Determining Statistically Robust Changes in Ungulate Browsing Pressure as a Basis for Adaptive Wildlife Management
Abstract
1. Introduction
- (i)
- How can the variability of browsing impact and its change over time be predicted and tested for statistical significance?
- (ii)
- Can the sample size of the established regeneration inventory be reduced and still serve wildlife managers as a valuable information source?
- (iii)
- What is the minimum observable significant change with respect to browsing impact in the Bavarian Forest National Park in applying its current forest inventory method?
2. Methods
2.1. Study Area
2.2. Wildlife Management
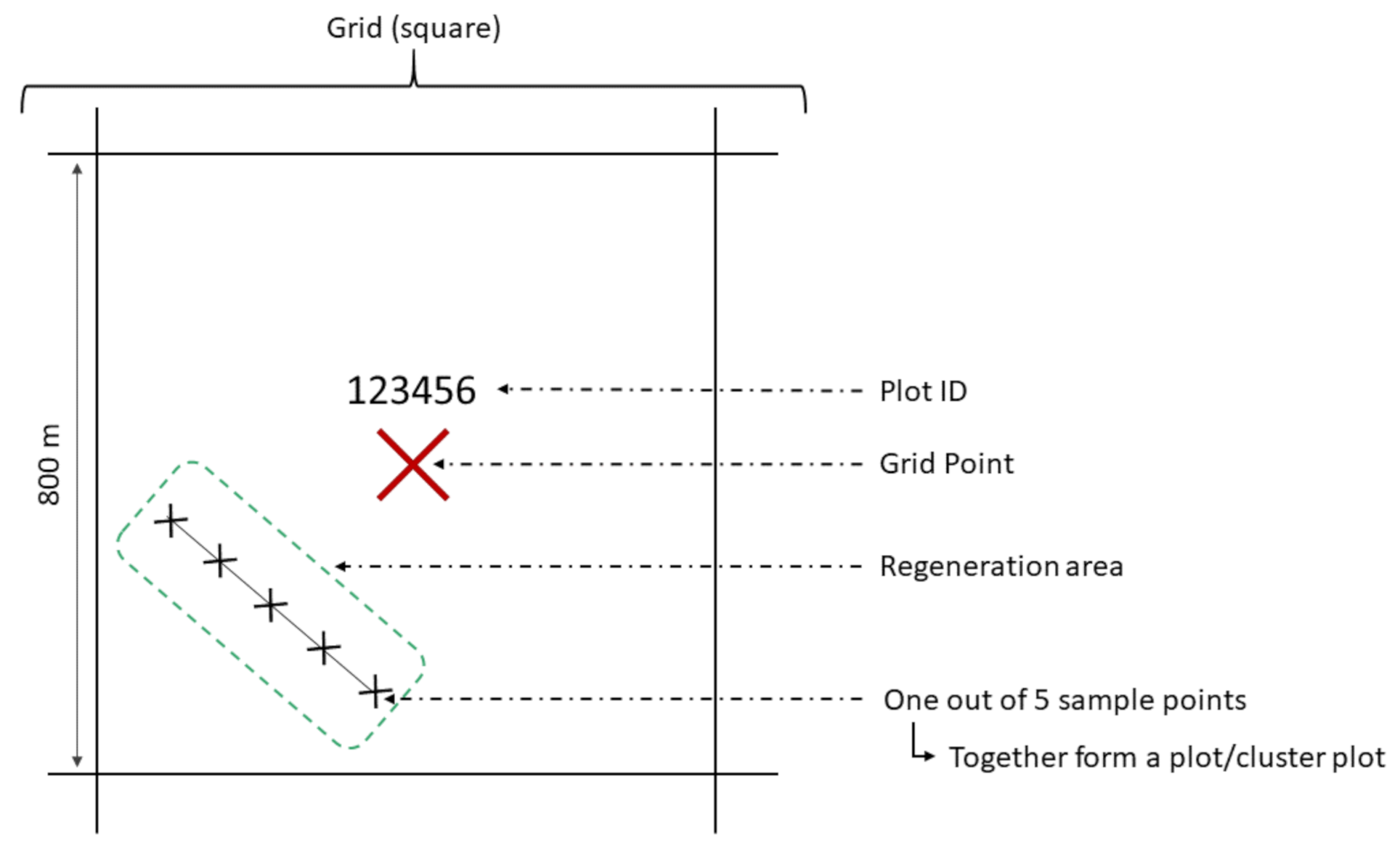
2.3. Inventory Method
2.4. Data Set
2.5. Data Analysis
2.5.1. Browsing Probability and Its Change
| = linear predictor for the browsing probability; | |
| = fixed intercept (the origin in our model); | |
| = cluster-specific (random) deviation from the fixed intercept; | |
| = random intercept for cluster (plot) i; | |
| = fixed slope parameter of covariate (year); | |
| = error term; | |
| = browsing probability. |
2.5.2. Sensitivity Analysis
2.5.3. Simulation of Ungulate Browsing
2.5.4. Computational Details
3. Results
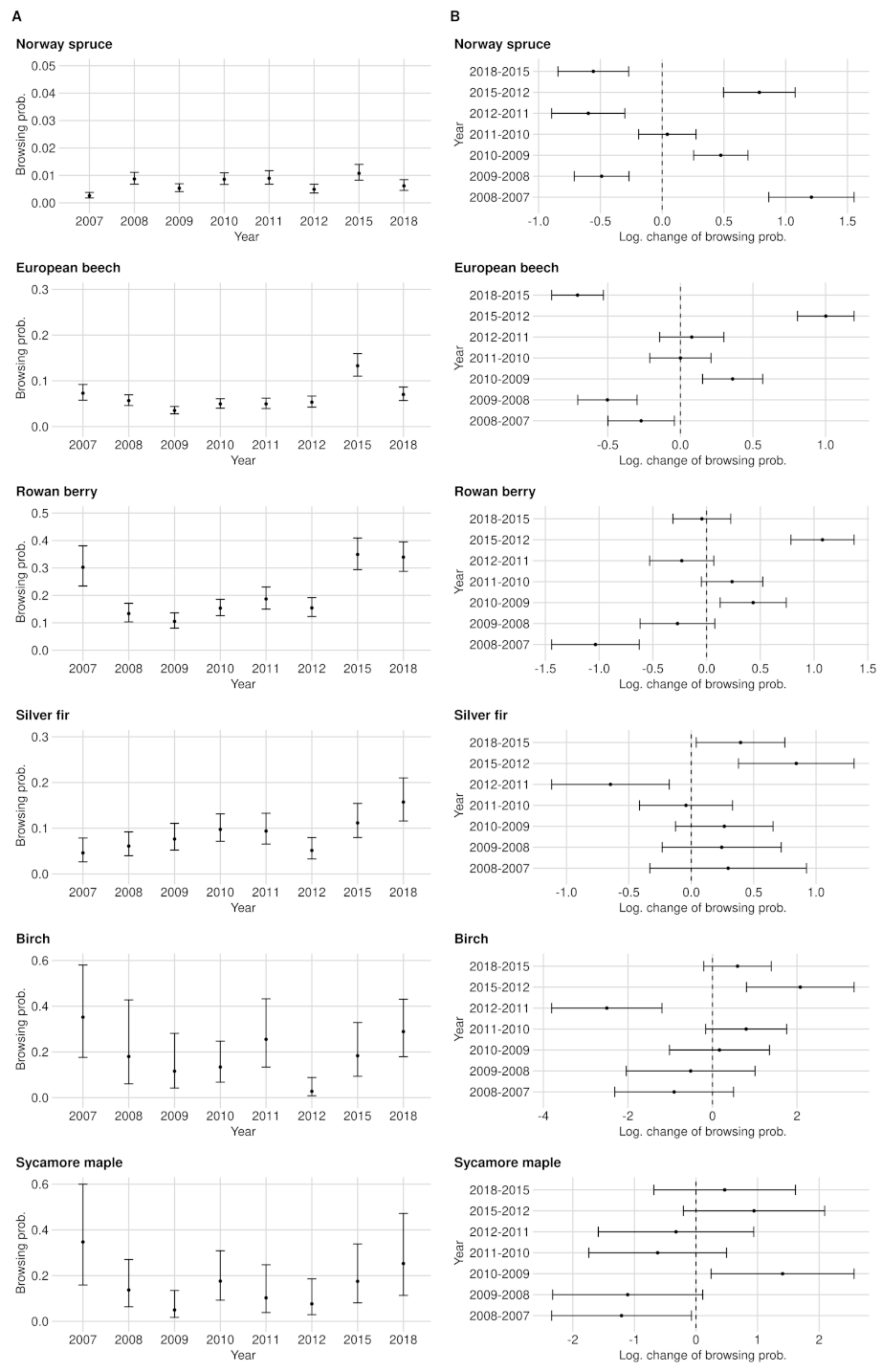
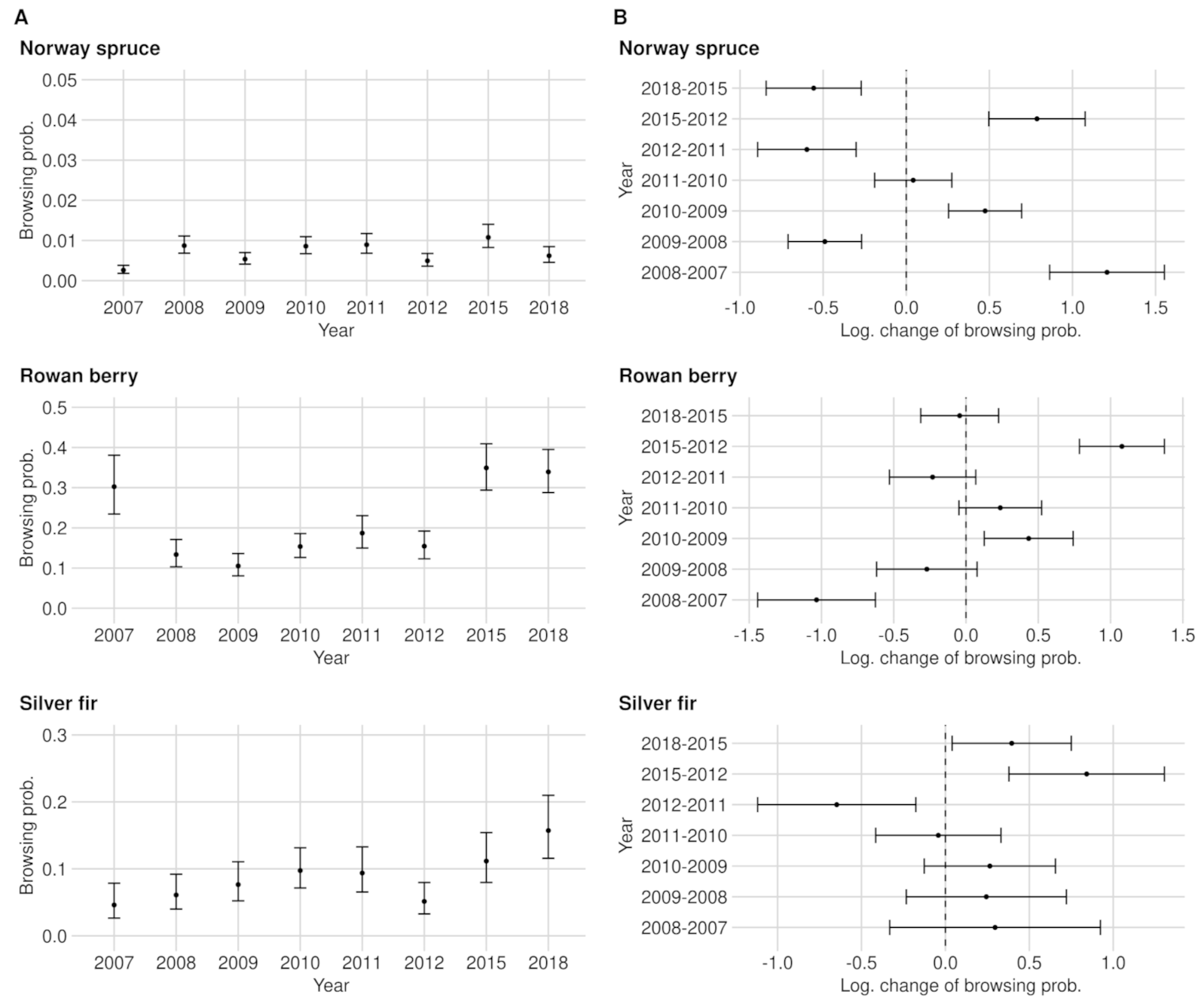
3.1. Browsing Probability Time Series
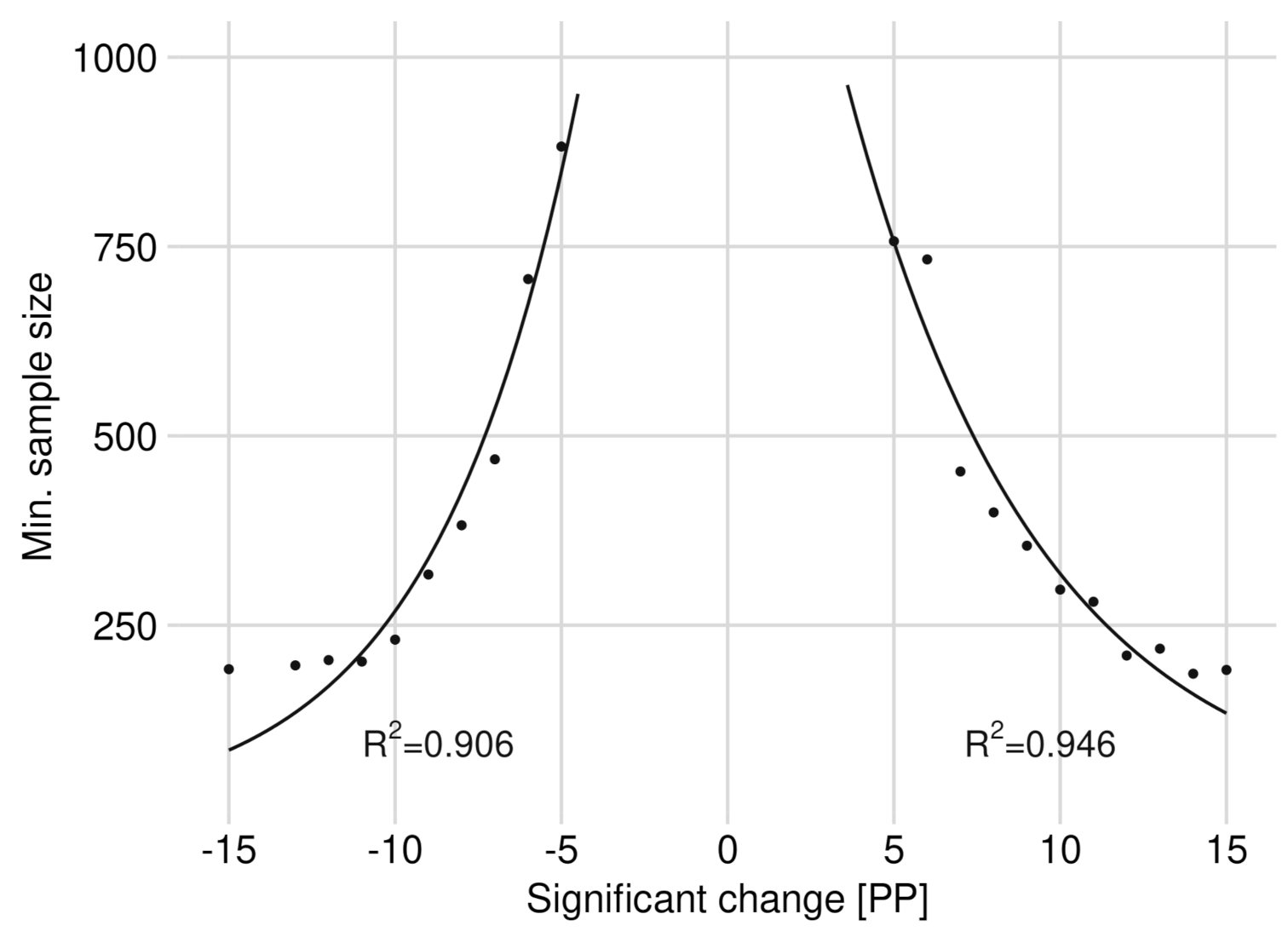
3.2. Significance of Changes and Sample Sizes
3.3. Simulation of Ungulate Browsing on Rowan
4. Discussion
4.1. Evaluating Ungulate Browsing Impact (Question I)
4.2. The Ideal Sample Size and Cost Efficiency (Question II and III)
4.3. Inventory Design and Further Improvements
4.4. Opportunities for Future Research
4.4.1. Which Tree Species Must Undergo a Browsing Change to Trigger an Intervention in the Wildlife Population?
- Act as an “early warning system” that reflects the influence of ungulate browsing of other tree species or, rather, a development of the ungulate densities in time;
- Be preferred by ungulates so that the indicator species sensitively indicates changes (large amplitude) rather than just indicating a change. Ideally, the species should be so sensitive that strong browsing quickly reduces its abundance;
- Be able to be monitored cost-efficiently; due to a high abundance and uniform distribution in the study area, an ideal browsing indicator species achieves a high estimation accuracy in data collection without great effort.
4.4.2. From Which Browsing Probability or Threshold Does the Population of a Tree Species Decrease in the Long Run?
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | Browsing probability; |
| BFNP | Bavarian Forest National Park; |
| RLG | Rachel-Lusen region (in the Bavarian Forest National Park); |
| FRG | Falkenstein-Rachel region (in the Bavarian Forest National Park); |
| NP | National park; |
| Obs. | Observation; |
| PP | Percentage point. |
Appendix A. Detailed Browsing Evaluation of the BFNP
Appendix A.1. Browsing Probability Time Series
Appendix A.1.1. Norway Spruce
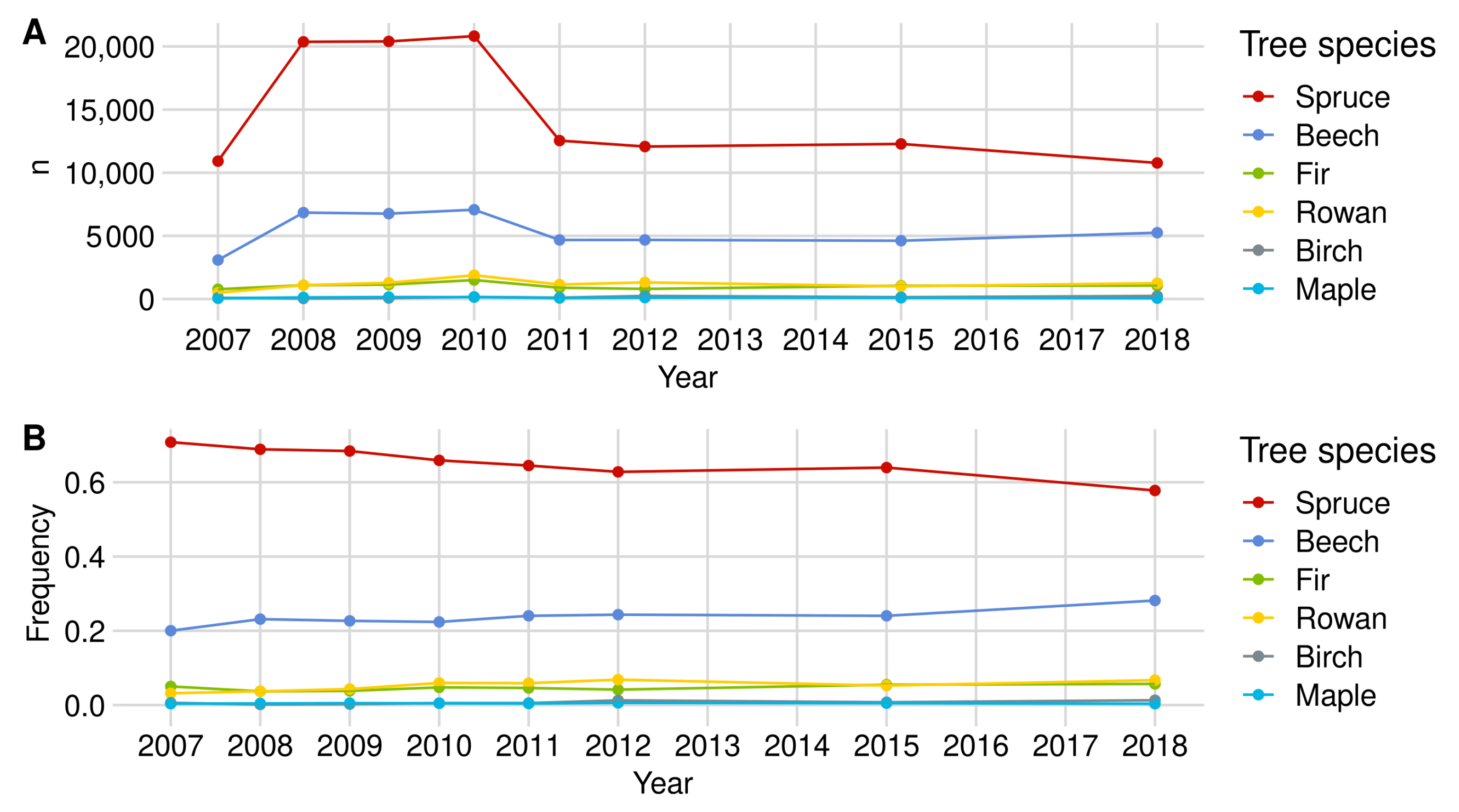
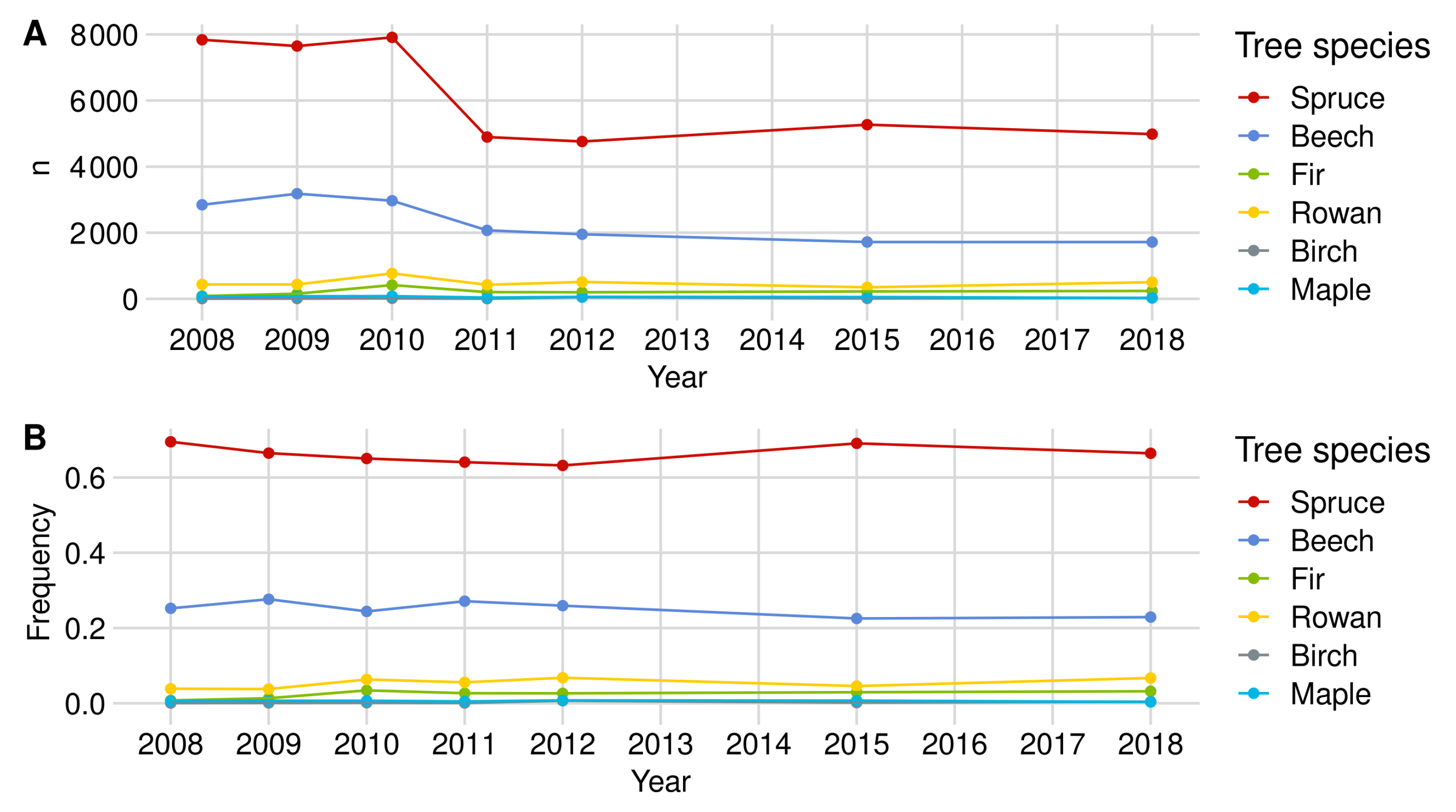
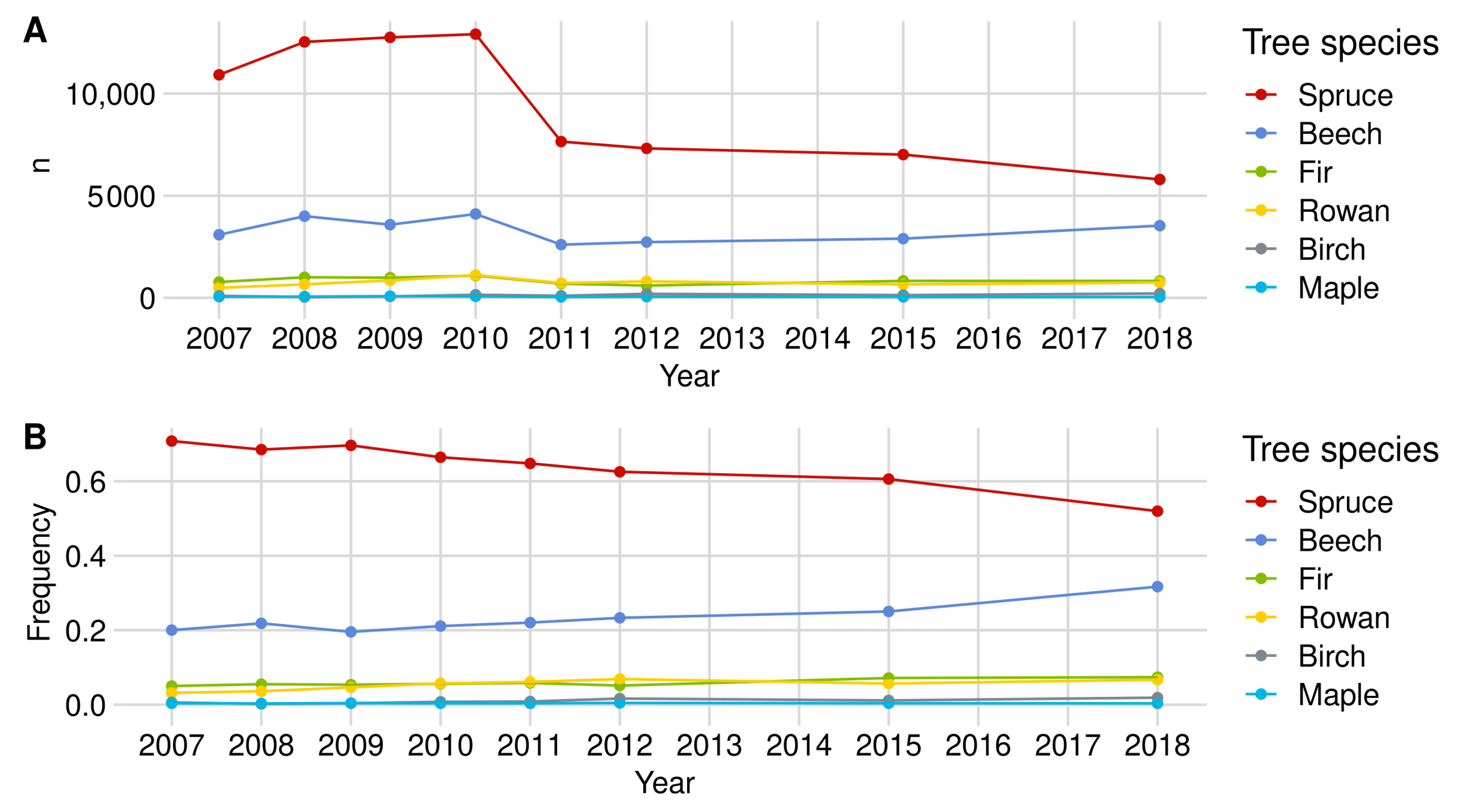
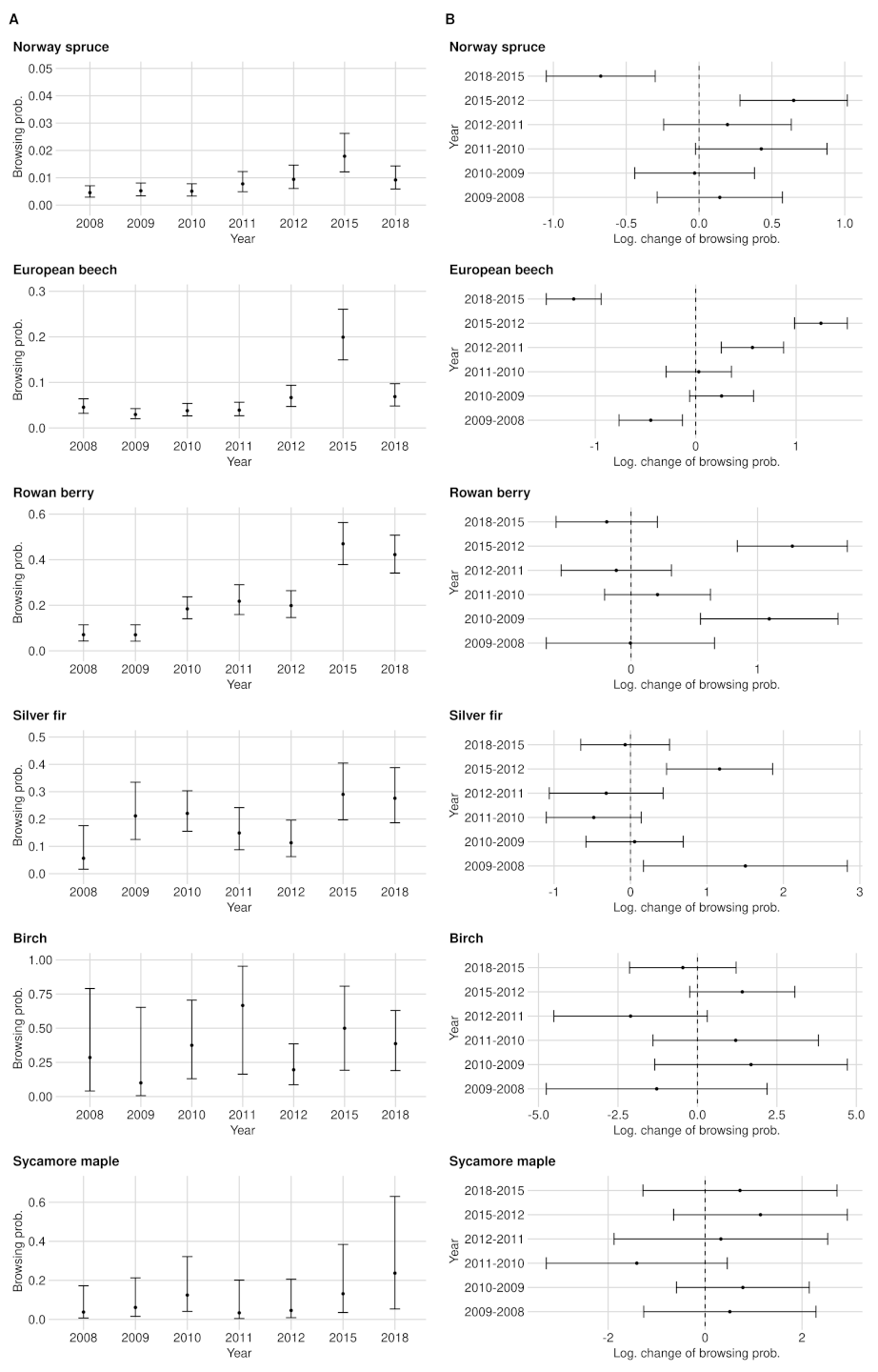
| Norway Spruce | European Beech | Rowan Berry | Silver Fir | Birch | Sycamore Maple | |
|---|---|---|---|---|---|---|
| 2007 | −5.94 *** | −2.54 *** | −0.84 *** | −3.03 *** | −0.61 | |
| 2008 | −4.73 *** | −2.81 *** | −1.87 *** | −2.74 *** | −1.52 *** | −1.84 *** |
| 2009 | −5.22 *** | −3.31 *** | −2.14 *** | −2.49 *** | −2.03 *** | −2.95 *** |
| 2010 | −4.75 *** | −2.95 *** | −1.71 *** | −2.23 *** | −1.87 *** | −1.54 *** |
| 2011 | −4.71 *** | −2.95 *** | −1.47 *** | −2.27 *** | −1.07 *** | −2.17 *** |
