Soil Carbon, Nitrogen, and Phosphorus Storages and Their Stoichiometry Due to Mixed Afforestation with Hippophae rhamnoides in the Loess Hilly Region, China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Material
2.3. Soil Sampling and Laboratory Analyses
2.4. Data Processing
3. Results
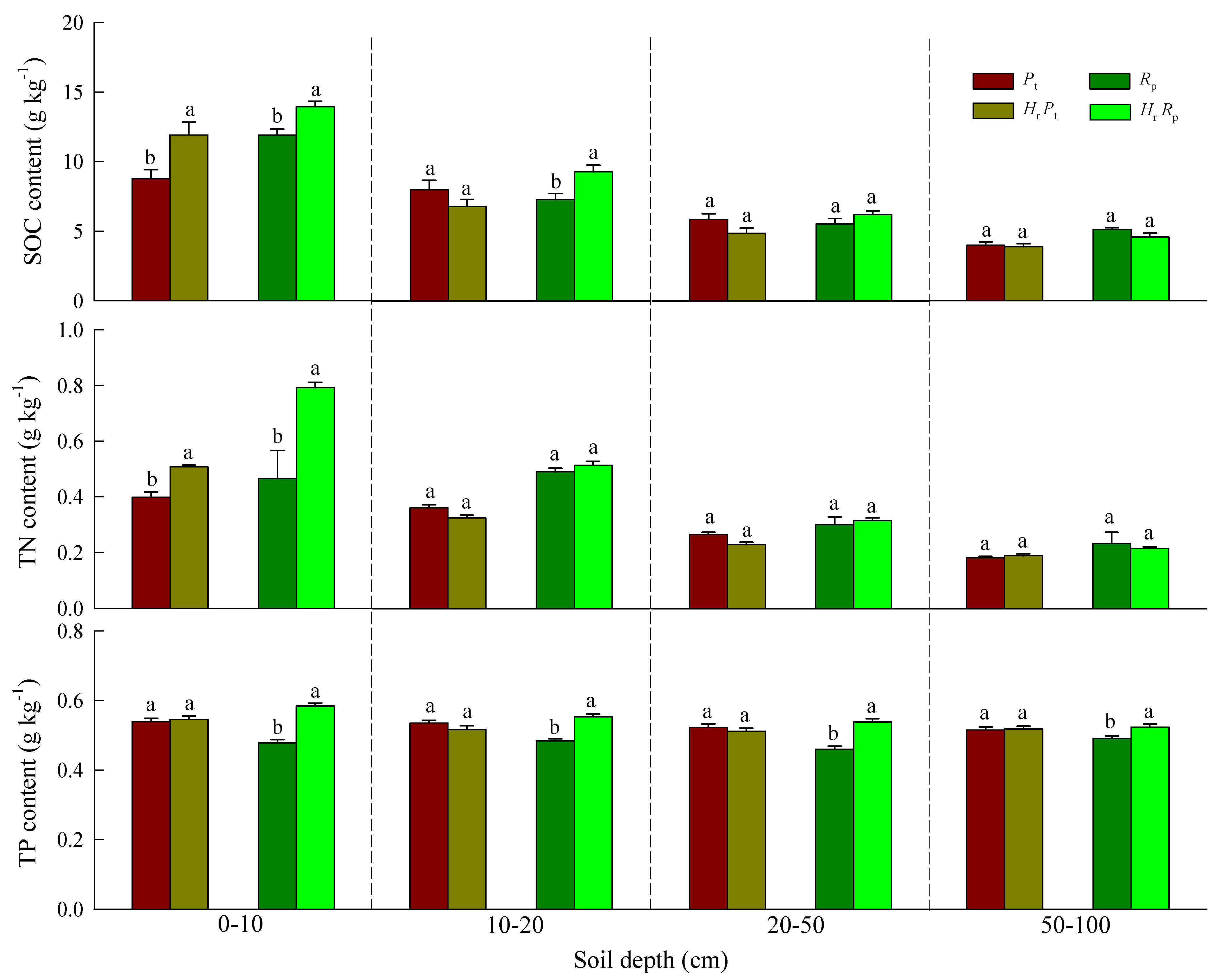
3.1. Soil Physical and Chemical Properties and SOC, TN, and TP Contents
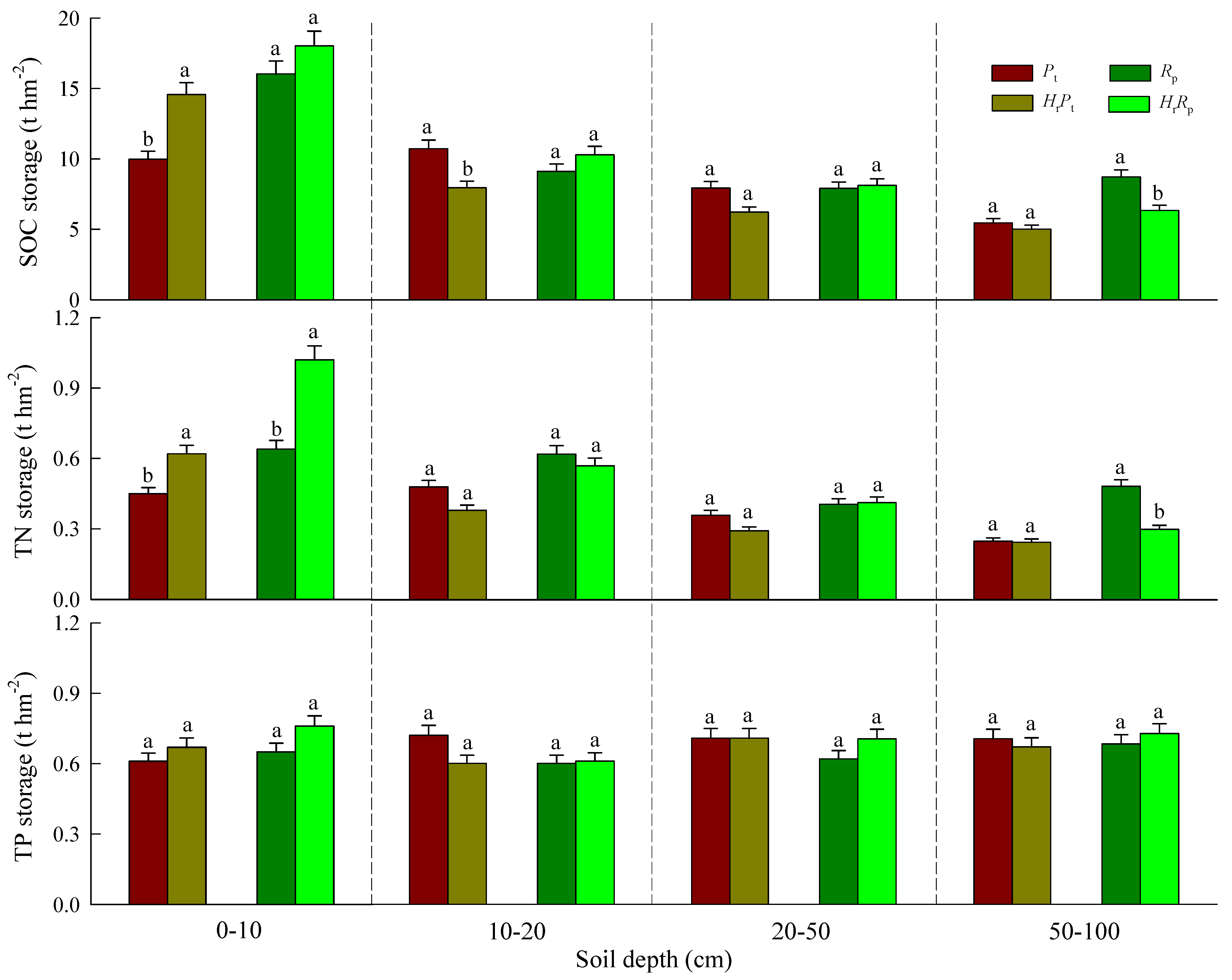
3.2. SOC, TN, and TP Storages
3.3. SOC, TN, and TP Stoichiometry
3.4. Correlations between the Soil Storages, Stoichiometry, and Physical and Chemical Properties
4. Discussions
4.1. Effects of Mixed Afforestation on the SOC, TN, and TP Storages
4.2. Effect of Mixed Afforestation on Stoichiometric Ratios
4.3. Influencing Factors of the Storage of SOC, TN, and TP, and the Stoichiometric Ratios
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reed, S.C.; Yang, X.; Thornton, P.E. Incorporating phosphorus cycling into global modeling efforts: A worthwhile, tractable endeavor. New Phytol. 2015, 208, 324–329. [Google Scholar] [CrossRef]
- Wang, Q.; Song, J.; Cao, L.; Li, X.; Yuan, H.; Li, N. Distribution and storage of soil organic carbon in a coastal wetland under the pressure of human activities. J. Soils Sediments 2017, 17, 11–22. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. The Role of Plants in the Effects of Global Change on Nutrient Availability and Stoichiometry in the Plant-Soil System. Plant Physiol. 2012, 160, 1741–1761. [Google Scholar] [CrossRef] [Green Version]
- Nejidat, A.; Potrafka, R.M.; Zaady, E. Successional biocrust stages on dead shrub soil mounds after severe drought: Effect of micro-geomorphology on microbial community structure and ecosystem recovery. Soil Biol. Biochem. 2016, 103, 213–220. [Google Scholar] [CrossRef]
- Lang, A.C.; von Oheimb, G.; Scherer-Lorenzen, M.; Yang, B.; Trogisch, S.; Bruelheide, H.; Ma, K.; Härdtle, W. Mixed afforestation of young subtropical trees promotes nitrogen acquisition and retention. J. Appl. Ecol. 2014, 51, 224–233. [Google Scholar] [CrossRef]
- Bing, H.; Wu, Y.; Zhou, J.; Sun, H.; Luo, J.; Wang, J.; Yu, D. Stoichiometric variation of carbon, nitrogen, and phosphorus in soils and its implication for nutrient limitation in alpine ecosystem of Eastern Tibetan Plateau. J. Soils Sediments 2016, 16, 405–416. [Google Scholar] [CrossRef]
