Shifts in Growth Responses to Climate and Exceeded Drought-Vulnerability Thresholds Characterize Dieback in Two Mediterranean Deciduous Oaks
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites, Species, and Climate
2.2. Dendrochronological Methods
2.3. Climate-Growth Relationships and Statistical Analyses
2.4. VS-Lite Forward Growth Model
3. Results
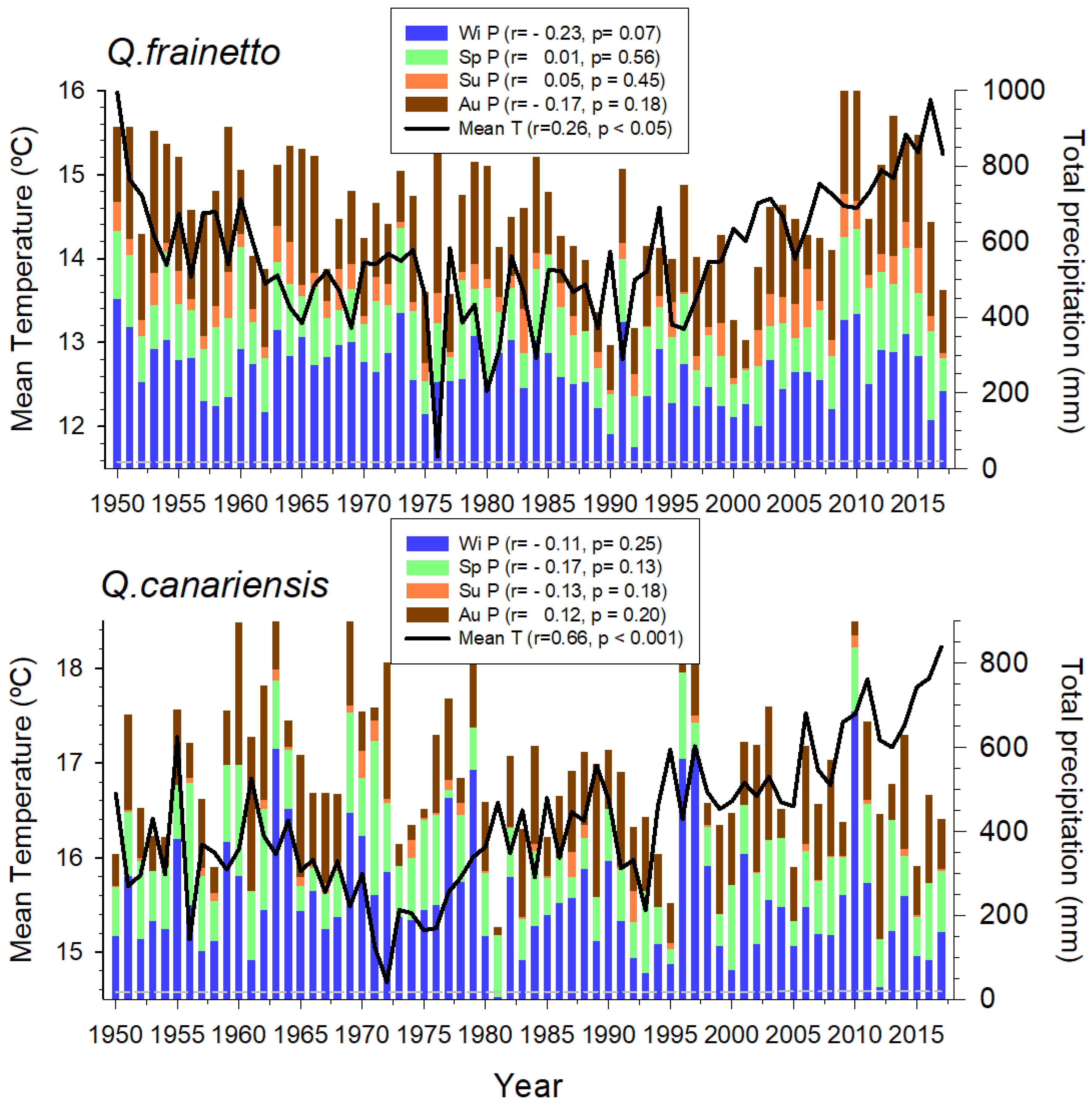
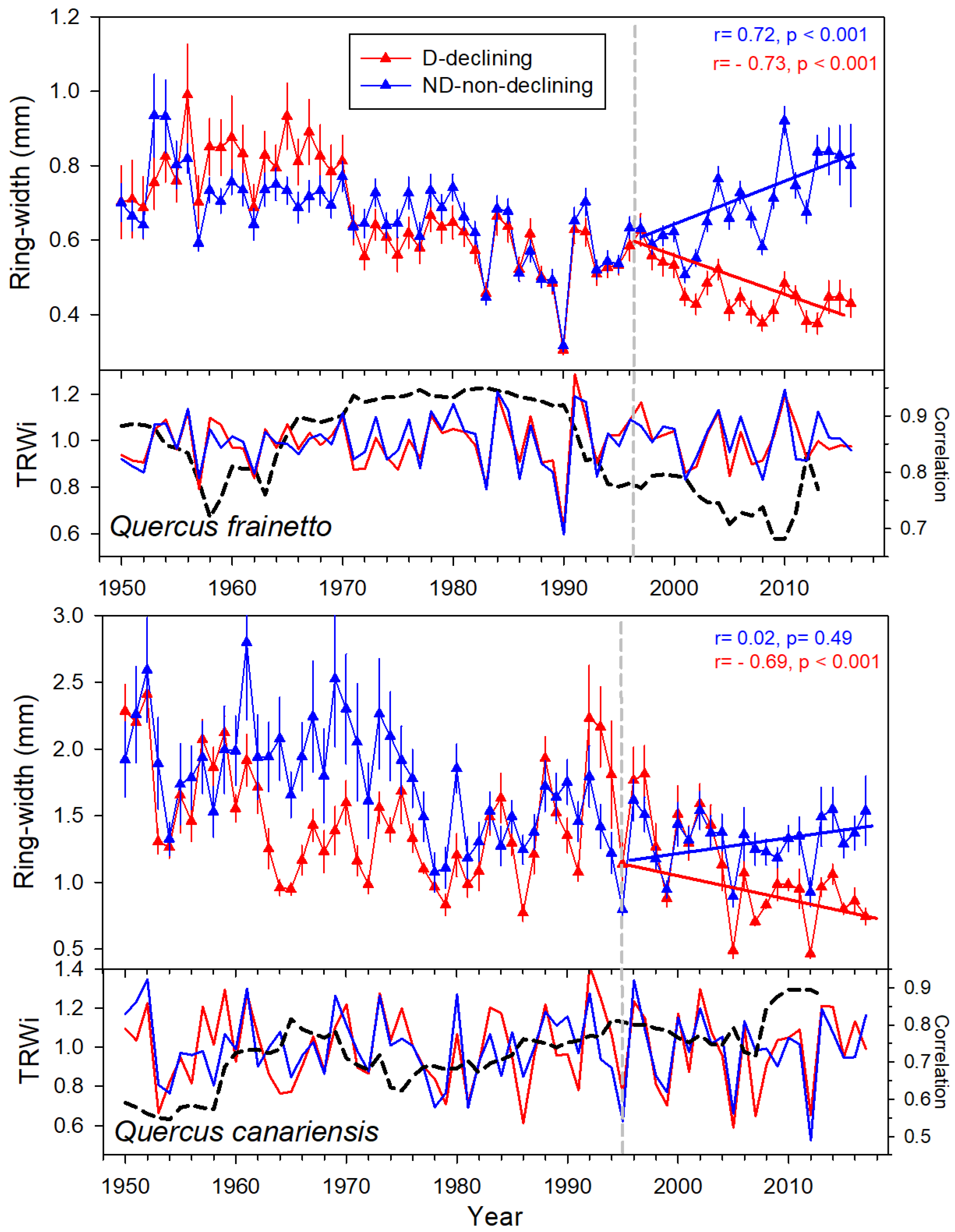
3.1. Climate Trends, Size, and Growth Patterns
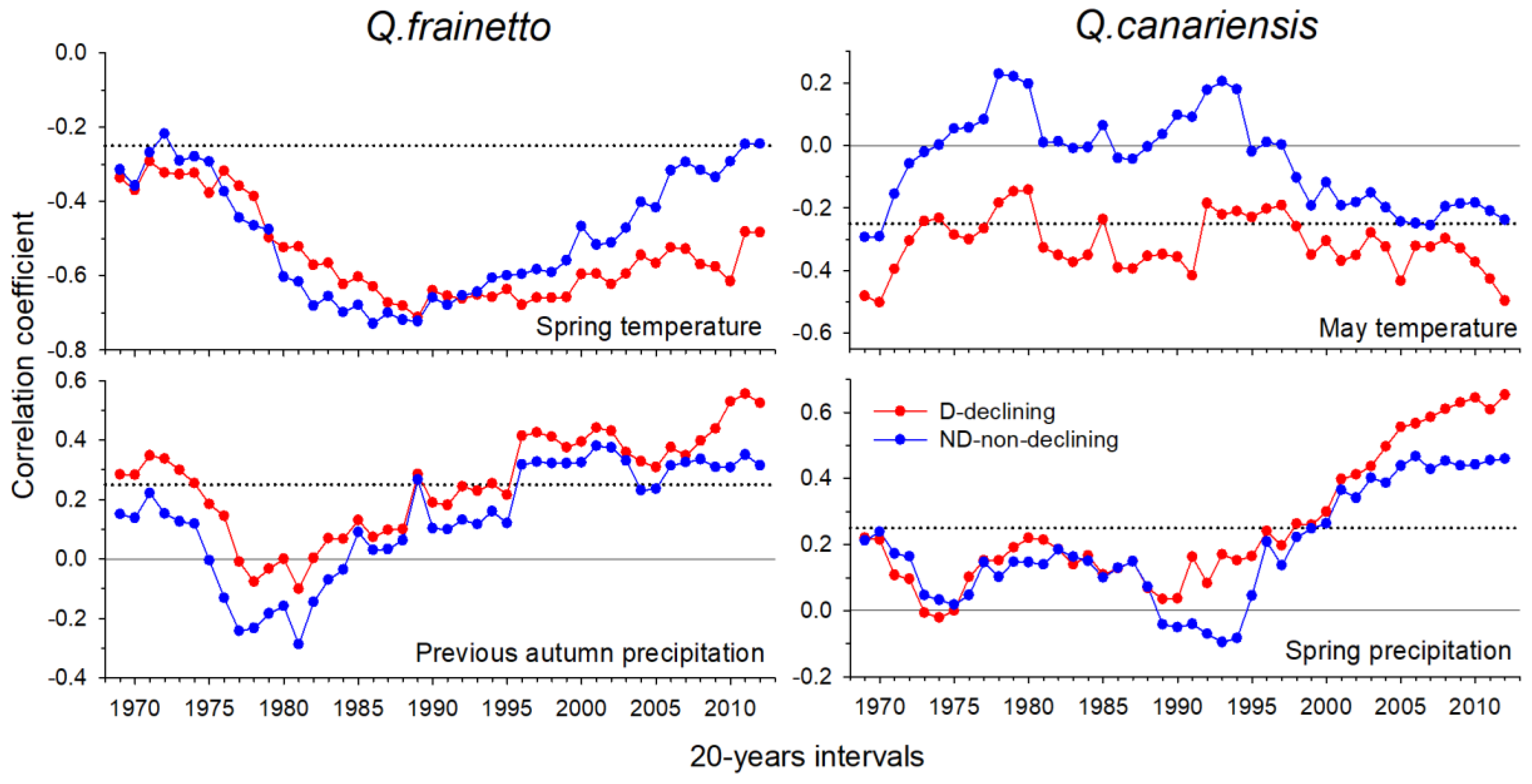
3.2. Climate-Growth Associations
3.3. Forward Growth Modeling for Oaks Dieback
4. Discussion
4.1. Climate Variability and Growth Trends
4.2. Warmer and Drier Conditions during the Early Growing Season Reduce Growth in D Trees
4.3. Changes in the Climatic Thresholds Leading to Forest Dieback
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sánchez-Salguero, R.; Navarro-Cerillo, R.M.; Camarero, J.J.; Fernández-Cancio, A. Selective drought-induced decline of pine species in southeastern Spain. Clim. Chang. 2012, 113, 767–785. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Sangüesa-Barreda, G.; Oliva, J.; Vicente-Serrano, S.M. To die or not to die: Early-warning signals of dieback in response to a severe drought. J. Ecol. 2015, 103, 44–57. [Google Scholar] [CrossRef]
- Pedersen, B.S. The role of stress in the mortality of midwestern oaks as indicated by growth prior to death. Ecology 1998, 79, 79–93. [Google Scholar] [CrossRef]
- Andersson, M.; Milberg, P.; Bergman, K.-O. Low pre-death growth rates of oak (Quercus robur L.)—Is oak death a long-term process induced by dry years? Ann. For. Sci. 2011, 68, 159–168. [Google Scholar] [CrossRef]
- Colangelo, M.; Camarero, J.J.; Ripullone, F.; Gazol, A.; Sánchez-Salguero, R.; Oliva, J.; Redondo, M.A. Drought decreases growth and increases mortality of coexisting native and introduced tree species in a temperate floodplain forest. Forests 2018, 9, 205. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolstroma, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.T.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manage. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Kane, J.; Anderegg, L.D.L. Consequences of widespread tree mortality triggered by drought and temperature stress. Nat. Clim. Chang. 2013, 3, 30–36. [Google Scholar] [CrossRef]
- IPCC. Climate Change and Land: An IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems. 2019; in press. [Google Scholar]
- Peñuelas, J.; Lloret, F.; Montoya, R. Severe drought effects on Mediterranean woody flora in Spain. For. Sci. 2001, 47, 214–218. [Google Scholar]
