Smaller and Isolated Grassland Fragments Are Exposed to Stronger Seed and Insect Predation in Habitat Edges
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Region and Sampling Design
2.2. Sentinel Prey
2.3. Seed Predation
2.4. Statistical Analysis
3. Results
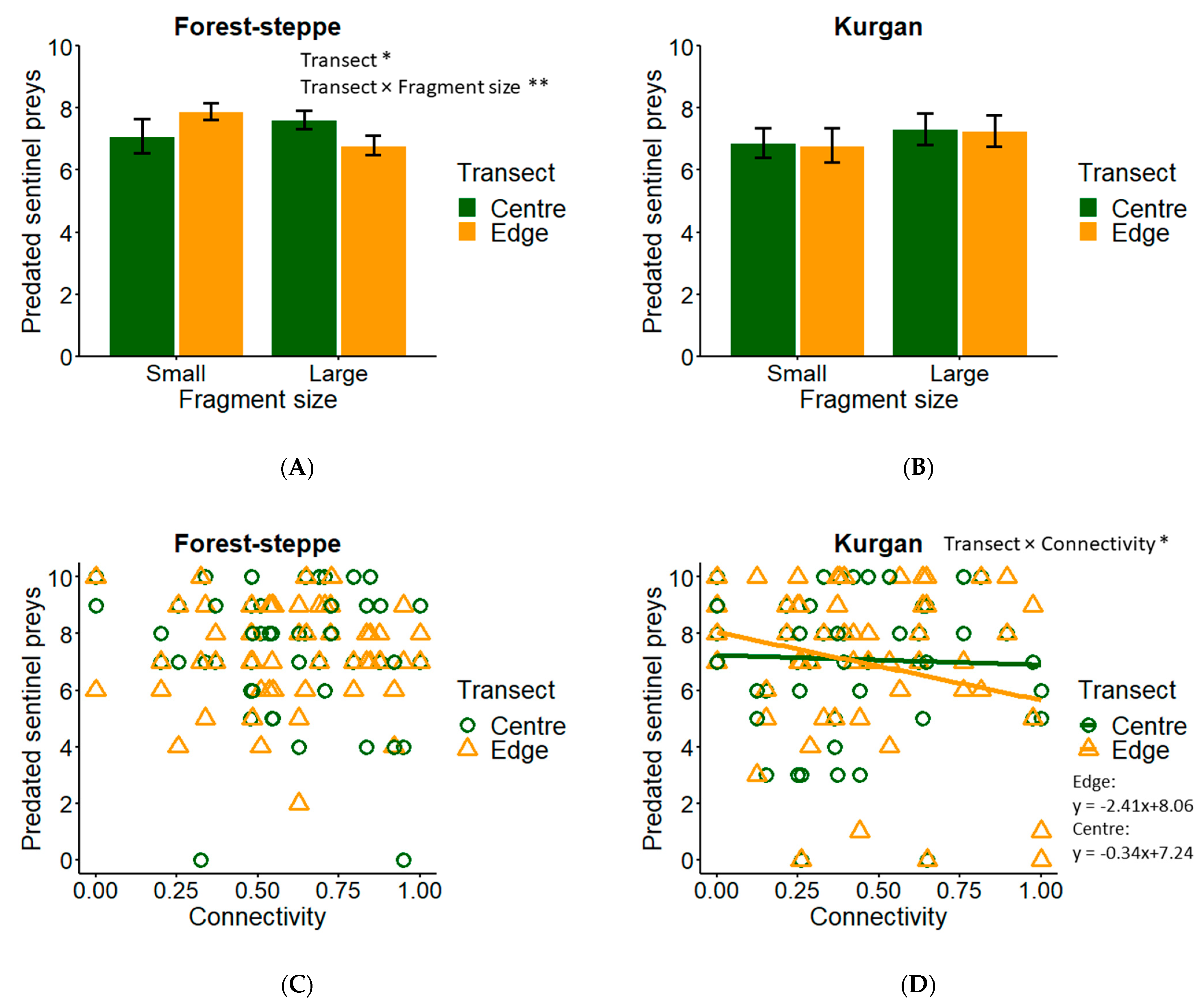
3.1. Sentinel Prey
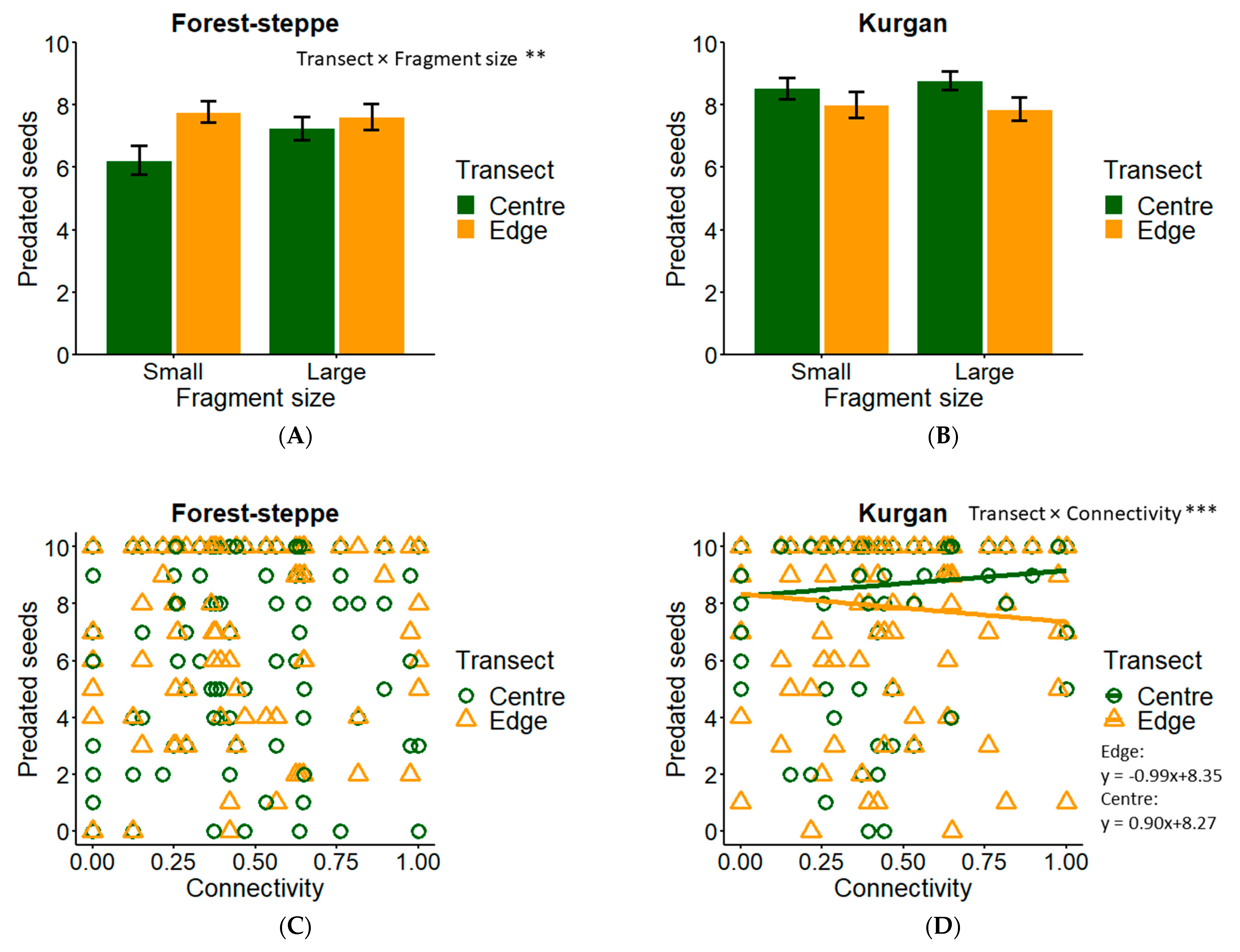
3.2. Seed Predation
4. Discussion
4.1. Predation in Forest-Steppe
4.2. Predation in Kurgan
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cardoso, P.; Barton, P.S.; Birkhofer, K.; Chichorro, F.; Deacon, C.; Fartmann, T.; Fukushima, C.S.; Gaigher, R.; Habel, J.C.; Hallmann, C.A.; et al. Scientists’ warning to humanity on insect extinctions. Biol. Conserv. 2020, 242, 108426. [Google Scholar] [CrossRef]
- Habel, J.C.; Trusch, R.; Schmitt, T.; Ochse, M.; Ulrich, W. Long-term large-scale decline in relative abundances of butterfly and burnet moth species across south-western Germany. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.; Lindenmayer, D.B. Landscape modification and habitat fragmentation: A synthesis. Glob. Ecol. Biogeogr. 2007, 16, 265–280. [Google Scholar] [CrossRef]
- MacArthur, R.H.; Wilson, E.O. The Theory of Island Biogeography; Princeton University Press: Princeton, NJ, USA, 1967. [Google Scholar]
- Laurance, W.F. Beyond island biogeography theory: Understanding habitat fragmentation in the real world. In The Theory of Island Biogeography Revisited; Losos, J.B., Ricklefs, R.E., Eds.; Princeton University Press: Princeton, NJ, USA, 2009; pp. 214–236. [Google Scholar]
- Ord, T.J.; Emblen, J.; Hagman, M.; Shofner, R.; Unruh, S. Manipulation of habitat isolation and area implicates deterministic factors and limited neutrality in community assembly. Ecol. Evol. 2017, 7, 5845–5860. [Google Scholar] [CrossRef] [PubMed]
- Baz, A.; Garcia-Boyero, A. The effects of forest fragmentation on butterfly communities in central Spain. J. Biogeogr. 1995, 129–140. [Google Scholar] [CrossRef]
- Franzén, M.; Schweiger, O.; Betzholtz, P.E. Species-area relationships are controlled by species traits. PLoS ONE 2012, 7, e37359. [Google Scholar] [CrossRef]
