Soil Respiration in Alder Swamp (Alnus glutinosa) in Southern Taiga of European Russia Depending on Microrelief
Abstract
1. Introduction
2. Materials and Methods
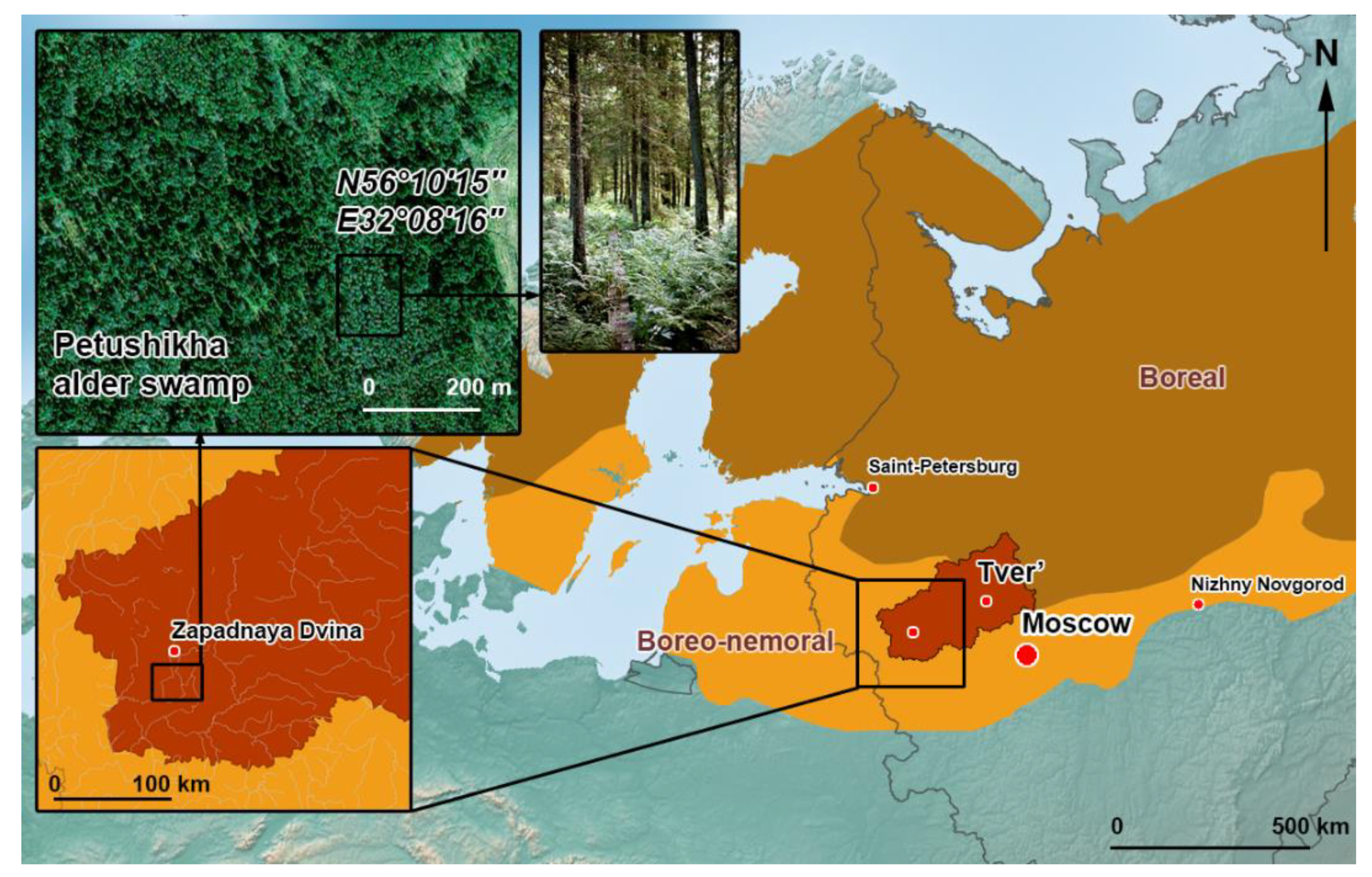
2.1. Study Location
2.2. Vegetation Cover
2.3. Soil Cover
2.4. Field Measurements
2.5. Description of Rsoil Model
2.6. Data Analysis
3. Results
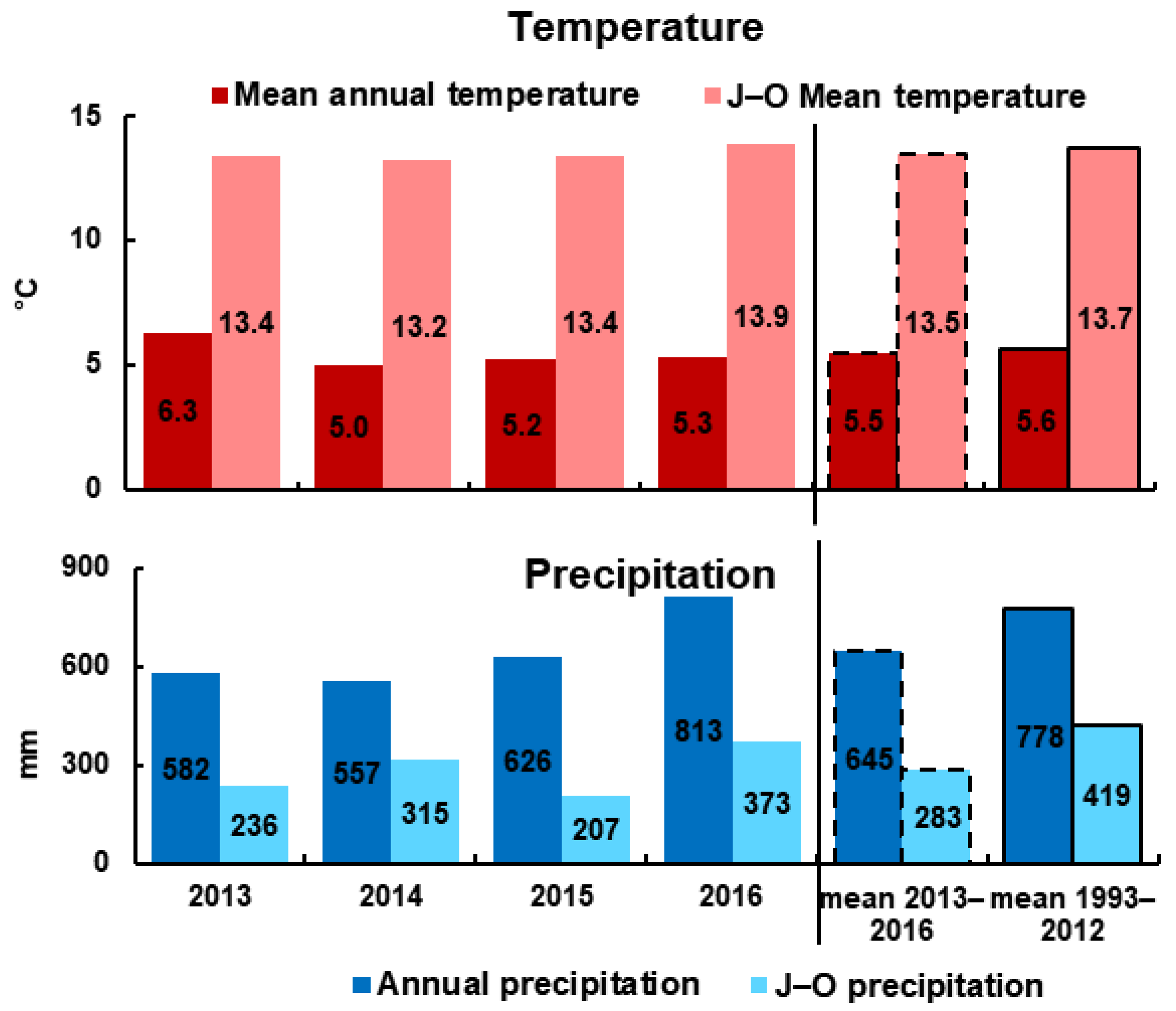
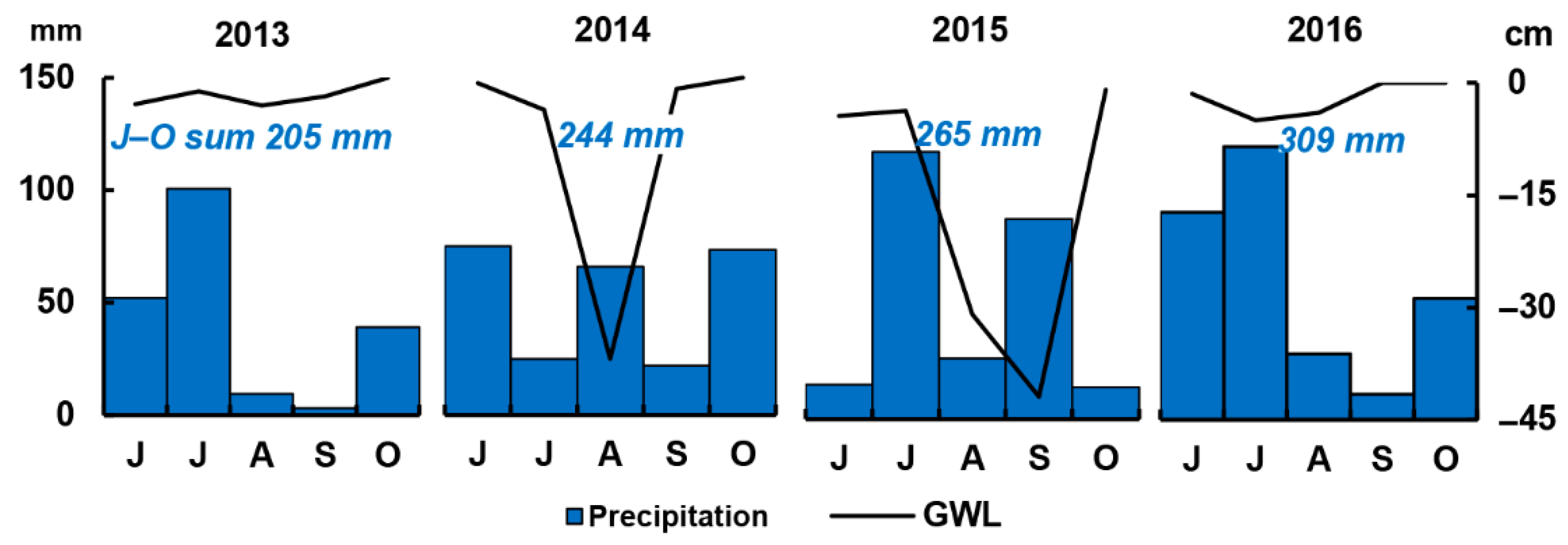
3.1. Meteorological and Environmental Conditions
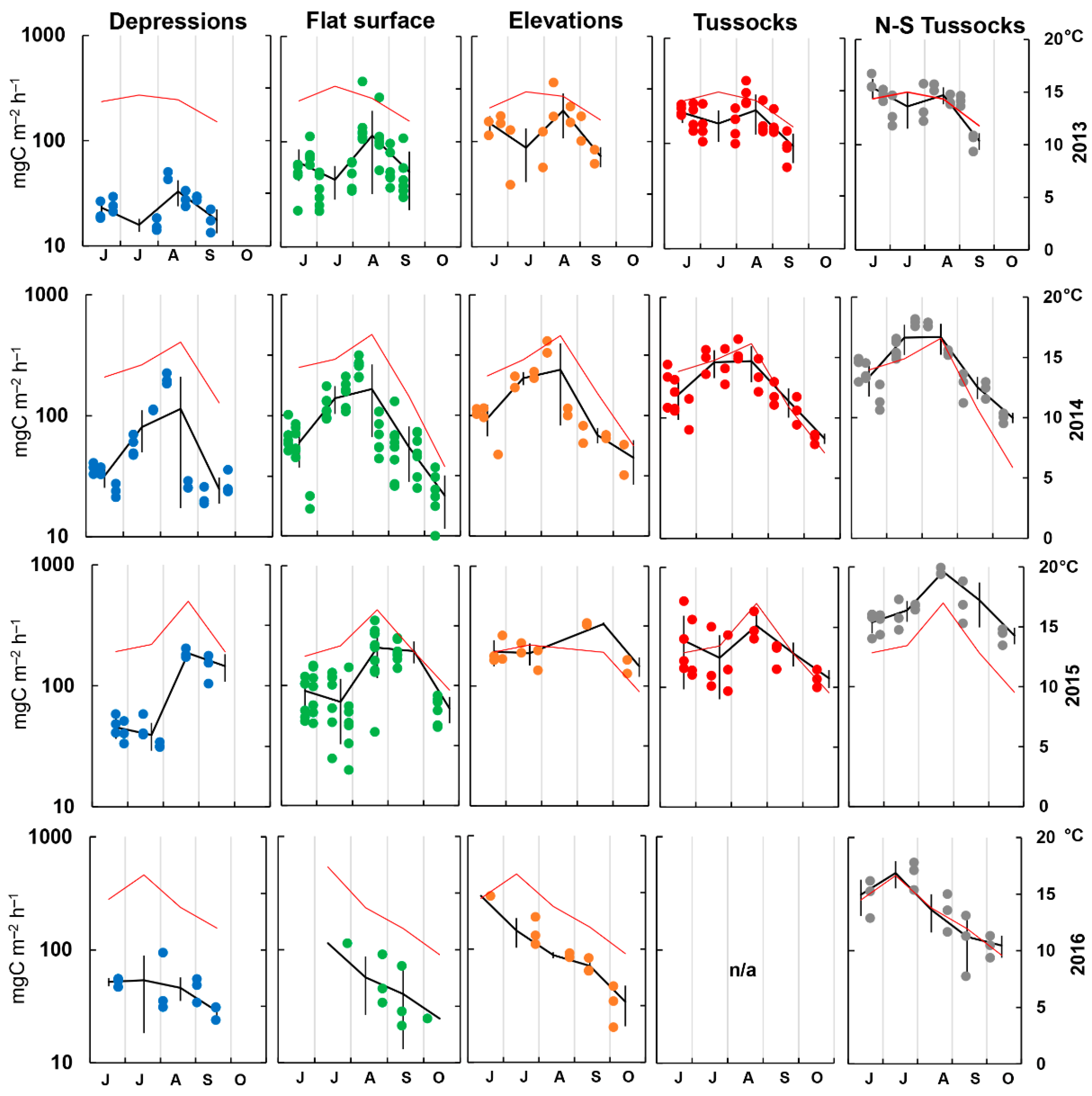
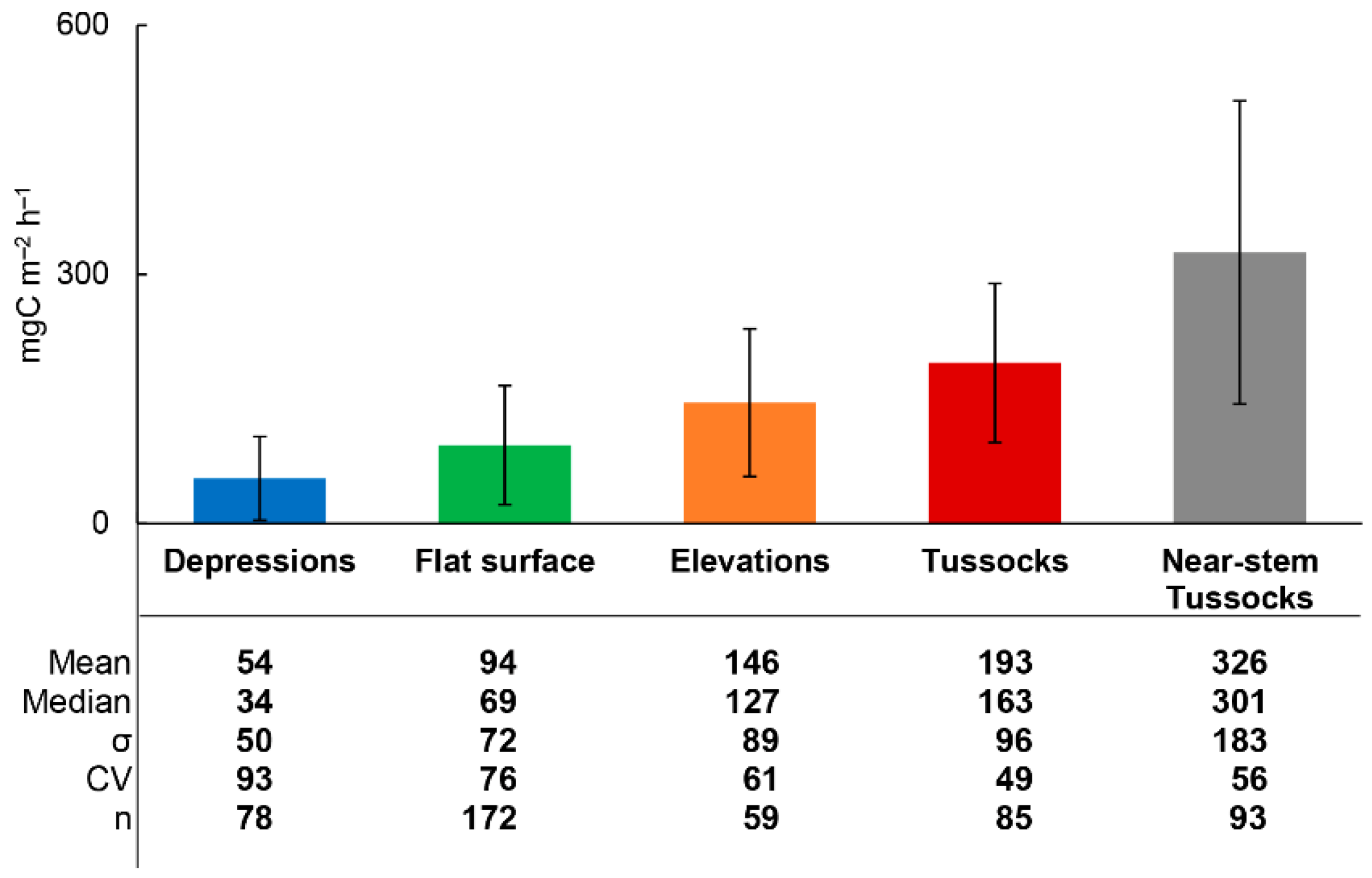
3.2. Rsoil Fluxes and Environmental Parameters
3.3. Rsoil Flux Data Analysis
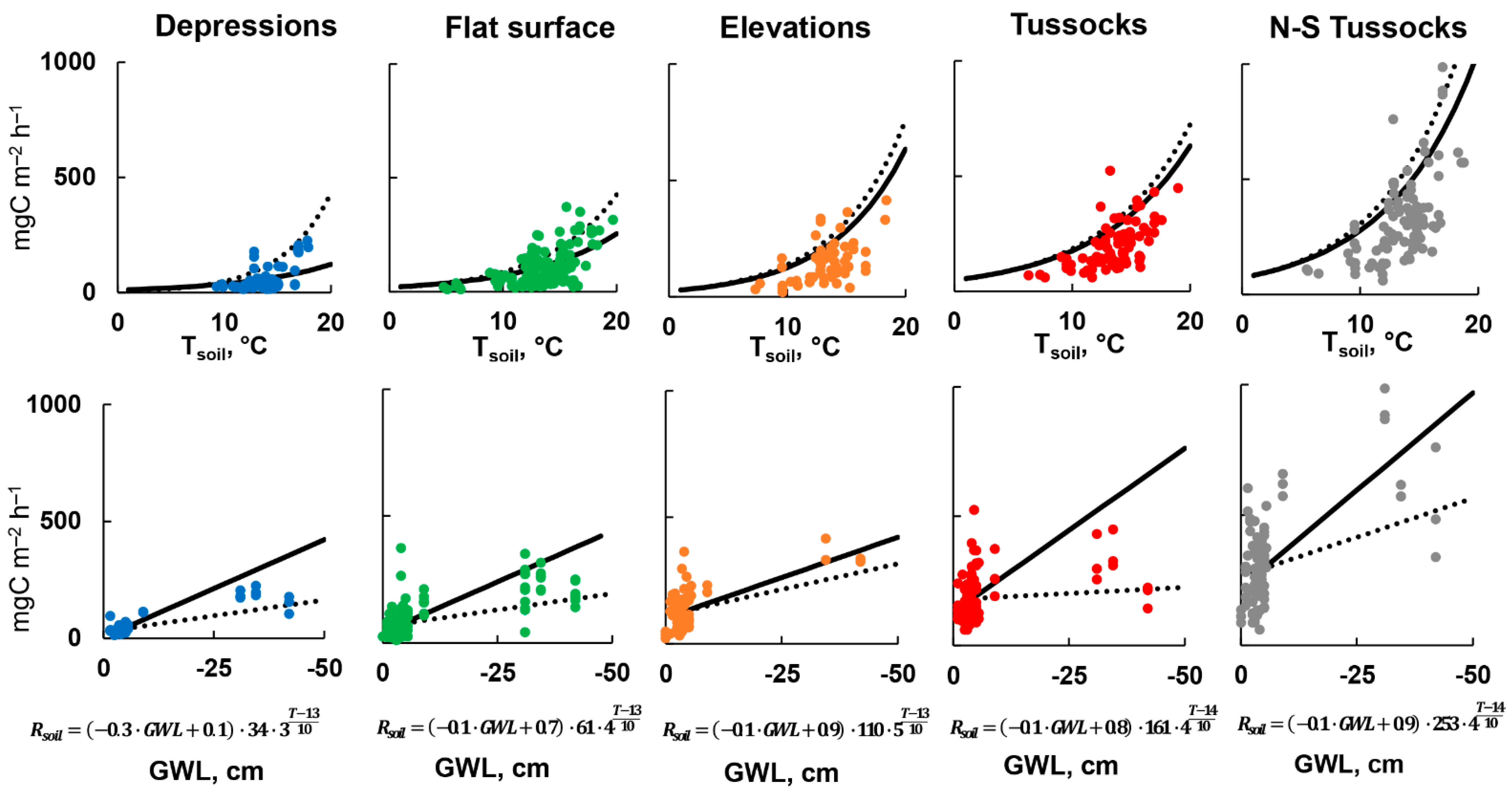
3.4. Parametrization of Rsoil Model
3.5. Cross Validation of Rsoil Model
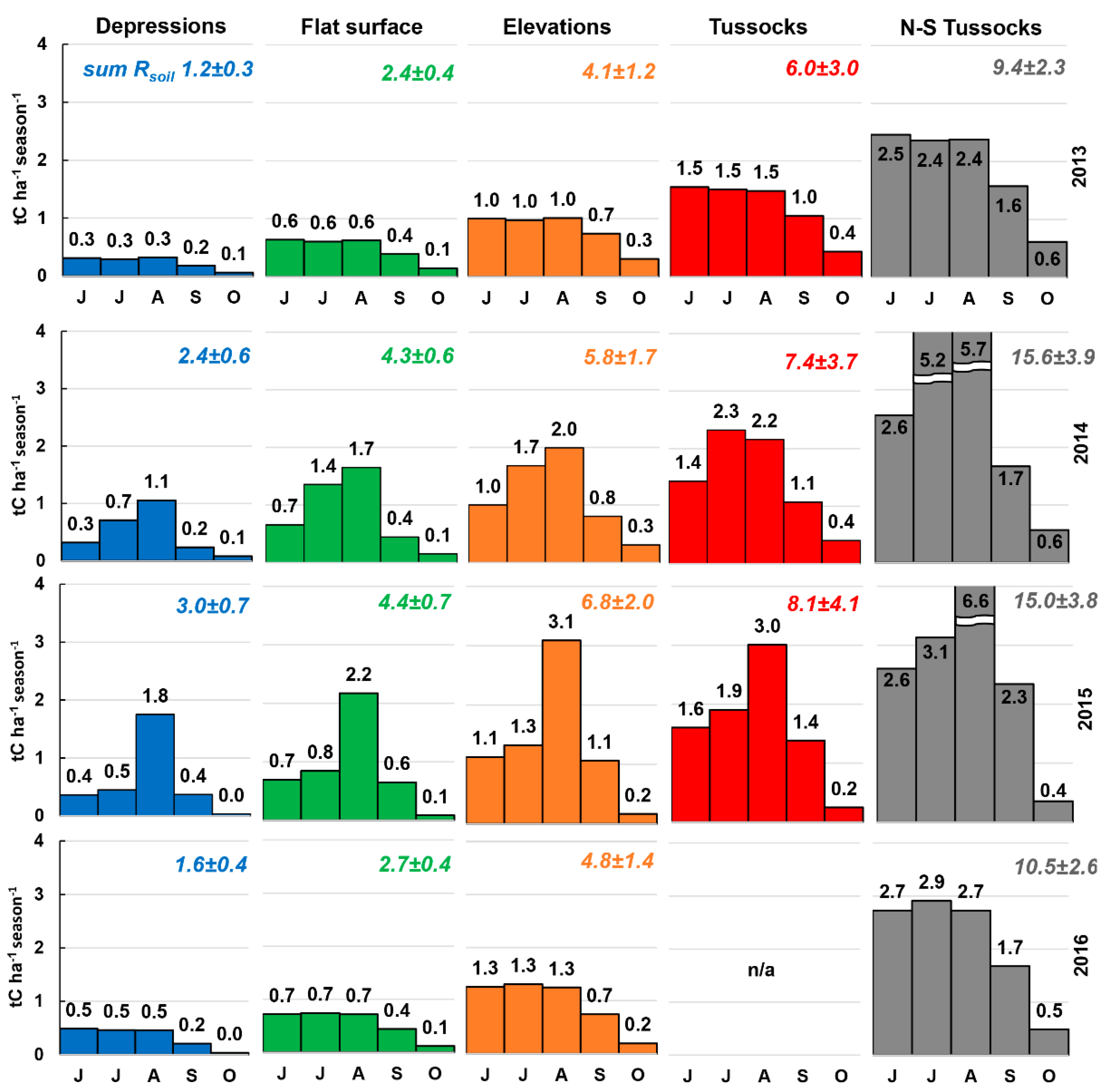
3.6. Total Rsoil per Season
4. Discussion
4.1. Rsoil Fluxes and Their Seasonal Dynamics
4.2. Influence of Extra-Dry Periods on Rsoil Model Parameterization
4.3. Influence of Extra Dry Periods on Seasonal Rsoil
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parish, F.; Sirin, A.; Charman, D.; Joosten, H.; Minayeva, T.; Silvius, M.; Stringer, L. Assessment on Peatlands, Biodiversity and Climate Change; Global Environment Centre and Wetlands International: Wageningen, The Netherlands, 2008. [Google Scholar]
- Yu, Z.C. Northern peatland carbon stocks and dynamics: A review. Biogeosciences 2012, 9, 4071–4085. [Google Scholar] [CrossRef]
- Loisel, J.; Yu, Z.; Beilman, D.W.; Camill, P.; Alm, J.; Amesbury, M.J.; Anderson, D.; Andersson, S.; Bochicchio, C.; Barber, K.; et al. A database and synthesis of northern peatland soil properties and Holocene carbon and nitrogen accumulation. Holocene 2014, 24, 1028–1042. [Google Scholar] [CrossRef]
- Drösler, M.; Verchot, L.; Freibauer, A.; Pan, G.; Evans, C.D.; Bourbonniere, R.A.; Alm, J.P.; Page, S.; Agus, F.; Hergoualc’h, K.; et al. Chapter 2: Drained inland organic soils. In 2013 Supplement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Wetlands; Hiraishi, T., Krug, T., Tanabe, K., Srivastava, N., Baasansuren, J., Fukuda, M., Troxler, T.G., Eds.; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2014. [Google Scholar]
- Joosten, H.; Sirin, A.; Couwenberg, J.; Laine, J.; Smith, P. The role of peatlands in climate regulation. In Peatland Restoration and Ecosystem Services; Cambridge University Press (CUP): Cambridge, UK, 2016; pp. 63–76. [Google Scholar]
