Identification of Alnus glutinosa L. and A. incana (L.) Moench. Hybrids in Natural Forests Using Nuclear DNA Microsatellite and Morphometric Markers
Abstract
1. Introduction
2. Materials and Methods
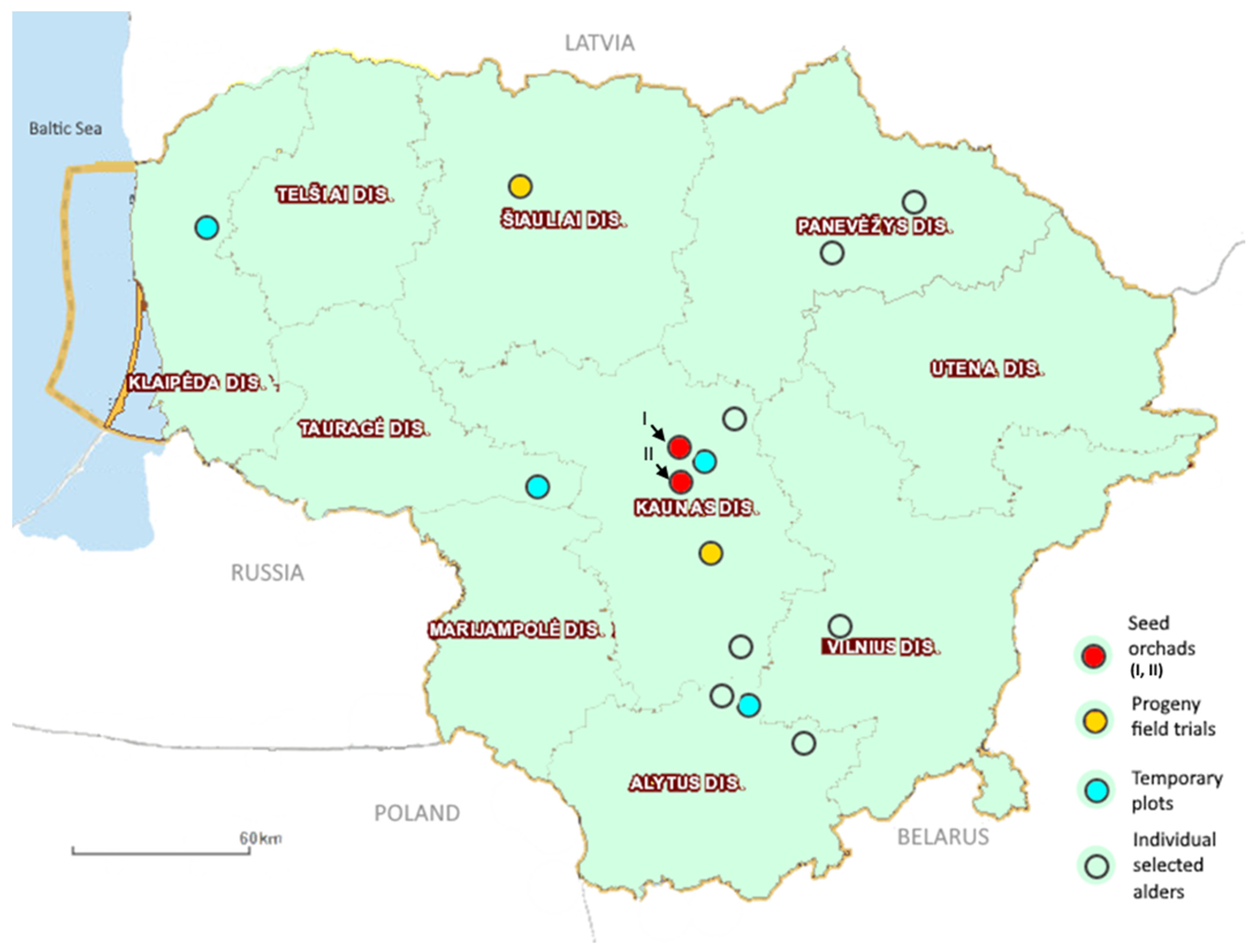
2.1. Material
2.2. Morphology Traits
2.3. DNA Extraction and Microsatellite Genotyping
2.4. Molecular Data Analysis
2.5. Statistical Analysis of Leaf Morphometric Traits
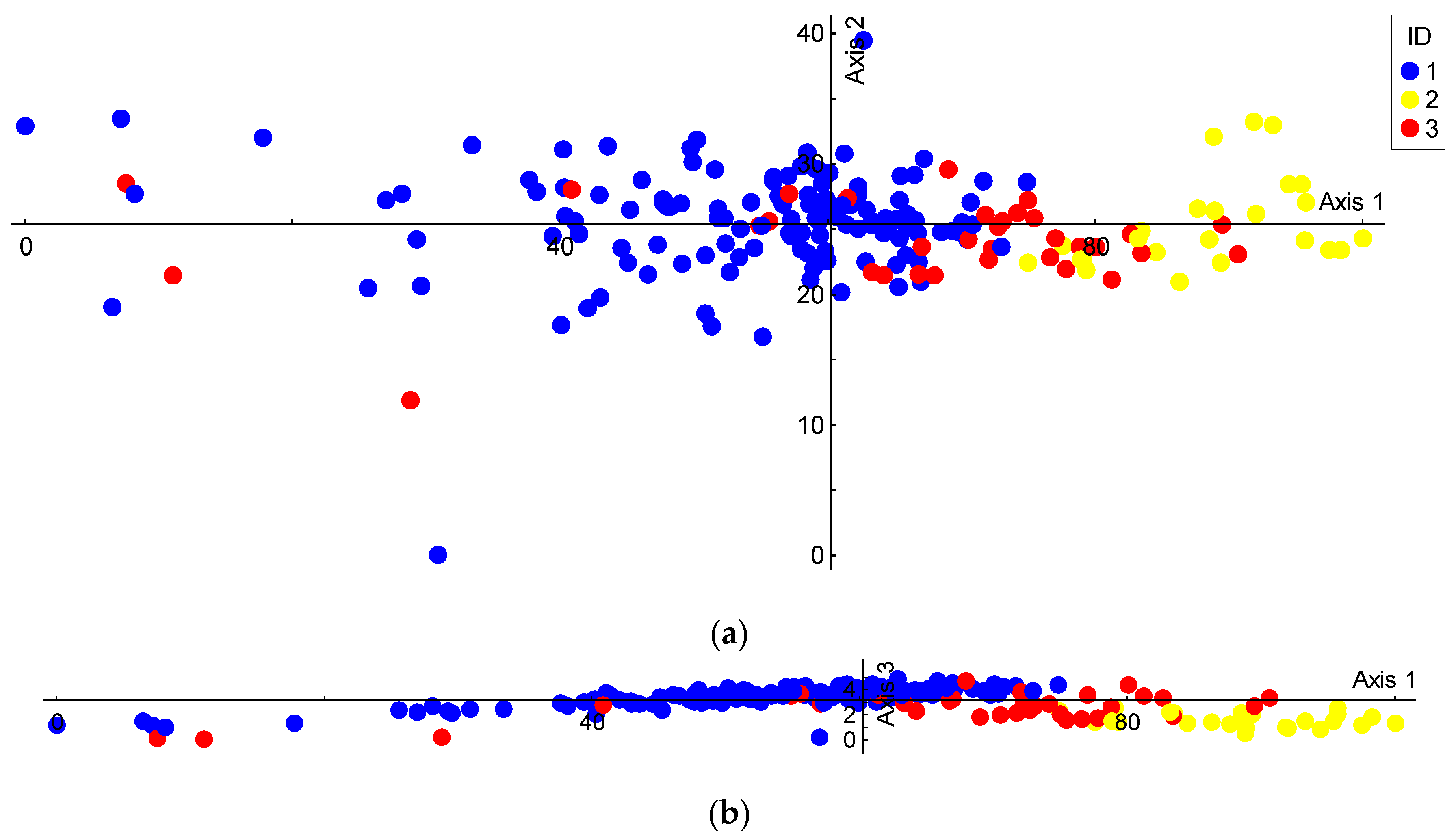
3. Results
3.1. Species Genetic Differentiation
3.2. Genetic Diversity
3.3. Leaf Morphology Variation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neale, D.B.; Wheeler, N.C. Hybridization and Introgression. In The Conifers: Genomes, Variation and Evolution; Neale, D.B., Wheeler, N.C., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 387–429. ISBN 978-3-319-46807-5. [Google Scholar]
- Anderson, E. Introgressive Hybridization. In Introgressive Hybridization; John Wiley & Sons: Hoboken, NJ, USA, 1949. [Google Scholar]
- Arnold, M.L. Natural Hybridization as an Evolutionary Process. Annu. Rev. Ecol. Syst. 1992, 23, 237–261. [Google Scholar] [CrossRef]
- Seehausen, O. Hybridization and Adaptive Radiation. Trends Ecol. Evol. 2004, 19, 198–207. [Google Scholar] [CrossRef]
- Tamošaitis, S.; Jurkšienė, G.; Petrokas, R.; Buchovska, J.; Kavaliauskienė, I.; Danusevičius, D.; Baliuckas, V. Dissecting Taxonomic Variants within Ulmus Spp. Complex in Natural Forests with the Aid of Microsatellite and Morphometric Markers. Forests 2021, 12, 653. [Google Scholar] [CrossRef]
- Choler, P.; Erschbamer, B.; Tribsch, A.; Gielly, L.; Taberlet, P. Genetic Introgression as a Potential to Widen a Species’ Niche: Insights from Alpine Carex curvula. Proc. Natl. Acad. Sci. USA 2004, 101, 171–176. [Google Scholar] [CrossRef]
- Martin, N.H.; Bouck, A.C.; Arnold, M.L. Detecting Adaptive Trait Introgression Between Iris Fulva and I. brevicaulis in Highly Selective Field Conditions. Genetics 2006, 172, 2481–2489. [Google Scholar] [CrossRef]
- Castric, V.; Bechsgaard, J.; Schierup, M.H.; Vekemans, X. Repeated Adaptive Introgression at a Gene under Multiallelic Balancing Selection. PLoS Genet. 2008, 4, e1000168. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Cui, M.-L.; Cubas, P.; Gillies, A.; Lee, K.; Chapman, M.A.; Abbott, R.J.; Coen, E. Regulatory Genes Control a Key Morphological and Ecological Trait Transferred Between Species. Science 2008, 322, 1116–1119. [Google Scholar] [CrossRef]
- Twyford, A.D.; Ennos, R.A. Next-Generation Hybridization and Introgression. Heredity 2012, 108, 179–189. [Google Scholar] [CrossRef]
- Arnold, M.L.; Hodges, S.A. Are Natural Hybrids Fit or Unfit Relative to Their Parents? Trends Ecol. Evol. 1995, 10, 67–71. [Google Scholar] [CrossRef]
- Arnold, M.L. Natural Hybridization and Evolution; Oxford University Press: Oxford, UK, 1997; ISBN 978-0-19-509975-1. [Google Scholar]
- Van Droogenbroeck, B.; Kyndt, T.; Romeijn-Peeters, E.; Van Thuyne, W.; Goetghebeur, P.; Romero-Motochi, J.P.; Gheysen, G. Evidence of Natural Hybridization and Introgression between Vasconcellea Species (Caricaceae) from Southern Ecuador Revealed by Chloroplast, Mitochondrial and Nuclear DNA Markers. Ann. Bot. 2006, 97, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.A.; Francisco-Ortega, J.; Jansen, R.K. Hybridization and the Extinction of Rare Plant Species. Conserv. Biol. 1996, 10, 10–16. [Google Scholar] [CrossRef]