| 2012 | −5.31 *** | −2.88 *** | −1.70 *** | −2.92 *** | −3.57 *** | −2.49 *** |
| 2015 | −4.52 *** | −1.87 *** | −0.62 *** | −2.07 *** | −1.49 *** | −1.55 *** |
| 2018 | −5.08 *** | −2.58 *** | −0.67 *** | −1.68 *** | −0.90 *** | −1.08 ** |
| AIC | 16,925.02 | 21,784.74 | ||||
| BIC | 17,012.29 | 21,862.76 | ||||
| Log Likelihood | −8453.51 | −10,883.37 | −4412.72 | −2471.14 | −481.87 | −353.87 |
| Num. obs. | 120,164 | 42,979 | 9474 | 8313 | 1117 | 850 |
| Num. groups: PlotID | 412 | 343 | 370 | 254 | 132 | 76 |
| Var: PlotID (Intercept) |
| Norway Spruce | European Beech | Rowan Berry | Silver Fir | Birch | Sycamore Maple | |
|---|---|---|---|---|---|---|
| 2008–2007 | 1.208 *** | −0.27 * | −1.034 *** | −1.209 * | ||
| 2009–2008 | −0.49 *** | −0.503 *** | ||||
| 2010–2009 | 0.474 *** | 0.36 *** | 0.434 ** | 1.406 ** | ||
| 2011–2010 | ||||||
| 2012–2011 | −0.598 *** | −0.647 ** | −2.499 *** | |||
| 2015–2012 | 0.786 *** | 1.001 *** | 1.079 *** | 0.842 *** | 2.077 *** | |
| 2018–2015 | −0.557 *** | −0.708 *** | 0.395 * | |||
| 2018–2008 | −0.342 ** | 0.229 * | 1.203 *** | 1.056 *** | ||
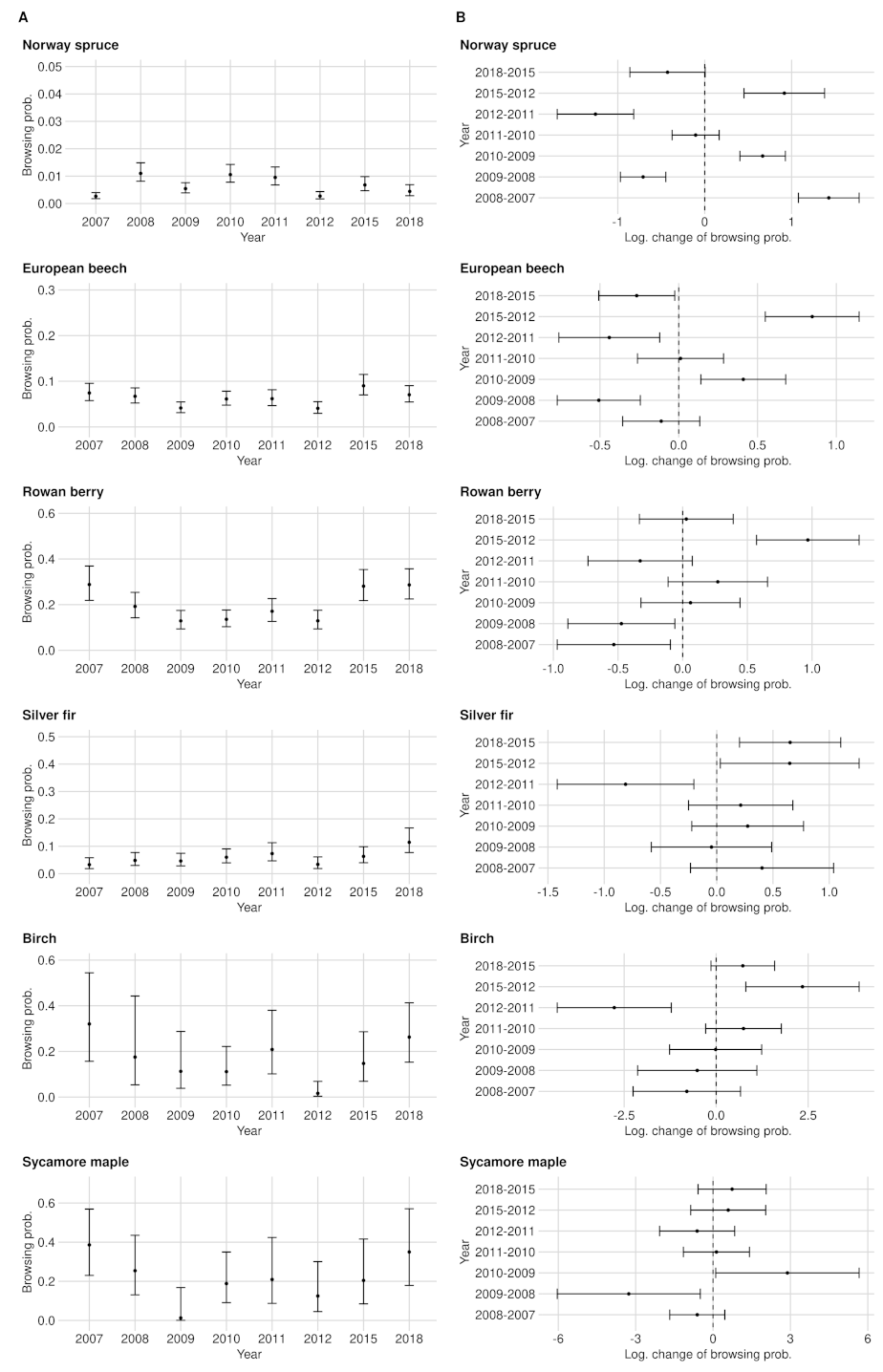
Appendix A.1.2. European Beech
Appendix A.1.3. Rowan Berry
Appendix A.1.4. Silver Fir
Appendix A.1.5. Birch
Appendix A.1.6. Sycamore Maple
Appendix A.2. Significance of Changes and Sample Sizes
Appendix A.2.1. Significant Changes of BP between 2012 and 2015
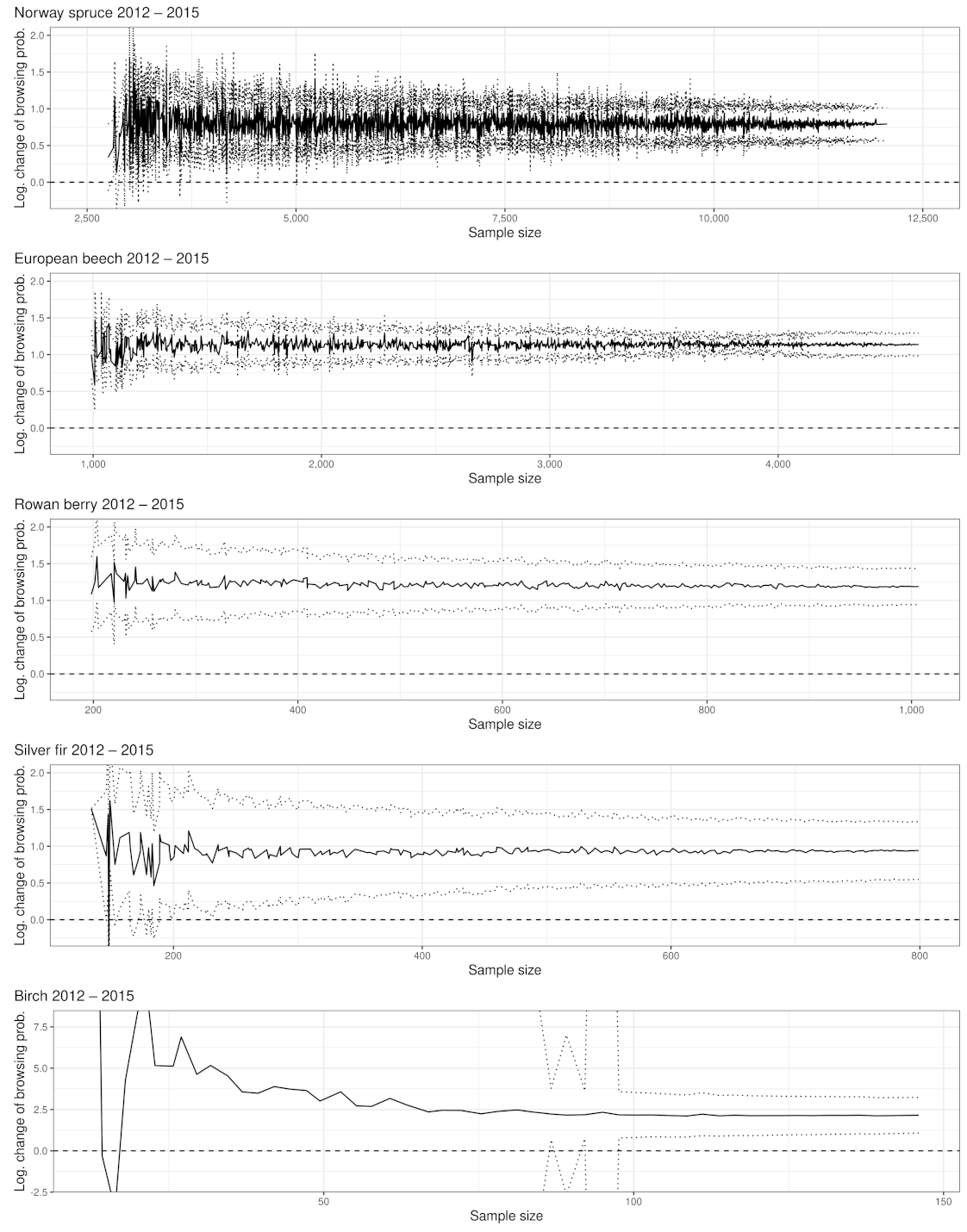
Appendix A.2.2. Significant Changes of BP between 2015 and 2018

Appendix B. Figures
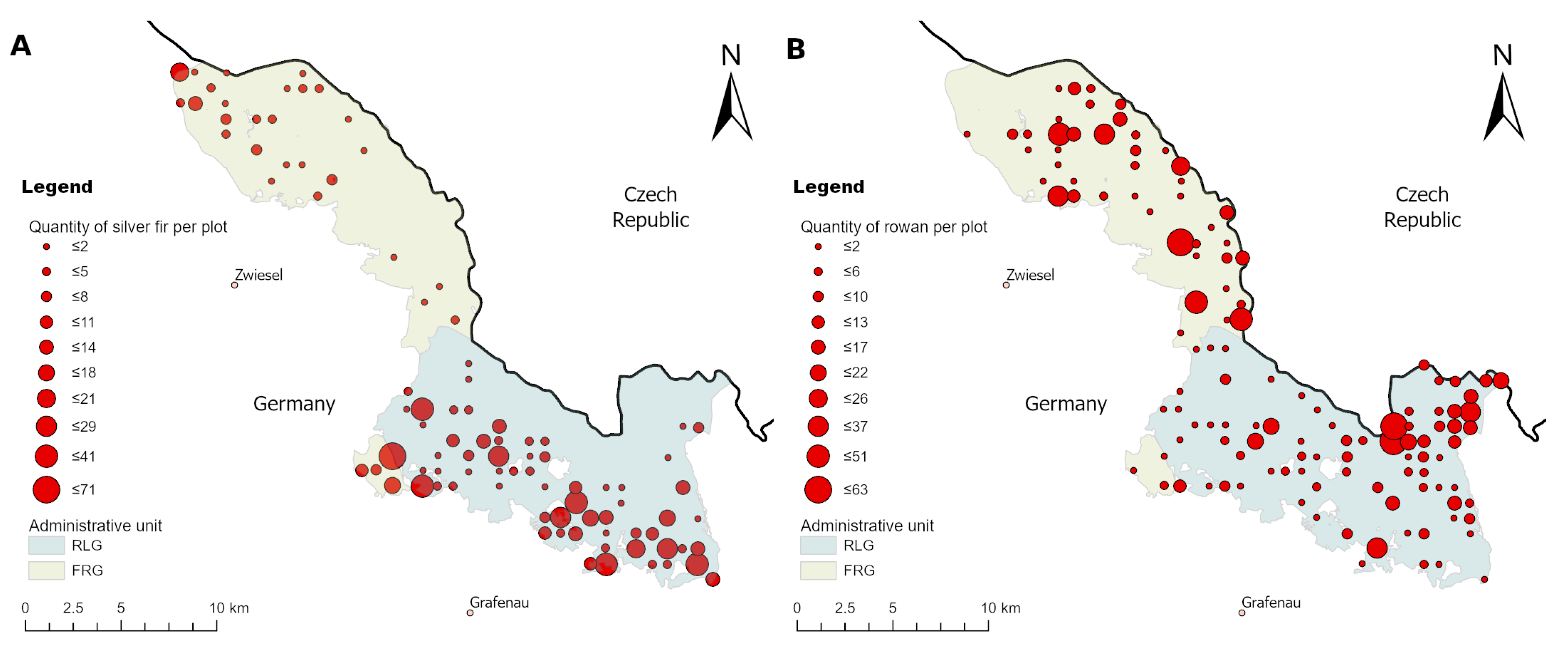
References
- Terborgh, J. Ecological Meltdown in Predator-Free Forest Fragments. Science 2001, 294, 1923–1926. [Google Scholar] [CrossRef]
- Pastor, J.; Cohen, Y. Herbivores, the Functional Diversity of Plants Species, and the Cycling of Nutrients in Ecosystems. Theor. Popul. Biol. 1997, 51, 165–179. [Google Scholar] [CrossRef]
- Gill, R.M.A. A Review of Damage by Mammals in North Temperate Forests: 3. Impact on Trees and Forests. Forestry 1992, 65, 363–388. [Google Scholar] [CrossRef]
- Möst, L.; Hothorn, T.; Müller, J.; Heurich, M. Creating a Landscape of Management: Unintended Effects on the Variation of Browsing Pressure in a National Park. For. Ecol. Manag. 2015, 338, 46–56. [Google Scholar] [CrossRef]
- Peringer, A.; Schulze, K.A.; Stupariu, I.; Stupariu, M.S.; Rosenthal, G.; Buttler, A.; Gillet, F. Multi-Scale Feedbacks between Tree Regeneration Traits and Herbivore Behavior Explain the Structure of Pasture-Woodland Mosaics. Landsc. Ecol. 2016, 31, 913–927. [Google Scholar] [CrossRef]
- Hothorn, T.; Müller, J. Large-Scale Reduction of Ungulate Browsing by Managed Sport Hunting. For. Ecol. Manag. 2010, 260, 1416–1423. [Google Scholar] [CrossRef]
- Fuller, R.J. Responses of Woodland Birds to Increasing Numbers of Deer: A Review of Evidence and Mechanisms. Forestry 2001, 74, 289–298. [Google Scholar] [CrossRef]
- Ammer, C. Impact of Ungulates on Structure and Dynamics of Natural Regeneration of Mixed Mountain Forests in the Bavarian Alps. For. Ecol. Manag. 1996, 88, 43–53. [Google Scholar] [CrossRef]
- Ripple, W.J.; Newsome, T.M.; Wolf, C.; Dirzo, R.; Everatt, K.T.; Galetti, M.; Hayward, M.W.; Kerley, G.I.H.; Levi, T.; Lindsey, P.A.; et al. Collapse of the World’s Largest Herbivores. Sci. Adv. 2015, 1, e1400103. [Google Scholar] [CrossRef]
- Apollonio, M.; Andersen, R.; Putman, R. (Eds.) European Ungulates and Their Management in the 21st Century; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2010. [Google Scholar]
- Bradshaw, R.H.; Hannon, G.E.; Lister, A.M. A Long-Term Perspective on Ungulate–Vegetation Interactions. For. Ecol. Manag. 2003, 181, 267–280. [Google Scholar] [CrossRef]
