Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Białowieża Forest District, Poland
Abstract
1. Introduction
2. Material and Methods
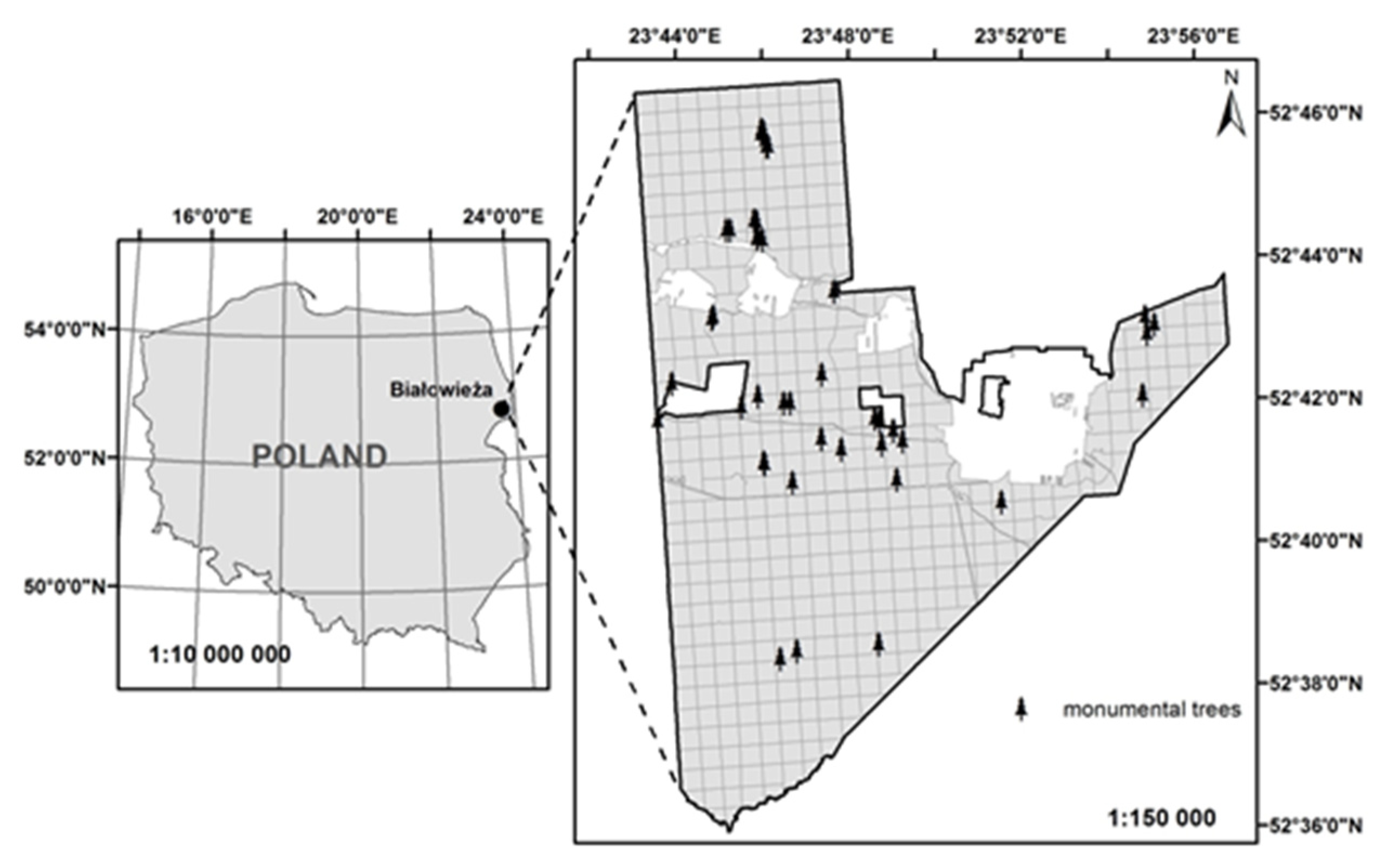
2.1. Study Area
2.2. Health State Assessment and Meteorological Data Analysis
- inventory data concerning location;
- crown canopy density and defoliation of the upper part of the crown for health assessment [15];
- multispectral airborne images of Norway spruce monumental trees obtained from Project LIFE+ ForBioSensing PL, ‘Comprehensive monitoring of stand dynamics in the Białowieża Forest’;
- meteorological data from 2008–2016 obtained from the Institute of Meteorology and Water Management in Białowieża.
