Compost and PGP-Based Biostimulant as Alternative to Peat and NPK Fertilization in Chestnut (Castanea Sativa Mill.) Nursery Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Compost and Biostimulant
2.2. Experimental Design
2.3. Morpho-Physiological Parameters of Seedlings
2.4. Water Deficit Assay
2.5. Cultivable Microorganisms
2.6. Statistical Analysis
3. Results
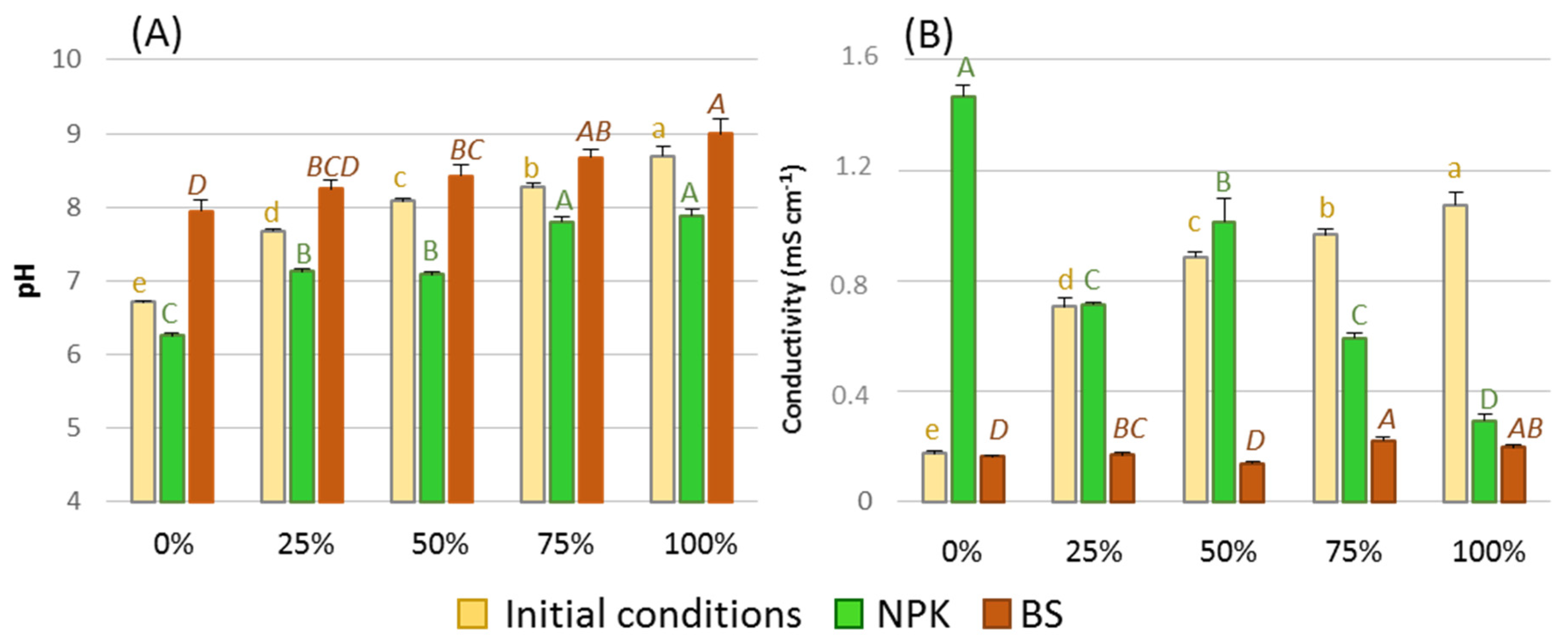
3.1. Chemical Characteristics of the Substrate
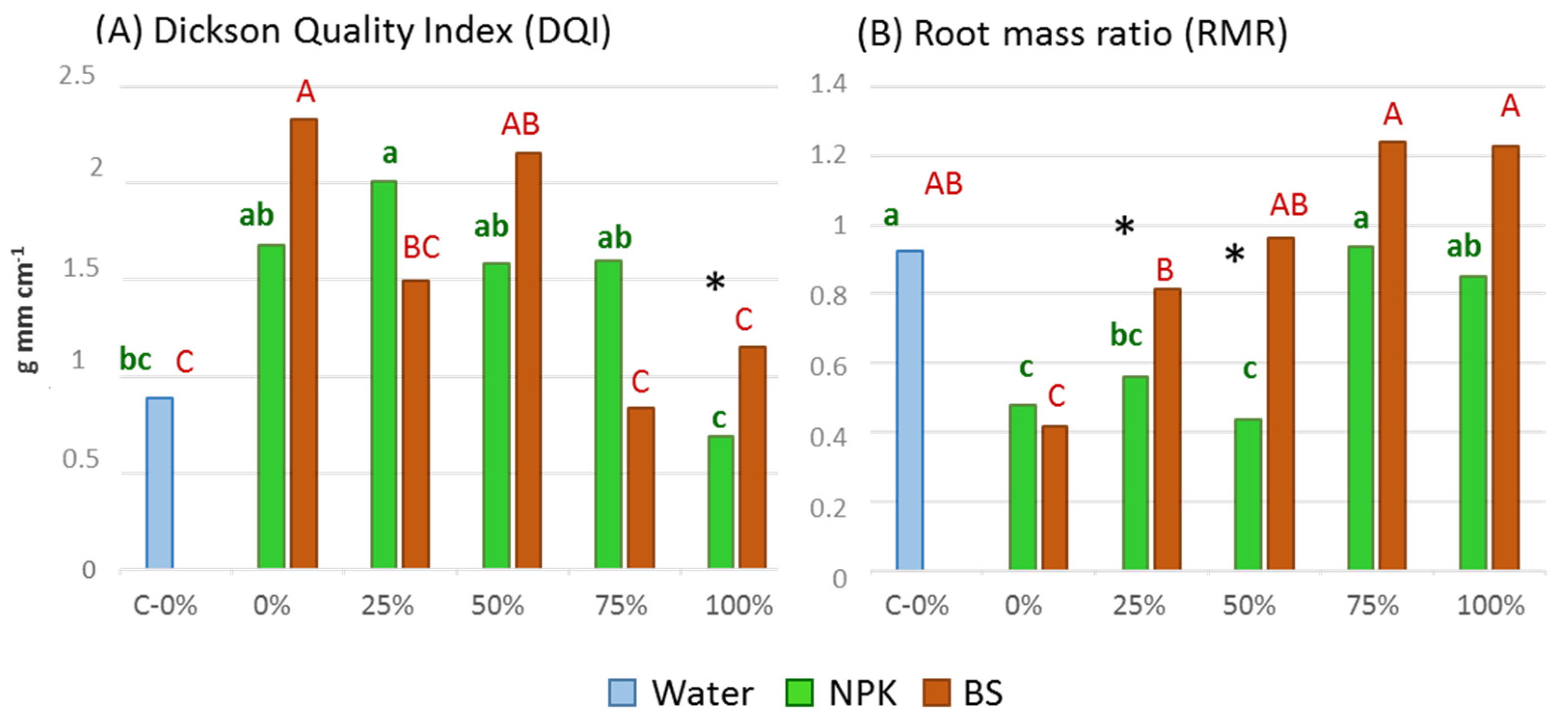
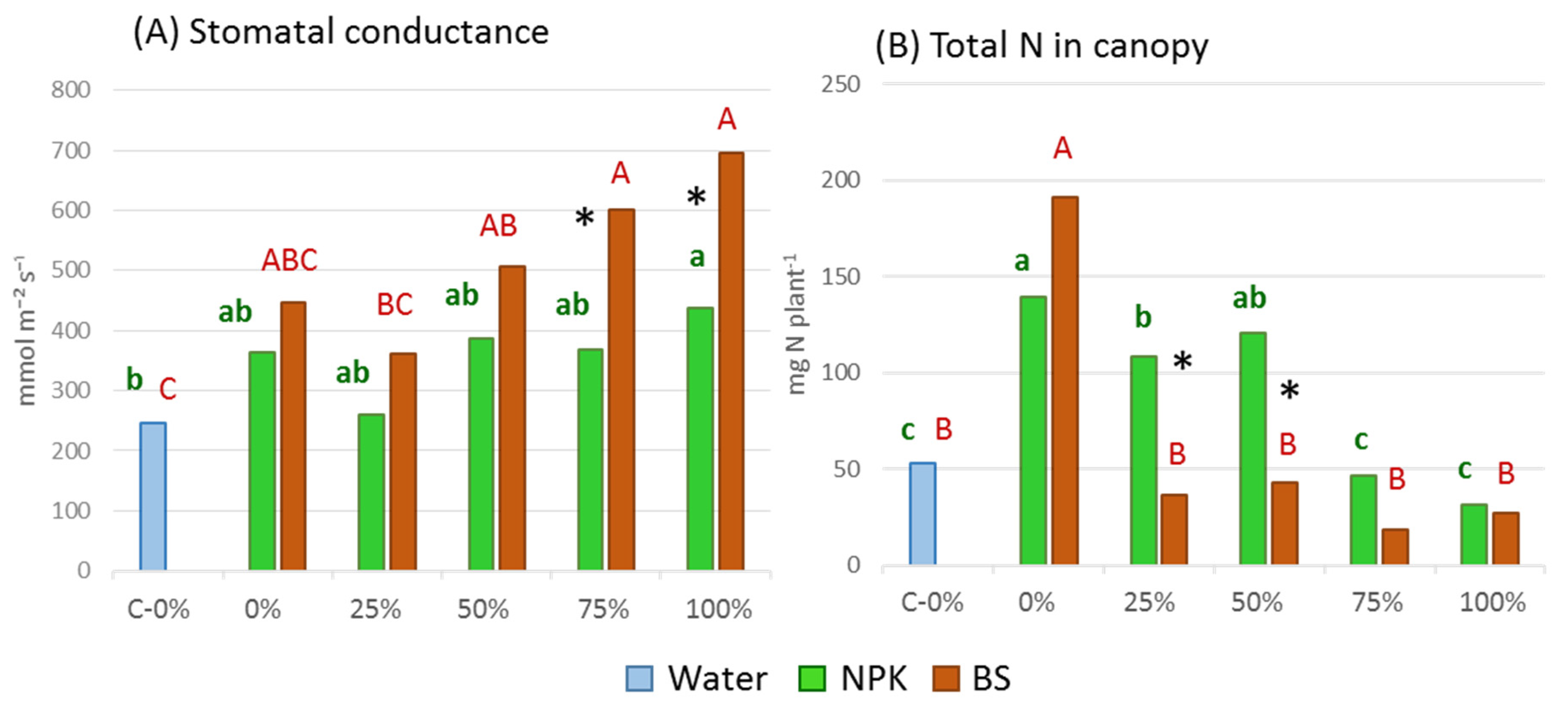
3.2. Plant Growth
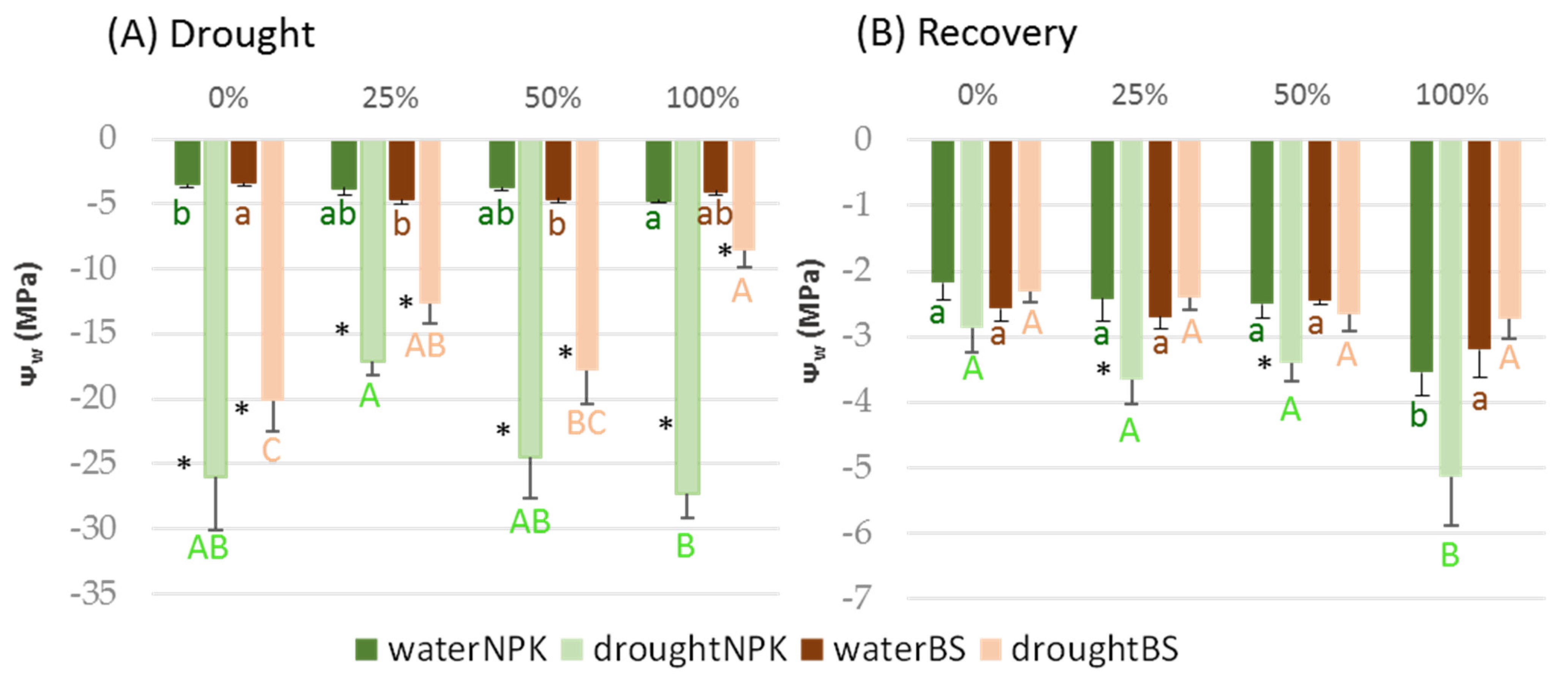
3.3. Plant Tolerance to Water Stress
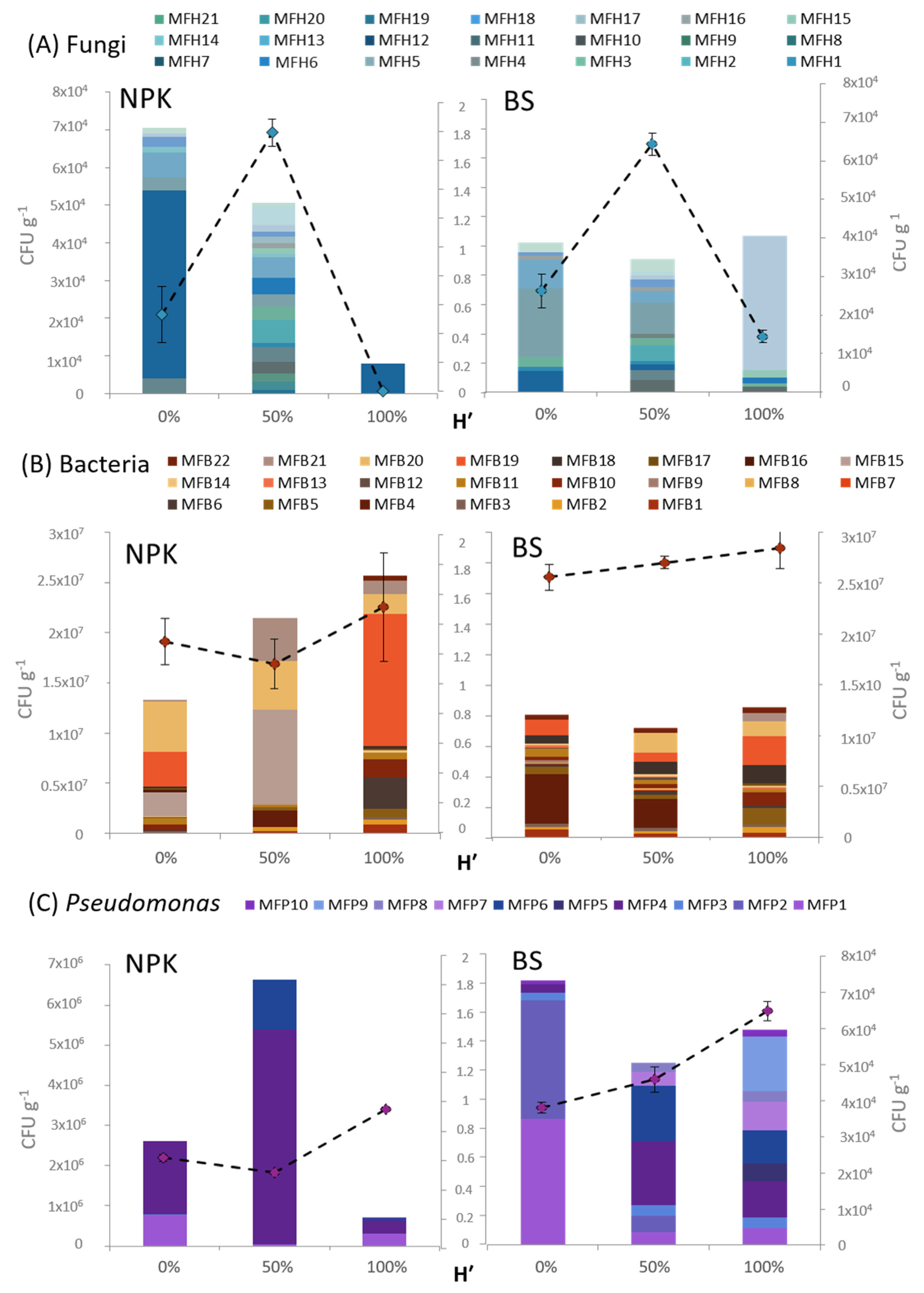
3.4. Rhizosphere Microbiota Analysis
4. Discussion
4.1. Use of Compost as a Substitute for Peat in Chestnut Tree Cultivation
4.2. Use of PGP-Based Biostimulant as an Alternative to Mineral Fertilization
4.3. Compost–Biostimulant Synergy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Conedera, M.; Krebs, P. History, present situation and perspective of chestnut cultivation in Europe. Acta Hort. 2008, 784, 23–27. [Google Scholar] [CrossRef]
