Associations of Phoretic Mites on Bark Beetles of the Genus Ips in the Black Sea Mountains of Turkey
Abstract
1. Introduction
2. Materials and Methods
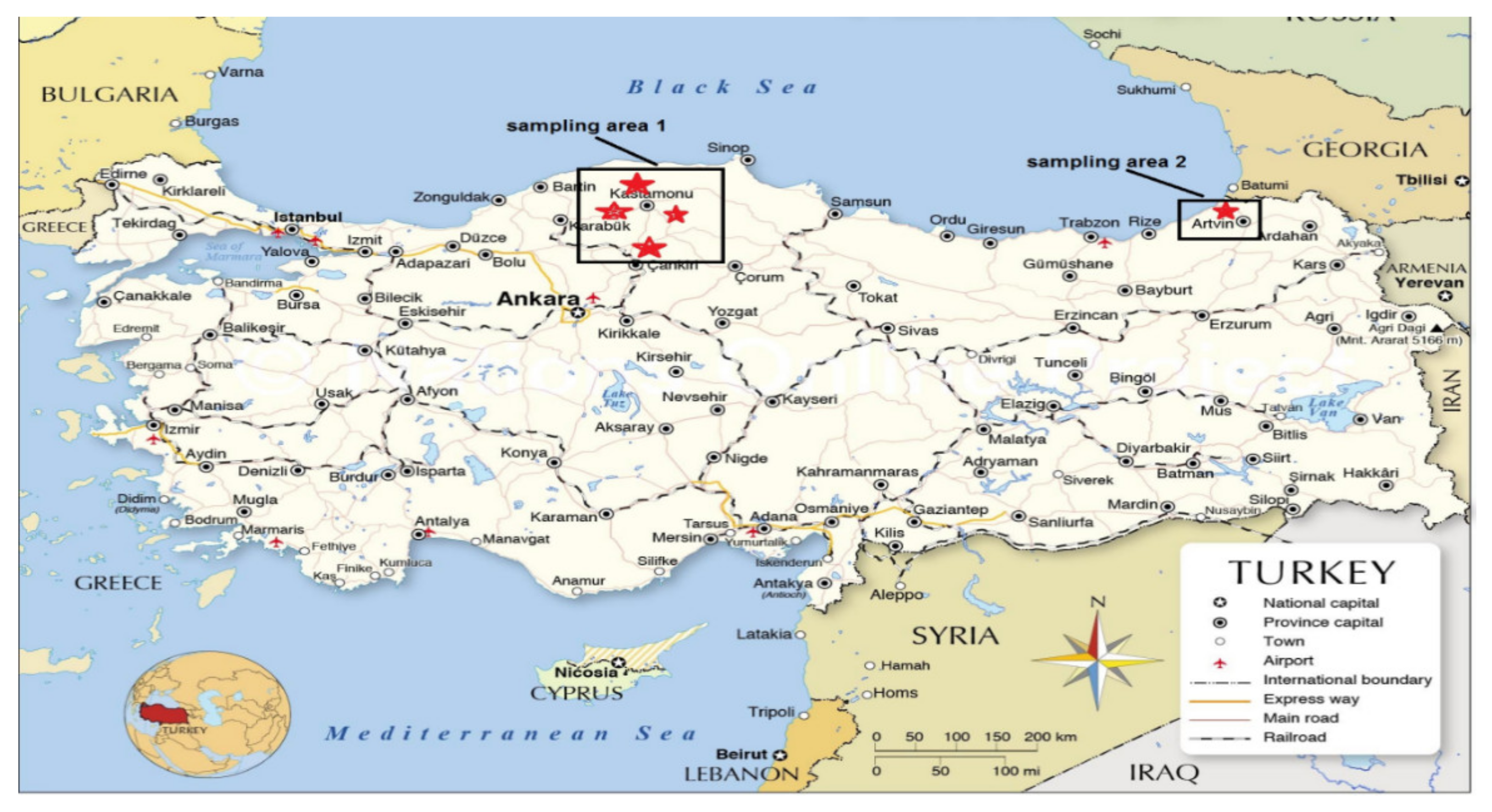
2.1. Study Area and Sampling Procedures
2.2. Sampling Procedures, Laboratory Treatment and Identification of Phoretic Mites
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vissa, S.; Hofstetter, R.W. The role of mites in bark and ambrosia beetle–fungal interactions. Insect Physiol. Ecol. 2017, 135–156. [Google Scholar] [CrossRef]
- Moser, J.C. Mite predators of the southern pine beetle. Ann. Entomol. Soc. Am. 1975, 68, 1113–1116. [Google Scholar] [CrossRef]
- Pernek, M.; Hrasovec, B.; Matosevic, D.; Pilas, I.; Kirisits, T.; Moser, J.C. Phoretic mites of three bark beetles (Pityokteines spp.) on Silver fir. J. Pest. Sci. 2008, 81, 35–42. [Google Scholar] [CrossRef]
- Hofsteter, R.W.; Moser, J.C. Role of mite sin insect-fungus associations. Ann. Rev. Entomol. 2014, 59, 537–557. [Google Scholar] [CrossRef]
- Hofstetter, R.W.; Dikins-Bookwalter, J.; Davis, T.D.; Klepzig, K.D. Symbiotic associations of bark beetles. In Bark Beetles: Biology and Ecology of Native and Invasive Species; Vega, F.E., Hofstetter, R.W., Eds.; Elsevier/Academic Press: London, UK, 2015; pp. 209–245. [Google Scholar]
- Hofstetter, R.W.; Moser, J.C.; Blomquist, S. Mites associated with bark beetles and their hypophoretic Ophiostomatoid fungi. In The Ophiostomatoid Fungi: Expanding Frontiers; Wingfield, S., Ed.; CBS-KNAW Fungal Biodiversity Centre: Utrecht, The Nederlands, 2013; pp. 165–176. [Google Scholar]
- Kontschán, J.; Szőcs, G.; Kiss, B.; Khaustov, A.A. Bark beetle associated trematurid mites (Acari: Uropodina: Trematuridae) from Asian Russia with description of a new species. Syst. Appl. Acarol. 2019, 24, 1592–1603. [Google Scholar]
- Schelhaas, M.J.; Nabuurg, G.J.; Schuck, A. Natural disturbances in the European forests in the 19th and 20th centuries. Glob. Chang. Biol. 2003, 9, 1620–1633. [Google Scholar] [CrossRef]
- Raffa, K.F.; Aukema, B.H.; Bentz, B.J.; Carroll, A.L.; Hickl, J.A.; Turner, M.G.; Romme, W.H. Cross-scale drivers of natural disturbances prone to anthropogenic amplification: The dynamics of bark beetle eruptions. Bioscience 2008, 58, 501–517. [Google Scholar] [CrossRef]
- Wermelinger, B. Ecology and management of the spruce bark beetle Ips typographus—A review of recent research. For. Ecol. Manag. 2004, 202, 67–82. [Google Scholar] [CrossRef]