2.3. Plant Material and Nuclear Loci Genotyping
2.4. Genetic Variation Based on Allelic Diversity
2.5. Genetic Differentiation between Spruce Populations
2.6. Spruce Populations Clustering
3. Results
3.1. Spruce Health Status Based on Ground and Remote Sensing Inventory
3.2. Analysis of Meteorological Data
3.3. Genetic VAriation within Populations
3.4. Genetic Differentiation between Spruce Populations
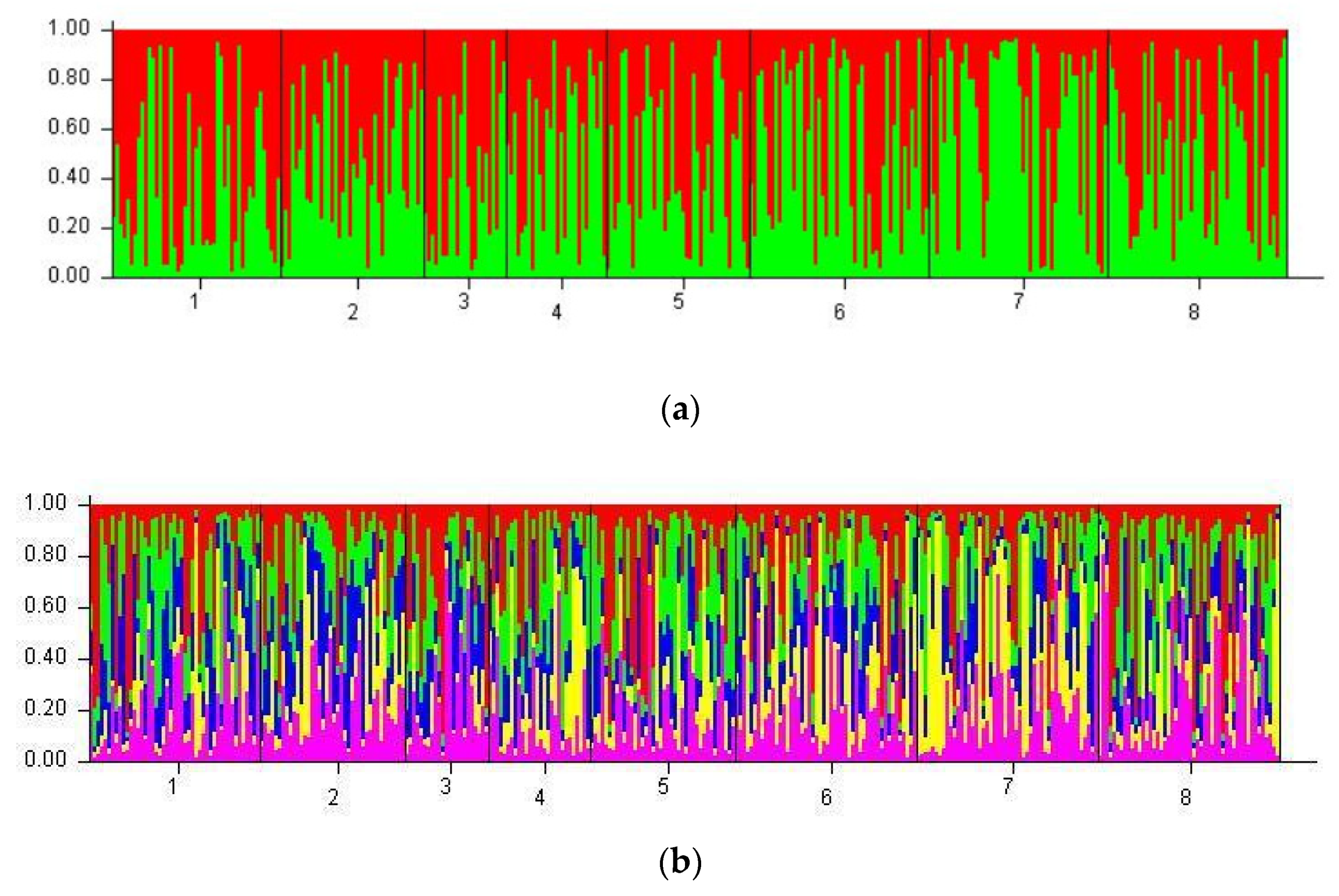
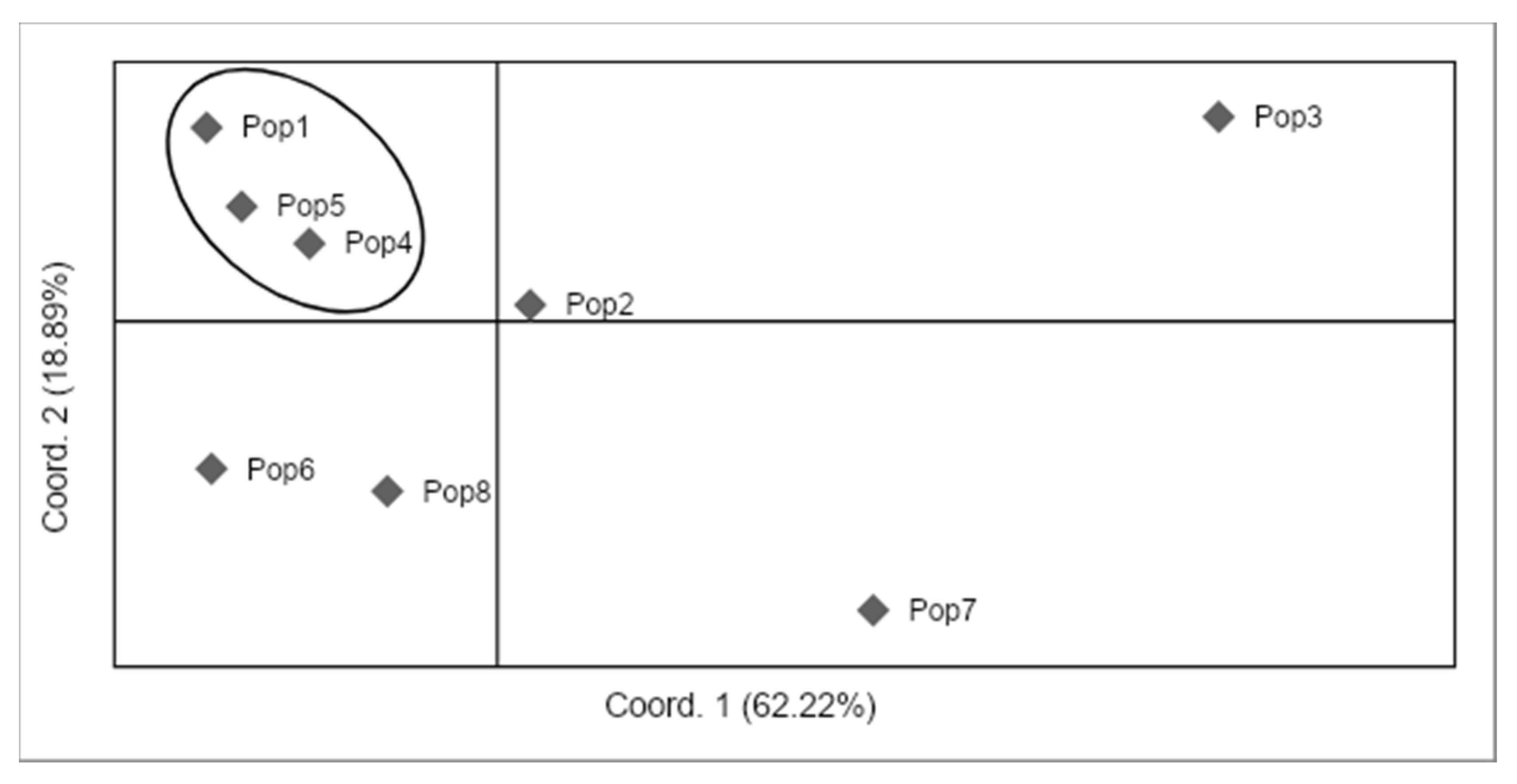
3.5. Population Clustering
4. Discussion
4.1. Old Forest Trees Maintain Ecosystem Biodiversity
4.2. Climatic Conditions and Insect Outbreaks
4.3. Health Status Evaluation of Spruces in BFD
4.4. Natural Selection, Gene Flow and Possible Changes in Biodiversity
4.5. Genetic Structure of Monumental Trees
4.6. STRUCTURE Model and Specific Allelic Patterns
4.7. Gene Diversity and Adaptive Potential
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okołów, C. Białowieża National Park: Know It, Understand It, Protect It; Białowieski Park Narodowy: Białowieski, Poland, 2009. [Google Scholar]
- Kowalczyk, B.; Mikowski, R.; Mikowski, L. Environmental Law in Poland; Wolters Kluwer: Philadelphia, PA, USA, 2019; p. 232. [Google Scholar]
- Schroeder, M.; Cocoş, D. Performance of the tree-killing bark beetles Ips typographus and Pityogenes chalcographus in non-indigenous lodgepole pine and their historical host Norway spruce. Agric. For. Entomol. 2018, 20, 347–357. [Google Scholar] [CrossRef]
