinity
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heuertz, M.; Carnevale, S.; Fineschi, S.; Sebastiani, F.; Hausman, J.F.; Paule, L.; Vendramin, G.G. Chloroplast DNA phylogeography of European ashes, Fraxinus sp. (Oleaceae): Roles of hybridization and life history traits. Mol. Ecol. 2006, 15, 2131–2140. [Google Scholar] [CrossRef] [PubMed]
- Musolin, D.L.; Selikhovkin, A.V.; Shabunin, D.A.; Zviagintsev, V.B.; Baranchiko, Y.B. Between ash dieback and emerald ash borer: Two asian invaders in Russia and the future of ash in Europe. Balt. For. 2017, 23, 316–333. [Google Scholar]
- Keča, N.; Tkaczyk, M.; Żółciak, A.; Stocki, M.; Kalaji, H.M.; Nowakowska, J.A.; Oszako, T. Survival of European ash seedlings treated with phosphite after infection with the Hymenoscyphus fraxineus and Phytophthora species. Forests 2018, 9, 442. [Google Scholar] [CrossRef]
- Dobrowolska, D.; Hein, S.; Oosterbaan, A.; Wagner, S.; Clark, J.; Skovsgaard, J.P. A review of European ash (Fraxinus excelsior L.): Implications for silviculture. Forestry 2011, 84, 133–148. [Google Scholar] [CrossRef]
- Fijołek, M. Atlas Dzikich Roślin Jadalnych (Atlas of Wild Edible Plants), 1st ed.; Wydawnictwo SBM: Warsaw, Poland, 2017; pp. 66–67. [Google Scholar]
- Farrell, E.P.; Führer, E.; Ryan, D.; Andersson, F.; Hüttl, R.; Piussi, P. European forest ecosystems: Building the future on the legacy of the past. For. Ecol. Manag. 2000, 132, 5–20. [Google Scholar] [CrossRef]
- Surówka, E.; Rapacz, M.; Janowiak, F. Climate change influences the interactive effects of simultaneous impact of abiotic and biotic stresses on plants. In Plant Ecophysiology and Adaptation under Climate Change: Mechanisms and Perspectives I; Hasanuzzaman, M., Ed.; Springer: Singapore, 2020; pp. 1–50. [Google Scholar]
- Kowalski, T.; Bilański, P. Fungi detected in the previous year’s leaf petioles of fraxinus excelsior and their antagonistic potential against Hymenoscyphus fraxineus. Forests 2021, 12, 1412. [Google Scholar] [CrossRef]
- Turczański, K.; Rutkowski, P.; Dyderski, M.K.; Wrońska-Pilarek, D.; Nowiński, M. Soil pH and organic matter content affects European ash (Fraxinus excelsior L.) crown defoliation and its impact on understory vegetation. Forests 2020, 11, 22. [Google Scholar] [CrossRef]
- Lenz, H.D.; Bartha, B.; Straßer, L.; Lemme, H. Development of ash dieback in south-eastern Germany and the increasing occurrence of secondary pathogens. Forests 2016, 7, 41. [Google Scholar] [CrossRef]
- Stocks, J.J.; Metheringham, C.L.; Plumb, W.J.; Lee, S.J.; Kelly, L.J.; Nichols, R.A.; Buggs, R.J.A. Genomic basis of European ash tree resistance to ash dieback fungus. Nat. Ecol. Evol. 2019, 3, 1686–1696. [Google Scholar] [CrossRef]
- Fones, H.N.; Mardon, C.; Gurr, J.G. A role for the asexual spores in infection of fraxinus excelsior by the ash-dieback fungus Hymenoscyphus fraxineus. Sci. Rep. 2016, 6, 1–10. [Google Scholar]
- Gross, A.; Sieber, T.N. Virulence of Hymenoscyphus albidus and native and introduced Hymenoscyphus fraxineus on Fraxinus excelsior and Fraxinus pennsylvanica. Plant Pathol. 2016, 65, 655–663. [Google Scholar] [CrossRef]
- Kowalski, T.; Holdenrieder, O. The teleomorph of Chalara fraxinea, the causal agent of ash dieback. For. Pathol. 2009, 39, 304–308. [Google Scholar] [CrossRef]
- Bakys, R.; Vasaitis, R.; Skovsgaard, J.P. Patterns and severity of crown dieback in young even-aged stands of european ash (Fraxinus excelsior L.) in relation to stand density, bud flushing phenotype, and season. Plant. Prot. Sci. 2013, 49, 120–126. [Google Scholar] [CrossRef]
- Han, J.-G.; Shrestha, B.; Hosoya, T.; Lee, K.-H.; Sung, G.-H.; Shin, H.-D. First report of the ash dieback pathogen Hymensocyphus fraxineus in Korea. Mycobiology 2014, 42, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Enderle, R.; Metzler, B.; Riemer, U.; Gerald, K. Ash dieback on sample points of the national forest inventory in south-western Germany. Forests 2018, 9, 25. [Google Scholar] [CrossRef]
- Ioos, R.; Kowalski, T.; Husson, C.; Holdenrieder, O. Rapid in planta detection of Chalara fraxinea by a real-time PCR assay using a dual-labelled probe. Eur. J. Plant Pathol. 2009, 125, 329–335. [Google Scholar] [CrossRef]
- Ustawa o Lasach. 1997, Dz. U. 1991 Nr 101 poz. 444 (The Act on Forests. 1997. NS. of Laws 1991 No. 101, item 444). Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU19911010444 (accessed on 1 December 2021).