- Soltis, P.S.; Soltis, D.E. The Role of Hybridization in Plant Speciation. Annu. Rev. Plant Biol. 2009, 60, 561–588. [Google Scholar] [CrossRef] [PubMed]
- Vít, P.; Wolfová, K.; Urfus, T.; Tájek, P.; Suda, J. Interspecific Hybridization between Rare and Common Plant Congeners Inferred from Genome Size Data: Assessing the Threat to the Czech Serpentine Endemic Cerastium alsinifolium. Preslia 2014, 86, 95–117. [Google Scholar]
- Claessens, H.; Oosterbaan, A.; Savill, P.; Rondeux, J. A Review of the Characteristics of Black Alder (Alnus Glutinosa (L.) Gaertn.) and Their Implications for Silvicultural Practices. For. Int. J. For. Res. 2010, 83, 163–175. [Google Scholar] [CrossRef]
- Durrant, T.H.; de Rigo, D.; Caudullo, G. Alnus Incana in Europe: Distribution, Habitat, Usage and Threats. In European Atlas of Forest Tree Species; San-Miguel Ayanz, J., de Rigo, D., Caudullo, G., Houston Durrant, T., Mauri, A., Eds.; EU Publication Office: Luxembourg, 2016; Volume 2. [Google Scholar]
- Hylander, N. Leaved and Small-Leaved Forms of Alnus Glutinosa and A. Incana. Sven. Bot. Tidskr. 1957, 51, 437–453. [Google Scholar]
- Mizushima, M. On a Hybrid of Alnus. J. Jpn. Bot. 1957, 32, 1–5. [Google Scholar]
- Steele, F.L. Introgression of Alnus Serrulata and Alnus Rugosa. Rhodora 1961, 63, 297–304. [Google Scholar]
- Furlow, J.J. The Systematics of the American Species of Alnus (Betulaceae). Rhodora 1979, 81, 1–121. [Google Scholar]
- Bobrov, E.G. Some Traits of Modern History of Flora and Vegetation of the Southern Part of the Russian Far East. Bot. Zhurnal 1980, 65, 172–184. [Google Scholar]
- Bousguet, I.; Cheliar, W.M.; Lalonde, M. Allozyme Divergence and Introgressive Hybridization between Alnus Crispa and Alnus Sinuata (Betulaceae). Am. J. Bot. 1989, 76, 228–229. [Google Scholar]
- Hall, R.B.; Burgess, D. Evaluation of Alnus Species and Hybrids. Biomass 1990, 22, 21–34. [Google Scholar] [CrossRef]
- Bousquet, J.; Cheliak, W.M.; Wang, J.; Lalonde, M. Genetic Divergence and Introgressive Hybridization Between Alnus Sinuata andA. Crispa (Betulaceae). Pl. Syst. Evol. 1990, 170, 107–124. [Google Scholar] [CrossRef]
- King, R.A.; Ferris, C. Chloroplast DNA and Nuclear DNA Variation in the Sympatric Alder Species, Alnus Cordata (Lois.) Duby and A. Glutinosa (L.) Gaertn. Biol. J. Linn. Soc. 2000, 70, 147–160. [Google Scholar] [CrossRef]
- Banaev, E.; Bažant, V. Study of Natural Hybridization between Alnus Incana (L.) Moench. and Alnus Glutinosa (L.) Gaertn. J. For. Sci. 2007, 53, 66–73. [Google Scholar] [CrossRef]
- Zhuk, A.; Veinberga, I.; Daugavietis, M.; Rungis, D. Cross-Species Amplification of Betula Pendula Roth. Simple Sequence Repeat Markers in Alnus Species. Balt. For. 2008, 14, 116–121. [Google Scholar]
- Jurkšienė, G.; Baliuckas, V. Natural Hybridization and Features between Alnus Incana (L.) Moenc. and Alnus Glutinosa (L.) Gaertn. in Lithuania. In Book of Abstracts, Proceedings of the Agrosym 2019: X International Scientific Agriculture Symposium, Jahorina, Bosnia and Herzegovina, 3–6 October 2019; Kovacevic, D., Ed.; University of East Sarajevo, Faculty of Agriculture: Sarajevo, Bosnia and Herzegovina, 2019. [Google Scholar]