- Milner, J.M.; Bonenfant, C.; Mysterud, A.; Gaillard, J.M.; Csanyi, S.; Stenseth, N.C. Temporal and Spatial Development of Red Deer Harvesting in Europe: Biological and Cultural Factors. J. Appl. Ecol. 2006, 43, 721–734. [Google Scholar] [CrossRef]
- Linnell, J.D.; Cretois, B.; Nilsen, E.B.; Rolandsen, C.M.; Solberg, E.J.; Veiberg, V.; Kaczensky, P.; Van Moorter, B.; Panzacchi, M.; Rauset, G.R.; et al. The Challenges and Opportunities of Coexisting with Wild Ungulates in the Human-Dominated Landscapes of Europe’s Anthropocene. Biol. Conserv. 2020, 244, 108500. [Google Scholar] [CrossRef]
- Ripple, W.J.; Estes, J.A.; Beschta, R.L.; Wilmers, C.C.; Ritchie, E.G.; Hebblewhite, M.; Berger, J.; Elmhagen, B.; Letnic, M.; Nelson, M.P.; et al. Status and Ecological Effects of the World’s Largest Carnivores. Science 2014, 343. [Google Scholar] [CrossRef]
- Ammer, C.; Vor, T.; Knoke, T.; Wagner, S. Der Wald-Wild-Konflikt: Analyse Und Lösungsansätze Vor Dem Hintergrund Rechtlicher, Ökologischer Und Ökonomischer Zusammenhänge. In Göttinger Forstwissenschaften; Niedersächsische Staats-und Universitätsbibliothek and Univ. Verl. Göttingen: Göttingen, Germany, 2010; Volume 5. [Google Scholar]
- Boulanger, V.; Baltzinger, C.; Saïd, S.; Ballon, P.; Picard, J.F.; Dupouey, J.L. Ranking Temperate Woody Species along a Gradient of Browsing by Deer. For. Ecol. Manag. 2009, 258, 1397–1406. [Google Scholar] [CrossRef]
- Eiberle, K. Über Den Einfluss Des Wildverbisses Auf Die Mortalität von Jungen Waldbäumen in Der Oberen Montanstufe. Schweiz. Z. Forstwes 1989, 140, 1031–1042. [Google Scholar]
- Rooney, T.P. Deer Impacts on Forest Ecosystems: A North American Perspective. Forestry 2001, 74, 201–208. [Google Scholar] [CrossRef]
- Vavra, M.; Parks, C.G.; Wisdom, M.J. Biodiversity, Exotic Plant Species, and Herbivory: The Good, the Bad, and the Ungulate. For. Ecol. Manag. 2007, 246, 66–72. [Google Scholar] [CrossRef]
- Gill, R.M.A. A Review of Damage by Mammals in North Temperate Forests: 1. Deer. Forestry 1992, 65, 145–169. [Google Scholar] [CrossRef]
- Bergquist, J.; Bergström, R.; Zakharenka, A. Responses of Young Norway Spruce (Picea Abies) to Winter Browsing by Roe Deer (Capreolus Capreolus): Effects on Height Growth and Stem Morphology. Scand. J. For. Res. 2003, 18, 368–376. [Google Scholar] [CrossRef]
- Harmer, R. The Effect of Plant Competition and Simulated Summer Browsing by Deer on Tree Regeneration. J. Appl. Ecol. 2001, 38, 1094–1103. [Google Scholar] [CrossRef]
- Kupferschmid, A.D.; Bugmann, H. Ungulate Browsing in Winter Reduces the Growth of Fraxinus and Acer Saplings in Subsequent Unbrowsed Years. Plant Ecol. 2008, 198, 121–134. [Google Scholar] [CrossRef][Green Version]
- Kuijper, D.P.J.; Jędrzejewska, B.; Brzeziecki, B.; Churski, M.; Jędrzejewski, W.; Żybura, H. Fluctuating Ungulate Density Shapes Tree Recruitment in Natural Stands of the Białowieża Primeval Forest, Poland. J. Veg. Sci. 2010, 21, 1082–1098. [Google Scholar] [CrossRef]
- Kupferschmid, A.D.; Bütikofer, L.; Hothorn, T.; Schwyzer, A.; Brang, P. Quantifying the Relative Influence of Terminal Shoot Browsing by Ungulates on Tree Regeneration. For. Ecol. Manag. 2019, 446, 331–344. [Google Scholar] [CrossRef]
- Kupferschmid, A.D.; Bütikofer, L.; Hothorn, T.; Schwyzer, A.; Brang, P. Ungulate Species and Abundance as Well as Environmental Factors Determine the Probability of Terminal Shoot Browsing on Temperate Forest Trees. Forests 2020, 11, 764. [Google Scholar] [CrossRef]
- Côté, S.D.; Rooney, T.P.; Tremblay, J.P.; Dussault, C.; Waller, D.M. Ecological Impacts of Deer Overabundance. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 113–147. [Google Scholar] [CrossRef]
- Stockton, S.A.; Allombert, S.; Gaston, A.J.; Martin, J.L. A Natural Experiment on the Effects of High Deer Densities on the Native Flora of Coastal Temperate Rain Forests. Biol. Conserv. 2005, 126, 118–128. [Google Scholar] [CrossRef]