- Rouault, G.; Candau, J.N.; Lieutier, F.; Nageleisen, L.N.; Martin, J.C.; Warzée, N. Effects of drought and heat on forest insect populations in relation to the drought in western Europe. Ann. For. Sci. 2006, 63, 613–624. [Google Scholar] [CrossRef]
- Mulock, P.; Christiansen, E. The threshold of successful attack by Ips typographus on Picea abies: A field experiment. For. Ecol. Manag. 1986, 14, 125–132. [Google Scholar] [CrossRef]
- Burkhard, B.; Bässler, C.; Thorn, S.; Noss, R.; Schröder, B.; Dieffenbach-Fries, H.; Foullois, N.; Müller, J. Bark Beetles Increase Biodiversity While Maintaining Drinking Water Quality. Conserv. Lett. 2015, 8, 272–281. [Google Scholar]
- Vanická, H.; Holuša, J.; Resnerová, K.; Ferenčík, J.; Potterf, M.; Vélea, A.; Grodzki, W. Interventions have limited effects on the population dynamics of Ips typographus and its natural enemies in the Western Carpathians (Central Europe). For. Ecol. Manag. 2020, 470–471, 118209. [Google Scholar] [CrossRef]
- Lindgren, B.S.; Raffa, K.F. Evolution of tree killing in bark beetles (Coleoptera: Curculionidae): Trade-offs between the maddening crowds and a sticky situation. Can. Entomol. 2013, 145, 471–495. [Google Scholar] [CrossRef]
- Cobb, T.P.; Hannam, K.D.; Kishchuk, B.E.; Langor, D.W.; Quideau, S.A.; Spence, J.R. Wood-feeding beetles and soil nutrient cycling in burned forests: Implications of post-fire salvage logging. Agric. For. Entomol. 2010, 12, 9–18. [Google Scholar] [CrossRef]
- Pernek, M.; Wirth, S.; Blomquist, S.R.; Avtzis, D.N.; Moser, J.C. New associations of phoretic mites on Pityokteines curvidens (Coleoptera, Curculionidae, Scolytinae). Cent. Eur. J. Biol. 2012, 7, 63–68. [Google Scholar] [CrossRef]
- Klepzig, K.D.; Moser, J.C.; Lombardero, F.J.; Hofstetter, R.W.; Ayres, M.P. Symbiosis and competition: Complex interactions among beetles, fungi and mites. Symbiosis 2001, 30, 83–96. [Google Scholar]
- Lombardero, M.J.; Matthew, P.A.; Hofstetter, R.W.; Moser, J.C.; Klepzig, K.D. Strong indirect interactions of Tarsonemus mites (Acarina: Tarsonemidae) and Dendroctonus frontalis (Coleoptera: Scolytidae). Oikos 2003, 102, 243–252. [Google Scholar] [CrossRef]
- Walter, D.E.; Lindquist, E.E.; Smith, I.M.; Cook, D.R.; Krantz, G.W. Order Trombidiformes. In A Manual of Acrology, 2nd ed.; Kranz, G.W., Lubbock, D.E., Eds.; Texas Tech University Press: Lubbock, TX, USA, 2009; pp. 83–94. [Google Scholar]