- Leifeld, J.; Menichetti, L. The underappreciated potential of peatlands in global climate change mitigation strategies. Nat. Commun. 2018, 9, 1–7. [Google Scholar] [CrossRef]
- Jurasinski, G.; Ahmad, S.; Anadon-Rosell, A.; Berendt, J.; Beyer, F.; Bill, R.; Blume-Werry, G.; Couwenberg, J.; Günther, A.; Joosten, H.; et al. From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach. Soil Syst. 2020, 4, 14. [Google Scholar] [CrossRef]
- Bubier, J.; Costello, A.; Moore, T.R.; Roulet, N.T.; Savage, K. Microtopography and methane flux in boreal peatlands, northern Ontario, Canada. Can. J. Bot. 1993, 71, 1056–1063. [Google Scholar] [CrossRef]
- Alm, J.; Korhola, A.; Turunen, J.; Saarnio, S.; Jungner, H.; Tolonen, K.; Silvola, J. Past and future atmospheric carbon gas (CO2, CH4) exchange in boreal peatlands. Int. Peat J. 1999, 9, 127–135. [Google Scholar]
- Godwin, C.M.; McNamara, P.J.; Markfort, C.D. Evening methane emission pulses from a boreal wetland correspond to con-vective mixing in hollows. J. Geophys. Res. Biogeosci. 2013, 118, 994–1005. [Google Scholar] [CrossRef]
- Frolking, S.; Roulet, N.T.; Moore, T.R.; Richard, P.J.H.; Lavoie, M.; Muller, S.D. Modeling Northern Peatland Decomposition and Peat Accumulation. Ecosystems 2001, 4, 479–498. [Google Scholar] [CrossRef]
- Kleinen, T.; Brovkin, V.; Schuldt, R.J. A dynamic model of wetland extent and peat accumulation: Results for the Holocene. Biogeosciences 2012, 9, 235–248. [Google Scholar] [CrossRef]
- Zhang, L.; Gałka, M.; Kumar, A.; Liu, M.; Knorr, K.-H.; Yu, Z.-G. Plant succession and geochemical indices in immature peatlands in the Changbai Mountains, northeastern region of China: Implications for climate change and peatland development. Sci. Total Environ. 2020, 773, 143776. [Google Scholar] [CrossRef]
- Matthews, E.; Fung, I. Methane emission from natural wetlands: Global distribution, area, and environmental characteristics of sources. Glob. Biogeochem. Cycles 1987, 1, 61–86. [Google Scholar] [CrossRef]
- Panikov, N.S.; Dedysh, S. Cold season CH4 and CO2 emission from boreal peat bogs (West Siberia): Winter fluxes and thaw activation dynamics. Glob. Biogeochem. Cycles 2000, 14, 1071–1080. [Google Scholar] [CrossRef]
- Moore, P.D. The future of cool temperate bogs. Environ. Conserv. 2002, 29, 3–20. [Google Scholar] [CrossRef]
