Variation in Carbon Content among the Major Tree Species in Hemiboreal Forests in Latvia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
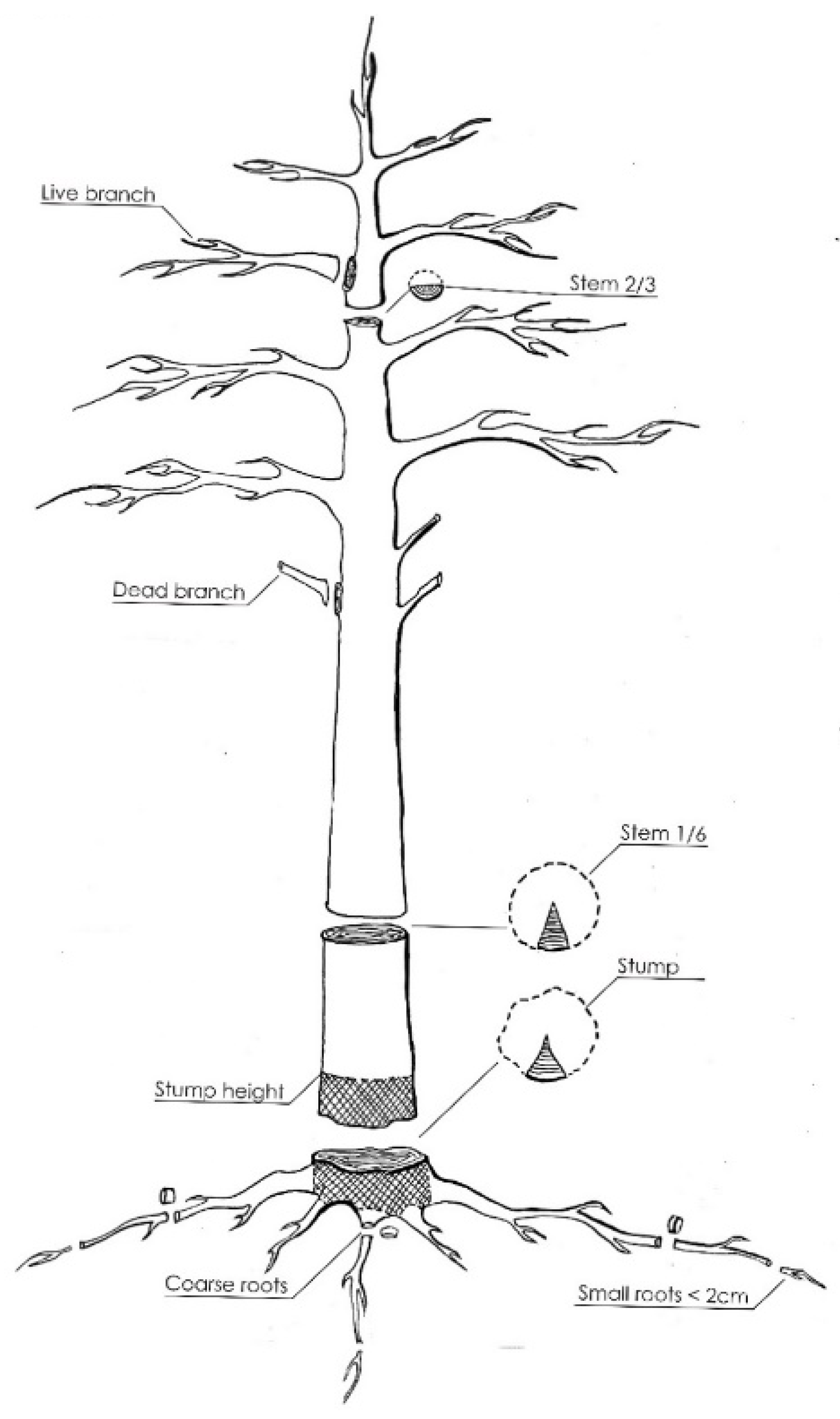
2.2. Sampling Design and Chemical Analysis
- Stump—monolith (both above- and belowground portions), nondifferentiated parts of some roots;
- Coarse roots—diameter greater than or equal to 2 cm;
- Small roots—diameter less than 2 cm.
