Influence of Copper and Biopolymer/Saqez Resin on the Properties of Poplar Wood
Abstract
1. Introduction
2. Material and Methods
2.1. Material
2.2. Methods
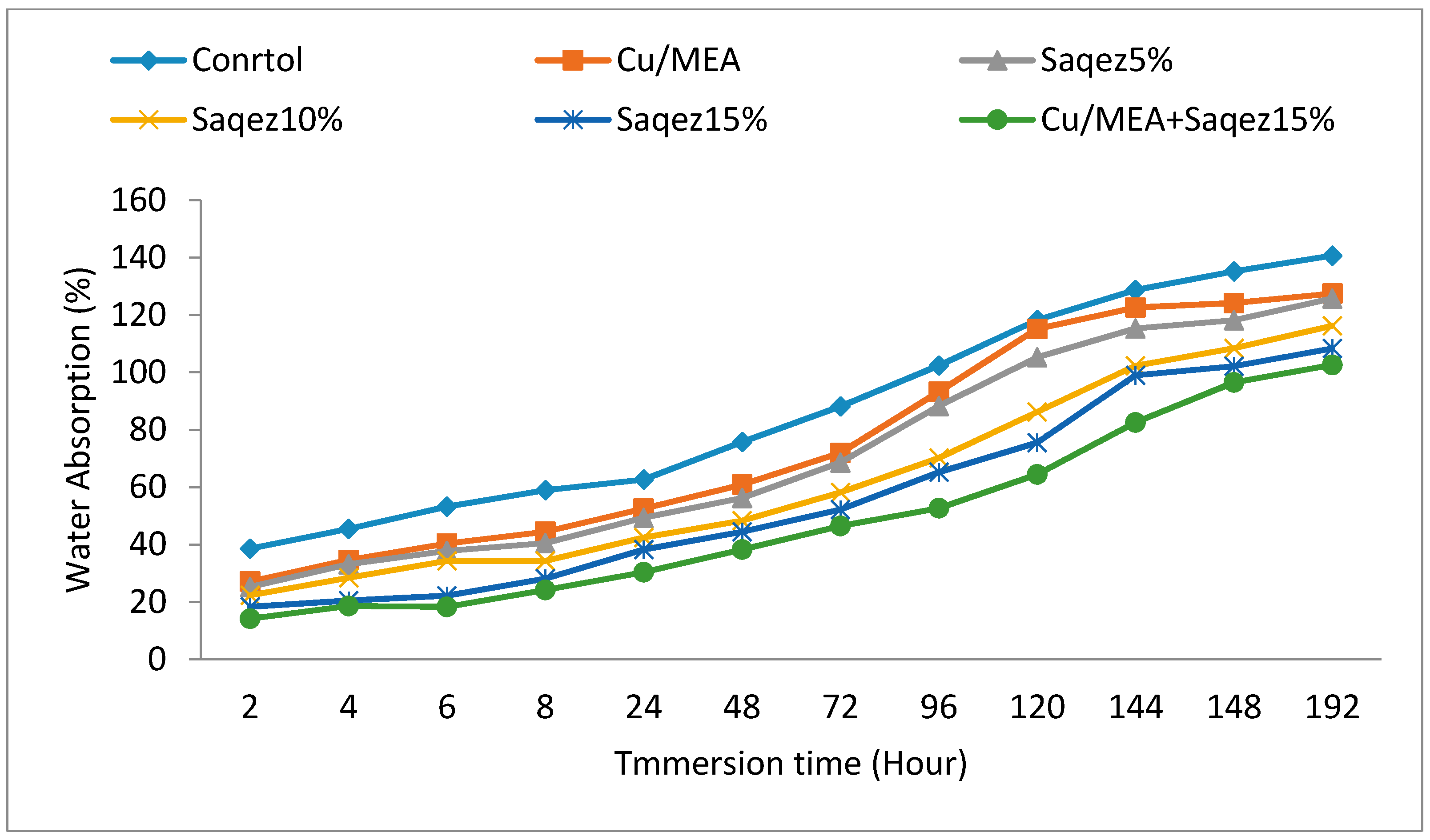
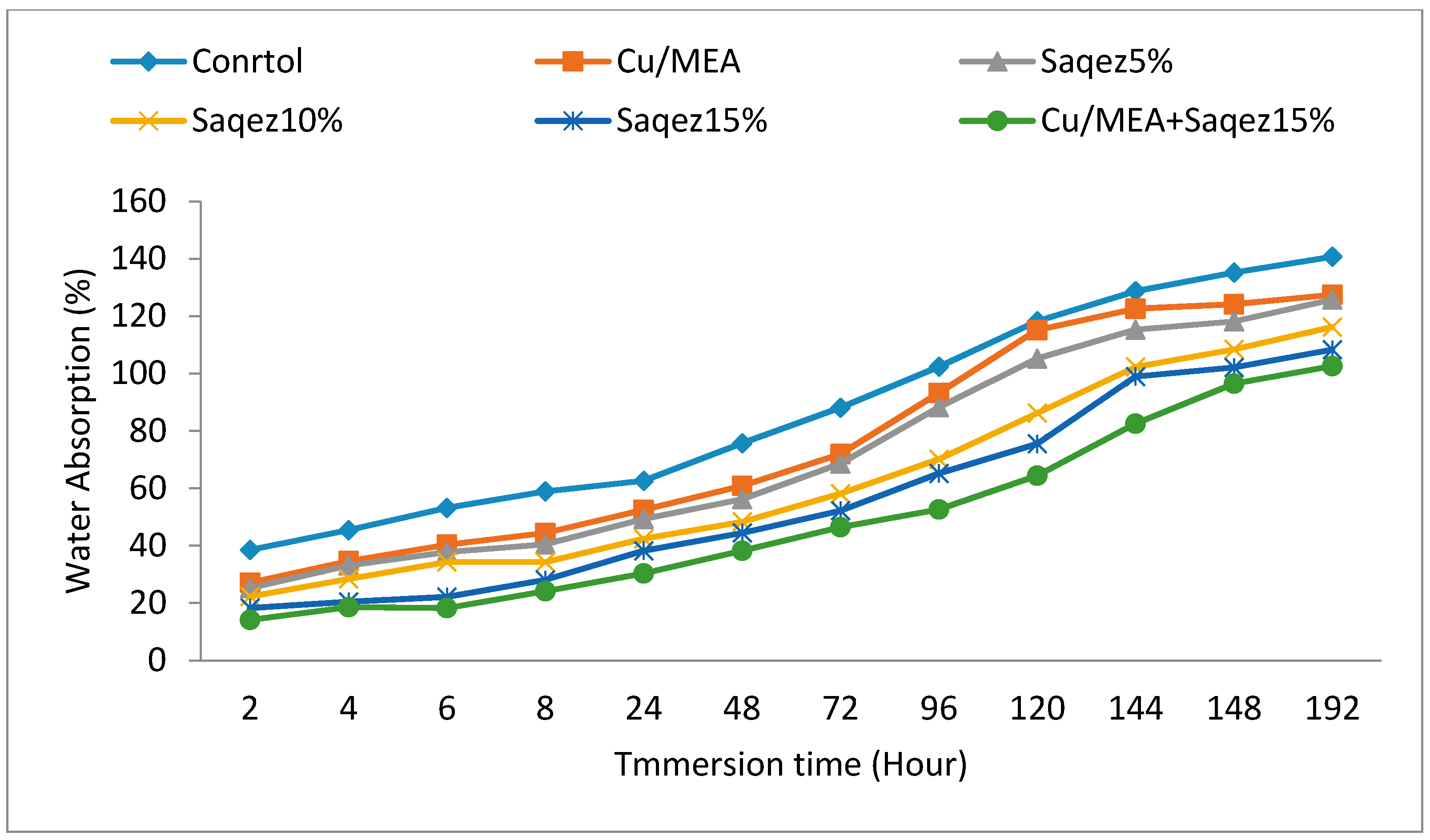
2.2.1. Water Absorption and Volumetric Swelling Measurements
2.2.2. Biological Durability
2.2.3. Statistical Analysis
3. Result and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cheung, K.C.K. Wooden structures. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1, pp. 1–14. [Google Scholar]
- Kutnik, M.; Suttie, E.; Brischke, C. European standards on durability and performance of wood and wood-based products—Trends and challenges. Wood Mater. Sci. Eng. 2014, 9, 122–133. [Google Scholar] [CrossRef]
- Lebow, S. Leaching of Wood Preservative Components and Their Mobility in the Environment: Summary of Pertinent Literature; General Technical Report FPL-GTR-93; US Department of Agriculture: Madison, WI, USA, 2006. [Google Scholar]
- Bahmani, M.; Fromm, J.; Schmidt, O.; Melcher, E. Residual metal content and metal distribution in chromium/copper-treated wood after field and laboratory leaching exposure. Eur. J. Wood Prod. 2015, 73, 377–384. [Google Scholar] [CrossRef]
- Bahmani, M.; Melcher, E.; Schmidt, O.; Fromm, J. Influence of exposure time, wood species and dimension on the remaining copper and chromium content in CC-treated wood after field and laboratory leaching tests. Holzforschung 2015, 69, 1143–1150. [Google Scholar] [CrossRef]
- Casado-Sanz, M.M.; Silva-Castro, I.; Ponce-Herrero, L.; Martín-Ramos, P.; Martín-Gil, J.; Acuña-Rello, L. White-rot fungi control on Populus spp. wood by pressure treatments with silver nanoparticles, chitosan oligomers and propolis. Forests 2019, 10, 885. [Google Scholar] [CrossRef]
- Žlahtič, M.; Mikac, U.; Serša, I.; Merela, M.; Humar, M. Distribution and penetration of tung oil in wood studied by magnetic resonance microscopy. Industr. Crop Prod. 2017, 96, 149–157. [Google Scholar] [CrossRef]