- Marini, L.; Økland, B.; Jönsson, A.M.; Bentz, B.; Carroll, A.; Forster, B.; Grégoire, J.-C.; Hurling, R.; Nageleisen, L.M.; Netherer, S.; et al. Climate drivers of bark beetle outbreak dynamics in Norway spruce forests. Ecography 2017, 49, 1426–1435. [Google Scholar] [CrossRef]
- Brauns, A. Forest Insects. In Occurrence in Stands and Habitats; PWRiL: Warsaw, Poland, 1975. [Google Scholar]
- Wermelinger, B.; Seifert, M. Temperature dependent reproduction on the spruce bark beetle Ips typographus, and analysis of the potential population growth. Ecol. Entomol. 1999, 24, 103–110. [Google Scholar] [CrossRef]
- Fahse, L.; Heurich, M. Simulation and analysis of outbreaks of bark beetle infestations and their management at the stand level. Ecol. Model. 2011, 222, 1833–1846. [Google Scholar] [CrossRef]
- Linnakoski, R.; Mahilainen, S.; Harrington, A.; Eriksson, M.; Mehtalo, L.; Pappinen, A.; Wingfield, M.J. Season succession of fungi associated with Ips typographus beetles and their phoretic mites in an outbreak region of Finland. PLoS ONE 2016, 11, e0155622. [Google Scholar] [CrossRef]
- Six, D.L.; Wingfield, M.J. The role of phytopathogenicity in bark beetle-fungus symbioses: A challenge to the classic paradigm. Ann. Rev. Entomol. 2011, 56, 255–272. [Google Scholar] [CrossRef]
- State Forest Report. State Forest Information Centre: Warsaw, Poland, 2017. Available online: https://www.bdl.lasy.gov.pl/portal/Media/Default/Publikacje/raport_o_stanie_lasow_2017.pdf (accessed on 22 February 2019).