- Hanski, I. Metapopulation Ecology; Oxford University Press Inc.: New York, NY, USA, 1999. [Google Scholar]
- Murcia, C. Edge effects in fragmented forests: Implications for conservation. Trends Ecol. Evol. 1995, 10, 58–62. [Google Scholar] [CrossRef]
- Ries, L.; Sisk, T.D. A predictive model of edge effects. Ecology 2004, 85, 2917–2926. [Google Scholar] [CrossRef]
- Rand, T.A.; Tylianakis, J.M.; Tscharntke, T. Spillover edge effects: The dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats. Ecol. Lett. 2006, 9, 603–614. [Google Scholar] [CrossRef]
- Boetzl, F.A.; Schneider, G.; Krauss, J. Asymmetric carabid beetle spillover between calcareous grasslands and coniferous forests. J. Insect Conserv. 2016, 20, 49–57. [Google Scholar] [CrossRef]
- Bianchi, F.J.; Booij, C.J.H.; Tscharntke, T. Sustainable pest regulation in agricultural landscapes: A review on landscape composition, biodiversity and natural pest control. Proc. R. Soc. B Biol. Sci. 2006, 273, 1715–1727. [Google Scholar] [CrossRef] [PubMed]
- Batáry, P.; Báldi, A.; Ekroos, J.; Gallé, R.; Grass, I.; Tscharntke, T. Biologia Futura: Landscape perspectives on farmland biodiversity conservation. Biol. Futur. 2020, 1–10. [Google Scholar] [CrossRef]
- Badenhausser, I.; Gross, N.; Mornet, V.; Roncoroni, M.; Saintilan, A.; Rusch, A. Increasing amount and quality of green infrastructures at different scales promotes biological control in agricultural landscapes. Agric. Ecosyst. Environ. 2020, 290, 106735. [Google Scholar] [CrossRef]
- Blitzer, E.J.; Dormann, C.F.; Holzschuh, A.; Klein, A.M.; Rand, T.A.; Tscharntke, T. Spillover of functionally important organisms between managed and natural habitats. Agric. Ecosyst. Environ. 2012, 146, 34–43. [Google Scholar] [CrossRef]
- Schneider, G.; Krauss, J.; Boetzl, F.A.; Fritze, M.A.; Steffan-Dewenter, I. Spillover from adjacent crop and forest habitats shapes carabid beetle assemblages in fragmented semi-natural grasslands. Oecologia 2016, 182, 1141–1150. [Google Scholar] [CrossRef]