2.3. Data Analysis
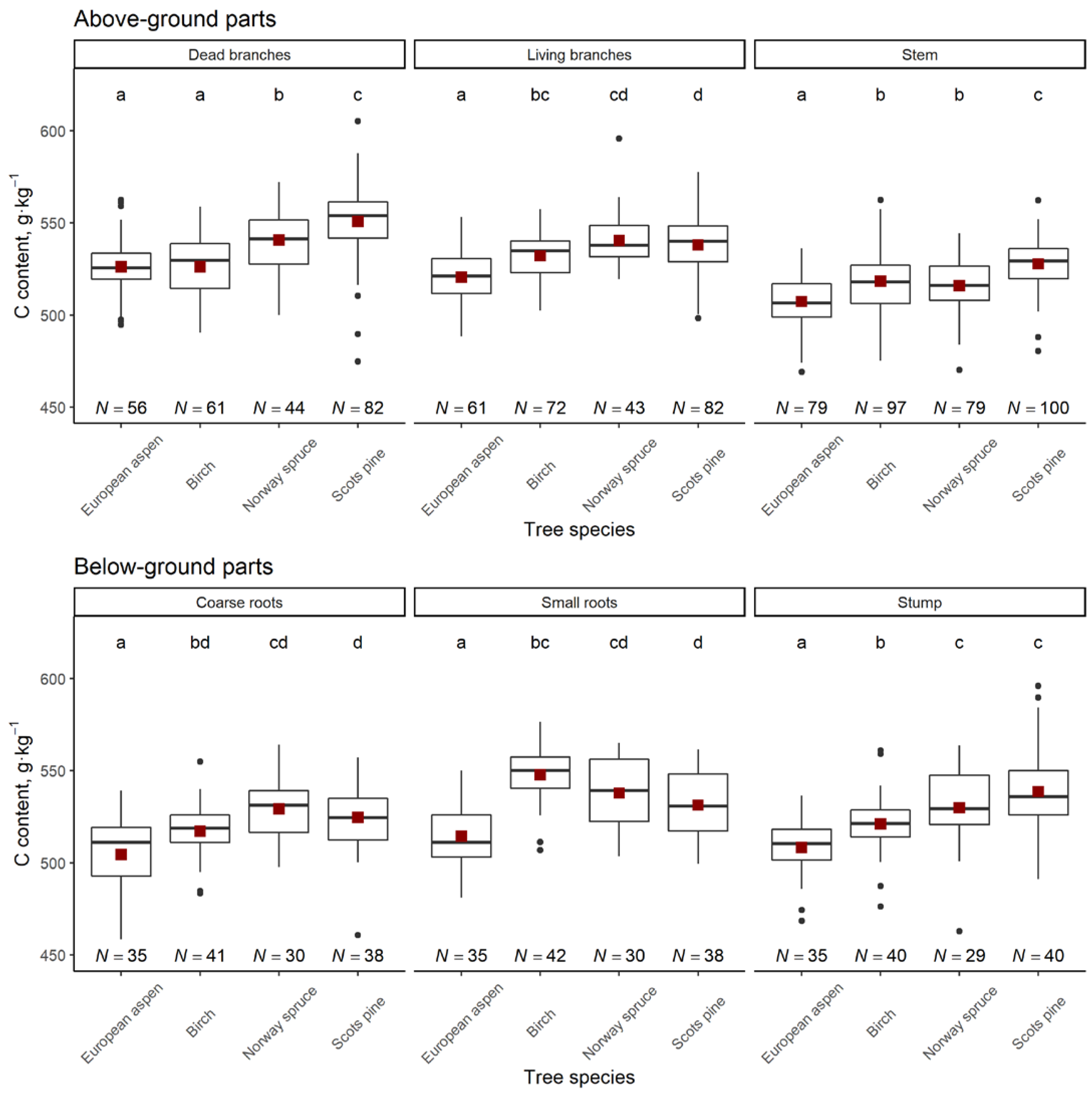
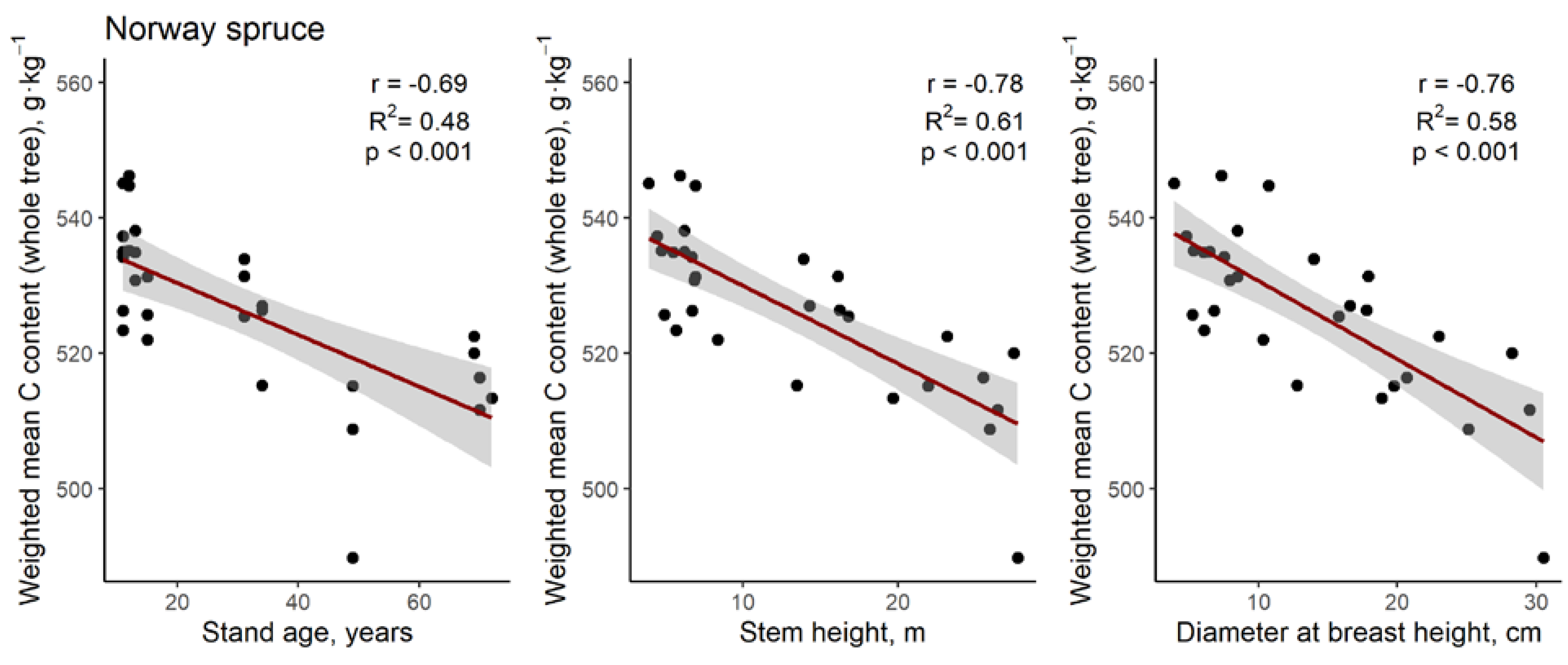
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef] [Green Version]
- Hudiburg, T.W.; Law, B.E.; Moomaw, W.R.; Harmon, M.E.; Stenzel, J.E. Meeting GHG reduction targets requires accounting for all forest sector emissions. Environ. Res. Lett. 2019, 14, 095005. [Google Scholar] [CrossRef]
- Anderson-Teixeira, K.J.; Herrmann, V.; Banbury Morgan, R.; Bond-Lamberty, B.; Cook-Patton, S.C.; Ferson, A.E.; Muller-Landau, H.C.; Wang, M.M.H. Carbon cycling in mature and regrowth forests globally. Environ. Res. Lett. 2021, 16, 053009. [Google Scholar] [CrossRef]
- Gower, S. Patterns and mechanisms of the forest carbon cycle. Annu. Rev. Environ. Resour. 2003, 28, 169–204. [Google Scholar] [CrossRef]
- Lamlom, S.H.; Savidge, R.A. A reassessment of carbon content in wood: Variation within and between 41 North American species. Biomass Bioenergy 2003, 25, 381–388. [Google Scholar] [CrossRef]
- Taub, D.R. Effects of rising atmospheric concentrations of carbon dioxide on plants. Nat. Educ. Knowl. 2010, 3, 21. [Google Scholar]
- U.S. EPA (United States Environmental Protection Agency). EPA’s Report on the Environment (ROE). Carbon Storage in Forests. Available online: https://www.epa.gov/report-environment (accessed on 18 August 2021).