- Heyman, O.; Gaston, G.G.; Kimerling, A.J.; Campbell, J.T. A per-segment approach to improving aspen mapping from high-resolution remote sensing imagery. J. For. 2003, 101, 29–33. [Google Scholar] [CrossRef]
- Skakun, R.S.; Wulder, M.A.; Franklin, S.E. Sensitivity of the thematic mapper enhanced wetness difference index to detect mountain pine beetle red-attack damage. Rem. Sens. Environ. 2003, 86, 433–443. [Google Scholar] [CrossRef]
- Erfanifard, Y.; Stereńczak, K.; Miścicki, S. Management strategies alter competitive interactions and structural properties of Norway spruce in mixed stands of Białowieża Forest, Poland. For. Ecol. Manag. 2019, 437, 87–98. [Google Scholar] [CrossRef]
- Zhou, Z.; Woodcock, C.E.; Olofsson, P. Continuous monitoring of forest disturbance using all available Landsat imagery. Rem. Sens. Environ. 2012, 122, 75–91. [Google Scholar] [CrossRef]
- Michel, A., Seidling, W., Prescher, A.K., Eds.; Technical Report of ICP Forests Report under the UNECE Convention on Long-Range Transboundary Air Pollution (CLRTAP). Available online: http://icp-forest.net (accessed on 31 January 2019).
- Zmarz, A.; Będkowski, K.; Miścicki, S.; Plutecki, W. Assessment of Norway spruce health using multispectral images acquired from unmanned aerial vehicle with non-metric cameras. Arch. Fotogram. Kartogr. Teledetekcji 2012, 23, 541–550. [Google Scholar]
- Sokołowski, A.W. Primeval Forests of Białowieża. Forest State Information Center; CILP: Warsaw, Poland, 2004; pp. 8–15. [Google Scholar]
- Eriksson, G.; Ekberg, I. An Introduction to Forest Genetics; SLU Repro: Uppsala, Sweden, 2001. [Google Scholar]
- Pfeiffer, A.; Olivieri, A.M.; Morgante, M. Identification and characterisation of microsatellites in Norway spruce (Picea abies K.). Genome 1997, 40, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, R.; Scotti, I.; Jansson, G.; Plomion, C.; Mathur, G. Inheritance and diversity of simple sequence repeat (SSR) microsatellite markers in various families of Picea abies. Hereditas 2003, 138, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Goudet, J. FSTAT, a Program to Estimate and Test Gene Diversities and Fixation Indices (Version 2.9.4). Available online: http://www2.unil.ch/popgen/softwares/fstat.htm (accessed on 31 January 2019).
- Nei, M. Molecular Evolutionary Genetics; Columbia University Press: New York, NY, USA, 1987. [Google Scholar]
- Weir, B.S.; Cockerham, C.C. Estimating F-statistics for the analysis of population structure. Evolution 1984, 38, 1358–1370. [Google Scholar]
- Chapuis, M.P.; Estoup, A. Microsatellite null alleles and estimation of population differentiation. Mol. Biol. Evol. 2007, 24, 621–631. [Google Scholar] [CrossRef]
- Weir, B.S. Genetic Data Analysis II; Sinauer Associates: Sunderland, MA, USA, 1996. [Google Scholar]
- Dempster, A.P.; Laird, N.M.; Rubin, D.B. Maximum likelihood from incomplete data via the EM algorithm. J. Roy. Stat. Soc. B 1977, 39, 1–38. [Google Scholar]
- Michalakis, Y.; Excoffier, L. A generic estimation of population subdivision using distances between alleles with special reference for microsatellite loci. Genetics 1996, 142, 1061–1064. [Google Scholar]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar]
- Corander, J.; Tang, J. Bayesian analysis of population structure based on linked molecular information. Math. Biosci. 2007, 205, 19–31. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Earl, D.A.; von Holdt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Cons. Gen. Res. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Felsenstein, J. PHYLIP (Phylogeny Inference Package) Version 3.6; Department of Genome Sciences, University of Washington: Seattle, DC, USA, 2005. [Google Scholar]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Nielsen, M.M.; Heurich, M.; Malmberg, B.; Brun, A. Automatic mapping of standing dead trees after an Insect outbreak using the window independent context segmentation method. J. For. 2014, 112, 564–571. [Google Scholar] [CrossRef]
