Abstract
Cupressaceae pollen is responsible for winter respiratory allergies in the Mediterranean area. Pollen grains of this diverse family share the same characteristics under light microscopy. Consequently, the partial contribution of each Cupressaceae species to the airborne pollen spectrum cannot be determined with conventional aerobiological methods. Studies on major aeroallergens offer better information on the allergic sensitization and appearance of symptoms in patients sensitized to airborne pollen and spores. Our aim was to determine if airborne Cupressaceae pollen concentrations correspond to aerodynamics of the major allergen of Cupressus (Cup a 1), as well as the aeroallergen correlation with different-sized particles. The air was sampled in two cities of Southern Spain (Granada and Córdoba) using the Hirst-type volumetric sampler for airborne pollen detection during 2006 to 2008 and the Andersen Cascade Impactor for aeroallergen detection during the main pollination period of Cupressaceae in the same period. The samples were analyzed by the methodology proposed by the Spanish Aerobiology Network (REA), the minimum requirements of the European Aeroallergen Society (EAS) for pollen, and by ELISA immunoassay to detect airborne Cup a 1. The distribution patterns of airborne Cupressaceae pollen and Cup a 1 were observed throughout the sampling period, although with some irregular oscillations. Cupressaceae pollen and Cup a 1 showed positive and significant correlation during the period of maximum concentrations (January to March). However, the results of this study showed that the period of exposure of aeroallergens depends on the Cupressus species and their abundance in cities. According to the allergy potential obtained, species like C. arizonica could release more allergens than other species like C. sempervirens.
1. Introduction
Cupressaceae pollen is an important cause of pollinosis. In the Mediterranean region, it is responsible for winter respiratory allergies in a period when few allergenic plants are flowering [1,2,3]. For example, in southeast France [4], Cupressaceae pollen is the fourth most sensitizing allergen source, after mites, grass pollen, and Alternaria. A clinical study in southern Spain revealed that 30–35% of the population are sensitive to this pollen type [5,6]. Moreover, a recent 15-year clinical survey from northern Italy documented an increase in cypress pollen sensitization and allergy [7].
To date, the extent of this allergy within the population may have been underestimated because its symptoms might have been confused with cooccurring winter respiratory tract infections [8]. In earlier epidemiological studies, this type of pollen allergy seemed to be practically exclusive to Mediterranean areas. However, increases in prevalence in some areas may be caused by more extensive use of some Cupressaceae species [9] for gardening and reforestation in addition to the global rise in temperatures, due both to a higher pollen concentration and longer pollen season [10].
The main species of Cupressaceae family are present on all continents, except Antarctica [11]. The most common species around the Mediterranean basin are cultivated as ornamental trees in urban spaces, i.e., Italian cypress (Cupressus sempervirens L.), Arizona cypress (C. arizonica Greene), Monterrey cypress (C. macrocarpa Hartweg), and Mexican cypress (C. lusitanica Miller). Some species are also used for forest cultivation, i.e., C. macrocarpa in the North of the province of Cordoba [12]. Autochthonous plants, such as common juniper (Juniperus communis L., J. oxycedrus L.) and savin juniper (J. sabina L., J. phoenicea L., J. thurifera L.), are also present in the province but usually far from the cities.
Pollen morphology within non-papillate Cupressaceae (including Cupressus) is mainly stenopalynous, with spheroidal shape, psilate exine sculpture, and a circular thinning of the exine in the leptoma area. Due to the lack of genus- or species-specific morphological features, Cupressaceae pollen is not differentiated in aerobiological studies using conventional light microscopy. All species of this family are wind-pollinated and produce large amounts of light pollen, which enables its wide dispersal. Consequently, the partial contribution of each Cupressaceae species to the airborne pollen spectrum cannot be determined [13]. Nonetheless, the information generated is very useful and important for the prevention and avoidance of allergenic symptoms.
In the last decades, some aerobiologists have focused their investigations on the behavior of airborne pollen and aeroallergens, as well as the seasonal variability and the influence of meteorological variables on aeroallergens’ concentrations [14,15]. The knowledge of the dynamic of these particles is contributing to acquire information on the levels of airborne biological pollutants and, in consequence, to understand the symptoms of patients allergic to pollen. Several methodologies have been used, such as Cyclone, ChemVol, or Andersen collector, to study the allergen load in the atmosphere [16,17,18,19,20]. All these studies indicated that dynamics of aeroallergen and airborne pollen are directly related.
Actually, four allergens have been identified and characterized in C. arizonica pollen. The most important is Cup a 1 (pectate lyase), a glycoprotein recognized as a major allergen, with a PM of 43 kDa [21]. Cup a 1 is a specific allergen characterized by its high-carbohydrate and low-protein content, due to the physical-chemical properties of the pollen grains [22]. It is synthesized in the Golgi complex and transported by vesicles to the exine, intine, and orbicules that this pollen type has abroad [21]. Immunolocation assays determined that it could be implicated in the first steps of male–female gametophyte recognition [23] or involved in modeling tissue and pollen tube growth [23]. The high sequence identity and, consequently, high degree of cross reactivity among Cupressaceae family species suggest the use of Cup a 1 allergen as a representative marker of the entire family [24,25].