- Favero, A.; Daigneault, A.; Sohngen, B. Forests: Carbon sequestration, biomass energy, or both? Sci. Adv. 2020, 6, eaay6792. [Google Scholar] [CrossRef] [Green Version]
- Thomas, S.C.; Martin, A.R. Carbon content of tree tissues: A synthesis. Forests 2012, 3, 332–352. [Google Scholar] [CrossRef] [Green Version]
- Gao, B.; Taylor, A.R.; Chen, H.Y.H.; Wang, J. Variation in total and volatile carbon concentration among the major tree species of the boreal forest. For. Ecol. Manag. 2016, 375, 191–199. [Google Scholar] [CrossRef]
- Zhou, L.; Li, S.; Liu, B.; Wu, P.; Heal, K.V.; Ma, X. Tissue-specific carbon concentration, carbon stock, and distribution in Cunninghamia lanceolata (Lamb.) Hook plantations at various developmental stages in subtropical China. Ann. For. Sci. 2019, 76, 70. [Google Scholar] [CrossRef]
- Matthews, G. The Carbon Content of Trees; Technical Paper; Forestry Commission: Edinburgh, UK, 1993; p. 28. [Google Scholar]
- Bert, D.G.; Danjon, F. Carbon concentration variations in the roots, stem and crown of mature Pinus pinaster (Ait.). For. Ecol. Manag. 2006, 222, 279–295. [Google Scholar] [CrossRef]
- Wang, X.W.; Weng, Y.H.; Liu, G.F.; Krasowski, M.J.; Yang, C.P. Variations in carbon concentration, sequestration and partitioning among Betula platyphylla provenances. For. Ecol. Manag. 2015, 358, 344–352. [Google Scholar] [CrossRef]
- Ma, S.; He, F.; Tian, D.; Zou, D.; Yan, Z.; Yang, Y.; Zhou, T.; Huang, K.; Shen, H.; Fang, J. Variations and determinants of carbon content in plants: A global synthesis. Biogeosciences 2018, 15, 693–702. [Google Scholar] [CrossRef] [Green Version]
- Zapletal, M.; Juráò, S.; Krpeð, V.; Michna, K.; Edwards-Jonáðová, M.; Cudlín, P. Effect of ozone flux on selected structural and antioxidant characteristics of a mountain Norway spruce forest. Balt. For. 2018, 24, 261–267. [Google Scholar]
- Juráň, S.; Grace, J.; Urban, O. Temporal changes in ozone concentrations and their impact on vegetation. Atmosphere 2021, 12, 82. [Google Scholar] [CrossRef]
- Martin, A.R.; Doraisami, M.; Thomas, S.C. Global patterns in wood carbon concentration across the world’s trees and forests. Nat. Geosci. 2018, 11, 915–920. [Google Scholar] [CrossRef]
- IPCC. 2006 IPCC Guidelines for National Greenhouse Gas Inventories. In IPCC National Greenhouse Gas Inventories Programme; Eggleston, H.S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Eds.; IGES: Kanagawa, Japan, 2006. [Google Scholar]
- Thomas, S.C.; Malczewski, G. Wood carbon content of tree species in Eastern China: Interspecific variability and the importance of the volatile fraction. J. Environ. Manag. 2007, 85, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Tolunay, D. Carbon concentrations of tree components, forest floor and understory in young Pinus sylvestris stands in north-western Turkey. Scand. J. For. Res. 2009, 24, 394–402. [Google Scholar] [CrossRef]