- Šmíd, J.; Douda, J.; Krak, K.; Mandák, B. Analyses of Hybrid Viability across a Hybrid Zone between Two Alnus Species Using Microsatellites and CpDNA Markers. Genes 2020, 11, 770. [Google Scholar] [CrossRef]
- Stanton, B.J.; Best, T.; Islam-Faridi, N.; Gantz, C.; Haiby, K.; Johnson, L.J.; Shuren, R.; Stanish, A.; Staton, M.; Weathers, T.C.; et al. Inter-Specific Hybridization of Alnusrubra and Alnus Rhombifolia:Preliminary Report of a New Taxon and DNA Marker Resources for Bioenergy Feedstockproduction. Tree Genet. Genomes 2020, 16, 70. [Google Scholar] [CrossRef]
- Villani, F.; Castellana, S.; Beritognolo, I.; Cherubini, M.; Chiocchini, F.; Battistelli, A.; Mattioni, C. Genetic Variability of Alnus Cordata (Loisel.) Duby Populations and Introgressive Hybridization with A. Glutinosa (L.) Gaertn. in Southern Italy: Implication for Conservation and Management of Genetic Resources. Forests 2021, 12, 655. [Google Scholar] [CrossRef]
- Kobendza, R. Meiszance Naturalne Olszy Szarej i Czarnej w Polsce (Alnus Incana Moench.× Alnus Glutinosa Gaertn.–Alnus Hybrida Alex. Braun.). Rocznik Dendrol. 1956, 56, 57–62. (In Polish) [Google Scholar]
- Kundzinsh, A.V. Hybrids of Alnus Glutinosa and A. Incana in the Forests of the Latvian SSR. Izv. AN Latv. SSR 1957, 2, 115. [Google Scholar]
- Voronova, A.; Lazdina, D.; Korica, A.; Veinberga, I.; Liepins, K.; Rungis, D. Evaluation of Allelic Content in an Experimental Alder (Alnus Spp.) Plantation. Acta Biol. Univ. Daugavp. 2015, 15, 227–240. [Google Scholar]
- Parfenov, V.I. Dependence of Distribution and Adaptation of Plant Species on the Area Borders; Nauka i Tekhnika: Minsk, Belarus, 1980. [Google Scholar]
- Parnell, J. Variation and Hybridisation of Alnus Miller in Ireland. Watsonia 1994, 20, 67–70. [Google Scholar]
- Olshansky, I.G. Genus Alnus Mill. (Betulaceae SF Gray) in the flora of Ukraine (Piд Alnus Mill. (Betulaceae SF Gray) y флopi Уkpaїни). Бioлoгічні Cucmeми 2014, 6, 72–77. [Google Scholar]
- Vander Mijnsbrugge, K. Morphological Dissection of Leaf, Bud and Infructescence Traits of the Interfertile Native A. Glutinosa and Non-Native A. Incana in Flanders (Northern Part of Belgium). Trees 2015, 29, 1661–1672. [Google Scholar] [CrossRef]
- Vidaković, V.; Stefanović, M.; Novaković, M.; Jadranin, M.; Popović, Z.; Matić, R.; Tešević, V.; Bojović, S. Inter-and Intraspecific Variability of Selected Diarylheptanoid Compounds and Leaf Morphometric Traits in Alnus Glutinosa and Alnus Incana. Holzforschung 2018, 72, 1031–1041. [Google Scholar] [CrossRef]
- Yurkevich, I.D.; Geltman, V.S.; Lovcij, N.F. Types and Associations of Black Alder Forests (Tunы u Accoцuaцuu Чернooльхoвых Лecoв); Nauka i Tiechnika: Minsk, Belarus, 1968. [Google Scholar]
- Lučnik, Z.I. Introduction of Trees and Shrubs in Shrubs in the Altai Krai; Kolos: Moscow, Russia, 1974. [Google Scholar]
- Ramanauskas, V. Dendrologija (Dendrology); Mintis: Vilnius, Lithuania, 1973. [Google Scholar]
- Danusevičius, D.; Kembrytė, R.; Buchovska, J.; Baliuckas, V.; Kavaliauskas, D. Genetic Signature of the Natural Gene Pool of Tilia Cordata Mill. in Lithuania: Compound Evolutionary and Anthropogenic Effects. Ecol. Evol. 2021, 11, 6260–6275. [Google Scholar] [CrossRef]