- Vompersky, S.E.; Sirin, A.A.; Sal’nikov, A.A.; Tsyganova, O.P.; Valyaeva, N.A. Estimation of the areas of peatland and palu-dified Shallow-peat forest in Russia. Lesovedenie 2011, 5, 3–11. (In Russian) [Google Scholar]
- Utkin, A.I.; Lindeman, G.V.; Nekrasov, V.N.; Simolin, A.V. Forest of Russia: An Encyclopedia; Great Russian Encyclopedia: Moscow, Russia, 1995; 447p. (In Russian) [Google Scholar]
- Yakovlev, F.S. Black alder in the Kivach nature reserve and adjacent areas. Proc. Kivach State Reserve 1973, 2, 23–31. (In Russian) [Google Scholar]
- Sirin, A.; Minayeva, T.; Yurkovskaya, T.; Kuznetsov, O.; Smagin, V.; Fedotov, Y. Russian Federation (European Part). In Mires and Peatlands of Europe: Status, Distribution, and Conservation; Joosten, H., Tanneberger, F., Moen, A., Eds.; Schweizerbart Science Publishers: Stuttgart, Germany, 2017; pp. 589–616. [Google Scholar]
- Bikbaev, I.G.; Martynenko, V.B.; Shirokikh, P.S.; Muldashev, A.A.; Baisheva, E.Z.; Minaeva, T.Y.; Sirin, A.A. Communities of the class Alnetea Glutinosae in the southern Ural region. Proceedings of the Samara Scientific. Cent. RAS 2017, 19, 110–119. (In Russian) [Google Scholar]
- Baginsky, V.F.; Katkov, N.N. Ecological features, structure and the forecast of changes of typological structure of black alder forests in Belarus. Eco–Potential 2013, 1, 84–92. (In Russian) [Google Scholar]
- Grigora, I.M. Alder forest swamps of Ukrainian Polesye and their typology. Lesovedenie 1976, 5, 12–21. (In Russian) [Google Scholar]
- Laivinsh, M.Y. Black alder forest communities (Carici elongatae Alnetum Koch. 1926) of lake islands in Latvia. Bot. J. 1985, 70, 1199–1208. (In Russian) [Google Scholar]
- Timofeev, D.I. Biological and ecological features of black alder forests in boreal forests. Lesovedenie 1993, 1, 35–41. (In Russian) [Google Scholar]
- Mander, Ü.; Maddison, M.; Soosaar, K.; Teemusk, A.; Kanal, A.; Uri, V.; Truu, J. The impact of a pulsing groundwater table on greenhouse gas emissions in riparian grey alder stands. Environ. Sci. Pollut. Res. 2015, 22, 2360–2371. [Google Scholar] [CrossRef]
- Wroński, K.T. Spatial variability of CO2 fluxes from meadow and forest soils in western part of Wzniesienia Łódzkie (Łódź Hills). For. Res. Pap. 2018, 79, 45–58. [Google Scholar] [CrossRef]
- Ullah, S.; Frasier, R.; Pelletier, L.; Moore, T.R. Greenhouse gas fluxes from boreal forest soils during the snow-free period in Quebec, Canada. Can. J. For. Res. 2009, 39, 666–680. [Google Scholar] [CrossRef]