- Ravazzi, C. Late quaternary history of spruce in southern Europe. Rev. Paleobot. Palynol. 2002, 120, 131–177. [Google Scholar] [CrossRef]
- Salimonti, A.; Simeone, V.; Cesari, G.; Lamaj, F.; Cattivelli, L.; Perri, E.; Desiderio, F.; Fanizzie, F.P.; Del Coco, L.; Zelasco, S. A first molecular investigation of monumental olive trees in Apulia region. Sci. Hortic. 2013, 162, 204–212. [Google Scholar] [CrossRef]
- Wojnicka-Półtorak, A.; Celiński, K.; Chudzińska, E. Temporal dynamics in the genetic structure of a natural population of Picea abies. Biologia 2016, 71, 875–884. [Google Scholar] [CrossRef]
- Nowakowska, J.A.; Łukaszewicz, J.; Borys, M.; Tereba, A.; Konecka, A.; Zawadzka, A.; Sułkowska, M.; Zajączkowski, P. Origin of the Norway spruce (Picea abies L. Karst.) stands from Białowieża Forest at the level of Białystok Regional Directorate of the State Forests based on mitochondrial DNA analysis. Sylwan 2017, 161, 40–51. [Google Scholar]
- Wojnicka-Półtorak, A.; Wachowiak, W.; Prus-Głowacki, W.; Celiński, K.; Korczyk, A. Genetic heterogeneity in age classes of naturally regenerated old growth forest of Picea abies (L.) Karst. Silv. Genet. 2014, 63, 185–190. [Google Scholar] [CrossRef]
- Mott, C.L. Environmental constraints to the geographic expansion of plant and animal species. Nat. Educ. Knowl. 2010, 3, 72. [Google Scholar]
- Schlyter, P.; Stjernquist, I.; Bärring, L.; Jönsson, A.M.; Nilsson, C. Assessment of the impacts of climate change and weather extremes on boreal forests in northern Europe, focusing on Norway spruce. Clim. Res. 2006, 31, 75–84. [Google Scholar] [CrossRef]
- Brandt, L.A.; Butler, P.R.; Handler, S.D.; Janowiak, M.K.; Shannon, P.D.; Swanston, C.W. Integrating science and management to assess forest ecosystem vulnerability to climate change. J. For. 2016, 115, 212–221. [Google Scholar] [CrossRef]
- Tiscar, P.A.; Lucas-Borja, M.E. Structure of old-growth and managed stands and growth of old trees in a Mediterranean Pinus nigra forest in southern Spain. Forestry 2016, 89, 201–207. [Google Scholar] [CrossRef]
- Mikusiński, G.; Bubnicki, J.W.; Churski, M.; Czeszczewik, D.; Walankiewicz, W.; Kuijper, D.P. Is the impact of loggings in the last primeval lowland forest in Europe underestimated? The conservation issues of Białowieża Forest. Biol. Cons. 2018, 227, 266–274. [Google Scholar] [CrossRef]
- Stereńczak, K.; Mielcarek, M.; Modzelewska, A.; Kraszewski, B.; Fassnacht, F.E.; Hilszczański, J. Intra-annual Ips typographus outbreak monitoring using multi-temporal GIS analysis based on hyperspectral and ALS data in the Białowieża Forests. For. Ecol. Manag. 2019, 442, 105–116. [Google Scholar] [CrossRef]
- Stereńczak, K.; Mielcarek, M.; Kamińska, A.; Kraszewski, B.; Piasecka, Ż.; Miścicki, S.; Heurich, M. Influence of selected habitat and stand factors on bark beetle Ips typographus (L.) outbreak in the Białowieża Forest. For. Ecol. Manag. 2020, 459, 117826. [Google Scholar] [CrossRef]
- Nebe, W. Über Beziehungen zwischen Klima Und Wachstum der Fichte (Picea abies L.) in ihrem europeischen Verbreitungsgebiet. Arch. Forstw. 1968, 17, 1219–1238. [Google Scholar]
- Gebauer, R.; Volarík, D.; Urban, J.; Børja, I.; Nagy, N.E.; Eldhuset, T.D.; Krokene, P. Effects of prolonged drought on the anatomy of sun and shade needles in young Norway spruce trees. Ecol. Evol. 2015, 5, 4989–4998. [Google Scholar] [CrossRef]
- Gebauer, R.; Volarík, D.; Urban, J.; Børja, I.; Nagy, N.E.; Eldhuset, T.D.; Krokene, P. Effects of mild drought on the morphology of sun and shade needles in 20-year-old Norway spruce trees. iForest-Biogeosciences For. 2019, 12, 27–34. [Google Scholar] [CrossRef]
- Grodzki, W. Mass outbreaks of the spruce bark beetle Ips typographus in the context of the controversies around the Białowieża Primeval Forest. For. Res. Papers 2016, 77, 324–331. [Google Scholar] [CrossRef]
