Drought Drives Growth and Mortality Rates in Three Pine Species under Mediterranean Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Field Sampling
2.3. Dendrochronological Analyses
2.4. Drought: Spatial and Temporal Patterns
2.5. Drought-Growth Relationships
2.6. Statistical Analyses
3. Results
3.1. Drought Severity and Mortality Patterns
3.2. Drought-Growth Relationships
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Babst, F.; Bouriaud, O.; Poulter, B.; Trouet, V.; Girardin, M.P.; Frank, D.C. Twentieth century redistribution in climatic drivers of global tree growth. Sci. Adv. 2019, 5, eaat4313. [Google Scholar] [CrossRef] [Green Version]
- Allen, C.D.; Breshear, D.; McDowell, N.G. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Williams, A.P.; Allen, C.D.; Macalady, A.K.; Griffin, D.; Woodhouse, C.A.; Meko, D.M.; Swetnam, T.W.; Rauscher, S.A.; Seager, R.; Grissino-Mayer, H.D.; et al. Temperature as a potent driver of regional forest drought stress and tree mortality. Nat. Clim. Chang. 2013, 3, 292–297. [Google Scholar] [CrossRef]
- Grossiord, C.; Buckley, T.N.; Cernusak, L.A.; Novick, K.A.; Poulter, B.; Siegwolf, R.T.W.; Sperry, J.S.; McDowell, N.G. Plant responses to rising vapor pressure deficit. New Phytol. 2020, 226, 1550–1566. [Google Scholar] [CrossRef] [Green Version]
- Zweifel, R.; Sterck, F.; Braun, S.; Buchmann, N.; Eugster, E.; Gessler, A.; Häni, M.; Peters, R.L.; Walthert, L.; Wilhelm, M.; et al. Why trees grow at night. New Phytol. 2021, 231, 2174–2185. [Google Scholar] [CrossRef]
- Arend, M.; Link, R.M.; Patthey, R.; Hoch, G.; Schuldt, B.; Kahmen, A. Rapid hydraulic collapse as cause of drought-induced mortality in conifers. Proc. Nat. Acad. Sci. USA 2021, 118, e2025251118. [Google Scholar] [CrossRef] [PubMed]
- Hember, R.A.; Kurz, W.A.; Coops, N.C. Relationships between individual-tree mortality and water-balance variables indicate positive trends in water stress-induced tree mortality across North America. Glob. Chang. Biol. 2017, 23, 1691–1710. [Google Scholar] [CrossRef]
- Senf, C.; Buras, A.; Zang, C.S.; Rammig, A.; Seidl, R. Excess forest mortality is consistently linked to drought across Europe. Nat. Comm. 2020, 11, 6200. [Google Scholar] [CrossRef] [PubMed]
- Batllori, E.; Lloret, F.; Aakala, T.; Anderegg, W.; Aynekulu, E.; Bendixsen, D.; Bentouati, A.; Bigler, C.; Burk, C.; Camarero, J.; et al. Forest and Woodland Replacement Patterns Following Drought-Related Mortality. Proc. Nat. Acad. Sci. USA 2020, 117, 29720–29729. [Google Scholar] [CrossRef]
- García-Valdés, R.; Vayreda, J.; Retana, J.; Martínez-Vilalta, J. Low forest productivity associated with increasing drought-tolerant species is compensated by an increase in drought-tolerance richness. Glob. Chang. Biol. 2021, 27, 2113–2127. [Google Scholar] [CrossRef]
- Ruiz-Benito, P.; Lines, E.R.; Gómez-Aparicio, L.; Zavala, M.A.; Coomes, D.A. Patterns and drivers of tree mortality in Iberian forests: Climatic effects are modified by competition. PLoS ONE 2013, 8, e56843. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Aparicio, L.; García-Valdés, R.; Ruiz-Benito, P.; Zavala, M.A. Disentangling the relative importance of climate, size and competition on tree growth in Iberian forests: Implications for management under global change. Glob. Chang. Biol. 2011, 17, 2400–2414. [Google Scholar] [CrossRef] [Green Version]
- Vicente-Serrano, S.M.; Lopez-Moreno, J.I.; Beguería, S.; Lorenzo-Lacruz, J.; Sanchez-Lorenzo, A.; García-Ruiz, J.M.; Azorin-Molina, C.; Morán-Tejeda, E.; Revuelto, J.; Trigo, R.; et al. Evidence of increasing drought severity caused by temperature rise in southern Europe. Environ. Res. Lett. 2014, 9, 044001. [Google Scholar] [CrossRef]
- Kjellström, E.; Nikulin, G.; Strandberg, G.; Christensen, O.; Jacob, D.; Keuler, K.; Lenderink, G.; van Meijgaard, E.; Schär, C.; Somot, S.; et al. European climate change at global mean temperature increases of 1.5 and 2 °C above pre-industrial conditions as simulated by the EURO-CORDEX regional climate models. Earth Syst. Dynam. 2018, 9, 459–478. [Google Scholar] [CrossRef] [Green Version]
- Spinoni, J.; Vogt, J.V.; Naumann, G.; Barbosa, P.; Dosio, A. Will drought events become more frequent and severe in Europe? Int. J. Climatol. 2018, 38, 1718–1736. [Google Scholar] [CrossRef] [Green Version]