- Sulman, B.N.; Desai, A.R.; Schroeder, N.M.; Ricciuto, D.; Barr, A.; Richardson, A.D.; Flanagan, L.B.; LaFleur, P.M.; Tian, H.; Chen, G.; et al. Impact of hydrological variations on modeling of peatland CO2 fluxes: Results from the North American Carbon Program site synthesis. J. Geophys. Res. Space Phys. 2012, 117, 1. [Google Scholar] [CrossRef]
- Kutsch, W.L.; Staack, A.; Wötzel, J.; Middelhoff, U.; Kappen, L. Field measurements of root respiration and total soil respira-tion in an alder forest. New Phytol. 2001, 150, 157–168. [Google Scholar] [CrossRef]
- Vasilevich, V.I.; Shchukina, K.V. Black alder forests of the northwest of European Russia. Bot. J. 2001, 86, 15–26. (In Russian) [Google Scholar]
- Parker, G.R.; Schneider, G. Biomass and Productivity of an Alder Swamp in Northern Michigan. Can. J. For. Res. 1975, 5, 403–409. [Google Scholar] [CrossRef]
- Bulatov, M.I. Distribution of black alder in the Moscow region. Lesovedenie 1980, 5, 108–109. (In Russian) [Google Scholar]
- Sarycheva, E.P. Spatial structure and species diversity of black alder forests of the Nerusso–Desnyanskiy Poleye. Bot. J. 1998, 83, 65–71. (In Russian) [Google Scholar]
- Katunova, V.V. Edapho-Phytocenotic Characteristics of Black Alder Forests in the Middle Zone of the European Part of Russia (on the Example of the Nizhny Novgorod Region and the Republic of Mordovia), Actual Problems of Forestry in the Nizhny Novgorod Volga Region and Ways to Solve Them, Nizhny Novgorod; Nizhny Novgorod State Agricultural Academy: Nizhny Novgorod, Russia, 2005; pp. 81–88. (In Russian) [Google Scholar]
- Šourková, M.; Frouz, J.; Šantrùčková, H. Accumulation of carbon, nitrogen and phosphorus during soil formation on alder spoil heaps after brown-coal mining, near Sokolov (Czech Republic). Geoderma 2005, 124, 203–214. [Google Scholar] [CrossRef]
- Kutenkov, S.A. Swamp black alder forests of Karelia. Lesovedenie 2010, 1, 12–21. [Google Scholar]
- Naumov, A.V.; Efremova, T.T.; Efremov, S.P. On the issue of carbon dioxide and methane emissions from bog soils of south-ern Vasyugane. Sib. Ekol. Zhurn. 1994, 3, 269–274. (In Russian) [Google Scholar]
- Glagolev, M.V.; Golovatskaya, E.A.; Shnyrev, N.A. Emission of greenhouse gases at the territory of west Siberia. Sib. Ekol. Zhurn. 2007, 2, 197–210. (In Russian) [Google Scholar]