- Woźniak, M.; Kwaśniewska-Sip, P.; Waśkiewicz, A.; Cofta, G.; Ratajczak, I. The possibility of propolis extract application in wood protection. Forests 2020, 11, 465. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.Y.Y.; Grant, R.I.; Friedman, M.; Elliott, C.; Situ, C. Antibacterial activities of naturally occurring compounds against Mycobacterium avium subsp. paratuberculosis. Appl. Environ. Microbiol. 2008, 74, 5986–5990. [Google Scholar] [CrossRef]
- Singh, T.; Singh, A. A review on natural products as wood protectant. Wood Sci. Technol. 2012, 46, 851–870. [Google Scholar] [CrossRef]
- Humar, M.; Lesar, B. Efficacy of linseed- and tung-oil-treated wood against wood-decay fungi and water uptake. Int. Biodeterior. Biodegrad. 2013, 85, 223–227. [Google Scholar] [CrossRef]
- Pánek, M.; Reinprecht, L.; Hulla, M. Ten essential oils for beech wood protection. Efficacy against wood-destroying fungi and moulds, and effect on wood discoloration. BioResources 2014, 9, 5588–5603. [Google Scholar] [CrossRef]
- De Medeiros, F.C.; Gouveia, F.N.; Bizzo, H.R.; Vieira, R.F.; Del Menezzi, C.H. Fungicidal activity of essential oils from Brazilian Cerrado species against wood decay fungi. Int. Biodeterior. Biodegrad. 2016, 114, 87–93. [Google Scholar] [CrossRef]
- Fidah, A.; Salhi, N.; Rahouti, M.; Kabouchi, B.; Ziani, M.; Aberchane, M.; Famiri, A. Natural durability of Cedrus atlantica wood related to the bioactivity of its essential oil against wood decaying fungi. Maderas Cienc. Tecnol. 2016, 18, 567–576. [Google Scholar] [CrossRef]
- Isman, M.B.; Miresmaill, S.; MacHial, C. Commercial opportunities for pesticides based on plant essential oils in agriculture, industry and consumer products. Phytochem. Rev. 2011, 10, 197–204. [Google Scholar] [CrossRef]
- Fatima, R.; Morrell, J.J. Ability of plant-derived oils to inhibit dampwood termite (Zootermopsis augusticollis) activity. Maderas Cienc. Tecnol. 2015, 17, 685–690. [Google Scholar] [CrossRef]