- Smal, H.; Ligęza, S.; Pranagal, J.; Urban, D.; Pietruczyk-Popławska, D. Changes in the stocks of soil organic carbon, total nitrogen and phosphorus following afforestation of post-arable soils: A chronosequence study. For. Ecol. Manag. 2019, 451, 117536. [Google Scholar] [CrossRef]
- Deng, L.; Shangguan, Z. Afforestation Drives Soil Carbon and Nitrogen Changes in China. Land Degrad. Dev. 2016, 28, 151–165. [Google Scholar] [CrossRef] [Green Version]
- Hoogmoed, M. Soil Carbon and Nitrogen Cycling Following Afforestation with Mixed-Species Tree Plantings. Ph.D. Thesis, Monash University, Melbourne, Australia, 2014. [Google Scholar]
- Forrester, D.I.; Bauhus, J.; Cowie, A. On the success and failure of mixed-species tree plantations: Lessons learned from a model system of Eucalyptus globulus and Acacia mearnsii. For. Ecol. Manag. 2005, 209, 147–155. [Google Scholar] [CrossRef]
- Cui, Q.F.; Cheng, Q.; Yang, X.T. Soil ecological stoichiometry characteristics of the Platycladus orientalis forest in the South Taihang Mountain. J. West China For. Sci. 2020, 49, 20–30. (In Chinese) [Google Scholar]
- Huang, Y.L.; Tian, Q.; Qin, G.H. Effects of a mixed plantation of Robinia pseudoacacia and Fraxinus velutina on soil bacterial structure and diversity in the Yellow River Delta. Acta Ecol. Sin. 2018, 38, 3859–3867. (In Chinese) [Google Scholar]
- Ding, X.J.; Xie, G.L.; Jing, R.Y. Decomposition characteristics of litters in different mixed forest of Robinia pseudoacacia in Yellow River Delta. J. Soil Water Conserv. 2016, 30, 249–253. [Google Scholar]
- Wang, T.; Xue, X.; Zhou, L.; Guo, J. Combating Aeolian Desertification in Northern China. Land Degrad. Dev. 2015, 26, 118–132. [Google Scholar] [CrossRef]
- Galiana, A.; Gnahoua, G.M.; Chaumont, J. Improvement of nitrogen fixation in Acacia mangium through inoculation with rhizobium. Agrofor. Syst. 1998, 40, 297–307. [Google Scholar] [CrossRef]
- Hoogmoed, M.; Cunningham, S.C.; Baker, P.J. Is there more soil carbon under nitrogen-fixing trees than under non-nitrogen-fixing trees in mixed-species restoration plantings? Agric. Ecosyst. Environ. 2014, 188, 80–84. [Google Scholar] [CrossRef]