- Radoglou, K.; Raftoyannis, Y.; Halivopoulos, G. The effects of planting date and seedling quality on field performance of Castanea sativa Mill. And Quercus frainetto Ten. Seedl. For. 2003, 76, 569–578. [Google Scholar] [CrossRef][Green Version]
- Landis, T.D. Mineral nutrition as an Index of Seedling Quality. In Evaluating Seedling Quality: Principles, Procedures, and Predictive Abilities of Major Tests; Duryea, M.L., Ed.; Forest Research Laboratory, Oregon State University: Corvallis, OR, USA, 1985; pp. 29–48. [Google Scholar]
- Veijalainen, A.M.; Juntunen, M.L.; Heiskanen, J.; Lilja, A. Growing Picea abies seedlings in peat and composted forest-nursery waste mixtures for forest regeneration. Scan. J. For. Res. 2007, 22, 390–397. [Google Scholar] [CrossRef]
- Krucker, M.; Hummel, R.L.; Cogger, C. Chrysanthemum production in composted and noncomposted organic waste substrates fertilized with nitrogen at two rates using surface and subirrigation. Hort. Sci. 2010, 45, 1695–1701. [Google Scholar] [CrossRef]
- Maher, M.; Prasad, M.; Raviv, M. Organic soilless media components. In Soilless Culture: Theory and Practice; Raviv, M., Lieth, J.H., Eds.; Academic Press: San Diego, CA, USA, 2008; pp. 459–504. [Google Scholar]
- Alexander, P.D.; Bragg, N.C.; Meade, R.; Padelopoulos, G.; Watts, O. Peat in horticulture and conservation: The UK response to a changing world. Mires Peat 2008, 3, 8. [Google Scholar]
- Medina, E.; Paredes, C.; Pérez-Murcia, M.D.; Bustamante, M.A.; Moral, R. Spent mushroom substrates as component of growing media for germination and growth of horticultural plant. Bioresour. Technol. 2009, 100, 4227–4232. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Wang, Q.; Zhang, J.; Wang, L.; Li, Y.; Yang, G. Minimum fertilization at the appearance of the first flower benefits cotton nutrient utilization of nitrogen, phosphorus and potassium. Sci. Rep. 2001, 10, 1–13. [Google Scholar]
- Van den Driessche, R. Changes in drought resistance and root growth capacity of container seedlings in response to nursery drought, nitrogen and potassium treatments. Can. J. Forest Res. 1992, 22, 740–749. [Google Scholar] [CrossRef]