- Su, Y.C.; Ho, C.L.; Wang, E.I.C.; Chang, S.T. Antifungal activities and chemical compositions of essential oils from leaves of four eucalypts. Taiwan Lin Ye Ke Xue 2006, 21, 49–61. [Google Scholar]
- Yang, V.W.; Clausen, C.A. Moldicidal Properties of Seven Essential Oils; Technical Report No. 06-30404; IRG Secretariat: Stockholm, Sweden, 2006. [Google Scholar]
- Mohareb, A.S.; Badawy, M.E.; Abdelgaleil, S.A. Antifungal activity of essential oils isolated from Egyptian plants against wood decay fungi. J. Wood Sci. 2013, 59, 499–505. [Google Scholar] [CrossRef]
- Bahmani, M.; Schmidt, O. Plant essential oils for environment-friendly protection of wood objects against fungi. Maderas Cienc. Tecnol. 2018, 20, 325–332. [Google Scholar] [CrossRef]
- Alfredsen, G.; Flaete, P.O. Tall oil performance after a decade of field exposure. In Proceedings of the Annual Meeting of the International Research Group on Wood Protection, Viña del Mar, Chile, 10–14 May 2015; p. 30672. [Google Scholar]
- Can, A.; Sivrikaya, H. Combined effects of copper and oil treatment on the properties of Scots pine wood. Drewno Prace Naukowe Doniesienia Komunikaty 2017, 60, 89–103. [Google Scholar]
- Dhyani, S.; Kamdem, D.P. Bioavailability and form of copper in wood treated with copper-based preservative. Wood Sci. Technol. 2012, 46, 1203–1213. [Google Scholar] [CrossRef]
- Pankras, S.; Cooper, P.A.; Wylie, S. Relationship between copper species in solution and leaching from alkaline copper quat (ACQ) treated wood. Holzforschung 2012, 66, 505–514. [Google Scholar] [CrossRef]
- Lee, M.J.; Cooper, P.A. Copper precipitation of Cu-monoethanolamine preservative in wood. Holzforschung 2012, 66, 1017–1024. [Google Scholar] [CrossRef]
- Humar, M.; Pohleven, F.; Žlindra, D. Influence of water, properties on leaching of copper-based preservatives from treated wood. Wood Res. 2006, 51, 69–76. [Google Scholar]