- Rieseberg, L.H.; Ellstrand, N.C.; Arnold, D.M. What Can Molecular and Morphological Markers Tell Us About Plant Hybridization? Crit. Rev. Plant Sci. 1993, 12, 213–241. [Google Scholar] [CrossRef]
- Rieseberg, L.H. The Role of Hybridization in Evolution: Old Wine in New Skins. Am. J. Bot. 1995, 82, 944–953. [Google Scholar] [CrossRef]
- Mejnartowicz, L. Morphology and Growth of Alnus Incana× Glutinosa Hybrids. Arbor. Kórnickie Rocz. XXVI 1982, 26, 15. [Google Scholar]
- Mejnartowicz, L. Evidence for Long-Term Heterosis Phenomenon in the Alnus Incana× Glutinosa F-1 Hybrids. Silvae Genet. 1999, 48. Available online: https://agris.fao.org/agris-search/search.do?request_locale=en&recordID=DE1999T30143&sourceQuery=&query=&sortField=&sortOrder=&countryResource=&agrovocString=&advQuery=¢erString=&enableField= (accessed on 24 September 2021).
- Bašić, N.; Selimović, E.; Pustahija, F. Morphological Identification of Nothospecies Alnus × Pubescens Tausch. and Their New Localities in Central Bosnia. Rad. Šumarskog Fak. Univ. Sarajev. 2014, 44, 15–24. [Google Scholar]
- Viljevac, B. Morfološki i Genetički Dokazi Hibridizacije Izmedju Bijele (Alnus Incana (L.) Moench) i Crne Johe (A. Glutinosa (L.) Gaertn.) u Podravini. Ph.D. Thesis, University of Zagreb, Faculty of Science, Department of Biology, Zagreb, Croatia, 2020. (In Croatian). [Google Scholar]
- Harrison, R.G.; Larson, E.L. Hybridization, Introgression, and the Nature of Species Boundaries. J. Hered. 2014, 105, 795–809. [Google Scholar] [CrossRef]
- Zalapa, J.E.; Brunet, J.; Guries, R.P. Original Article: The Extent of Hybridization and Its Impact on the Genetic Diversity and Population Structure of an Invasive Tree, Ulmus Pumila (Ulmaceae). Evol. Appl. 2010, 3, 157–168. [Google Scholar] [CrossRef]
- Brunet, J.; Zalapa, J.E.; Pecori, F.; Santini, A. Hybridization and Introgression between the Exotic Siberian Elm, Ulmus Pumila, and the Native Field Elm, U. Minor, in Italy. Biol. Invasions 2013, 15, 2717–2730. [Google Scholar] [CrossRef]
- Buiteveld, J.; Vanden Broeck, A.; Cox, K.; Collin, E. Human Impact on the Genetic Diversity of Dutch Field Elm (Ulmus Minor) Populations in the Netherlands: Implications for Conservation. Plant Ecol. Evol. 2016, 149, 165–176. [Google Scholar] [CrossRef]
- Hirsch, H.; Brunet, J.; Zalapa, J.E.; von Wehrden, H.; Hartmann, M.; Kleindienst, C.; Schlautman, B.; Kosman, E.; Wesche, K.; Renison, D.; et al. Intra- and Interspecific Hybridization in Invasive Siberian Elm. Biol. Invasions 2017, 19, 1889–1904. [Google Scholar] [CrossRef]
- Oliveira, E.J.; Pádua, J.G.; Zucchi, M.I.; Vencovsky, R.; Vieira, M.L.C. Origin, Evolution and Genome Distribution of Microsatellites. Genet. Mol. Biol. 2006, 29, 294–307. [Google Scholar] [CrossRef]
- Barbará, T.; Palma-Silva, C.; Paggi, G.M.; Bered, F.; Fay, M.F.; Lexer, C. Cross-Species Transfer of Nuclear Microsatellite Markers: Potential and Limitations. Mol. Ecol. 2007, 16, 3759–3767. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.R.; Burke, J.M. EST-SSRs as a Resource for Population Genetic Analyses. Heredity 2007, 99, 125–132. [Google Scholar] [CrossRef]
- Wang, M.L.; Barkley, N.A.; Jenkins, T.M. Microsatellite Markers in Plants and Insects. Part I: Applications of Biotechnology. Genes Genomes Genom. 2009, 3, 54–67. [Google Scholar]