- Allombert, S.; Gaston, A.J.; Martin, J.L. A Natural Experiment on the Impact of Overabundant Deer on Songbird Populations. Biol. Conserv. 2005, 126, 1–13. [Google Scholar] [CrossRef]
- Popma, J.M.; Nadelhoffer, K.J. Deer Browsing Effects on Temperate Forest Soil Nitrogen Cycling Shift from Positive to Negative across Fertility Gradients. Can. J. For. Res. 2020, 50, 1281–1288. [Google Scholar] [CrossRef]
- Prietzel, J.; Ammer, C. Montane Bergmischwälder Der Bayerischen Kalkalpen: Reduktion Der Schalenwilddichte Steigert Nicht Nur Den Verjüngungserfolg, Sondern Auch Die Bodenfruchtbarkeit. Allg. Forst-Und Jagdztg. 2008, 179, 104–112. [Google Scholar]
- Pastor, J.; Dewey, B.; Naiman, R.J.; McInnes, P.F.; Cohen, Y. Moose Browsing and Soil Fertility in the Boreal Forests of Isle Royale National Park. Ecology 1993, 74, 467–480. [Google Scholar] [CrossRef]
- Garland, G.; Banerjee, S.; Edlinger, A.; Miranda Oliveira, E.; Herzog, C.; Wittwer, R.; Philippot, L.; Maestre, F.T.; Heijden, M.G.A. A Closer Look at the Functions behind Ecosystem Multifunctionality: A Review. J. Ecol. 2021, 109, 600–613. [Google Scholar] [CrossRef]
- Knoke, T.; Messerer, K.; Paul, C. The Role of Economic Diversification in Forest Ecosystem Management. Curr. For. Rep. 2017, 3, 93–106. [Google Scholar] [CrossRef]
- Knoke, T.; Kindu, M.; Jarisch, I.; Gosling, E.; Friedrich, S.; Bödeker, K.; Paul, C. How Considering Multiple Criteria, Uncertainty Scenarios and Biological Interactions May Influence the Optimal Silvicultural Strategy for a Mixed Forest. For. Policy Econ. 2020, 118, 102239. [Google Scholar] [CrossRef]
- Andrus, R.A.; Hart, S.J.; Veblen, T.T. Forest Recovery Following Synchronous Outbreaks of Spruce and Western Balsam Bark Beetle Is Slowed by Ungulate Browsing. Ecology 2020, 101. [Google Scholar] [CrossRef]
- Seidl, R.; Müller, J.; Hothorn, T.; Bässler, C.; Heurich, M.; Kautz, M. Small Beetle, Large-Scale Drivers: How Regional and Landscape Factors Affect Outbreaks of the European Spruce Bark Beetle. J. Appl. Ecol. 2015, 53, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest Disturbances under Climate Change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef]
- Thom, D.; Rammer, W.; Seidl, R. The Impact of Future Forest Dynamics on Climate: Interactive Effects of Changing Vegetation and Disturbance Regimes. Ecol. Monogr. 2017, 87, 665–684. [Google Scholar] [CrossRef]
- Knoke, T.; Gosling, E.; Thom, D.; Chreptun, C.; Rammig, A.; Seidl, R. Economic Losses from Natural Disturbances in Norway Spruce Forests–A Quantification Using Monte-Carlo Simulations. Ecol. Econ. 2021, 185, 107046. [Google Scholar] [CrossRef]
- Clasen, C.; Griess, V.C.; Knoke, T. Financial Consequences of Losing Admixed Tree Species: A New Approach to Value Increased Financial Risks by Ungulate Browsing. For. Policy Econ. 2011, 13, 503–511. [Google Scholar] [CrossRef]
- Imperio, S.; Ferrante, M.; Grignetti, A.; Santini, G.; Focardi, S. Investigating Population Dynamics in Ungulates: Do Hunting Statistics Make up a Good Index of Population Abundance? Wildl. Biol. 2010, 16, 205–214. [Google Scholar] [CrossRef]
- Clasen, C.; Knoke, T. Site Conditions Have an Impact on Compensation Payments for the Loss of Tree Species in Mixed Forests. Forestry 2013, 86, 533–542. [Google Scholar] [CrossRef][Green Version]
- Ward, A.I.; White, P.C.; Smith, A.; Critchley, C.H. Modelling the Cost of Roe Deer Browsing Damage to Forestry. For. Ecol. Manag. 2004, 191, 301–310. [Google Scholar] [CrossRef]