- Freeman, M.H.; Nicholas, D.D.; Schultz, T.P. Nonarsenical wood protection: Alternatives for chromated copper arsenate, creosote and pentachlorophenol. In Environmental Impacts of Treated Wood; Townsend, T.G., Solo-Gabriele, H., Eds.; CRC/Taylor and Francis: Boca Raton, FL, USA, 2006; pp. 19–36. [Google Scholar]
- Schultz, T.P.; Nicholas, D.D.; McIntyre, C.R. Recent patents and developments in biocidal wood protection systems for exterior applications. Recent Pat. Mater. Sci. 2008, 1, 128. [Google Scholar] [CrossRef]
- Wang, J.; Zhong, H.; Ma, E.; Cao, J. Properties of wood treated with compound systems of paraffin wax emulsion and copper azole. Eur. J. Wood Prod. 2018, 76, 315–323. [Google Scholar] [CrossRef]
- Mahdavi, A. The economic, social, and ecological impacts of wild pistachio (Pistacia atlantica Desf.) oleo-gum resin extraction cooperatives in Zagros forests, Ilam Province, Iran. For. Trees Livelihoods 2015, 24, 275–284. [Google Scholar] [CrossRef]
- Ghalem, B.R.; Benali, M. Antimicrobial activity determination of gum of Pistacia atlantica Desf. oil. Afr. J. Microbiol. Res. 2010, 4, 2457–2460. [Google Scholar]
- Mousavi, R. Non-wood forest products, the utilisation, and harvesting methods in Sardasht, North West Iran. Int. J. For. Soil Eros. 2012, 2, 133–136. [Google Scholar]
- Balatinecz, J.J.; Kretschmann, D.E. Properties and utilization of poplar wood. In Poplar Culture in North America Part A; NRC Research Press: Ottawa, QC, Canada, 2001; pp. 277–291. [Google Scholar]
- Brischke, C.; Meyer, L.; Alfredsen, G.; Humar, M.; Francis, L.; Flæte, P.O.; Larsson-Brelid, P. Natural durability of timber exposed above ground—A survey. Drvn. Ind. 2013, 64, 113–129. [Google Scholar] [CrossRef]
- International Organization for Standardization. Physical and Mechanical Properties of Wood-Test Methods for Small Clear Wood Specimens-Part 14: Determination of Volumetric Shrinkage; ISO 13061-14:2016; ISO: Geneva, Switzerland, 2016; p. 5. [Google Scholar]
- European Committee for Standardization (CEN). Wood Preservatives Test Method for Determining the Protective Effectiveness Against Wood Destroying Basidiomycetes Determination of the Toxic Values; CEN EN 113:1996; CEN: Brussel, Belgium, 1996; p. 28. [Google Scholar]