- Desprez-Loustau, M.L.; Marcais, B.; Nageleisen, L.M.; Piou, D.; Vannini, A. Interactive effects of drought and pathogens in forest trees. Ann. For. Sci. 2006, 63, 597–612. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzbergerg, T.; Riglingh, A.; Breshearsi, D.D.; Hogg, E.H.T.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought. New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Wulder, M.A.; Dymond, C.C.; White, J.C.; Leckie, D.G.; Caroll, A.L. Surveying mountain pine beetle damage of forests: A review of remote sensing opportunities. For. Ecol. Manag. 2006, 221, 27–41. [Google Scholar] [CrossRef]
- Senf, C.; Seidl, R.; Hostert, P. Remote sensing of forest insect disturbances: Current state and future directions. Int. J. Appl. Earth Obs. Geoinf. 2017, 60, 49–60. [Google Scholar] [CrossRef]
- Heurich, M.; Ochs, T.; Andresen, T.; Schneider, T. Object-oriented image analyses for semi-automatic detection of dead trees following a spruce bark beetle (Ips typographus) outbreak. Eur. J. For. Res. 2010, 129, 313–324. [Google Scholar] [CrossRef]
- Polewski, P.; Yao, W.; Heurich, M.; Krzystek, P.; Stilla, U. Detection of single standing dead trees from aerial color infrared imagery by segmentation with shape and intensity priors. ISPRS Ann. Photogramm. Remote Sens. Spat. Inf. Sci. 2015, 2, 181–188. [Google Scholar] [CrossRef]
- Tollefsrud, M.M.; Latałowa, M.; van der Knaap, W.O.; Brochmann, C.; Sperisen, C. Late Quaternary history of North Eurasian Norway spruce (Picea abies) and Siberian spruce (Picea obovata) inferred from macrofossils, pollen and cytoplasmic DNA variation. J. Biogeogr. 2015, 42, 1431–1442. [Google Scholar] [CrossRef]
- Humphrey, J.W. Benefits to biodiversity from developing old-growth conditions in British upland spruce plantations: A review and recommendations. For. Intern. J. For. Res. 2005, 78, 33–53. [Google Scholar] [CrossRef]
- Westergren, M.; Bozic, G.; Kraigher, H. Genetic diversity of core vs. peripheral Norway spruce native populations at a local scale in Slovenia. iForest 2018, 11, 104–110. [Google Scholar] [CrossRef]
- Greenbaum, G.; Templeton, A.R.; Zarmi, Y.; Bar-David, S. Allelic richness following population founding events–a stochastic modeling framework incorporating gene flow and genetic drift. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Allendorf, F.W. Genetic drift and the loss of alleles versus heterozygosity. Zoo. Biol. 1986, 5, 181–190. [Google Scholar] [CrossRef]
- Hughes, R.A.; Inouye, B.D.; Johnson, M.T.J.; Underwood, N.; Vellend, M. Ecological consequences of genetic diversity. Ecol. Lett. 2008, 11, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Szulkin, M.; Bierne, N.; David, P. Heterozygosity-fitness correlations: A time for reappraisal. Evolution 2010, 64, 1202–1217. [Google Scholar] [CrossRef] [PubMed]
- Szczecińska, M.; Sramko, G.; Wołosz, K.; Sawicki, J. Genetic diversity and population structure of the rare and endangered plant species Pulsatilla patens (L.) Mill. in East Central Europe. PLoS ONE 2016, 11, e0151730. [Google Scholar] [CrossRef]
- Martín, M.A.; Mattioni, C.; Cherubini, M.; Villani, F.; Martín, L.M. A comparative study of European chestnut varieties in relation to adaptive markers. Agrofor. Syst. 2017, 91, 97–109. [Google Scholar] [CrossRef]
- Belletti, P.; Ferrazzini, D.; Piotti, A.; Monteleone, I.; Ducci, F. Genetic variation and divergence in Scots pine (Pinus sylvestris L.) within its natural range in Italy. Eur. J. For. Res. 2012, 131, 1127–1138. [Google Scholar] [CrossRef]
- Rajora, O.P.; Pluhar, S.A. Genetic diversity impacts of forest fires, forest harvesting, and alternative reforestation practices in black spruce (Picea mariana). Theor. Appl. Genet. 2003, 106, 1203–1212. [Google Scholar] [CrossRef]
- Alcaide, F.; Solla, A.; Cherubini, M.; Mattioni, C.; Cuenca, B.; Camisón, Á.; Martín, M.Á. Adaptive evolution of chestnut forests to the impact of ink disease in Spain. J. Syst. Evol. 2020. [Google Scholar] [CrossRef]