- Glagolev, M.V.; Chistotin, M.V.; Shnyrev, N.A.; Sirin, A.A. Summer–autumn emission of carbon dioxide and methane from drained peatlands, changed during economic use and natural bogs (on the example of a site in the Tomsk region). Agrokhimiya 2008, 5, 46–58. (In Russian) [Google Scholar]
- Naumov, A.V. Soil Respiration: Components, Ecological Functions, Geographic Patterns; Publishing House of SB RAS: Novosibirsk, Russia, 2009; 208p. (In Russian) [Google Scholar]
- Glagolev, M.; Kleptsova, I.; Filippov, I.; Maksyutov, S.; Machida, T. Regional methane emission from West Siberia mire land-scapes. Environ. Res. Lett. 2011, 6, 045214. [Google Scholar] [CrossRef]
- Kim, H.-S.; Maksyutov, S.; Glagolev, M.V.; Machida, T.; Patra, P.; Sudo, K.; Inoue, G. Evaluation of methane emissions from West Siberian wetlands based on inverse modeling. Environ. Res. Lett. 2011, 6, 035201. [Google Scholar] [CrossRef][Green Version]
- Glukhova, T.V.; Vompersky, S.E.; Kovalev, A.G. Emission of CO2 from the surface of oligotrophic bogs with due account for their microrelief in the southern taiga of European Russia. Eurasian Soil Sci. 2013, 46, 1172–1181. [Google Scholar] [CrossRef]
- Bohn, T.J.; Melton, J.R.; Ito, A.; Kleinen, T.; Spahni, R.; Stocker, B.D.; Zhang, B.; Zhu, X.; Schroeder, R.; Glagolev, M.V.; et al. WETCHIMP-WSL: Intercomparison of wetland methane emissions models over West Siberia. Biogeosciences 2015, 12, 3321–3349. [Google Scholar] [CrossRef]
- Vompersky, S.E.; Sirin, A.A.; Glukhov, A.I. Formation and Regime of Runoff during Hydroforestry; Nauka: Moscow, Russia, 1988; 168p. (In Russian) [Google Scholar]
- Stuiver, M.; Reimer, P.J. Extended 14C database and revised CALIB radio–carbon calibration program. Radiocarbon 1993, 35, 215–230. [Google Scholar] [CrossRef]
- Lloyd, J.; Taylor, J.A. On the Temperature Dependence of Soil Respiration. Funct. Ecol. 1994, 8, 315. [Google Scholar] [CrossRef]
- Panikov, N.S.; Blagodatsky, S.A.; Blagodatskaya, J.V.; Glagolev, M.V. Determination of microbial mineralization activity in soil by modified Wright and Hobbie method. Biol. Fertil. Soils 1992, 14, 280–287. [Google Scholar] [CrossRef]
- Glagolev, M.V.; Sabrekov, A.F. On a problems related to a concept of soil thermal diffusivity and estimation of its dependence on soil moisture. Environ. Dyn. Glob. Clim. Chang. 2019, 10, 68–85. [Google Scholar] [CrossRef]