- Lloret, F.; Siscart, D.; Dalmases, C. Canopy recovery after drought dieback in holm-oak Mediterranean forests of Catalonia (NE Spain). Glob. Chang. Biol. 2004, 10, 2092–2099. [Google Scholar] [CrossRef]
- Di Filippo, A.; Alessandrini, A.; Biondi, F.; Blasi, S.; Portoghesi, L.; Piovesan, G. Climate change and oak growth decline: Dendroecology and stand productivity of a Turkey oak (Quercus cerris L.) old stored coppice in Central Italy. Ann. For. Sci. 2010, 67, 706. [Google Scholar] [CrossRef]
- Linares, J.C.; Camarero, J.J. From pattern to process: Linking intrinsic water-use efficiency to drought-induced forest decline. Glob. Chang. Biol. 2012, 18, 1000–1015. [Google Scholar] [CrossRef]
- Colangelo, M.; Camarero, J.J.; Battipaglia, G.; Borghetti, M.; De Micco, V.; Gentilesca, T.; Ripullone, F. A multi-proxy assessment of dieback causes in a Mediterranean oak species. Tree Physiol. 2017, 37, 617–631. [Google Scholar] [CrossRef]
- Colangelo, M.; Camarero, J.J.; Borghetti, M.; Gazol, A.; Ripullone, F. Size matters a lot: Drought-affected Italian oaks are smaller and show lower growth prior to tree death. Front. Plant Sci. 2017, 8, 135. [Google Scholar] [CrossRef]
- Sangüesa-Barreda, G.; Camarero, J.J.; Oliva, J.; Montes, F.; Gazol, A. Past logging, drought and pathogens interact and contribute to forest dieback. Agric. For. Meteorol. 2015, 208, 85–94. [Google Scholar] [CrossRef]
- Barbero, M.; Loisel, R.; Quèzel, P. Biogeography, ecology and history of Mediterranean Quercus ilex ecosystems. Vegetatio 1992, 99–100, 19–34. [Google Scholar] [CrossRef]
- Gentilesca, T.; Camarero, J.J.; Colangelo, M.; Nolè, A.; Ripullone, F. Drought-induced oak decline in the western Mediterranean region: An overview on current evidences, mechanisms and management options to improve forest resilience. iFor. Biogeosci. For. 2017, 10, 796–806. [Google Scholar] [CrossRef]
- Adams, H.D.; Zeppel, M.J.; Anderegg, W.R.; Hartmann, H.; Landhäusser, S.M.; Tissue, D.T.; Huxman, T.E.; Hudson, P.J.; Franz, T.E.; Allen, C.D.; et al. A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nat. Ecol. Evol. 2016, 1, 1285–1291. [Google Scholar] [CrossRef]
- Corcuera, L.; Camarero, J.J.; Gil-Pelegrín, E. Effects of a severe drought on growth and wood-anatomical properties of Quercus faginea. IAWA J. 2004, 25, 185–204. [Google Scholar] [CrossRef]
- Corcuera, L.; Camarero, J.J.; Gil-Pelegrín, E. Effects of a severe drought on Quercus ilex radial growth and xylem anatomy. Trees 2004, 18, 83–92. [Google Scholar]
- Montserrat-Martí, G.; Camarero, J.J.; Palacio, S.; Pérez-Rontomé, C.; Milla, R.; Albuixech, J.; Maestro, M. Summer-drought constrains the phenology and growth of two co-existing Mediterranean oaks with contrasting leaf habit: Implications for their persistence and reproduction. Trees 2009, 23, 787–799. [Google Scholar] [CrossRef]