- Nawrot-Chorabik, K.; Marcol-Rumak, N.; Latowski, D. Investigation of biocontrol potential of two ash endophytes against Hymenoscyphus fraxineus using in vitro plant—fungus dual cultures. Forests 2021, 12, 1750. [Google Scholar] [CrossRef]
- Bálint, M.; Tiffin, P.; Hallström, B.; O’Hara, R.B.; Olson, M.S.; Fankhauser, J.D.; Piepenbring, M.; Schmitt, I. Host genotype shapes the foliar fungal microbiome of balsam poplar (Populus balsamifera). PLoS ONE 2013, 8, e53987. [Google Scholar] [CrossRef] [PubMed]
- Nawrot-Chorabik, K. Interactions between embryogenic callus of Abies alba and Heterobasidion spp. in dual cultures. Biol. Plant. 2014, 58, 363–369. [Google Scholar] [CrossRef]
- Hrib, J.; Rypacek, V. A simple callus test to determine the aggressiveness of wood-destroying fungi. Eur. J. For. Pathol. 1981, 11, 270–274. [Google Scholar] [CrossRef]
- Nawrot-Chorabik, K.; Pietrzykowski, M. Ecophysiological aspects of in vitro biotechnological studies using somatic embryogenesis of callus tissue toward protecting forest ecosystems. J. For. Res. 2019, 30, 1159–1166. [Google Scholar] [CrossRef]
- Bhusal, N.; Lee, M.; Reum Han, A.; Han, A.; Kim, H.S. Responses to drought stress in Prunus sargentii and Larix kaempferi seedlings using morphological and physiological parameters. For. Ecol. Manag. 2020, 465, 118099. [Google Scholar] [CrossRef]
- Tseng, S.; Liang, C.; Chia, T.; Ton, S. Changes in the composition of the soil bacterial community in heavy metal-contaminated farmland. Int. J. Environ. Res. Public Health 2021, 18, 8661. [Google Scholar] [CrossRef] [PubMed]
- Caracciolo, A.B.; Terenzi, V. Rhizosphere microbial communities and heavy metals. Microorganisms 2021, 9, 1462. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Bukhari, S.S.; Mustafa, A.; Ditta, A.; Alamri, S.; El-Esawi, M.A.; Rafique, M.; Ashraf, H.; Siddiqui, N.H. Mitigation of nickel toxicity and growth promotion in sesame through the application of a bacterial endophyte and zeolite in nickel contaminated soil. Int. J. Environ. Res. Public Health 2020, 17, 8859. [Google Scholar] [CrossRef]