- Camarero, J.J.; Gazol, A.; Sangüesa-Barreda, G.; Oliva, J.; Vicente-Serrano, S.M. To die or not to die: Early warnings of tree dieback in response to a severe drought. J. Ecol. 2015, 103, 44–57. [Google Scholar] [CrossRef] [Green Version]
- Gazol, A.; Camarero, J.J.; Sánchez-Salguero, R.; Vicente-Serrano, S.M.; Serra-Maluquer, X.; Gutiérrez, E.; de Luis, M.; Sangüesa-Barreda, G.; Novak, K.; Rozas, V. Drought legacies are short, prevail in dry conifer forests and depend on growth variability. J. Ecol. 2020, 108, 2473–2484. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Viguera, B.; Cabrera, M.; Cañellas, I. Drought induced decline could portend widespread pine mortality at the xeric ecotone in managed mediterranean pine-oak woodlands. For. Ecol. Manag. 2014, 320, 70–82. [Google Scholar] [CrossRef]
- Camarero, J.; Gazol, A.; Sangüesa-Barreda, G.; Cantero, A.; Sánchez-Salguero, R.; Sánchez-Miranda, A.; Granda, E.; Serra-Maluquer, X.; Ibáñez, R. Forest growth responses to drought at short- and long-term scales in Spain: Squeezing the stress memory from tree rings. Front. Ecol. Evol. 2018, 6, 9. [Google Scholar] [CrossRef] [Green Version]
- González-de-Andrés, C.; Camarero, J.J. Disentangling mechanisms of drought-induced dieback in Pinus nigra Arn. from growth and wood isotope patterns. Forests 2020, 11, 1339. [Google Scholar] [CrossRef]
- Savi, T.; Casolo, V.; Dal Borgo, A.; Rosner, S.; Torboli, V.; Stenni, B.; Bertoncin, P.; Martellos, S.; Pallavicini, A.; Nardini, A. Drought-induced dieback of Pinus nigra: A tale of hydraulic failure and carbon starvation. Cons. Phys. 2020, 7, coz012. [Google Scholar]
- Heres, A.; Camarero, J.J.; López, B.C.; Martínez-Vilalta, J. Declining hydraulic performances and low carbon investments in tree rings predate Scots pine drought-induced mortality. Trees 2014, 28, 1737–1750. [Google Scholar] [CrossRef]
- Herguido, E.; Granda, E.; Benavides, R.; García-Cervigón, A.I.; Camarero, J.J.; Valladares, F. Contrasting growth and mortality responses to climate warming of two pine species in a continental Mediterranean ecosystem. For. Ecol. Manag. 2016, 363, 149–158. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Férriz, M.; García-Garrido, S.; Aguín, O.; Elvira-Recuenco, M.; Hernandez-Escribano, L.; Martin-Benito, D.; Raposo, R. Synergistic abiotic and biotic stressors explain widespread decline of Pinus pinaster in a mixed forest. Sci. Total Environ. 2019, 685, 963–975. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Fernández, D.; Viana-Soto, A.; Camarero, J.J.; Zavala, M.A.; Tijerín, J.; García, M. Using spectral indices as early warning signals of forest dieback: The case of drought-prone Pinus pinaster forests. Sci. Total Environ. 2021, 793, 148578. [Google Scholar] [CrossRef] [PubMed]
- Bigler, C.; Bräker, O.; Bugmann, H.; Dobbertin, M.; Rigling, A. Drought as an inciting mortality factor in Scots pine stands of the Valais, Switzerland. Ecosystems 2006, 9, 330–343. [Google Scholar] [CrossRef] [Green Version]
- Preisler, Y.; Tatarinov, F.; Grünzweig, J.M.; Bert, D.; Ogée, J.; Wingate, L.; Rotenberg, E.; Rohatyn, S.; Her, N.; Moshe, I.; et al. Mortality versus survival in drought-affected Aleppo pine forest depends on the extent of rock cover and soil stoniness. Funct. Ecol. 2019, 33, 901–912. [Google Scholar] [CrossRef]
- Serra-Maluquer, X.; Granda, E.; Camarero, J.J.; Vilà-Cabrera, A.; Jump, A.; Sánchez-Salguero, R.; Sangüesa-Barreda, G.; Imbert, B.; Gazol, A. Impacts of recurrent dry and wet years alter long-term tree growth trajectories. J. Ecol. 2021, 3, 1561–1574. [Google Scholar] [CrossRef]
- Andregg, L.D.L.; Andregg, W.R.L.; Berry, J.A. Tree physiology review: Not all droughts are created equal: Translating meteorological drought into woody plant mortality. Tree Physiol. 2013, 33, 701–712. [Google Scholar] [CrossRef] [Green Version]
- Trugman, A.T.; Anderegg, L.D.; Anderegg, W.R.; Das, A.J.; Stephenson, N.L. Why is tree drought mortality so hard to predict? Trends Ecol. Evol. 2021, 36, 520–532. [Google Scholar] [CrossRef]
- Dobbertin, M. Tree growth as indicator of tree vitality and of tree reaction to environmental stress: A review. Eur. J. For. 2005, 124, 319–333. [Google Scholar] [CrossRef]