- Prat, D.; Leger, C.; Bojovic, S. Genetic Diversity among Alnus Glutinosa (L.) Gaertn. Populations. Acta Oecologica 1992, 13, 469–477. [Google Scholar]
- King, R.A.; Ferris, C. Chloroplast DNA Phylogeography of Alnus Glutinosa (L.) Gaertn. Mol. Ecol. 1998, 7, 1151–1161. [Google Scholar] [CrossRef]
- Mejnartowicz, L. Genetic Variation within and among Naturally Regenerating Populations of Alder [Alnus Glutinosa]. Acta Societatis Botanicorum Poloniae 2008, 77, 105–110. [Google Scholar] [CrossRef][Green Version]
- Lepais, O.; Muller, S.D.; Saad-Limam, S.B.; Benslama, M.; Rhazi, L.; Belouahem-Abed, D.; Daoud-Bouattour, A.; Gammar, A.M.; Ghrabi-Gammar, Z.; Bacles, C.F.E. High Genetic Diversity and Distinctiveness of Rear-Edge Climate Relicts Maintained by Ancient Tetraploidisation for Alnus Glutinosa. PLoS ONE 2013, 8, e75029. [Google Scholar] [CrossRef] [PubMed]
- Havrdová, A.; Douda, J.; Krak, K.; Vít, P.; Hadincová, V.; Zákravský, P.; Mandák, B. Higher Genetic Diversity in Recolonized Areas than in Refugia of Alnus Glutinosa Triggered by Continent-Wide Lineage Admixture. Mol. Ecol. 2015, 24, 4759–4777. [Google Scholar] [CrossRef]
- Cubry, P.; Gallagher, E.; O’Connor, E.; Kelleher, C.T. Phylogeography and Population Genetics of Black Alder (Alnus Glutinosa (L.) Gaertn.) in Ireland: Putting It in a European Context. Tree Genet. Genomes 2015, 11, 99. [Google Scholar] [CrossRef]
- Mandák, B.; Havrdová, A.; Krak, K.; Hadincová, V.; Vít, P.; Zákravský, P.; Douda, J. Recent Similarity in Distribution Ranges Does Not Mean a Similar Postglacial History: A Phylogeographical Study of the Boreal Tree Species Alnus Incana Based on Microsatellite and Chloroplast DNA Variation. New Phytol. 2016, 210, 1395–1407. [Google Scholar] [CrossRef] [PubMed]
- Poljak, I.; Idžojtić, M.; Šapić, I.; Korijan, P.; Vukelić, J. Diversity and Structure of Croatian Continental and Alpine-Dinaric Populations of Grey Alder (Alnus Incana /L./ Moench Subsp. Incana); Isolation by Distance and Environment Explains Phenotypic Divergence. Šumarski List 2018, 142, 19–31. [Google Scholar] [CrossRef]
- Ager, A.A.; Heilman, P.E.; Stettler, R.F. Genetic Variation in Red Alder (Alnusrubra) in Relation to Native Climate and Geography. Can. J. For. Res. 1993, 23, 1930–1939. [Google Scholar] [CrossRef]
- Dang, Q.L.; Xie, C.Y.; Ying, C.; Guy, R.D. Genetic Variation of Ecophysiological Traits in Red Alder (Alnusrubra Bong.). Can. J. For. Res. 1994, 24, 2150–2156. [Google Scholar] [CrossRef]
- Hamann, A.; El-Kassaby, Y.A.; Koshy, M.P.; Namkoong, G. Multivariate Analysis of Allozymic and Quantitative Trait Variation in Alnus Rubra: Geographic Patterns and Evolutionary Implications. Can. J. For. Res. 1998, 28, 1557–1565. [Google Scholar] [CrossRef]
- Xie, C.-Y.; El-Kassaby, Y.A.; Ying, C.C. Genetics of Red Alder (Alnus Rubra Bong.) Populations in British Columbia and Its Implications for Gene Resources Management. New For. 2002, 24, 97–112. [Google Scholar] [CrossRef]
- Paus-Glowacki, W.; Meinartowicz, L. Serological Investigation of Alnus Incana x Glutinosa Hybrids and Their Parental Species. Silvae Genet. 1992, 41, 2. [Google Scholar]