- McDowell, N.; Williams, A.; Xu, C.; Pockman, W.T.; Dickman, L.T.; Sevanto, S.; Pangle, R.; Limousin, J.; Plaut, J.; Mackay, D.S.; et al. Multi-scale predictions of massive conifer mortality due to chronic temperature rise. Nat. Clim. Chang. 2016, 6, 295–300. [Google Scholar] [CrossRef]
- Greenwood, S.; Ruiz-Benito, P.; Martınez-Vilalta, J.; Lloret, F.; Kitzberger, T.; Allen, C.D.; Kraft, N.J. Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area. Ecol. Lett. 2017, 20, 539–553. [Google Scholar] [CrossRef]
- Tessier, L.; Nola, P.; Serre-Bachet, F. Deciduous Quercus in the Mediterranean region—Tree-ring/climate relationships. New Phytol. 1994, 126, 355–367. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Fonti, P.; Cherubini, P.; Martín-Benito, D.; Chaar, H.; Cañellas, I. Xylem hydraulic adjustment and growth response of Quercus canariensis Willd. to climatic variability. Tree Physiol. 2012, 32, 401–403. [Google Scholar] [CrossRef]
- Cailleret, M.; Jansen, S.; Robert, E.M.R.; Desoto, L.; Aakala, T.; Antos, J.A.; Beikircher, B.; Bigler, C.; Bugmann, H.; Caccianiga, M.; et al. A synthesis of radial growth patterns preceding tree mortality. Glob. Chang. Biol. 2017, 23, 1675–1690. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J. Greater sensitivity to hotter droughts underlies juniper dieback and mortality in Mediterranean shrublands. Sci. Total Environ. 2020. [Google Scholar] [CrossRef]
- Vaganov, E.A.; Hughes, M.K.; Shashkin, A.V. Growth Dynamics of Conifer Tree Rings; Springer: Berlin, Germany, 2006; p. 358. ISBN 978-3-540-26086-8. [Google Scholar]
- Tolwinski-Ward, S.E.; Evans, M.N.; Hughes, M.K.; Anchukaitis, K.J. An efficient forward model of the climate controls on interannual variation in tree-ring width. Clim. Dyn. 2011, 36, 2419–2439. [Google Scholar] [CrossRef]
- Tolwinski-Ward, S.E.; Anchukaitis, K.J.; Evans, M.N. Bayesian parameter estimation and interpretation for an intermediate model of tree-ring width. Clim. Past 2013, 9, 1481–1493. [Google Scholar] [CrossRef]
- Levanic, T.; Cater, M.; McDowell, N.G. Associations between growth, wood anatomy, carbon isotope discrimination and mortality in a Quercus robur forest. Tree Physiol. 2011, 31, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Chatziphilippidis, G.; Spyroglou, G. Sustainable Management of Coppice Forests in Greece. In Towards the Sustainable Use of Europe’s Forest Ecosystem and Landscape Research: Scientific Challenges and Opportunities Proc 25–27 June 2003 Tours; Anderson, F., Birot, Y., Pāivinen, R., Eds.; EFI Proceedings: Joensuu, Finland, 2004; Volume 49, pp. 61–70. [Google Scholar]