- Madeira, F.; Tscharntke, T.; Elek, Z.; Kormann, U.G.; Pons, X.; Rösch, V.; Samu, F.; Scherber, C.; Batáry, P. Spillover of arthropods from cropland to protected calcareous grassland–the neighbouring habitat matters. Agric. Ecosyst. Environ. 2016, 235, 127–133. [Google Scholar] [CrossRef]
- Fahrig, L.; Arroyo-Rodríguez, V.; Bennett, J.R.; Boucher-Lalonde, V.; Cazetta, E.; Currie, D.J.; Eigenbrod, F.; Ford, A.T.; Harrison, S.P.; Jaeger, J.A.; et al. Is habitat fragmentation bad for biodiversity? Biol. Conserv. 2019, 230, 179–186. [Google Scholar] [CrossRef]
- Riggi, L.G.; Berggren, Å. Small field islands systems include a large proportion of the regional orthopteran species pool in arable landscapes. J. Insect Conserv. 2020, 24, 695–703. [Google Scholar] [CrossRef]
- Tscharntke, T.; Steffan-Dewenter, I.; Kruess, A.; Thies, C. Contribution of small habitat fragments to conservation of insect communities of grassland–cropland landscapes. Ecol. Appl. 2002, 12, 354–363. [Google Scholar] [CrossRef]
- Rösch, V.; Tscharntke, T.; Scherber, C.; Batáry, P. Biodiversity conservation across taxa and landscapes requires many small as well as single large habitat fragments. Oecologia 2015, 179, 209–222. [Google Scholar] [CrossRef]
- Zabel, J.; Tscharntke, T. Does fragmentation of Urtica habitats affect phytophagous and predatory insects differentially? Oecologia 1998, 116, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Isbell, F.; Cowles, J.M. Biodiversity and ecosystem functioning. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 471–493. [Google Scholar] [CrossRef]
- Rossetti, M.R.; Tscharntke, T.; Aguilar, R.; Batáry, P. Responses of insect herbivores and herbivory to habitat fragmentation: A hierarchical meta-analysis. Ecol. Lett. 2017, 20, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Brudvig, L.A.; Damschen, E.I.; Haddad, N.M.; Levey, D.J.; Tewksbury, J.J. The influence of habitat fragmentation on multiple plant–animal interactions and plant reproduction. Ecology 2015, 96, 2669–2678. [Google Scholar] [CrossRef]
- Bagchi, R.; Brown, L.M.; Elphick, C.S.; Wagner, D.L.; Singer, M.S. Anthropogenic fragmentation of landscapes: Mechanisms for eroding the specificity of plant–herbivore interactions. Oecologia 2018, 187, 521–533. [Google Scholar] [CrossRef]