- Farrell, M.; Jones, D.L. Food waste composting: Its use as a peat replacement. Waste Manag. 2010, 30, 1495–1501. [Google Scholar] [CrossRef]
- Veijalainen, A.M.; Heiskanen, J.; Juntunen, M.L.; Lilja, A. Tree-seedling compost as a component in Sphagnum peat-based growing media for conifer seedlings: Physical and chemical properties. Acta Hort. 2008, 779, 431–438. [Google Scholar] [CrossRef]
- Wilson, S.B.; Mecca, L.K.; Stoffella, P.J. Evaluation of compost as a viable medium amendment for containerized perennial production. Acta Hort. 2004, 659, 697–703. [Google Scholar] [CrossRef]
- Raviv, M. Production of high-quality composts for horticultural purposes—A minireview. Hort. Technol. 2005, 15, 52–57. [Google Scholar] [CrossRef]
- DuJardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hort. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Odoh, C.K.; Eze, C.N.; Akpi, U.K.; Unah, V.U. Plant growth promoting rhizobacteria (PGPR): A novel agent for sustainable food production. Am. J. Agr. Biol. Sci. 2019, 14, 35–54. [Google Scholar]
- Nihorimebere, V.; Ongena, M.; Smargiassi, M.; Thonart, P. Beneficial effect of the rhizosphere microbial community for plant growth and health. Biotechnol. Agron. Soc. Environ. 2011, 15, 327–337. [Google Scholar]
- Yang, J.; Kloepper, J.W.; Ryu, J. Rhizosphere bacteria help plants tolerate abiotic stress. Trend Plant Sci. 2009, 14, 1–4. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Kloepper, J. Agricultural uses of plant biostimulants. Plant Soil. 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Otero, M.; Salcedo, I.; Txarterina, K.; González-Murua, C.; Duñabeitia, M.K. Quality assessment of Pinus radiata production under sustainable nursery management based on compost tea. J. Plant Nutr. Soil. Sci. 2019, 182, 1–11. [Google Scholar] [CrossRef]
- Dickson, A.; Leaf, A.L.; Hosner, J.F. Quality appraisal of white spruce and white pine seedling stock in nurseries. Forest. Chron. 1960, 36, 10–13. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v7: User Manual/Tutorial. PRIMER-EPlymouth. 2015. Available online: http://updates.primer-e.com/primer7/manuals/User_manual_v7a.pdf (accessed on 4 June 2021).