- Sanders, T.G.M.; Pitman, R.; Broadmeadow, M.S.J. Species-specific climate response of oaks (Quercus spp.) under identical environmental conditions. iFor. Biogeosci. For. 2004, 7, 61–69. [Google Scholar] [CrossRef]
- Costa, M.; Morla, C.; Sáinz, H. Los Bosques Ibéricos: Una Interpretación Geobotánica; Editorial Planeta: Barcelona, Spain, 1997; p. 598. [Google Scholar]
- Cornes, R.C.; Van der Schrier, G.; Van den Besselaar, E.J.; Jones, P.D. An Ensemble Version of the E-OBS Temperature and Precipitation Datasets. J. Geophys. Res. Atmos. 2018, 123, 9391–9409. [Google Scholar] [CrossRef]
- Wazen, N.; Garavaglia, V.; Picard, N.; Besacier, C.; Fady, B. Distribution maps of twenty-four Mediterranean and European ecologically and economically important forest tree species compiled from historical data collections. Ann. Silvicul. Res. 2020, 44, 95–101. [Google Scholar]
- Bordács, S.; Zhelev, P.; Schirone, B. EUFORGEN Technical Guidelines for genetic conservation and use for Hungarian oak (Quercus frainetto). In European Forest Genetic Resources Programme (EUFORGEN); European Forest Institute: Rome, Italy, 2019; p. 6. [Google Scholar]
- Dobbertin, M. Tree growth as indicator of tree vitality and of tree reaction to environmental stress: A review. Eur. J. For. Res. 2005, 124, 319–333. [Google Scholar] [CrossRef]
- Fritts, H.C. Tree Rings and Climate; Academic Press: London, UK, 2001; p. 567. [Google Scholar]
- Holmes, R.L. Computer-assisted quality control in tree–ring dating and measurement. Tree Ring Bull. 1983, 43, 69–78. [Google Scholar]
- Briffa, K.R.; Jones, P.D. Basic chronology statistics and assessment. In Methods of Dendrochronology: Applications in the Environmental Sciences; Kluwer Academic Publishers: Berlin, Germany, 1990; pp. 137–152. [Google Scholar]
- Wigley, T.M.L.; Briffa, K.R.; Jones, P.D. On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J. Clim. Appl. Meteorol. 1984, 23, 201–213. [Google Scholar] [CrossRef]
- Bunn, A.G. A dendrochronology program library in r (dplR). Dendrochronologia 2008, 26, 115–124. [Google Scholar] [CrossRef]
- Zang, C.; Biondi, F. Treeclim: An R package for the numerical calibration of proxy-climate relationships. Ecography 2015, 38, 431–436. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Huang, J.; Van den Dool, H.M.; Georgakakos, K.P. Analysis of model-calculated soil moisture over the United States (1931–1993) and applications to long-range temperature forecasts. J. Clim. 1996, 9, 1350–1362. [Google Scholar] [CrossRef]