- Rosario, R.M.T.; Hunter, P.E. The genus Myrmozercon Berlese, with descriptions of two new species (Acari: Mesostigmata: Laelapidae). J. Parasitol. 1988, 74, 466–470. [Google Scholar] [CrossRef]
- Kiełczewski, B.; Moser, J.C.; Wiśniewski, J. Surveying the acarofauna associated with Polish Scolytidae. Bulletin de la société des amis des sciences et des lettres de Poznań. Ser. D Sci. Biol. 1983, 22, 151–159. [Google Scholar]
- Zeydanli, U. Western Asia: Northern Turkey Extending along the Southern Black Sea Region. 2020. Available online: https://www.worldwildlife.org/ecoregions/pa0515 (accessed on 5 November 2020).
- Williams, L.; Zazanashvili, N.; Sanadiradze, G.; Kandaurov, A. An Ecoregional Conservation Plan for Caucasus; WWF: Brussels, Belgium, 2006; p. 220. [Google Scholar]
- Schütt, H. The Turkish Land Snails 1758–2005: Vollständig Revidierte und Erweiterte Auflage. Nat. Wiss. 2005, 1, 559. [Google Scholar]
- Yüksek, T.; Ölmez, Z. A general assessment of climate, soil structure, forest areas, growing stock and some forestry applications of Artvin region. Artvin Çoruh Üniversitesi Orman Fakültesi Dergisi 2002, 3, 50–62. [Google Scholar]
- Knížek, M. Platypodinae and Scolytinae. In Catalogue of Palaearctic Coleoptera; Löbl, I., Smetana, A., Curculionoidea, I., Eds.; Apollo Books: Stenstrup, Denmark, 2011; Volume 7, pp. 201–251. [Google Scholar]
- Sarikaya, O.; Avci, M. Predators of Scolytinae (Coleoptera: Curculionidae) species of the coniferous forests in the Western Mediterranean Region, Turkey. Turk. Entomoloji Derg. 2009, 33, 253–264. [Google Scholar]
- Sarıkaya, O.; Knižek, M. Scolytus koenigi Schevyrew, 1890: A New Record for Turkish Scolytinae (Coleoptera: Curculionidae) Fauna. J. Entomol. Res. Soc. 2013, 15, 95–99. [Google Scholar]
- Maslov, A.D. Koroed-Tipograf i Usykhanie Elovykh Lesov. Moscow Vserosiyskiy Nauchnoissledovatelskiy Inst. Lesovod. Mekhanizatsii Lesn. Khozyaystva 2010, 138, 320. [Google Scholar]
- Yüksel, B.; Tozlu, G.; Şentürk, M. Sarıkamış Sarıçam (Pinus sylvestris L.) Ormanlarında Etkin Zarar Yapan Kabuk Böcekleri ve Bunlara Karşı Alınabilecek Önlemler. Doğu Akdeniz Ormancılık Araştırma Müdürlüğü. Tek. Bül. 2000, 1, 1–66. [Google Scholar]