- Sjölund, M.J.; Jump, A.S. Coppice management of forests impacts spatial genetic structure but not genetic diversity in European beech (Fagus sylvatica L.). For. Ecol. Manag. 2015, 336, 65–71. [Google Scholar] [CrossRef]
- Ramakrishnan, U. Detecting past population bottlenecks using temporal genetic data. Mol. Ecol. 2005, 14, 2915–2922. [Google Scholar] [CrossRef] [PubMed]
- Meirmans, P.G. Using the AMOVA framework to estimate a standardized genetic differentiation measure. Evolution 2006, 60, 2399–2402. [Google Scholar] [CrossRef] [PubMed]
- Hamrick, J.L.; Godt, M.J.W. Allozyme diversity in plant species. In Plant Population Genetics, Breeding and Genetic Resources; Brown, H.D., Clegg, M.T., Kahler, A.L., Weir, B.S., Eds.; Sinauer: Sunderland, MA, USA, 1989; pp. 43–63. [Google Scholar]
- Mousadik, A.; Agroforesterie, L.; Sciences, F. High level of genetic differentiation for allelic richness among populations of the argan tree [Argania spinosa (L.) Skeels] endemic to Morocco. Theor. Appl. Genet. 1996, 92, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Nowakowska, J.A. Mitochondrial and nuclear DNA differentiation of Picea abies populations in Poland. Dendrobiology 2009, 61 (Suppl), 119–129. [Google Scholar]
- Jombart, T.; Devillard, S.; Balloux, F. Discriminant analysis of principal components: A new method for the analysis of genetically structured populations. BMC Genet. 2010, 11, 94. [Google Scholar] [CrossRef] [PubMed]
- Kalinowski, S.T. The computer program STRUCTURE does not reliably identify the main genetic clusters within species: Simulations and implications for human population structure. Heredity 2010, 106, 625–632. [Google Scholar] [CrossRef]
- Stojnić, S.; Avramidou, E.V.; Fussi, B.; Westergren, M.; Orlović, S.; Matović, B.; Trudić, B.; Kraigher, H.; Aravanopoulos, F.A.; Konnert, M. Assessment of Genetic Diversity and Population Genetic Structure of Norway Spruce (Picea abies (L.) Karsten) at Its Southern Lineage in Europe. Implications for Conservation of Forest Genetic Resources. Forests 2019, 10, 258. [Google Scholar] [CrossRef]
- Bínová, Z.; Korecký, J.; Dvořák, J.; Bílý, J.; Zádrapová, D.; Jansa, V.; Lstibůrek, M. Genetic Structure of Norway Spruce Ecotypes Studied by SSR Markers. Forests 2020, 11, 110. [Google Scholar] [CrossRef]
- Lefèvre, F. Human impacts on forest genetic resources in the temperate zone: An updated review. For. Ecol. Manag. 2004, 197, 257–271. [Google Scholar] [CrossRef]

| Health Status | Crown Color | Tree Crown Condition Description | Tree Crown Condition Image |
|---|---|---|---|
| 1—Very high | Saturated red | Healthy, full needles, with no visible color changes |  |
| 2—High | Red lightened | Reduction in foliage, parts of needles with visible discoloration |  |
| 3—Weak | White (with a slight addition of red) | Significant reduction in foliage, many remaining needles are discolored |  |
| 4—Dead | Green | Dead trees |  |
| Population Code | Forest District | Population Name | Number of Trees | Geographic Location | Mean Age (years 2) | Area (ha) |
|---|---|---|---|---|---|---|
| 1 | Białowieża | Monumental trees | 47 | 52°38′/46′ N, 23°43′/54′ E | 225 | 12.59 |
| 2 | Białowieża | BNP 1, parcel 256dg | 40 | 52°46′ N, 23°50′ E | 150 | 30.17 |
| 3 | Białowieża | BNP, parcel 284dg | 23 | 52°45′ N, 23°51′ E | 140 | 25.95 |
| 4 | Białowieża | Teremiski | 28 | 52°44′52′′ N, 23°46′30′′ E | 188 | 4.72 |
| 5 | Białowieża | Grudki | 40 | 52°40′40′′ N, 23°47′06′′ E | 149 | 20.87 |
| 6 | Hajnówka | Długi Bród | 50 | 52°38′30′′ N, 23°37′05′′ E | 162 | 9.90 |
| 7 | Browsk | Krynica | 50 | 52°52′46′′ N, 23°32′55′′ E | 132 | 3.96 |
| 8 | Browsk | Jelonka | 50 | 52°48′00′′ N, 23°47′50′′ E | 133 | 14.21 |
| Health Status 1 | Year | |||
|---|---|---|---|---|
| 2014 | 2015 | 2016 | 2017 | |
| 1—Very high | 5 | 1 | 1 | 1 |