Proteins and glycoproteins from pollen can function as allergens, environmental particles interacting with the human immune system to produce an allergic response in predisposed individuals [26]. However, there is still no evidence whether the differences in symptoms could be explained by differences in the number or size of particles carrying airborne allergens [27]. Airborne particles’ matter (PM) is defined as a heterogeneous mixture of solid and liquid particles suspended in air that varies continuously in size and chemical composition in space and time [28]. Although the World Health Organization concluded that health risks are present at any level of particles [29], the Environmental Protection Agency (EPA) determined that particles may be classified in two size categories based on their size: PM10, as particulate matter with an aerodynamic diameter of 10 μm, and PM2.5, as fine particulate matter with an aerodynamic diameter of 2.5 μm [30]. The aim of the present study was to study the seasonal allergenic load in the atmosphere of Granada and Córdoba (southern Spain) in relation to Cupressaceae pollen and to demonstrate if there is a parallel dynamic between concentrations of this pollen type and airborne Cup a 1 allergen. Moreover, we determined the allergen distribution based on EPA categories (PM10 and PM2.5) to establish whether this distribution could be related with the allergy symptoms.
2. Materials and Methods
2.1. Area of Study
The aerobiological study was carried out in two cities of southern Spain: the University of Granada in the city center (37°11′ N, 03°35′ W; 685 m.a.s.l.) and the University of Córdoba in the outskirts of the northeastern city (37°50′ N, 04°45′ W; 123 m.a.s.l.). Although the climate in both cities is Mediterranean (with moderate annual temperature and summer drought), both cities are characterized by huge climatic contrasts’ oscillations in temperature (summer–winter and day–night) [31].
The main species of Cupressus present in these cities are C. sempervirens and C. arizonica, although it is possible to find C. lusitanica and C. macrocarpa as ornamental plants used occasionally in both urban spaces and surrounding. Although the flowering start of these species varies, their flowering periods are usually overlapped. Juniperus species also appear in both provinces [32], but far from the cities, and their pollen is not well represented in the urban air. Some papers have reported that Cupressaceae pollen season usually starts in this region during autumn, depending on the different species [2,3,6].
2.2. Sampling of Airborne Pollen and Allergens
For this study, an aerobiological monitoring of Cupressaceae pollen and Cup a 1 allergen was performed. Volumetric Hirst-type Spore Traps were used for pollen monitoring [33]. This sampler is capable of collecting pollen, spores, and other particles suspended in the air, with an aspiration of 10 L/min, comparable with the average human minute ventilation. In both cities, Hirst samplers were placed at 22–23 m above ground level. The counting method was that recommended by the Spanish Aerobiology Network (REA) [34] and the minimum requirements of the European Aerobiology Society (EAS) [35]. Terminology used in this paper follows the International Association for the Aerobiology (IAA) and EAS recommendations [36]. Due to the flowering period of the different species, in this study the agronomic years (September to August) were considered. The daily pollen data are expressed as daily average of pollen per cubic meter of air (pollen/m3). Aerobiological data are expressed in daily pollen and Annual Pollen Integral (APIn). Airborne pollen was studied from September 1993 to the end of August 2019. In this study, we calculated the Main Pollen Season (MPS) following Andersen, 1991 [37]. We considered the pollen season started when the sum of the daily mean pollen concentrations reached 2.5% of the annual sum and the end when the sum reached 97.5%.
The aeroallergen (Cup a 1) monitoring was performed from 15 January to 31 March, in the case of Granada during 2006 and 2008 and in Córdoba in 2007 and 2008, by means of a cascade impact collector [38]. The sampling took place 11:00 h–17:00 h when pollen concentrations were highest [6,39]. These collectors distributed the particles in different stages of size-fractions. The air flow through the impactor was controlled by a pump that drew in air at 30 L/min (Lanzoni SPS 3001, Italy). The size discrimination of the particles was possible by the variation in the air velocity, which was led sequentially through a series of fiberglass Whatman® filters (glass microfiber filters; type: GF/A) of descending pore size [38]. The samples were analyzed by ELISA technique [40]. Cup a 1 allergen was quantified using polyclonal antibody (Bial-Aristegui, Bilbao, Spain) [41]. The standard curve was drawn from dilutions of Cup a 1 allergen purified from C. arizonica pollen extract by affinity chromatography (Bial-Aristegui, Bilbao, Spain; [41]). For a reliable comparison of the results for the two samples of two cities, these collectors were positioned adjacent to the Hirst samplers. The results in all cases are expressed in nanograms of allergen per cubic meter of air (ng/m3). Aeroallergen data are expressed in daily allergen and Allergen Seasonal Integral (ASIn).