- Lavergne, A.; Daux, V.; Villalba, R.; Barichivich, J. Temporal changes in climatic limitation of tree-growth at upper treeline forests: Contrasted responses along the west-to-east humidity gradient in Northern Patagonia. Dendrochronologia 2015, 36, 49–59. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J.; Gutiérrez, E.; González Rouco, F.; Gazol, A.; Sangüesa-Barreda, G.; Andreu-Hayles, L.; Linares, J.C.; Seftigen, K. Assessing forest vulnerability to climate warming using a process-based model of tree growth: Bad prospects for rear-edges. Glob. Chang. Biol. 2017, 23, 2705–2719. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salguero, R.; Camarero, J.J.; Carrer, M.; Gutiérrez, E.; Alla, A.Q.; Andreu-Hayles, L.; Hevia, A.; Koutavas, A.; Martínez-Sancho, E.; Nola, P.; et al. Climate extremes and predicted warming threaten Mediterranean Holocene firs forests refugia. Proc. Natl. Acad. Sci. USA 2017, 114, E10142–E10150. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Wei, C.; Liu, X.; Zhang, L. Qinghai spruce (Picea crassifolia) and Chinese pine (Pinus tabuliformis) show high vulnerability and similar resilience to early-growing-season drought in the Helan Mountains, China. Ecol. Indic. 2020, 110, 105871. [Google Scholar] [CrossRef]
- Camarero, J.J.; Sangüesa-Barreda, G.; Vergarechea, M. Prior height, growth, and wood anatomy differently predispose to drought-induced dieback in two Mediterranean oak species. Ann. For. Sci. 2016, 73, 341–351. [Google Scholar] [CrossRef]
- McDowell, N.G.; Beerling, D.J.; Breshears, D.D.; Fisher, R.A.; Raffa, K.F.; Stitt, M. The interdependence of mechanisms underlying climate-driven vegetation mortality. Trends Ecol. Evol. 2011, 26, 523–532. [Google Scholar] [CrossRef]
- Zweifel, R.; Rigling, A.; Dobbertin, M. Species-specific stomatal response of trees to drought—A link to vegetation dynamics? J. Veg. Sci. 2009, 20, 442–454. [Google Scholar] [CrossRef]
- Villar, R.; Ruiz-Benito, P.; Enrique, G.; Poorter, H.; Cornelissen, J.H.; Quero, J.L. Growth and growth-related traits for a range of Quercus species grown as seedlings under controlled conditions and for adult plants from the field. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus Quercus L.; Gil-Pelegrín, E., Peguero-Pina, J.J., Sancho-Knapik, D., Eds.; Springer: Cham, Switzerland, 2017; pp. 393–417. [Google Scholar]
- Forner, A.; Valladares, F.; Bonal, D.; Granier, A.; Grossiord, C.; Aranda, I. Extreme droughts affecting Mediterranean tree species’ growth and water-use efficiency: The importance of timing. Tree Physiol. 2018, 38, 1127–1137. [Google Scholar] [CrossRef]
- Gričar, J.; Zavadlav, S.; Jyske, T.; Lavrič, M.; Laakso, T.; Hafner, P.; Eler, K.; Vodnik, D. Effect of soil water availability on intra-annual xylem and phloem formation and non-structural carbohydrate pools in stem of Quercus pubescens. Tree Physiol. 2019, 39, 222–233. [Google Scholar] [CrossRef]