- Wilson, S.B.; Stoffella, P.J.; Graetz, D.A. Development of compost-based media for containerized perennials. Sci. Hort. 2002, 93, 311–320. [Google Scholar] [CrossRef]
- Davis, A.S.; Jacobs, D.F.; Wightman, K.E.; Birge, Z.K.D. Organic matter added to bareroot nursery beds influences soil properties and morphology of Fraxinus pennsylvanica and Quercus rubra seedlings. New For. 2006, 31, 293–303. [Google Scholar] [CrossRef]
- Albano, J.P.; Miller, W.B. Marigold cultivars vary in susceptibility to iron toxicity. Hort. Sci. 1998, 33, 1180–1182. [Google Scholar] [CrossRef]
- Smith, B.R.; Fisher, P.R.; Argo, W.R. Growth and pigment content of container-grown impatiens and petunia in relation to root substrate pH and applied micronutrient concentration. Hort. Sci. 2004, 39, 1421–1425. [Google Scholar] [CrossRef]
- Cristiano, G.; Camposeo, S.; Fracchiolla, M.; Vivaldi, G.A.; De Lucia, B.D.L.; Cazzato, E. Salinity differentially affects growth and ecophysiology of two mastic tree (Pistacia lentiscus L.) accessions. Forests 2016, 7, 156. [Google Scholar] [CrossRef]
- Yadav, K.D.; Ahammed, M.M. Integrated composting-vermicomposting process for stabilization of human faecal slurry. Ecol. Eng. 2012, 47, 24–29. [Google Scholar] [CrossRef]
- Grossnickle, S.C. Importance of root growth in overcoming planting stress. New For. 2005, 30, 273–294. [Google Scholar] [CrossRef]
- Ashraf, R.; Shahid, F.; Adam, T.A. Association of fungi, bacteria and Actinomycetes with different composts. Pak. J. Bot. 2007, 39, 2141–2151. [Google Scholar]
- Khan, Z.; Kandel, S.L.; Ramos, D.N.; Ettl, G.J.; Kim, S.H.; Doty, S.L. Increased biomass of nursery-grown Douglas-fir seedlings upon inoculation with diazotrophic endophytic consortia. Forests 2015, 6, 3582–3593. [Google Scholar] [CrossRef]
- Probanza, A.; Lucas García, J.A.; Ruiz Palomino, M.; Ramos, B.; Gutiérrez Mañero, F.J. Pinus pinea L. seedling growth and bacterial rhizosphere structure after inoculation with PGPR Bacillus (B. licheniformis CECT 5106 and B. pumilus CECT 5105). Appl. Soil Ecol. 2002, 20, 75–84. [Google Scholar] [CrossRef]
- Jacobs, D.F.; Timmer, V.R. Fertilzer-induced changes in rhizosphere electrical conductivity: Relation to forest tree seedling root system growth and function. New For. 2005, 30, 147–166. [Google Scholar] [CrossRef]
- Bhattacharyya, P.N.; Jha, D.K. Plant growth-promoting rhizobacteria (PGPR): Emergence in agriculture. World J. Microbiol. Biotechnol. 2012, 28, 1327–1350. [Google Scholar] [CrossRef]
- Yan, Y.; Yang, J.; Dou, Y. Nitrogen fixation island and rhizosphere competence traits in the genome of root-associated Pseudomonas stutzeri A1501. Proc. Nat. Acad. Sci. USA 2008, 21, 7564–7569. [Google Scholar] [CrossRef]
- Iqbal, S.; Hameed, S.; Shahid, M.; Hussain, K.; Ahmad, N.; Niaz, M. In vitro characterization of bacterial endophytes from tomato (Solanum lycopersicum L.) for phytobeneficial traits. App. Ecol. Environ. Res. 2018, 16, 1037–1051. [Google Scholar] [CrossRef]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Patten, C.L.; Glick, B.R. Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. App. Environ. Microbiol. 2002, 68, 3795–3801. [Google Scholar] [CrossRef]
- Kudoyarova, G.; Arkhipova, T.; Korshunova, T.; Bakaeva, M.; Loginov, O.; Dodd, C. Phytohormone mediation interactions between plants and non-symbiotic growth promoting bacteria under edaphic stresses. Front. Plant Sci. 2019, 10, 1368. [Google Scholar] [CrossRef]
- Gutiérrez-Mañero, F.J.; Ramos-Solano, B.; Probanza, A.N.; Mehouachi, J.R.; Tadeo, F.; Talon, M. The plant-growth-promoting rhizobacteria Bacillus pumilus and Bacillus licheniformis produce high amounts of physiologically active gibberellins. Phys. Plant. 2001, 111, 206–211. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; Cortés-Penagos, C.; López-Bucio, J. Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol. 2009, 149, 1579–1592. [Google Scholar] [CrossRef]