- Bashir, M.A.; Naveed, M.; Ashraf, S.; Mustafa, A.; Ali, Q.; Rafique, M.; Alamri, S.; Siddiqui, M.H. Performance of Zea mays L. cultivars in tannery polluted soils: Management of chromium phytotoxicity through theapplication of biochar and compost. Physiol. Plant. 2021, 173, 129–147. [Google Scholar] [CrossRef] [PubMed]
- Niekerk, L.A.; Carelse, M.F.; Bakare, O.O.; Mavumengwana, V.; Keyster, M.; Gokul, A. The relationship between cadmium toxicity and the modulation of epigenetic traits in plants. Int. J. Mol. Sci. 2021, 22, 7046. [Google Scholar] [CrossRef]
- Nas, F.S.; Ali, M. The effect of lead on plants in terms of growing and biochemical parameters. MOJ Ecol. Environ. Sci. 2018, 3, 265–268. [Google Scholar]
- Severoglu, Z. Use of biomonitor European ash (Fraxinus excelsior L.) tree for monitoring traffic related heavy metal pollution in bishkek/kyrgyzstan: Impacts on plant nutrition. Fresenius Environ. Bull. 2018, 27, 9662–9673. [Google Scholar]
- Chuifan, Z.; Huang, M.; Ren, H.; Yu, Y.; Wu, J.; Ma, X. Bioaccumulation and detoxification mechanisms for lead uptake identified in Rhus chinensis Mill. Seedlings. Ecotoxicol. Environ. Saf. 2017, 142, 59–68. [Google Scholar]
- Narayani, M.; Srivastava, S. Elicitation: A stimulation of stress in in vitro plant cell/tissue cultures for enhancement of secondary metabolite production. Phytochem. Rev. 2017, 16, 1227–1252. [Google Scholar] [CrossRef]
- Gorji, T.; Yildirim, A.; Sertel, E.; Tanik, A. Remote sensing approaches and mapping methods for monitoring soil salinity under different climate regimes. Int. J. Geoinform. 2019, 6, 33–49. [Google Scholar] [CrossRef]
- Foyer, C.H.; Ruban, A.V.; Noctor, G. Viewing oxidative stress through the lens of oxidative signalling rather than damage. Biochem. J. 2017, 474, 877–883. [Google Scholar] [CrossRef]
- Isayenkov, S.V.; Maathuis, F.J.M. Plant salinity stress: Many unanswered questions remain. Front. Plant Sci. 2019, 10, 80. [Google Scholar] [CrossRef] [PubMed]
- Hussain, B.; Lucas, S.J.; Ozturk, L.; Budak, H. Mapping QTLs conferring salt tolerance and micronutrient concentrations at seedling stage in wheat. Sci. Rep. 2017, 7, 15662. [Google Scholar] [CrossRef]
- Smirnoff, N.; Arnaud, D. Hydrogen peroxide metabolism and functions in plants. New Phytol. 2019, 221, 1197–1214. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 1–13. [Google Scholar] [CrossRef]