- O’Connor, B.M. Acarine-fungal relationships: The evolution of symbiotic associations. In Fungus-Insect Relationships: Perspectives in Ecology and Evolution; Wheeler, P., Blackwell, M., Eds.; Columbia University Press: New York, NY, USA, 1984; pp. 354–381. [Google Scholar]
- Kinn, D.N. Notes on the life cycle and habits of Digamasellus quadrisetus (Mesostigmata: Digamasellidae). Ann. Entomol. Soc. Am. 1967, 60, 862–865. [Google Scholar] [CrossRef]
- Paraschiv, M.; Isaia, G. Disparity of Phoresy in Mesostigmatid Mites upon their specific arrier Ips typographus (Coleoptera: Scolytinae). Insects 2020, 11, 771. [Google Scholar] [CrossRef]
- Paraschiv, M.; Martinez-Ruiz, C.; Fernandez, M.M. Dynamic associations between Ips sexdentatus (Coleoptera: Scolytinae) and its phoretic mites in a Pinus pinaster forest in northwest Spain. Exp. Appl. Acarol. 2018, 75, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Khaustov, A.A.; Klimov, P.B.; Trach, V.A.; Bobylev, A.N.; Salavatulin, V.M.; Khaustov, V.A.; Tolstikov, A.V. Review of mites (Acari) associated with the European spruce bark beetle, Ips typographus (Coleoptera: Curculionidae: Scolytinae) in Asian Russia. Acarina 2018, 26, 3–79. [Google Scholar] [CrossRef]
- Jacot, A.P. Acarina as possible vectors of the Dutch elm disease. J. Econ. Entomol. 1934, 27, 858–859. [Google Scholar] [CrossRef]
- Walter, D.E.; Proctor, H.C. Mites: Ecology, Evolution, and Behaviour; CABI Publishing: New York, NY, USA, 1999. [Google Scholar]
- Hunter, P.E. Five new mites of the subfamily Ereynetinae (Acarina: Ereynetidae). Fla. Entomol. 1964, 47, 181–193. [Google Scholar] [CrossRef]
- Schaeffer, S.; Koblmueller, S. Unexpected diversity in the host-generalist oribatid mite Paraleius leontonychus (Oribatida, Scheloribatidae) phoretic on Palearctic bark beetles. PEERJ 2020, 8, e9710. [Google Scholar] [CrossRef] [PubMed]
- Kinn, D.N. Incidence of pinewood nematode dauerlarvae and phoretic mites associated with long-horned beetles in central Louisiana. Can. J. For. Res. 1987, 17, 187–190. [Google Scholar] [CrossRef]
- André, H.M.; Fain, A. Phylogeny, ontogeny and adaptive radiation in the superfamily Tydeoidea (Acari: Actinedida), with a reappraisal of morphological characters. Zool. J. Linn. Soc. 2000, 130, 405–448. [Google Scholar] [CrossRef]
- O’Connor, B.; Klimov, P. Family Ereynetidae Oudemans. 2004. Available online: http://insects.ummz.lsa.umich.edu/beemites/Species_Accounts/Ereynetidae.htm (accessed on 5 December 2020).