- Li, X.; Sun, K.; Li, F.Y. Variation in leaf nitrogen and phosphorus stoichiometry in the nitrogen-fixing Chinese sea-buckthorn (Hippophae rhamnoides L. subsp. sinensis Rousi) across northern China. Ecol. Res. 2014, 29, 723–731. [Google Scholar] [CrossRef]
- Kasel, S.; Singh, S.; Sanders, G.J.; Bennett, L.T. Species-specific effects of native trees on soil organic carbon in biodiverse plantings across north-central Victoria, Australia. Geoderma 2011, 161, 95–106. [Google Scholar] [CrossRef]
- Xu, H.; Qu, Q.; Li, P.; Guo, Z.; Wulan, E.; Xue, S. Stocks and Stoichiometry of Soil Organic Carbon, Total Nitrogen, and Total Phosphorus after Vegetation Restoration in the Loess Hilly Region, China. Forests 2019, 10, 27. [Google Scholar] [CrossRef] [Green Version]
- Xie, Z.; Zhu, J.; Liu, G.; Cadisch, G.; Hasegawa, T.; Chen, C.; Sun, H.; Tang, H.; Zeng, Q. Soil organic carbon stocks in China and changes from 1980s to 2000s. Glob. Chang. Biol. 2007, 13, 1989–2007. [Google Scholar] [CrossRef]
- Deng, L.; Liu, G.B.; Shangguan, Z.P. Land use conversion and changing soil carbon stocks in China’s ‘Grain-for-Green’ Program: A synthesis. Glob. Chang. Biol. 2014, 20, 3544–3556. [Google Scholar] [CrossRef]
- Pérez-Cruzado, C.; Mansilla-Salinero, P.; Rodríguez-Soalleiro, R.; Merino, A. Influence of tree species on carbon sequestration in afforested pastures in a humid temperate region. Plant Soil 2012, 353, 333–353. [Google Scholar] [CrossRef]
- Silver, W.L.; Kueppers, L.M.; Lugo, A.E.; Ostertag, R.; Matzek, V. Carbon sequestration and plant community dynamics following reforestation of tropical pasture. Ecol. Appl. 2004, 14, 1115–1127. [Google Scholar] [CrossRef]
- Fyles, J.; Fyles, I. Interaction of Douglas-fir with red alder and salal foliage litter during decomposition. Can. J. For. Res. 1993, 23, 358–361. [Google Scholar] [CrossRef]
- Zhu, Q.K.; Zhao, Y.M.; Wang, L.L.; Ma, H.; Liu, L.L. Influence of stand mixed pattern on growth of Hippophae rhamniodes L. subsp. sinensis in semi-arid region of Loess Plateau in North Shaanxi, China. J. Northeast For. Univ. 2013, 41, 1–4. (In Chinese) [Google Scholar]
- He, X.-H.; Critchley, C.; Bledsoe, C. Nitrogen Transfer Within and Between Plants Through Common Mycorrhizal Networks (CMNs). Crit. Rev. Plant Sci. 2003, 22, 531–567. [Google Scholar] [CrossRef]