- Hossain, M.M.; Sultana, F.; Islam, S. Plant Growth-Promoting Fungi (PGPF): Phytostimulation and Induced Systemic Resistance. In Plant-Microbe Interactions in Agro-Ecological Perspectives, Microbial Interactions and Agro-ecological Impacts; Singh, D.P., Singh, H.B., Prabha, R., Eds.; Springer: Singapore, 2017; Volume 2, pp. 135–191. [Google Scholar]
- Colla, G.; Rouphael, Y.; Di Mattia, E.; El-Nakhel, C.; Cardarelli, M. Co-inoculation of Glomus intraradices and Trichoderma atroviride acts as a biostimulant to promote growth, yield and nutrient uptake of vegetable crops. J. Sci. Food Agric. 2015, 95, 1706–1715. [Google Scholar] [CrossRef]
- Reddy, P.P. Plant Growth Promoting Rhizobacteria for Horticultural Crop Protection, Challenges, Future Prospects and Conclusions; Springer: New Delhi, India, 2014; pp. 296–303. [Google Scholar]
- Amprayn, K.; Rose, M.; Kecskés, M.; Pereg, L.; Nguyen, H.; Kennedy, I. Plant growth promoting characteristics of soil yeast (Candida tropicalis HY) and its effectiveness for promoting rice growth. Appl. Soil Ecology. 2012, 61, 295–299. [Google Scholar] [CrossRef]
- Sarabia, M.; Cornejo, P.; Azcón, R.; Carreón-Abud, Y.; Larsen, J. Mineral phosphorus fertilization modulates interactions between maize, rhizosphere yeasts and arbuscular mycorrhizal fungi. Rhizosphere 2017, 4, 325–341. [Google Scholar] [CrossRef]
- Nutaratat, P.; Srisuk, N.; Arunrattiyakorn, P.; Limtong, S. Plant growth-promoting traits of epiphytic and endophytic yeasts isolated from rice and sugar cane leaves in Thailand. Fungal Biol. 2014, 118, 683–694. [Google Scholar] [CrossRef]
- Babu, A.G.; Kim, S.W.; Yadav, D.J.; Hyum, U.; Adhikari, M.; Lee, Y.S. Penicillium menonorum: A novel fungus to promote growth and nutrient management in cucumber plants. Mycobiology 2015, 43, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Xing, S.; Ma, H.; Du, Z.; Ma, B. Cytokinin-producing, plant growth-promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. App. Microbiol. Biotech. 2013, 97, 9155–9164. [Google Scholar] [CrossRef] [PubMed]
- Sandhya, V.Z.A.S.; Grover, M.; Reddy, G.; Venkateswarlu, B. Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-p45. Biol. Fert. Soil. 2009, 46, 17–26. [Google Scholar] [CrossRef]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M.; SkZ, A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Arshad, M.; Shaharoona, B.; Mahmood, T. Inoculation with Pseudomonas spp. containing ACC-deaminase partially eliminates the effects of drought stress on growth, yield, and ripening of pea (Pisum sativum L.). Pedosphere 2008, 18, 611–620. [Google Scholar] [CrossRef]
- Arzanesh, M.H.; Alikhani, H.A.; Khavazi, K.; Rahimian, H.A.; Miransari, M. Wheat (Triticum aestivum L.) growth enhancement by Azospirillum sp. under drought stress. World J. Microbiol. Biotech. 2011, 27, 197–205. [Google Scholar] [CrossRef]
- Shoresh, M.; Harman, G.E.; Mastouri, F. Induced systemic resistance and plant responses to fungal biocontrol agents. Ann. Rev. Phyto. 2010, 48, 21–43. [Google Scholar] [CrossRef]
- Chepsergon, J.; Mwamburi, D.L.; Kassim, M.K. Mechanism of Drought Tolerance in Plants Using Trichoderma spp. Int. J. Sci. Res. 2014, 3, 1592–1595. [Google Scholar]
- Martínez-Medina, A.; Alguacil, M.D.M.; Pascual, J.A.; Van Wees, S.C. Phytohormone profiles induced by Trichoderma isolates correspond with their biocontrol and plant growth-promoting activity on melon plants. J. Chem. Ecol. 2014, 40, 804–815. [Google Scholar] [CrossRef] [PubMed]
- Arif, M.S.; Shahzad, S.M.; Riaz, M.; Yasmeen, T.; Shahzad, T.; Akhtar, M.J.; Bragazza, L.; Buttler, A. Nitrogen-enriched compost application combined with plant growth-promoting rhizobacteria (PGPR) improves seed quality and nutrient use efficiency of sunflower. J. Plant Nutr. Soil Sci. 2017, 180, 464–473. [Google Scholar] [CrossRef]


| Compost | Biostimulant (BS) | |
|---|---|---|
| Dry matter (%) | 87.74 | 0.91 |
| Moisture (%) | 12.26 | 99.09 |
| Organic matter (%) | 69.3 | nd |
| Total N (%) | 2.85 | 0.1449 |
| Ammonium (mg/L) | 71.2 | 0.0183 |
| Nitrate (mg/L) | <1.0 | nd |
| Organic C (%) | 40.2 | n.m. |
| C/N | 14.1 | 14.1 |
| pH (1/5 v/v) | 7.59 | 7.84 |
| Conductivity (mS/cm) (1/5 v/v) | 11.2 | 14.3 |
| Density (g/cm3) | 0.35 | 1.011 |