- Poljak, I.; Idžojtić, M.; Šapić, I.; Vukelić, J.; Zebec, M. Variability of Populations of White (Alnus Incana /L./ Moench) and Black Alder (A. Glutinosa /L./ Gaertn.) In the Mura and Drava Area According to Leaf Morphological Characteristics (Varijabilnost Populacija Bijele (Alnus Incana/L./Moench) i Crne Johe (A. Glutinosa/L./Gaertn.) Na Području Mure i Drave Prema Morfološkim Obilježjima Listova). Šumarski List 2014, 138, 7–16. [Google Scholar]
- Bousquet, J.; Cheliak, W.M.; Lalonde, M. Allozyme Variability in Natural Populations of Green Alder (Alnus Crispa) in Quebec. Genome 1987, 29, 345–352. [Google Scholar] [CrossRef]
- Peev, C.I.; Vlase, L.; Antal, D.S.; Dehelean, C.A.; Szabadai, Z. Determination of Some Polyphenolic Compounds in Buds of Alnus and Corylus Species by HPLC. Chem. Nat. Compd. 2007, 43, 259–262. [Google Scholar] [CrossRef]
- Dumolin, S.; Demesure, B.; Petit, R.J. Inheritance of Chloroplast and Mitochondrial Genomes in Pedunculate Oak Investigated with an Efficient PCR Method. Theoret. Appl. Genetics 1995, 91, 1253–1256. [Google Scholar] [CrossRef]
- Wu, B.; Lian, C.; Hogetsu, T. Development of Microsatellite Markers in White Birch (Betula Platyphylla Var. Japonica). Mol. Ecol. Notes 2002, 2, 413–415. [Google Scholar] [CrossRef]
- Ogyu, K.; Tsuda, Y.; Sugaya, T.; Yoshimaru, H.; Ide, Y. Identification and Characterization of Microsatellite Loci in Betula Maximowicziana Regel. Molecular Ecology Notes 2003, 3, 268–269. [Google Scholar] [CrossRef]
- Kulju, K.K.M.; Pekkinen, M.; Varvio, S. Twenty-Three Microsatellite Primer Pairs for Betula Pendula (Betulaceae). Molecular Ecol. Notes 2004, 4, 471–473. [Google Scholar] [CrossRef]
- Gürcan, K.; Mehlenbacher, S.A. Development of Microsatellite Marker Loci for European Hazelnut (Corylus Avellana L.) from ISSR Fragments. Mol. Breeding 2010, 26, 551–559. [Google Scholar] [CrossRef]
- Lance, S.L.; Jones, K.L.; Hagen, C.; Glenn, T.C.; Jones, J.M.; Gibson, J.P. Development and Characterization of Nineteen Polymorphic Microsatellite Loci from Seaside Alder, Alnus Maritima. Conserv. Genet. 2009, 10, 1907–1910. [Google Scholar] [CrossRef]
- Tsuda, Y.; Ueno, S.; Ide, Y.; Tsumura, Y. Development of 14 EST-SSRs for Betula Maximowicziana and Their Applicability to Related Species. Conserv. Genet. 2009, 10, 661–664. [Google Scholar] [CrossRef]
- Lepais, O.; Bacles, C.F.E. De Novo Discovery and Multiplexed Amplification of Microsatellite Markers for Black Alder (Alnus Glutinosa) and Related Species Using SSR-Enriched Shotgun Pyrosequencing. J. Hered. 2011, 102, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of Population Structure Using Multilocus Genotype Data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the Number of Clusters of Individuals Using the Software Structure: A Simulation Study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; vonHoldt, B.M. STRUCTURE HARVESTER: A Website and Program for Visualizing STRUCTURE Output and Implementing the Evanno Method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Blair, A.C.; Hufbauer, R.A. Hybridization and Invasion: One of North America’s Most Devastating Invasive Plants Shows Evidence for a History of Interspecific Hybridization. Evol. Appl. 2010, 3, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, N.M.; Mayzel, J.; Jakobsson, M.; Rosenberg, N.A.; Mayrose, I. Clumpak: A Program for Identifying Clustering Modes and Packaging Population Structure Inferences across K. Mol. Ecol. Resour. 2015, 15, 1179–1191. [Google Scholar] [CrossRef]