2.3. Meteorological Data
Daily series of temperature (maximum, mean, and minimum) and precipitation were used. Data were provided by the Andalusia Network of Agroclimatic Information (RIAA).
2.4. Statistical Analysis
Spearman’s correlation coefficients between Cup a 1 allergen and Cupressaceae pollen were calculated during the allergen study period. This analysis was carried out by using the IBM SPSS Statistic version 19.0.3.
3. Results
The main meteorological parameters, airborne pollen, and Cup a 1, during studied period, are shown in Table 1. Some differences between both cities studied were observed. In Granada, the weather was colder and drier (14.4–15.2 °C, 257.4–204.7 mm) than in Córdoba (16.9–17.0 °C, 521.0–660.0 mm) through the study time.

Table 1.
Meteorological data, Cupressaceae pollen, and Cup a 1 allergens’ parameter in Granada and Córdoba. APIn is considered from 1 September to 31 August, while SPIn is 15 January to 31 March.
As Table 1 shows, the aerobiological pattern of this pollen type was different. In Granada, while the period 2005–2006 presented the shortest pollen season (203 days) and lowest Annual Pollen Integral (APIn) (11,678 pollen/m3), 2007–2008 presented the longest presence (225 days) and highest APIn (26,487 pollen/m3). The peak day pollen concentration was also higher in the second year (1535 pollen/m3) than in the first (950 pollen/m3). In relationship with the MPS, both years were similar with a length of 94–101 days, starting in the middle of January and ending in April. Pollen season in Córdoba was longer (214 days) and lower in APIn (5901 pollen/m3) in 2006–2007 than in the 2007–2008 period, with pollen season (175 days) and APIn (13,061 pollen/m3). Again, the peak day pollen concentration was higher in 2007–2008 (1231 pollen/m3) than 2006–2007 (716 pollen/m3).
The comparative study of the two cities showed that higher pollen concentrations were detected during 2007–2008 in both sites than during prior periods. However, during 2007–2008, the peak pollen concentration day occurred with only one day of difference in both cities (16 February in Granada and 17 February in Córdoba).
Regarding the aeroallergen, higher concentrations where detected during years with lower airborne pollen in both cities (Table 1). The Allergen Season Integral (ASIn) and peak allergenic concentrations recorded differences. In Granada 2005–2006, the ASIn registered was 205,244.5 ng/m3 of Cup a 1, while it was detected at 49,653.5 ng/m3 in 2007–2008. On the other hand, in Córdoba during 2005–2006 the ASIn reached 78,419.8 ng/m3 but in 2007–2008 it registered 30,051.9 ng/m3 of Cup a 1. Comparing both cities during 2007–2008, as was previously observed on pollen, the peak day in Granada occurred one day before, on 16 February with 3730.1 ng/m3, while in Córdoba it occurred on 17 February, with 3830.1 ng/m3.
Regarding the results of pollen potency (pollen vs. allergens), in the same city this value was similar but between cities Granada registered lower pollen potency than Córdoba (21.3–2.1 and 42.2–37.3, during both years, respectively).
Finally, the study of Cup a 1 allergen with the different particle sizes showed that the highest allergen load was localized in PM2.5 in both cities (ranging from 94.9% to 74.3% in Granada and 96.0% to 90.3% in Córdoba).
Taking into account the average concentration and the absolute maximum of the Cupressaceae airborne pollen for 26 years (1993/94–2018/19), Figure 1 shows the annual pattern of Cupressaceae pollen in both cities. This pollen type presented a long pollen season, i.e., from September to June–July, with peaks in the curve from February and March. Granada registered a higher pollen concentration than Córdoba, with an explosive increase during February. Instead, in Córdoba, the important concentrations were registered since January with maximum in February. In addition, we observed a small peak, more remarkable in Granada, during October in both cities, with absolute maximum over 200 pollen grains/m3.
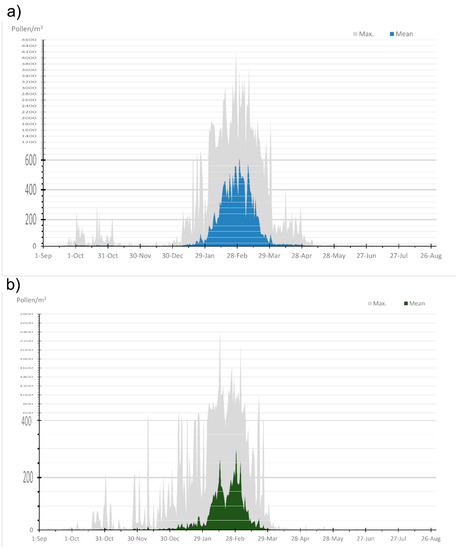
Figure 1.
Daily average pollen and absolute maximum concentration of Cupressaceae pollen during 26 years of study period (1993–1994 to 2018–2019) expressed in pollen/m3: (a) Granada; (b) Córdoba.