- Forrester, D.I.; Bauhus, J.; Cowie, A.; Vanclay, J. Mixed-species plantations of Eucalyptus with nitrogen-fixing trees: A review. For. Ecol. Manag. 2006, 233, 211–230. [Google Scholar] [CrossRef] [Green Version]
- Fang, X.-M.; Zhang, X.-L.; Zong, Y.-Y.; Zhang, Y.; Wan, S.-Z.; Bu, W.-S.; Chen, F.-S. Soil phosphorus functional fractions and tree tissue nutrient concentrations influenced by stand density in subtropical Chinese fir plantation forests. PLoS ONE 2017, 12, e0186905. [Google Scholar] [CrossRef]
- Feng, D.; Bao, W.; Pang, X. Consistent profile pattern and spatial variation of soil C/N/P stoichiometric ratios in the subalpine forests. J. Soils Sediments 2017, 17, 2054–2065. [Google Scholar] [CrossRef]
- Tessier, J.T.; Raynal, D.J. Vernal nitrogen and phosphorus retention by forest understory vegetation and soil microbes. Plant Soil 2003, 256, 443–453. [Google Scholar] [CrossRef]
- Dong, C.; Qiao, Y.; Cao, Y.; Chen, Y.; Wu, X.; Xue, W. Seasonal Variations in Carbon, Nitrogen and Phosphorus Stoichiometry of a Robinia pseudoacacia Plantation on the Loess Hilly Region, China. Forests 2021, 12, 214. [Google Scholar] [CrossRef]
- Yue, K.; Fornara, D.; Yang, W.; Peng, Y.; Li, Z.; Wu, F.; Peng, C. Effects of three global change drivers on terrestrial C:N:P stoichiometry: A global synthesis. Glob. Chang. Biol. 2017, 23, 2450–2463. [Google Scholar] [CrossRef]
- Chen, F.-S.; Feng, X.; Liang, C. Endogenous versus exogenous nutrient affects C, N, and P dynamics in decomposing litters in mid-subtropical forests of China. Ecol. Res. 2012, 27, 923–932. [Google Scholar] [CrossRef]
- Tian, H.; Chen, G.; Zhang, C.; Melillo, J.M.; Hall, C.A.S. Pattern and variation of C:N:P ratios in China’s soils: A synthesis of observational data. Biogeochemistry 2010, 98, 139–151. [Google Scholar] [CrossRef]
- Li, M.; Zhou, X.; Zhang, Q.; Cheng, X. Consequences of afforestation for soil nitrogen dynamics in central China. Agric. Ecosyst. Environ. 2014, 183, 40–46. [Google Scholar] [CrossRef]
- De Feudis, M.; Cardelli, V.; Massaccesi, L.; Lagomarsino, A.; Fornasier, F.; Westphalen, D.J.; Cocco, S.; Corti, G.; Agnelli, A. Influence of Altitude on Biochemical Properties of European Beech (Fagus sylvatica L.) Forest Soils. Forests 2017, 8, 213. [Google Scholar] [CrossRef] [Green Version]
- Güsewell, S.; Jewell, P.L.; Edwards, P.J. Effects of heterogeneous habitat use by cattle on nutrient availability and litter decomposition in soils of an Alpine pasture. Plant Soil 2005, 268, 135–149. [Google Scholar] [CrossRef]