- Liu, J.; Wilson, M.; Hu, G.; Liu, J.; Wu, J.; Yu, M. How does habitat fragmentation affect the biodiversity and ecosystem functioning relationship? Landsc. Ecol. 2018, 33, 341–352. [Google Scholar] [CrossRef]
- Fleury, M.; Galetti, M. Forest fragment size and microhabitat effects on palm seed predation. Biol. Conserv. 2006, 131, 1–13. [Google Scholar] [CrossRef]
- González-Fernández, J.; De la Peña, F.; Hormaza, J.; Boyero, J.; Vela, J.; Wong, E.; Trigo, M.M.; Montserrat, M. Alternative food improves the combined effect of an omnivore and a predator on biological pest control. A case study in avocado orchards. Bull. Entomol. Res. 2009, 99, 433–444. [Google Scholar] [CrossRef]
- Garcia, D.; Chacoff, N.P. Scale-dependent effects of habitat fragmentation on hawthorn pollination, frugivory, and seed predation. Conserv. Biol. 2007, 21, 400–411. [Google Scholar] [CrossRef]
- Tong, X.; Zhang, Y.X.; Wang, R.; Inbar, M.; Chen, X.Y. Habitat fragmentation alters predator satiation of acorns. J. Plant Ecol. 2017, 10, 67–73. [Google Scholar] [CrossRef]
- Elzinga, J.A.; Turin, H.; Van Damme, J.M.; Biere, A. Plant population size and isolation affect herbivory of Silene latifolia by the specialist herbivore Hadena bicruris and parasitism of the herbivore by parasitoids. Oecologia 2005, 144, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Eötvös, C.B.; Magura, T.; Lövei, G.L. A meta-analysis indicates reduced predation pressure with increasing urbanization. Landsc. Urban Plan. 2018, 180, 54–59. [Google Scholar] [CrossRef]
- Auld, T.D.; Denham, A.J. The role of ants and mammals in dispersal and post-dispersal seed predation of the shrubs Grevillea (Proteaceae). Plant Ecol. 1999, 144, 201–213. [Google Scholar] [CrossRef]
- Rusch, A.; Birkhofer, K.; Bommarco, R.; Smith, H.G.; Ekbom, B. Predator body sizes and habitat preferences predict predation rates in an agroecosystem. Basic Appl. Ecol. 2015, 16, 250–259. [Google Scholar] [CrossRef]