- Anderson, E.C.; Thompson, E.A. A Model-Based Method for Identifying Species Hybrids Using Multilocus Genetic Data. Genetics 2002, 160, 1217–1229. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic Analysis in Excel. Population Genetic Software for Teaching and Research-an Update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Nei, M. Genetic Distance between Populations. Am. Nat. 1972, 106, 283–292. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. Genalex 6: Genetic Analysis in Excel. Population Genetic Software for Teaching and Research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Kamvar, Z.N.; Tabima, J.F.; Grünwald, N.J. Poppr: An R Package for Genetic Analysis of Populations with Clonal, Partially Clonal, and/or Sexual Reproduction. PeerJ 2014, 2, e281. [Google Scholar] [CrossRef]
- Goudet, J. FSTAT, a Program to Estimate and Test Gene Diversities and Fixation Indices, Version 2.9.3. 2001. Available online: http://www2.unil.ch/popgen/softwares/fstat.htm (accessed on 20 May 2021).
- Jombart, T. An Introduction to Adegent 2.0. 0. R-Tutorial; Imperial College London, MRC Centre for Outbreak Analysis and Modelling: London, UK, 2015. [Google Scholar]
- Jombart, T.; Collins, C. A Tutorial for Discriminant Analysis of Principal Components (DAPC) Using Adegenet 2.0.0; Imperial College London, MRC Centre for Outbreak Analysis and Modelling: London, UK, 2015; Volume 43. [Google Scholar]
- Nei, M. Estimation of Average Heterozygosity and Genetic Distance from a Small Number of Individuals. Genetics 1978, 89, 583–590. [Google Scholar] [CrossRef]
- Addinsoft XLSTAT Statistical and Data Analysis Solution; Addinsoft: New York, NY, USA, 2020.
- McCune, B.; Mefford, M.J. PC-ORD. Multivariate Analysis of Ecological Data; MjM Software: Gleneden Beach, OR, USA, 2006. [Google Scholar]
- Li, X.; Wei, G.; El-Kassaby, Y.A.; Fang, Y. Hybridization and Introgression in Sympatric and Allopatric Populations of Four Oak Species. BMC Plant Biol. 2021, 21, 266. [Google Scholar] [CrossRef] [PubMed]
- Cannon, C.H.; Scher, C.L. Exploring the Potential of Gametic Reconstruction of Parental Genotypes by F1 Hybrids as a Bridge for Rapid Introgression. Genome 2017, 60, 713–719. [Google Scholar] [CrossRef]
- Kundzinsh, A.V. Experiments on Artificial Hybridization of Alder. In Gain in Forest Productivity; Zinatne: Riga, Latvia, 1968; pp. 69–99. [Google Scholar]

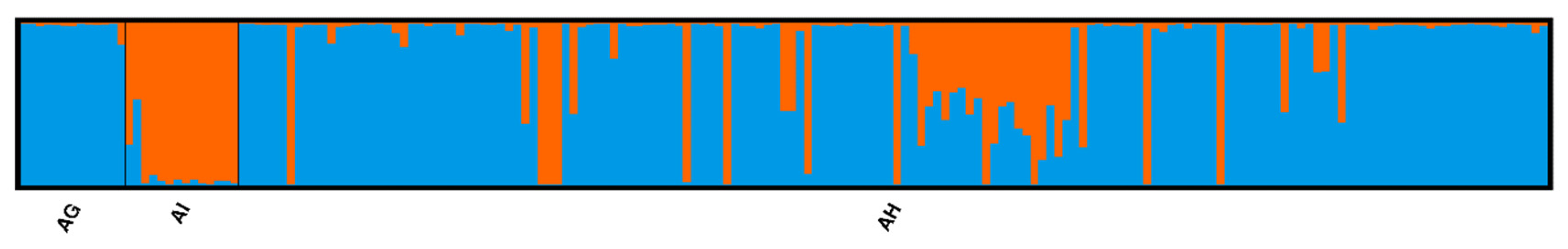
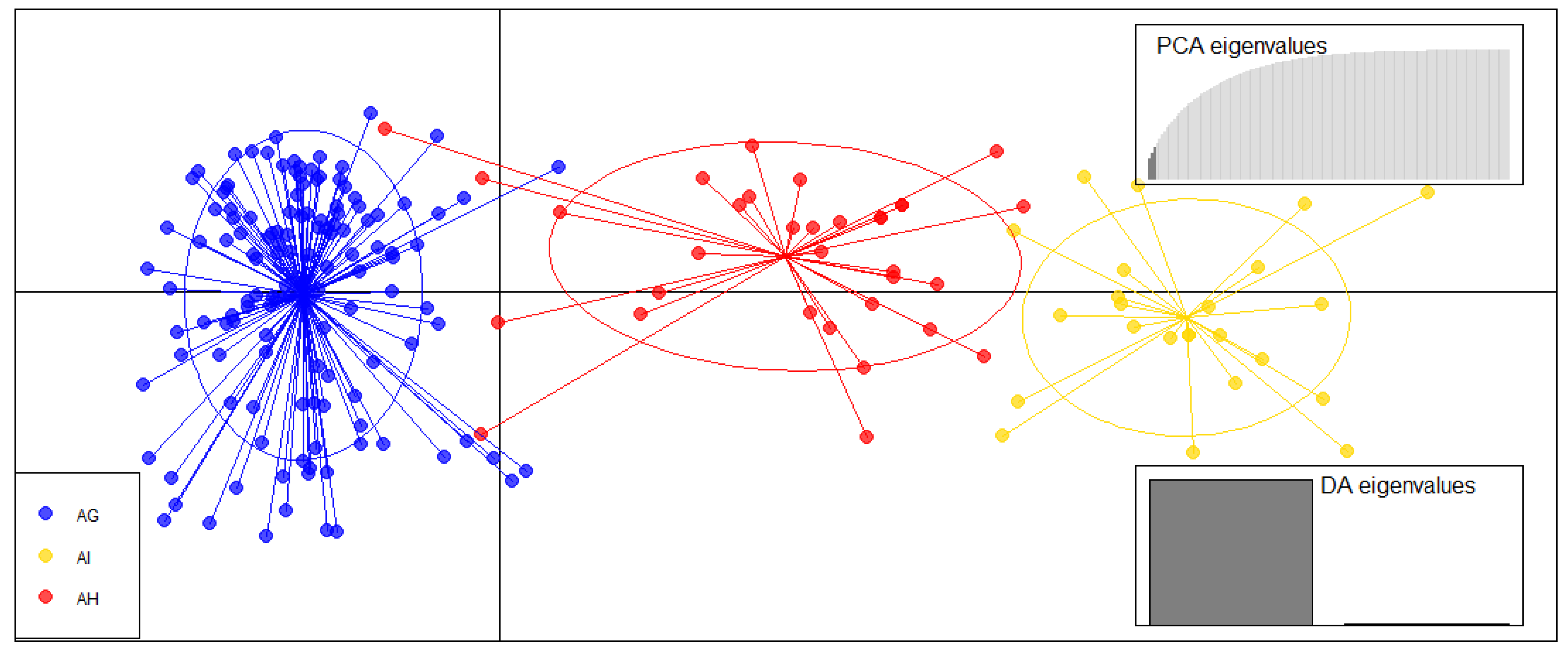
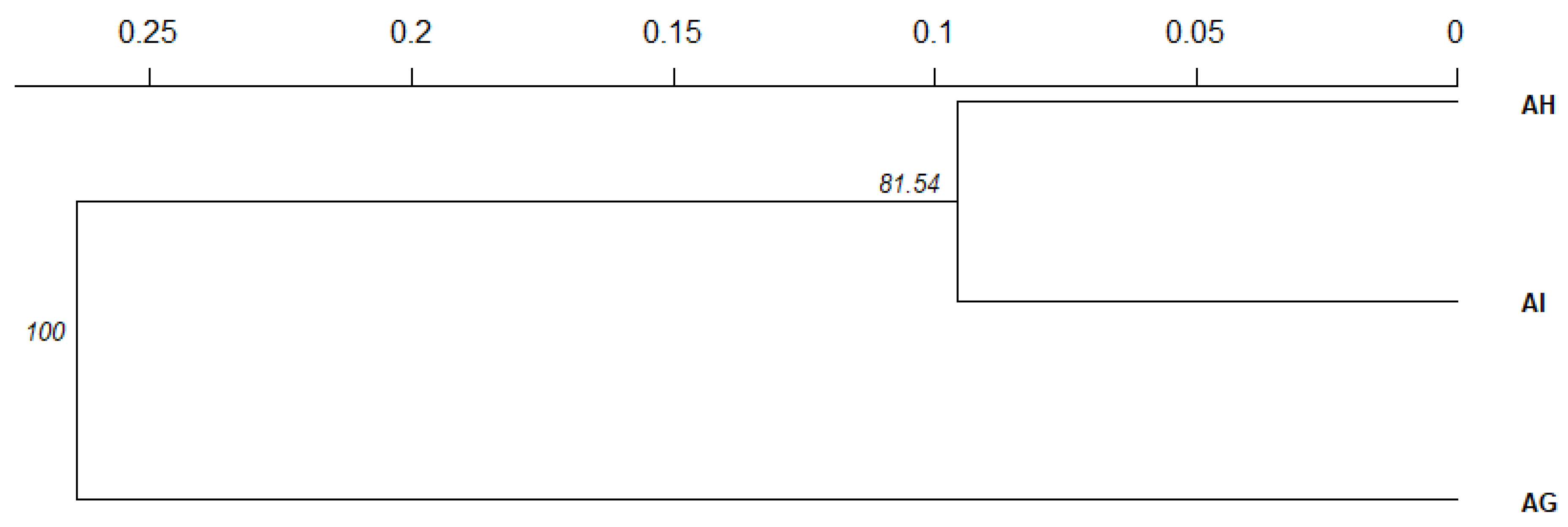


| Speaces Based on Basyan Clustering Analysis | Abbreviations | N1 | Na | Ne | Np | Ar | Ho | uHe | FIS |
|---|---|---|---|---|---|---|---|---|---|
| Alnus glutinosa | AG | 132 | 10.07 | 4.39 | 34.00 | 7.28 | 0.589 | 0.636 | 0.057 |
| A. incana | AI | 24 | 5.07 | 2.96 | 2.00 | 5.03 | 0.498 | 0.586 | 0.197 |
| A. × hybrid | AH | 33 | 8.13 | 4.47 | 3.00 | 7.58 | 0.764 | 0.726 | −0.066 |
| Total mean | 7.76 | 3.94 | 13.0 | 6.63 | 0.617 | 0.649 | 0.063 |
| Morphological Traits 1 | Alnus glutinosa | Alnus incana | Alnus × hybrid | R2 | F | Pr > F |
|---|---|---|---|---|---|---|
| Min–Max Mean ± Standart Deviation (n − 1) | ||||||