The aeroallergen dynamic of Cup a 1 in both cities was characterized by its continued presence during the studied period, although with irregular oscillations (Figure 2). In both cities, and during the two years, the period with higher pollen and aeroallergens’ concentrations coincided; it also occurred for peak days.
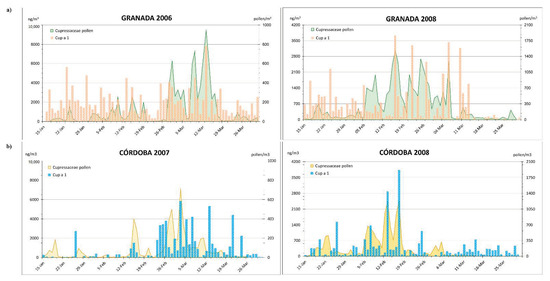
Figure 2.
Dynamic of Cupressaceae pollen (pollen/m3) and Cup a 1 (ng/m3) distributed in PM10 and PM2.5 in Granada (a) and Córdoba (b).
In Granada, positive and significant correlations between Cupressaceae pollen and Cup a 1 during 2005–2006 and 2007–2008 were registered (0.200 and 0.282; p < 0.05, respectively). In Córdoba, positive and significative correlations between both variables (0.515; p < 0.01) occurred during 2006–2007 but not for 2007–2008 (Table 2).

Table 2.
Correlation coefficients for Cupressaceae pollen, Cup a 1 allergen, and allergens in PM10 and PM 2.5, ** p < 0.01; * p < 0.05. The shadow and bold format indicate the results more relevant.
In relation with the PM particles, Cupressaceae pollen showed some significant correlation with PM2.5, while Cup a 1 registered significant correlation with both PM sizes in both cities and years.
4. Discussion
The study of airborne Cupressaceae pollen type showed that only summer can be considered a period free of this pollen type. In this study we considered agronomic years (September to August). The higher concentration, from January to March, corresponded to ornamental or forest cultivation trees (Cupressus sp.); lower concentrations were detected during autumn, probably due to autochthonous species of Juniperus sp., well distributed in the provinces [6,39]. The different pollination behavior of this genera makes it difficult to establish the main pollen season (MPS). In this study, MPS was determined. The results showed that in Granada the MPS was similar in the studied periods, starting in the second week of January and ending in the third week of April. However, in Córdoba the MPS was different to Granada and between the studied periods in the same city (Table 1). In Córdoba, the starting appeared one week and one month before and the ending was one month earlier than in Granada. As Hidalgo et al. [42] showed, in Córdoba, three species of Cupressus are present: C. arizonica, C. macrocarpa, and C. sempervirens. The male phenology of their flower showed that pollen release in C. arizonica takes place roughly three weeks earlier than in the other species and in C. macrocarpa slightly earlier than in C. sempervirens. As Table 1 shows, the start of the MPS could indicate the different flowering times of these species. In Córdoba, during the 2006–2007 period, the MPS started on 5 January, probably due to C. macrocarpa, while in 2007–2008, MPS started one month before (6 December), probably due to C. arizonica. In Granada, the main Cupressus sp. is C. sempervirens. The start of the MPS in the two periods was in the second week of January. Although there was a different pollination period for these Cupressus species, all of them were characterized by a short production in time but very abundant, making this airborne pollen the most frequent during winter, as reported in other cities of Spain, such as Badajoz [43], Toledo [44], Salamanca [45], Santiago de Compostela [46], and in other countries in the Mediterranean Basin, such as Italy [47], France [48], Portugal [1], or Turkey [49]. On the other hand, the presence of a small peak in October reaffirmed the different flowering period of Cupressaceae species, especially in autochthonous species of Juniperus [6].
Otherwise, Cupressaceae pollen type is an important cause of pollinosis during winter in the Mediterranean Basin [50]. Although the appearance of the allergic symptoms, such as rhinitis and asthma, depend on the daily pollen concentration threshold [51], they are very difficult to establish. As shown by Charpin et al. [24], researchers of different countries use threshold levels based on their scientific experiences. However, in the atmosphere, there are other particles that probably can provoke allergic symptoms [52]. This study showed similar dynamics for Cupressaceae pollen and Cup a 1, with significant correlations between these variables observed in both cities during the two years (except in Córdoba 2008–2009). These results agree with other aeroallergen studies performed with different taxa, i.e., allergens from grass [53], birch [54], pellitory [20], and olive [15]. However, regarding the annual intensity in both variables, the pollen integral (APIn) and the allergen integral (ASIn), opposite results were observed, i.e., years with higher APIn but lower ASIn and vice versa. This observation occurred in both Granada and Córdoba (Table 1). According to Plaza et al. [55] and De Linares et al. [20], some taxa (e.g., grasses and pellitory) under stress processes or extreme situations can produce a decrease in flowering intensity. In the case of Cupressaceae, similar results have been obtained.