- van Beeck Calkoen, S.T.; Mühlbauer, L.; Andrén, H.; Apollonio, M.; Balčiauskas, L.; Belotti, E.; Carranza, J.; Cottam, J.; Filli, F.; Gatiso, T.T.; et al. Ungulate Management in European National Parks: Why a More Integrated European Policy Is Needed. J. Environ. Manag. 2020, 260, 110068. [Google Scholar] [CrossRef] [PubMed]
- Nationalparkverwaltung Bayerischer Wald. Nationalparkplan: Leitbild Und Ziele; Nationalparkverwaltung Bayerischer Wald: Grafenau, Germany, 2010. [Google Scholar]
- Heurich, M.; Baierl, F.; Günther, S.; Sinner, K.F. Management and Conservation of Large Mammals in the Bavarian Forest National Park. Silva Gabreta 2011, 17, 1–18. [Google Scholar]
- Nichols, J.; Williams, B. Monitoring for Conservation. Trends Ecol. Evol. 2006, 21, 668–673. [Google Scholar] [CrossRef]
- Johnson, B.L. The Role of Adaptive Management as an Operational Approach for Resource Management Agencies. Conserv. Ecol. 1999, 3, 1–11. [Google Scholar] [CrossRef]
- Williams, B.K.; Szaro, R.C.; Shapiro, C.D. Adaptive Management: The US Department of the Interior Technical Guide; Technical Report; US Department of the Interior: Washington, DC, USA, 2009. [Google Scholar]
- Chevrier, T.; Saïd, S.; Widmer, O.; Hamard, J.P.; Saint-Andrieux, C.; Gaillard, J.M. The Oak Browsing Index Correlates Linearly with Roe Deer Density: A New Indicator for Deer Management? Eur. J. Wildl. Res. 2012, 58, 17–22. [Google Scholar] [CrossRef]
- Heurich, M. Ergebnisse der Verjüngungsinventur 2015; Nationalparkverwaltung Bayerischer Wald: Grafenau, Germany, 2015. [Google Scholar]
- Reimoser, F.; Armstrong, H.; Suchant, R. Measuring Forest Damage of Ungulates: What Should Be Considered. For. Ecol. Manag. 1999, 120, 47–58. [Google Scholar] [CrossRef]
- Morellet, N.; Gaillard, J.M.; Hewison, A.J.M.; Ballon, P.; Boscardin, Y.; Duncan, P.; Klein, F.; Maillard, D. Indicators of Ecological Change: New Tools for Managing Populations of Large Herbivores: Ecological Indicators for Large Herbivore Management. J. Appl. Ecol. 2007, 44, 634–643. [Google Scholar] [CrossRef]
- Moog, M. Bewertung von Wildschäden im Wald: Modelle, Methoden, Bewertung, 1st ed.; Neumann-Neudamm: Melsungen, Germany, 2008. [Google Scholar]
- Rüegg, D.; Nigg, H. Mehrstufige Verjüngungskontrollen Und Grenzwerte Für Die Verbissintensität|Comparitive Regeneration Control and Limiting Value of Browsing Damage Intensity. Schweiz. Z. Forstwes. 2003, 154, 314–321. [Google Scholar] [CrossRef][Green Version]
- Huber, M.O.; Schwyzer, A.; Kupferschmid, A.D. A Comparison Between Plot-Count and Nearest-Tree Method in As-Sessing Tree Regeneration Features. Curr. Trends For. Res. 2018, 10, 2638-0013. [Google Scholar]
- Gaillard, J.M.; Duncan, P.; van Wieren, S.E.; Loison, A.; Klein, F.; Maillard, D. Managing Large Herbivores in Theory and Practice: Is the Game the Same for Browsing and Grazing Species. In The Ecology of Browsing and Grazing; Heldmaier, G., Jackson, R.B., Lange, O.L., Mooney, H.A., Schulze, E.D., Sommer, U., Caldwell, M.M., Gordon, I.J., Prins, H.H.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; Volume 195, pp. 293–307. [Google Scholar] [CrossRef]
- Szwagrzyk, J.; Gazda, A.; Muter, E.; Pielech, R.; Szewczyk, J.; Zięba, A.; Zwijacz-Kozica, T.; Wiertelorz, A.; Pachowicz, T.; Bodziarczyk, J. Effects of Species and Environmental Factors on Browsing Frequency of Young Trees in Mountain Forests Affected by Natural Disturbances. For. Ecol. Manag. 2020, 474, 118364. [Google Scholar] [CrossRef]
- Senn, J.; Suter, W. Ungulate Browsing on Silver Fir (Abies Alba) in the Swiss Alps: Beliefs in Search of Supporting Data. For. Ecol. Manag. 2003, 181, 151–164. [Google Scholar] [CrossRef]
- Reimoser, F.; Gossow, H. Impact of Ungulates on Forest Vegetation and Its Dependence on the Silvicultural System. For. Ecol. Manag. 1996, 88, 107–119. [Google Scholar] [CrossRef]