- Munns, R.; Gilliham, M. Salinity tolerance of crops—What is the cost? New Phytol. 2015, 208, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Dumanović, J.; Nepovimova, E.; Natić, M.; Kuča, K.; Jaćević, V. The significance of reactive oxygen species and antioxidant defense system in plants: A concise overview. Front. Plant Sci. 2021, 11, 552969. [Google Scholar] [CrossRef] [PubMed]
- Libik, M.; Konieczny, R.; Pater, B.; Ślesak, I.; Miszalski, Z. Differences in the activities of some antioxidant enzymes and in H2O2 content during rhizogenesis and somatic embryogenesis in callus cultures of the ice plant. Plant Cell Rep. 2005, 23, 834–841. [Google Scholar] [CrossRef]
- Konieczny, R.; Banaś, A.K.; Surówka, E.; Michalec, Ż.; Miszalski, Z.; Libik-Konieczny, M. Pattern of antioxidant enzyme activities and hydrogen peroxide content during developmental stages of rhizogenesis from hypocotyl explants of Mesembryanthemum crystallinum L. Plant Cell Rep. 2014, 33, 165–177. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meratan, A.A.; Ghaffari, S.M.; Niknam, V. In vitro organogenesis and antioxidant enzymes activity in Acanthophyllum sordidum. Biol. Plant. 2009, 53, 5–10. [Google Scholar] [CrossRef]
- Vatankhah, E.; Niknam, V.; Ebrahimzadeh, H. Activity of antioxidant enzyme during in vitro organogenesis in Crocus sativus. Biol. Plant. 2010, 3, 509–514. [Google Scholar] [CrossRef]
- Nawrot-Chorabik, K.; Osmenda, M.; Słowiński, K.; Latowski, D.; Tabor, S.; Woodward, S. Stratification, scarification and application of phytohormones promote dormancy breaking and germination of pelleted scots pine (Pinus sylvestris L.) seeds. Forests 2021, 12, 621. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 495–497. [Google Scholar] [CrossRef]
- Kowalski, T.; Bilański, P. Hymenoscyphus pusillus, a new species on leaves of Fraxinus pennsylvanica in Poland. For. Pathol. 2019, 49, e12481. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitationof microgram quantities of protein utilizing the principle ofprotein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. In Methods in Enzymology; Academic Press: London, UK, 1984; Volume 105, pp. 121–126. [Google Scholar]