In order to assess the risk period for people allergic to pollen, several studies have shown that airborne pollen concentration do not always represent the total risk period. This statement is because during periods without pollen there are probably smaller particles in the atmosphere. The presence of these smaller particles can vary, attending to different environmental and botanical factors, as well as with the year and location of collection [56]. For this reason, one important factor to study was the pollen potency (allergen/pollen grain), defined as the actual allergen release capacity of any pollen in the study [57]. Our results showed that the pollen potency was different in the studied cities. While in Granada it ranged from 21.1–21.3 during both periods, in Córdoba it was higher (42.2–37.3). As commented before, in these areas several species of Cupressus are present. While in Granada the main species is C. sempervirens, in Córdoba C. sempervirens, C. arizonica, and C. macrocarpa are well represented [12]. In fact, the MPS start in Córdoba was related to the last two species [42]. With this information, we concluded that, although Granada registered higher pollen concentration than Córdoba, the higher pollen potency in Cordoba probably supported that C. arizonica and C. macrocarpa were responsible for higher allergen emission. With this observation, it could be interesting to study the real allergenic capacity of these three species, due to the symptom risks being dependent on pollen concentrations and also on the different species of Cupressus in the urban areas.
The present study classified the aeroallergens according to size in EPA categories (PM10 and PM2.5). Our results showed major concentrations in the range of PM2.5. Some studies based on immunocytochemical methods [23,57] showed that in certain Cupressus species, the orbicules present in pollen exine contained allergen load. According to some studies, these orbicules are released and become part of the submicron fraction in the air [58]. This fact could explain why, in our study, the highest allergen concentrations were recorded in PM2.5.
Spearman correlation analysis showed a positive and significant correlation between Cupressaceae pollen vs. Cup a 1 with PM10 and PM2.5 in both studied years, except in 2008 in Córdoba, where Cup a 1 correlated only with PM2.5. During 2007–2008 in Córdoba, the MPS started in December and the duration was shorter (175) than other studied periods. This different behavior could be due to a major contribution of C. arizonica pollen. In our opinion, further airborne allergenic studies should be realized to understand the questions not answered in this study.
5. Conclusions
In conclusion, the main pollen season corresponded to different species of Cupressus sp. (common of the urban green spaces) from January to March, when it registered a short production in time but very abundant pollen release. However, the pollen season depends on the different species of Cupressus present in the cities. This study also supported that the main allergen load does not always derive from the pollen alone. Higher allergens were detected in the respirable fraction (PM2.5), probably of the orbicules, released from the pollen cones before and concurrently with the pollen.
Author Contributions
All authors participated in the design and interpretation of the data, wrote the paper, and participated in the revisions of it. C.D.L., A.M.V., and M.P.P. performed experiments and analysis. All authors have read and agreed to the published version of the manuscript.
Funding
This research was carried out within the framework of the research project funding by the Ministry of Science and Technology I + D + I of Spain (CGL2006-1648-CO3-02 and FENOMED) and also the Andalusian Government for RNM 0110 PAIDI.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy.
Acknowledgments
The authors wish to thank the Ministry of Science and Technology I+D+I CGL2006-1648-CO3-02 and FENOMED for financing this study and also the Andalusian Government for RNM 0110 PAIDI. Moreover, our thanks to Marta Gómez-Domenech for helping in the study carried out in Córdoba.
Conflicts of Interest
The authors declare that they have no conflict of interest. Funders had no role in the design of the study. They also did not participate in the collection, analysis, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Gomes, C.; Ribeiro, H.; Abreu, I. Aerobiology of Cupressaceae in Porto City, Portugal. Aerobiologia 2019, 35, 97–103. [Google Scholar] [CrossRef]
- Martínez-Bracero, M.; Alcázar, P.; Díaz de la Guardia, C.; González-Minero, F.J.; Ruiz, L.; Trigo Pérez, M.M.; Galán, C. Pollen Calendars: A guide to common airborne pollen in Andalusia. Aerobiologia 2015, 31, 549–557. [Google Scholar] [CrossRef]
- Velasco-Jiménez, M.J.; Alcázar, P.; Díaz de la Guardia, C.; Trigo, M.M.; De Linares, C.; Recio, M.; Galán, C. Pollen season trends in winter flowering trees in South Spain. Aerobiologia 2020, 36, 220–234. [Google Scholar] [CrossRef]
- Charpin, D.; Calleja, M.; Lahoz, C.; Pichot, C.; Waisel, Y. Allergy to cypress pollen. Allergy 2005, 60, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Guerra, F.; Daza, J.C.; Miguel, R.; Galan, C.; Dominguez, E.; Sanchez-Gijo, P. Sensitivity to Cupressus. Allergenic significance in Cordoba (Spain). J. Investig. Allergol. Clin. Immunol. 1996, 6, 117–120. [Google Scholar]
- Díaz de la Guardia, C.; Alba Sánchez, M.F.; De Linares Fernández, C.; Nieto-Lugilde, D.; López Caballero, J. Aerobiological and allergenic analysis of Cupressaceae pollen in Granada (Southern Spain). J. Investig. Allergol. Clin. Immunol. 2006, 16, 24–33. [Google Scholar]
- Asero, R.; Ceriotti, V.; Bonini, M. Cypress pollen allergy in Milan: The story of an ongoing growth. Eur. Ann. Allergy Clin. Immunol. 2020. Available online: http://www.eurannallergyimm.com/cont/online-first/825/original-articlebrcypress-pollen-allergy-milan-story-ongoing-3705allasp1.pdf (accessed on 30 December 2020).