- European Committee for Standardization (CEN). Wood Preservatives—Accelerated Ageing of Treated Wood Prior to Biological Testing—Leaching Procedure; CEN EN 84; CEN: Brussels, Belgium, 2002; p. 7. [Google Scholar]
- Liu, M.; Hui, Z.; Erni, M.; Ru, L. Resistance to fungal decay of paraffin wax emulsion/copper azole compound system treated wood. Int. Biodeterior. Biodegrad. 2018, 129, 61–66. [Google Scholar] [CrossRef]
- Treu, A.; Larnøy, E.; Militz, H. Process related copper leaching during a combined wood preservation process. Eur. J. Wood Prod. 2011, 69, 263–269. [Google Scholar] [CrossRef][Green Version]
- O’Leary, P.; Hodges, P.A. The relationship between full penetration uptake and swelling of different fluids. Wood Sci. Technol. 2001, 35, 217–227. [Google Scholar] [CrossRef]
- Kozlevčar, B.; Humar, M.; Strauch, P.; Leban, I. Fixation of copper(II) ions in aqueous solution to lignin model compound vanillin in an absence of the nitrogen donor ligands; structural and EPR correlation. Z. Naturforsch. B 2005, 60, 1273–1277. [Google Scholar] [CrossRef]
- Meyer-Veltrup, L.; Brischke, C.; Alfredsen, G.; Humar, M.; Flæte, P.O.; Isaksson, T.; Brelid, P.L.; Westin, M.; Jermer, J. The combined effect of wetting ability and durability on outdoor performance of wood: Development and verification of a new prediction approach. Wood Sci. Technol. 2017, 51, 615–637. [Google Scholar] [CrossRef]
- Banks, W.B. Factors affecting the introduction of preservatives into wood. Pestic. Sci. 1972, 3, 219–227. [Google Scholar] [CrossRef]
- Schmidt, O. Wood and Tree Fungi: Biology, Damage, Protection, and Use; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Humar, M.; Kržišnik, D.; Lesar, B.; Thaler, N.; Ugovšek, A.; Zupančič, K.; Žlahtič, M. Thermal modification of wax-impregnated wood to enhance its physical, mechanical, and biological properties. Holzforschung 2017, 71, 57–64. [Google Scholar] [CrossRef]