- Allgood, G.S.; Perry, J.J. Oxygen defense systems in obligately thermophilic bacteria. Can. J. Microbiol. 1985, 31, 1006–1010. [Google Scholar] [CrossRef]
- Erfmeier, A.; Haldan, K.L.; Beckmann, L.M.; Behrens, M.; Rotert, J.; Schrautzer, J. Ash dieback and its impact in near-natural forest remnants—A plant community-based inventory. Front. Plant Sci. 2019, 10, 658. [Google Scholar] [CrossRef]
- Pereira, C.; Montalbán, I.A.; Pedrosa, A.; Tavares, J.; Pestryakov, A.; Bogdanchikova, N.; Canhoto, J.; Moncaleán, P. Regeneration of Pinus halepensis (Mill.) through organogenesis from apical shoot buds. Forests 2021, 12, 363. [Google Scholar] [CrossRef]
- Fenning, T.M. The use of tissue culture and in-vitro approaches for the study of tree diseases. Tissue Organ Cult. 2019, 136, 415–430. [Google Scholar] [CrossRef]
- Miura, K.; Tada, Y. Regulation of water, salinity, and cold stress responses by salicylic acid. Front. Plant Sci. 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, J.; Qin, G.; Cui, H.; Wang, Q. Salicylic acid analogues with biological activity may induce chilling tolerance of maize (Zea mays) seeds. Botany 2012, 90, 845–855. [Google Scholar] [CrossRef]
- Zengin, F. Effects of exogenous salicylic acid on growth characteristics and bio-chemical content of wheat seeds under arsenic stress. J. Environ. Biol. 2015, 36, 249. [Google Scholar]
- Hattori, H. Influence of heavy metals on soil microbial activities. Soil. Sci Plant Nut. 1992, 38, 93–100. [Google Scholar] [CrossRef]
- Raddi, S.; Mariotti, B.; Martini, S.; Pierguidi, A. Salinity tolerance in Fraxinus angustifolia vahl.: Seed emergence in field and germination trials. Forests 2019, 10, 940. [Google Scholar] [CrossRef]
- Corpas, F.J.; del Rio, L.A.; Palma, J.M. Plant peroxisomes at the crossroad of NO and H2O2 metabolism. J. Integr. Plant Biol. 2019, 61, 803–816. [Google Scholar]
- Mur, L.A.J.; Mandon, J.; Persijn, S.; Cristescu, S.M.; Moshkov, I.E.; Novikova, G.V.; Hall, M.A.; Harren, F.J.M.; Hebelstrup, K.H.; Gupta, K.J. Nitric oxide in plants: An assessment of the current state of knowledge. AoB Plants 2013, 5, 1–17. [Google Scholar] [CrossRef]