- Cailleret, M.; Jansen, S.; Robert, E.M.R.; Desoto, L.; Aakala, T.; Antos, J.A.; Beikircher, B.; Bigler, C.; Bugmann, H.; Caccianiga, M.; et al. A synthesis of radial growth patterns preceding tree mortality. Glob. Chang. Biol. 2017, 23, 1675–1690. [Google Scholar] [CrossRef] [PubMed]
- DeSoto, L.; Cailleret, M.; Sterck, F.; Jansen, S.; Kramer, K.; Robert, E.M.R.; Aakala, T.; Amoroso, M.M.; Bigler, C.; Camarero, J.J.; et al. Low growth resilience to drought is related to future mortality risk in trees. Nat. Commun. 2020, 11, 545. [Google Scholar] [CrossRef]
- Colangelo, M.; Camarero, J.J.; Borghetti, M.; Gazol, A.; Ripullone, F. Size matters a lot: Drought-affected Italian oaks are smaller and show lower growth prior to tree death. Front. Plant Sci. 2017, 8, 135. [Google Scholar] [CrossRef] [Green Version]
- Navarro-Cerrillo, R.M.; Sánchez-Salguero, R.; Manzanedo, R.D.; Camarero, J.J.; Fernández Cancio, A. Site and age condition the growth responses to climate and drought of relict Pinus nigra subsp. salzmannii populations in Southern Spain. Tree-Ring Res. 2014, 70, 145–155. [Google Scholar] [CrossRef] [Green Version]
- Ne’eman, G.; Osem, Y. Pines and Their Mixed Forest Ecosystems in the Mediterranean Basin; Springer: Cham, Switzerland, 2021; pp. 237–325. [Google Scholar]
- Fritts, H.C. Tree-Rings and Climate; Academic Press: London, UK, 1976. [Google Scholar]
- Yamaguchi, D.K. A simple method for cross-dating increment cores from living trees. Can. J. For. Res. 1991, 21, 414–416. [Google Scholar] [CrossRef]
- Mast, J.N.; Veblen, T.T. A dendrochronological method of studying tree mortality patterns. Phys. Geogr. 1994, 15, 529–542. [Google Scholar] [CrossRef]
- Larsson, L.A.; Larsson, P.O. CDendro and CooRecorder (v. 9.3.1); Cybis Elektronik and Data AB: Saltsjöbaden, Sweden, 2018. [Google Scholar]
- Holmes, R.L. Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull. 1983, 43, 68–78. [Google Scholar]
- Wigley, T.M.L.; Briffa, K.R.; Jones, P.D. On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J. Clim. Appl. Meteorol. 1984, 23, 201–213. [Google Scholar] [CrossRef]
- Bunn, A.G. Statistical and visual crossdating in R using the dplR library. Dendrochronologia 2010, 28, 251–258. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A multiscalar drought index sensitive to global warming: The standardized precipitation evapotranspiration index. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef] [Green Version]
- Vicente-Serrano, S.M.; Tomas-Burguera, M.; Beguería, S.; Reig, F.; Latorre, B.; Peña-Gallardo, M.; Yolanda Luna, M.; Morata, A.; González-Hidalgo, J.C. A high-resolution dataset of drought indices for Spain. Data 2017, 2, 22. [Google Scholar] [CrossRef] [Green Version]
- Pasho, E.; Camarero, J.J.; de Luis, M.; Vicente-Serrano, S.M. Impacts of drought at different time scales on forest growth across a wide climatic gradient in north-eastern Spain. Agric. For. Meterorol. 2011, 151, 1800–1811. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Sánchez-Salguero, R.; Sangüesa-Barreda, G.; Díaz-Delgado, R.; Casals, R. Dieback and mortality of junipers caused by drought: Dissimilar growth and wood isotope patterns preceding shrub death. Agric. For. Meteorol. 2020, 291, 108078. [Google Scholar] [CrossRef]
- Villaescusa, R.; Díaz, R. Segundo Inventario Forestal Nacional (1986–1996); ICONA, Ministerio de Medio Ambiente: Madrid, Spain, 1998.
- Zang, C.; Biondi, F. Treeclim: An R package for the numerical calibration of proxy-climate relationships. Ecography 2015, 38, 431–436. [Google Scholar] [CrossRef]
- Pinheiro, J.C.; Bates, D.M. Linear Mixed-Effects Models: Basic Concepts and Examples. In Mixed-Effects Models in S and S-PLUS. Statistics and Computing; Springer: New York, NY, USA, 2000; pp. 3–56. [Google Scholar] [CrossRef]
- Spyroglou, I.; Skalák, J.; Balakhonova, V.; Benedikty, Z.; Rigas, A.G.; Hejátko, J. Mixed models as a tool for comparing groups of time series in plant sciences. Plants 2021, 10, 362. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 15 December 2020).
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. R Core Team nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1-151. 2020. Available online: https://CRAN.R-project.org/package=nlme (accessed on 1 March 2021).