- Lövei, G.L.; Ferrante, M. A review of the sentinel prey method as a way of quantifying invertebrate predation under field conditions. Insect Sci. 2017, 24, 528–542. [Google Scholar] [CrossRef]
- Imboma, T.S.; Gao, D.P.; You, M.S.; You, S.; Lövei, G.L. Predation Pressure in Tea (Camellia sinensis) Plantations in Southeastern China Measured by the Sentinel Prey Method. Insects 2020, 11, 212. [Google Scholar] [CrossRef]
- Deák, B.; Valkó, O.; Török, P.; Tóthmérész, B. Factors threatening grassland specialist plants—A multi-proxy study on the vegetation of isolated grasslands. Biol. Conserv. 2016, 204, 255–262. [Google Scholar] [CrossRef]
- Erdős, L.; Tölgyesi, C.; Horzse, M.; Tolnay, D.; Hurton, Á.; Schulcz, N.; Körmöczi, L.; Lengyel, A.; Bátori, Z. Habitat complexity of the Pannonian forest-steppe zone and its nature conservation implications. Ecol. Complex. 2014, 17, 107–118. [Google Scholar] [CrossRef]
- Deák, B.; Tóthmérész, B.; Valkó, O.; Sudnik-Wójcikowska, B.; Bragina, T.M.; Moysiyenko, I.I.; Bragina, T.M.; Apostolova, I.; Dembicz, I.; Bykov, N.I. Cultural monuments and nature conservation: The role of kurgans in maintaining steppe vegetation. Biodivers. Conserv. 2016, 25, 2473–2490. [Google Scholar] [CrossRef]
- Deák, B.; Tóth, C.A.; Bede, Á.; Apostolova, I.; Bragina, T.M.; Báthori, F.; Bán, M. Eurasian Kurgan Database–a citizen science tool for conserving grasslands on historical sites. Hacquetia 2019, 18, 179–187. [Google Scholar] [CrossRef]
- Fekete, G.; Molnár, Z.; Kun, A.; Botta-Dukát, Z. On the structure of the Pannonian forest-steppe: Grasslands on sand. Acta Zool. Acad. Sci. Hung. 2002, 48 (Suppl. 1), 137–150. [Google Scholar]
- Bölöni, J.; Molnár, Z.; Kun, A.; Biró, M. Általános Nemzeti Élőhely-Osztályozási Rendszer (Á-NÉR 2007); MTA ÖBKI: Vácrátót, Hungary, 2007; p. 184. [Google Scholar]
- Hanski, I.; Ovaskainen, O. The metapopulation capacity of a fragmented landscape. Nature 2000, 404, 755–758. [Google Scholar] [CrossRef] [PubMed]
- Quantum GIS Development Team. Quantum GIS Geographic Information System. Open Source Geospatial Foundation Project. 2019. Available online: http://qgis.osgeo.org (accessed on 10 March 2019).