- Striegl, R.G.; Wickland, K.P. Effects of a clear-cut harvest on soil respiration in a jack pine-lichen woodland. Can. J. For. Res. 1998, 28, 534–539. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Biryukova, O.; Kuznetzova, T.; Mölter, K.; Kandeler, E.; Stahr, K. Carbon partitioning in plant and soil, car-bon dioxide fluxes and enzyme activities as affected by cutting ryegrass. Biol. Fertil. Soils 2002, 35, 348–358. [Google Scholar]
- Tanaka, K.; Hashimoto, S. Plant canopy effects on soil thermal and hydrological properties and soil respiration. Ecol. Model. 2006, 196, 32–44. [Google Scholar] [CrossRef]
- Qian, J.H.; Doran, J.W.; Walters, D.T. Maize plant contributions to root zone available carbon and microbial transformations of nitrogen. Soil Biol. Biochem. 1997, 29, 1451–1462. [Google Scholar] [CrossRef]
- Helal, H.M.; Sauerbeck, D.R. Influence of plant roots on C and P metabolism in soil. Plant Soil 1984, 76, 175–182. [Google Scholar] [CrossRef]
- Helal, H.M.; Sauerbeck, D. Effect of plant roots on carbon metabolism of soil microbial biomass. J. Plant Nutr. Soil Sci. 1986, 149, 181–188. [Google Scholar] [CrossRef]
- Krauss, K.W.; Whitbeck, J.L. Soil Greenhouse Gas Fluxes during Wetland Forest Retreat along the Lower Savannah River, Georgia (USA). Wetlands 2011, 32, 73–81. [Google Scholar] [CrossRef]
- Kimball, J.S.; Thornton, P.E.; White, M.A.; Running, S.W. Simulating forest productivity and surface-atmosphere carbon ex-change in the BOREAS study region. Tree Physiol. 1997, 17, 589–599. [Google Scholar] [CrossRef]
- Sundari, S.; Hirano, T.; Yamada, H.; Kusin, K.; Limin, S. Effect of groundwater level on soil respiration in tropical peat swamp forests. J. Agric. Meteorol. 2012, 68, 121–134. [Google Scholar] [CrossRef]
- Glagolev, M.V.; Ilyasov, D.V.; Terentyeva, I.E.; Sabrekov, A.F.; Krasnov, O.A.; Maksutov, S.S. Methane and carbon dioxide fluxes in the waterlogged forests of Western Siberian southern and middle taiga subzones. Opt. Atmos. Okeana 2017, 30, 301–309. (In Russian) [Google Scholar]
- Jovani-Sancho, A.J.; Cummins, T.; Byrne, K.A. Soil respiration partitioning in afforested temperate peatlands. Biogeochemistry 2018, 141, 1–21. [Google Scholar] [CrossRef]
- Kurganova, I.; De Gerenyu, V.L.; Rozanova, L.; Sapronov, D.; Myakshina, T.; Kudeyarov, V. Annual and seasonal CO2 fluxes from Russian southern taiga soils. Tellus B Chem. Phys. Meteorol. 2003, 55, 338–344. [Google Scholar] [CrossRef]