- Sharifi, M.S.; Hazell, S.L. GC-MS analysis and antimicrobial activity of the essential oil of the trunk exudates from Pistacia atlantica kurdica. J. Pharm. Sci. Res. 2011, 3, 1364. [Google Scholar]
- Mahmoud Kia, M. Heat Modification of Poplar Wood Using Rapeseed Oil Resin of Persian Turoentine for Urban Furniture Application. Ph.D. Thesis, University of Tehran, Tehran, Iran, July 2018. [Google Scholar]
- Glisić, S.B.; Milojević, S.Z.; Dimitrijević, S.I.; Orlović, A.M.; Skala, D.U. Antimicrobial activity of the essential oil and different fractions of Juniperus communis L. and a comparison with some commercial antibiotics. J. Serb. Chem. Soc. 2007, 72, 311–320. [Google Scholar] [CrossRef]
- Da Silva, M.R.; Brito, J.O.; Govone, J.S.; de Oliveira Machado, G.; Calil, C.; Christoforo, A.L.; Lahr, F.A.R. Chemical and mechanical properties changes in Corymbia citriodora wood submitted to heat treatment. Int. Biodeterior. Biodegrad. 2015, 5, 98–104. [Google Scholar]
- Salem, M.Z.; Zidan, Y.E.; El Hadidi, N.M.; Mansour, M.M.; Elgat, W.A.A. Evaluation of usage three natural extracts applied to three commercial wood species against five common molds. Int. Biodeterior. Biodegrad. 2016, 110, 206–226. [Google Scholar] [CrossRef]
- Humar, M.; Lesar, B. Fungicidal properties of individual components of copper-ethanolamine-based wood preservatives. Int. Biodeterior. Biodegrad. 2008, 62, 46–50. [Google Scholar] [CrossRef]


| Test Groups | Preservative Solution (%) | ||
|---|---|---|---|
| Cu (%) | Saqez Resin (%) | ||
| A | Control | 0 | 0 |
| B | Copper/MEA * | 0/5 | 0 |
| C | Saqez | 0 | 5 |
| 0 | 10 | ||
| 0 | 15 | ||
| D | Copper/MEA + Saqez | 0/5 | 15 |
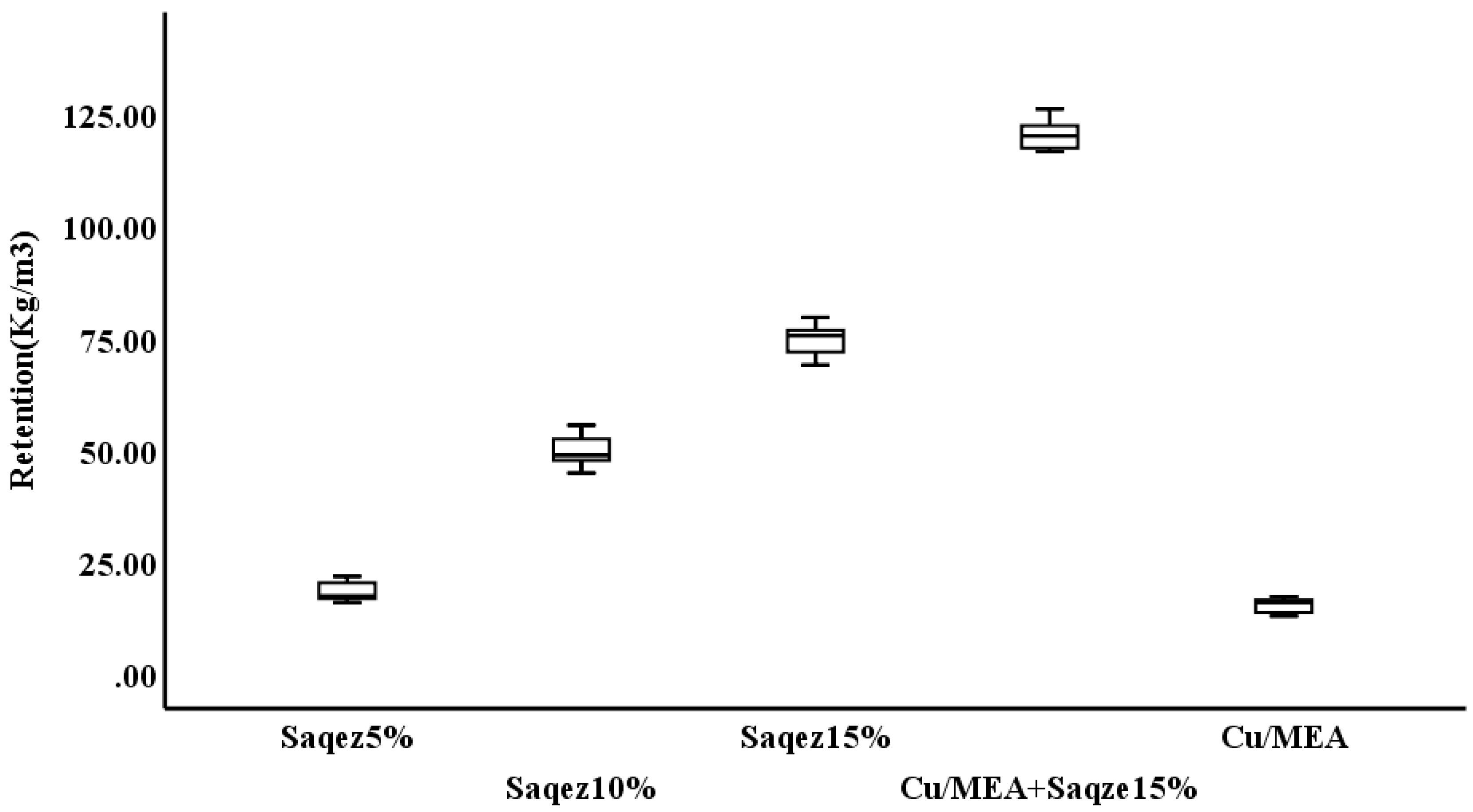
| Treatment | Retention * (kg/m3) | Shapiro–Wilk Test p-Value | ANOVA p-Value | DMRT ** | |||
|---|---|---|---|---|---|---|---|
| Saqez 5% | 18.42 ± 3.09 | 0.434 | 0.000 | a | |||
| Saqez 10% | 49.85 ± 5.20 | 0.910 | b | ||||
| Saqez 15% | 74.55 ± 5.19 | 0.911 | c | ||||