- Kinn, D.N. The Life Cycle and Behavior of Cercoleipus coelonotus (Acarina: Mesostigmata), Including a Survey of Phoretic Mite Associates of California Scolytidae; University of California, Publications in Entomology: Berkeley, CA, USA, 1971; Volume 65, p. 66. [Google Scholar]

| Mite Species, Order, Family and Phoretic Stage | Location on Bark Beetle | Total Number of Mites on BB | Number of Mites in Alcohol Sediments | Number of Mites in Lactophenol Sediments | Total Number of Mites | Percent of Total Mites (n = 63) | Additional Information on Sites of Mite Species Collection |
|---|---|---|---|---|---|---|---|
| Dendrolaelaps quadrisetus Mesostigmata, Digamasellidae Deutonymph | Under elytra of Ips sexdentatus Ventral abdomen of Ips acuminatus Ventral thorax and head of Ips typographus | 10 | - | 1 | 11 | 17.5 | Kastamonu Province: Taşköprü Disctrict, Kapaklı Forests (41°24 N, 34°19 E), 1203 m, on Pinus nigra L. 22 June 2019; Daday District, Çamkonak Forests (41°23 N, 34°13 E), 1494 m, on Pinus nigra L. 30 May 2018; Küre District, Masruf Forests (41°43 N, 33°39 E), 1272 m, on Pinus nigra L. 30 May 2018; Artvin Province (41°11 N, 41°48 E), 708 m, on Picea abies L., 24 May 2019 |
| Ereynetes sp. Trombidiformes, Ereynetidae Female | Leg of Ips acuminatus | 1 | - | - | 1 | 1.6 | Kastamonu Province: Küre District, Masruf Forests (41°43 N, 33°39 E), 1272 m, on Pinus sylvestris L. 30 May 2018 |
| Histiostoma piceae Sarcoptiformes, Histiostomatidae Deutonymph | Under elytra of Ips sexdentatus | 1 | - | - | 1 | 1.6 | Kastamonu Province: Ilgaz Mountains (41°22′ N, 34°32′ E), 1407 m, on Pinus sylvestris L., 20 April 2018 |
| Paraleiuscf. leontonychus Sarcoptiformes, Hemileiidae | Galleries of BB | - | 1 | - | 1 | 1.6 | Kastamonu Province: Taşköprü District, Kapaklı Forests (41°25′ N, 34°18′ E), 917 m, on Pinus nigra L. 21 July 2019 |
| Pleuronectocelaeno barbara Mesostigmata, Neotenogyniidae Female | Under elytra of Ips sexdentatus | 1 | - | - | 1 | 1.6 | Kastamonu Province: Taşköprü District, Kapaklı Village Forest Area (41°24′ N, 34°19′ E), 1312 m, on Pinus nigra L., 1 July 2019 |
| Proctolaelaps hystricoides Mesostigmata, Melicharidae Female | Ips acuminatus in galleries of BB | 20 | - | 2 | 22 | 34.9 | Kastamonu Province: Küre District, Masruf Forests (41°43 N, 33°39 E), 1272 m, on Pinus nigra L. 30 May 2018 |
| Schizostethus simulatrix Mesostigmata, Melicharidae Deutonymph | Elytral declivity of Ips typographus | 12 | - | - | 12 | 19.0 | Artin Province (41°11 N, 41°48 E), 708 m, on Picea abies L. 24 May 2019 |
| Trichouropoda lamellosa Mesostigmata, Uropodidae Deutonymph | Elytral declivity of Ips typographus Dorsal thorax of Ips sexdentatus | 10 | - | - | 10 | 15.9 | Kastamonu Province: Küre District, Masruf Forests (41°43 N, 33°39 E), 1219 m, on Pinus nigra L. 17 July 2019; Daday District, Çamkonak Forests (41°23 N, 33°13 E), 1494 m, on Pinus nigra L. 30 May 2018; Artin Province (41°11 N, 41°48 E), 708 m, on Picea abies L., 24 May 2019 |
| Trichouropoda lamellosa | Unknown | - | 1 | - | 1 | 1.6 | |
| Uroobovella ipidis Mesostigmata, Urodinychidae Deutonymph | Dorsal thorax of Ips typographus | 3 | - | - | 3 | 4.7 | Kastamonu Province: Küre District, Masruf Forests (41°43 N, 33°39 E), 1272 m, on Pinus nigra L., 17 July 2019 |
| Total | 58 | 2 | 3 | 63 | 100.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cilbircioğlu, C.; Kovač, M.; Pernek, M. Associations of Phoretic Mites on Bark Beetles of the Genus Ips in the Black Sea Mountains of Turkey. Forests 2021, 12, 516. https://doi.org/10.3390/f12050516
Cilbircioğlu C, Kovač M, Pernek M. Associations of Phoretic Mites on Bark Beetles of the Genus Ips in the Black Sea Mountains of Turkey. Forests. 2021; 12(5):516. https://doi.org/10.3390/f12050516
Chicago/Turabian StyleCilbircioğlu, Cihan, Marta Kovač, and Milan Pernek. 2021. "Associations of Phoretic Mites on Bark Beetles of the Genus Ips in the Black Sea Mountains of Turkey" Forests 12, no. 5: 516. https://doi.org/10.3390/f12050516
APA StyleCilbircioğlu, C., Kovač, M., & Pernek, M. (2021). Associations of Phoretic Mites on Bark Beetles of the Genus Ips in the Black Sea Mountains of Turkey. Forests, 12(5), 516. https://doi.org/10.3390/f12050516