- Bhusal, N.; Lee, M.; Lee, H.; Adhikari, A.; Han, A.R.; Kim, H.S. Evaluation of morphological, physiological, and biochemical traits for assessing drought resistance in eleven tree species. Sci. Total. Environ. 2021, 779, 146466. [Google Scholar] [CrossRef]
- Khaleghi, A.; Naderi, R.; Brunetti, C.; Maserti, B.E.; Babalar, M. Morphological, physiochemical and antioxidant responses of Maclura pomifera to drought stress. Sci. Rep. 2019, 9, 19250. [Google Scholar] [CrossRef]
- Bauwe, H.; Hagemann, M.; Kern, R.; Timm, S. Photorespiration has a dual origin and manifold links to central metabolism. Curr. Opin. Plant Biol. 2012, 15, 269–275. [Google Scholar] [CrossRef]
- Pandey, V.P.; Awasthi, M.; Singh, S.; Tiwari, S.; Dwivedi, U.N. A Comprehensive review on function and application of plant peroxidases. Biochem. Anal. Biochem. 2017, 6, 1. [Google Scholar] [CrossRef]
- Racchi, M.; Bagnoli, F.; Balla, I.; Danti, S. Differential activity of catalase and superoxide dismutase in seedlings and in vitro micropropagated oak (Quercus robur L.). Plant Cell Rep. 2001, 20, 169–174. [Google Scholar]
- Rousseau, C.; Belin, E.; Bove, E.; Rousseau, D.; Fabre, F.; Berruyer, R.; Guillaumes, J.; Manceau, C.; Jacques, M.-A.; Boureau, T. High throughput quantitative phenotyping of plant resistance using chlorophyll fluorescence image analysis. Plant Methods 2013, 17, 1–13. [Google Scholar] [CrossRef]
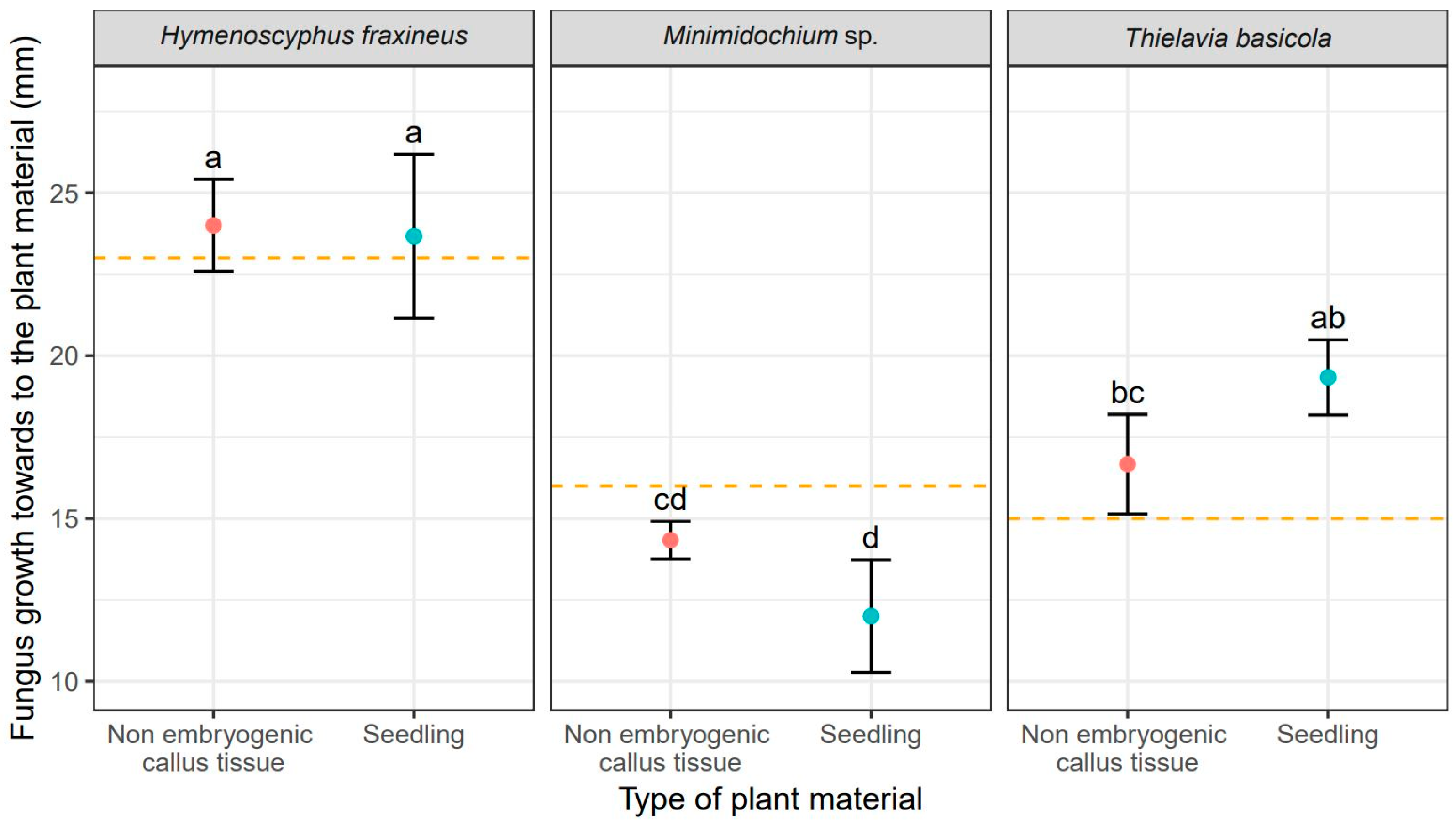

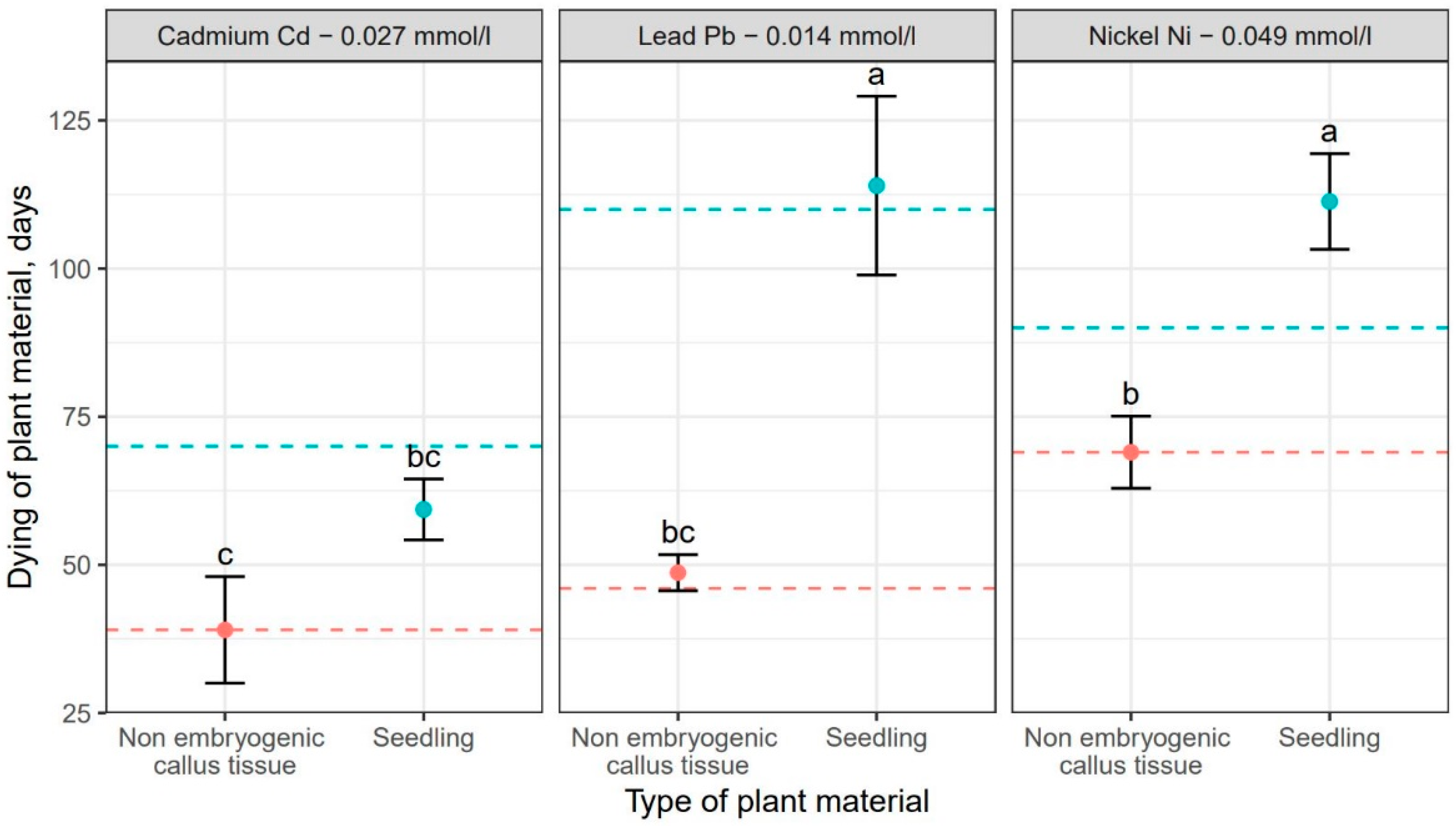

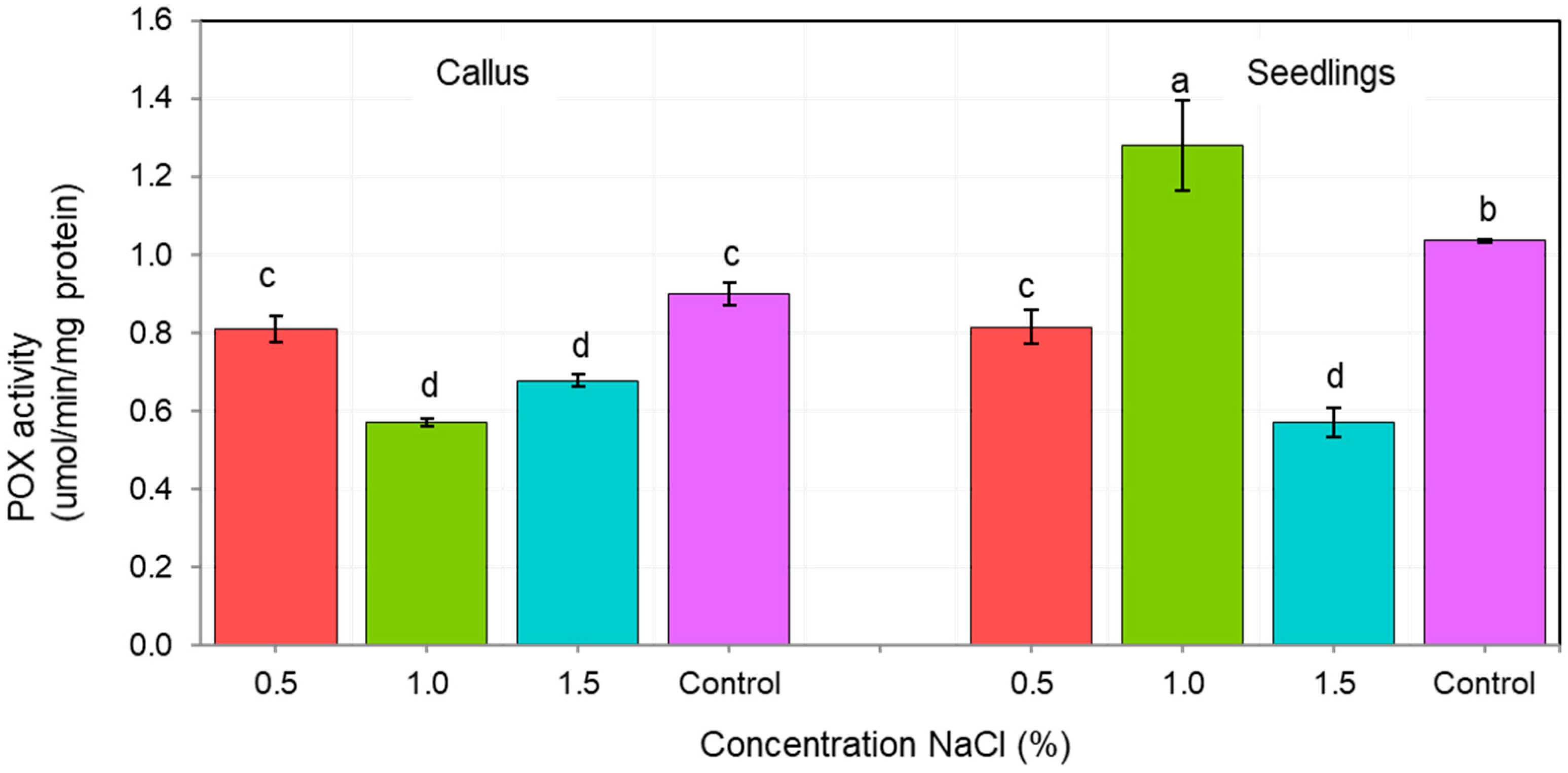
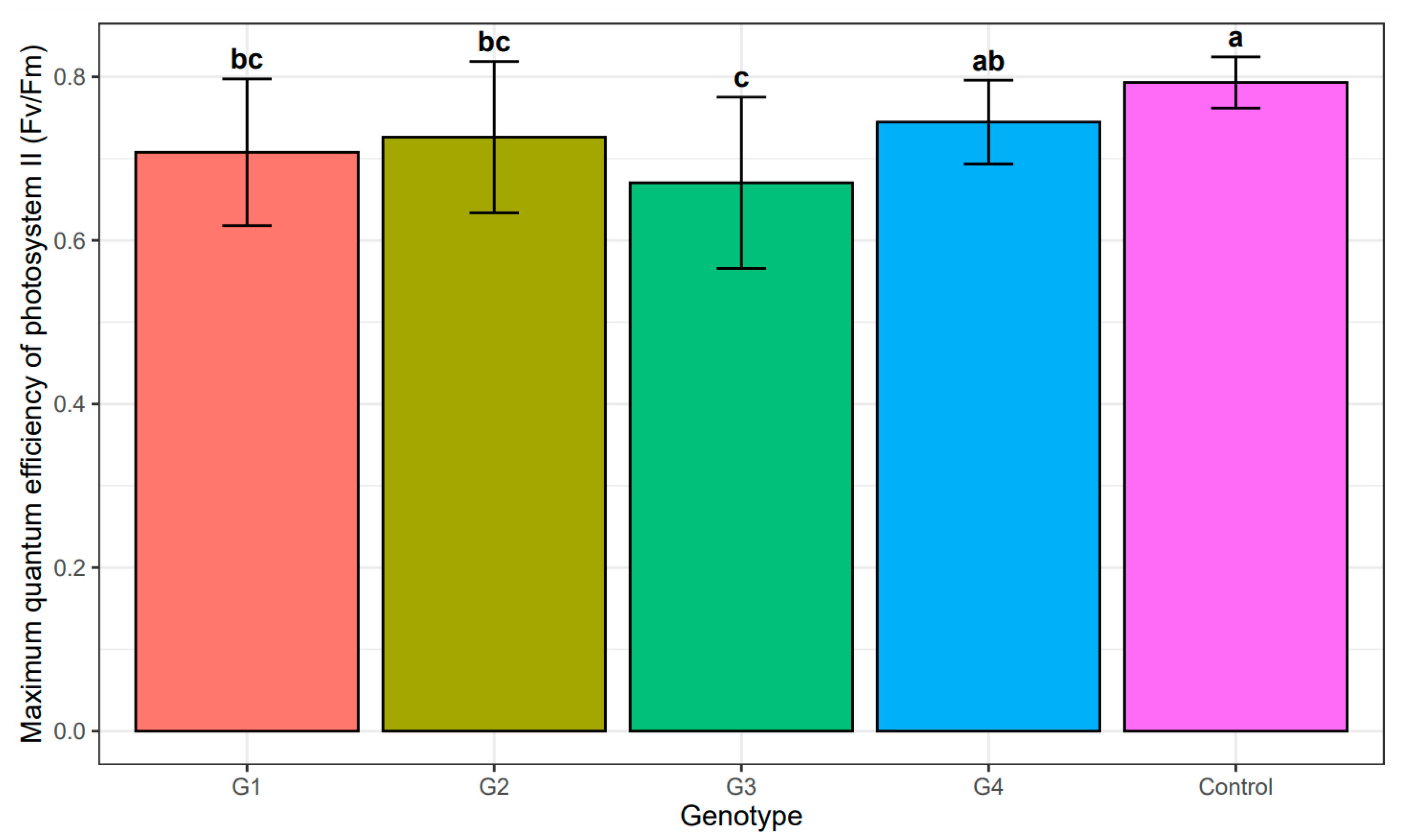
| Sources of Variance | Df | Sum Sq | Mean Sq | F Value | Pr (>F) | |
|---|---|---|---|---|---|---|
| Fungus growth towards plant material (mm) | ||||||
| Fungus | 2 | 308.37 | 154.18 | 59.163 | <0.001 | *** |
| Plant material | 1 | 0.00 | 0.00 | 0.001 | 0.976 | |
| Fungus × Plant material | 2 | 18.96 | 9.48 | 3.638 | 0.061 | |
| Residuals | 11 | 28.67 | 2.61 | |||
| Fungus growth opposite the plant material (mm) | ||||||
| Fungus | 2 | 276.91 | 138.45 | 71.390 | <0.001 | *** |
| Plant material | 1 | 7.74 | 7.74 | 3.989 | 0.071 | |
| Fungus × Plant material | 2 | 0.96 | 0.48 | 0.249 | 0.784 | |
| Residuals | 11 | 21.33 | 1.94 | - | - | |
| Fungus | Non-Embryonic Callus Tissue | Seedling | Average |
|---|---|---|---|