- Ying, J.; Weng, Y.; Oswald, B.P.; Zhang, H. Variation in carbon concentrations and allocations among Larix olgensis populations growing in three field environments. Ann. For. Sci. 2019, 76, 99. [Google Scholar] [CrossRef] [Green Version]
- Martin, A.R.; Gezahegn, S.; Thomas, S.C. Variation in carbon and nitrogen concentration among major woody tissue types in temperate trees. Can. J. For. Res. 2015, 45, 744–757. [Google Scholar] [CrossRef]
- EEA. European Forest Types. Categories and Types for Sustainable Forest Management Reporting and Policy; EEA Technical Report No 9/2006; EEA: Copenhagen, Denmark, 2007; p. 114. [Google Scholar]
- Liepiņš, J.; Lazdiņš, A.; Liepiņš, K. Equations for estimating above- and belowground biomass of Norway spruce, Scots pine, birch spp. and European aspen in Latvia. Scand. J. For. Res. 2018, 33, 58–70. [Google Scholar] [CrossRef]
- The R Project for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 18 August 2021).
- Janssens, I.A.; Sampson, D.A.; Cermák, J.; Meiresonne, L.; Riguzzi, F.; Overloop, S.; Ceulemans, R. Above- and belowground phytomass and carbon storage in a Belgian Scots pine stand. Ann. For. Sci. 1999, 56, 81–90. [Google Scholar] [CrossRef]
- Savidge, R.A. Biochemistry of Seasonal Cambial Growth and Wood Formation-An Overview of the Challenges. In Cell and Molecular Biology of Wood Formation; Savidge, R.A., Barnett, J.R., Napier, R., Eds.; BIOS Scientific Publishers Ltd.: Oxford, UK, 2000; p. 30. [Google Scholar]
- Laiho, R.; Laine, J. Tree stand biomass and carbon content in an age sequence of drained pine mires in southern Finland. For. Ecol. Manag. 1997, 93, 161–169. [Google Scholar] [CrossRef]
- Bembenek, M.; Giefing, D.F.; Jelonek, T.; Karaszewski, Z.; Kruszyk, R.; Tomczak, A.; Woszczyk, M.; Mederski, P.S. Carbon content in juvenile and mature wood of Scots pine (Pinus sylvestris L.). Balt. For. 2015, 21, 279–284. [Google Scholar]
- Wegiel, A.; Polowy, K. Aboveground carbon content and storage in mature Scots pine stands of different densities. Forests 2020, 11, 240. [Google Scholar] [CrossRef] [Green Version]
- Zobel, B.J.; Sprague, J.R. Juvenile Wood in Forest Trees; Springer Series in Wood Science; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Martin, A.R.; Thomas, S.C.; Zhao, Y. Size-dependent changes in wood chemical traits: A comparison of neotropical saplings and large trees. AoB PLANTS 2013, 5, plt039. [Google Scholar] [CrossRef]
- Ma, S.; Eziz, A.; Tian, D.; Yan, Z.; Cai, Q.; Jiang, M.; Ji, C.; Fang, J. Tree size and age induced stem carbon content variations cause an uncertainty in forest carbon stock estimation. Biogeosci. Discuss. 2019. [preprint]. [Google Scholar] [CrossRef]
- Skovsgaard, J.P.; Stupak, I.; Vesterdal, L. Distribution of biomass and carbon in even-aged stands of Norway spruce (Picea abies (L.) Karst.): A case study on spacing and thinning effects in northern Denmark. Scand. J. For. Res. 2006, 21, 470–488. [Google Scholar] [CrossRef]