- Kormann, U.; Rösch, V.; Batáry, P.; Tscharntke, T.; Orci, K.M.; Samu, F.; Scherber, C. Local and landscape management drive trait-mediated biodiversity of nine taxa on small grassland fragments. Divers. Distrib. 2015, 21, 1204–1217. [Google Scholar] [CrossRef]
- Howe, A.; Lövei, G.L.; Nachman, G. Dummy caterpillars as a simple method to assess predation rates on invertebrates in a tropical agroecosystem. Entomol. Exp. Appl. 2009, 131, 325–329. [Google Scholar] [CrossRef]
- Purger, J.J.; Kurucz, K.; Tóth, Á.; Batáry, P. Coating plasticine eggs can eliminate the overestimation of predation on artificial ground nests. Bird Study 2012, 59, 350–352. [Google Scholar] [CrossRef]
- Low, P.A.; Sam, K.; McArthur, C.; Posa, M.R.C.; Hochuli, D.F. Determining predator identity from attack marks left in model caterpillars: Guidelines for best practice. Entomol. Exp. Appl. 2014, 152, 120–126. [Google Scholar] [CrossRef]
- Brown, J.H.; Grover, J.J.; Davidson, D.W.; Lieberman, G.A. A preliminary study of seed predation in desert and montane habitats. Ecology 1975, 56, 987–992. [Google Scholar] [CrossRef]
- Linabury, M.C.; Turley, N.E.; Brudvig, L.A. Insects remove more seeds mammals in first-year prairie restorations. Restor. Ecol. 2019, 27, 1300–1306. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B. Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Richards, S.A. Dealing with overdispersed count data in applied ecology. J. Appl. Ecol. 2008, 45, 218–227. [Google Scholar] [CrossRef]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Barton, K. MuMIn: R Functions for Model Selection and Model Averaging. R Package Version 0.12.0. 2009. Available online: http://r-forge.r-project.org/projects/mumin (accessed on 2 June 2020).
- Laurance, W.F.; Yensen, E. Predicting the impacts of edge effects in fragmented habitats. Biol. Conserv. 1991, 55, 77–92. [Google Scholar] [CrossRef]
- Orrock, J.L.; Damschen, E.I. Corridors cause differential seed predation. Ecol. Appl. 2005, 15, 793–798. [Google Scholar] [CrossRef]
- Murphy, S.M.; Battocletti, A.H.; Tinghitella, R.M.; Wimp, G.M.; Ries, L. Complex community and evolutionary responses to habitat fragmentation and habitat edges: What can we learn from insect science? Curr. Opin. Insect Sci. 2016, 14, 61–65. [Google Scholar] [CrossRef]
- Wimp, G.M.; Ries, L.; Lewis, D.; Murphy, S.M. Habitat edge responses of generalist predators are predicted by prey and structural resources. Ecology 2019, 100, e02662. [Google Scholar] [CrossRef]
- Gallé, R.; Geppert, C.; Földesi, R.; Tscharntke, T.; Batáry, P. Arthropod functional traits shaped by landscape-scale field size, local agri-environment schemes and edge effects. Basic Appl. Ecol. 2020, 48, 102–111. [Google Scholar] [CrossRef]
- Martinson, H.M.; Fagan, W.F. Trophic disruption: A meta-analysis of how habitat fragmentation affects resource consumption in terrestrial arthropod systems. Ecol. Lett. 2014, 17, 1178–1189. [Google Scholar] [CrossRef]
- Wimp, G.M.; Murphy, S.M.; Lewis, D.; Ries, L. Do edge responses cascade up or down a multi-trophic food web? Ecol. Lett. 2011, 14, 863–870. [Google Scholar] [CrossRef]
- Ries, L.; Fagan, W.F. Habitat edges as a potential ecological trap for an insect predator. Ecol. Entomol. 2003, 28, 567–572. [Google Scholar] [CrossRef]
- Thomson, L.J.; Hoffmann, A.A. Spatial scale of benefits from adjacent woody vegetation on natural enemies within vineyards. Biol. Control 2013, 64, 57–65. [Google Scholar] [CrossRef]
- Craig, M.T.; Orrock, J.L.; Brudvig, L.A. Edge-mediated patterns of seed removal in experimentally connected and fragmented landscapes. Landsc. Ecol. 2011, 26, 1373–1381. [Google Scholar] [CrossRef]