| P | 0.00–5.50 0.30 ± 0.59 | 1.00–4.00 3.66 ± 0.74 | 0.00–4.00 1.88 ± 1.16 | 0.719 | 237.570 | <0.0001 |
| N | 5.00–8.83 7.21 ± 0.65 | 8.00–11.80 10.04 ± 1.05 | 6.40–10.20 8.17 ± 0.91 | 0.612 | 146.400 | <0.0001 |
| W | 64.46–159.75 93.21 ± 18.13 | 33.84–67.03 48.22 ± 9.94 | 46.39–151.85 77.40 ± 25.22 | 0.397 | 61.242 | <0.0001 |
| E/B | 0.26–0.67 0.40 ± 0.09 | 0.07–0.34 0.20 ± 0.08 | 0.15–0.59 0.31 ± 0.11 | 0.373 | 55.207 | <0.0001 |
| E | 3.73–58.50 26.55 ± 7.86 | 4.74–20.00 11.47 ± 4.22 | 7.58–49.69 19.20 ± 8.81 | 0.326 | 44.913 | <0.0001 |
| D/A | 0.46–0.70 0.57 ± 0.04 | 0.39–0.58 0.49 ± 0.05 | 0.45–0.61 0.53 ± 0.04 | 0.320 | 43.756 | <0.0001 |
| B/A | 0.66–1.03 0.82 ± 0.07 | 0.62–0.85 0.73 ± 0.06 | 0.67–0.95 0.82 ± 0.07 | 0.134 | 14.404 | <0.0001 |
| D | 4.95–68.20 46.52 ± 8.82 | 23.24–57.30 39.47 ± 7.93 | 25.71–62.27 40.34 ± 7.59 | 0.113 | 11.840 | <0.0001 |
| B | 5.75–112.35 66.78 ± 13.62 | 39.35–75.53 59.16 ± 10.09 | 43.69–87.20 62.43 ± 10.80 | 0.046 | 4.459 | 0.013 |
| I | 2.08–38.88 24.94 ± 6.00 | 14.86–39.02 22.65 ± 5.24 | 10.38–35.90 22.63 ± 5.79 | 0.032 | 3.048 | 0.050 |
| A | 7.83–122.98 81.81 ± 14.96 | 54.69–103.86 81.00 ± 13.76 | 50.30–106.43 76.58 ± 13.21 | 0.018 | 1.717 | 0.182 |
| I/A | 0.05–0.70 0.31 ± 0.07 | 0.19–0.45 0.28 ± 0.06 | 0.12–0.48 0.30 ± 0.08 | 0.016 | 1.495 | 0.227 |
| Morphologic Traits | PC 1 | PC 2 | PC 3 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| R | R2 | tau | R | R2 | tau | R | R2 | tau | |
| W | −0.998 1 | 0.995 | −0.958 | −0.068 | 0.005 | 0.057 | −0.002 | 0.000 | −0.033 |
| E | −0.845 | 0.714 | −0.672 | 0.535 | 0.286 | 0.427 | −0.001 | 0.000 | 0.018 |
| N | 0.502 | 0.252 | 0.386 | 0.044 | 0.002 | −0.057 | −0.741 | 0.549 | −0.509 |
| P | 0.586 | 0.343 | 0.351 | −0.017 | 0.000 | −0.044 | −0.734 | 0.538 | −0.548 |
| D/A | −0.457 | 0.209 | −0.342 | 0.086 | 0.007 | 0.111 | 0.347 | 0.012 | 0.195 |
| E/B | −0.870 | 0.757 | −0.703 | 0.112 | 0.012 | 0.166 | 0.083 | 0.007 | 0.040 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jurkšienė, G.; Tamošaitis, S.; Kavaliauskas, D.; Buchovska, J.; Danusevičius, D.; Baliuckas, V. Identification of Alnus glutinosa L. and A. incana (L.) Moench. Hybrids in Natural Forests Using Nuclear DNA Microsatellite and Morphometric Markers. Forests 2021, 12, 1504. https://doi.org/10.3390/f12111504
Jurkšienė G, Tamošaitis S, Kavaliauskas D, Buchovska J, Danusevičius D, Baliuckas V. Identification of Alnus glutinosa L. and A. incana (L.) Moench. Hybrids in Natural Forests Using Nuclear DNA Microsatellite and Morphometric Markers. Forests. 2021; 12(11):1504. https://doi.org/10.3390/f12111504
Chicago/Turabian StyleJurkšienė, Girmantė, Sigitas Tamošaitis, Darius Kavaliauskas, Jurata Buchovska, Darius Danusevičius, and Virgilijus Baliuckas. 2021. "Identification of Alnus glutinosa L. and A. incana (L.) Moench. Hybrids in Natural Forests Using Nuclear DNA Microsatellite and Morphometric Markers" Forests 12, no. 11: 1504. https://doi.org/10.3390/f12111504
APA StyleJurkšienė, G., Tamošaitis, S., Kavaliauskas, D., Buchovska, J., Danusevičius, D., & Baliuckas, V. (2021). Identification of Alnus glutinosa L. and A. incana (L.) Moench. Hybrids in Natural Forests Using Nuclear DNA Microsatellite and Morphometric Markers. Forests, 12(11), 1504. https://doi.org/10.3390/f12111504