- Corcuera, L.; Camarero, J.J.; Sisó, S.; Gil-Pelegrín, E. Radial-growth and wood-anatomical changes in overaged Quercus pyrenaica coppice stands: Functional responses in a new Mediterranean landscape. Trees 2006, 20, 91–98. [Google Scholar] [CrossRef]
- Alla, A.Q.; Camarero, J.J. Contrasting responses of radial growth and wood anatomy to climate in a Mediterranean ring-porous oak: Implications for its future persistence or why the variance matters more than the mean. Eur. J. For. Res. 2012, 131, 1537–1550. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Martín, P.; Saz, M.A.; Gea-Izquierdo, G.; Cañellas, I.; Gil-Pelegrín, E. Evidence of vulnerability segmentation in a deciduous Mediterranean oak (Quercus subpyrenaica E. H. del Villar). Trees 2015, 29, 1917–1927. [Google Scholar] [CrossRef]
- Ripullone, F.; Camarero, J.J.; Colangelo, M.; Voltas, J. Variation in the access to deep soil water pools explains tree-to-tree differences in drought-triggered dieback of Mediterranean oaks. Tree Physiol. 2020, 40, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Altman, J.; Hédl, R.; Szabó, P.; Mazůrek, P.; Riedl, V.; Müllerovám, J.; Kopecký, M.; Doležal, J. Tree-rings mirror management legacy: Dramatic response of standard oaks to past coppicing in Central Europe. PLoS ONE 2013, 8, e55770. [Google Scholar] [CrossRef]
- Cuny, H.E.; Rathgeber, C.B.K.; Lebourgeois, F.; Fortin, M.; Fournier, M. Life strategies in intra-annual dynamics of wood formation: Example of three conifer species in a temperate forest in north-east France. Tree Physiol. 2012, 32, 612–625. [Google Scholar] [CrossRef]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processes and long-term consequences. Ann. For. Sci. 2006, 63, 625–644. [Google Scholar] [CrossRef]
- Fichot, R.; Chamaillard, S.; Depardieu, C.; Le Thiec, D.; Cochard, H.; Barigah, T.S.; Brignolas, F. Hydraulic efficiency and coordination with xylem resistance to cavitation, leaf function, and growth performance among eight unrelated Populus deltoids × Populus nigra hybrids. J. Exp. Bot. 2011, 62, 2093–2106. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sisó, S.; Sancho-Knapik, D.; Díaz-Espejo, A.; Flexas, J.; Galmés, J.; Gil-Pelegrín, E. Leaf morphological and physiological adaptations of a deciduous oak (Quercus faginea Lam.) to the Mediterranean climate: A comparison with a closely related temperate species (Quercus robur L.). Tree Physiol. 2016, 36, 287–299. [Google Scholar] [CrossRef]
- Palacio, S.; Camarero, J.J.; Maestro, M.; Alla, A.Q.; Lahoz, E.; Montserrat-Martí, G. Are storage and tree growth related? Seasonal nutrient and carbohydrate dynamics in evergreen and deciduous Mediterranean oaks. Trees 2018, 32, 777–790. [Google Scholar] [CrossRef]