- Caimmi, D.; Raschetti, P.; Pons, H.; Dhivert-Donnadieu, P.J.; Bousquet, J.; Demoly, P. Epidemiology of Cypress Pollen Allergy in Montpellier. J. Investig. Allergol. Clin. Immunol. 2012, 22, 6. [Google Scholar]
- Kay, A.B.; Bousquet, J.; Holt, P.G.; Kaplan, A.P. Allergy and Allergic Diseases; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Ariano, R.; Canonica, G.W.; Passalacqua, G. Possible role of climate changes in variations in pollen seasons and allergic sensitizations during 27 years. Ann. Allergy Asthma Immunol. 2010, 104, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Farjon, A. A monograph of Cupressaceae and Sciadopitys; Kew Royal Botanic Gardens: Richmond, UK, 2005. [Google Scholar]
- Galán, C.; Domínguez, E. La Botánica. In La Ciencia en la Córdoba Andalusí, 1st ed.; Real Academia de Córdoba, Litopress Córdoba: Córdoba, Spain, 2020; pp. 93–128. [Google Scholar]
- Flonard, M.; Lo, E.; Levetin, E. Increasing Juniperus virginiana L. pollen in the Tulsa atmosphere: Long-term trends, variability, and influence of meteorological conditions. Int. J. Biometeorol. 2018, 62, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Galán, C.; Fuillerat, M.J.; Comtois, P.; Domínguez-Vilches, E. Predictive study of Cupressaceae pollen season onset, severity, máximum value and maximum value date. Aerobiologia 1998, 14, 1995–1999. [Google Scholar] [CrossRef]
- Plaza, M.P.; Alcázar, P.; Galán, C. Correlation between airborne Olea europaea pollen concentrations and levels of the major allergen Ole e 1 in Córdoba, Spain, 2012–2014. Int. J. Biometeorol. 2016, 60, 1841–1847. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rajo, F.J.; Jato, V.; González-Parrado, Z.; Elvira-Rendueles, B.; Moreno-Grau, S.; Vega-Maray, A.; Fernández-González, D.; Asturias, J.A.; Suárez-Cervera, M. The combination of airborne pollen and allergen quantification to reliably assess the real pollinosis risk in different bioclimatic areas. Aerobiologia 2011, 27, 1–12. [Google Scholar] [CrossRef]
- Plaza, M.P.; Alcázar, P.; Velasco-Jiménez, M.J.; Galán, C. Aeroallergens: A comparative study of two monitoring methods. Aerobiologia 2017, 33, 363–373. [Google Scholar] [CrossRef]
- Alcázar, P.; Galán, C.; Torres, C.; Domínguez-Vilches, E. Detection of Airborne Allergen (Pla a 1) in Relation to Platanus Pollen in Córdoba, South Spain’. Ann. Agric. Environ. Med. 2015, 22, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Celenk, S. Detection of Reactive Allergens in Long-Distance Transported Pollen Grains: Evidence from Ambrosia. Atmos. Environ. 2019, 209, 212–219. [Google Scholar] [CrossRef]
- De Linares, C.; Alcázar, P.; Valle, A.M.; Díaz de la Guardia, C.; Galán, C. Parietaria major allergens vs. pollen in the air we breathe’. Environ. Res. 2019, 176, 108514. [Google Scholar] [CrossRef]
- Di Felice, G.; Caiaffa, M.F.; Bariletto, G.; Afferni, C.; Di Paola, R.; Mari, A.; Palumbo, S.; Tinghino, R.; Sallusto, F.; Tursi, A.; et al. Allergens of Arizona cypress (Cupressus arizonica) pollen: Characterization of the pollen extract and identification of the allergenic components. J. Allergy Clin. Immunol. 1994, 94, 547–555. [Google Scholar] [CrossRef]
- Di Felice, G.; Barletta, B.; Tinghino, R.; Pini, C. Cupressaceae pollinosis: Identification, purification and cloning of relevant allergens. Int. Arch. Allergy Immunol. 2001, 125, 280–289. [Google Scholar] [CrossRef]
- Suárez-Cervera, M.; Takahashi, Y.; Vega-Maray, A.; Seoane-Camba, J.A. Immunocytochemical localization of Cry j 1, the major allergen of Cryptomeria japonica (Taxodiaceae) in Cupressus arizonica and Cupressus sempervirens (Cupressaceae) pollen grains. Sex. Plant Reprod. 2003, 16, 9–15. [Google Scholar] [CrossRef]
- Charpin, D.; Pichot, C.; Belmonte, J.; Sutra, J.P.; Zidkova, J.; Chanez, P.; Poncet, P. Cypress pollinosis: From tree to clinic. Clin. Rev. Allergy Immunol. 2019, 56, 174–195. [Google Scholar] [CrossRef]