- Magnusson, A.; Skaug, H.; Nielsen, A.; Berg, C.; Kristensen, K. glmmTMB: Generalized Linear Mixed Models Using Template Model Builder. R Package Version 1.4.2. 2020. Available online: https://cran.r-project.org/web/packages/glmm (accessed on 1 April 2021).
- Hyndman, R.J.; Athanasopoulos, G.; Bergmeir, C.; Caceres, G.; Chhay, L. Package ‘Forecast’. R Package Version 8.14. 2020. Available online: https://cran.r-project.org/web/packages/forecast (accessed on 1 March 2021).
- Burnham, K.P.; Anderson, D.R. Model selection and multimodel inference. In A Practical Information-Theoretic Approach; Springer: New York, NY, USA, 2002. [Google Scholar]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. R Package Version 1.6.2-1. 2018. Available online: https://cran.r-project.org/web/packages/emmeans (accessed on 1 March 2021).
- Bartoń, K. MuMIn: Multi-Model Inference. R Package Version 1.43.17. 2020. Available online: https://cran.r-project.org/web/packages/MuMIn (accessed on 15 December 2020).
- Nakagawa, S.; Johnson, P.C.D.; Schielzeth, H. The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J. R. Soc. 2017, 14, 20170213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vicente-Serrano, S.M.; Domínguez-Castro, F.; Murphy, C.; Hannaford, J.; Reig, F.; Peña-Angulo, D.; Tramblay, Y.; Trigo, R.M.; mac Donald, N.; Luna, M.Y.; et al. Long-term variability and trends in meteorological droughts in Western Europe (1851–2018). Int. J. Climatol. 2021, 41, E690–E717. [Google Scholar] [CrossRef]
- Gazol, A.; Sangüesa-Barreda, G.; Granda, E.; Camarero, J.J. Tracking the impact of drought on functionally different woody plants in a Mediterranean scrubland ecosystem. Plant Ecol. 2017, 218, 1009–1020. [Google Scholar] [CrossRef]
- Gao, S.; Liu, R.; Zhou, T.; Fang, W.; Yi, C.; Lu, R.; Zhao, X.; Luo, H. Dynamic responses of tree-ring growth to multiple dimensions of drought. Glob. Chang. Biol. 2018, 24, 5380–5390. [Google Scholar] [CrossRef] [Green Version]
- Camarero, J.J.; Gazol, A.; Sangüesa-Barreda, G.; Vergarechea, M.; Alfaro-Sánchez, R.; Cattaneo, N.; Vicente-Serrano, S.M. Tree growth is more limited by drought in rear-edge forests most of the times. For. Ecosyst. 2021, 8, 25. [Google Scholar] [CrossRef]