| Fungus growth towards plant material (mm) | |||
| Hymenocyphus fraxineus | 24.00 | 23.70 | 23.80 (a) 1 |
| Thielavia basicola | 16.70 | 19.30 | 18.00 (b) 1 |
| Minimidochium sp. | 14.30 | 12.00 | 13.17 (c) 1 |
| Average | 17.76 (a) 2 | 18.30 (a) 2 | - |
| Fungus growth opposite the plant material (mm) | |||
| Hymenocyphus fraxineus | 8.00 | 9.00 | 8.60 (b) 3 |
| Thielavia basicola | 6.70 | 8.70 | 7.67 (b) 3 |
| Minimidochium sp. | 16.00 | 17.00 | 16.50 (a) 3 |
| Average | 10.50 (a) 4 | 11.55 (a) 4 | - |
| Metal | Non-Embryonic Callus Tissue | Seedling | Average |
|---|---|---|---|
| Dieback (days) | Dieback (days) | ||
| Nickel 0.049 | 69.00 | 106.00 | 87.5 (a) 1 |
| Lead 0.014 | 48.00 | 113.00 | 80.5 (a) 1 |
| Cadmium 0.027 | 39.00 | 62.00 | 50.5 (b) 1 |
| Avarge | 52.00 (b) 2 | 93.70 (a) 2 | - |
| Sources of Variance | Df | Sum Sq | Mean Sq | F Value | Pr (>F) | |
|---|---|---|---|---|---|---|
| Metal | 2 | 6181.00 | 3091.00 | 41.58 | <0.001 | *** |
| Plants material | 1 | 10,417.00 | 10,417.00 | 140.13 | <0.001 | *** |
| Metal × Plant material | 2 | 1829.00 | 915.00 | 12.30 | <0.001 | *** |
| Residuals | 18 | 1338.00 | 74.00 | - | - |
| Sources of Variance | Df | Sum Sq | Mean Sq | F Value | Pr (>F) |
|---|---|---|---|---|---|
| All studied genotypes and control | |||||
| Genotype | 4 | 0.1434 | 0.0358 | 6.308 | <0.001 |
| Residuals | 88 | 0.5001 | 0.0057 | ||
| Genotype G1 | |||||
| Concentration | 2 | 0.0805 | 0.0402 | 25.1885 | <0.001 |
| Residuals | 10 | 0.0160 | 0.0016 | ||
| Genotype G2 | |||||
| Concentration | 2 | 0.0165 | 0.0082 | 0.9588 | 0.402 |
| Residuals | 18 | 0.1546 | 0.0086 | ||
| Genotype G3 | |||||
| Concentration | 2 | 0.0166 | 0.0083 | 0.7277 | 0.503 |
| Residuals | 12 | 0.1369 | 0.0114 | ||
| Genotype G4 | |||||
| Concentration | 2 | 0.0151 | 0.0076 | 3.5016 | 0.049 |
| Residuals | 21 | 0.0453 | 0.0022 | ||
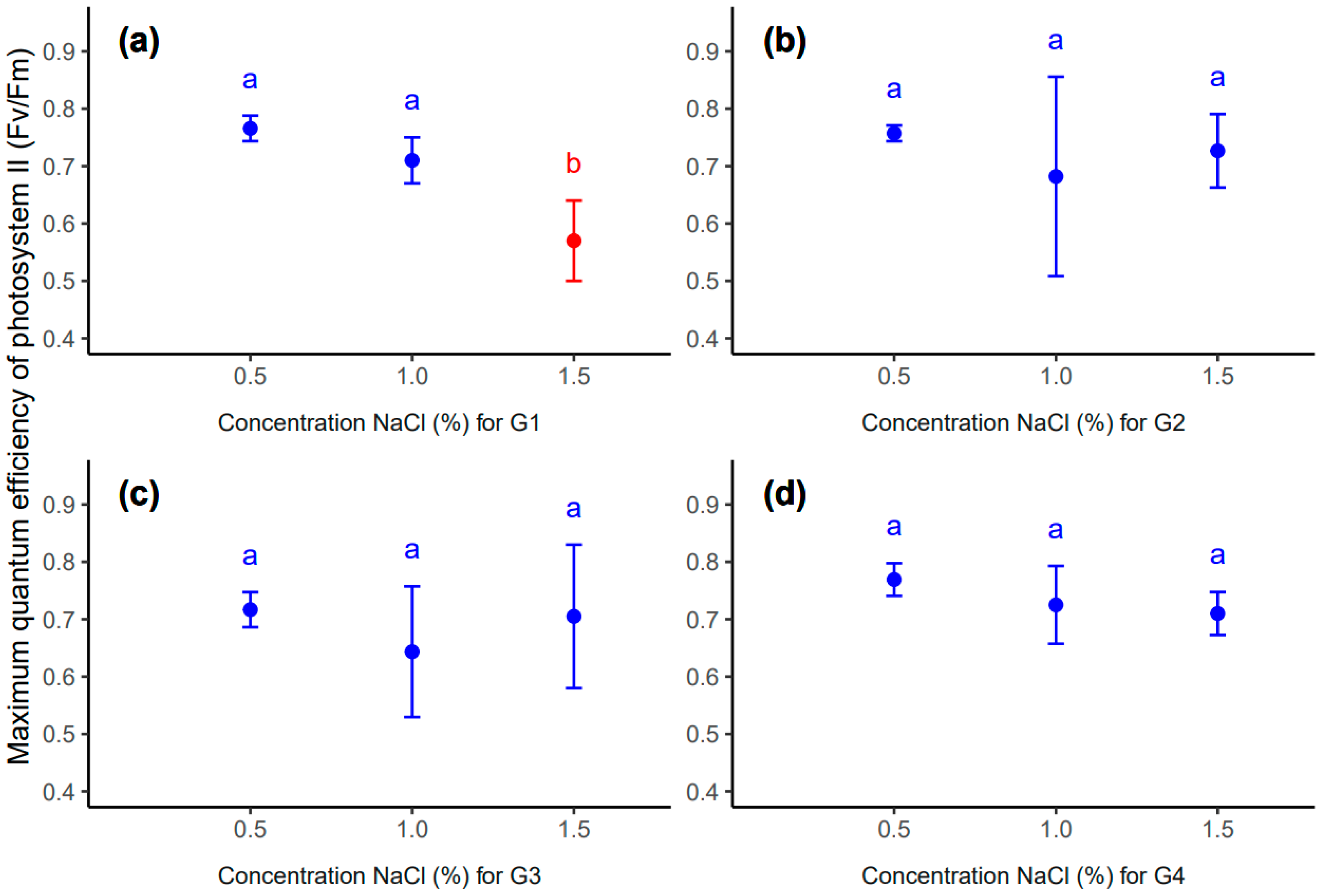
| Concentration NaCl | Value | SD | n | Min | Max | Q25 | Q50 | Q75 | Groups | |
|---|---|---|---|---|---|---|---|---|---|---|
| Genotype G1 | ||||||||||
| 0.5 | 0.77 | 0.02 | 7 | 0.74 | 0.80 | 0.75 | 0.76 | 0.78 | a 1 | |
| 1.0 | 0.71 | 0.04 | 3 | 0.67 | 0.75 | 0.69 | 0.71 | 0.73 | a 1 | |