- McCoy, M.W.; Barfield, M.; Holt, R.D. Predator shadows: Complex life histories as generators of spatially patterned indirect interactions across ecosystems. Oikos 2009, 118, 87–100. [Google Scholar] [CrossRef]
- Tölgyesi, C.; Császár, P.; Torma, A.; Török, P.; Bátori, Z.; Gallé, R. Think twice before using narrow buffers: Attenuating mowing-induced arthropod spillover at forest–grassland edges. Agric. Ecosyst. Environ. 2018, 255, 37–44. [Google Scholar] [CrossRef]
- Schneider, G.; Krauss, J.; Steffan-Dewenter, I. Predation rates on semi-natural grasslands depend on adjacent habitat type. Basic Appl. Ecol. 2013, 14, 614–621. [Google Scholar] [CrossRef]
- Rand, T.A.; Louda, S.M. Spillover of agriculturally subsidized predators as a potential threat to native insect herbivores in fragmented landscapes. Conserv. Biol. 2006, 20, 1720–1729. [Google Scholar] [CrossRef] [PubMed]
- Rand, T.A.; Tscharntke, T. Contrasting effects of natural habitat loss on generalist and specialist aphid natural enemies. Oikos 2007, 116, 1353–1362. [Google Scholar] [CrossRef]
- Labruyere, S.; Petit, S.; Ricci, B. Annual variation of oilseed rape habitat quality and role of grassy field margins for seed eating carabids in arable mosaics. Agric. For. Entomol. 2018, 20, 234–245. [Google Scholar] [CrossRef]

| Model a | Variable | Relative Importance (%) b | Multimodel Estimate ± 95% c | |
|---|---|---|---|---|
| Insect predation (FS) | Fragment size (L/S) | 100 | −0.215 | ±0.538 |
| Transect (C/E) | 100 | −0.449 | ±0.427 * | |
| Fragment size × transect | 100 | 0.895 | ±0.538 ** | |
| Connectivity | 36 | 0.050 | ±0.928 | |
| Connectivity × transect | 8 | 0.300 | ±1.140 | |
| Connectivity × fragment size | 7 | 0.251 | ±1.689 | |
| Insect predation (KU) | Connectivity | 67 | −0.418 | ±1.887 |
| Transect (C/E) | 62 | 0.405 | ±0.651 | |
| Connectivity × Transect | 49 | −1.303 | ±1.014 * | |
| Fragment size (L/S) | 33 | −0.258 | ±1.087 | |
| Fragment size × transect | 4 | −0.077 | ±0.573 | |
| Connectivity × Fragment size | 4 | −0.425 | ±3.402 | |
| Seed predation (FS) | Fragment size (L/S) | 100 | −0.521 | ±0.651 |
| Transect (C/E) | 100 | 0.208 | ±0.313 | |
| Fragment size × Transect | 100 | 0.635 | ±0.387 ** | |
| Connectivity | 38 | 0.071 | ±1.203 | |
| Connectivity × transect | 8 | 0.164 | ±0.827 | |
| Connectivity × fragment size | 8 | −0.380 | ±2.125 | |
| Seed predation (KU) | Connectivity | 100 | 1.281 | ±1.589 |
| Transect (C/E) | 100 | 0.098 | ±0.439 | |
| Connectivity × transect | 100 | −1.731 | ±0.872 *** | |
| Fragment size (L/S) | 42 | 0.064 | ±1.037 | |
| Fragment size × transect | 15 | 0.227 | ±0.469 | |
| Connectivity × Fragment size | 11 | −0.547 | ±2.797 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuli-Révész, K.; Korányi, D.; Lakatos, T.; Szabó, Á.R.; Batáry, P.; Gallé, R. Smaller and Isolated Grassland Fragments Are Exposed to Stronger Seed and Insect Predation in Habitat Edges. Forests 2021, 12, 54. https://doi.org/10.3390/f12010054
Kuli-Révész K, Korányi D, Lakatos T, Szabó ÁR, Batáry P, Gallé R. Smaller and Isolated Grassland Fragments Are Exposed to Stronger Seed and Insect Predation in Habitat Edges. Forests. 2021; 12(1):54. https://doi.org/10.3390/f12010054
Chicago/Turabian StyleKuli-Révész, Kitti, Dávid Korányi, Tamás Lakatos, Ágota Réka Szabó, Péter Batáry, and Róbert Gallé. 2021. "Smaller and Isolated Grassland Fragments Are Exposed to Stronger Seed and Insect Predation in Habitat Edges" Forests 12, no. 1: 54. https://doi.org/10.3390/f12010054
APA StyleKuli-Révész, K., Korányi, D., Lakatos, T., Szabó, Á. R., Batáry, P., & Gallé, R. (2021). Smaller and Isolated Grassland Fragments Are Exposed to Stronger Seed and Insect Predation in Habitat Edges. Forests, 12(1), 54. https://doi.org/10.3390/f12010054