- Martínez-Vilalta, J.; Piñol, J. Drought-induced mortality and hydraulic architecture in pine populations of the NE Iberian Peninsula. For. Ecol. Manag. 2002, 161, 247–256. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J.; Gutiérrez, E.; González Rouco, F.; Gazol, A.; Sangüesa-Barreda, G.; Andreu-Hayles, L.; Linares, J.C.; Seftigen, K. Assessing forest vulnerability to climate warming using a process-based model of tree growth: Bad prospects for rear-edges. Glob. Chang. Biol. 2017, 23, 2705–2719. [Google Scholar] [CrossRef] [PubMed]
- Gazol, A.; Camarero, J.J.; Sangüesa-Barreda, G.; Vicente-Serrano, S.M. Post-drought resilience after forest die-off: Shifts in regeneration, composition, growth and productivity. Front. Plant Sci. 2018, 9, 1546. [Google Scholar] [CrossRef]
- Voltas, J.; Camarero, J.J.; Carulla, D.; Aguilera, M.; Oriz, A.; Ferrio, J.P. A retrospective, dual-isotope approach reveals individual predispositions to winter-drought induced tree dieback in the southernmost distribution limit of Scots pine. Plant Cell Environ. 2013, 36, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- Bhusal, N.; Han, S.G.; Yoon, T.M. Impact of drought stress on photosynthetic response, leaf water potential, and stem sap flow in two cultivars of bi-leader apple trees (Malus × domestica Borkh.). Sci. Hort. 2019, 246, 535–543. [Google Scholar] [CrossRef]
- Levanic, T.; Cater, M.; McDowell, N.G. Associations between growth, wood anatomy, carbon isotope discrimination and mortality in a Quercus robur forest. Tree Physiol. 2011, 31, 298–308. [Google Scholar] [CrossRef] [Green Version]
- Irvine, J.; Perks, M.P.; Magnani, F.; Grace, J. The response of Pinus sylvestris to drought: Stomatal control of transpiration and hydraulic conductance. Tree Phys. 1998, 18, 393–402. [Google Scholar] [CrossRef]
- Picon, C.; Guehl, J.M.; Ferhi, A. Leaf gas exchange and carbon isotope composition responses to drought in a drought-avoiding (Pinus pinaster) and a drought-tolerant (Quercus petraea) species under present and elevated atmospheric CO2 concentrations. Plant Cell Environ. 1996, 19, 182–190. [Google Scholar] [CrossRef]