- Hagen, R.; Kühl, N.; Hanewinkel, M.; Suchant, R. Number and Height of Unbrowsed Saplings Are More Appropriate than the Proportion of Browsed Saplings for Predicting Silvicultural Regeneration Success. Ann. For. Sci. 2021, 78, 21. [Google Scholar] [CrossRef]
- Kennel, E. Die Überlebensprognose: Statistische Wertung von Verbissbefunden. AFZ-Der Wald 2003, 25, 1302–1306. [Google Scholar]
- Clasen, C.; Heurich, M.; Glaesener, L.; Kennel, E.; Knoke, T. What Factors Affect the Survival of Tree Saplings under Browsing, and How Can a Loss of Admixed Tree Species Be Forecast? Ecol. Model. 2015, 305, 1–9. [Google Scholar] [CrossRef]
- Eiberle, K.; Nigg, H. Grundlagen Zur Beurteilung Des Wildverbisses Im Gebirgswald. Schweiz. Z. Forstwes. 1987, 138, 747–785. [Google Scholar]
- Odermatt, O. Zur Bewertung von Wildverbiss. Die Methode Eiberle. Schweiz. Z. Forstwes 1996, 147, 177–199. [Google Scholar]
- Rooney, T.P.; Gross, K. A Demographic Study of Deer Browsing Impacts on Trillium Grandiflorum. Plant Ecol. 2003, 168, 267–277. [Google Scholar] [CrossRef]
- Edenius, L.; Ericsson, G. Effects of Ungulate Browsing on Recruitment of Aspen and Rowan: A Demographic Approach. Scand. J. For. Res. 2015, 1–6. [Google Scholar] [CrossRef]
- Richardson, S.; Mill, A.C.; Davis, D.; Jam, D.; Ward, A.I. A Systematic Review of Adaptive Wildlife Management for the Control of Invasive, Non-native Mammals, and Other Human–Wildlife Conflicts. Mammal Rev. 2020, 50, 147–156. [Google Scholar] [CrossRef]
- Polley, H. Survey Instructions for the 3rd National Forest Inventory (2011–2012) 2nd Revised Version, May 2011 with 4. Corrigendum (21.03. 2014); Bundesministerium Für Ernährung, Landwirtschaft Und Verbraucherschutz Ref. 535; Federal Ministry of Food; Agriculture, and Consumer Protection: Bonn, Germany, 2011. [Google Scholar]
- McWilliams, W.H.; Westfall, J.A.; Brose, P.H.; Dey, D.C.; Hatfield, M.; Johnson, K.; Laustsen, K.M.; Lehman, S.L.; Morin, R.S.; Nelson, M.D.; et al. A Regeneration Indicator for Forest Inventory and Analysis: History, Sampling, Estimation, Analytics, and Potential Use in the Midwest and Northeast United States. Gen. Tech. Rep. NRS-148. Newt. Sq. PA US Dep. Agric. For. Serv. North. Res. Station. 2015, 148, 1–74. [Google Scholar] [CrossRef]
- Holmström, H.; Kallur, H.; Ståhl, G. Cost-plus-Loss Analyses of Forest Inventory Strategies Based on kNN-Assigned Reference Sample Plot Data. Silva Fenn. 2003, 37. [Google Scholar] [CrossRef]
- Kangas, A.S.; Horne, P.; Leskinen, P. Measuring the Value of Information in Multicriteria Decisionmaking. For. Sci. 2010, 56, 558–566. [Google Scholar]
- Cochran, W.G. Sampling Techniques, 3rd ed.; Wiley Series in Probability and Mathematical Statistics; Wiley: New York, NY, USA, 1977. [Google Scholar]
- Knoke, T.; Kindu, M.; Schneider, T.; Gobakken, T. Inventory of Forest Attributes to Support the Integration of Non-Provisioning Ecosystem Services and Biodiversity into Forest Planning—From Collecting Data to Providing Information. Curr. For. Rep. 2021, 7, 38–58. [Google Scholar] [CrossRef]
- Grussu, G.; Testolin, R.; Saulei, S.; Farcomeni, A.; Yosi, C.K.; De Sanctis, M.; Attorre, F. Optimum Plot and Sample Sizes for Carbon Stock and Biodiversity Estimation in the Lowland Tropical Forests of Papua New Guinea. Forestry 2016, 89, 150–158. [Google Scholar] [CrossRef]
- Guadagnoli, E.; Velicer, W.F. Relation of Sample Size to the Stability of Component Patterns. Psychol. Bull. 1988, 103, 265–275. [Google Scholar] [CrossRef]
- Heurich, M.; Neufanger, M. Die Wälder des Nationalparks Bayerischer Wald: Ergebnisse der Waldinventur 2002/2003 Im Geschichtlichen Und Waldökologischen Kontext; Nationalpark Bayerischer Wald, Nationalparkverwaltung Bayerischer Wald: Grafenau, Germany, 2005; Volume 16. [Google Scholar]