- Liao, C.; Luo, Y.; Fang, C.; Chen, J.; Li, B. The effects of plantation practice on soil properties based on the comparison between natural and planted forests: A meta-analysis. Glob. Ecol. Biogeogr. 2011, 21, 318–327. [Google Scholar] [CrossRef]
- Pizzeghello, D.; Francioso, O.; Concheri, G.; Muscolo, A.; Nardi, S. Land Use Affects the Soil C Sequestration in Alpine Environment, NE Italy. Forests 2017, 8, 197. [Google Scholar] [CrossRef] [Green Version]
- Rachid, C.; Balieiro, F.; Peixoto, R.; Pinheiro, Y.; Piccolo, M.D.C.; Chaer, G.; Rosado, A. Mixed plantations can promote microbial integration and soil nitrate increases with changes in the N cycling genes. Soil Biol. Biochem. 2013, 66, 146–153. [Google Scholar] [CrossRef]
- Chen, C.R.; Condron, L.M.; Davis, M.R.; Sherlock, R.R. Effects of afforestation on phosphorus dynamics and biological properties in a New Zealand grassland soil. Plant Soil 2000, 220, 151–163. [Google Scholar] [CrossRef]
- Hoogmoed, M.; Cunningham, S.C.; Thomson, J.R.; Baker, P.J.; Beringer, J.; Cavagnaro, T.R. Does afforestation of pastures increase sequestration of soil carbon in Mediterranean climates? Agric. Ecosyst. Environ. 2012, 159, 176–183. [Google Scholar] [CrossRef]
- Song, B.-L.; Yan, M.-J.; Hou, H.; Guan, J.-H.; Shi, W.-Y.; Li, G.-Q.; Du, S. Distribution of soil carbon and nitrogen in two typical forests in the semiarid region of the Loess Plateau, China. Catena 2016, 143, 159–166. [Google Scholar] [CrossRef]


| Sample Plot | Tree Species | Altitude (m) | Stand Age (a) | Tree Height (m) | DBH/DGH (cm) | Crown Diameter of (EW × SN) (m × m) |
|---|---|---|---|---|---|---|
| Pt | P. tabuliformis | 1189 | 17 | 3.69 ± 0.09 | 7.43 ± 0.27 | (2.96 ± 0.12) × (3.36 ± 0.11) |
| Rp | R. pseudoacacia | 1287 | 17 | 7.59 ± 0.65 | 9.40 ± 1.50 | (4.36 ± 0.39) × (4.24 ± 0.32) |
| HrPt | P. tabuliformis | 1270 | 17 | 4.01 ± 0.17 | 7.11 ± 0.48 | (2.01 ± 0.09) × (2.04 ± 0.11) |
| H. rhamnoides | 3.08 ± 0.14 | 5.43 ± 0.33 | (2.62 ± 0.12) × (2.60 ± 0.14) | |||
| HrRp | R. pseudoacacia | 1302 | 17 | 5.92 ± 0.52 | 7.80 ± 1.02 | (1.79 ± 0.10) × (1.80 ± 0.09) |
| H. rhamnoides | 2.69 ± 0.10 | 4.63 ± 0.32 | (3.36 ± 0.35) × (3.25 ± 0.34) |