| Cu/MEA + Saqez 15% | 120.56 ± 4.82 | 0.689 | d | ||||
| Cu/MEA | 15.24 ± 2.32 | 0.914 | a | ||||
| Treatment | WPG * (%) | Shapiro–Wilk Test p-Value | ANOVA p-Value | DMRT ** | |||
|---|---|---|---|---|---|---|---|
| Saqez5% | −0.81 ± 0.05 | 0.148 | 0.000 | a | |||
| Saqez10% | 6.17 ± 1.61 | 0.721 | b | ||||
| Saqez15% | 9.66 ± 1.47 | 0.327 | c | ||||
| Cu/MEA + Saqez15% | 23.21 ± 3.21 | 0.380 | d | ||||
| Cu/MEA | 2.53 ± 0.12 | 0.346 | e | ||||
| Leaching Test | Treatment | Mass Loss * (%) | Shapiro–Wilk Test p-Value | ANOVA p-Value | DMRT ** | |||
|---|---|---|---|---|---|---|---|---|
| Before Leaching | Control | 39.30 ± 2.17 | 0.86 | 0.000 | a | |||
| Saqez5% | 30.06 ± 2.64 | 0.37 | b | |||||
| Saqez10% | 28.15 ± 2.95 | 0.94 | b | |||||
| Saqez15% | 20.43 ± 2.74 | 0.49 | c | |||||
| Cu/MEA + Saqez15% | 0.92 ± 0.21 | 0.72 | d | |||||
| Cu/MEA | 1.10 ± 0.23 | 0.84 | d | |||||
| After leaching | Control | 44.77 ± 3.88 | 0.58 | 0.000 | a | |||
| Saqez5% | 35.22 ± 2.05 | 0.39 | b | |||||
| Saqez10% | 33.74 ± 2.58 | 0.80 | b | |||||
| Saqez15% | 26.43 ± 2.99 | 0.29 | c | |||||
| Cu/MEA + Saqez15% | 1.55 ± 0.051 | 0.98 | d | |||||
| Cu/MEA | 3.51 ± 0.40 | 0.18 | d | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, H.; Bahmani, M.; Rahimi, S.; Humar, M. Influence of Copper and Biopolymer/Saqez Resin on the Properties of Poplar Wood. Forests 2020, 11, 667. https://doi.org/10.3390/f11060667
Dong H, Bahmani M, Rahimi S, Humar M. Influence of Copper and Biopolymer/Saqez Resin on the Properties of Poplar Wood. Forests. 2020; 11(6):667. https://doi.org/10.3390/f11060667
Chicago/Turabian StyleDong, Huijun, Mohsen Bahmani, Sohrab Rahimi, and Miha Humar. 2020. "Influence of Copper and Biopolymer/Saqez Resin on the Properties of Poplar Wood" Forests 11, no. 6: 667. https://doi.org/10.3390/f11060667
APA StyleDong, H., Bahmani, M., Rahimi, S., & Humar, M. (2020). Influence of Copper and Biopolymer/Saqez Resin on the Properties of Poplar Wood. Forests, 11(6), 667. https://doi.org/10.3390/f11060667