- Poorter, L.; Castilho, C.V.; Schietti, J.; Oliveira, R.S.; Costa, F.R.C. Can traits predict individual growth performance? A test in a hyperdiverse tropical forest. New Phytol. 2018, 219, 109–121. [Google Scholar] [CrossRef]
- Magnani, F.; Mencuccini, M.; Grace, J. Age-related decline in stand productivity: The role of structural acclimation under hydraulic constraints. Plant Cell Environ. 2000, 23, 251–263. [Google Scholar] [CrossRef]
- Manion, P.D. Tree Disease Concepts; Prentice Hall: Upper Saddle River, NJ, USA, 1981; p. 409. [Google Scholar]
- Gómez-Aparicio, L.; García-Valdés, R.; Ruiz-Benito, P.; Zavala, M.A. Disentangling the relative importance of climate, size and competition on tree growth in Iberian forests: Implications for forest management under global change. Glob. Chang. Biol. 2011, 17, 2400–2414. [Google Scholar] [CrossRef]
- Drobyshev, I.; Linderson, H.; Sonesson, K. Relationship between crown condition and tree diameter growth in southern Swedish oaks. Environ. Monit. Assess. 2007, 128, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Matías, L.; Abdelaziz, M.; Godoy, O.; Gómez-Aparicio, L. Disentangling the climatic and biotic factors driving changes in the dynamics of Quercus suber populations across the species’ latitudinal range. Divers. Distrib. 2019, 25, 524–535. [Google Scholar] [CrossRef]
- Matías, L.; Pérez-Ramos, I.M.; Gómez-Aparicio, L. Are northern-edge populations of cork oak more sensitive to drought than those of the southern-edge? Environ. Exp. Bot. 2019, 163, 78–85. [Google Scholar] [CrossRef]
- Rodríguez-Calcerrada, J.; Sancho-Knapik, D.; Martin-StPaul, N.K.; Limousin, J.-M.; McDowell, N.C.; Gil-Pelegrín, E. Drought-Induced Oak Decline—Factors Involved, Physiological Dysfunctions, and Potential Attenuation by Forestry Practices. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus Quercus L.; Gil-Pelegrín, E., Peguero-Pina, J.J., Sancho-Knapik, D., Eds.; Springer: Cham, Switzerland, 2017; pp. 393–417. [Google Scholar]
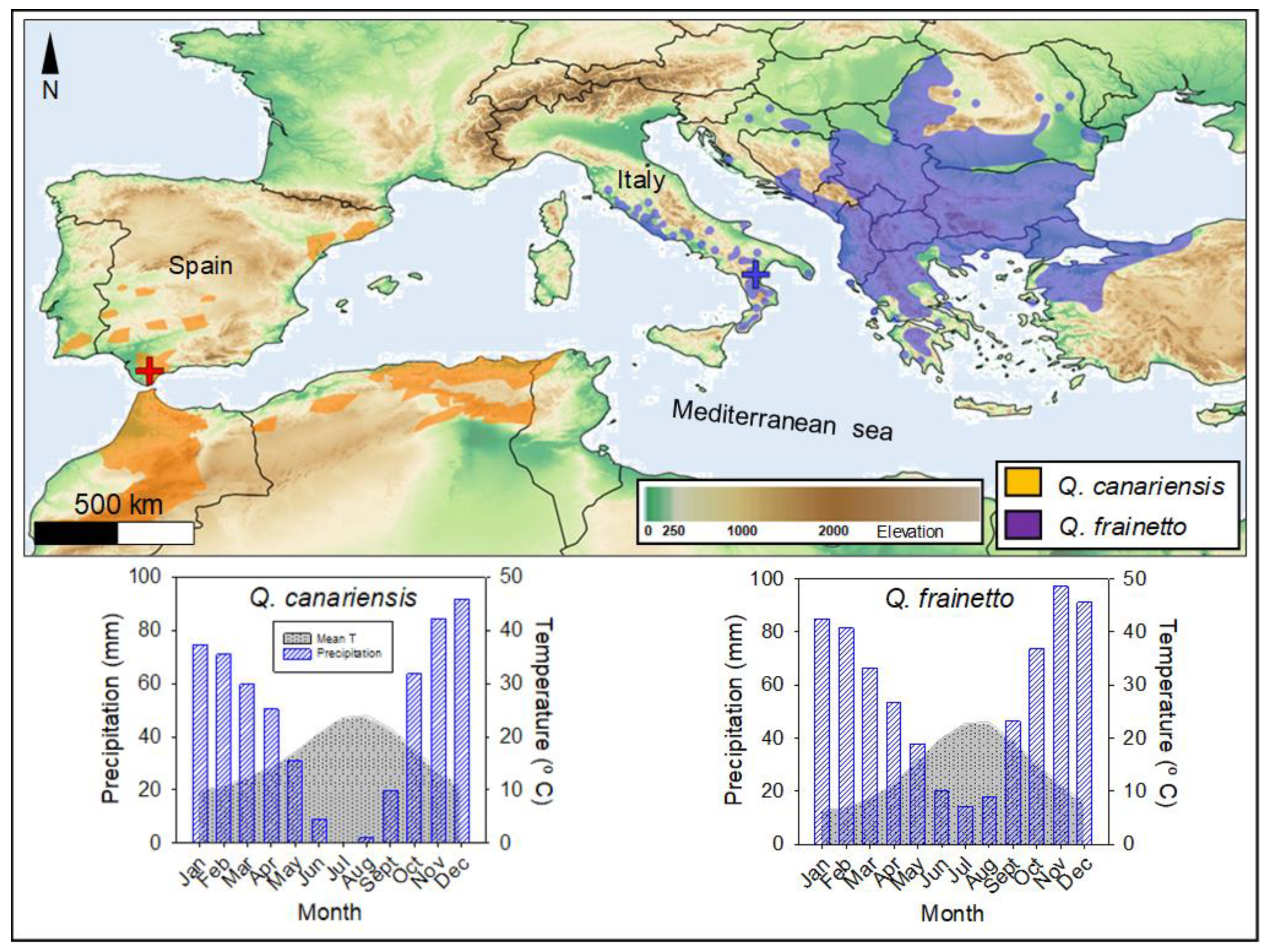

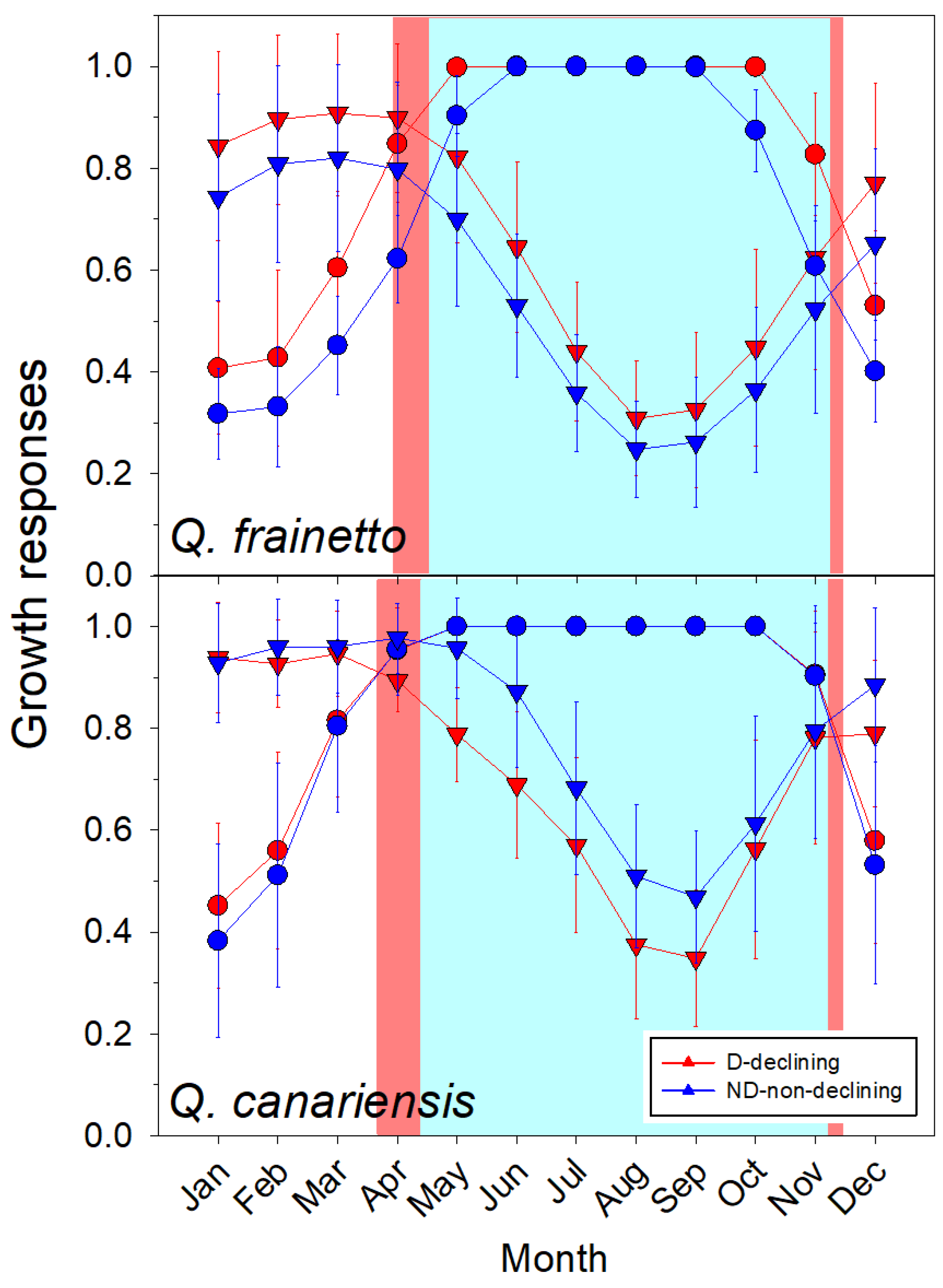
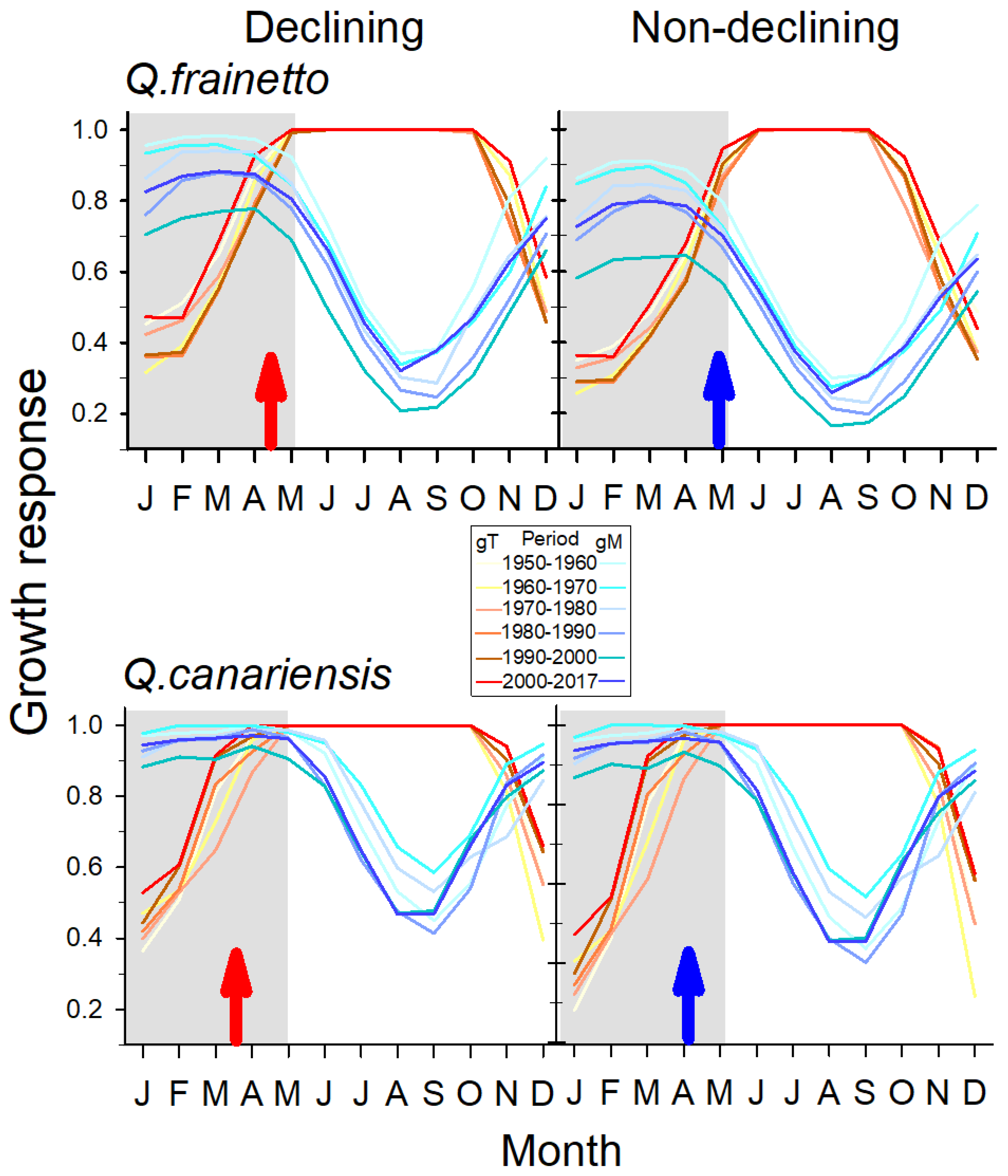
| Variable | Q. frainetto | Q. canariensis |
|---|---|---|
| Site name | San Paolo Albanese | Gamir-Alcornocales |
| Latitude (N) | 40°00′50″ | 36°34′12″ |
| Longitude (W/E) | 16°21′01″ E | 5°32′02″ W |
| Elevation range (m a.s.l.) | 850–1170 | 490–530 |
| Aspect | SW | SE |
| Slope (%) | 25–30 | 15 |
| Annual precipitation (mm) | 584 ± 17 | 560 ± 23 |
| Spring precipitation (mm) | 132 ± 6 | 141 ± 8 |
| Mean temperature (°C) | 14.1 ± 0.5 | 16.4 ± 0.1 |