| Soil Depth (cm) | Sample Plot | SWC (m3 m−3) | Field Capacity (%) | BD (g cm−3) | Soil Total Porosity (%) | Nitrate N (g kg−1) | Ammonium N (g kg−1) | Available P (g kg−1) |
|---|---|---|---|---|---|---|---|---|
| 0–10 | Pt | 0.14 ± 0.002 | 17.94 ± 0.24 | 1.23 ± 0.01 | 51.49 ± 0.59 | 0.21 ± 0.01 b | 1.86 ± 0.11 | 0.31 ± 0.02 b |
| HrPt | 0.13 ± 0.004 | 17.14 ± 0.20 | 1.22 ± 0.03 | 51.86 ± 0.60 | 0.39 ± 0.02 a | 1.49 ± 0.09 | 0.41 ± 0.02 a | |
| Rp | 0.11 ± 0.001 | 13.72 ± 0.16 b | 1.35 ± 0.02 | 46.94 ± 0.54 b | 0.86 ± 0.05 | 1.13 ± 0.06 b | 0.31 ± 0.02 b | |
| HrRp | 0.11 ± 0.003 | 15.47 ± 0.18 a | 1.29 ± 0.01 | 50.49 ± 0.58 a | 0.85 ± 0.05 | 1.93 ± 0.11 a | 0.50 ± 0.02 a | |
| 10–20 | Pt | 0.12 ± 0.002 | 15.51 ± 0.31 b | 1.34 ± 0.02 a | 47.11 ± 0.82 b | 0.26 ± 0.04 | 1.86 ± 0.32 | 0.36 ± 0.06 |
| HrPt | 0.13 ± 0.005 | 18.21 ± 0.32 a | 1.17 ± 0.03 b | 51.77 ± 0.90 a | 0.33 ± 0.06 | 1.41 ± 0.24 | 0.37 ± 0.06 | |
| Rp | 0.09 ± 0.002 | 15.72 ± 0.27 b | 1.25 ± 0.02 a | 50.99 ± 0.88 b | 0.56 ± 0.10 | 1.14 ± 0.20 | 0.19 ± 0.03 b | |
| HrRp | 0.12 ± 0.005 | 19.01 ± 0.33 a | 1.11 ± 0.03 b | 55.41 ± 0.96 a | 0.46 ± 0.08 | 1.42 ± 0.25 | 0.37 ± 0.06 a | |
| 20–50 | Pt | 0.13 ± 0.002 | 15.45 ± 0.22 | 1.35 ± 0.02 | 47.66 ± 0.69 | 0.15 ± 0.02 | 1.73 ± 0.20 | 0.30 ± 0.04 |
| HrPt | 0.11 ± 0.004 | 16.56 ± 0.24 | 1.28 ± 0.03 | 50.27 ± 0.73 | 0.15 ± 0.02 | 1.27 ± 0.15 | 0.37 ± 0.04 | |
| Rp | 0.09 ± 0.001 | 14.39 ± 0.21 | 1.35 ± 0.02 | 47.38 ± 0.68 | 0.45 ± 0.05 | 1.34 ± 0.16 | 0.18 ± 0.02 b | |
| HrRp | 0.11 ± 0.006 | 15.93 ± 0.23 | 1.31 ± 0.03 | 47.83 ± 0.73 | 0.37 ± 0.04 | 1.32 ± 0.15 | 0.33 ± 0.04 a | |
| 50–100 | Pt | 0.11 ± 0.003 | 15.51 ± 0.31 | 1.37 ± 0.03 | 48.38 ± 0.98 | 0.12 ± 0.03 | 1.72 ± 0.40 | 0.35 ± 0.08 |
| HrPt | 0.08 ± 0.005 | 16.08 ± 0.37 | 1.29 ± 0.04 | 50.91 ± 1.07 | 0.17 ± 0.04 | 1.36 ± 0.32 | 0.36 ± 0.08 | |
| Rp | 0.10 ± 0.002 | 16.06 ± 0.32 | 1.39 ± 0.03 | 45.39 ± 0.92 | 0.33 ± 0.07 | 1.16 ± 0.23 | 0.21 ± 0.05 | |
| HrRp | 0.08 ± 0.009 | 16.29 ± 0.29 | 1.38 ± 0.04 | 46.79 ± 1.01 | 0.27 ± 0.06 | 1.28 ± 0.33 | 0.29 ± 0.07 |
| Soil Depth (cm) | SOC (g kg−1) | TN (g kg−1) | TP (g kg−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| ME | TS | ME × TS | ME | TS | ME × TS | ME | TS | ME × TS | |
| 0–10 | 5.13 * | 19.52 *** | 3.22 | 57.60 *** | 40.19 *** | 13.84 *** | 35.74 *** | 1.41 | 27.67 *** |