- Matricardi, P.M.; Kleine-Tebbe, H.J.; Hoffmann, R.; Valenta, C.; Hilger, S.; Hofmaier, R.C.; Aalberse, I.; Agache, R.; Asero, B.; Ballmer-Weber, D.; et al. EAACI Molecular Allergology User’s Guide. Pediatr. Allergy Immunol. 2016, 27, 1–250. [Google Scholar]
- D’Amato, G.; Liccardi, G.; D’amato, M.; Holgate, S. Environmental risk factors and allergic bronchial asthma. Clin. Exp. Allergy 2005, 35, 1113–1124. [Google Scholar] [CrossRef]
- Ribeiro, H.; Guimarães, F.; Duque, L.; Noronha, F.; Abreu, I. Characterization of particulate matter on airborne pollen grains. Environ. Pollut. 2015, 206, 7–16. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Health Effects of Particulate Matter. Policy Implications for Countries in Eastern Europe, Caucasus and Central Asia; WHO Regional Office for Europe: Copenhagen, Denmark, 2013; Available online: https://www.euro.who.int/__data/assets/pdf_file/0006/189051/Health-effects-of-particulate-matter-final-Eng.pdf (accessed on 21 February 2021).
- World Health Organization. The World Health Report 2000: Health Systems: Improving Performance; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Esworthy, R. Air Quality: EPA’s 2013 Changes to the Particulate Matter (PM) Standard; Library of Congress, Congressional Research Service: Washington, DC, USA, 2013. [Google Scholar]
- Sánchez-Morales, J.; Pardo-Igúzquiza, E.; Rodríguez-Tovar, F.J. Spatial Distribution of Climatic Cycles in Andalusia (Southern Spain). In International Conference on Time Series and Forecasting; Springer: Cham, Switzerland, 2018; pp. 243–255. [Google Scholar]
- Castroviejo, S. Flora Iberica 1–8, 10–15, 17–18, 21; Real Jardín Botánico, CSIC: Madrid, Spain, 1993. [Google Scholar]
- Hirst, J.M. An automatic volumetric spore-trap. Ann. Appl. Biol. 1952, 39, 257–265. [Google Scholar] [CrossRef]
- Galán, C.; Cariñanos, P.; Alcázar, P.; Domínguez, E. Manual de Calidad y Gestión de la Red Española de Aerobiología (Quality Manual and Management Spanish Aerobiology Network); Servicio de Publicaciones de la Universidad de Córdoba: Córdoba, Spain, 2007. [Google Scholar]
- Galán, C.; Smith, M.; Thibaudon, M.; Frenguelli, G.; Oteros, J.; Gehrig, R.; Berger, U.; Clot, B.; Brandao, R.; The EAS QC Working Group. Pollen monitoring: Minimum requirements and reproducibility of analysis. Aerobiologia 2014, 30, 385–395. [Google Scholar] [CrossRef]
- Galán, C.; Ariatti, A.; Bonini, M.M.; Clot, B.; Crouzy, B.; Dahl, A.; Fernández-González, D.; Frenguelli, G.; Gehrig, R.; Isard, S.; et al. Recommended terminology for aerobiological studies. Aerobiologia 2017, 33, 293–295. [Google Scholar] [CrossRef]
- Andersen, T.B. A model to predict the beginning of the pollen season. Grana 1991, 30, 269–275. [Google Scholar] [CrossRef]
- Andersen, A.A. New sampler for the collection, sizing, and enumeration of viable airborne particles. J. Bacteriol. 1958, 76, 471. [Google Scholar] [CrossRef]
- Galán, C.; Tormo, R.; Cuevas, J.; Infante, F.; Dominguez, E. Theoretical daily variation patterns of airborne pollen in the South-West of Spain. Grana 1991, 30, 201–209. [Google Scholar] [CrossRef]
- De Linares, C.; Nieto-Lugilde, D.; Alba, F.; Díaz de la Guardia, C.; Galán, C.; Trigo, M.M. Detection of airborne allergen (Ole e 1) in relation to Olea europaea pollen in S Spain. Clin. Exp. Allergy 2007, 37, 125–132. [Google Scholar] [CrossRef]
- Arilla, M.C.; Ibarrola, I.; Garcia, R.; De La Hoz, B.; Martinez, A.; Asturias, J.A. Quantification of the major allergen from cypress (Cupressus arizonica) pollen, Cup a 1, by monoclonal antibody-based ELISA. Int. Arch. Allergy Immunol. 2004, 134, 10–16. [Google Scholar] [CrossRef]
- Hidalgo, P.J.; Galán, C.; Domínguez, E. Male phenology of three species of Cupressus: Correlation with airborne pollen. Trees 2003, 17, 336–344. [Google Scholar] [CrossRef]