- Heurich, M.; Reinelt, A.; Fahse, L. Waldentwicklung Im Bergwald Nach Windwurf Und Borkenkäferbefall: Die Buchdruckermassenvermehrung Im Nationalpark Bayerischer Wald. Wiss. Reihe Bayrischen Staatsforstverwalt. 2001, 14, 9–48. [Google Scholar]
- Heurich, M.; Englmaier, K.H. The Development of Tree Species Composition in the Rachel–Lusen Region of the Bavarian Forest National Park. Silva Gabreta 2010, 16, 165–186. [Google Scholar]
- Stalling, H. Untersuchungen zur Spät- und Postglazialen Vegetationsgeschichte Im Bayerischen Wald. Ph.D. Thesis, Georg-August-Universität Göttingen, Göttingen, Germany, 1987. [Google Scholar]
- Sommerfeld, A.; Rammer, W.; Heurich, M.; Hilmers, T.; Müller, J.; Seidl, R. Do Bark Beetle Outbreaks Amplify or Dampen Future Bark Beetle Disturbances in Central Europe? J. Ecol. 2020, 734–749. [Google Scholar] [CrossRef]
- van der Knaap, W.O.; van Leeuwen, J.F.N.; Fahse, L.; Szidat, S.; Studer, T.; Baumann, J.; Heurich, M.; Tinner, W. Vegetation and Disturbance History of the Bavarian Forest National Park, Germany. Veg. Hist. Archaeobotany 2020, 29, 277–295. [Google Scholar] [CrossRef]
- Wermelinger, B. Ecology and Management of the Spruce Bark Beetle Ips Typographus—A Review of Recent Research. For. Ecol. Manag. 2004, 202, 67–82. [Google Scholar] [CrossRef]
- Bače, R.; Svoboda, M.; Pouska, V.; Janda, P.; Červenka, J. Natural Regeneration in Central-European Subalpine Spruce Forests: Which Logs Are Suitable for Seedling Recruitment? For. Ecol. Manag. 2012, 266, 254–262. [Google Scholar] [CrossRef]
- Zeppenfeld, T.; Svoboda, M.; DeRose, R.J.; Heurich, M.; Müller, J.; Čížková, P.; Starý, M.; Bače, R.; Donato, D.C. Response of Mountain Picea Abies Forests to Stand-Replacing Bark Beetle Outbreaks: Neighbourhood Effects Lead to Self-Replacement. J. Appl. Ecol. 2015, 52, 1402–1411. [Google Scholar] [CrossRef]
- Moser, B.; Schütz, M.; Hindenlang, K.E. Importance of Alternative Food Resources for Browsing by Roe Deer on Deciduous Trees: The Role of Food Availability and Species Quality. For. Ecol. Manag. 2006, 226, 248–255. [Google Scholar] [CrossRef]
- Ewald, J.; Braun, L.; Zeppenfeld, T.; Jehl, H.; Heurich, M. Estimating the Distribution of Forage Mass for Ungulates from Vegetation Plots in Bavarian Forest National Park. Tuexenia 2014, 34, 53–70. [Google Scholar] [CrossRef]
- Nationalparkverwaltung Bayerischer Wald. Jahresbericht 2017; Nationalparkverwaltung Bayerischer Wald: Grafenau, Germany, 2017. [Google Scholar]
- Gerner, J.; Heurich, M.; Günther, S.; Schraml, U. Red Deer at a Crossroads—An Analysis of Communication Strategies Concerning Wildlife Management in the ‘Bayerischer Wald’ National Park, Germany. J. Nat. Conserv. 2011, 19, 319–326. [Google Scholar] [CrossRef]
- Kennel, E. Folgerungen Aus Den Ergebnissen Der Verbissinventur in Bayern 1989. Allg. Forstz. 1989, 44, 1100–1101. [Google Scholar]
- Kennel, E. Was Kann Das Vegetationsgutachten Zum Nachhaltigen Management Eines Waldverträglichen Schalenwildbestandes Leisten? Ber. ANL 1998, 22, 51–58. [Google Scholar]
- Bayerische Forstverwaltung. Anweisung für die Erstellung Forstliches Gutachten zur Situation der Waldverjüngung 2018; Bayerisches Staatsministerium für Ernährung, Landwirtschaft und Forsten: München, Germany, 2018. [Google Scholar]
- Bayerische Forstverwaltung. Anweisung Für das Stichprobenverfahren zur Erfassung der Seltenen Baumarten und Erstellung der Revierweisen Aussagen zur Verbisssituation; Bayerisches Staatsministerium für Ernährung, Landwirtschaft und Forsten: München, Germany, 2009. [Google Scholar]
- Zeppenfeld, T. Evaluierung Der Sonderaufnahme: Seltene Baumarten in Der Verbissinventur—Auswirkungen Auf Den Stichprobenumfang. (Unpublished).