| Parameter, Unit | Value | Tree Species | |||
|---|---|---|---|---|---|
| Norway Spruce | Scots Pine | Birch | European Aspen | ||
| Number of sample trees | total number | 81 | 102 | 105 | 84 |
| Age 1, years | average | 41 | 54 | 35 | 23 |
| range | 9–97 | 6–141 | 8–92 | 5–76 | |
| Stem height, m | average ± S.E. | 16.6 ± 1.0 | 17.3 ± 0.9 | 18.1 ± 0.8 | 16.6 ± 0.9 |
| range | 2.8–30.8 | 1.9–34.5 | 4.9–32.3 | 3.7–29.9 | |
| Diameter at breast height, cm | average ± S.E. | 17.5 ± 1.0 | 19.0 ± 0.9 | 14.7 ± 0.7 | 13.8 ± 0.9 |
| range | 2.4–36.3 | 1.5–45.3 | 2.7–37.2 | 2.8–34.1 | |
| Tree Part | Values | Weighted Mean C Content in Tree, g·kg−1 | |||
|---|---|---|---|---|---|
| Norway Spruce | Scots Pine | Birch | European Aspen | ||
| Aboveground parts | average ± S.E. | 524.4 ± 1.4 a | 530.4 ± 1.3 b | 520.6 ± 1.4 c | 510.2 ± 1.3 d |
| median | 524.2 | 531.3 | 520.4 | 509.8 | |
| range | 483.9–551.7 | 467.2–562.9 | 487.8–559.7 | 480.9–534.6 | |
| Belowground parts | average ± S.E. | 529.9 ± 2.6 a | 531.5 ± 2.4 a | 527.9 ± 1.7 a | 507.4 ± 2.1 b |
| median | 529.0 | 529.4 | 528.9 | 508.4 | |
| range | 497.2–559.3 | 486.5–567.0 | 502.9–549.6 | 482.1–531.9 | |
| Whole | average ± S.E. | 526.5 ± 2.3 a | 533.2 ± 1.6 b | 521.4 ± 1.5 c | 509.0 ± 1.6 d |
| median | 526.4 | 535.5 | 521.5 | 507.9 | |
| range | 489.8–546.2 | 502.1–554.7 | 501.3–550.5 | 490.0–527.4 | |
| Values | Weighted Mean C Content for Forest Stands, g·kg−1 | |||
|---|---|---|---|---|
| Stands Dominated by Norway Spruce | Stands Dominated by Scots Pine | Stands Dominated by Birch | Stands Dominated by European Aspen | |
| Average ± S.E. | 525.6 ± 0.1 a | 531.3 ± 0.1 b | 521.4 ± 0.1 c | 512.7 ± 0.1 d |
| Median | 526.4 | 532.1 | 521.4 | 512.0 |
| Range | 518.7–529.6 | 518.4–533.2 | 513.9–527.9 | 509.0–523.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bārdule, A.; Liepiņš, J.; Liepiņš, K.; Stola, J.; Butlers, A.; Lazdiņš, A. Variation in Carbon Content among the Major Tree Species in Hemiboreal Forests in Latvia. Forests 2021, 12, 1292. https://doi.org/10.3390/f12091292
Bārdule A, Liepiņš J, Liepiņš K, Stola J, Butlers A, Lazdiņš A. Variation in Carbon Content among the Major Tree Species in Hemiboreal Forests in Latvia. Forests. 2021; 12(9):1292. https://doi.org/10.3390/f12091292
Chicago/Turabian StyleBārdule, Arta, Jānis Liepiņš, Kaspars Liepiņš, Jeļena Stola, Aldis Butlers, and Andis Lazdiņš. 2021. "Variation in Carbon Content among the Major Tree Species in Hemiboreal Forests in Latvia" Forests 12, no. 9: 1292. https://doi.org/10.3390/f12091292
APA StyleBārdule, A., Liepiņš, J., Liepiņš, K., Stola, J., Butlers, A., & Lazdiņš, A. (2021). Variation in Carbon Content among the Major Tree Species in Hemiboreal Forests in Latvia. Forests, 12(9), 1292. https://doi.org/10.3390/f12091292