- Borghetti, M.; Cinnirella, S.; Magnani, F.; Saracino, A. Impact of long-term drought on xylem embolism and growth in Pinus halepensis Mill. Trees Struct. Funct. 1998, 12, 187–195. [Google Scholar] [CrossRef]
- McDowell, N.G.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Ye, Q.; Liu, H.; Brodribb, T.J. Wood density predicts mortality threshold for diverse trees. New Phytol. 2021, 229, 3053–3057. [Google Scholar] [CrossRef] [PubMed]
- Carnicer, J.; Domingo-Marimon, C.; Ninyerola, M.; Camarero, J.J.; Bastos, A.; López-Parages, J.; Blanquer, L.; Rodríguez-Fonseca, B.; Lenton, T.M.; Dakos, V.; et al. Regime shifts of Mediterranean forest carbon uptake and reduced resilience driven by multidecadal ocean surface temperature. Glob. Chang. Biol. 2019, 25, 2825–2840. [Google Scholar] [CrossRef] [PubMed]
- Keen, R.M.; Voelker, S.L.; Bentz, B.J.; Wang, S.Y.S.; Ferrell, R. Stronger influence of growth rate than severity of drought stress on mortality of large ponderosa pines during the 2012–2015 California drought. Oecologia 2020, 194, 359–370. [Google Scholar] [CrossRef]
- Ripullone, F.; Camarero, J.J.; Colangelo, M.; Voltas, J. Variation in the access to deep soil water pools explains tree-to-tree differences in drought-triggered dieback of Mediterranean oaks. Tree Phys. 2020, 40, 591–604. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Schwalm, C.; Biondi, F.; Camarero, J.J.; Koch, G.; Litvak, M.; Ogle, K.; Shaw, J.D.; Shevliakova, E.; Williams, A.P.; et al. Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science 2015, 349, 528–532. [Google Scholar] [CrossRef] [Green Version]
- Borghetti, M.; Colangelo, M.; Ripullone, F.; Rita, A. Ondate di siccità e calore, spunti per una selvicoltura adattativa. For. J. Silvic. For. Ecol. 2021, 18, 49–57. [Google Scholar] [CrossRef]


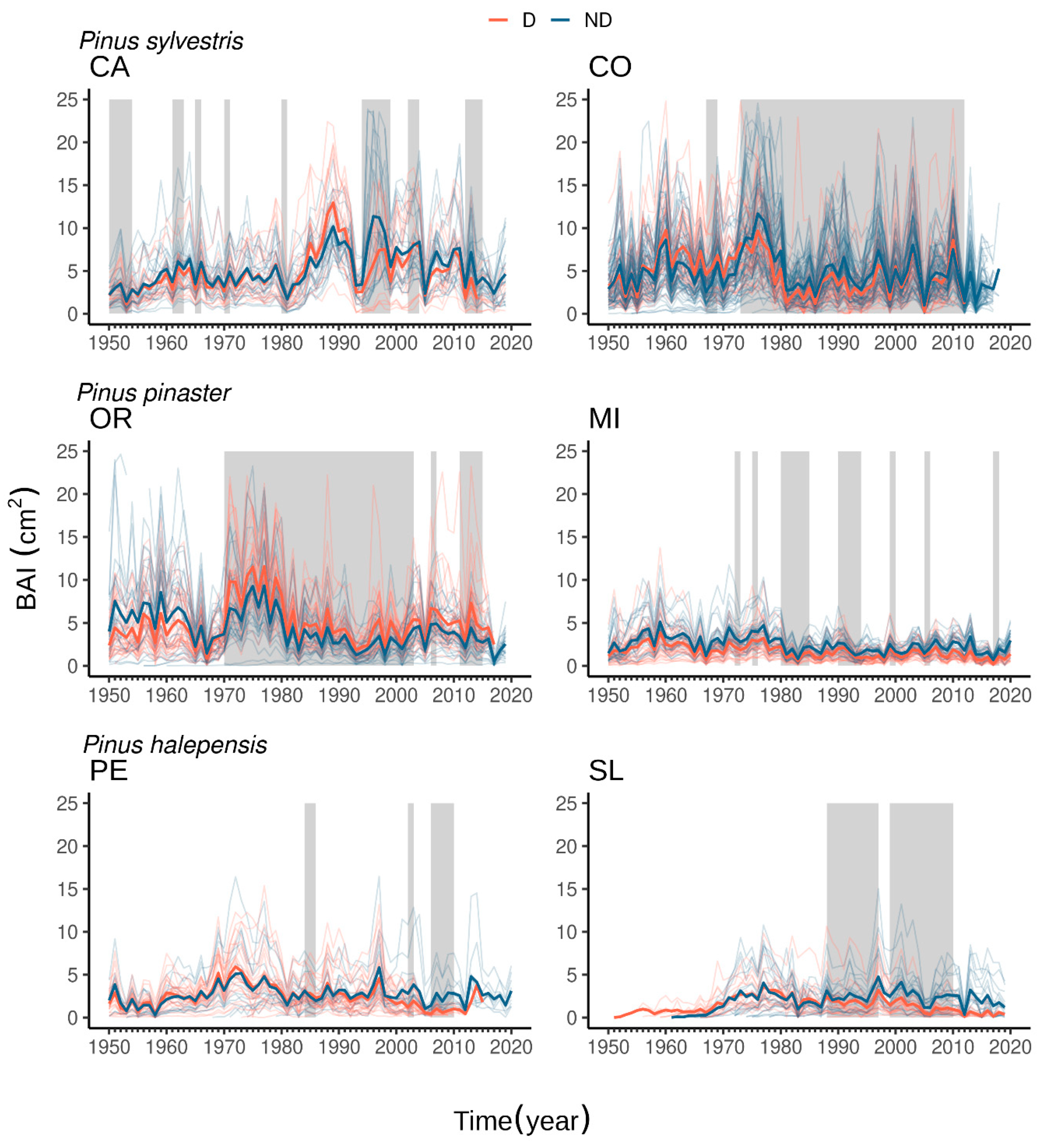
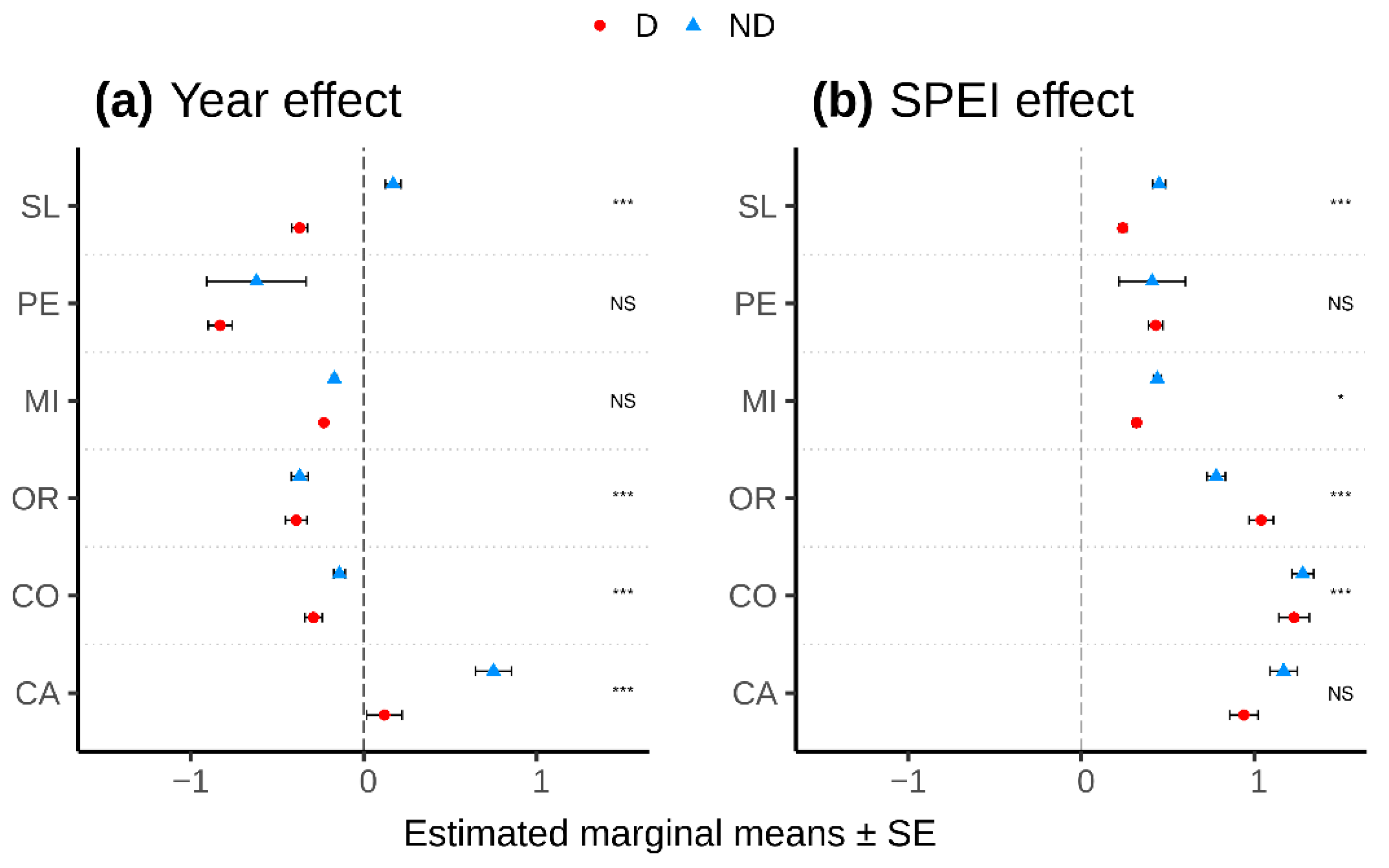
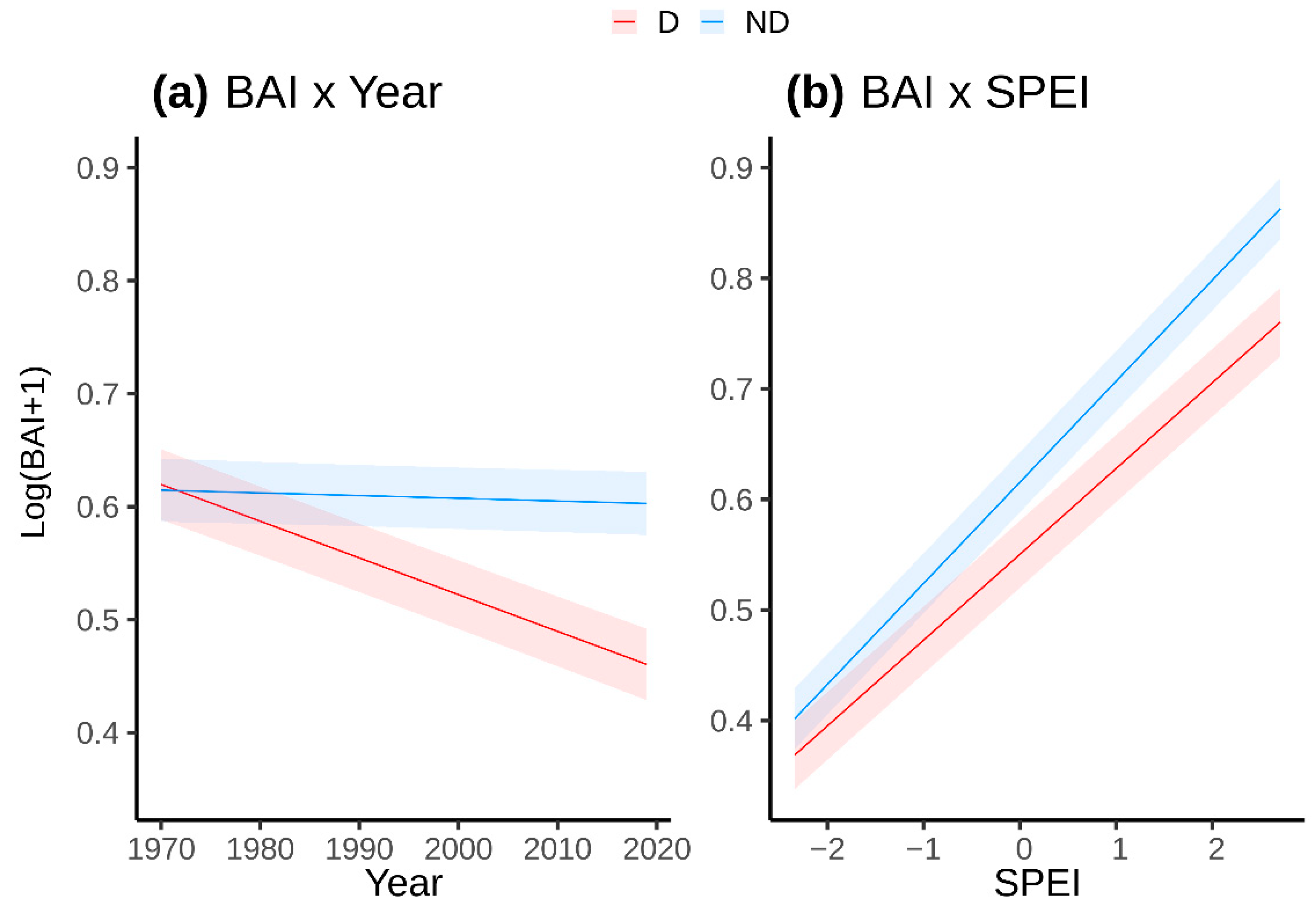
| Species | Site (Code) | Latitude N | Longitude (−W, +E) | Elevation (m a.s.l.) | Temperature (°C) | Precipitation (mm) | Undergrowth Species 1 |