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Fahrmeir, L.; Kneib, T.; Lang, S.; Marx, B. Regression: Models, Methods and Applications; Springer: Berlin, Germany, 2013. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4. J. Stat. Softw. 2015, 67. [Google Scholar] [CrossRef]
- Hedeker, D.; Gibbons, R.D.; Flay, B.R. Random-Effects Regression Models for Clustered Data with an Example from Smoking Prevention Research. J. Consult. Clin. Psychol. 1994, 62, 757–765. [Google Scholar] [CrossRef]
- Neuhaus, J.M.; Kalbfleisch, J.D. Between-and within-Cluster Covariate Effects in the Analysis of Clustered Data. Biometrics 1998, 54, 638–645. [Google Scholar] [CrossRef]
- Bretz, F.; Hothorn, T.; Westfall, P. Multiple Comparison Procedures in Linear Models. In COMPSTAT 2008; Brito, P., Ed.; Physica-Verlag HD: Heidelberg, Germany, 2008; pp. 423–431. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4. [Google Scholar] [CrossRef]
- Cederlund, G.; Bergqvist, J.; Kjellander, P.; Gill, R.; Gaillard, J.; Boisaubert, B.; Ballon, P.; Duncan, P. Managing Roe Deer and Their Impact on the Environment: Maximising the Net Benefits to Society. In The European Roe Deer: The Biology of Success; Scandinavian University Press: Oslo, Norway, 1998; Volume 376, pp. 337–372. [Google Scholar]
- McRoberts, R.E.; Tomppo, E.O. Remote Sensing Support for National Forest Inventories. Remote Sens. Environ. 2007, 110, 412–419. [Google Scholar] [CrossRef]
- Carignan, V.; Villard, M.A. Selecting Indicator Species to Monitor Ecological Integrity: A Review. Environ. Monit. Assess. 2002, 78, 45–61. [Google Scholar] [CrossRef]
- Killip, S.; Mahfoud, Z.; Pearce, K. What Is an Intracluster Correlation Coefficient? Crucial Concepts for Primary Care Researchers. Ann. Fam. Med. 2004, 2, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Sokal, R.; Oden, N. Spatial Autocorrelation in Biology: 1. Methodology. Biol. J. Linn. Soc. 1978, 10, 199–228. [Google Scholar] [CrossRef]
- Agresti, A.; Coull, B.A. Approximate Is Better than “Exact” for Interval Estimation of Binomial Proportions. Am. Stat. 1998, 52, 119–126. [Google Scholar] [CrossRef]
- Hoen, H.F.; Solberg, B. Potential and Economic Efficiency of Carbon Sequestration in Forest Biomass Through Silvicultural Management. For. Sci. 1994, 40, 429–451. [Google Scholar] [CrossRef]
- von Gadow, K. Forsteinrichtung: Analyse und Entwurf der Waldentwicklung; Universitätsdrucke, Univ. Verl. Göttingen: Göttingen, Germany, 2005. [Google Scholar]
- Motta, R. Ungulate Impact on Rowan (Sorbus aucuparia L.) and Norway Spruce (Picea abies (L.) Karst.) Height Structure in Mountain Forests in the Eastern Italian Alps. For. Ecol. Manag. 2003, 181, 139–150. [Google Scholar] [CrossRef]
- Prodan, M. Punktstichprobe Fur Die Forsteinrichtung. Forst. Holzwirt. 1968, 23, 225–226. [Google Scholar]
- Kleinn, C.; Vilčko, F. A New Empirical Approach for Estimation in K-Tree Sampling. For. Ecol. Manag. 2006, 237, 522–533. [Google Scholar] [CrossRef]
- Staupendahl, K. Die Modifizierte 6-Baum-Stichprobe–Ein Geeignetes Verfahren Zur Erfassung von Waldbeständen. Allg. Forst. J. Ztg. 2008, 179, 21–33. [Google Scholar]
- Lynch, T.B.; Rusydi, R. Distance Sampling for Forest Inventory in Indonesian Teak Plantations. For. Ecol. Manag. 1999, 113, 215–221. [Google Scholar] [CrossRef]
- Picard, N.; Kouyaté, A.M.; Dessard, H. Tree Density Estimations Using a Distance Method in Mali Savanna. For. Sci. 2005, 51, 7–18. [Google Scholar] [CrossRef]
- Mandallaz, D. Sampling Techniques for Forest Inventories; Applied Environmental Statistics; Chapman and Hall/CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Kleinn, C.; Vilčko, F. Design-Unbiased Estimation for Point-to-Tree Distance Sampling. Can. J. For. Res. 2006, 36, 1407–1414. [Google Scholar] [CrossRef]
- Kuijper, D.; Cromsigt, J.; Churski, M.; Adam, B.; Jędrzejewska, B.; Jędrzejewski, W. Do Ungulates Preferentially Feed in Forest Gaps in European Temperate Forest? For. Ecol. Manag. 2009, 258, 1528–1535. [Google Scholar] [CrossRef]
- Churski, M.; Bubnicki, J.W.; Je, B. Brown World Forests: Increased Ungulate Browsing Keeps Temperate Trees in Recruitment Bottlenecks in Resource Hotspots. New Phytol. 2016, 214, 158–168. [Google Scholar] [CrossRef]
- Bergquist, J.; Löf, M.; Örlander, G. Effects of Roe Deer Browsing and Site Preparation on Performance of Planted Broadleaved and Conifer Seedlings When Using Temporary Fences. Scand. J. For. Res. 2009, 24, 308–317. [Google Scholar] [CrossRef]
- Harmer, R. Survival and New Shoot Production by Artificially Browsed Seedlings of Ash, Beech, Oak and Sycamore Grown under Different Levels of Shade. For. Ecol. Manag. 1999, 116, 39–50. [Google Scholar] [CrossRef]
- Kuijper, D.P.J.; Cromsigt, J.P.G.M.; Jędrzejewska, B.; Miścicki, S.; Churski, M.; Jędrzejewski, W.; Kweczlich, I. Bottom-up versus Top-down Control of Tree Regeneration in the Białowieża Primeval Forest, Poland. J. Ecol. 2010, 98, 888–899. [Google Scholar] [CrossRef]
- Long, Z.T.; Pendergast, T.H.; Carson, W.P. The Impact of Deer on Relationships between Tree Growth and Mortality in an Old-Growth Beech-Maple Forest. For. Ecol. Manag. 2007, 252, 230–238. [Google Scholar] [CrossRef]
- Čermák, P.; Horsák, P.; Špiřík, M.; Mrkva, R. Relationships between Browsing Damage and Woody Species Dominance. J. For. Sci. 2009, 55, 23–31. [Google Scholar] [CrossRef]
- Knoke, T.; Paul, C.; Rammig, A.; Gosling, E.; Hildebrandt, P.; Härtl, F.; Peters, T.; Richter, M.; Diertl, K.H.; Castro, L.M.; et al. Accounting for Multiple Ecosystem Services in a Simulation of Land-use Decisions: Does It Reduce Tropical Deforestation? Glob. Chang. Biol. 2020, 26, 2403–2420. [Google Scholar] [CrossRef]
- Eiberle, K.; Nigg, H. Criteria for Permissible Browse Impact on Sycamore Maple (Acer Pseudoplatanus) in Mountain Forests. Experientia 1987, 43, 127–133. [Google Scholar] [CrossRef]
- Rentch, J.S.; Fortney, R.H.; Stephenson, S.L.; Adams, H.S.; Grafton, W.N.; Anderson, J.T. Vegetation-Site Relationships of Roadside Plant Communities in West Virginia, USA. J. Appl. Ecol. 2005, 42, 129–138. [Google Scholar] [CrossRef]
- Bakker, J.D. Increasing the Utility of Indicator Species Analysis. J. Appl. Ecol. 2008, 45, 1829–1835. [Google Scholar] [CrossRef]
- Anderson, R.C. Height of White-Flowered Trillium (Trillium Grandiflorum) as an Index of Deer Browsing Intensity. Ecol. Appl. 1994, 4, 104–109. [Google Scholar] [CrossRef]
- Kremen, C. Assessing the Indicator Properties of Species Assemblages for Natural Areas Monitoring. Ecol. Appl. Publ. Ecol. Soc. Am. 1992, 2, 203–217. [Google Scholar] [CrossRef]
- Williams, B.K.; Nichols, J.D.; Conroy, M.J. Analysis and Management of Animal Populations; Elsevier Science: Burlington, MA, USA, 2002. [Google Scholar]
- Leder, B. Die Vogelbeere–Eine Unterschätzte Baumart Unserer Mittelgebirge. In Beiträge zur Vogelbeere; Bayerische Landesanstalt für Wald und Forstwirtschaft, Ed.; Berichte Aus Der Bayerischen Landesanstalt Für Wald- Und Forstwirtschaft: Freising, Germany, 1998. [Google Scholar]
- Walters, M.B.; Farinosi, E.J.; Willis, J.L. Deer Browsing and Shrub Competition Set Sapling Recruitment Height and Interact with Light to Shape Recruitment Niches for Temperate Forest Tree Species. For. Ecol. Manag. 2020, 467, 118134. [Google Scholar] [CrossRef]
- Ammer, C.; Stimm, B.; Mosandl, R. Ontogenetic Variation in the Relative Influence of Light and Belowground Resources on European Beech Seedling Growth. Tree Physiol. 2008, 28, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Pépin, D.; Renaud, P.C.; Boscardin, Y.; Goulard, M.; Mallet, C.; Anglard, F.; Ballon, P. Relative Impact of Browsing by Red Deer on Mixed Coniferous and Broad-Leaved Seedlings—An Enclosure-Based Experiment. For. Ecol. Manag. 2006, 222, 302–313. [Google Scholar] [CrossRef]
- Čermák, P. Browsing and Peeling Damage on Woody Plants in the Czech Republic. In Folia Forestalia Bohemica: Sborník Původních Vědeckých Prací a Monografií; Lesnická práce: Kostelec nad Černými lesy, Czech Republic, 2007; Volume 6. [Google Scholar]
- Wallgren, M.; Bergström, R.; Bergqvist, G.; Olsson, M. Spatial Distribution of Browsing and Tree Damage by Moose in Young Pine Forests, with Implications for the Forest Industry. For. Ecol. Manag. 2013, 305, 229–238. [Google Scholar] [CrossRef]
- Connell, J.H. Apparent versus “Real” Competition in Plants. In Perspectives on Plant Competition; Academic Press: New York, NY, USA, 1990; pp. 9–26. [Google Scholar] [CrossRef]
- Seagle, S.W.; Liang, S.Y. Application of a Forest Gap Model for Prediction of Browsing Effects on Riparian Forest Succession. Ecol. Model. 2001, 144, 213–229. [Google Scholar] [CrossRef]
- De Jager, N.R.; Drohan, P.J.; Miranda, B.M.; Sturtevant, B.R.; Stout, S.L.; Royo, A.A.; Gustafson, E.J.; Romanski, M.C. Simulating Ungulate Herbivory across Forest Landscapes: A Browsing Extension for LANDIS-II. Ecol. Model. 2017, 350, 11–29. [Google Scholar] [CrossRef]
- Seidl, R.; Rammer, W.; Scheller, R.M.; Spies, T.A. An Individual-Based Process Model to Simulate Landscape-Scale Forest Ecosystem Dynamics. Ecol. Model. 2012, 231, 87–100. [Google Scholar] [CrossRef]
- Jorritsma, I.; van Hees, A.; Mohren, G. Forest Development in Relation to Ungulate Grazing: A Modeling Approach. For. Ecol. Manag. 1999, 120, 23–34. [Google Scholar] [CrossRef]
- Weisberg, P.J.; Bonavia, F.; Bugmann, H. Modeling the Interacting Effects of Browsing and Shading on Mountain Forest Tree Regeneration (Picea Abies). Ecol. Model. 2005, 185, 213–230. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bödeker, K.; Ammer, C.; Knoke, T.; Heurich, M. Determining Statistically Robust Changes in Ungulate Browsing Pressure as a Basis for Adaptive Wildlife Management. Forests 2021, 12, 1030. https://doi.org/10.3390/f12081030
Bödeker K, Ammer C, Knoke T, Heurich M. Determining Statistically Robust Changes in Ungulate Browsing Pressure as a Basis for Adaptive Wildlife Management. Forests. 2021; 12(8):1030. https://doi.org/10.3390/f12081030
Chicago/Turabian StyleBödeker, Kai, Christian Ammer, Thomas Knoke, and Marco Heurich. 2021. "Determining Statistically Robust Changes in Ungulate Browsing Pressure as a Basis for Adaptive Wildlife Management" Forests 12, no. 8: 1030. https://doi.org/10.3390/f12081030
APA StyleBödeker, K., Ammer, C., Knoke, T., & Heurich, M. (2021). Determining Statistically Robust Changes in Ungulate Browsing Pressure as a Basis for Adaptive Wildlife Management. Forests, 12(8), 1030. https://doi.org/10.3390/f12081030