| 10–20 | 0.56 | 2.82 | 8.75 ** | 0.29 | 1.98 | 3.50 | 9.12 ** | 0.75 | 27.68 *** |
| 20–50 | 0.67 | 3.42 | 2.64 | 1.40 | 3.04 | 3.73 | 13.38 *** | 3.89 | 23.34 *** |
| 50–100 | 0.99 | 2.75 | 3.80 | 2.17 | 1.99 | 3.12 | 5.10 * | 1.45 | 3.61 |
| Soil Depth (cm) | SOC Storage (t hm−2) | TN Storage (t hm−2) | TP Storage (t hm−2) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| ME | TS | ME × TS | ME | TS | ME × TS | ME | TS | ME × TS | |
| 0–10 | 14.62 ** | 30.41 ** | 2.28 | 45.26 *** | 50.27 *** | 7.06 * | 4.54 | 2.37 | 0.51 |
| 10–20 | 2.06 | 0.44 | 12.70 ** | 3.35 | 2.04 | 0.88 | 2.04 | 2.04 | 2.83 |
| 20–50 | 2.92 | 4.59 | 4.82 | 1.91 | 1.20 | 2.92 | 0.18 | 0.26 | 3.16 |
| 50–100 | 13.95 ** | 37.10 *** | 6.53 * | 23.73 ** | 56.27 *** | 21.75 ** | 0.02 | 0.18 | 0.96 |
| Sample Plot | SOC Storage (t hm−2) | TN Storage (t hm−2) | TP Storage (t hm−2) |
|---|---|---|---|
| Pt | 86.46 ± 5.00 | 3.92 ± 0.23 | 6.87 ± 0.40 |
| HrPt | 85.19 ± 4.92 | 3.88 ± 0.22 | 6.50 ± 0.38 |
| Rp | 104.47 ± 6.03 b | 5.39 ± 0.31 b | 6.83 ± 0.37 |
| HrRp | 108.34 ± 6.26 a | 5.86 ± 0.34 a | 7.01 ± 0.41 |
| ME | 0.05 | 0.59 | 0.12 |
| TS | 13.60 ** | 37.84 *** | 0.00 |
| ME×TS | 6.21 * | 0.83 | 1.69 |
| Soil Depth (cm) | C:N | C:P | N:P | ||||||
|---|---|---|---|---|---|---|---|---|---|
| ME | TS | ME × TS | ME | TS | ME × TS | ME | TS | ME × TS | |
| 0–10 | 8.76 ** | 0.27 | 16.59 *** | 3.27 | 18.64 *** | 6.68 * | 28.43 *** | 35.95 *** | 3.45 |
| 10–20 | 0.20 | 1.86 | 2.11 | 0.00 | 1.88 | 2.16 | 2.96 | 1.03 | 0.74 |
| 20–50 | 0.09 | 1.53 | 0.01 | 2.36 | 2.84 | 0.14 | 1.91 | 2.06 | 0.01 |
| 50–100 | 0.07 | 0.01 | 0.31 | 2.30 | 1.11 | 3.89 | 3.35 | 3.62 | 3.85 |
| Forest Type | Soil Property | BD | SWC | Total Porosity | Field Capacity | SOC | TN | TP | Nitrate N | Ammonium N | Available P |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pure forest | Cs | −0.937 ** | 0.999 ** | 0.939 ** | 0.816 * | ||||||
| Ns | −0.831 * | −0.804 * | 0.981 ** | 0.993 ** | 0.928 ** | −0.757 * | −0.746 * | ||||
| Ps | 0.887 ** | 0.846 * | 0.867 * | ||||||||
| C:N | −0.791 * | 0.979 ** | |||||||||
| C:P | −0.850 * | 0.963 ** | 0.999 ** | 0.955 ** | −0.807 * | −0.796 * | |||||
| N:P | −0.915 ** | 0.917 ** | 0.995 ** | 0.987 ** | −0.882 * | −0.873 * | |||||
| Mixed forest | Cs | −0.820 * | 0.970 ** | 0.919 ** | 0.946 ** | 0.904 ** | 0.979 ** | ||||