- Kurganova, I.N.; Lopes de Gerenyu, V.O.; Myakshina, T.N.; Sapronov, D.V.; Romashkin, I.V.; Zhmurin, V.A.; Kudeyarov, V.N. Natural and Model Assessments of Respiration of Forest Sod–Podzolic Soil in the Prioksko–Terrasny Biosphere Reserve. Lesovedenie 2019, 5, 435–448. (In Russian) [Google Scholar]
- Kozlov, M. Environmental Research Planning: Theory and Practical Guidelines; Litres: Moscow, Russia, 2018. (In Russian) [Google Scholar]
- IPCC. Climate Change 2007: Synthesis Report. In Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2007; 104p. [Google Scholar]
- Sheikh, M.A.; Kumar, M.; Todaria, N.P.; Bhat, J.A.; Kumar, A.; Pandey, R. Contribution of Cedrus deodara forests for climate mitigation along altitudinal gradient in Garhwal Himalaya, India. Mitig. Adapt. Strat. Glob. Chang. 2021, 26, 1–19. [Google Scholar] [CrossRef]

| Year | DEP | FL | EL | TUS | STUS |
|---|---|---|---|---|---|
| c, d, R2, n | |||||
| 2013 | 0.3, 6.9, 0.7, 20 | 0.4, 6.8, 0.5, 48 | 0.2, 9.3, 0.2, 16 | 0.3, 6.8, 0.4, 32 | 0.4, 6.7, 0.6, 24 |
| 2014 | 0.5, 1.4, 0.4, 28 | 0.8, −5.8, 0.6, 66 | 0.5, 2.2, 0.3, 19 | 0.7, −4.0, 0.6, 31 | 0.9, −8.7, 0.7, 33 |
| 2015 | 3.7, −76, 0.5, 18 | 0.8, −5.9, 0.5, 50 | 0.7, −2.7, 0.9, 12 | 0.8, −6.1, 0.6, 22 | 0.8, −6.2, 0.6, 21 |
| 2016 | 0.6, −2.8, 0.7, 12 | 0.5, −0.1, 0.8, 8 | 0.6, −1.1, 0.9, 12 | − | 0.6, −0.6, 0.9, 15 |
| Parameter | DEP | CV | FL | CV | EL | CV | TUS | CV | STUS | CV |
|---|---|---|---|---|---|---|---|---|---|---|
| a | −0.07 | 8 | −0.04 | 7 | −0.03 | 15 | −0.01 | 38 | −0.02 | 14 |
| b | 0.69 | 5 | 0.90 | 2 | 1.0 | 4 | 1.0 | 3 | 1.0 | 3 |
| Rref | 40 | 2 | 73 | 1 | 120 | 2 | 171 | 2 | 277 | 2 |
| Q10 | 8.8 | 10 | 4.9 | 5 | 5.0 | 9 | 4.0 | 7 | 4.0 | 6 |
| Tref | 13.2 | - | 13.4 | - | 13.2 | - | 13.5 | - | 13.5 | - |
| Ecosystem Type | Location | Rsoil, mgC m−2 h−1 | Reference |
|---|---|---|---|
| Eutrophic forested fen | Russia (West Siberia) | 128 | [38] |
| Eutrophic fen | 25–370 | [39] | |
| Periodically waterlogged forest | 174–414 | [60] | |
| Alder swamp | Germany | 100–1008 | [30] |
| Eutrophic fen | USA | 150–350 | [29] |
| Swamp fen | Ireland | 50–400 | [61] |
| Swamp fen | USA | 29–59 | [57] |
| Forest with alder shrub | Canada | 42 | [58] |
| Alder swamp | Canada | 48–69 | [28] |
| Swamp fen | Indonesia | 115–220 | [59] |
| Forest | European Russia | 62–264 | [62] |