| Total calcium (mg/L) | 233 | 0.0131 |
| Sulphate (mg/L) | 402 | 0.0634 |
| Phosphate (mg/L) | 35.7 | nd |
| Magnesium (mg/L) | 60.40 | 0.0508 |
| Carbonate (mg/L) | <5.0 | n.m. |
| Bicarbonate (mg/L) | 1740 | n.m. |
| Chloride (mg/L) | 2140 | n.m. |
| Potassium (mg/L) | 3170 | 0.457 |
| Sodium (mg/L) | 349 | 2.037 |
| Humic acids (%) | 18.40 | nd |
| Fulvic acids (%) | 3.82 | nd |
| Total humic extract (%) | 22.2 | nd |
| Escherichia coli (MPN/g) | <10 | nd |
| Salmonella (25 g) | nd | nd |
| Root Volume | Height | Root- Collar Diameter | Canopy Area | SLA | Leaf DW | Stem DW | Root DW | Total DW | ||||||||||
| sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | |
| substrate | *** | 0.381 | *** | 0.792 | *** | 0.96 | *** | 0.716 | ns | 0.158 | *** | 0.843 | *** | 0.679 | *** | 0.377 | *** | 0.723 |
| fertilization | ns | 0.044 | *** | 0.320 | ns | 0.002 | ns | 0.002 | ns | 0.028 | ns | 0.054 | ** | 0.118 | ns | 0.06 | ns | 0.024 |
| substrate × Fert | ** | 0.214 | *** | 0.485 | *** | 0.309 | ns | 0.119 | ns | 0.121 | *** | 0.421 | *** | 0.345 | ns | 0.110 | ** | 0.260 |
| RMR | DQI | Root/Shoot | Leaf N (%) | N Canopy | SC | Soil pH | Soil EC | |||||||||||
| sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | sig | ɳ2p | |||
| substrate | *** | 0.449 | *** | 0.337 | *** | 0.581 | *** | 0.403 | *** | 0.798 | ** | 0.297 | *** | 0.842 | *** | 0.935 | ||
| fertilization | *** | 0.199 | ns | 0.004 | ** | 0.147 | *** | 0.543 | *** | 0.211 | ** | 0.186 | *** | 0.884 | *** | 0.983 | ||
| substrate × Fert | ns | 0.107 | * | 0.181 | * | 0.215 | * | 0.246 | *** | 0.468 | ns | 0.063 | *** | 0.284 | *** | 0.916 | ||
| Fungi | Bacteria | Pseudomonas | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fertilization | Fertilization | Fertilization | ||||||||||||
| Source | Df | Sum Sq | Pseudo-F | p (perm) | Source | Df | Sum Sq | Pseudo-F | p (perm) | Source | Df | Sum Sq | Pseudo-F | p (perm) |
| Fertilization | 1 | 9589.4 | 2.6385 | 0.017 | Fertilization | 1 | 9579 | 5.0969 | 0.001 | Fertilization | 1 | 27,036 | 12.376 | 0.001 |
| Residuals | 34 | 1.24 × 105 | Residuals | 16 | 30,070 | Residuals | 34 | 74,274 | ||||||
| Total | 35 | 1.33 × 105 | Total | 17 | 39,649 | Total | 35 | 1.01 × 105 | ||||||
| Substrate | Substrate | Substrate | ||||||||||||
| Source | Df | Sum Sq | Pseudo-F | p (perm) | Source | Df | Sum Sq | Pseudo-F | p (perm) | Source | Df | Sum Sq | Pseudo-F | p (perm) |
| Substrate | 2 | 43,984 | 8.1383 | 0.001 | Substrate | 2 | 9682.6 | 2.4234 | 0.016 | Substrate | 2 | 14,683 | 2.7968 | 0.004 |
| Residuals | 33 | 89,175 | Residuals | 15 | 29,967 | Residuals | 33 | 86,627 | ||||||
| Total | 35 | 1.33 × 105 | Total | 17 | 39,649 | Total | 35 | 1.01 × 105 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuertes-Mendizábal, T.; Huérfano, X.; Ortega, U.; González-Murua, C.; Estavillo, J.M.; Salcedo, I.; Duñabeitia, M.K. Compost and PGP-Based Biostimulant as Alternative to Peat and NPK Fertilization in Chestnut (Castanea Sativa Mill.) Nursery Production. Forests 2021, 12, 850. https://doi.org/10.3390/f12070850
Fuertes-Mendizábal T, Huérfano X, Ortega U, González-Murua C, Estavillo JM, Salcedo I, Duñabeitia MK. Compost and PGP-Based Biostimulant as Alternative to Peat and NPK Fertilization in Chestnut (Castanea Sativa Mill.) Nursery Production. Forests. 2021; 12(7):850. https://doi.org/10.3390/f12070850
Chicago/Turabian StyleFuertes-Mendizábal, Teresa, Ximena Huérfano, Unai Ortega, Carmen González-Murua, José María Estavillo, Isabel Salcedo, and Miren K. Duñabeitia. 2021. "Compost and PGP-Based Biostimulant as Alternative to Peat and NPK Fertilization in Chestnut (Castanea Sativa Mill.) Nursery Production" Forests 12, no. 7: 850. https://doi.org/10.3390/f12070850
APA StyleFuertes-Mendizábal, T., Huérfano, X., Ortega, U., González-Murua, C., Estavillo, J. M., Salcedo, I., & Duñabeitia, M. K. (2021). Compost and PGP-Based Biostimulant as Alternative to Peat and NPK Fertilization in Chestnut (Castanea Sativa Mill.) Nursery Production. Forests, 12(7), 850. https://doi.org/10.3390/f12070850