| Mean spring temperature (°C) | 11.9 ± 0.2 | 14.6 ± 0.2 |
| Variable | Q. frainetto | Q. canariensis | ||
|---|---|---|---|---|
| ND | D | ND | D | |
| DBH (cm) | 32.3 ± 0.8 | 31.9 ± 0.9 | 48.5 ± 1.8b | 55.8 ± 5.5a |
| H (m) | 14.3 ± 0.6a | 9.9 ± 0.5b | 11.7 ± 0.2 | 11.1 ± 0.5 |
| Defoliation (%) | 8 ± 0.8b | 66 ± 2.8a | 10 ± 2b | 73 ± 4a |
| No. trees/No. radii | 24/34 | 27/34 | 11/11 | 11/9 |
| Age (years) 1,2 | 140 ± 3a | 132 ± 6b | 107 ± 4b | 117 ± 5a |
| Timespan | 1851–2016 | 1831–2016 | 1897–2017 | 1873–2017 |
| RW ± SE (mm) | 0.70 ± 0.01a | 0.61 ± 0.02b | 1.48 ± 0.04a | 1.35 ± 0.05b |
| AC | 0.75 ± 0.01 | 0.77 ± 0.02 | 0.62 ± 0.05 | 0.68 ± 0.06 |
| MS | 0.23 ± 0.02 | 0.23 ± 0.01 | 0.29 ± 0.02 | 0.32 ± 0.01 |
| IC | 0.51 ± 0.02 | 0.46 ± 0.02 | 0.47 ± 0.03a | 0.36 ± 0.09b |
| EPS | 0.951 | 0.928 | 0.930 | 0.898 |
| Periods | Variable | Q. frainetto | Q. canariensis | ||
|---|---|---|---|---|---|
| ND | D | ND | D | ||
| 1950–2016 | r | 0.38 | 0.36 | 0.46 | 0.33 |
| T1/T2 (°C) | 2.0/14.3 | 2.6/11.3 | 7.1/13.3 | 7.6/13.4 | |
| M1/M2 (v/v) | 0.079/0.369 | 0.085/0.416 | 0.009/0.246 | 0.018/0.263 | |
| 1950–1983 | r | 0.29 | 0.36 | 0.32 | 0.26 |
| T1/T2 (°C) | 2.5/12.9 | 3.6/10.9 | 5.4/15.4 | 6.7/13.7 | |
| M1/M2 (v/v) | 0.065/0.460 | 0.081/0.462 | 0.045/0.101 | 0.027/0.111 | |
| 1984–2016 | r | 0.45 | 0.36 | 0.60 | 0.49 |
| T1/T2 (°C) | 2.6/17.5 | 3.2/11.7 | 6.66/13.6 | 8.67/15.3 | |
| M1/M2 (v/v) | 0.094/0.216 | 0.069/0.477 | 0.014/0.219 | 0.077/0.267 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Salguero, R.; Colangelo, M.; Matías, L.; Ripullone, F.; Camarero, J.J. Shifts in Growth Responses to Climate and Exceeded Drought-Vulnerability Thresholds Characterize Dieback in Two Mediterranean Deciduous Oaks. Forests 2020, 11, 714. https://doi.org/10.3390/f11070714
Sánchez-Salguero R, Colangelo M, Matías L, Ripullone F, Camarero JJ. Shifts in Growth Responses to Climate and Exceeded Drought-Vulnerability Thresholds Characterize Dieback in Two Mediterranean Deciduous Oaks. Forests. 2020; 11(7):714. https://doi.org/10.3390/f11070714
Chicago/Turabian StyleSánchez-Salguero, Raúl, Michele Colangelo, Luis Matías, Francesco Ripullone, and J. Julio Camarero. 2020. "Shifts in Growth Responses to Climate and Exceeded Drought-Vulnerability Thresholds Characterize Dieback in Two Mediterranean Deciduous Oaks" Forests 11, no. 7: 714. https://doi.org/10.3390/f11070714
APA StyleSánchez-Salguero, R., Colangelo, M., Matías, L., Ripullone, F., & Camarero, J. J. (2020). Shifts in Growth Responses to Climate and Exceeded Drought-Vulnerability Thresholds Characterize Dieback in Two Mediterranean Deciduous Oaks. Forests, 11(7), 714. https://doi.org/10.3390/f11070714