| Ns | 0.999 ** | 0.981 ** | 0.854 * | 0.974 ** | 0.913 ** | ||||||
| Ps | −0.995 ** | 0.979 ** | 0.945 ** | 0.873 * | |||||||
| C:N | −0.871 * | 0.822 * | 0.997 ** | −0.804 * | −0.825 * | ||||||
| C:P | −0.761 * | 0.990 ** | 0.954 ** | −0.908 ** | 0.943 ** | 0.954 ** | |||||
| N:P | −0.731 * | 0.992 ** | 0.999 ** | −0.767 * | 0.997 ** | 0.842 * |
| Forest Type | Soil Property | Equation | R2 |
|---|---|---|---|
| Pure forest | Cs | Cs = 22.79SOC − 85.46 | 0.996 |
| Ns | Ns = 11.53SWC + 22.61TN − 5.33 | 0.998 | |
| Ps | Ps = 0.43X1 + 4.87X4 − 15.69 | 0.987 | |
| C:N | C:N = 0.80X1 − 17.31 | 0.948 | |
| C:P | C:P = 0.12X2 − 39.42TP − 0.88 | 0.998 | |
| N:P | N:P = 2.91TN − 0.01X1 + 0.27 | 0.986 | |
| Mixed forest | Cs | Cs = 142.89BD − 165.37SWC + 9.07SOC − 145.37 | 0.989 |
| Ns | Ns = 4.20BD + 6.01TN + 4.08TP + 2.50X3 − 6.23 | 0.996 | |
| Ps | Ps = 2.48BD + 24.38TP + 0.30X2 − 10.51X3 − 10.72 | 0.999 | |
| C:N | C:N = 14.26BD + 0.90X1 − 21.29TN − 0.87X2 − 19.24 | 0.993 | |
| C:P | C:P = 0.69SOC − 30.25TP + 12.45X3 + 1.25 | 0.999 | |
| N:P | N:P = 1.08TN + 0.36X3 + 0.21 | 0.995 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Niu, Y.; Xun, M.; Jin, J.; Tang, Y.; Chen, Y. Soil Carbon, Nitrogen, and Phosphorus Storages and Their Stoichiometry Due to Mixed Afforestation with Hippophae rhamnoides in the Loess Hilly Region, China. Forests 2021, 12, 1718. https://doi.org/10.3390/f12121718
Wu X, Niu Y, Xun M, Jin J, Tang Y, Chen Y. Soil Carbon, Nitrogen, and Phosphorus Storages and Their Stoichiometry Due to Mixed Afforestation with Hippophae rhamnoides in the Loess Hilly Region, China. Forests. 2021; 12(12):1718. https://doi.org/10.3390/f12121718
Chicago/Turabian StyleWu, Xu, Yaobin Niu, Mengyao Xun, Junyi Jin, Yakun Tang, and Yunming Chen. 2021. "Soil Carbon, Nitrogen, and Phosphorus Storages and Their Stoichiometry Due to Mixed Afforestation with Hippophae rhamnoides in the Loess Hilly Region, China" Forests 12, no. 12: 1718. https://doi.org/10.3390/f12121718
APA StyleWu, X., Niu, Y., Xun, M., Jin, J., Tang, Y., & Chen, Y. (2021). Soil Carbon, Nitrogen, and Phosphorus Storages and Their Stoichiometry Due to Mixed Afforestation with Hippophae rhamnoides in the Loess Hilly Region, China. Forests, 12(12), 1718. https://doi.org/10.3390/f12121718