|---|---|---|---|---|---|---|---|
| Pinus sylvestris | Hereña (HE) | 42.77 | −2.92 | 580 | 11.0 | 852 | Qf, Qi, Jp |
| Calomarde (CA) | 40.37 | −1.56 | 1340 | 12.2 | 390 | Qf, Jt, Jo | |
| Corbalán (CO) | 40.28 | −0.78 | 1303 | 12.6 | 421 | Pn, Qf, Qi, Jp | |
| Pinus pinaster | Orera (OR) | 41.31 | −1.45 | 884 | 13.2 | 405 | Qi |
| Miedes (MI) | 41.27 | −1.43 | 963 | 12.8 | 418 | Qi, Au | |
| Mora de Rubielos (MR) | 40.23 | −0.73 | 1186 | 12.2 | 462 | Qi, Pn, Jp, Jo | |
| Pinus halepensis | Sierra de Luna (SL) | 41.98 | −0.84 | 493 | 13.4 | 485 | Jo, Jp, Qc, Pl |
| Peñaflor (PE) | 41.78 | −0.72 | 375 | 15.5 | 376 | Jp, Jt |
| Tree Species | Site | Basal Area (m2 ha−1) | Density (Ind. ha−1) | Mortality (%) | Dbh (cm) | No. Sampled Dead/Living Trees | Age at 1.3 m (years) |
|---|---|---|---|---|---|---|---|
| P. sylvestris | HE | 4.26 | 799 | 25.0 | 20.5 ± 1.7 | 2/30 | 107 ± 2 |
| CA | 6.20 | 784 | 37.1 | 25.0 ± 1.5 | 9/13 | 133 ± 5 | |
| CO | 4.00 | 126 | 95.0 | 27.3 ± 1.0 | 38/57 | 138 ± 6 | |
| P. pinaster | OR | 6.32 | 779 | 50.0 | 28.3 ± 0.9 | 17/16 | 87 ± 2 |
| MI | 5.73 | 791 | 41.2 | 21.7 ± 1.3 | 15/15 | 82 ± 2 | |
| MR | 2.05 | 608 | 21.1 | 13.9 ± 1.7 | 2/22 | 85 ± 6 | |
| P. halepensis | SL | 2.12 | 577 | 26.3 | 12.8 ± 2.4 | 13/13 | 53 ± 3 |
| PE | 4.50 | 179 | 44.0 | 32.3 ± 1.4 | 26/14 | 78 ± 5 |
| Species | Site | Status | Basal Area Increment (cm2) | Tree-Ring Width (mm) | First-Order Autocorrelation | Mean Sensitivity | rbar | Period with EPS > 0.85 |
|---|---|---|---|---|---|---|---|---|
| P. sylvestris | CA | ND | 2.91 ± 0.18 | 0.88 ± 0.50 | 0.63 | 0.371 | 0.51 | 1845–2019 |
| D | 3.54 ± 0.20 | 0.76 ± 0.46 | 0.66 | 0.400 | 0.50 | 1895–2019 | ||
| CO | ND | 3.11 ± 0.17 | 1.01 ± 0.62b | 1.01 | 0.436 | 0.56 | 1844–2018 | |
| D | 3.80 ± 0.14 | 0.71 ± 0.47a | 0.60 | 0.492 | 0.62 | 1838–2012 | ||
| P. pinaster | OR | ND | 4.14 ± 0.21 | 1.24 ± 1.11 | 0.73 | 0.472 | 0.60 | 1920–2019 |
| D | 4.85 ± 0.21 | 1.25 ± 1.00 | 0.72 | 0.452 | 0.63 | 1917–2016 | ||
| MI | ND | 2.10 ± 0.11 | 0.88 ± 0.63 | 0.61 | 0.429 | 0.60 | 1921–2020 | |