- Silva-Palacios, I.; Fernández-Rodríguez, S.; Durán-Barroso, P.; Tormo-Molina, R.; Maya-Manzano, J.M.; Gonzalo-Garijo, Á. Temporal modelling and forecasting of the airborne pollen of Cupressaceae on the southwestern Iberian Peninsula. Int. J. Biometeorol. 2016, 60, 297–306. [Google Scholar] [CrossRef]
- Sabariego, S.; Cuesta, P.; Fernández-González, F.; Pérez-Badia, R. Models for forecasting airborne Cupressaceae pollen levels in central Spain. Int. J. Biometeorol. 2012, 56, 253–258. [Google Scholar] [CrossRef]
- Rodríguez de la Cruz, D.; Sánchez-Reyes, E.; Sánchez-Sánchez, J. A contribution to the knowledge of Cupressaceae airborne pollen in the middle west of Spain. Aerobiologia 2015, 31, 435–444. [Google Scholar] [CrossRef]
- Aira, M.J.; Dopazo, A.; Jato, M.V. Aerobiological monitoring of Cupressaceae pollen in Santiago de Compostela (NW Iberian Peninsula) over six years. Aerobiologia 2001, 17, 319–325. [Google Scholar] [CrossRef]
- Nicoleta, I. Aerobiological monitoring of Taxaceae/Cupressaceae pollen in Timisoara. J. Hortic. For. Biotechnol. 2009, 13, 163–170. [Google Scholar]
- Pichot, C.; Calleja, M.; Penel, V.; Bues-Charbit, M.; Charpin, D. Inference of the pollen penetration and remanence into dwellings using seasonal variation of indoor/outdoor pollen counts. Aerobiologia 2015, 31, 315–322. [Google Scholar] [CrossRef]
- Uguz, U.; Guvensen, A.; Tort, N.S. Annual and intradiurnal variation of dominant airborne pollen and the effects of meteorological factors in Çeşme (Izmir, Turkey). Environ. Monit. Assess. 2017, 189, 530. [Google Scholar] [CrossRef]
- D’Amato, G.; Cecchi, L.; Bonini, S.; Nunes, C.; Annesi-Maesano, I.; Behrendt, H.; Van Cauwenberge, P. Allergenic pollen and pollen allergy in Europe. Allergy 2007, 62, 976–990. [Google Scholar] [CrossRef] [PubMed]
- Rakotozandry, T.; Cassagne, E.; Martin, S.; Alauzet, P.; Navarro, I.; Delcroux, C.; Caillaud, D.; Besancenot, J.; Thibaudon, M.; Charpin, D. Exposure to Cypress Pollens and Subsequent Symptoms: A Panel Study. Int. Arch. Allergy Immunol. 2019, 180, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Cecchi, L. From pollen count to pollen potency: The molecular era of aerobiology. Eur. Respir. J. 2013, 42, 898–900. [Google Scholar] [CrossRef]
- Buters, J.; Prank, M.; Sofiev, M.; Pusch, G.; Albertini, R.; Annesi-Maesano, I.; Antunes, C.; Behrendt, M.D.; Berger, U.; Brandao, R.; et al. Variation of the group 5 grass pollen allergen content of airborne pollen in relation to geographic location and time in season. J. Allergy Clin. Immunol. 2015, 136, 87–95. [Google Scholar] [CrossRef]
- Buters, J.T.; Thibaudon, M.; Smith, M.; Kennedy, R.; Rantio-Lehtimäki, A.; Albertini, R.; Reese, G.; Weber, B.; Galán, C.; Brandao, R.; et al. Release of Bet v 1 from birch pollen from 5 European countries. Results from the HIALINE study. Atmos. Environ. 2012, 55, 496–505. [Google Scholar] [CrossRef]
- Plaza, M.P.; Alcázar, P.; Hernández Ceballos, M.A.; Galán, C. Mismatch in Aeroallergens and airborne grass pollen concentrations. Atmos. Environ. 2016, 144, 361–369. [Google Scholar] [CrossRef]
- Jochner, S.; Lüpke, M.; Laube, J.; Weichenmeier, I.; Pusch, G.; Traidl-Hoffmann, C.; Menzel, A. Seasonal variation of birch and grass pollen loads and allergen release at two sites in the German Alps. Atmos. Environ. 2015, 122, 83–93. [Google Scholar] [CrossRef]
- Canini, A.; Giovinazzi, J.; Iacovacci, P.; Pini, C.; Caiola, M.G. Localisation of a carbohydrate epitope recognised by human IgE in pollen of Cupressaceae. J. Plant Res. 2004, 117, 147–153. [Google Scholar] [CrossRef]
- Emberlin, J.C. Plant allergens on pauci-micronic airborne particles. Clin. Exp. Allergy 1995, 25, 202–205. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).