| 1.5 | 0.57 | 0.07 | 3 | 0.50 | 0.64 | 0.53 | 0.57 | 0.60 | b 1 | |
| G1 Total | 0.71 | 0.09 | 13 | 0.50 | 0.80 | 0.67 | 0.75 | 0.76 | bc 2 | |
| Genotype G2 | ||||||||||
| 0.5 | 0.76 | 0.01 | 7 | 0.73 | 0.77 | 0.76 | 0.76 | 0.76 | a 3 | |
| 1.0 | 0.68 | 0.17 | 5 | 0.41 | 0.82 | 0.61 | 0.76 | 0.81 | a 3 | |
| 1.5 | 0.73 | 0.06 | 9 | 0.60 | 0.80 | 0.68 | 0.74 | 0.78 | a 3 | |
| G2 Total | 0.73 | 0.09 | 21 | 0.41 | 0.82 | 0.72 | 0.76 | 0.77 | bc 2 | |
| Genotype G3 | ||||||||||
| 0.5 | 0.72 | 0.03 | 3 | 0.69 | 0.75 | 0.70 | 0.71 | 0.73 | a 4 | |
| 1.0 | 0.64 | 0.11 | 9 | 0.41 | 0.76 | 0.63 | 0.68 | 0.71 | a 4 | |
| 1.5 | 0.70 | 0.12 | 3 | 0.58 | 0.83 | 0.64 | 0.70 | 0.77 | a 4 | |
| G3 Total | 0.67 | 0.10 | 15 | 0.41 | 0.83 | 0.64 | 0.70 | 0.72 | c 2 | |
| Genotype G4 | ||||||||||
| 0.5 | 0.77 | 0.03 | 12 | 0.73 | 0.81 | 0.74 | 0.78 | 0.79 | a 5 | |
| 1.0 | 0.72 | 0.07 | 8 | 0.61 | 0.80 | 0.69 | 0.73 | 0.78 | a 5 | |
| 1.5 | 0.71 | 0.04 | 4 | 0.67 | 0.76 | 0.69 | 0.70 | 0.72 | a 5 | |
| G4 Total | 0.74 | 0.05 | 24 | 0.61 | 0.81 | 0.71 | 0.75 | 0.79 | ab 2 | |
| Control | 0.79 | 0.03 | 20 | 0.72 | 0.83 | 0.78 | 0.80 | 0.82 | a 2 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nawrot-Chorabik, K.; Sułkowska, M.; Osmenda, M.; Mohytych, V.; Surówka, E.; Latowski, D. The Impact of Biotic and Abiotic Stress Factors on Development of European Ash Tissue Cultures. Forests 2022, 13, 59. https://doi.org/10.3390/f13010059
Nawrot-Chorabik K, Sułkowska M, Osmenda M, Mohytych V, Surówka E, Latowski D. The Impact of Biotic and Abiotic Stress Factors on Development of European Ash Tissue Cultures. Forests. 2022; 13(1):59. https://doi.org/10.3390/f13010059
Chicago/Turabian StyleNawrot-Chorabik, Katarzyna, Małgorzata Sułkowska, Małgorzata Osmenda, Vasyl Mohytych, Ewa Surówka, and Dariusz Latowski. 2022. "The Impact of Biotic and Abiotic Stress Factors on Development of European Ash Tissue Cultures" Forests 13, no. 1: 59. https://doi.org/10.3390/f13010059
APA StyleNawrot-Chorabik, K., Sułkowska, M., Osmenda, M., Mohytych, V., Surówka, E., & Latowski, D. (2022). The Impact of Biotic and Abiotic Stress Factors on Development of European Ash Tissue Cultures. Forests, 13(1), 59. https://doi.org/10.3390/f13010059