| 2—High | 29 | 24 | 9 | 9 |
| 3—Weak | 1 | 5 | 3 | 2 |
| 4—Dead | 12 | 17 | 34 | 35 |
| Health Status of Tree a | Number of Trees | |||||
|---|---|---|---|---|---|---|
| Partial Nature Reserves | Valuable Fragments of Native Forest | Water Protection Areas | ||||
| 2014 | 2017 | 2014 | 2017 | 2014 | 2017 | |
| Healthy (1 + 2) | 19 | 5 | 12 | 3 | 3 | 2 |
| Weak (3) | - | - | 1 | 1 | - | 1 |
| Dead (4) | 4 | 18 | 6 | 15 | 2 | 2 |
| Total | 23 | 23 | 19 | 19 | 5 | 5 |
| Year | Annual Precipitation [mm] | Precipitation of Growing Season [mm] | Rprep1 | Average Annual Temperature [°C] | Average Temperature of Growing Season [°C] |
|---|---|---|---|---|---|
| 2000 | 533.33 | 304.05 | 0.57 | 8.13 | 13.19 |
| 2001 | 605.20 | 453.93 | 0.75 | 7.20 | 13.55 |
| 2002 | 625.61 | 412.98 | 0.66 | 7.78 | 13.71 |
| 2003 | 548.96 | 367.81 | 0.67 | 7.01 | 12.88 |
| 2004 | 660.95 | 416.40 | 0.63 | 7.05 | 12.54 |
| 2005 | 633.49 | 399.10 | 0.63 | 6.94 | 12.85 |
| 2006 | 567.18 | 442.40 | 0.78 | 7.23 | 13.87 |
| 2007 | 692.92 | 450.47 | 0.65 | 8.00 | 13.36 |
| 2008 | 666.61 | 413.33 | 0.62 | 8.15 | 13.10 |
| 2009 | 786.06 | 518.83 | 0.66 | 7.22 | 12.96 |
| 2010 | 870.12 | 704.81 | 0.81 | 6.81 | 13.60 |
| 2011 | 637.22 | 503.44 | 0.79 | 7.74 | 13.79 |
| 2012 | 675.50 | 540.46 | 0.80 | 7.23 | 13.61 |
| 2013 | 721.89 | 534.29 | 0.74 | 7.70 | 13.71 |
| 2014 | 608.38 | 450.28 | 0.74 | 8.01 | 13.41 |
| 2015 | 512.57 | 358.86 | 0.70 | 8.79 | 13.43 |
| 2016 | 842.50 | 674.07 | 0.80 | 7.51 | 13.39 |
| Average 2000–2011 | 652.30 | 448.96 | 0.69 | 7.44 | 13.28 |
| Average 2012–2016 | 672.17 | 511.59 | 0.76 | 7.85 | 13.51 |
| Population | N | Na | Ne | AR(11) | Apriv | I | HO | HE | Gd | FIS |
|---|---|---|---|---|---|---|---|---|---|---|
| Pop1 | 47 | 15.909 | 7.656 | 8.895 | 0.909 | 1.979 | 0.564 | 0.707 | 0.716 | 0.211 *** |
| Pop2 | 40 | 13.182 | 7.124 | 8.565 | 0.545 | 1.913 | 0.578 | 0.712 | 0.724 | 0.203 *** |
| Pop3 | 23 | 9.909 | 5.780 | 8.113 | 0.455 | 1.745 | 0.627 | 0.703 | 0.726 | 0.147 *** |
| Pop4 | 28 | 12.364 | 6.917 | 8.744 | 0.273 | 1.891 | 0.550 | 0.713 | 0.730 | 0.246 *** |
| Pop5 | 40 | 14.455 | 7.519 | 8.760 | 0.636 | 1.930 | 0.564 | 0.702 | 0.714 | 0.214 *** |
| Pop6 | 50 | 13.818 | 7.265 | 8.432 | 0.273 | 1.928 | 0.541 | 0.718 | 0.727 | 0.255 *** |
| Pop7 | 50 | 14.273 | 6.989 | 8.509 | 0.182 | 1.916 | 0.519 | 0.702 | 0.713 | 0.264 *** |
| Pop8 | 50 | 15.273 | 7.562 | 8.689 | 0.364 | 1.971 | 0.587 | 0.722 | 0.731 | 0.196 *** |
| Mean | 41 | 13.648 | 7.102 | 8.588 | 0.455 | 1.909 | 0.566 | 0.710 | 0.723 | 0.217 *** |
| Source of Variability | d.f. 1 | Sum of Squares | Mean Square | Estimated Variance | Variation Share (%) | p Value 2 |
|---|---|---|---|---|---|---|
| Among populations | 7 | 53.532 | 7.647 | 0.030 | 1 | 0.007 |
| Within populations | 648 | 2611.123 | 8.0088 | 4.044 | 99 | 0.300 |
| Total | 655 | 2664.655 | 4.074 | 100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowakowska, J.A.; Hsiang, T.; Patynek, P.; Stereńczak, K.; Olejarski, I.; Oszako, T. Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Białowieża Forest District, Poland. Forests 2020, 11, 647. https://doi.org/10.3390/f11060647
Nowakowska JA, Hsiang T, Patynek P, Stereńczak K, Olejarski I, Oszako T. Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Białowieża Forest District, Poland. Forests. 2020; 11(6):647. https://doi.org/10.3390/f11060647
Chicago/Turabian StyleNowakowska, Justyna Anna, Tom Hsiang, Paulina Patynek, Krzysztof Stereńczak, Ireneusz Olejarski, and Tomasz Oszako. 2020. "Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Białowieża Forest District, Poland" Forests 11, no. 6: 647. https://doi.org/10.3390/f11060647
APA StyleNowakowska, J. A., Hsiang, T., Patynek, P., Stereńczak, K., Olejarski, I., & Oszako, T. (2020). Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Białowieża Forest District, Poland. Forests, 11(6), 647. https://doi.org/10.3390/f11060647