| D | 1.52 ± 0.07 | 0.74 ± 0.61 | 0.71 | 0.461 | 0.54 | 1921–2020 | ||
| P. halepensis | PE | ND | 2.74 ± 0.13 | 1.08 ± 0.66 | 0.52 | 0.442 | 0.50 | 1946–2020 |
| D | 2.17 ± 0.12 | 0.97 ± 0.68 | 0.65 | 0.447 | 0.49 | 1916–2015 | ||
| SL | ND | 2.34 ± 0.12 | 1.13 ± 0.78 | 0.60 | 0.480 | 0.48 | 1969–2019 | |
| D | 1.64 ± 0.12 | 0.96 ± 0.77 | 0.76 | 0.486 | 0.39 | 1966–2015 |
| Species | Site | Intercept | Year | Dbh | Status | SPEI | SPEI × Status | Year × Status | Akaike Weight | R2m (%) | R2c (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| P. sylvestris | CA | −1.252 | 0.001 | 0.011 | −6.183 | 0.091 | – | 0.003 | 0.67 | 17.7 | 37.1 |
| CO | 5.147 | −0.002 | – | −2.659 | 0.127 | −0.012 | 0.001 | 0.97 | 20.8 | 59.6 | |
| P. pinaster | OR | 5.924 | −0.002 | 0.015 | −0.089 | 0.103 | −0.015 | – | 0.66 | 34.3 | 62.2 |
| MI | 5.500 | −0.003 | 0.016 | −1.746 | 0.058 | 0.014 | 0.001 | 0.99 | 36.5 | 59.7 | |
| P. halepensis | PE | 20.180 | −0.010 | – | – | 0.063 | – | – | 0.21 | 29.7 | 66.4 |
| SL | 10.736 | −0.005 | – | −14.143 | 0.049 | 0.024 | 0.007 | 0.53 | 14.9 | 44.4 | |
| All sites | – | 7.025 | −0.003 | 0.011 | −5.967 | 0.067 | 0.012 | 0.003 | 0.99 | 11.6 | 61.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valeriano, C.; Gazol, A.; Colangelo, M.; Camarero, J.J. Drought Drives Growth and Mortality Rates in Three Pine Species under Mediterranean Conditions. Forests 2021, 12, 1700. https://doi.org/10.3390/f12121700
Valeriano C, Gazol A, Colangelo M, Camarero JJ. Drought Drives Growth and Mortality Rates in Three Pine Species under Mediterranean Conditions. Forests. 2021; 12(12):1700. https://doi.org/10.3390/f12121700
Chicago/Turabian StyleValeriano, Cristina, Antonio Gazol, Michele Colangelo, and Jesús Julio Camarero. 2021. "Drought Drives Growth and Mortality Rates in Three Pine Species under Mediterranean Conditions" Forests 12, no. 12: 1700. https://doi.org/10.3390/f12121700
APA StyleValeriano, C., Gazol, A., Colangelo, M., & Camarero, J. J. (2021). Drought Drives Growth and Mortality Rates in Three Pine Species under Mediterranean Conditions. Forests, 12(12), 1700. https://doi.org/10.3390/f12121700