| 101 | [62] | ||
| Alder swamp | 54–326 | Current study |
| Parameter | DEP | CV | FL | CV | EL | CV | TUS | CV | STUS | CV |
|---|---|---|---|---|---|---|---|---|---|---|
| a | −0.25 | 11 | −0.13 | 9 | −0.06 | 26 | −0.08 | 17 | −0.06 | 24 |
| b | 0.11 | 95 | 0.66 | 7 | 0.94 | 7 | 0.80 | 7 | 0.88 | 6 |
| Rref | 34 | 2 | 61 | 1 | 110 | 2 | 161 | 2 | 253 | 2 |
| Q10 | 3.3 | 12 | 3.6 | 6 | 5.1 | 10 | 3.6 | 8 | 3.8 | 7 |
| Tref | 13.2 | − | 13.4 | − | 13.2 | − | 13.5 | − | 13.5 | − |
| Year | DEP | σ | FL | σ | EL | σ | TUS | σ | STUS | σ | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2013 | Rsoil | 0.7 | 0.9 | 1.9 | 0.4 | 3.8 | 1.7 | 5.2 | 1.8 | 8.5 | 3.3 |
| diff., % | −39 | −20 | −5 | −14 | −9 | ||||||
| 2014 | Rsoil | 1.7 | 2.0 | 3.4 | 0.8 | 5.0 | 2.3 | 7.6 | 2.6 | 13.3 | 5.2 |
| diff., % | −31 | −21 | −13 | +3 | −15 | ||||||
| 2015 | Rsoil | 1.0 | 1.1 | 2.6 | 0.6 | 5.0 | 2.3 | 6.8 | 2.3 | 11.3 | 4.4 |
| diff., % | −68 | −42 | −26 | −16 | −25 | ||||||
| 2016 | Rsoil | 1.0 | 1.1 | 2.2 | 0.5 | 4.7 | 2.1 | − | − | 9.6 | 3.7 |
| diff., % | −42 | −19 | −2 | −9 | |||||||
| mean | Rsoil | 1.4 | 2.5 | 4.6 | 6.5 | 10.7 | |||||
| diff., % | −45 | −25 | −12 | −9 | −14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glukhova, T.V.; Ilyasov, D.V.; Vompersky, S.E.; Golovchenko, A.V.; Manucharova, N.A.; Stepanov, A.L. Soil Respiration in Alder Swamp (Alnus glutinosa) in Southern Taiga of European Russia Depending on Microrelief. Forests 2021, 12, 496. https://doi.org/10.3390/f12040496
Glukhova TV, Ilyasov DV, Vompersky SE, Golovchenko AV, Manucharova NA, Stepanov AL. Soil Respiration in Alder Swamp (Alnus glutinosa) in Southern Taiga of European Russia Depending on Microrelief. Forests. 2021; 12(4):496. https://doi.org/10.3390/f12040496
Chicago/Turabian StyleGlukhova, Tamara V., Danil V. Ilyasov, Stanislav E. Vompersky, Alla V. Golovchenko, Natalia A. Manucharova, and Alexey L. Stepanov. 2021. "Soil Respiration in Alder Swamp (Alnus glutinosa) in Southern Taiga of European Russia Depending on Microrelief" Forests 12, no. 4: 496. https://doi.org/10.3390/f12040496
APA StyleGlukhova, T. V., Ilyasov, D. V., Vompersky, S. E., Golovchenko, A. V., Manucharova, N. A., & Stepanov, A. L. (2021). Soil Respiration in Alder Swamp (Alnus glutinosa) in Southern Taiga of European Russia Depending on Microrelief. Forests, 12(4), 496. https://doi.org/10.3390/f12040496






